- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
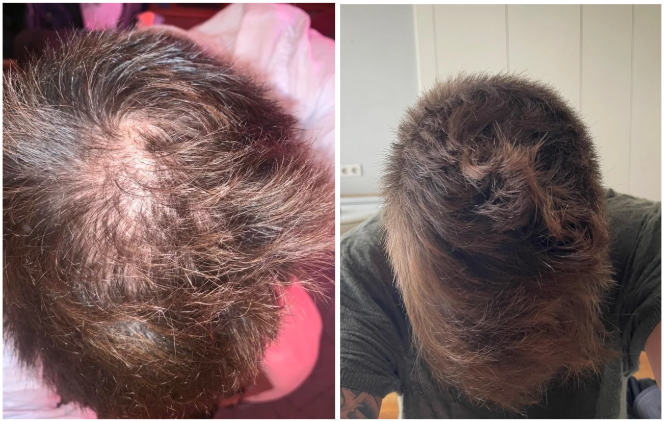
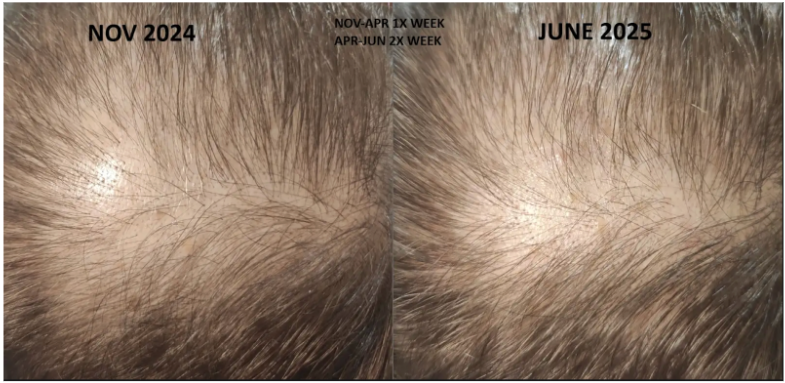
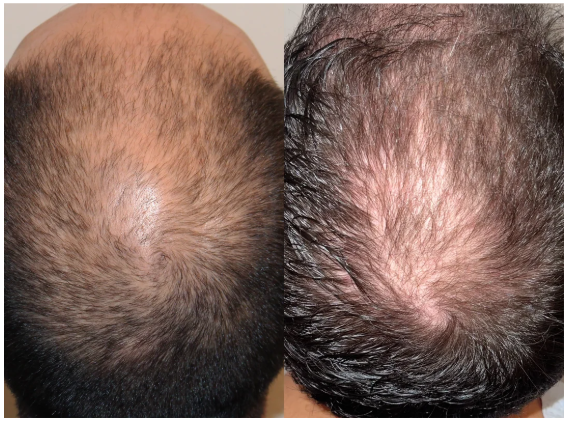
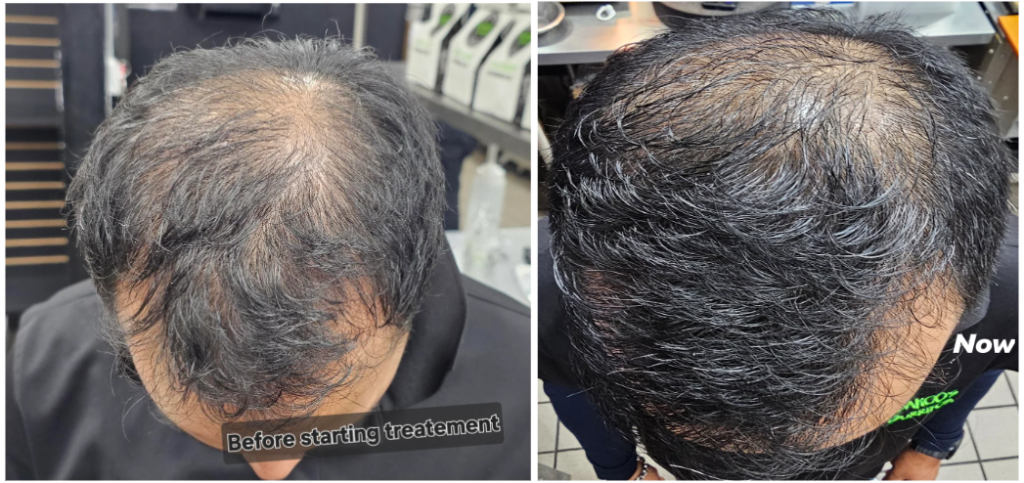
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Does Anavar Cause Hair Loss?
-
10 Best Shampoos for Hair Loss in 2026
-
XYON Review: Do Their Products Actually Regrow Hair?
-
Minoxidil Before and After Photos [2026] | Does It Work?
-
How to Get Finasteride: Is It Over the Counter?
-
Keeps Review: The Truth About Their Hair Loss Treatments?
-
7 Best Oils for Hair Growth
-
Hims Hair Growth Reviews: The Pros, Cons, and Real Results
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
Articles
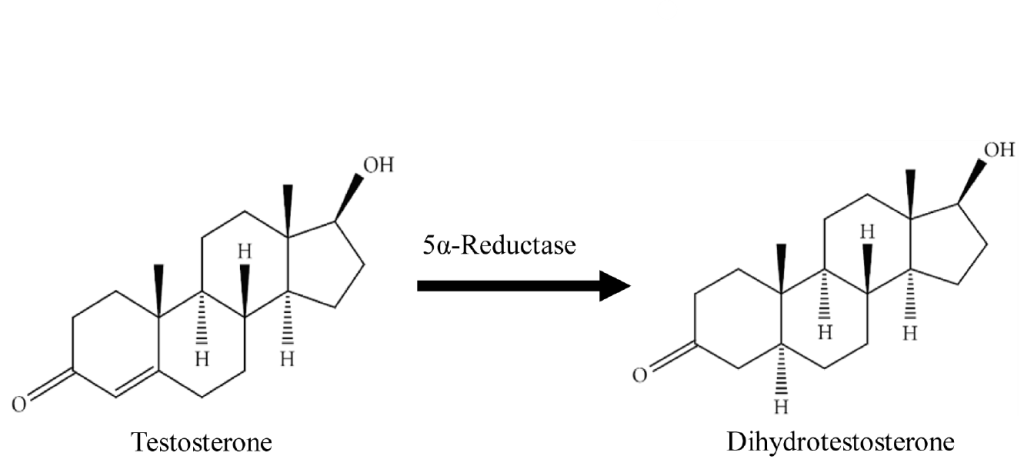
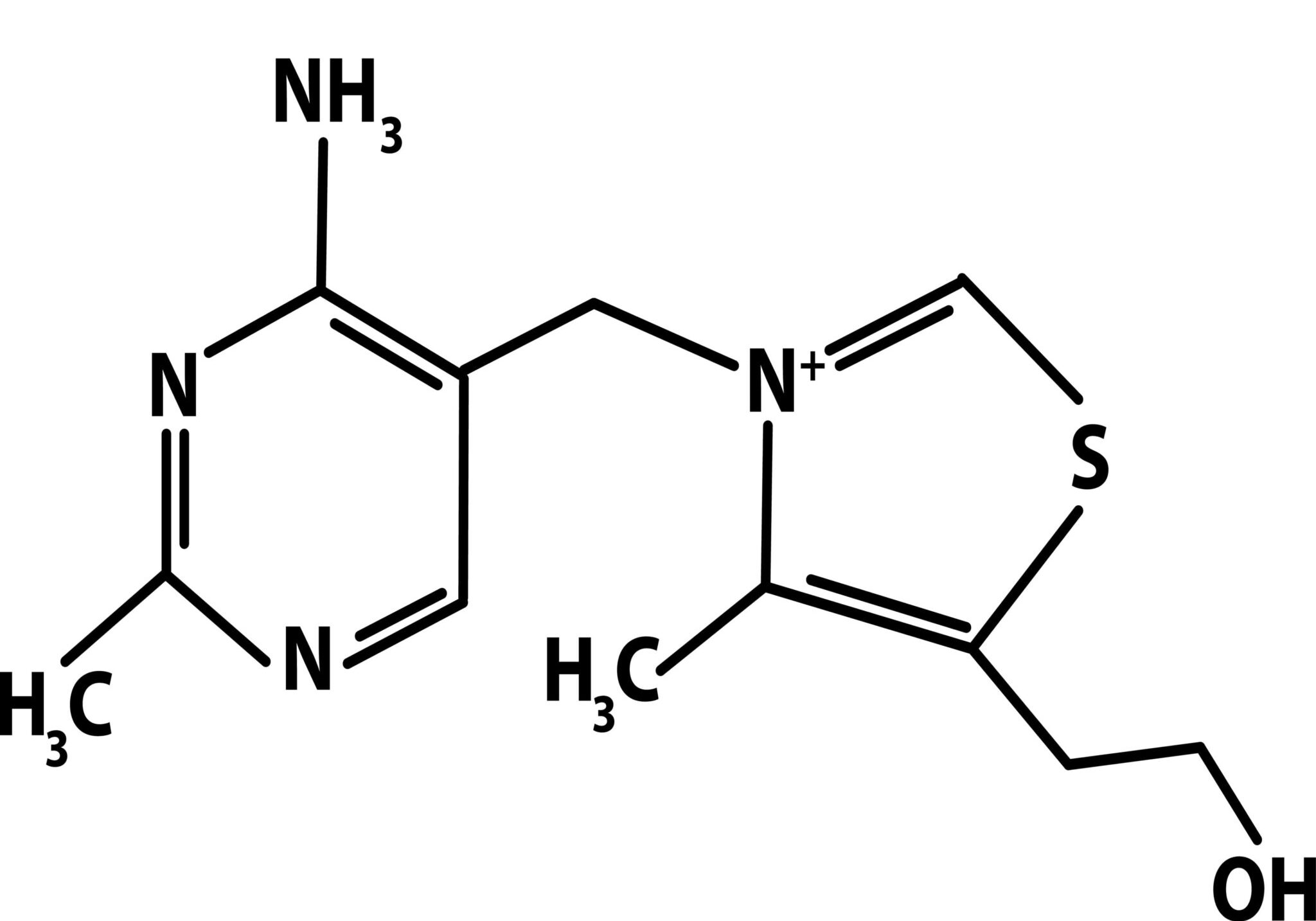
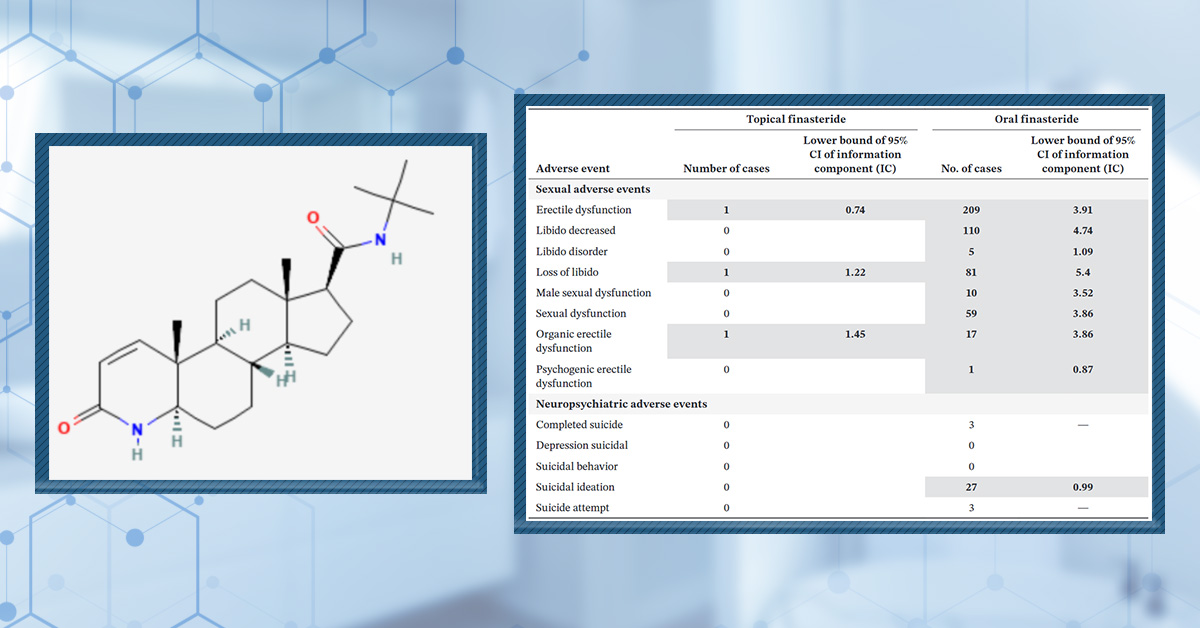
Oral finasteride has established itself as a highly effective treatment for androgenic alopecia (AGA) and is widely prescribed around the globe. Its success can be largely attributed to its proven ability to slow hair loss and stimulate regrowth by inhibiting the conversion of testosterone to DHT, the hormone chiefly responsible for follicular miniaturization.
However, despite its well-documented efficacy, oral finasteride has become equally notorious for its potentially systemic side effects. Some men experience adverse effects such as decreased libido, erectile dysfunction, mood changes, and even persistent symptoms after discontinuation, a phenomenon called post-finasteride syndrome.[1]Diviccaro, S., Melcangi, R.C., Giatti, S. (2019). Post-finasteride syndrome: an emerging clinical problem. Neurobiology of Stress. 12. 100209. Available at: https://doi.org/10.1016/j.ynstr.2019.100209 This reputation has prompted many to seek alternatives that are both effective and safer for long-term use.
Recently, topical finasteride formulations have garnered attention as a promising alternative. By delivering the medication directly to the scalp, topical finasteride aims to minimize systemic absorption and thereby reduce the likelihood of side effects while still providing the benefits of DHT inhibition where it matters most. This alternative approach has piqued the interest of both patients wary of systemic issues and clinicians seeking tailored treatments.
In this article, we will examine whether topical finasteride can serve as a viable alternative to its oral counterpart. The main focus will be on answering three questions:
- Can topical finasteride effectively regrow hair in those affected by AGA?
- Does it match the efficacy results seen with oral finasteride?
- Can it reduce hair loss while offering a more favorable side effect profile?
Ulo offers finasteride options that range from low to high dose finasteride – allowing you to be flexible in your treatment choices.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

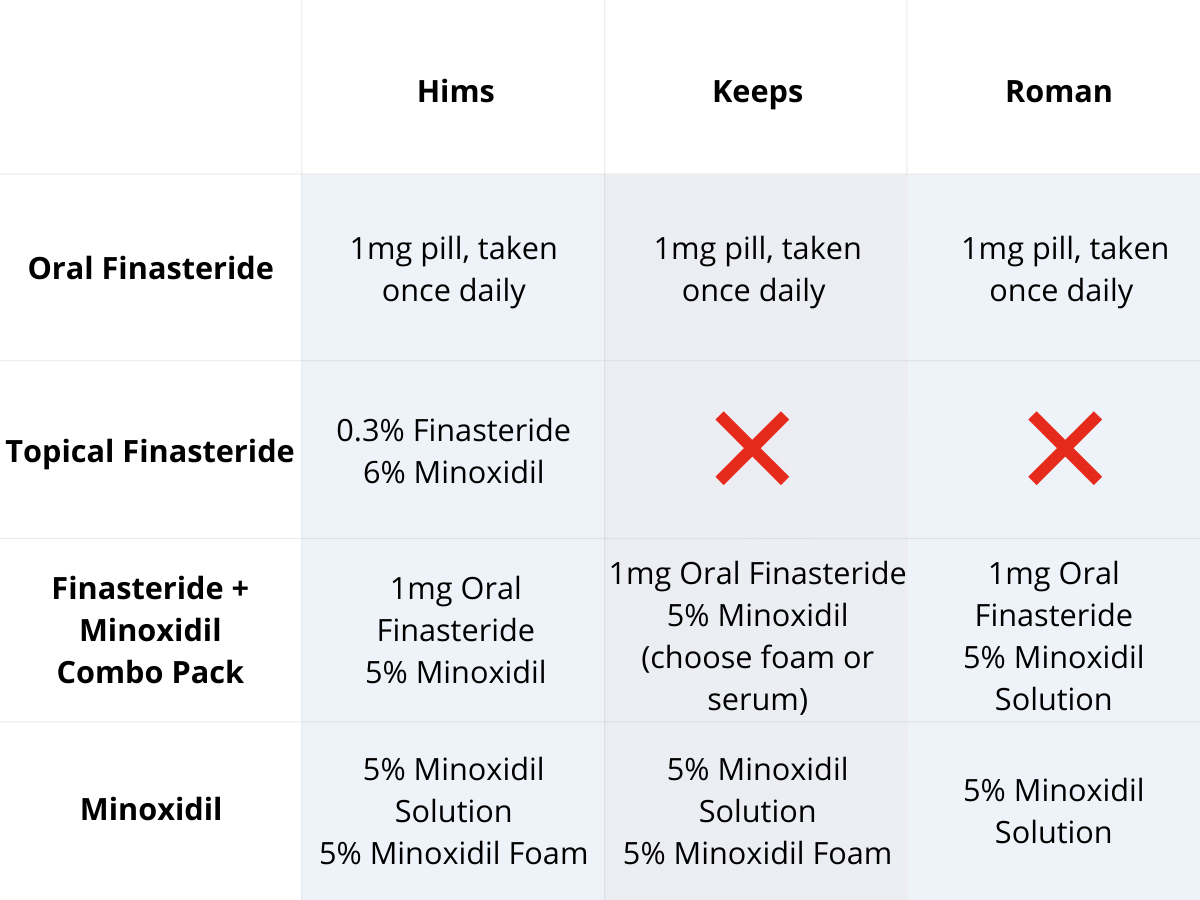
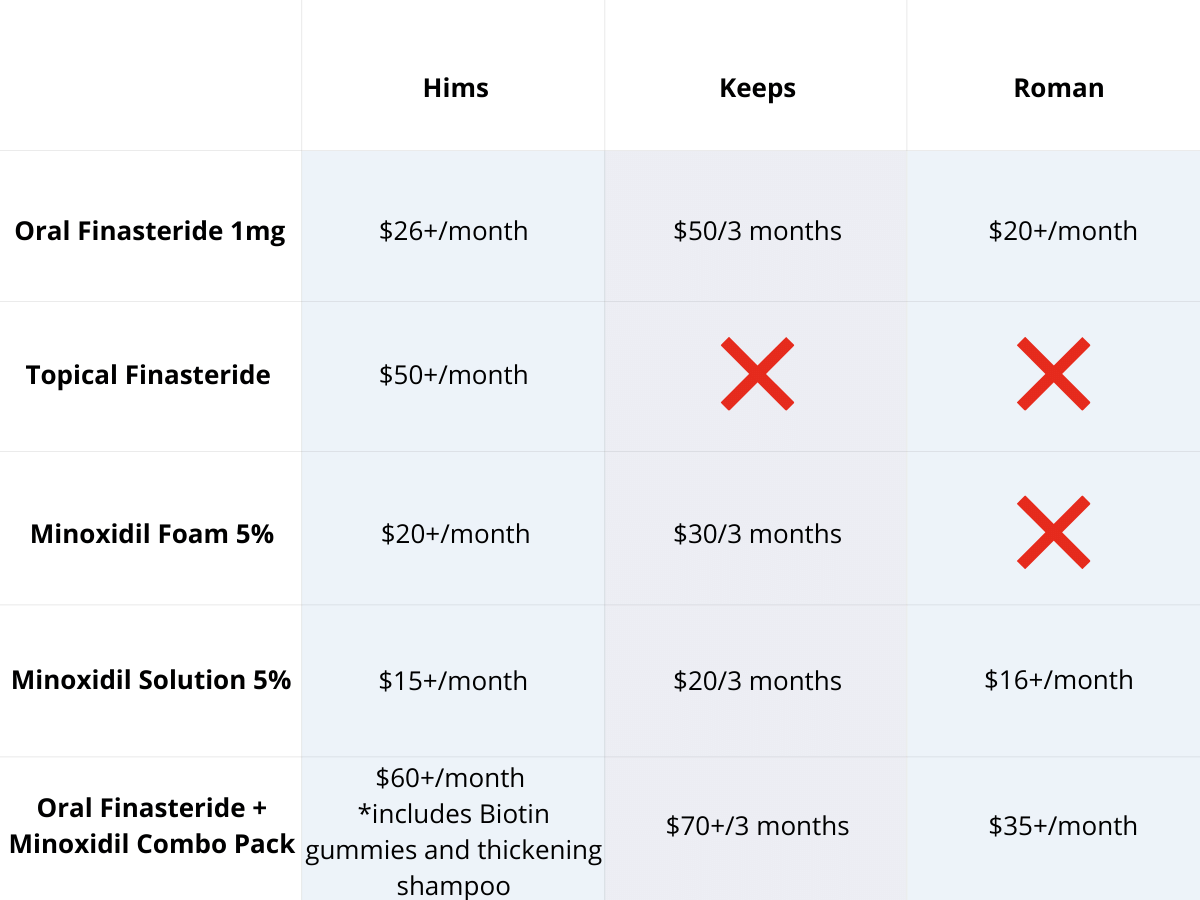
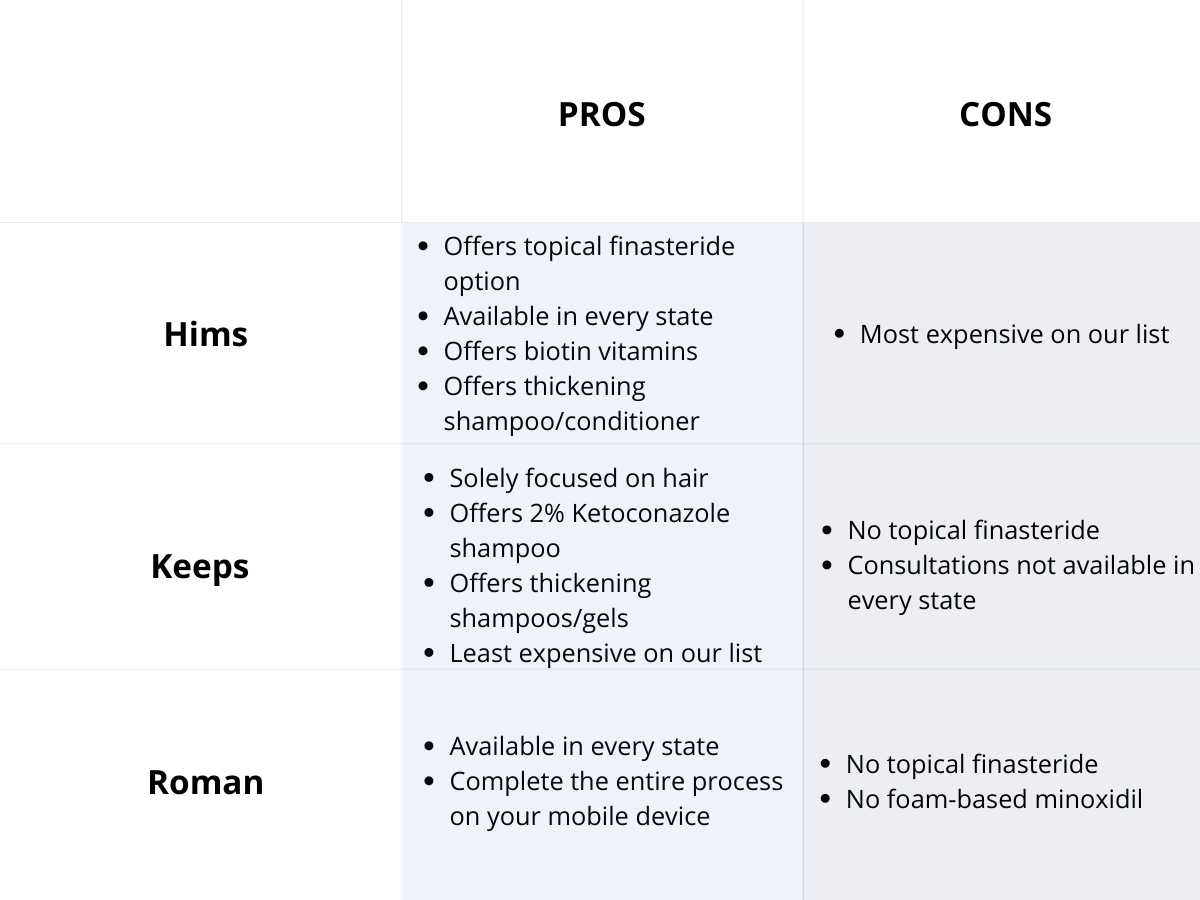
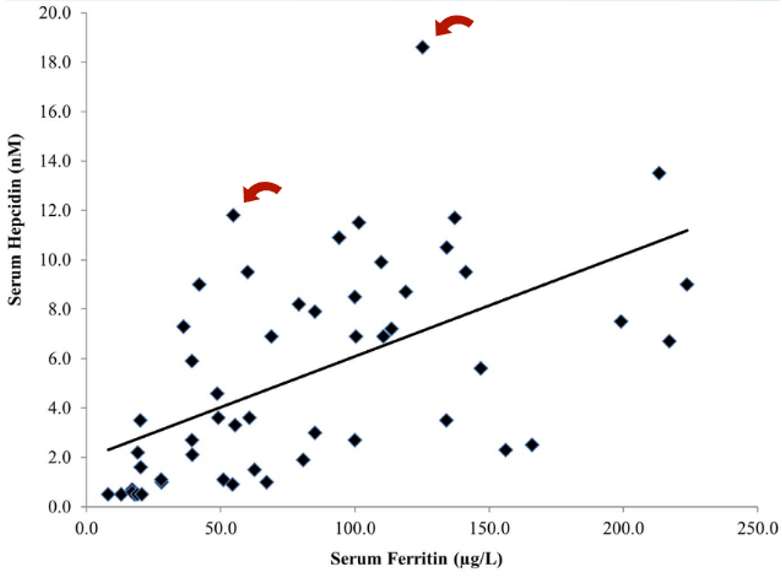
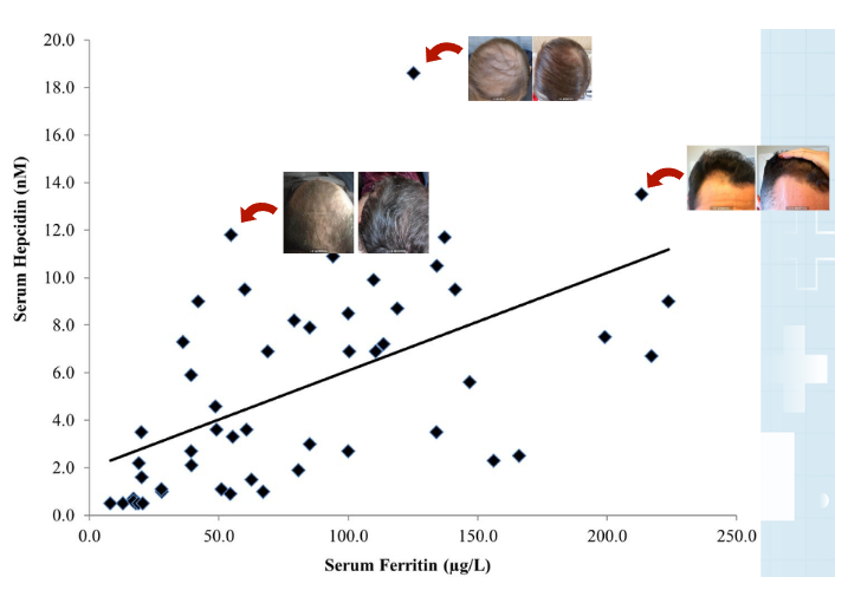
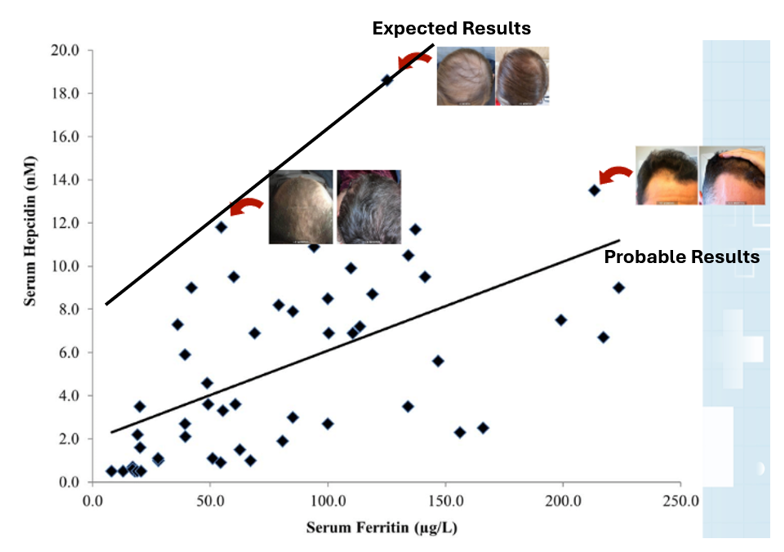
Oral vs. Topical Finasteride
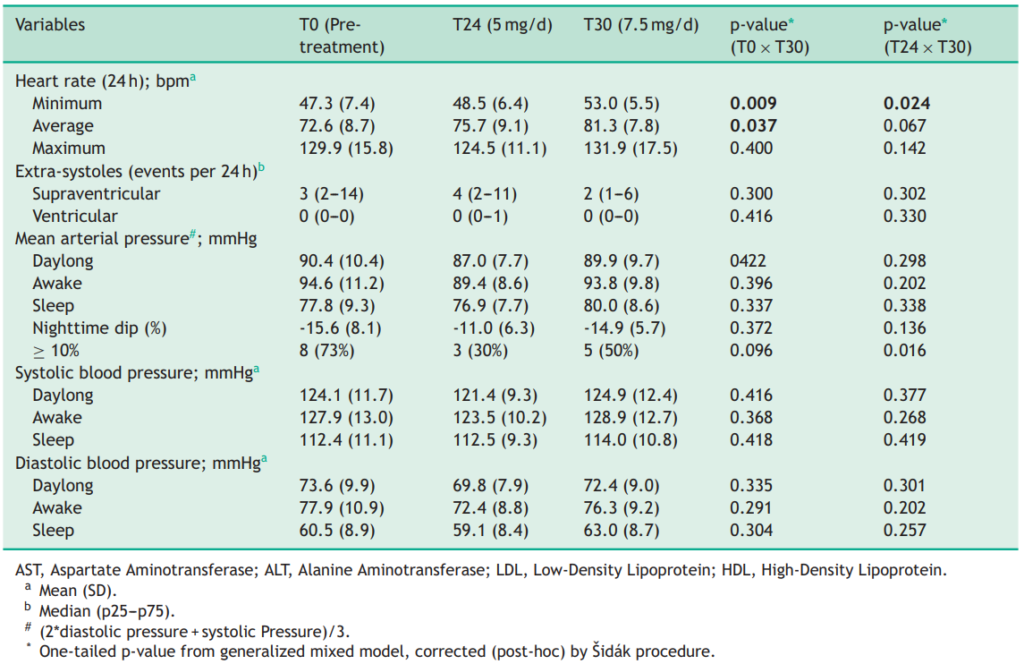
Some research directly compares oral finasteride (1 mg daily) with various concentrations of topical finasteride, aiming to match hair regrowth efficacy while minimizing systemic side effects. Let’s take a look at a couple of examples:
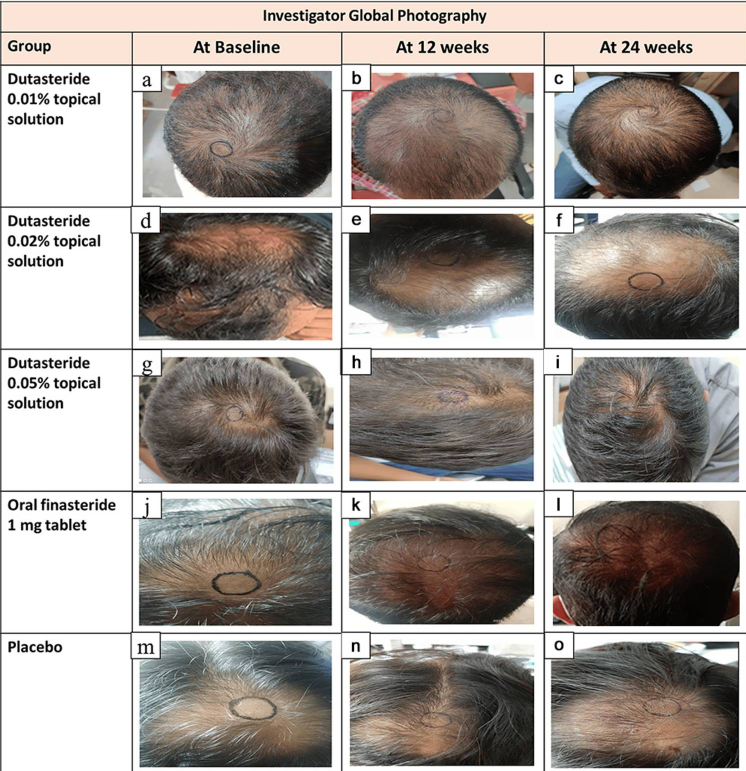
Study 1 – Piraccini et al., 2022 (Phase III RCT, 24 weeks)
- Compared topical finasteride (0.25% spray) to oral finasteride (1 mg/day) and placebo in men with AGA.[2]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. Efficacy and safety of … Continue reading
- The increase in hair count on a defined scalp area was similar for topical and oral finasteride, both outperforming the placebo, suggesting comparable clinical benefit.
- Systemic finasteride (oral) reduced mean serum DHT by 55.6%, whereas topical finasteride reduced it by 34.5%. Plasma finasteride exposure from topical application was over 100 times lower than from oral administration.
- Both had similar low rates of mild side effects, but sexual side effects, but sexual side effects were more associated with oral treatment.
- Caveats: The “target area” measured may not represent the full scalp effects; although systemic absorption, albeit at a much lower level with topical application, still occurred and varied by formulation.
Study 2 – Bhura, 2013 (RCT, 8 months)
- Parallel-group comparison in male AGA, tracking hair density and systemic DHT.[3]Bhura, M. (2013). Comparative Analysis of Topical and Oral Finasteride in Androgenetic Alopecia: Impact on Hair Growth, Systemic Absorption, and Adverse Effects. Indian Journal of Basic and Applied … Continue reading
- Oral finasteride resulted in slightly superior increases in hair number and thickness, although the difference was not statistically significant.
- Oral finasteride significantly reduced systemic DHT and caused more sexual side effects; topical finasteride led to minimal serum DHT changes and almost no systemic side effects, with most reactions being local (e.g., irritation).
- Those worried about long-term systemic effects preferred topical use, despite slightly less pronounced gains.
Study 3 – Hajheydari, 2009 (RCT, 6 months)
- Compared a 1% topical gel + placebo tablet with oral finasteride 1 mg/day + placebo gel.[4]Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. Indian Journal of Dermatology, … Continue reading
- Both the gel and tablet produced statistically significant increases in total hair count and terminal hair count.
- There were no significant differences in hair thickness, hair count, or size of the bald area between the two groups.
Mechanism of Action
Finasteride Pharmacodynamics
Finasteride is a competitive inhibitor of 5α-reductase, primarily targeting the type II isoenzyme at therapeutic doses, which predominates in the hair follicles and prostate. As mentioned above, this enzyme catalyzes the conversion of testosterone to DHT.
At higher tissue concentrations, finasteride can also inhibit type I 5α-reductase, which is primarily found in the skin and sebaceous glands; however, its clinical significance for hair loss at standard doses is limited. By selectively inhibiting the type II enzyme, finasteride effectively lowers local and systemic DHT levels.[5]Finn, D.A., Beadles-Bohling, A., Beckley, E.H., Ford, M.M., Gililland, K.R., Gorin-Meyer, R.E., Wiren, K.M. (2006). 12(1). 53-76. A New Look at the 5α-reductase Inhibitor Finasteride. CNS Drug … Continue reading
What is Finasteride’s Effect on DHT Suppression?
Finasteride’s effect on DHT suppression is characterized by a logarithmic dose-response curve, meaning that even very low systemic levels can produce a marked reduction in both serum and scalp DHT. Most of the drug’s inhibitory action is achieved at low doses, with additional dosing yielding only marginal further effect.
As a result, even minimal “leakage” of topically applied finasteride into the bloodstream can decrease DHT levels elsewhere in the body.
Pharmacokinetic studies show that inhibition of type-II 5ɑ-reductase reaches saturation at typical clinical dosage, while type-1 enzyme inhibition requires much higher concentrations. This nonlinear pharmacodynamic profile explains both the strong efficacy and the wide safety margin of topical finasteride: a small amount achieves most of the desired effect, so careful formulation is crucial to maximize scalp delivery while minimizing unwanted systemic exposure.[6]Suzuki, R., Satoh, H., Ohtani, H., Hori, S., Sawada, Y. (2010). Saturable Binding of Finasteride to Steroid 5ɑ-reductase as Determinant of Nonlinear Pharmacokinetics. Drug Metabolism and … Continue reading
What the Science Says
Topical finasteride has been studied across a 200-fold concentration range, from 0.005% solutions to 1% gels. Collectively, these trials demonstrate meaningful reductions in shedding and measurable regrowth; however, systemic exposure increases with dose and vehicle potency.
Study Concentration & Vehicle Hair Growth-Outcomes Systemic/Serum Findings Mazzarella 1997, single-blind, 52 men/women, 16 months. 0.005% hydro-alcoholic solution 73% reported “high effectiveness”; wash-test hair counts improved; slowed shedding by month 6 No significant change in plasma DHT or testosterone; absorption was negligible. Tanglertsampan 2012 RCT, 33 men, 24 weeks. 0.1% lotion + 3% minoxidil The combination arm gained more hairs/cm2 and thicker shafts than minoxidil alone. Local irritation mild; systemic parameters not monitored. Datta 2021 double-blind trial, 35 participants completed, 6 months. 0.1% lotion +5% minoxidil vs oral 1 mg. The topical combination was non-inferior to oral minoxidil for reducing the Hamilton-Norwood stage. Sexual adverse events only occurred in the oral group; topical was well-tolerated. Caserini 2016 OK study, 50 men, 1 week; Piraccini 2022 phase III, 323 completers, 24 weeks. 0.25% solution. -70% scalp DHT after once-daily 1 mL; +20.2 hairs in 1 cm2 target area at 24 weeks – numerically equal to oral 1 mg. 100-200 μL doses reduced serum DHT by only 24-26%; 400 μL treatment reduced levels by 44-48%; Cmax was more than 100 times lower than oral. Rossi 2020 retrospective, 69 women, 12-18 months. 0.5% lotion (postmenopausal women) The Finasteride + minoxidil group scored higher on a 7-point global scale compared to the 17ɑ-estradiol + minoxidil group. No androgenic side effects reported. Hajheydari 2009 DB-RCT, 45 men, 6 months. 1% gel Increases in total and terminal hair counts matched those of oral 1 mg; the bald area remained unchanged. Serum DHT not assayed; clinical side-effects minimal. Low-Dose Takeaways
Even a micro-dose of 0.005% twice daily (~0.1 mg/day) curbed shedding and improved density without measurable systemic suppression, making it an attractive option for highly risk-averse users.[7]Mazzarella, F., Loconsole, F., Cammisa, A., Mastrolonardo, M., Vena, G.A. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy course. … Continue reading
Mid-Range (0.1-0.25%)
Adding 0.1% finasteride to minoxidil amplifies regrowth compared to minoxidil alone, while standalone 0.25% sprays deliver oral-level scalp DHT blockade, maintaining serum exposure roughly one-tenth that of tablets. Dose and volume are critical.[8]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading
High-Dose (0.5-1%)
Topical finasteride options at 0.5% have been shown to extend benefits to female pattern hair loss with good tolerability.[9]Rossi, A., Magri, F., D’Arino, A., Pigliacelli, F., Muscianese, M., Leoncini, P., Caro, G., Federico, A., Fortuna, M.C., Carlesimo, M. (2020). Efficacy of Topical Finasteride 0.5% vs 17ɑ-Estradiol … Continue reading
Why Formulation and Dose Matter
- The vehicle drives absorption: according to the studies, hydro-alcoholic sprays penetrate faster than gels or liposomes, explaining divergent serum DHT curves at equal nominal strengths.
- Daily drug load, not percentage alone, predicts systemic spill over; 0.025% at 400 μL approaches the systemic impact of oral dosing, whereas 100 μL does not.
- Combination therapy often allows for a lower finasteride concentration to achieve equal cosmetic benefits, thereby minimizing exposure.
Minimizing Systemic Absorption
While topical finasteride offers an option for minimizing systemic exposure, several key variables influence the amount that enters the bloodstream, and this can occur surprisingly quickly.
Key Factors Affecting Systemic Absorption
- Carrier Agent: The vehicle used to deliver finasteride dramatically alters absorption. Alcohol-based (ethosomal) carriers enhance drug penetration through the skin and into deeper layers like the dermis much more than conventional liposomes, which tend to remain more superficial. One study found that ethosomes resulted in approximately six times greater dermal accumulation than liposomes, highlighting the direct impact of carrier choice on systemic exposure.[10]Rao, Y., Zheng, F., Zhang, X., Gao, J., Liang, W. (2008). In Vitro Percutaneous Permeation and Skin Accumulation of Finasteride Using Vesicular Ethosomal Carriers. AAPS PharmSciTech. 9(3). 860-865. … Continue reading
- Time on Scalp Before Washing: The longer finasteride remains on the scalp before washing, the greater the chance for absorption through the skin. Washing off too soon reduces drug uptake, but leaving it for extended periods increases both local efficacy and systemic exposure. Guidelines often recommend allowing at least 4-6 hours on the scalp, though this can vary by formulation.
- Frequency of Use: If you can keep topical finasteride on your scalp for at least 10-12 hours, once-daily applications of low-dose topical finasteride might be beneficial. For those who can only tolerate finasteride for 4-6 hours, twice-daily applications may be better. You can read more about how often topical finasteride should be applied here.
- Individual Skin Permeability: Personal factors, including skin thickness, barrier integrity, genetics, and even scalp conditions, can strongly influence how much finasteride penetrates the scalp. Individuals with more permeable skin may absorb a greater amount of the drug systemically, increasing the risk of side effects.[11]Brito, S., Baek, M., Bin, B-H. (2024). Skin Structure, Physiology, and Pathology in Topical and Transdermal Drug Delivery. Pharmaceutics. 16(11). 1403. Available at: … Continue reading
How To Maximize Gains and Minimize Risk
To achieve the best results and reduce the likelihood of side effects, there are several strategies you can employ:
- Establish a Baseline with Serum DHT Testing
Before starting topical finasteride, get a baseline measurement of your serum DHT levels through a blood test. This serves as a reference to assess the extent to which systemic DHT is affected by your treatment.
- Retest After 30 Days
After one month of consistent topical use, repeat the serum DHT test under the same conditions (preferably in the morning, fasted, and at a similar time of day). This helps you gauge systemic absorption and adjust your regimen if your serum DHT levels drop excessively. Hormone levels fluctuate based on daily rhythms, food intake, and stress, so always test under similar circumstances: morning, fasted, and ideally before applying the day’s finasteride.
- Avoid Confounding Supplements
Avoid supplements that may directly affect DHT, such as those that increase DHT (e.g., creatine) or quercetin (which may lower it). This ensures your test results and progress are a direct reflection of the topical finasteride.[12]van der Merwe, J., Brooke, N.E., Myburgh, K.H. (2009). Three weeks of creatine monohydrate supplementation affects dihydrotestosterone to testosterone ratio in college-aged rugby players. Clinical … Continue reading,[13]Ma, Z., Nguyen, T.H., Huynh, T.H., Do, P.T., Huynh, H. (2004). Reduction of rat prostate weight by combined quercetin-finasteride treatment is associated with cell cycle deregulation. Journal of … Continue reading
- Be Patient – It Takes Time
Visible improvements often require 12-24 months. Don’t make hasty adjustments if you don’t see immediate changes; hair cycles are slow.
- Track Your Progress Objectively
Use standardized, high-quality photo documentation, same angle, lighting, and distance each time, to objectively monitor changes in hair density and coverage.
Combination Therapies
Robust evidence supports the use of combination therapies for AGA, with multi-modal approaches consistently outperforming monotherapy in terms of efficacy and speed of regrowth.
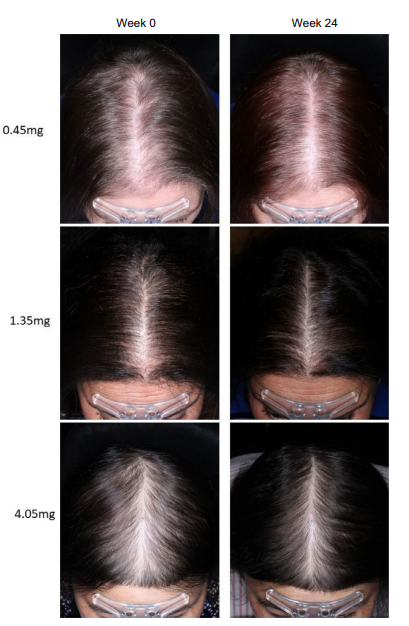
Finasteride + Minoxidil: Studies show that pairing topical finasteride with 5% minoxidil yields superior hair density and patient satisfaction compared to either agent alone, particularly after 24 weeks of treatment.[14]Apoorva, V.B., Vibhu, M., Singh, R.H., Smita, T. (2023). Comparative Efficacy of Topical Finasteride (0.25%) in Combination with Minoxidil (5%) Against 5% Minoxidil or 0.25% Finasteride Alone in Male … Continue reading,[15]Rossi, A., Caro, G. (2023). Efficacy of the association of topical minoxidil and topical finasteride compared to their use in monotherapy in men with androgenetic alopecia: A prospective, randomized, … Continue reading,[16]Abeck, F., Hansen, I., Kott, J., Schroder, F., Garrahy, E., Veneroso, J., Runger, A., Torster, L., Schneider, S.W., von Buren, J. (2024). Patient-reported outcomes of topical finasteride/minoxidil … Continue reading This combination is effective for both new users and those who have experienced shedding after discontinuing oral finasteride.
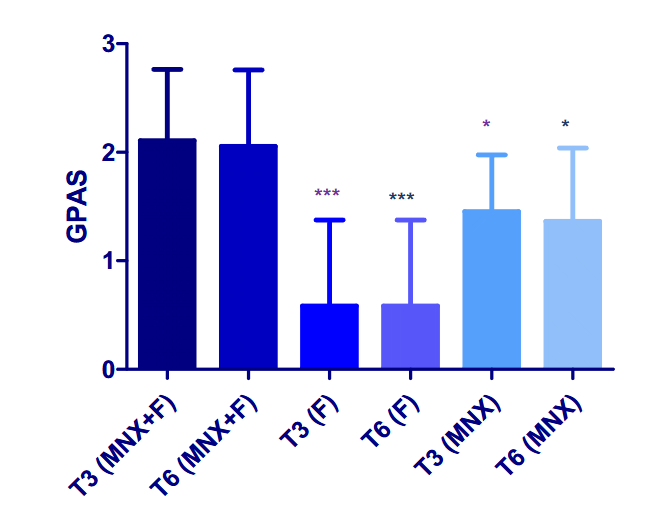
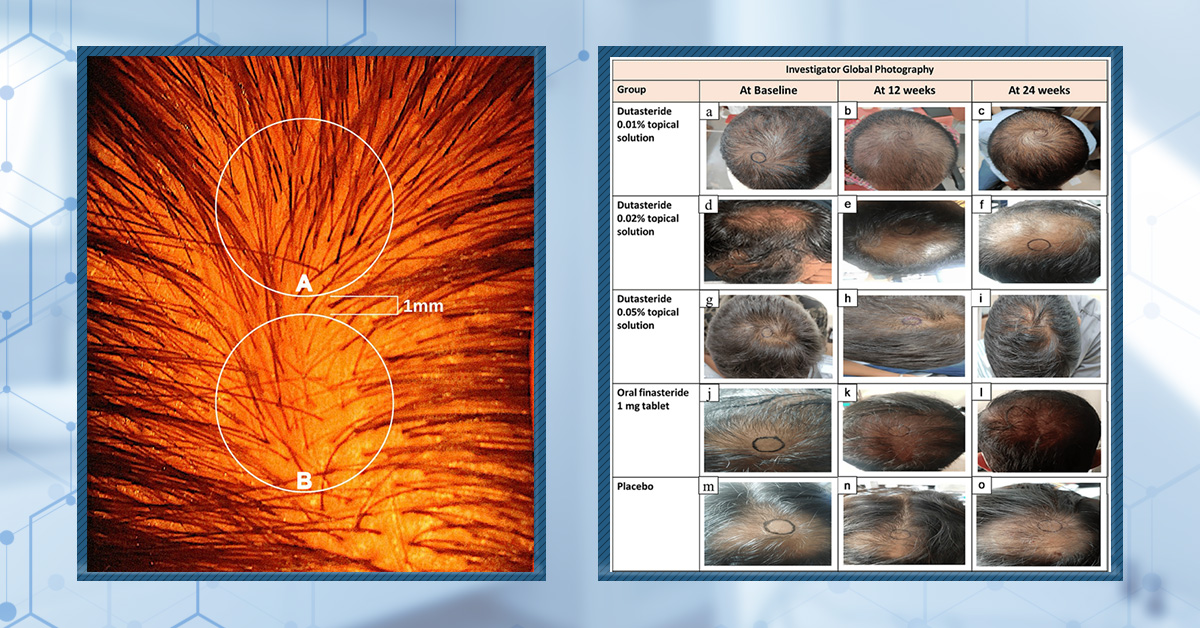
Figure 1: Effect of combination minoxidil and finasteride or monotherapy treatment on global photographic assessment score at T3 (3 months) and T6 (six months).[17]Rossi, A., Caro, G. (2023). Efficacy of the association of topical minoxidil and topical finasteride compared to their use in monotherapy in men with androgenetic alopecia: A prospective, randomized, … Continue reading Image obtained in line with the Creative Commons License.
Finasteride + Microneedling: Adding microneedling to topical therapy can significantly enhance outcomes. Clinical trials demonstrated that combining microneedling with minoxidil and/or finasteride increased hair density and shaft diameter more than minoxidil alone, with effects noticeable within just 12 weeks.[18]Chang, Y., Zhang, W., Zhou, J., Lv, L., He, Q., Chen, Y., Wang, P., Zhai, Q. (2025). Clinical efficacy of microneedle combined with 5% Minoxidil solution and finasteride in the treatment of … Continue reading,[19]Adistri, K., Sirait, S.P., Rihatmadja, R., Legiawati, L., Indriatmi, W., Saldi, S.R.F. (2024). Effectiveness and safety of the combination therapy of micro-needling and minoxidil in androgenetic … Continue reading
Triple Topical Therapy: Preliminary studies on formulations that combine finasteride, dutasteride, and minoxidil show promising results, with visible regrowth as early as three months in some cases.[20]Rafi, A.W., Katz, R.M. (2011). Pilot Study of 15 Patients Receiving a New Treatment Regimen for Androgenic Alopecia: The Effects of Atopy on AGA. ISRN Dermatology. 241953. Available at: … Continue reading
So, combination therapy appears to be more effective than monotherapy. Therefore, leveraging two or more topical therapies or adding microneedling can support hair regrowth outcomes.
Who Should (or Shouldn’t) Use Topical Finasteride?
Good Candidates:
- Men diagnosed with AGA.
- Those who tolerate oral finasteride but seek to minimize systemic risks.
Not Ideal For:
- People with hair loss from causes other than AGA (e.g., alopecia areata, telogen effluvium).
- Those who have previously shown a poor response to finasteride.
- Anyone trying to conceive, or with infants, toddlers, or pregnant individuals in close contact.
- Users who are unable or unwilling to follow consistent application routines or medical monitoring.
Final Verdict
Topical finasteride is a legitimate option for hair regrowth. When formulated and used properly, it can rival oral finasteride’s effectiveness with a lower risk of systemic side effects. Success hinges on the right delivery method, correct dilution, and consistent application. While not flawless, it excels as part of a comprehensive regimen, especially when paired with therapies like minoxidil or microneedling.
For those hesitant about oral medication, topical finasteride offers a practical, lower-risk compromise, provided users carefully follow evidence-based protocols to optimize both safety and results. Used strategically, it is a potent addition to hair loss treatment plans.
References[+]
References ↑1 Diviccaro, S., Melcangi, R.C., Giatti, S. (2019). Post-finasteride syndrome: an emerging clinical problem. Neurobiology of Stress. 12. 100209. Available at: https://doi.org/10.1016/j.ynstr.2019.100209 ↑2 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III randomized, controlled clinical trial. JEADV. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑3 Bhura, M. (2013). Comparative Analysis of Topical and Oral Finasteride in Androgenetic Alopecia: Impact on Hair Growth, Systemic Absorption, and Adverse Effects. Indian Journal of Basic and Applied Medical Research. 3(1). 436-444 ↑4 Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. Indian Journal of Dermatology, Venereology, and Leprology. 75(1). 47-51. Available at: https://doi.org/10.4103/0378-6323.45220 ↑5 Finn, D.A., Beadles-Bohling, A., Beckley, E.H., Ford, M.M., Gililland, K.R., Gorin-Meyer, R.E., Wiren, K.M. (2006). 12(1). 53-76. A New Look at the 5α-reductase Inhibitor Finasteride. CNS Drug Reviews. Available at: https://doi.org/10.1111/j.1529-3458.2006.00053.x ↑6 Suzuki, R., Satoh, H., Ohtani, H., Hori, S., Sawada, Y. (2010). Saturable Binding of Finasteride to Steroid 5ɑ-reductase as Determinant of Nonlinear Pharmacokinetics. Drug Metabolism and Pharmacokinetics. 25(2). 208-213. Available at: https://doi.org/10.2133/dmpk.25.208 ↑7 Mazzarella, F., Loconsole, F., Cammisa, A., Mastrolonardo, M., Vena, G.A. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy course. Journal of Dermatological Treatment. 8. 189-192. ↑8 Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. International Journal of Clinical Pharmacology and Therapeutics. 54(1). 19-27. Available at: https://doi.org/10.5414/CP202467 ↑9 Rossi, A., Magri, F., D’Arino, A., Pigliacelli, F., Muscianese, M., Leoncini, P., Caro, G., Federico, A., Fortuna, M.C., Carlesimo, M. (2020). Efficacy of Topical Finasteride 0.5% vs 17ɑ-Estradiol 0.05% in the Treatment of Postmenopausal Female Pattern Hair Loss: A Retrospective, Single-Blind Study of 119 Patients. Dermatology Practical & Conceptual. 10(2). E2020039. Available at: https://doi.org/10.5826/dpc.1002a39 ↑10 Rao, Y., Zheng, F., Zhang, X., Gao, J., Liang, W. (2008). In Vitro Percutaneous Permeation and Skin Accumulation of Finasteride Using Vesicular Ethosomal Carriers. AAPS PharmSciTech. 9(3). 860-865. Available at: https://doi.org/10.1208/s12249-008-9124-y ↑11 Brito, S., Baek, M., Bin, B-H. (2024). Skin Structure, Physiology, and Pathology in Topical and Transdermal Drug Delivery. Pharmaceutics. 16(11). 1403. Available at: https://doi.org/10.3390/pharmaceutics16111403 ↑12 van der Merwe, J., Brooke, N.E., Myburgh, K.H. (2009). Three weeks of creatine monohydrate supplementation affects dihydrotestosterone to testosterone ratio in college-aged rugby players. Clinical Journal of Sports Medicine. 19(5). 399-404. Available at: https://doi.org/10.1097/JSM.0b013e3181b8b52f. ↑13 Ma, Z., Nguyen, T.H., Huynh, T.H., Do, P.T., Huynh, H. (2004). Reduction of rat prostate weight by combined quercetin-finasteride treatment is associated with cell cycle deregulation. Journal of Endocrinology. 181(3). 493-507. Available at: https://doi.org/10.1677/joe.0.1810493 ↑14 Apoorva, V.B., Vibhu, M., Singh, R.H., Smita, T. (2023). Comparative Efficacy of Topical Finasteride (0.25%) in Combination with Minoxidil (5%) Against 5% Minoxidil or 0.25% Finasteride Alone in Male Androgenetic Alopecia: A Pilot, Randomized, Open-Label Study. International Journal of Trichology. 15(2). 56-62. Available at: https://doi.org/10.4103/ijt_ijt_72_22 ↑15 Rossi, A., Caro, G. (2023). Efficacy of the association of topical minoxidil and topical finasteride compared to their use in monotherapy in men with androgenetic alopecia: A prospective, randomized, controlled, assessor-blinded, 3-arm, pilot trial. Journal of Cosmetic Dermatology. 23(2). 502-509. Available at: https://doi.org/10.1111/jocd.15953 ↑16 Abeck, F., Hansen, I., Kott, J., Schroder, F., Garrahy, E., Veneroso, J., Runger, A., Torster, L., Schneider, S.W., von Buren, J. (2024). Patient-reported outcomes of topical finasteride/minoxidil treatment for male androgenetic alopecia: A retrospective study using telemedical data. Journal of Cosmetic Dermatology. 23(9). 2956-2963. Available at: https://doi.org/10.1111/jocd.16360 ↑17 Rossi, A., Caro, G. (2023). Efficacy of the association of topical minoxidil and topical finasteride compared to their use in monotherapy in men with androgenetic alopecia: A prospective, randomized, controlled, assessor-blinded, 3-arm, pilot trial. Journal of Cosmetic Dermatology. 23(2). 502-509. Available at: https://doi.org/10.1111/jocd.15953 ↑18 Chang, Y., Zhang, W., Zhou, J., Lv, L., He, Q., Chen, Y., Wang, P., Zhai, Q. (2025). Clinical efficacy of microneedle combined with 5% Minoxidil solution and finasteride in the treatment of androgenetic alopecia in males. Archives of Dermatological Research. 317(428). Available at: https://doi.org/10.1007/s00403-025-03891-y ↑19 Adistri, K., Sirait, S.P., Rihatmadja, R., Legiawati, L., Indriatmi, W., Saldi, S.R.F. (2024). Effectiveness and safety of the combination therapy of micro-needling and minoxidil in androgenetic alopecia of Indonesian men: a randomized controlled trial. Dermatology Reports. 16(3). 9945. Available at: https://doi.org/10.4081/dr.2024.9945 ↑20 Rafi, A.W., Katz, R.M. (2011). Pilot Study of 15 Patients Receiving a New Treatment Regimen for Androgenic Alopecia: The Effects of Atopy on AGA. ISRN Dermatology. 241953. Available at: https://doi.org/10.5402/2011/241953 Topical minoxidil is a first-line treatment for female pattern hair loss (FPHL) and other conditions such as telogen effluvium and traction alopecia. Many women of reproductive age use minoxidil and may wonder about its safety when trying to conceive or in early pregnancy.
While most reproductive safety discussions focus on oral or systemic medications, even topically applied drugs can raise questions about potential effects on fertility or early fetal development. Although systemic absorption from topical formulations is low, uncertainty persists regarding potential hormonal or embryonic effects.
This article examines available evidence to evaluate whether topical minoxidil poses any risk to female fertility or conception. By reviewing preclinical studies and clinical and pharmacological data, we aim to clarify whether there is any risk associated with topical minoxidil when trying to conceive.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Topical Minoxidil: Mechanism and Pharmacology
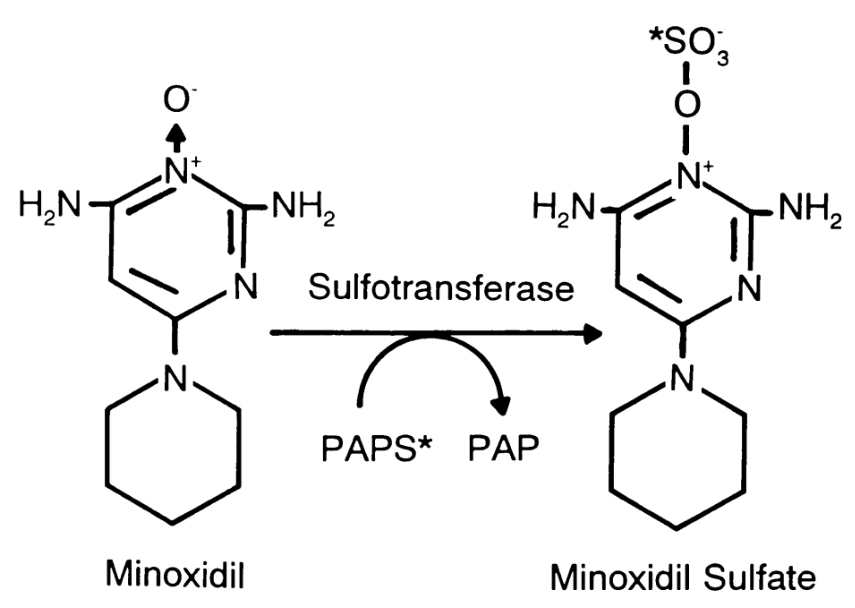
Minoxidil may promote hair growth through several complementary mechanisms. After topical application, the drug is enzymatically converted by sulfotransferase (SULT1A1) into minoxidil sulfate, its active form. This metabolite enhances potassium channel activity within follicular cells, promoting cell proliferation and prolonging the anagen (growth) phase of the hair cycle. Additionally, minoxidil increases vascularity around the follicle by stimulating vascular endothelial growth factor (VEGF), improving oxygen and nutrient delivery to support stronger, thicker hair growth.[1]Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786. Available at: … Continue reading
There are important pharmacological distinctions between topical and oral formulations. Topical minoxidil acts locally on the scalp with minimal systemic absorption, which significantly reduces the risk of adverse effects. In contrast, oral minoxidil can cause systemic side effects, such as fluid retention, weight gain, increased blood pressure (hypertension), and other cardiovascular changes.[2]StatPearls Editorial Team. (no date). Minoxidil. In: StatPearls [Internet]. StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: October 2025)
Female Fertility: What Matters When Trying to Conceive?
Female fertility relies on coordinated hormonal cycles, healthy egg development, and a receptive uterine environment. Follicle-stimulating hormone (FSH) and luteinizing hormone (LH) regulate ovulation, while estrogen and progesterone prepare the uterus for implantation and support early pregnancy.[3]Thiyagarajan, D. K., Basit, H., & Jeanmonod, R. (2024 Jan 1). Physiology, menstrual cycle. In: StatPearls [Internet]. StatPearls Publishing. Available at: … Continue reading In addition to hormonal balance, healthy reproductive anatomy is important: clear fallopian tubes, a healthy uterus, and a cervix free of significant abnormalities are required for fertilization and embryo implantation.[4]Mayo Clinic Staff. (no date). Female infertility – Symptoms & causes. Available at: https://www.mayoclinic.org/diseases-conditions/female-infertility/symptoms-causes/syc-20354308/ (Accessed: … Continue reading
Of course, women, or individuals who are female at birth, looking to conceive also need to consider the impact of medications following conception. The safe use of medications is essential to support maternal health and healthy fetal development while minimizing risks of complications and birth defects (teratogenic effects).
Systemic medications can interfere with fertility and pregnancy by disrupting hormonal balance or exerting teratogenic effects. For example, antiandrogens such as spironolactone, cyproterone acetate, and finasteride, sometimes prescribed for female hair loss, are contraindicated when trying to conceive due to their potential impact on fetal development.[5]BinJadeed, H. F., & Alajlan, A. (2021). Pregnancy and neonatal outcome with maternal exposure to finasteride: a case series. Journal of Dermatology and Dermatologic Surgery. 25(2). 84-86. … Continue reading,[6]Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. … Continue reading Animal models suggest these agents interfere with androgen signaling, which is essential for normal sexual differentiation, particularly in male fetuses.[7]Conley, J. M., Lambright, C. S., Evans, N., Cardon, M., Furr, J., Wilson, V. S., & Gray Jr, L. E. (2018). Mixed “antiandrogenic” chemicals at low individual doses produce reproductive tract … Continue reading In contrast, nutritional supplements like iron, vitamin D, and biotin are generally safe and can support overall reproductive health.
Systemic Absorption of Topical Minoxidil: How Much Gets In?
Topical minoxidil is formulated to act locally on the scalp, with only a small proportion entering systemic circulation – typically around 1–2%.[8]Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786. Available at: … Continue reading Pharmacokinetic studies show that this absorption rate can vary depending on several factors, including formulation strength (2% vs. 5%), frequency of application, and the integrity of the scalp barrier. Plasma minoxidil levels in individuals using topical formulations generally remain well below those associated with systemic pharmacologic or reproductive effects observed in animal studies.[9]Singh, S., Patil, A., Kianfar, N., Waśkiel-Burnat, A., Rudnicka, L., Sinclair, R., & Goldust, M. (2022). Does topical minoxidil at concentrations higher than 5% provide additional clinical … Continue reading,[10]Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: … Continue reading
By contrast, oral minoxidil is almost completely absorbed, leading to significantly higher plasma concentrations and systemic exposure. This explains why oral therapy, though often more potent, also carries a greater risk of systemic side effects, including edema, tachycardia, and generalized hypertrichosis.[11]Randolph, M., & Tosti, A. (2021). Oral minoxidil treatment for hair loss: a review of efficacy and safety. Journal of the American Academy of Dermatology. 84(3). 737-746. Available at: … Continue reading
Does Topical Minoxidil Affect Female Fertility, Conception, or Pregnancy?
When considering the impact of medications on women’s fertility and ability to conceive, it is also essential to review the evidence regarding potential effects during pregnancy, as treatments will inevitably continue to be taken into early pregnancy, and systemic effects may be long-lasting. Here, we review preclinical and clinical evidence on whether topical minoxidil affects female fertility and pregnancy.
Preclinical Evidence
Minoxidil is classified as a non-hormonal treatment, suggesting it is unlikely to impact hormonal cycles that impact fertility. However, due to the relationship between hair growth and hormonal pathways, some preclinical research has focused on how minoxidil affects androgen pathways.
One cell-based study suggests that minoxidil can directly interact with and weaken androgen receptor signaling. However, this study used higher concentrations of minoxidil than would be used in hair loss treatment, and for practical purposes, topical minoxidil remains classified as a non-hormonal treatment.[12]Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: … Continue reading
However, in an animal model using golden Syrian hamsters, 5% topical minoxidil did not prevent the androgen-dependent growth of pigmented spots, suggesting that minoxidil does not have any effect on androgen response.[13]Nuck, B. A., Fogelson, S. L., & Lucky, A. W. (1987). Topical minoxidil does not act as an antiandrogen in the flank organ of the golden Syrian hamster. Archives of Dermatology. 123(1). 59-61. … Continue reading
There is a significant lack of research using preclinical animal models investigating the impact of topical minoxidil on female fertility and during pregnancy. However, one small-scale study did examine the effect of 5% topical minoxidil on embryonic mortality in rats. They found no impact of daily minoxidil application over the course of 20 days of pregnancy compared to the control group. However, the sample size was relatively small (20 rats in total), and no molecular or cellular analysis was performed to look for potential mechanisms that could cause embryonic lethality.[14]Turkina, V. A., Chemodurova, N. Y., Pryzyglei, H. V., & Kuzminov, Y. B. (2021). Potential embryotoxic effect study of minoxidil-containing lotion in experiment with female rats. Світ … Continue reading
Clinical Evidence
Unfortunately, a lack of research into the impact of medications on women and fetuses during pregnancy is a systemic issue within medicine. Pregnant women are generally excluded from clinical trials due to safety concerns, as well as the confounding impact of widespread hormonal changes associated with pregnancy.[15]Stock, S. J., & Norman, J. E. (2019). Medicines in pregnancy. F1000Research. 8(F1000Faculty Rev):911. Available at: https://doi.org/10.12688/f1000research.17535.1 These issues extend to women trying to conceive, and most clinical trials have explicit protocols that remove individuals from clinical trials after they become pregnant.[16]Phelan, A. L., Kunselman, A. R., Chuang, C. H., Raja-Khan, N. T., & Legro, R. S. (2016). Exclusion of women of childbearing potential in clinical trials of type 2 diabetes medications: a review … Continue reading
The safety of medications for pregnant women and their children is typically established after drugs have come to market, through observational studies and pharmacovigilance, which looks for cases where problems have arisen. While such processes are good at spotting signals of high risk, these forms of evidence are less powerful than clinical trials and cannot detect less impactful effects.
As such, determining the safety of drugs for pregnant women is often done using scarce information. As a result, many medications are contraindicated (their use is advised against) for pregnant women due to a lack of evidence, not because of evidence.[17]Thomas, S. H., & Yates, L. M. (2012). Prescribing without evidence – pregnancy. British Journal of Clinical Pharmacology. 74(4). 691-697. Available at: … Continue reading
Multiple reviews of the safety of dermatological treatments in pregnant women advise against the use of monoxidil.[18]Koh, Y. P., Tian, E. A., & Oon, H. H. (2019). New changes in pregnancy and lactation labelling: Review of dermatologic drugs. International Journal of Women’s Dermatology. 5(4). 216-226. … Continue reading,[19]Truong, T. M., Yaghi, M., & Murase, J. (2023). Dermatologic Drug Safety in Pregnancy. Journal of Dermatology for Physician Assistants. 17(3). 1-12. Available at: … Continue reading,[20]Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777–2786. Available at: … Continue reading However, this advice appears to be based on two case studies where topical minoxidil was theorized as the cause of the birth defects. Case studies present a single individual (or group of individuals) where an event or phenomenon has occurred. They are a weak form of evidence and are typically used as the basis for larger studies.
In one of these case studies, the pregnancy of a 28-year-old woman who was applying topical minoxidil was terminated due to the presence of fetal malformation. Topical minoxidil was suggested as a possible cause of teratogenesis.[21]Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., & Moroni, F. (2003). Topically applied minoxidil may cause fetal malformation: a case report. Birth Defects Research Part A: Clinical … Continue reading The other study noted a case of a rare condition called caudal regression syndrome in a fetus where the mother had been using topical minoxidil, though other causes were also proposed.[22]Rojansky, N., Fasouliotis, S. J., Ariel, I., & Nadjari, M. (2002). Extreme caudal agenesis. Possible drug-related etiology?. Journal of the American Academy of Dermatology. 47(3). 241-245. … Continue reading These cases were published in 2002 and 2003, and both advised that further studies were warranted.
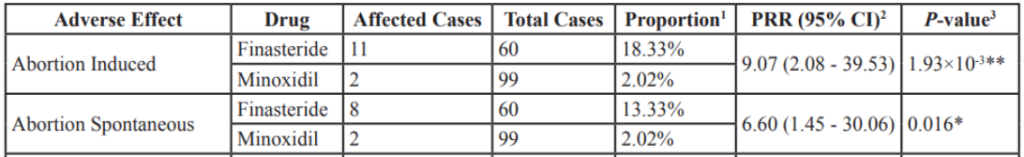
In a more systematic approach, one study analysed FDA Adverse Event Reporting System (FAERS) data, which collects evidence of adverse effects related to medications.[23]Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. … Continue reading The study used data collected between 2004 and 2014. Over the ten years analyzed, 2 cases of induced abortion and 2 cases of spontaneous abortion were reported in women using minoxidil. This represents around 2% of cases reported. For comparison, induced abortion represented around 18% of reported adverse effects related to finasteride, for which it was the most commonly reported issue. No cases related to sexual dysfunction or loss of libido were reported.
Figure 1: Comparison between adverse events reported by female alopecia areata patients exposed to finasteride and minoxidil. Adapted from Table 4.[24]Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. … Continue reading Image used in accordance with the PMC Creative Commons Licence.
It is due to the lack of evidence and the potential issues that these case studies raise that minoxidil is typically contraindicated for women who are trying to conceive or are pregnant. There is no evidence to suggest that minoxidil has any impact on female fertility. Larger observational and controlled studies would be required to make evidence-based conclusions regarding the safety of minoxidil for women trying to conceive.
Summary Consensus
There is no direct evidence linking topical minoxidil to reduced fertility. The use of topical minoxidil is advised against for women trying to conceive due to a small number of reported cases where topical minoxidil was associated with fetal abnormalities.
Counseling and Clinical Recommendations
The Food and Drug Administration (FDA) classifies topical minoxidil as a pregnancy category C drug. This means studies have shown a risk to the fetus, but there are no adequate, controlled studies in pregnant women. Such drugs are generally only prescribed if there is a significant benefit to the mother that outweighs potential risks. As such, a review published by the American Academy of Dermatology in 2025 advises against using minoxidil while pregnant.[25]Olsen, E. A., Sinclair, R., Hordinsky, M., Mesinkovska, N. A., Sadick, N., Shapiro, J., & Bergfeld, W. (2025). Summation and recommendations for the safe and effective use of topical and oral … Continue reading In the UK, the National Institute for Health and Care Excellence (NICE) also advises against the use of topical minoxidil when pregnant or breastfeeding.[26]National Institute for Health and Care Excellence. (no date). Female-pattern hair loss (female androgenetic alopecia) — Prescribing information: Topical minoxidil. Available at: … Continue reading
Does topical minoxidil cause infertility?
There is no evidence to suggest that topical minoxidil can impact fertility in women. There are a very limited number of cases reported where minoxidil has been associated with impotence in men.[27]Rietschel, R. L., & Duncan, S. H. (1987). Safety and efficacy of topical minoxidil in the management of androgenetic alopecia. Journal of the American Academy of Dermatology. 16(3). 677-685. … Continue reading
Does minoxidil interfere with female hormones or ovulation?
There is limited research into the effect of minoxidil on female hormones or ovulation. However, minoxidil is not a hormonal drug, and there is limited evidence to suggest that it interferes significantly with hormones at levels associated with topical treatment.
Should women stop minoxidil months before trying to conceive?
While there is no evidence that minoxidil can impact women’s ability to conceive, it is not advised to use the treatment while pregnant. Therefore, it is recommended to stop using minoxidil while trying to conceive.[28]Olsen, E. A., Sinclair, R., Hordinsky, M., Mesinkovska, N. A., Sadick, N., Shapiro, J., & Bergfeld, W. (2025). Summation and recommendations for the safe and effective use of topical and oral … Continue reading
Can minoxidil exposure cause miscarriage or birth defects?
There are limited case reports that associate topical minoxidil with birth defects and fetal malformation, leading to termination. These findings have not been expanded on by larger trials or observational studies.[29]Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., & Moroni, F. (2003). Topically applied minoxidil may cause fetal malformation: a case report. Birth Defects Research Part A: Clinical … Continue reading,[30]Rojansky, N., Fasouliotis, S. J., Ariel, I., & Nadjari, M. (2002). Extreme caudal agenesis. Possible drug-related etiology?. Journal of the American Academy of Dermatology. 47(3). 241-245. … Continue reading
Do all hair loss drugs have the same reproductive risks?
Different hair loss treatments have fundamentally different mechanisms. Finasteride and dutasteride are hormonally active and may impact male fertility and fetal development.[31]BinJadeed, H. F., & Alajlan, A. (2021). Pregnancy and neonatal outcome with maternal exposure to finasteride: a case series. Journal of Dermatology and Dermatologic Surgery. 25(2). 84-86. … Continue reading,[32]Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. … Continue reading Risks associated with minoxidil are significantly less well established, and adverse reproductive events have been shown to be significantly lower than finasteride.[33]Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. … Continue reading
Latest Research and Knowledge Gaps
There is a significant gap in our knowledge of whether minoxidil affects pregnancy and the mechanisms by which it might do so. Data from preclinical models has focused on hair cycle dynamics rather than reproductive parameters, with only limited research on its impact on androgenic hormonal pathways.
Both of the case studies that associated topical minoxidil with adverse pregnancy outcomes are over 20 years old, with no follow-up studies to better understand these reports. Clinical trials or observational studies would be necessary to make evidence-based assessments on the safety of minoxidil during pregnancy and its impact on female fertility.
Final Thoughts
Current evidence does not show that topical minoxidil impairs fertility or conception. However, because pregnancy safety data remain incomplete, discontinuation is recommended when planning conception – a precautionary measure rather than a proven necessity.
For women experiencing distressing hair loss, treatment decisions should balance cosmetic benefit with potential but unproven reproductive risk, ideally in consultation with a dermatologist or reproductive health specialist.
References[+]
References ↑1, ↑8 Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑2 StatPearls Editorial Team. (no date). Minoxidil. In: StatPearls [Internet]. StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: October 2025) ↑3 Thiyagarajan, D. K., Basit, H., & Jeanmonod, R. (2024 Jan 1). Physiology, menstrual cycle. In: StatPearls [Internet]. StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK500020/ (Accessed: October 2025) ↑4 Mayo Clinic Staff. (no date). Female infertility – Symptoms & causes. Available at: https://www.mayoclinic.org/diseases-conditions/female-infertility/symptoms-causes/syc-20354308/ (Accessed: October 2025) ↑5, ↑31 BinJadeed, H. F., & Alajlan, A. (2021). Pregnancy and neonatal outcome with maternal exposure to finasteride: a case series. Journal of Dermatology and Dermatologic Surgery. 25(2). 84-86. Available at: https://doi.org/10.4103/jdds.jdds_33_21 ↑6, ↑23, ↑24, ↑33 Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. Available at: https://doi.org/10.18632/oncotarget.12617 ↑7 Conley, J. M., Lambright, C. S., Evans, N., Cardon, M., Furr, J., Wilson, V. S., & Gray Jr, L. E. (2018). Mixed “antiandrogenic” chemicals at low individual doses produce reproductive tract malformations in the male rat. Toxicological Sciences. 164(1). 166-178. Available at: https://doi.org/10.1093/toxsci/kfy069 ↑9 Singh, S., Patil, A., Kianfar, N., Waśkiel-Burnat, A., Rudnicka, L., Sinclair, R., & Goldust, M. (2022). Does topical minoxidil at concentrations higher than 5% provide additional clinical benefit?. Clinical and Experimental Dermatology. 47(11). 1951-1955. Available at: https://doi.org/10.1111/ced.15338 ↑10, ↑12 Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: /https://doi.org/10.18632/oncotarget.1886 ↑11 Randolph, M., & Tosti, A. (2021). Oral minoxidil treatment for hair loss: a review of efficacy and safety. Journal of the American Academy of Dermatology. 84(3). 737-746. Available at: https://doi.org/10.1016/j.jaad.2020.06.1009 ↑13 Nuck, B. A., Fogelson, S. L., & Lucky, A. W. (1987). Topical minoxidil does not act as an antiandrogen in the flank organ of the golden Syrian hamster. Archives of Dermatology. 123(1). 59-61. Available at: https://doi.org/10.1001/archderm.1987.01660250065019 ↑14 Turkina, V. A., Chemodurova, N. Y., Pryzyglei, H. V., & Kuzminov, Y. B. (2021). Potential embryotoxic effect study of minoxidil-containing lotion in experiment with female rats. Світ медицини та біології. (2). 248-251. Available at: https://doi.org/10.26724/2079-8334-2021-2-76-248-251 ↑15 Stock, S. J., & Norman, J. E. (2019). Medicines in pregnancy. F1000Research. 8(F1000Faculty Rev):911. Available at: https://doi.org/10.12688/f1000research.17535.1 ↑16 Phelan, A. L., Kunselman, A. R., Chuang, C. H., Raja-Khan, N. T., & Legro, R. S. (2016). Exclusion of women of childbearing potential in clinical trials of type 2 diabetes medications: a review of protocol-based barriers to enrollment. Diabetes Care. 39(6). 1004-1009. Available at: https://doi.org/10.2337/dc15-2723 ↑17 Thomas, S. H., & Yates, L. M. (2012). Prescribing without evidence – pregnancy. British Journal of Clinical Pharmacology. 74(4). 691-697. Available at: https://doi.org/10.1111/j.1365-2125.2012.04332.x ↑18 Koh, Y. P., Tian, E. A., & Oon, H. H. (2019). New changes in pregnancy and lactation labelling: Review of dermatologic drugs. International Journal of Women’s Dermatology. 5(4). 216-226. Available at: https://doi.org/10.1016/j.ijwd.2019.05.002 ↑19 Truong, T. M., Yaghi, M., & Murase, J. (2023). Dermatologic Drug Safety in Pregnancy. Journal of Dermatology for Physician Assistants. 17(3). 1-12. Available at: https://doi.org/10.58744/001c.88954 ↑20 Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777–2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑21, ↑29 Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., & Moroni, F. (2003). Topically applied minoxidil may cause fetal malformation: a case report. Birth Defects Research Part A: Clinical and Molecular Teratology. 67(12). 997-1001. Available at: https://doi.org/10.1002/bdra.10095 ↑22, ↑30 Rojansky, N., Fasouliotis, S. J., Ariel, I., & Nadjari, M. (2002). Extreme caudal agenesis. Possible drug-related etiology?. Journal of the American Academy of Dermatology. 47(3). 241-245. Available at: https://pubmed.ncbi.nlm.nih.gov/11933692/ ↑25, ↑28 Olsen, E. A., Sinclair, R., Hordinsky, M., Mesinkovska, N. A., Sadick, N., Shapiro, J., & Bergfeld, W. (2025). Summation and recommendations for the safe and effective use of topical and oral minoxidil. Journal of the American Academy of Dermatology. 93(2). 457-465. Available at: https://doi.org/10.1016/j.jaad.2025.04.016 ↑26 National Institute for Health and Care Excellence. (no date). Female-pattern hair loss (female androgenetic alopecia) — Prescribing information: Topical minoxidil. Available at: https://cks.nice.org.uk/topics/female-pattern-hair-loss-female-androgenetic-alopecia/prescribing-information/topical-minoxidil/ (Accessed: October 2025) ↑27 Rietschel, R. L., & Duncan, S. H. (1987). Safety and efficacy of topical minoxidil in the management of androgenetic alopecia. Journal of the American Academy of Dermatology. 16(3). 677-685. Available at: https://doi.org/10.1016/S0190-9622(87)70087-5 ↑32 Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. Available at: https://doi.org/10.18632/oncotarget.12617 Topical minoxidil is one of the most widely used treatments for androgenic alopecia (AGA) in both men and women. While its efficacy and safety as a hair growth stimulant are well established, some men express concern about whether continuing hair loss treatments is safe when trying to conceive. These concerns often stem from known reproductive warnings associated with other medications, particularly finasteride, which directly influences hormonal pathways. Minoxidil, however, works through entirely different mechanisms, enhancing blood flow, prolonging the hair growth phase, and stimulating follicular activity.
This article examines the available scientific evidence to determine whether topical minoxidil has any measurable effect on male fertility. By reviewing preclinical studies, clinical trials, and pharmacologic data, we aim to clarify whether there is any credible link between minoxidil use and changes in reproductive hormones, sperm quality, or conception outcomes.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Does Minoxidil Work?
Topical minoxidil stimulates hair growth primarily by increasing blood flow and nutrient delivery to hair follicles. Once applied to the scalp, it’s converted by the enzyme sulfotransferase (SULT1A1) into its active metabolite, minoxidil sulfate, which promotes follicular cell proliferation and prolongs the anagen (growth) phase of the hair cycle. It also upregulates vascular endothelial growth factor (VEGF) and activates potassium channels, both of which create a more favorable microenvironment for hair growth.[1]Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786. Available at: … Continue reading
Many people choose topical minoxidil over oral formulations because it offers similar local benefits with minimal systemic absorption. This greatly reduces the risk of side effects such as water retention, hypertrichosis (excess body hair), or cardiovascular symptoms sometimes seen with oral minoxidil. As an over-the-counter option with a well-established safety profile, topical minoxidil remains the most accessible first-line treatment for AGA.
Male Fertility: What Matters When Trying to Conceive?
Male fertility is defined as the ability to produce viable sperm capable of fertilizing an egg, and is impacted by a wide range of factors. These include hormonal regulation of sperm production, urogenital abnormalities, genetic factors, and environmental factors such as diet, obesity, and cigarette smoking. Semen quality is the best single snapshot of male reproductive potential. Sperm count influences the statistical odds that enough sperm reach the egg; sperm motility determines whether sperm can traverse cervical mucus, the uterus, and the fallopian tube; and morphology reflects the proportion of intact sperm likely to bind and penetrate the egg. These parameters work together, so clinicians interpret them as a profile rather than in isolation.[2]StatPearls; Male Infertility. Available at: https://www.ncbi.nlm.nih.gov/books/NBK562258/ (Accessed: October 2025)
Importantly, medications or drugs can influence many aspects of sperm production and function. Certain prescription drugs, such as anabolic steroids, testosterone supplements, finasteride, spironolactone, and some antidepressants or antihypertensives, can suppress the hormonal pathways that regulate spermatogenesis, reducing sperm count or motility. Others may interfere with sperm maturation, DNA integrity, or seminal fluid composition, leading to poorer fertilization potential. Even topical or seemingly localized medications can, in rare cases, exert mild systemic effects that alter reproductive hormones or oxidative balance.[3]Semet, M., Paci, M., Saïas‐Magnan, J., Metzler‐Guillemain, C., Boissier, R., Lejeune, H., & Perrin, J. (2017). The impact of drugs on male fertility: a review. Andrology. 5(4). 640-663. … Continue reading
For this reason, men actively trying to conceive are often advised to review all current medications and supplements with a clinician to assess potential reproductive impacts and, if necessary, explore safer alternatives or temporary adjustments.
Systemic Absorption of Topical Minoxidil: How Much Gets In?
Topical minoxidil is designed to act locally on the scalp, with only a small fraction of the drug entering the bloodstream. Studies estimate that around 1-2% of applied minoxidil is systemically absorbed through intact skin, though this can vary depending on factors such as scalp condition, formulation type, and frequency of use.
For instance, alcohol-based liquid solutions may enhance penetration slightly more than foam preparations, while damaged or inflamed skin can increase absorption. The scalp’s stratum corneum serves as the main barrier, limiting systemic exposure and ensuring that most of the drug remains localized to hair follicles.[4]Gupta, A. K., Talukder, M., Venkataraman, M., & Bamimore, M. A. (2022). Minoxidil: a comprehensive review. Journal of Dermatological Treatment. 33(4). 1896-1906. Available at: … Continue reading
In contrast, oral minoxidil is fully absorbed into systemic circulation and metabolized in the liver, resulting in significantly higher plasma concentrations of active minoxidil sulfate. This explains why oral therapy, though often more potent, also carries a greater risk of systemic side effects, including edema, tachycardia, and generalized hypertrichosis.[5]Randolph, M., & Tosti, A. (2021). Oral minoxidil treatment for hair loss: a review of efficacy and safety. Journal of the American Academy of Dermatology. 84(3). 737-746. Available at: … Continue reading Topical use, by comparison, delivers similar follicular benefits with a much lower risk profile, making it the preferred option for those seeking targeted hair regrowth.
Does Topical Minoxidil Affect Male Fertility?
Topical minoxidil might raise concerns for men trying to conceive, as its activity and impact are related to hormonally sensitive pathways. To explore this question properly, we can look at the evidence from laboratory models, human studies, and systematic analyses to understand any potential risks.
Preclinical Evidence
Other hair loss treatments, such as finasteride, act on hormonal pathways to alter the activity of androgens like testosterone. They may therefore pose a risk to male fertility. As such, it’s important to establish the impact of minoxidil on hormonal factors such as androgen response.
Animal models, particularly those involving rodents and primates, have primarily examined hair cycle dynamics rather than reproductive parameters. These studies consistently show that minoxidil promotes the transition of hair follicles from the telogen (resting) to anagen (growth) phase, enlarges follicular structures, and increases local blood flow.
One study using golden Syrian hamsters showed that 5% topical minoxidil did not prevent the androgen-dependent growth of pigmented spots, suggesting that minoxidil does not have any effect on androgen response.[6]Nuck, B. A., Fogelson, S. L., & Lucky, A. W. (1987). Topical minoxidil does not act as an antiandrogen in the flank organ of the golden Syrian hamster. Archives of Dermatology. 123(1). 59-61. … Continue reading
In contrast, a more recent, small-scale study using rats investigated the impact of topical minoxidil on sperm health. They found that the experimental group, receiving 10% minoxidil, had a 12-13% decline in testicular weight compared to the controls, as well as increases in markers related to oxidative stress within the testicles.[7]Turkina, V., Chemodurova, N., Hrushka, O., & Pryzyhlei, H. (2020). Topical effect of minoxidil-containing lotion on morphofunctional indicators of male rats’ reproductive system. … Continue reading Unfortunately, the authors do not specify the volume of 10% minoxidil used, making it impossible to make a concrete comparison to normal dosing in humans. This makes the translatability of these toxicology studies more challenging.
Research using cell-based models, including human hair dermal papilla cells, suggests that minoxidil can directly interact with and weaken androgen receptor signaling. However, this study used higher concentrations of minoxidil than would be used in hair loss treatment, and for practical purposes, topical minoxidil remains classified as a non-hormonal treatment.[8]Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: … Continue reading
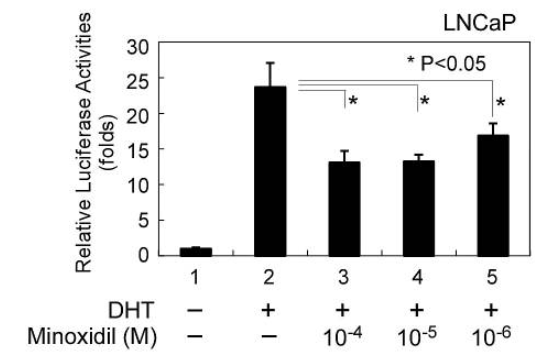
Figure 1: In vitro studies show a potential effect of minoxidil on androgen receptor activity. Adapted from:[9]Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: … Continue reading Image used in accordance with the Creative Commons Attribution Licence.
Clinical Evidence
While preclinical studies can provide insights into minoxidil activity, evidence for its effect and potential risks should be derived primarily from studies in humans. In clinical settings, topical minoxidil has been studied extensively for AGA, with trials ranging from short-term safety assessments to large randomized controlled trials. Across numerous studies, using 2%, 3%, 5%, and even 10% topical concentrations, the most commonly reported adverse events have been mild scalp irritation, contact dermatitis, or unwanted hair growth on non-target areas.[10]Olsen, E. A., Dunlap, F. E., Funicella, T., Koperski, J. A., Swinehart, J. M., Tschen, E. H., & Trancik, R. J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical … Continue reading,[11]Hasanzadeh, H., Nasrollahi, S. A., Halavati, N., Saberi, M., & Firooz, A. (2016). Efficacy and safety of 5% minoxidil topical foam in male pattern hair loss treatment and patient satisfaction. … Continue reading,[12]Ghonemy, S., Alarawi, A., & Bessar, H. (2021). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a … Continue reading
One randomized trial of 149 subjects reported two cases of impotence during the study, which disappeared following discontinuation of minoxidil treatment.[13]Rietschel, R. L., & Duncan, S. H. (1987). Safety and efficacy of topical minoxidil in the management of androgenetic alopecia. Journal of the American Academy of Dermatology. 16(3). 677-685. … Continue reading This was the only case of sexual side effects we could find, and it seems to represent a rare complaint. Notably, however, none of these trials have reported reproductive or hormonal disturbances.
One randomized, controlled study that compared the impact of topical minoxidil alone to a solution containing 0.25% finasteride and 3% minoxidil explicitly monitored sexual side effects. The trial assessed levels of dihydrotestosterone (DHT), a marker for androgen suppression that may be relevant to male fertility. The report found a modest decrease in DHT only in the group who also applied finasteride, suggesting that minoxidil did not impact androgen activity.[14]Suchonwanit, P., Srisuwanwattana, P., Chalermroj, N., & Khunkhet, S. (2018). A randomized, double-blind controlled study of the efficacy and safety of topical solution of 0.25% finasteride … Continue reading
The largest comparative analysis to date, using the FDA Adverse Event Reporting System (FAERS) data, reinforces these findings. Over ten years of reporting revealed that finasteride was associated with markedly higher rates of erectile dysfunction, decreased libido, and ejaculatory disorders. In contrast, minoxidil, both topical and oral, was not associated with reproductive toxicity in either men or women. Dermatologic reactions such as irritation or dermatitis were the only consistent signals. This post-marketing evidence, covering thousands of patients, supports the conclusion that minoxidil does not interfere with male sexual function or fertility.[15]Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. … Continue reading
It is worth noting that most clinical studies do not measure semen parameters, testosterone, or dihydrotestosterone (DHT) levels in participants, so while there is no evidence of harm, data on direct fertility endpoints remain limited. However, trials that have assessed hormonal changes indirectly, such as topical finasteride-minoxidil combinations, report only minimal and clinically insignificant reductions in DHT, well within physiologic ranges and unlikely to impact spermatogenesis. Collectively, this body of evidence supports that topical minoxidil, when used at therapeutic concentrations, exerts its effects locally on hair follicles without meaningful systemic hormonal consequences.
Comparison to Other Hair Loss Treatments
Compared with other medical treatments for hair loss, topical minoxidil has a distinctly favorable reproductive safety profile. Finasteride and dutasteride, both 5-α-reductase inhibitors, work by suppressing the conversion of testosterone to DHT, a potent androgen involved in follicular miniaturization. While effective, this mechanism directly influences sex hormone pathways and has been associated in some users with reduced libido, erectile dysfunction, decreased semen volume, and abnormal sperm morphology.[16]Gupta, A. K., Venkataraman, M., Talukder, M., & Bamimore, M. A. (2022). Finasteride for hair loss: a review. Journal of Dermatological Treatment. 33(4). 1938-1946. Available at: … Continue reading,[17]Estill, M. C., Ford, A., Omeira, R., & Rodman, M. (2023). Finasteride and dutasteride for the treatment of male androgenetic alopecia: a review of efficacy and reproductive adverse effects. … Continue reading
Topical minoxidil, in contrast, exerts no effect on androgen synthesis or receptor binding, acts locally, and is absorbed systemically at rates of roughly 1-2%. Clinical and pharmacologic studies have not detected significant changes in serum hormones, testicular function, or sperm parameters associated with its use. For men attempting to conceive, topical minoxidil is therefore considered the safer option when pharmacologic hair restoration is desired.
Summary Consensus
No systematic reviews or meta-analyses have found an association between topical minoxidil and male reproductive dysfunction. Across clinical trials and pharmacovigilance datasets (such as FAERS), the consensus remains that topical minoxidil poses minimal to no risk to male fertility. Its non-hormonal mechanism stands in contrast to antiandrogenic drugs like finasteride or dutasteride, which are known to suppress DHT levels and can cause sexual side effects in some users.
The absence of evidence of reproductive toxicity for topical minoxidil is notable given its widespread use over several decades. Still, it is important to acknowledge that the absence of evidence is not definitive proof of absence, and few studies have directly measured sperm quality or fertility outcomes, leaving a modest evidence gap for future research.
Counseling and Clinical Recommendations
Professional society guidelines, including those from the American Academy of Dermatology (AAD), European Academy of Dermatology and Venereology (EADV), and National Institute for Health and Care Excellence (NICE), all recognize topical minoxidil as a first-line treatment for AGA. None include warnings about male fertility or recommend discontinuing treatment when trying to conceive. Recommendations against use in pregnancy and breastfeeding are directed exclusively at women due to potential fetal exposure, not paternal risk.[18]Olsen, E. A., Sinclair, R., Hordinsky, M., Mesinkovska, N. A., Sadick, N., Shapiro, J., & Bergfeld, W. (2025). Summation and recommendations for the safe and effective use of topical and oral … Continue reading
In clinical practice, dermatologists generally reassure male patients that topical minoxidil can be safely continued when planning conception. They may, however, emphasize good application hygiene, using the prescribed dose, applying only to the scalp, washing hands afterward, and allowing the product to dry fully before physical contact to prevent unintended transfer to partners or infants. Because systemic absorption is minimal, these precautions are primarily to avoid topical irritation rather than systemic effects.
Myths and Misconceptions About Topical Minoxidil and Fertility
Given the links between minoxidil activity and hormonally sensitive pathways related to hair loss, some men may have concerns about using the medication when trying to conceive. Let’s look at some of the potential misconceptions about topical minoxidil and fertility.
Topical minoxidil causes infertility
There is some limited evidence from animal studies suggesting that topical minoxidil may affect sperm quality.[19]Turkina, V., Chemodurova, N., Hrushka, O., & Pryzyhlei, H. (2020). Topical effect of minoxidil-containing lotion on morphofunctional indicators of male rats’ reproductive system. … Continue reading However, there is no evidence that topical minoxidil reduces sperm count, motility, or morphology. Clinical trials of 3%, 5%, and 10% minoxidil report vanishingly few reproductive or hormonal side effects. The drug acts locally on the scalp, with systemic absorption of only about 1–2%, far below levels that could influence reproductive physiology. Neither animal studies nor human trials have identified any fertility-related toxicity associated with topical minoxidil use.
Topical minoxidil alters male hormone levels
Studies suggest that minoxidil does not interfere with androgen production, metabolism, or receptor binding at doses relevant for hair loss. It promotes hair growth through vascular and cellular mechanisms, such as increasing VEGF expression and opening potassium channels, not by modifying testosterone or dihydrotestosterone (DHT) pathways.
However, some research using cell-based models has suggested that minoxidil might impact androgen receptor activity at high concentrations.[20]Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: … Continue reading
Topical minoxidil causes erectile dysfunction
Sexual side effects are very rarely reported in any major clinical trials or pharmacovigilance data concerning topical minoxidil, with only two cases of impotence associated with topical minoxidil reported in the literature. Erectile dysfunction, decreased libido, and ejaculatory changes are associated with oral 5-α-reductase inhibitors (such as finasteride), not with topical minoxidil.[21]Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. … Continue reading
Fertility risks are the same for all hair loss drugs
Different hair loss treatments have fundamentally different mechanisms. Finasteride and dutasteride lower DHT and can influence sexual function and semen parameters in some users.[22]Estill, M. C., Ford, A., Omeira, R., & Rodman, M. (2023). Finasteride and dutasteride for the treatment of male androgenetic alopecia: a review of efficacy and reproductive adverse effects. … Continue reading Minoxidil, on the other hand, does not interact with the androgen pathway and has no known reproductive risks. Therefore, the fertility profile of topical minoxidil is significantly safer than that of antiandrogenic agents.
Final Thoughts
Topical minoxidil remains one of the safest and most accessible treatments for male pattern hair loss, with decades of research supporting its efficacy and tolerability. Unlike hormone-modulating therapies such as finasteride, minoxidil works through non-androgenic pathways and shows no evidence of affecting male fertility, hormone levels, or sexual function. While future studies could further clarify long-term reproductive outcomes, the current consensus is clear: topical minoxidil is a fertility-neutral, first-line option for men looking to manage hair loss safely.
References[+]
References ↑1 Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑2 StatPearls; Male Infertility. Available at: https://www.ncbi.nlm.nih.gov/books/NBK562258/ (Accessed: October 2025) ↑3 Semet, M., Paci, M., Saïas‐Magnan, J., Metzler‐Guillemain, C., Boissier, R., Lejeune, H., & Perrin, J. (2017). The impact of drugs on male fertility: a review. Andrology. 5(4). 640-663. Available at: https://doi.org/10.1111/andr.12366 ↑4 Gupta, A. K., Talukder, M., Venkataraman, M., & Bamimore, M. A. (2022). Minoxidil: a comprehensive review. Journal of Dermatological Treatment. 33(4). 1896-1906. Available at: https://doi.org/10.1080/09546634.2021.1945527 ↑5 Randolph, M., & Tosti, A. (2021). Oral minoxidil treatment for hair loss: a review of efficacy and safety. Journal of the American Academy of Dermatology. 84(3). 737-746. Available at: https://doi.org/10.1016/j.jaad.2020.06.1009 ↑6 Nuck, B. A., Fogelson, S. L., & Lucky, A. W. (1987). Topical minoxidil does not act as an antiandrogen in the flank organ of the golden Syrian hamster. Archives of Dermatology. 123(1). 59-61. Available at: https://doi.org/10.1001/archderm.1987.01660250065019 ↑7, ↑19 Turkina, V., Chemodurova, N., Hrushka, O., & Pryzyhlei, H. (2020). Topical effect of minoxidil-containing lotion on morphofunctional indicators of male rats’ reproductive system. Український журнал сучасних проблем токсикології. (2). 27-31. Available at: https://protox.medved.kiev.ua/index.php/en/issues/2020/2/item/633-topical-effect-of-minoxidil-containing-lotion-on-morphofunctional-indicators-of-male-rats-reproductive-system ↑8 Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: /https://doi.org/10.18632/oncotarget.1886 ↑9, ↑20 Hsu, C. L., Liu, J. S., Lin, A. C., Yang, C. H., Chung, W. H., & Wu, W. G. (2014). Minoxidil may suppress androgen receptor‐related functions. Oncotarget. 5(8). 2187-2197. Available at: /https://doi.org/10.18632/oncotarget.1886 ↑10 Olsen, E. A., Dunlap, F. E., Funicella, T., Koperski, J. A., Swinehart, J. M., Tschen, E. H., & Trancik, R. J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical minoxidil and placebo in the treatment of androgenetic alopecia in men. Journal of the American Academy of Dermatology. 47(3). 377-385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑11 Hasanzadeh, H., Nasrollahi, S. A., Halavati, N., Saberi, M., & Firooz, A. (2016). Efficacy and safety of 5% minoxidil topical foam in male pattern hair loss treatment and patient satisfaction. Acta Dermatovenerol Alp Pannonica Adriat. 25(3). 41-44. Available at: https://doi.org/10.15570/actaapa.2016.12 ↑12 Ghonemy, S., Alarawi, A., & Bessar, H. (2021). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic evaluation. Journal of Dermatological Treatment. 32(2). 236-241. Available at: https://doi.org/10.1080/09546634.2019.1654070 ↑13 Rietschel, R. L., & Duncan, S. H. (1987). Safety and efficacy of topical minoxidil in the management of androgenetic alopecia. Journal of the American Academy of Dermatology. 16(3). 677-685. Available at: https://doi.org/10.1016/S0190-9622(87)70087-5 ↑14 Suchonwanit, P., Srisuwanwattana, P., Chalermroj, N., & Khunkhet, S. (2018). A randomized, double-blind controlled study of the efficacy and safety of topical solution of 0.25% finasteride admixed with 3% minoxidil vs. 3% minoxidil solution in the treatment of male androgenetic alopecia. Journal of the European Academy of Dermatology and Venereology. 32(12). 2257-2263. Available at: https://doi.org/10.1111/jdv.15171 ↑15, ↑21 Wu, M., Yu, Q., & Li, Q. (2016). Differences in reproductive toxicology between alopecia drugs: an analysis on adverse events among female and male cases. Oncotarget. 7(50). 82074-82084. Available at: https://doi.org/10.18632/oncotarget.12617 ↑16 Gupta, A. K., Venkataraman, M., Talukder, M., & Bamimore, M. A. (2022). Finasteride for hair loss: a review. Journal of Dermatological Treatment. 33(4). 1938-1946. Available at: https://doi.org/10.1080/09546634.2021.1959506 ↑17 Estill, M. C., Ford, A., Omeira, R., & Rodman, M. (2023). Finasteride and dutasteride for the treatment of male androgenetic alopecia: a review of efficacy and reproductive adverse effects. Georgetown Medical Review. 7(1). Available at: https://doi.org/10.52504/001c.88531 ↑18 Olsen, E. A., Sinclair, R., Hordinsky, M., Mesinkovska, N. A., Sadick, N., Shapiro, J., & Bergfeld, W. (2025). Summation and recommendations for the safe and effective use of topical and oral minoxidil. Journal of the American Academy of Dermatology. 93(2). 457-465. Available at: https://doi.org/10.1016/j.jaad.2025.04.016 ↑22 Estill, M. C., Ford, A., Omeira, R., & Rodman, M. (2023). Finasteride and dutasteride for the treatment of male androgenetic alopecia: a review of efficacy and reproductive adverse effects. Georgetown Medical Review. 7(1). Available at: https://doi.org/10.52504/001c.88531 Finasteride is widely used for androgenic alopecia (AGA) and benign prostate hyperplasia (BPH). It’s generally well tolerated, with most users experiencing no significant adverse effects. However, clinicians in eye clinics have reported occurrences of dry eye that could be related to the use of finasteride.[1]NZ Optics. (2022). Finasteride and dry eye disease. Available at: https://www.nzoptics.co.nz/live-articles/finasteride-and-dry-eye-disease/ (Accessed: October 2025)
Dry eye disease (DED) is a common eye disorder characterized by insufficient tear production or poor tear quality. This can cause inadequate lubrication of the eye’s surface, leading to discomfort, irritation, and potentially damage to the eye. The condition is multifactorial, meaning it can be caused by a number of different factors. One important cause is increased tear evaporation, leading to evaporative dry eye. The quality and structure of tears are regulated by hormonal pathways, meaning that the anti-androgenic activity of finasteride could drive the development of the condition.[2]Golden, M. I., Meyer, J. J., Zeppieri, M., & Patel, B. C. (2024). Dry Eye Syndrome. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: … Continue reading
In this article, we will examine the research that links finasteride to dry eye disease and explore the potential mechanisms by which it may be contributing to the condition.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Finasteride and the Eye
Finasteride is a 5α-reductase inhibitor and works by reducing the conversion of testosterone to dihydrotestosterone (DHT). DHT drives the shrinking and weakening of hair follicles in AGA, meaning that finasteride activity can counteract these effects by decreasing the amount of DHT. Finasteride is typically administered orally, meaning it can have systemic effects on multiple parts of the body. This is particularly important in tissues that are sensitive to androgens like DHT.[3]Zito, P. M., Bistas, K. G., Patel, P., & Syed, K. (2024). Finasteride. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: … Continue reading
Figure 1. 5α-reductase converts testosterone into dihydrotestosterone. Finasteride is a 5α-reductase inhibitor, meaning it prevents or reduces this activity. Image adapted from Figure 1.[4]Azizi, A., Mumin, N. H., & Shafqat, N. (2021). Phytochemicals with anti 5-alpha-reductase activity: A prospective for prostate cancer treatment. F1000Research. 10. 221. Available at: … Continue reading Image used under Creative Commons License.
The meibomian glands are one such tissue. They are special sebaceous (oil-producing) glands located within the upper and lower eyelids, with each eye containing 20 – 40 of these glands. They produce meibum, a lipid-rich secretion that forms the outermost layer of the tear film and plays a critical role in maintaining ocular surface health. With every blink, meibum is expressed from the gland orifices, forming a thin, uniform layer over the eye. Meibum’s main function is to prevent evaporation of the tear film by creating a stable oil barrier, ensuring long-lasting lubrication of the eye’s surface.[5]Sabeti, S., Kheirkhah, A., Yin, J., & Dana, R. (2020). Management of meibomian gland dysfunction: a review. Survey of Ophthalmology. 65(2). 205-217. Available at: … Continue reading
Figure 2. The meibomian glands are arranged in parallel rows within the upper and lower eyelids. They produce meibum, which forms the lipid layer of the tear that coats the surface of the eye. Image adapted from Figure 3.[6]Dhamdhere, K., & Badawi, D. (2021). A blink-assisted, cornea-sparing wearable eyelid device for the effective penetration of therapeutic thermal energy into the meibomian glands for the treatment … Continue reading Image used under Creative Commons License.
Meibomian glands are directly regulated by androgens, via receptors present in their epithelial cells. Androgens, such as DHT, can bind to these receptors, directly influencing the synthesis of lipids essential for tear formation. Importantly, DHT is also synthesized in meibomian glands by 5α-reductase. This is where the connection to finasteride comes in: inhibition of 5α-reductase by the drug could stop DHT activity in the meibomian glands, stopping the production of lipids required to prevent tear evaporation. As such, while finasteride doesn’t target the eye, its impact on androgen pathways can have indirect effects.[7]Traish, A. M. (2020). Health risks associated with long-term finasteride and dutasteride use: it’s time to sound the alarm. The World Journal of Men’s Health. 38(3). 323-337. Available at: … Continue reading
Even partial androgen suppression, like oral finasteride for hair, could be enough to impact tear production, particularly in someone who has other risk factors associated with DED, such as older age, prolonged screen time, certain medications (antihistamines, antidepressants, diuretics, beta-blockers), autoimmune or connective tissue diseases, and environmental exposures such as wind, dry climates, or smoke.
Do DHT-Reducing Therapies Lead to Dry Eye?
A role for androgens in regulating meibomian gland function was proposed in a paper published in 2000.[8]Krenzer, K. L., Reza Dana, M., Ullman, M. D., Cermak, J. M., Tolls, D. B., Evans, J. E., & Sullivan, D. A. (2000). Effect of androgen deficiency on the human meibomian gland and ocular surface. … Continue reading Patients who were taking antiandrogen drugs for issues related to the prostate were assessed on symtpoms of DED and the contents of their meibomian gland secretions. They were compared to a control group that was on such medications. They found that antiandrogen therapy changes the composition of lipids in tears, while also decreasing the quality of tears and increasing participants’ susceptibility to light sensitivity, painful eyes, and blurred vision. This study laid the foundation for our current understanding of how DHT-lowering therapies might cause DED.
Preclinical Data
Animal models have been developed to understand the mechanisms that lead to DED, including a potential role for DHT-lowering therapies as suggested by these clinical findings. A 2014 study utilized a rat model to investigate the impact of finasteride on tear production.[9]Singh, S., Moksha, L., Sharma, N., Titiyal, J. S., Biswas, N. R., & Velpandian, T. (2014). Development and evaluation of animal models for sex steroid deficient dry eye. Journal of … Continue reading They found that oral administration of 1.6 mg/kg finasteride every day significantly decreased tear flow and increased the speed at which tears break up after 10 days in both male and female rats.
This study was supported by further research published in 2017, which used the same concentration of finasteride in rats for 40 days.[10]Li, K., Zhang, C., Yang, Z., Wang, Y., & Si, H. (2017). Evaluation of a novel dry eye model induced by oral administration of finasteride. Molecular Medicine Reports. 16(6). 8763-8770. Available … Continue reading They also found that tear production was reduced in finasteride-treated rats, and makers for tear break-up were increased. The impact of finasteride on tear production in rats is so reliable that it has been used to induce DED in models investigating potential treatments for the condition.[11]Zhang, C., Li, K., Yang, Z., Wang, Y., & Si, H. (2016). The effect of the aqueous extract of Bidens pilosa L. on androgen deficiency dry eye in rats. Cellular Physiology and Biochemistry. 39(1). … Continue reading
Figure 3. Finasteride treatment (Fin – right-hand group) increases fluorescein staining in the tears of rats, an indicator of tear break-up. Adapted from Figure 1.[12]Li, K., Zhang, C., Yang, Z., Wang, Y., & Si, H. (2017). Evaluation of a novel dry eye model induced by oral administration of finasteride. Molecular Medicine Reports. 16(6). 8763-8770. Available … Continue reading Image used under Creative Commons License.
Notably, these animal models use considerably higher concentrations of finasteride than would be recommended for hair loss – an equivalent dose for the average American man would be around 126 mg per day. In comparison, the standard regimen for AGA is only 1 mg per day. However, they do demonstrate the capacity for finasteride to impact tear production.
Clinical Data
Preclinical data, as well as a deeper understanding of the androgen-associated causes of DED, led to some concern about the potential adverse effects of finasteride.[13]Traish, A. M. (2020). Health risks associated with long-term finasteride and dutasteride use: it’s time to sound the alarm. The World Journal of Men’s Health. 38(3). 323-337. Available at: … Continue reading
Recently, clinical researchers have further explored this link. A retrospective case series looked at 116 patients at an eye clinic with a history of DED who were taking finasteride.[14]Nguyen, B. J., Meer, E., Gupta, A. S., Jinpeng, G., Ying, G. S., Bunya, V. Y., Macchi, I., & Massaro-Giordano, M. (2022). The effect of finasteride on dry eye disease. Investigative Ophthalmology … Continue reading
They found that a significantly greater proportion of patients had MGD, conjunctival abnormalities, and corneal abnormalities during follow-up examinations compared to their initial assessment, a period of four and a half years on average.
They also found a trend toward increased disease severity in patients on a higher dose of finasteride, although these findings were not statistically significant. It is hard to draw any conclusions from this research, as it only looked at individuals who both took finasteride and had DED. A retrospective study or clinical trial that included individuals who did not take finasteride would provide significantly greater insights.
Anecdotal reports from an eye clinic in New Zealand have indicated that increasingly, patients presenting with DED are also taking finasteride.[15]NZ Optics. (2022). Finasteride and dry eye disease. Available at: https://www.nzoptics.co.nz/live-articles/finasteride-and-dry-eye-disease/ (Accessed: October 2025) Data on the impact of finasteride on DED is, therefore, lacking, and robust clinical studies are required to better understand the potential link.
Some conclusions can also be drawn from clinical studies investigating other antiandrogenic drugs. Such medications are often used against prostate cancer, which depends on androgens such as testosterone and DHT for its growth and survival. A retrospective study compared a group of 1791 patients with prostate cancer who were taking a range of androgen deprivation therapies with those who weren’t.[16]Chien, H. W., Lin, C. W., Lee, C. Y., Huang, J. Y., Yang, S. F., & Wang, K. (2022). The use of androgen deprivation therapy for prostate cancer and its effect on the subsequent dry eye disease: a … Continue reading They found no significant difference in the development of DED.
A similar study compared a smaller group of prostate cancer patients on antiandrogens (31 in total) to those not on such medications. However, it investigated tear formation in greater depth.[17]Kurna, S. A., Hacisalihoglu, A. O., Altun, A., Ozel, N. O., Uruc, F., Kanar, H. S., & Arsan, A. K. (2022). Effects of systemic anti-androgen drugs on the ocular surface. Journal Français … Continue reading
They found a significant increase in markers for tear break-up and complaints of dry eye. Importantly, neither of these studies distinguishes between forms of antiandrogen therapy, nor do they specifically investigate the role of finasteride.
Summary Consensus
Data from animal models provide a clear mechanism by which finasteride could impact tear production and cause MGD and DED. However, the limited availability of clinical data makes it challenging to draw evidence-based conclusions about the potential adverse effects of finasteride. Clinical studies using a wide range of antiandrogen therapies in a different cohort (prostate cancer patients) also provide conflicting information. It’s important to remember that the absence of evidence doesn’t mean definitive proof of absence, and you should still consider the potential effects on your eyes when taking finasteride.
How Might Finasteride Cause Dry Eye?
- Finasteride reduces DHT: Finasteride blocks 5α-reductase, lowering dihydrotestosterone (DHT) levels in the body.
- Decreased androgen activity in eyelid glands: Lower DHT means less stimulation of the meibomian glands, which rely on androgens to produce the oils that keep tears in place.
- Reduced meibum quality and quantity: With reduced androgen influence, these glands produce less oil, thereby decreasing the stability of the tear film.
- Gland obstruction and dysfunction: Poor-quality meibum can clog gland openings, leading to meibomian gland dysfunction and reduced tear protection.
- Evaporative tear loss and dry eye symptoms: Tears evaporate more quickly, causing dryness, irritation, and blurred vision.
Secondary effects – Inflammation and surface damage: Ongoing tear film instability can trigger inflammation, discomfort, and damage to the eye’s surface over time.
Who Might Be Most At Risk of DED?
If you’re concerned about the risk of developing DED while taking finasteride, it’s important to consider other risk factors that could have an additive effect.[18]Britten-Jones, A. C., Wang, M. T., Samuels, I., Jennings, C., Stapleton, F., & Craig, J. P. (2024). Epidemiology and risk factors of dry eye disease: considerations for clinical management. … Continue reading Those at risk include:
Peri-and Post Menopausal Women – The hormonal changes of menopause lead to reduced secretion and quality in both lacrimal and meibomian glands, the core tear-producing tissues
Older People – The risk sharply increases after age 40, with prevalence rising further with each decade due to reduced tear production and gland function.
Contact Lens Wearers – Long-term use is linked to higher DED risk due to tear film disruption.
Screen Users – Reduced blink rate and incomplete blinks from device use elevate risk, especially in professional and urban populations.
Individuals on Certain Medications – Antihistamines, antidepressants, hormone therapies, and diuretics reduce tear secretion or alter tear composition.
People Exposed to Dry, Polluted, or Urban Environments – Low ambient humidity, air conditioning, pollution, and smoke contribute to increased evaporation and instability of the tear film.
How Can I Manage or Prevent Dry Eye While on Finasteride?
Dry eye can often be managed through a combination of lifestyle changes, environmental adjustments, proper eye care, and medical support.[19]Donthineni, P. R., Shanbhag, S. S., & Basu, S. (2021). An evidence-based strategic approach to prevention and treatment of dry eye disease, a modern global epidemic. Healthcare. 9(1). 89. … Continue reading If you’re taking finasteride and notice eye discomfort or dryness, these steps may help minimize symptoms and protect long-term eye health.
- Avoid smoky environments and consider quitting smoking. Both active and passive smoke can worsen dryness and irritation.
- Take regular eye breaks. Blinking more often and positioning screens slightly below eye level can also help reduce tear evaporation.
- Clean your eyelids daily using a warm compress followed by gentle cleansing with a mild, non-irritating lid cleanser. This can help keep oil glands functioning correctly, especially if you have blepharitis or meibomian gland dysfunction (MGD).
- Apply lubricating eye drops to keep the eyes moist and comfortable.
- Boost your intake of omega-3s through foods such as salmon, mackerel, walnuts, and flaxseed, or consider supplements under your doctor’s guidance. These healthy fats support meibomian gland function and tear quality.
- Protect your eyes from UV rays and dust by wearing sunglasses outdoors. Avoid rubbing your eyes, get sufficient sleep, and maintain a balanced diet rich in vitamins A, C, and E.
- If dryness, irritation, or visual changes continue, see an ophthalmologist or optometrist. They can assess tear quality and meibomian gland health, and may recommend advanced treatments such as prescription eye drops.
What’s Still Unknown
Although preclinical and clinical evidence suggest a possible link between finasteride and DED, many questions remain unanswered. Animal models suggest a causal connection but cannot replicate human physiology, and typically administer much higher doses than those that would be administered in humans. Large, controlled trials investigating the use of finasteride at the 1 mg hair-loss dose are required to draw evidence-based conclusions about the role of finasteride in DED.
Final Thoughts
Finasteride is an effective, well-established, and typically well-tolerated treatment for hair. However, concerns about links to meibomian gland dysfunction and DED are rising. The eye is a hormonally responsive organ, and systemic medications can have subtle but meaningful effects on ocular health. While animal studies and hormonal mechanisms support a potential link to dry eye disease, human data remain limited and inconclusive, especially at the low doses used for androgenic alopecia. For now, users should monitor for symptoms like dryness or irritation and seek ophthalmologic advice if they occur.
References[+]
References ↑1, ↑15 NZ Optics. (2022). Finasteride and dry eye disease. Available at: https://www.nzoptics.co.nz/live-articles/finasteride-and-dry-eye-disease/ (Accessed: October 2025) ↑2 Golden, M. I., Meyer, J. J., Zeppieri, M., & Patel, B. C. (2024). Dry Eye Syndrome. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK470411/ (Accessed: October 2025) ↑3 Zito, P. M., Bistas, K. G., Patel, P., & Syed, K. (2024). Finasteride. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK513329/ (Accessed: October 2025) ↑4 Azizi, A., Mumin, N. H., & Shafqat, N. (2021). Phytochemicals with anti 5-alpha-reductase activity: A prospective for prostate cancer treatment. F1000Research. 10. 221. Available at: https://doi.org/10.12688/f1000research.51066.3 ↑5 Sabeti, S., Kheirkhah, A., Yin, J., & Dana, R. (2020). Management of meibomian gland dysfunction: a review. Survey of Ophthalmology. 65(2). 205-217. Available at: https://doi.org/10.1016/j.survophthal.2019.08.007 ↑6 Dhamdhere, K., & Badawi, D. (2021). A blink-assisted, cornea-sparing wearable eyelid device for the effective penetration of therapeutic thermal energy into the meibomian glands for the treatment of dry eye disease. Journal of Clinical & Experimental Ophthalmology. 12 (S12):003. Available at: https://www.longdom.org/open-access/a-blinkassisted-corneasparing-wearable-eyelid-device-for-the-effectivepenetration-of-therapeutic-thermal-energy-into-the.pdf (Accessed: October 2025) ↑7, ↑13 Traish, A. M. (2020). Health risks associated with long-term finasteride and dutasteride use: it’s time to sound the alarm. The World Journal of Men’s Health. 38(3). 323-337. Available at: https://doi.org/10.5534/wjmh.200012 ↑8 Krenzer, K. L., Reza Dana, M., Ullman, M. D., Cermak, J. M., Tolls, D. B., Evans, J. E., & Sullivan, D. A. (2000). Effect of androgen deficiency on the human meibomian gland and ocular surface. The Journal of Clinical Endocrinology & Metabolism. 85(12). 4874-4882. Available at: https://doi.org/10.1210/jcem.85.12.7072 ↑9 Singh, S., Moksha, L., Sharma, N., Titiyal, J. S., Biswas, N. R., & Velpandian, T. (2014). Development and evaluation of animal models for sex steroid deficient dry eye. Journal of Pharmacological and Toxicological Methods. 70(1). 29-34. Available at: https://doi.org/10.1016/j.vascn.2014.03.004 ↑10 Li, K., Zhang, C., Yang, Z., Wang, Y., & Si, H. (2017). Evaluation of a novel dry eye model induced by oral administration of finasteride. Molecular Medicine Reports. 16(6). 8763-8770. Available at: https://doi.org/10.3892/mmr.2017.7754 ↑11 Zhang, C., Li, K., Yang, Z., Wang, Y., & Si, H. (2016). The effect of the aqueous extract of Bidens pilosa L. on androgen deficiency dry eye in rats. Cellular Physiology and Biochemistry. 39(1). 266-277. Available at: https://doi.org/10.1159/000445622 ↑12 Li, K., Zhang, C., Yang, Z., Wang, Y., & Si, H. (2017). Evaluation of a novel dry eye model induced by oral administration of finasteride. Molecular Medicine Reports. 16(6). 8763-8770. Available at: https://doi.org/10.3892/mmr.2017.7754 ↑14 Nguyen, B. J., Meer, E., Gupta, A. S., Jinpeng, G., Ying, G. S., Bunya, V. Y., Macchi, I., & Massaro-Giordano, M. (2022). The effect of finasteride on dry eye disease. Investigative Ophthalmology & Visual Science. 63(7). 427. Available at: https://doi.org/10.1167/iovs.63.7.427 ↑16 Chien, H. W., Lin, C. W., Lee, C. Y., Huang, J. Y., Yang, S. F., & Wang, K. (2022). The use of androgen deprivation therapy for prostate cancer and its effect on the subsequent dry eye disease: a population-based cohort study. International Journal of Medical Sciences. 19(7). 1103-1109. Available at: https://doi.org/10.7150/ijms.73417 ↑17 Kurna, S. A., Hacisalihoglu, A. O., Altun, A., Ozel, N. O., Uruc, F., Kanar, H. S., & Arsan, A. K. (2022). Effects of systemic anti-androgen drugs on the ocular surface. Journal Français d’Ophtalmologie. 45(6). 619-627. Available at: https://doi.org/10.1016/j.jfo.2021.06.007 ↑18 Britten-Jones, A. C., Wang, M. T., Samuels, I., Jennings, C., Stapleton, F., & Craig, J. P. (2024). Epidemiology and risk factors of dry eye disease: considerations for clinical management. Medicina. 60(9). 1458. Available at: https://doi.org/10.3390/medicina60091458 ↑19 Donthineni, P. R., Shanbhag, S. S., & Basu, S. (2021). An evidence-based strategic approach to prevention and treatment of dry eye disease, a modern global epidemic. Healthcare. 9(1). 89. Available at: https://doi.org/10.3390/healthcare9010089 Testosterone replacement therapy (TRT) can be a life-changing treatment for many men, restoring healthy hormone levels and renewing vitality after declines associated with low testosterone. However, many encounter a harsh irony: the same treatment that revitalizes them may also lead to accelerated hair loss, known as androgenetic alopecia (AGA). This frustrating dilemma can compel some men to choose between their appearance and their health.
Why do TRT patients often experience hair thinning just months into treatment? The culprit is testosterone’s conversion to DHT, the hormone that drives male pattern hair loss. Fortunately, new developments in comprehensive care address the testosterone-to-hair-loss pathway, allowing men to enjoy both hormone optimization and healthy hair. These clinically rigorous, customizable, and medically supervised treatments eliminate the either-or paradox.
Perfect Hair Health’s PhD experts have teamed up with Ulo, a leader in men’s wellness and hair care, to share five evidence-based techniques for preserving hair while on TRT. With Ulo’s unique expertise at the intersection of hormone optimization and hair restoration, patients gain access to solutions that are both proven and personalized.
Read on to learn how Ulo can help you maintain both hormonal health and hair density.
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

The Science of Testosterone and Hair Loss
Before exploring solutions, we will briefly explain the science behind TRT and hair loss, including relevant medical research that supports prevention-based strategies for preserving hair during TRT.
Testosterone Metabolism and DHT Formation
To understand the link between TRT and hair loss, we turn to the molecular process that converts testosterone into dihydrotestosterone (DHT), the hormone that causes follicular degradation and balding. Two types of 5α-reductase enzymes are responsible for deriving DHT from testosterone: Type I (primarily located in the skin and sebaceous glands) and Type II (mainly located in the hair follicles of the crown and temporal regions).[1]Owecka, B., Tomaszewska, A., Dobrzeniecki, K., Owecki, M. (2024). The Hormonal Background of Hair Loss in Non-Scarring Alopecias. Biomedicines. 12(3). 513. Available at: … Continue reading
Androgenetic alopecia (AGA) can occur naturally in susceptible individuals, whose hair follicles have high concentrations of androgen receptors and 5α-reductase enzymes. However, adding exogenous T through TRT can expedite this process. TRT’s elevation of testosterone levels can raise DHT to two or three times higher than starting levels, depending on the dosage and mode of application. The conversion of T to DHT is especially prevalent in the crown and temporal areas of the scalp, where it causes hair thinning and loss.[2]Owecka, B., Tomaszewska, A., Dobrzeniecki, K., Owecki, M. (2024). The Hormonal Background of Hair Loss in Non-Scarring Alopecias. Biomedicines. 12(3). 513. Available at: … Continue reading,[3]Stocks, B., Asempa, O. (2025). Does testosterone replacement therapy cause hair loss? Baylor College of Medicine. Available at: … Continue reading
Genetic predisposition plays a significant role, as variations in androgen receptor genes determine individual sensitivity to DHT. This explains why patients on identical TRT regimens can experience varying levels of hair loss, with some experiencing dramatic loss and others experiencing little or no effect. In those with hereditary androgen sensitivity, the hair follicles respond poorly to even slight elevations in local DHT levels on the scalp.[4]Owecka, B., Tomaszewska, A., Dobrzeniecki, K., Owecki, M. (2024). The Hormonal Background of Hair Loss in Non-Scarring Alopecias. Biomedicines. 12(3). 513. Available at: … Continue reading
Why TRT Accelerates Pattern Hair Loss
As previously noted, TRT increases testosterone and leads to higher levels of its derivative hormone, DHT, which binds to hair follicles and causes hair thinning and loss in genetically susceptible men. Spikes in DHT and associated hair loss can occur even with moderate TRT, as raising testosterone into the normal range can represent a significant increase from severe deficiency.[5]Borst, S.E., Shuster, J.J., Zou, B., Ye, F., Jia, H., Wokhlu, A., Yarrow, J.F. (2014). Cardiovascular risks and elevation of serum DHT vary by route of testosterone administration: a systemic review … Continue reading
Episodic TRT protocols like weekly injections can create testosterone peaks over 1,200 ng/dL for up to 48 hours after injection, leading to surges in scalp DHT production during these windows. Such DHT spikes can damage the follicles and contribute to rapid hair loss.[6]Stocks, B., Asempa, O. (2025). Does testosterone replacement therapy cause hair loss? Baylor College of Medicine. Available at: … Continue reading,[7]Shoskes, J.J., Wilson, M.K., Spinner, M.L. (2016). Pharmacology of testosterone replacement therapy preparations. Translational Andrology and Urology. 5(6). 834-843. Available at: … Continue reading
Disruption of hair follicle cycles can begin in the first 3-6 weeks of TRT. However, noticeable hair loss typically doesn’t emerge for several months, as early damage is concealed by hair growth cycles. Studies suggest higher TRT doses correlate with more rapid hair loss in susceptible patients.[8]Saad, F., Aversa, A., Isidori, A.M., Zafalon, L., Zitzmann, M., Gooren, L. (2011). Onset of effects of testosterone treatment and time span until maximum effects are achieved. European Journal of … Continue reading,[9]Fu, D., Huang, J., Li, K., Chen, Y., He, Y., Sun, Y., Guo, Y., Du, L., Qu, Q., Miao, Y., Hu, Z. (2021). Dihydrotestosterone-induced hair regrowth inhibition by activating androgen receptor in C57BL6 … Continue reading
Balancing Hormonal Benefits with Hair Protection
Although DHT can harm the hair follicles, it plays a vital role in various biological processes, including sexual function, bone mineralization, and muscle development. Therefore, the aim of hair loss treatment during TRT is not to eliminate the hormone. Rather, the therapeutic goal is to lower DHT to a level that minimizes follicular damage while preserving systemic functions. Striking this balance in individuals often requires sophisticated strategies that exceed standardized DHT suppression protocols.[10]Owecka, B., Tomaszewska, A., Dobrzeniecki, K., Owecki, M. (2024). The Hormonal Background of Hair Loss in Non-Scarring Alopecias. Biomedicines. 12(3). 513. Available at: … Continue reading
While some patients may be tempted to terminate TRT to protect their hair, research findings support a more measured approach that takes TRT’s broader benefits into account. Patients who elect to stop taking testosterone for hair preservation may experience a return of detrimental symptoms: sexual dysfunction, depression, fatigue, muscle loss, and more. The psychological and physiological impacts of low T can outweigh cosmetic concerns.
Given these considerations, practitioners advise strategic early intervention over reactionary care, as hair restoration becomes much more challenging after considerable follicular shrinkage. Early discussion with healthcare providers about hair preservation strategies helps maintain hair health throughout the hormone optimization process.[11]Owecka, B., Tomaszewska, A., Dobrzeniecki, K., Owecki, M. (2024). The Hormonal Background of Hair Loss in Non-Scarring Alopecias. Biomedicines. 12(3). 513. Available at: … Continue reading
With the science behind TRT and hair loss clear, let’s look at the latest, evidence-based ways patients can protect their hair.
Expert Solutions for TRT-Related Hair Loss
Hair loss associated with TRT often has distinct features, like episodic spikes in testosterone and DHT conversion rates. Although conventional hair loss interventions can be useful, protocols may benefit from personalization according to TRT regimens.
Ulo is a unique provider due to its innovative hair loss treatments and dual expertise in both hair restoration and hormone optimization. This combined clinical experience enables them to tailor treatments to individual patient needs, such as timing factors and treatment responses specific to TRT. All Ulo treatment protocols are physician-crafted and include close follow-up care to monitor for side effects and make adjustments as needed.
Treatment Options Overview
This overview outlines Ulo’s comprehensive strategy for hair loss intervention, from advanced prescription formulas to targeted nutritional support. All offerings are transparently priced with customizable features to account for TRT-related factors and individual patient considerations.
Product Category Mechanism of Action Delivery Method Formula Details Price Oral Medications Systemic 5α-reductase inhibition and vasodilation Oral capsule, tablet, or softgel Finasteride, dutasteride, and minoxidil for systemic DHT blocking and growth stimulation $24-65/month Topical Finasteride Localized Type II 5α-reductase inhibition with minimal systemic exposure Solution Concentration range (0.005%-0.2%); customizable adjuvants; irritant-free $64+/month Topical Dutasteride Localized dual enzyme (Types I & II) inhibition for maximum scalp DHT suppression Solution Concentration range (0.02%-0.2%); customizable adjuvants; irritant-free $84+/month Growth Booster Topicals Vasodilation, anti-inflammatory action without DHT manipulation Solution Customizable combinations of minoxidil, tretinoin, caffeine, melatonin, and cetirizine $54/month OTC Support Products Nutritional optimization, scalp environment enhancement, and daily follicular support Serum, supplements, shampoo, and conditioner Evidence-based supplements targeting TRT-specific deficiencies; irritant-free hair care products $29+/month Method #1: Oral Medications for Systemic Effects
Why Choose Oral Treatment?
This systemic approach provides several key advantages for men on testosterone therapy:
Benefit: Details: ✓ Comprehensive treatment Systemic DHT blocking plus vasodilation for growth stimulation ✓ Maximum potency Up to 93% DHT reduction with dutasteride ✓ Convenience Once-daily dosing ✓ Customizable Combination options with growth stimulators How Do Oral Medications Work to Treat Hair Loss?
DHT blockers like finasteride and dutasteride treat hair loss by suppressing the 5α-reductase enzymes that metabolize testosterone into DHT. Oral preparations have systemic effects and therefore offer extensive treatment. They can be used in conjunction with oral growth stimulators, such as minoxidil, to minimize hair loss associated with TRT.
Finasteride inhibits the Type II 5α-reductase enzyme to reduce DHT levels by up to 70%. In contrast, dutasteride is a more potent intervention as a dual inhibitor of both Type I and Type II 5α-reductase enzymes, achieving up to 93% lower DHT levels. This thorough DHT suppression can make dutasteride useful for advanced hair loss cases as well as high-dose TRT patients who may not respond to topical treatments.[12]Arif, T., Dorjay, K., Adil, M., Sami, M. (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). 31-35. Available at: … Continue reading
Studies indicate that dutasteride’s therapeutic effects may continue to build beyond the 12 months at which many finasteride users experience a plateau, further establishing dutasteride as the most potent treatment option. While oral DHT blockers offer powerful treatment, their systemic effects contribute to a greater risk of serious side effects, so strict medical oversight is required.[13]Harcha, W.G., Martinez, J.B., Tsai, T-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy … Continue reading
Ulo’s Oral Medication Approach
Ulo’s oral treatments offer effective systemic intervention through various formulations designed for optimal bioavailability, featuring customizable options. Their oral Dutasteride Rx delivers a 0.5 mg dose of dual enzyme-blocking treatment in a softgel format designed to enhance absorption, overcoming the poor bioavailability of powder formulas touted by competitors.
Their gentler oral Finasteride Rx also offers systemic DHT suppression in a once-daily capsule, while an oral Minoxidil Rx (1.25 mg or 2.5 mg) tablet formula increases systemic circulation and upregulates hair growth factors to treat hair loss as an alternative to DHT-blocking medications.
Ulo also offers oral preparations that synergistically combine DHT blockers with minoxidil for effects that are shown to exceed those of monotherapies: Dutasteride + Minoxidil Rx and Finasteride + Minoxidil Rx, which come with optional adjuvants like vitamin D and folic acid to address deficiencies. Rather than combining oral dutasteride and minoxidil into a single capsule, as competitors do, Ulo maintains a separate soft gel and powder format for each, enabling proper absorption.
Key Takeaway: Oral medications such as finasteride, dutasteride, and minoxidil provide the strongest systemic protection against hair loss during TRT, but their broad effects mean they require careful medical supervision to manage potential side effects.
Method #2: Topical Finasteride for Precision DHT Blocking
Why Choose Topical Finasteride?
Topical finasteride offers these strategic benefits for TRT patients:
Benefit: Details: ✓ Targeted action Scalp-focused DHT reduction ✓ Flexible dosing Low-dose (0.005%) and full-strength (0.2%) options ✓ Safer alternative to oral Comparable efficacy to oral without systemic exposure side effects ✓ Customizable Optional minoxidil and adjuvant combinations How Does Topical Finasteride Work?
Topical finasteride delivers targeted DHT suppression directly to the scalp, minimizing effects on the rest of the body. Studies show that topical finasteride can reduce scalp DHT to a therapeutic effect with little impact on serum DHT levels. In contrast, oral finasteride reduces DHT levels throughout the entire system. For men on TRT, maintaining systemic DHT is essential for bone, muscle, and sexual health; therefore, topical finasteride is typically the preferred option.[14]Kinter KJ, Amraei R, Anekar AA. Biochemistry, Dihydrotestosterone. [Updated 2023 Jul 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: … Continue reading,[15]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray … Continue reading
Topical formulations also allow more precise dosing with flexible concentrations that can be tailored to patient needs and responses. Low-dose formulas (0.005%) may be more appropriate for early-stage or moderate TRT patients, while higher-strength options (0.2%) can offer more aggressive treatment for substantial hair loss or intensive TRT protocols. Topical solutions can easily incorporate elective adjuvants, such as absorption enhancers and anti-inflammatories, for synergistic benefits.[16]Kinter KJ, Amraei R, Anekar AA. Biochemistry, Dihydrotestosterone. [Updated 2023 Jul 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: … Continue reading
Ulo’s Topical Finasteride System
Ulo’s topical finasteride solution provides targeted scalp treatment with an optimized delivery base that is free from potential irritants, such as propylene glycol. Their fully customizable Finasteride + Minoxidil Plus Rx is available in finasteride doses of 0.005% for gentler therapy and 0.2% for stronger intervention.
The robust formulation includes a wide range of synergistic add-on choices that address multiple hair loss pathways: up to 7% minoxidil for growth stimulation, tretinoin for better absorption, and multiple antioxidants for comprehensive support. This adaptability enables treatment tailored to patient needs rather than fixed protocols. Physician-crafted and supervised topical protocols are considered safe and effective for prolonged use to complement TRT journeys.
Key Takeaway: Topical finasteride allows TRT patients to lower scalp DHT while preserving systemic androgen function, making it a preferred option for balancing hair health with hormonal benefits.
Method #3: Topical Dutasteride for Comprehensive Scalp Protection
Why Choose Topical Dutasteride?
This powerful topical approach delivers distinct advantages:
Benefit: Details: ✓ Dual enzyme blocking Inhibits both Type I and Type II 5α-reductase ✓ Max scalp DHT reduction Superior enzyme inhibition for aggressive protocol ✓ Safe and targeted treatment Potent effects without systemic exposure ✓ Concentration control Low-dose (0.02%) to full-strength (0.2%) options ✓ Personalized add-ons Adjuvants can be tailored to individual needs How Does Topical Dutasteride Work?
Topical dutasteride leverages the dual enzyme inhibition and localized efficacy discussed in Methods 1 and 2 to powerfully suppress scalp DHT levels while maintaining systemic DHT. This approach can be beneficial for patients on intensive TRT protocols that don’t respond to single-enzyme-blocking treatments.
The superior targeted DHT suppression of topical dutasteride makes it an excellent choice for severe hair loss and TRT scenarios. It is the most potent topical DHT blocker available. Like topical finasteride, its concentration flexibility and easy incorporation of adjuvants enable more highly tailored and adaptable treatment compared to oral administration.[17]Arif, T., Dorjay, K., Adil, M., Sami, M. (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). 31-35. Available at: … Continue reading,[18]Harcha, W.G., Martinez, J.B., Tsai, T-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy … Continue reading,[19]Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through microneedling or oral 0.5 mg) in … Continue reading
Ulo’s Topical Dutasteride Excellence
Ulo’s topical dutasteride offerings include both low-dose (0.02%) and full-strength (0.2%) versions of their Dutasteride + Minoxidil Plus Rx solution, delivering potent dual enzyme inhibition for precision scalp treatment. While the lower concentration may suit maintenance or preventative protocols, the highly concentrated formula may benefit severe hair loss patients and aggressive TRT regimens.
Like Ulo’s topical finasteride products, their dutasteride selections are fully customizable with synergistic add-ons that include minoxidil (up to 7%), caffeine, tretinoin, cetirizine, and melatonin. The clinically backed formulas are designed to deeply penetrate the scalp and follicles without the use of irritants, corticosteroids, or unproven ingredients seen in competing premium brands.
The platform’s Build Your Own Topical feature offers complete customization across the full range of topical electives, including a choice of finasteride or dutasteride with individualized adjuvant combinations.
Key Takeaway: Topical dutasteride delivers the most powerful scalp-targeted DHT reduction available and is well-suited for men with significant hair loss or intensive TRT regimens that do not respond to other approaches.
Method #4: Growth Booster Topicals for Non-Hormonal Treatment
Why Choose Growth Booster Topicals?
Non-hormonal growth stimulation provides these unique benefits:
Benefit: Details: ✓ Hormone-free treatment Growth stimulation without hormonal manipulation ✓ Anti-inflammatory Addresses scalp inflammation contributing to hair loss ✓ High-strength formulas 7% minoxidil with absorption enhancers ✓ Versatile use Standalone or in combination with DHT blockers How Do Growth Booster Topicals Work?
Growth stimulators are useful for comprehensive hair restoration during TRT to address non-hormonal hair loss pathways outside of DHT’s purview. Minoxidil is one of the most commonly used medications, acting as a vasodilator to boost scalp circulation while upregulating growth factors to extend the anagen phase of each hair cycle.[20]Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through microneedling or oral 0.5 mg) in … Continue reading,[21]Koralewicz, M.M., Szatkowska, O.A. (2024). Topical solutions for androgenetic alopecia: evaluating efficacy and safety. Forum Dermatologicum. 10(3). 71-78. Available at: … Continue reading
Calming agents like cetirizine, melatonin, and caffeine address scalp inflammation, another key contributor to follicular damage and hair loss. Caffeine also enhances follicular nutrient supply to act as a growth stimulant, while melatonin’s antioxidant protection reduces shedding.[22]Koralewicz, M.M., Szatkowska, O.A. (2024). Topical solutions for androgenetic alopecia: evaluating efficacy and safety. Forum Dermatologicum. 10(3). 71-78. Available at: … Continue reading
The powerful combination of growth stimulation with anti-inflammatory action can provide standalone benefits or be applied as a complementary therapy with DHT blockers. Studies show that minoxidil is effectively combined with DHT blockers, offering greater outcomes than single-agent therapies. Growth boosters are therefore vital to the success of TRT hair loss prevention protocols.[23]Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through microneedling or oral 0.5 mg) in … Continue reading,[24]Koralewicz, M.M., Szatkowska, O.A. (2024). Topical solutions for androgenetic alopecia: evaluating efficacy and safety. Forum Dermatologicum. 10(3). 71-78. Available at: … Continue reading
Ulo’s Growth Booster Solutions
Ulo offers two customizable growth stimulation topicals that can be used alone or in conjunction with DHT blockers, depending on the prescribed protocol. Their Minoxidil + Tretinoin Plus Rx solution features a high 7% minoxidil in combination with tretinoin for optimal absorption. Adjuvants like cetirizine, melatonin, and caffeine may be added for anti-inflammatory support, antioxidant protection, and enhanced microcirculation.
For patients interested in calming the scalp without actives like minoxidil, the Cetirizine Plus Rx solution delivers a proven dose of 1% cetirizine. This potent anti-inflammatory also has anti-histamine properties to support scalp health and hair growth. Caffeine and melatonin may be added for well-rounded, non-hormonal treatment.
Like all Ulo topicals, these selections offer superior scalp penetration without irritating carriers or excipients, making them ideal for extended daily use in patients with sensitive skin or long-term combination therapy needs.
Key Takeaway: Growth booster topicals like minoxidil, caffeine, and melatonin support follicle strength through circulation, inflammation control, and antioxidant activity, and they can be used alone or combined with DHT blockers for greater results.
Method #5: OTC Support Products for Foundational Care
Why Choose Premium OTC Products?
Quality over-the-counter support offers these foundational advantages:
Benefit: Details: ✓ Nutritional support Addresses deficiencies contributing to hair loss ✓ Scalp optimization Creates an ideal environment for treatment absorption ✓ Natural ingredients Active botanicals and peptides ✓ Complete system Serum, supplements, shampoo, and conditioner How Do OTC Products Work?
High-quality, non-prescription products can also help target underlying hair health factors without hormonal suppression. These include nutritional support formulas that help address common deficiencies linked to hair loss. Such formulas may contain active botanical agents with anti-inflammatory or growth-promoting effects, providing gentle systemic support.[25]Koralewicz, M.M., Szatkowska, O.A. (2024). Topical solutions for androgenetic alopecia: evaluating efficacy and safety. Forum Dermatologicum. 10(3). 71-78. Available at: … Continue reading
Specialty hair care products, such as shampoos, conditioners, and serums, nourish and balance the scalp, providing an optimal environment for prescription medications to take hold and support overall treatment objectives. Non-prescription hair growth serums may contain botanical extracts and peptide-based therapeutics that soothe and strengthen the follicles as a complement to medical protocols.
Ulo’s OTC Support Range
Ulo’s four non-prescription haircare products support foundational hair health in synergy with the brand’s extensive prescription range. Their Hair Growth Serum contains three proprietary peptide blends that demonstrated comparable efficacy to minoxidil in a medically supervised study. Natural active ingredients such as saw palmetto, rosemary oil, and adenosine provide gentle DHT suppression, enhanced circulation, and growth stimulation, respectively.
The nutritional Hair Growth Supplement provides extensive hair and scalp nourishment with proven ingredients like vitamin D, biotin, hydrolyzed fish collagen, and saw palmetto. Antioxidants such as chlorophyll and astaxanthin combat oxidative stress from within.
Lastly, the Thickening Shampoo and Conditioner provide gentle daily scalp optimization with natural actives like caffeine to boost circulation, keratin to strengthen strands, and saw palmetto to suppress DHT levels. The PhD scientist-crafted formulas are free of known irritants and artificial fragrances, made instead with nourishing oils and amino acids that encourage natural hair growth and work to improve prescription treatment response.
Key Takeaway: Over-the-counter serums, supplements, and hair care products build the foundation for long-term hair preservation by supporting scalp health, addressing nutritional needs, and enhancing the effects of prescription treatments.
Why Ulo Stands Out for TRT Patients
Managing hair health during TRT requires clinical expertise to address unique hormonal challenges that generic treatments often miss. Ulo’s multidisciplinary care, personalized protocols, advanced formulations, and high-quality standards make it a leader in TRT-focused hair restoration telehealth. Below is a summary of what sets Ulo apart.
✓ Comprehensive Medical Oversight
Ulo partners exclusively with licensed and board-certified physicians to provide safe and effective care that meets the highest regulatory and safety standards. Full medical consultations and evaluations at onboarding enable Ulo’s qualified doctors to craft tailored treatment plans, while ongoing monitoring protocols establish positive outcomes.
✓ Dual Expertise in TRT and Hair Loss
Ulo operates as a leading telehealth provider with state-of-the-art services in both hair restoration and TRT, employing physicians with experience in both areas. The specialists at Ulo are uniquely qualified to address TRT-related hair loss with practical insights and actionable solutions.
✓ True Customization for TRT Needs
While most competitors tout fixed, one-size-fits-all formulations, Ulo, in contrast, carries truly customizable treatment options with adjustable doses and elective add-ons to meet individual patient needs. This is an important feature for TRT patients, who may require medications that vary from standard formulas and can be adjusted to changing responses over time.
✓ Pharmaceutical-Grade Quality Standards
Ulo ensures product quality by sourcing all medications from certified domestic pharmacies. Formulations are designed for peak bioavailability with innovative delivery systems, free of harsh additives like propylene glycol, which can be found in many hair loss products.
✓ Transparent Pricing and Customer Support
Ulo’s fixed-rate monthly subscription pricing avoids the rising costs and hidden fees that are common among telemedicine competitors. This allows patients to budget for long-term integrated care. Subscriptions cover all essential services, including physician consultations, follow-ups, prescription deliveries, and more. The platform’s responsive customer service ensures uninterrupted treatment and full support throughout the process.
Ulo vs. Standard Hair Loss Providers
The chart below illustrates the main differences between Ulo’s expert model and standard telehealth hair loss providers, showcasing how Ulo is uniquely equipped to meet the hair restoration needs of TRT patients.
Feature Ulo Standard Providers Medical Oversight Board-certified physicians with TRT and hair loss expertise Generic telehealth consultations Customization True concentration flexibility (0.005%-0.2%); adjuvant options Fixed formulations; limited options Formulation Quality Pharmaceutical-grade; soft gel dutasteride for optimal bioavailability Standard compounding; limited bioavailability Ingredient Safety Irritant-free; no propylene glycol or corticosteroids Often contains propylene glycol and harsh excipients TRT Knowledge Dual expertise in hormone optimization and hair restoration Hair loss focus only; limited TRT understanding Pricing Model Transparent flat-rate monthly subscriptions Hidden fees and escalating costs are common Quality Assurance Certificates of analysis; FDA-approved pharmacy sourcing Limited transparency; variable quality standards Combination Options Extensive adjuvant library (tretinoin, caffeine, melatonin, cetirizine) Basic combinations; limited synergistic ingredients Follow-up Care Unlimited physician access; regular monitoring protocols Limited follow-up; basic customer service Treatment Coordination Can coordinate with existing TRT protocols No hormone therapy coordination Safety Considerations
Hair restoration therapies, especially hormonal medications like DHT blockers, can interact with TRT and require strict medical oversight to monitor for adverse reactions. Among the different treatment methods discussed, safety profiles vary. Here is a brief overview of their safety findings. All medical concerns should ultimately be discussed with a qualified clinician.
Prescription treatments display differing safety profiles based on the delivery method, whether oral or topical. Oral DHT blockers have systemic exposure and therefore carry the risk of systemic side effects like sexual dysfunction, mood disorders, and gynecomastia; all of which require prompt medical evaluation. Oral minoxidil requires cardiovascular monitoring due to its circulatory effects.
Topical DHT blockers have limited systemic exposure and therefore milder associated side effects, often no more than minor localized scalp irritation that tends to resolve within 2-4 weeks. Regular follow-up exams are required every 3-6 months during the first year of prescribed hair loss treatment.
Over-the-counter products, such as shampoos and nutritional supplements, carry minimal risk; however, patients with allergies or sensitivities should carefully read the ingredients before use. OTC treatments can sometimes interact with medications or exacerbate underlying conditions, so it’s best to consult a healthcare professional for guidance.
Contraindications for prescription DHT blockers include severe cardiovascular conditions, active scalp infections, and liver disease. Women who are pregnant or plan to have a future pregnancy should avoid DHT blockers due to possible birth defects.
Consult your doctor for a complete list of all possible side effects, contraindications, and risk mitigation strategies.
Conclusion: Battling the Hair vs. Health Trade-Off
Accelerated hair loss can be one of the most frustrating and disruptive elements of TRT, calling for sophisticated approaches that go beyond conventional hair loss treatment. TRT creates unique challenges that demand advanced intervention strategies.
Thanks to the wide-ranging methods discussed in this guide, men no longer have to choose between hair preservation and hormone optimization on TRT. With dual expertise, qualified medical oversight, and tailored formulations that meet leading quality standards, Ulo rises to meet the varied needs of TRT patients where generic providers fall short.
Don’t allow hair loss to interfere with your journey to hormonal health and restored vitality. Ulo’s scientific approach and clinical experience make them the clear choice for men seeking to maintain their hair while optimizing their hormone therapy. Consult with a licensed Ulo physician today to craft a personalized treatment plan.
Discover your unique hair preservation strategy with Ulo.
Frequently Asked Questions
Does Ulo offer testosterone replacement therapy?
Yes, Ulo has testosterone replacement therapy offerings that include injections, topicals, and oral preparations. Like Ulo’s hair restoration services, their TRT program is medically rigorous and tailored to individual needs. Ulo’s multidisciplinary expertise gives them unique insight, enabling them to properly address both sides of the hormone equation when treating TRT-related hair loss.
Should I discuss hair loss concerns and treatment options with my doctor when starting TRT?
Yes, all hair concerns should be discussed with your clinician before starting TRT to enable proactive treatment planning and fully informed care decisions. Your doctor will assess your health profile and treatment goals to decide whether hair loss intervention is an appropriate choice and, if so, which method is best for you.
Which hair loss treatments require a prescription?
Prescriptions are required for DHT blockers like dutasteride and finasteride, growth stimulators like minoxidil, and some customized topicals. All require proper diagnostic and follow-up care protocols that meet clinical safety standards. Options that don’t require a prescription include over-the-counter nutritional supplements, serums, shampoos, and conditioners with unrestricted active ingredients like botanical extracts and peptides.
Can I combine hair loss treatment methods?
Yes, some hair loss treatments can be combined for synergistic effects. Popular combinations include minoxidil with DHT blockers (dutasteride or finasteride), alongside nutritional support. Treatments should only be combined under close medical supervision to avoid harmful interactions.
When do most hair loss treatments show results?
Although results differ depending on the patient and treatment type, most users experience initial effects within the first 3-6 months and more substantial benefits over 6-18 months. Consistent daily administration is required to successfully achieve and maintain hair loss treatment benefits.
What are common hair loss treatment side effects?
Potential side effects vary based on the type of treatment and are usually controllable with the right medical care. Topical treatments can produce mild and temporary irritation at the application site. Although rare, oral DHT-blocking medications can lead to systemic side effects like sexual dysfunction and mood disturbances requiring immediate medical attention. Thoroughly discuss all potential side effects and suitable monitoring protocols with your provider.
How much do Ulo’s hair loss treatments typically cost?
Ulo’s price points vary by treatment type and level of customization. Pricing reflects flat-rate monthly subscription fees that not only cover the cost of the medication but also include essential telehealth services like doctor consultations, follow-ups, and prescription deliveries. Their prescription oral treatments range from $24 to $65 per month, while topicals range from about $49 to $94 per month. OTC support products start at $29 per month.
What should I expect during my first consultation with Ulo?
The Ulo onboarding process starts with a thorough medical intake form that covers your health profile, treatment goals, and current medications. You will be partnered with one of Ulo’s licensed physicians, who, after reviewing your information, will collaborate with you to design a treatment plan tailored to your specific needs. During the initial consultation, your doctor will thoroughly discuss treatment options, realistic expectations, safety, and follow-up protocols to guarantee safe and effective care.
References[+]
References ↑1, ↑10, ↑11 Owecka, B., Tomaszewska, A., Dobrzeniecki, K., Owecki, M. (2024). The Hormonal Background of Hair Loss in Non-Scarring Alopecias. Biomedicines. 12(3). 513. Available at: https://doi.org/10.3390/biomedicines12030513 ↑2, ↑4 Owecka, B., Tomaszewska, A., Dobrzeniecki, K., Owecki, M. (2024). The Hormonal Background of Hair Loss in Non-Scarring Alopecias. Biomedicines. 12(3). 513. Available at: https://doi.org/10.3390/biomedicines12030513 ↑3, ↑6 Stocks, B., Asempa, O. (2025). Does testosterone replacement therapy cause hair loss? Baylor College of Medicine. Available at: https://blogs.bcm.edu/2025/08/11/does-testosterone-replacement-therapy-cause-hair-loss/ (Accessed: November 2025) ↑5 Borst, S.E., Shuster, J.J., Zou, B., Ye, F., Jia, H., Wokhlu, A., Yarrow, J.F. (2014). Cardiovascular risks and elevation of serum DHT vary by route of testosterone administration: a systemic review and meta-analysis. BMC Medicine. 12. 211. Available at: https://doi.org/10.1186/s12916-014-0211-5 ↑7 Shoskes, J.J., Wilson, M.K., Spinner, M.L. (2016). Pharmacology of testosterone replacement therapy preparations. Translational Andrology and Urology. 5(6). 834-843. Available at: https://doi.org/10.21037/tau.2016.07.10 ↑8 Saad, F., Aversa, A., Isidori, A.M., Zafalon, L., Zitzmann, M., Gooren, L. (2011). Onset of effects of testosterone treatment and time span until maximum effects are achieved. European Journal of Endocrinology. 165(5). 675-685. Available at: https://doi.org/10.1530/EJE-11-0221 ↑9 Fu, D., Huang, J., Li, K., Chen, Y., He, Y., Sun, Y., Guo, Y., Du, L., Qu, Q., Miao, Y., Hu, Z. (2021). Dihydrotestosterone-induced hair regrowth inhibition by activating androgen receptor in C57BL6 mice simulates androgenetic alopecia. Biomedicine & Pharmacology. 137. 111247. Available at: https://doi.org/10.1016/j.biopha.2021.111247 ↑12 Arif, T., Dorjay, K., Adil, M., Sami, M. (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). 31-35. Available at: https://doi.org/10.2174/1574884712666170310111125 ↑13 Harcha, W.G., Martinez, J.B., Tsai, T-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. Journal of the American Academy of Dermatology. 70(3). 489-498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑14, ↑16 Kinter KJ, Amraei R, Anekar AA. Biochemistry, Dihydrotestosterone. [Updated 2023 Jul 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557634/ (Accessed: November 2025) ↑15 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology & Venereology. 36(2). Available at: https://doi.org/10.1111/jdv.17738 ↑17 Arif, T., Dorjay, K., Adil, M., Sami, M. (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). 31-35. Available at: https://doi.org/10.2174/1574884712666170310111125 ↑18 Harcha, W.G., Martinez, J.B., Tsai, T-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. Journal of the American Academy of Dermatology. 70(3). 489-498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑19 Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through microneedling or oral 0.5 mg) in treatment of androgenetic alopecia. QJM: An International Journal of Medicine, 117(Supplement 1), hcae175.207. Available at: https://doi.org/10.1093/qjmed/hcae175.207 ↑20, ↑23 Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through microneedling or oral 0.5 mg) in treatment of androgenetic alopecia. QJM: An International Journal of Medicine, 117(Supplement 1), hcae175.207. Available at: https://doi.org/10.1093/qjmed/hcae175.207 ↑21, ↑22, ↑24, ↑25 Koralewicz, M.M., Szatkowska, O.A. (2024). Topical solutions for androgenetic alopecia: evaluating efficacy and safety. Forum Dermatologicum. 10(3). 71-78. Available at: https://doi.org/10.5603/fd.101208 Minoxidil is a widely used medication for treating androgenic alopecia (AGA) in both men and women. Originally developed as a treatment for high blood pressure, minoxidil was found to improve hair growth outcomes as a side effect, making its topical form one of only two (the other being finasteride) FDA-approved treatments for AGA.
Minoxidil is primarily available in two forms: topical and oral. The topical form, which includes both solutions and foams, is more commonly used and can be purchased over the counter. Oral minoxidil, however, is prescription only and is typically given at doses of 2.5 mg or 5 mg for hair loss treatment.
While minoxidil is generally considered safe, it’s important to understand its potential side effects and who might be more at risk from them. In this article, we will examine the side effects of topical and oral minoxidil and discuss how you can adjust your treatment regimen to mitigate these.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Does Minoxidil Work?
Minoxidil was originally developed as an antihypertensive medication. However, when treated participants started experiencing hypertrichosis (excessive hair growth), studies were conducted to find out if it could improve hair regrowth outcomes in humans (spoiler alert – it could!).[1]Bryan, J. (2011). How minoxidil was transformed from an antihypertensive to hair-loss drug. The Pharmaceutical Journal. Available at: … Continue reading
Minoxidil’s mechanism of action is not fully understood, but it is thought to work through two main mechanisms:
- Vasodilatory effects: It activates vascular endothelial growth factor (VEGF), increasing blood flow, oxygen, and growth factor delivery to the hair follicle.[2]Alhayaza, G., Hakami, A., AlMarzouk, L.H., Al Qurashi, A.A., Alghamdi, G., Alharithy, R. (2023). Topical minoxidil reported hair discoloration: a cross-sectional study. Dermatology Reports. 16(1). … Continue reading
- Potassium channel activation: This prolongs the growth (anagen) phase and shortens the resting (telogen) phase.
Research also shows that minoxidil can act on androgenic receptors, suppressing the expression of the androgen receptor and CYP17A1 and boosting the activity of CYP19A1. This decreases the formation and binding of dihydrotestosterone and enhances the production of estradiol, which may also benefit those with AGA.[3]Shen, Y., Zhu, Y., Zhang, L., Sun, J., Xie, B., Zhang, H., Song, X. (2023). New Target for Minoxidil in the Treatment of Androgenetic Alopecia. Drug Design, Development and Therapy. 17. 2537-2547. … Continue reading
How is Minoxidil Activated in the Body?
Minoxidil needs to be converted into its active form, minoxidil sulfate, by sulfotransferase enzymes before it can effectively stimulate hair growth.[4]Dhurat, R., Daruwalla, S., Pai, S., Kovacevic, M., McCoy, J., Shapiro, J., Sinclair, R., Vano-Galvan, S., Goren, A. (2021). SULT1A1 (Minoxidil Sulfotransferase) enzyme booster significantly improves … Continue reading This conversion primarily occurs in the scalp for topical minoxidil and in the liver for oral minoxidil.
Figure 1: The conversion of minoxidil to minoxidil sulfate by sulfotransferase.[5]Anderson, R.J., Kudlacek, P.E., Clemens, D.L. (1998). Sulfation of minoxidil by multiple human cytosolic sulfotransferases. Chemico-Biological Interactions. 109. 53-67. Available at: … Continue reading
However, enzyme activity varies significantly among individuals, meaning that some people naturally produce higher levels of sulfotransferase, allowing for better activation and increased effectiveness of the drug, while others have lower enzyme activity, which can limit its impact. You can read our article on how your genetics can influence sulfotransferase activity here.
Topical Minoxidil Vs. Oral Minoxidil: Activation Pathways
Topical minoxidil and oral minoxidil differ in application and effectiveness. Topical minoxidil is applied directly to the scalp, while oral minoxidil is taken as a pill.
The efficacy of these two treatment routes differs because of their distinct metabolic pathways.
Topical Minoxidil
Topical minoxidil is applied directly to the scalp, presenting a challenge for drug activation. The drug requires conversion to its active form, minoxidil sulfate, by sulfotransferase enzymes in the scalp. However, research has shown that 40-60% of people may lack sufficient enzyme levels to effectively activate the medication. This enzymatic variability means that for many users, topical minoxidil may not be 100% effective.
Around 1.4% of topical minoxidil is systemically absorbed, further limiting minoxidil efficacy. To counteract this limitation, researchers have explored combining minoxidil with treatments like microneedling or retinoic acid. It was found that with these combinations, minoxidil efficacy can significantly increase.[6]Lama, S.B.C., Pérez-González, L.A., Kosoglu, M.A., Dennis, R., Ortega-Quijano, D. (2024). Physical Treatments and Therapies for Androgenetic Alopecia. Journal of Clinical Medicine. 13(15). 4534. … Continue reading
Oral Minoxidil
Oral minoxidil, however, is metabolized in the liver, where sulfotransferase enzymes are abundant. The activated minoxidil sulfate then enters the bloodstream, where it reaches the hair follicle. Studies consistently demonstrate higher response rates for oral minoxidil compared to topical.[7]Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2022). Minoxidil: a comprehensive review. Journal of Dermatological Treatment. 33(4). 1896-1906. Available at: … Continue reading However, because oral minoxidil involves systemic absorption, it comes with a broader potential for side effects.
What Are the Side Effects of Topical Minoxidil?
Common Side Effects
While topical minoxidil typically has a great safety profile, there are some side effects that people can experience.
Common side effects include:
- Scalp Irritation and Contact Dermatitis: Redness, itching, flaking, and burning sensations.
These are the most frequently reported side effects from minoxidil. One retrospective study found that 6.4% of men reported mainly irritant and allergic reactions to minoxidil.[8]Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelb). 13(5). 1157-1169. Available at: … Continue reading These effects are typically due to an allergic reaction to propylene glycol rather than the minoxidil itself.[9]Lessmann, H., Schnuch, A., Geier, J., Uter, W. (2005). Skin-sensitizing and irritant properties of propylene glycol. Contact Dermatitis. 53(5). 247-259. Available at: … Continue reading
To mitigate the negative skin effects of topical minoxidil, you can do several things. Starting with a lower concentration, such as 2% instead of 5%, can reduce the risk of irritation while still providing benefits. Additionally, reducing application frequency from twice to once daily can limit exposure. Switching to a foam-based minoxidil product can be particularly effective, as these formulations typically don’t contain propylene glycol, which is often responsible for uncomfortable side effects like irritation, redness, and scalp burning. Furthermore, incorporating a moisturizer into your scalp care routine can help keep the skin hydrated and comfortable, further alleviating potential irritation.
- Shedding Phase: Temporary increase in hair loss when starting treatment.
One study reported that 55% of topical minoxidil users experience minoxidil shedding.[10]Ghonemy, S., Bessar, H., Alarawi, A. (2019). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic … Continue reading This is considered a normal side effect and usually indicates that the treatment is working. The shedding phase typically begins 2 to 4 weeks after starting treatment and subsides within 6 to 8 weeks as the hair cycle normalizes. After a few months of continuous use, most users should start seeing visible new hair growth.[11]Kaiser, M., Abdin, R., Gaumond, S.I., Issa, T. N., Jiminez, J.J. (2023). Treatment of androgenetic alopecia: current guidance and unmet needs. Clinical Cosmetic and Investigational Dermatology. 16. … Continue reading
- Facial Hypertrichosis: Unwanted excess facial hair.
Women, especially those over 50 or with pre-existing facial hair, seem to be at higher risk of developing hypertrichosis from topical minoxidil use, and as with other side effects, it is more common when using the 5% than the 2% concentration.[12]Dawber, R.P.R, Rundegren, J. (2003). Hypertrichosis in females applying minoxidil topical solution and in normal controls. Journal of the European Academy of Dermatology and Venereology. 17(3). … Continue reading
Figure 2: A 42 year old woman with generalized hypertrichosis after using 5% topical minoxidil for two weeks.[13]Gargallo V, Gutierrez C, Vanaclocha F, Guerra-Tapia A. Hipertricosis generalizada secundaria a minoxidil tópico. Actas Dermosifiliogr. 2015;106:599–600. Available at: … Continue reading
There are several theories for why hypertrichosis occurs in people using topical minoxidil.
- Product application: The product wasn’t applied carefully enough or was not completely dried before bed, leading to product transfer from the pillow to the face. However, this doesn’t account for hypertrichosis seen away from the site of application and is unlikely to cause the effects seen in case studies.
- Percutaneous absorption: Although topical minoxidil is primarily intended for local effects, a small amount can be absorbed systemically through the skin. This can lead to circulating minoxidil reaching hair follicles in distant areas of the body.
- Follicular hypersensitivity: Some people may have hair follicles that are exceptionally responsive to minoxidil. In these cases, even minimal systemic exposure might be sufficient to stimulate hair growth in distant follicles. This hypersensitivity appears to occur randomly, depending on the person.
- Dose-dependent response: Higher concentrations are more likely to cause hypertrichosis.
While hypertrichosis can occur, it is generally reversible once minoxidil treatment is stopped and typically resolves within 3-4 months.
There are a number of ways that hypertrichosis can be avoided or treated once it occurs.
Starting with a lower concentration can reduce the risk, especially for women and those with a history of excess facial hair. Careful application to the scalp only, allowing proper drying time, and adhering to the recommended dosage can also minimize systemic absorption and unintended spread.
Switching to a foam version may help those experiencing side effects. If you are experiencing mild hypertrichosis and don’t want to stop using minoxidil, you could use hair removal methods while continuing treatment.
For more severe cases, spironolactone (~25 mg daily) ando/or low-dose bicalutamide (~10 mg daily) have shown promise in managing minoxidil-induced hypertrichosis. However, these medications should be used under medical supervision.[14]Darendeliler, F., Bas, F., Balaban, S., Bundak, R., Demirkol, D., Saka, N., Gunoz, H. (1996). Spironolactone therapy in hypertrichosis. European Journal of Endocrinology. 135(5).604-608. Available … Continue reading,[15]Moussa, A., Kazmi, A., Bakhari, L., Sinclair, R.D. (2022). Bicalutamide improves minoxidil-induced hypertrichosis in female pattern hair loss: a retrospective review of 35 patients. Journal of the … Continue reading
- Headaches
Some people also experience headaches after using topical minoxidil. One study found that in users applying 2% minoxidil solution, 0.6% reported headaches, compared to 3% of participants using 5% minoxidil solution.[16]Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786. Available at: … Continue reading
Some people may simply experience headaches due to the smell of the product they are using, in which case, switching to an alcohol-free or unscented alternative may help. Others may be particularly sensitive to minoxidil and its vasodilatory effects, which might contribute to headaches. In this case, switching to a lower concentration or consulting with a healthcare provider might be preferable.
Some of our members have also switched to nanoxidil, an analogue of minoxidil that may offer a better safety profile (you can read more about the research quality of nanoxidil here), and found that their headaches resolved.
The above side effects are more often seen in the 5% than 2% solutions and are typically considered to be non-serious.[17]Nestor, M.S., Ablon, G., Gade, A., Han, H., Fischer, D.L. (2021). Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics. Journal of … Continue reading
Some of the rarer side effects include:
- Water Retention
While less common than with oral minoxidil, topical minoxidil application can lead to water retention. Some topical minoxidil users have reported under-eye bags, which may be due to increased water retention near the application areas. There are also anecdotal reports of “puffy face” from topical minoxidil application, suggesting localized fluid retention.[18]Gungor, S., Kocaturk, E., Topal, I.O. (2015). Frontal Edema Due to Topical Application of %5 Minoxidil Solution Following Mesotherapy Injections. International Journal of Trichology. 7(2). 86-87. … Continue reading
Anecdotally, there have also been reports of swollen feet and weight gain from using topical minoxidil; however, we couldn’t find these reports reflected in peer-reviewed literature. These effects may also stop with continued use of the drug, so some people keep an eye on the symptoms and wait to see if they go away.
However, you can try reducing the concentration or frequency of usage if the symptoms continue longer than you are comfortable with. Furthermore, excessive salt intake can exacerbate symptoms of edema. Limiting salt intake may help you resolve the edema without having to stop using minoxidil.[19]Patel, P., Nessel, T.A., Kumar, D. (2023). In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: … Continue reading
- Cardiovascular Issues
Some people also experience cardiovascular side effects. One case study found that applying large amounts of 2% topical minoxidil led to hypotension (low blood pressure) and feelings of faintness.[20]Ponomareva, M.A., Romanova, M.A., Shapshnikova, A.A., Piavchenko, G.A. (2024). Topical Minoxidil Overdose in a Young Man with Androgenetic Alopecia: A Case Report. Cureus. 16(6). E62382. Available … Continue reading Another case also documented low blood pressure and fainting after applying 12.5% topical minoxidil daily.[21]Dubrey, S.W., vanGriethuysen, J., Edwards, C.M.B. A hairy fall: syncope resulting from topical application of minoxidil. BMJ Case Reports. 1-2. Available at: https://doi.org/10.1136/bcr-2015-210945
Other people may experience heart palpitations, feelings of the heart beating rapidly or “skipping a beat”. While this is rare, one study found that 3.5% of women developed heart palpitations or a rapid heart rate after usage of 2% topical minoxidil solution compared to 1.8% who were using a 5% foam.[22]Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., Bartels, N.G. (2011). A randomized, single-blind trial of 5% minoxidil foam once daily versus 2% minoxidil solution twice daily in the … Continue reading
Therefore, we would recommend seeking medical advice and potentially finding a new treatment for hair loss if you experience these side effects.
So we’ve covered the side effects of topical minoxidil, but what about oral?
What Are the Side Effects of Oral Minoxidil?
Oral minoxidil side effects are similar to those of topical minoxidil, and both forms exhibit dose-dependent effects. However, oral administration leads to significantly greater systemic exposure to the drug than topical application. As a result, oral minoxidil typically produces more pronounced hair regrowth and more pronounced systemic side effects. It should be noted that use of oral minoxidil to improve hair regrowth is an off-label use of the drug and it is not FDA-approved for this indication.
Common Side Effects
- Hair Shedding
Like with topical minoxidil, you may experience increased hair shedding when you start taking minoxidil. This is considered to be a normal part of the hair cycle remodeling process, typically beginning two to four weeks after starting treatment and subsiding within six to eight weeks as the hair cycle normalizes.
- Hypertrichosis
While rare with topical minoxidil use, one of the most frequently reported side effects of oral minoxidil is hypertrichosis, which involves excessive hair growth on various parts of the body. This effect is dose-dependent, with some studies showing that increasing the dosage of oral minoxidil by just 1 mg daily is associated with a 17.6% increased risk of hypertrichosis.[23]Gupta, A.K., Hall, D.C., Talukder, M., Bamimore, M.A. (2022). There is a Positive Dose-Dependent Association between Low-Dose Oral Minoxidil and Its Efficacy for Androgenetic Alopecia: Findings from … Continue reading
Hypertrichosis frequently occurs on the face (sideburns, temples, upper lip, and chin), with rarer cases of generalized hypertrichosis occurring over the whole body.[24]Desai, D.D., Nohria, A., Brinks, A., Needle, C., Shapiro, J., Lo Sicco, K.I. (2024). Minoxidil-induced hypertrichosis: Pathophysiology, clinical implications, and therapeutic strategies. JAAD … Continue reading
- Fluid Retention
Fluid retention is another common side effect of oral minoxidil, occurring in about 1.3% of patients.[25]Trueb, R.M., Caballero-Uribe, N., Luu, N.N.C., Dmitriev, A. (2022). Serious complication of low-dose oral minoxidil for hair loss. JAAD Case Reports. 30. 97-98. Available at: … Continue reading This can manifest as mild swelling in the face, hands, or feet. In some cases, it may lead to rapid weight gain.
This weight gain can be significant and sudden, with some patients experiencing up to 20 pounds of weight gain (as water weight) in as little as one month.[26]Patel.K., Omar, J. (2023). Low dose oral minoxidil causing peripheral edema and rapid weight gain. Journal of General Internal Medicine. 38(Suppl 3). S592. Available at: … Continue reading
Fluid retention can manifest in several ways:
- Peripheral edema: Swelling in the extremities, particularly in the legs, ankles, and feet.
- Facial swelling: Puffiness or swelling in the face.
- Overall weight gain: A sudden increase in body weight, often 5 pounds or more.
This rapid retention can be concerning for patients and may lead to additional health risks if left unmanaged. Excess fluid in the body can potentially lead to congestive heart failure if not properly addressed.
To mitigate these side effects, there are a number of options:
- Use of diuretics: Doctors can prescribe a diuretic (water pill) alongside minoxidil to help counteract fluid retention. This is typically a low-dose loop diuretic.
- Sodium restriction: Limiting sodium intake can help reduce fluid retention. Patients are often not advised about this when starting minoxidil, which can exacerbate the problem.
- Dose adjustment: If fluid retention becomes severe, temporarily discontinuing minoxidil or significantly reducing the dose may be necessary. This allows the edema to resolve naturally before restarting at a lower dose.
- Regular monitoring: Daily weight monitoring can help identify fluid retention early, making it easier to manage.
- Gradual titration: Slowly increasing the dose of minoxidil over time, rather than starting with 5 mg, can help minimize side effects.
If these symptoms don’t resolve when trying these strategies, then it’s recommended to visit your doctor and potentially stop taking minoxidil.
- Lightheadedness, Tachycardia (high heart rate), Headache and Insomnia
Oral minoxidil can cause several cardiovascular and neurological side effects, including lightheadedness, tachycardia, headache, and insomnia.[27]Trueb, R.M., Caballero-Uribe, N., Luu, N.N.C., Dmitriev, A. Serious complication of low-dose oral minoxidil for hair loss. JAAD Case Reports. 30. 97-98. Available at: … Continue reading These effects are generally dose-dependent and more common at higher doses.
Lightheadedness has been reported to occur in 1.7% of patients, tachycardia in 0.9%, headache in 0.4%, and insomnia in 0.2% of patients. These can all be symptoms of decreased blood pressure due to minoxidil’s vasodilatory properties.
Headaches and insomnia are reported in around 0.4% and 0.2% of patients, respectively, using low-dose oral minoxidil. While the exact mechanism of these side effects is not clear, it is thought that it could be related to the vasodilatory effects of the drug.
What Are The Cardiovascular Concerns of Oral Minoxidil Usage?
The severity and frequency of cardiovascular side effects are closely tied to the dosage of oral minoxidil. A meta-regression analysis found a positive dose-dependent correlation between low-dose oral minoxidil and the risk of cardiovascular adverse events.
Low Dose (0.25-1.25 mg/day)
At lower doses, oral minoxidil is generally well-tolerated. Women typically start with doses ≤ 1 mg, which minimizes the risk of significant side effects. Lower doses are considered to be a safer starting point for most patients, and even very low doses (0.25 mg/day) have shown efficacy in some studies.[28]Ramírez-Marín, H.A., Tosti, A. (2022). Role of oral minoxidil in patterned hair loss. Indian Dermatology Online Journal. 13(6). 729-733. Available at: https://doi.org/10.4103/idoj.idoj_246_22 If you are experiencing side effects at higher doses, you can reduce your dose at home using a pill cutter.
Moderate Dose (2.5-5 mg/day)
Men may be prescribed up to 5 mg of minoxidil daily, which can increase the likelihood of side effects such as dizziness and fluid retention. A recent study examined the effects of 7.5 mg/day oral minoxidil in patients with normal blood pressure and AGA.[29]Sanabria, B.D., Perdomo, Y.C., Miot, H.A., Ramos, P.M. (2024). Oral minoxidil 7.5 mg for hair loss increases heart rate with no change in blood pressure in 24 h ambulatory blood pressure monitoring. … Continue reading The results showed a mild increase in heart rate but no significant changes in blood pressure, suggesting that doses slightly higher than the typical 5 mg can be tolerated. However, this should only be considered under medical supervision.
Figure 3: Heart rate and blood pressure monitoring of 11 adult males with AGA after 24 weeks (T24) of treatment with 5 mg/day of oral minoxidil and after 6 weeks (T30) of treatment with 7.5 mg/day of oral minoxidil.[30]Sanabria, B.D., Perdomo, Y.C., Miot, H.A., Ramos, P.M. (2024). Oral minoxidil 7.5 mg for hair loss increases heart rate with no change in blood pressure in 24 h ambulatory blood pressure monitoring. … Continue reading
High Dose (10 mg/day and above)
Doses above 10 mg daily are associated with a higher risk of serious cardiac events and are typically not recommended for hair loss treatment. The hypotensive effect of oral minoxidil becomes more significant at these higher doses and is often prescribed alongside beta blockers and diuretics to manage the side effects.
How Can I Mitigate Oral Minoxidil Side Effects?
We have covered a number of ways to mitigate oral minoxidil side effects, but there are some further ways that you can adjust the use of minoxidil to reduce your risk.
Split-Dosing
Splitting the daily dosage of oral minoxidil into two administrations, one in the morning and one in the evening, can potentially optimize its efficacy while minimizing side effects. This approach is based on the pharmacokinetics of oral minoxidil, which has a relatively short half-life of approximately 3-4 hours.[31]Vano-Galvan, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Saceda-Corralo, D., Rodrigues-Barata, R., Jiminez-Cauhe, J., Koh, W.L., Poa, J.E., Jerjen, R., de Carvalho, L.T., John, J.M., … Continue reading
By dividing the total daily dose, you can maintain more consistent blood levels of minoxidil throughout the day, potentially leading to more stable hair growth stimulation. For example, if 5 mg is prescribed daily, taking 2.5 mg in the morning and 2.5 mg in the evening may be more beneficial than a single 5 mg dose.
Sublingual Minoxidil
Some recommend using sublingual minoxidil as an alternative to traditional oral minoxidil. Sublingual administration involves a tablet that dissolves under the tongue. One 2021 randomized, double-blind, placebo-controlled phase 1b clinical trial investigated this delivery method.[32]Bokhari, L., Jones, L.N., Sinclair, R.D. (2021). Sublingual minoxidil for the treatment of male and female pattern hair loss: a randomized, double-blind, placebo-controlled, phase 1B clinical trial. … Continue reading The study tested daily doses of 0.45 mg to 4.05 mg of sublingual minoxidil.
This method offers several advantages:
- Bypasses first-pass metabolism in the liver.
- It allows for direct entry into the bloodstream, which can travel inactivated to the scalp and be activated by sulfotransferase at the site.
- Reduces systemic side effects.
- Improves bioavailability.
Key findings from the study included a dose-dependent improvement in hair parameters, with reduced side effects compared to oral minoxidil and no significant effect on blood pressure. In the blood, peak serum concentrations of minoxidil were only 10% of those seen with typical oral minoxidil.
Figure 4: Effect of different doses of sub-lingual minoxidil on hair regrowth outcomes after 24 weeks.[33]Bokhari, L., Jones, L.N., Sinclair, R.D. (2021). Sublingual minoxidil for the treatment of male and female pattern hair loss: a randomized, double-blind, placebo-controlled, phase 1B clinical trial. … Continue reading
At the 24-week follow-up, approximately 45% of patients in the 0.45 mg sublingual minoxidil group experienced improvements in frontal hair density, and 55% showed vertex improvement. Higher doses (4.05 mg) led to further results, with nearly 67% of patients experiencing improvements in both frontal and vertex hair density.
Sublingual minoxidil appears to be particularly beneficial for people concerned about the side effects of oral minoxidil. The medication was undetectable in plasma after 24 hours, and the mean peak minoxidil plasma concentration was significantly below the threshold associated with changes in blood pressure.
While the results are promising, it should be noted that this is the only study using sublingual minoxidil for AGA, and further studies with larger patient numbers are needed.
Switch To Topical Minoxidil
There is a chance that none of these options will work out for you, so you can try to switch to topical minoxidil. The side effects are more manageable for topical treatments, meaning that you can increase the dose and try to pair them with other treatments like microneedling or retinoic acid to further improve hair growth outcomes.
Can I Use Minoxidil If I Am Planning a Family?
Medical professionals generally advise against using both topical and oral minoxidil for both men and women when planning a family and for women during pregnancy and while breastfeeding.
While there is limited data on human pregnancies, animal studies have shown potential risks, including evidence of increased fetal resorption at high doses, one case report of fetal malformation associated with topical minoxidil use, and neonatal hypertrichosis reported following exposure during pregnancy.[34]Drugs. (2023). Minoxidil pregnancy and breastfeeding warnings. Drugs.com. Available at: https://www.drugs.com/pregnancy/minoxidil.html (Accessed: February 2025),[35]Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., Moroni, F. (2003). Topically applied minoxidil may cause fetal malformation: a case report. Birth defects research. Part A, Clinical and … Continue reading
There is a significant lack of well-controlled studies on minoxidil use during pregnancy and lactation. However, given the animal studies, medical professionals typically recommend avoiding minoxidil use when planning pregnancy, during pregnancy, and while breastfeeding, using adequate contraception if taking minoxidil, and discontinuing minoxidil use before attempting to conceive.[36]National Institute of Health and Care Excellence. (2021). Topical minoxidil. NICE. Available at: … Continue reading
Does Minoxidil Affect Male Fertility?
Current research suggests that minoxidil has minimal to no direct impact on male fertility. However, some research has linked minoxidil with oxidative stress and morphological changes to the testicles, which could indicate a potential negative impact.[37]Santana, F.F.V., Lozi, A.A., Goncalves, R.V., Silva, J.D., Matta, S.L.P.D. (2023). Comparative effects of finasteride and minoxidil on the male reproductive organs: A systematic review of in vitro … Continue reading
If you’re thinking about trying minoxidil and you are trying to conceive or have a pregnant partner, then it is advisable to talk to a medical professional before starting any treatment.
What if I’m Still Experiencing Side Effects?
Fortunately, minoxidil is just one of several treatment options available. If you’ve tried all of them and still experience side effects, you might consider exploring alternative therapies.
We have a wealth of information available so you can weigh your options and find out exactly how each treatment works and what your regrowth roadmap might look like. If you have any questions, reach out in the dedicated discussion thread below.
Final Thoughts
While minoxidil remains one of the most widely used treatments for AGA, both topical and oral formulations present unique challenges. The choice of which to use should be weighed carefully against the side effects, varying from mild scalp irritation to more significant cardiovascular effects. Ultimately, while the research supports minoxidil’s efficacy, it is not the only option out there, and if it isn’t working for you, then it is important to find the right one.
References[+]
References ↑1 Bryan, J. (2011). How minoxidil was transformed from an antihypertensive to hair-loss drug. The Pharmaceutical Journal. Available at: https://pharmaceutical-journal.com/article/news/how-minoxidil-was-transformed-from-an-antihypertensive-to-hair-loss-drug#:~:text=DAMN%2DO%20was%20effective%20in,seen%20in%20canine%20toxicity%20studies.&text=Despite%20the%20adverse%20effects%2C%20demand,week%20limit%20on%20treatment%20duration.&text=Owing%20to%20the%20drug’s%20effectiveness,of%20hypertrichosis%20began%20to%20emerge. (Accessed: February 2025) ↑2 Alhayaza, G., Hakami, A., AlMarzouk, L.H., Al Qurashi, A.A., Alghamdi, G., Alharithy, R. (2023). Topical minoxidil reported hair discoloration: a cross-sectional study. Dermatology Reports. 16(1). 9745. Available at: https://doi.org/10.4081/dr.2023.9745 ↑3 Shen, Y., Zhu, Y., Zhang, L., Sun, J., Xie, B., Zhang, H., Song, X. (2023). New Target for Minoxidil in the Treatment of Androgenetic Alopecia. Drug Design, Development and Therapy. 17. 2537-2547. Available at: https://doi.org/10.2147/DDDT.S427612 ↑4 Dhurat, R., Daruwalla, S., Pai, S., Kovacevic, M., McCoy, J., Shapiro, J., Sinclair, R., Vano-Galvan, S., Goren, A. (2021). SULT1A1 (Minoxidil Sulfotransferase) enzyme booster significantly improves response to topical minoxidil for hair growth. 21(1). 343-346. Available at: https://doi.org/10.1111/jocd.14299 ↑5 Anderson, R.J., Kudlacek, P.E., Clemens, D.L. (1998). Sulfation of minoxidil by multiple human cytosolic sulfotransferases. Chemico-Biological Interactions. 109. 53-67. Available at: https://doi.org/10.1016/S0009-2797(97)00120-8 ↑6 Lama, S.B.C., Pérez-González, L.A., Kosoglu, M.A., Dennis, R., Ortega-Quijano, D. (2024). Physical Treatments and Therapies for Androgenetic Alopecia. Journal of Clinical Medicine. 13(15). 4534. Available at: https://doi.org/10.3390/jcm13154534 ↑7 Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2022). Minoxidil: a comprehensive review. Journal of Dermatological Treatment. 33(4). 1896-1906. Available at: https://doi.org/10.1080/09546634.2021.1945527 ↑8 Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelb). 13(5). 1157-1169. Available at: https://doi.org/10.1007/s13555-023-00919-x ↑9 Lessmann, H., Schnuch, A., Geier, J., Uter, W. (2005). Skin-sensitizing and irritant properties of propylene glycol. Contact Dermatitis. 53(5). 247-259. Available at: https://doi.org/10.1111/j.0105-1873.2005.00693.x. ↑10 Ghonemy, S., Bessar, H., Alarawi, A. (2019). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic evaluation. Journal of Dermatological Treatment. 32(2). 236-241. Available at: https://doi.org/10.1080/09546634.2019.1654070 ↑11 Kaiser, M., Abdin, R., Gaumond, S.I., Issa, T. N., Jiminez, J.J. (2023). Treatment of androgenetic alopecia: current guidance and unmet needs. Clinical Cosmetic and Investigational Dermatology. 16. 1387-1406. Available at: https://doi.org/10.2147/CCID.S385861 ↑12 Dawber, R.P.R, Rundegren, J. (2003). Hypertrichosis in females applying minoxidil topical solution and in normal controls. Journal of the European Academy of Dermatology and Venereology. 17(3). 271-275. Available at: https://doi.org/10.1046/j.1468-3083.2003.00621.x. ↑13 Gargallo V, Gutierrez C, Vanaclocha F, Guerra-Tapia A. Hipertricosis generalizada secundaria a minoxidil tópico. Actas Dermosifiliogr. 2015;106:599–600. Available at: https://doi.org/10.1016/j.adengl.2015.06.019 ↑14 Darendeliler, F., Bas, F., Balaban, S., Bundak, R., Demirkol, D., Saka, N., Gunoz, H. (1996). Spironolactone therapy in hypertrichosis. European Journal of Endocrinology. 135(5).604-608. Available at: https://doi.org/10.1530/eje.01350604 ↑15 Moussa, A., Kazmi, A., Bakhari, L., Sinclair, R.D. (2022). Bicalutamide improves minoxidil-induced hypertrichosis in female pattern hair loss: a retrospective review of 35 patients. Journal of the American Acadamy of Dermatology. 87(2). 488-490. Available at: https://doi.org/10.1016/j.jaad.2021.10.048 ↑16 Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑17 Nestor, M.S., Ablon, G., Gade, A., Han, H., Fischer, D.L. (2021). Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics. Journal of Cosmetic Dermatology. 20. 3759-3781. Available at: https://doi.org/10.1111/jocd.14537 ↑18 Gungor, S., Kocaturk, E., Topal, I.O. (2015). Frontal Edema Due to Topical Application of %5 Minoxidil Solution Following Mesotherapy Injections. International Journal of Trichology. 7(2). 86-87. Available at: https://doi.org/10.4103/0974-7753.160124 ↑19 Patel, P., Nessel, T.A., Kumar, D. (2023). In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: February 2025) ↑20 Ponomareva, M.A., Romanova, M.A., Shapshnikova, A.A., Piavchenko, G.A. (2024). Topical Minoxidil Overdose in a Young Man with Androgenetic Alopecia: A Case Report. Cureus. 16(6). E62382. Available at: https://doi.org/10.7759/cureus.62382 ↑21 Dubrey, S.W., vanGriethuysen, J., Edwards, C.M.B. A hairy fall: syncope resulting from topical application of minoxidil. BMJ Case Reports. 1-2. Available at: https://doi.org/10.1136/bcr-2015-210945 ↑22 Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., Bartels, N.G. (2011). A randomized, single-blind trial of 5% minoxidil foam once daily versus 2% minoxidil solution twice daily in the treatment of androgenetic alopecia in women. Journal of the American Academy of Dermatology. 65(6). 1126-1134. Available at: https://doi.org/10.1016/j.jaas.2010.09.724 ↑23 Gupta, A.K., Hall, D.C., Talukder, M., Bamimore, M.A. (2022). There is a Positive Dose-Dependent Association between Low-Dose Oral Minoxidil and Its Efficacy for Androgenetic Alopecia: Findings from a Systematic Review with Meta-Regression Analyses. Skin Appendage Disorders. 8(5). 355-361. Available at: https://doi.org/10.1159/000525137 ↑24 Desai, D.D., Nohria, A., Brinks, A., Needle, C., Shapiro, J., Lo Sicco, K.I. (2024). Minoxidil-induced hypertrichosis: Pathophysiology, clinical implications, and therapeutic strategies. JAAD Reviews. 2. 41-49. Available at: https://doi.org/10.1016/j.jdrv.2024.08.002 ↑25 Trueb, R.M., Caballero-Uribe, N., Luu, N.N.C., Dmitriev, A. (2022). Serious complication of low-dose oral minoxidil for hair loss. JAAD Case Reports. 30. 97-98. Available at: https://doi.org/10.1016/j.jdcr.2022.09.035 ↑26 Patel.K., Omar, J. (2023). Low dose oral minoxidil causing peripheral edema and rapid weight gain. Journal of General Internal Medicine. 38(Suppl 3). S592. Available at: https://scholarlycommons.henryford.com/internalmedicine_mtgabstracts/162/ (Accessed: February 2024 ↑27 Trueb, R.M., Caballero-Uribe, N., Luu, N.N.C., Dmitriev, A. Serious complication of low-dose oral minoxidil for hair loss. JAAD Case Reports. 30. 97-98. Available at: https://doi.org/10.1016/j.jdcr.2022.09.035 ↑28 Ramírez-Marín, H.A., Tosti, A. (2022). Role of oral minoxidil in patterned hair loss. Indian Dermatology Online Journal. 13(6). 729-733. Available at: https://doi.org/10.4103/idoj.idoj_246_22 ↑29 Sanabria, B.D., Perdomo, Y.C., Miot, H.A., Ramos, P.M. (2024). Oral minoxidil 7.5 mg for hair loss increases heart rate with no change in blood pressure in 24 h ambulatory blood pressure monitoring. Anais Brasileiros de Dermatologia. 99(5). 734-736. Available at: https://doi.org/10.1016/j.abd.2023.08.016 ↑30 Sanabria, B.D., Perdomo, Y.C., Miot, H.A., Ramos, P.M. (2024). Oral minoxidil 7.5 mg for hair loss increases heart rate with no change in blood pressure in 24 h ambulatory blood pressure monitoring. Anais Brasileiros de Dermatologia. 99(5). 734-736. Available at: https://doi.org/10.1016/j.abd.2023.08.016 ↑31 Vano-Galvan, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Saceda-Corralo, D., Rodrigues-Barata, R., Jiminez-Cauhe, J., Koh, W.L., Poa, J.E., Jerjen, R., de Carvalho, L.T., John, J.M., Salas-Callo, C.I., Vincenzi, C., Yin, L., Lo-Sicco, K., Waskiel-Burnat, A., Starace, M., Zamorano, J.L., Jaen-Olasolo, P., Piraccini, B.M., Rudnicka, L., Shapiro, J., Tosti, A., Sinclair, R., Bhoyrul, B. (2021). Safety of low-dose oral minoxidil for hair loss: A multicenter study of 1404 patients. Journal of the American Academy of Dermatology. 84(6). 1644-1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑32 Bokhari, L., Jones, L.N., Sinclair, R.D. (2021). Sublingual minoxidil for the treatment of male and female pattern hair loss: a randomized, double-blind, placebo-controlled, phase 1B clinical trial. Journal of the European Academy of Dermatology and Venereology. (36)1. E62-e66. Available at: https://doi.org/10.1111/jdv.17623 ↑33 Bokhari, L., Jones, L.N., Sinclair, R.D. (2021). Sublingual minoxidil for the treatment of male and female pattern hair loss: a randomized, double-blind, placebo-controlled, phase 1B clinical trial. Journal of the European Academy of Dermatology and Venereology. (36)1. E62-e66. Available at: https://doi.org/10.1111/jdv.17623 ↑34 Drugs. (2023). Minoxidil pregnancy and breastfeeding warnings. Drugs.com. Available at: https://www.drugs.com/pregnancy/minoxidil.html (Accessed: February 2025) ↑35 Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., Moroni, F. (2003). Topically applied minoxidil may cause fetal malformation: a case report. Birth defects research. Part A, Clinical and molecular teratology. 67(12). 997-1001. Available at: https://doi.org/10.1002/bdra.10095 ↑36 National Institute of Health and Care Excellence. (2021). Topical minoxidil. NICE. Available at: https://cks.nice.org.uk/topics/female-pattern-hair-loss-female-androgenetic-alopecia/prescribing-information/topical-minoxidil/ (Accessed: February 2025) ↑37 Santana, F.F.V., Lozi, A.A., Goncalves, R.V., Silva, J.D., Matta, S.L.P.D. (2023). Comparative effects of finasteride and minoxidil on the male reproductive organs: A systematic review of in vitro and in vivo evidence. Toxicology and Applied Pharmacology. 478. 11670. Available at: https://doi.org/10.1016/j.taap.2023.116710. Research continues to show that oral minoxidil is an effective off-label treatment for men with pattern hair loss. The general rule-of-thumb: the bigger the dose, the better hair regrowth. But there’s a catch…
Higher dosages of oral minoxidil come at a risk of higher risk of side effects: excessive body hair growth, limb swelling, low blood pressure, and even heart palpitations.
Knowing this, is there a “best dose” for oral minoxidil (in mg) for men with pattern hair loss? More specifically, which dose of oral minoxidil maximizes our chances for hair regrowth and minimizes our risks of adverse events?
This Quick Win uncovers the latest research. The short answer: studies show that 2.5mg daily seems to be a tolerable, effective dose for most men with pattern hair loss. But the right dose for you will depend on (1) your severity of hair loss, and (2) your tolerance with side effects (not all of them are bad).
Note: Quick Wins are short articles focused on answering one question about hair loss. Given their specificity, these articles are written in a more scientific tone. If you’re new to hair loss education, start with these articles.
Interested in Oral Minoxidil?
Low-dose oral minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Oral minoxidil: research at a glance
To date, there have been fewer than 10 studies published on oral minoxidil for androgenic alopecia (AGA). Doses studied range from 0.25mg to 5.0mg daily, and study durations (at least the ones we evaluated) range from 24-52 weeks.
Across studies, there is a clear trend: the higher the dose of oral minoxidil, the better the hair regrowth. But this relationship doesn’t tell the whole story – as these higher dosages seem to confer with higher reports of side effects.
So, here’s what you should know before starting any daily dose of oral minoxidil.
#1: Oral minoxidil dosages from 0.25mg to 5mg improve pattern hair loss
The three most recent (and most robust) studies on oral minoxidil all varied dosing by 0.25mg, 2.5mg, and 5.0mg daily. All of them showed benefit – with higher dosages demonstrating visual improvements. Just see these photos of a male who took 5mg of oral minoxidil daily for 3 months.[1]Jimenez-Cauhe, Juan et al. Effectiveness and safety of low-dose oral minoxidil in male androgenetic alopecia. Journal of the American Academy of Dermatology, Volume 81, Issue 2, 648 – 649
Male with AGA: improvement with 5mg oral minoxidil over 3 months
So, let’s organize the findings of these three studies by dosage. Then, let’s evaluate their results in terms of:
- Response rates (i.e., the percent of people who experienced an improvement), and…
- Side effects (i.e., unintended effects of the drug)
Do we see any trends in data? Specifically, is there a “sweet spot” where most men can maximize their chances of hair regrowth from oral minoxidil while minimizing their risk of serious side effects?
Yes.
Oral minoxidil for AGA: study results by dosage
See each study’s summaries.
Daily Dose (Duration)
Response Rate
Side Effects
0.25 mg
(6+ months)[2]Pirmez, Rodrigo et al. Very-low-dose oral minoxidil in male androgenetic alopecia: A study with quantitative trichoscopic documentation. Journal of the American Academy of Dermatology, Volume 82, … Continue reading60%
90%:
increased beard growth (52%), increased body hair (20%), hair shedding (16%), pedal edema (4%)2.5 mg to 5 mg
(6-12 months)[3]Jimenez-Cauhe, Juan et al. Effectiveness and safety of low-dose oral minoxidil in male androgenetic alopecia. Journal of the American Academy of Dermatology, Volume 81, Issue 2, 648 – 64990%
29.3%:
increased body hair (24.3%), lower limb edema (4.8%), hair shedding (2.4%)5 mg
(6 months)[4]Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Journal of the American Academy of Dermatology, Volume 72, Issue 5, AB113100%
100%:
increased body hair growth (93%), pedal edema (10%), heart / EKG alterations (10%)At first glance, the risk of side effects across even small dosages seems ridiculously high (90%+).
However, not all of these side effects are bad.
For instance, of the side effects reported in these studies, the overwhelming majority of them constituted increased body/facial hair growth. Most men aren’t going to care about this. In fact, man men might even prefer more body or facial hair.
Secondly, increased hair shedding was generally only reported at the beginning of each study. This is because, when starting minoxidil (topical or oral), the drug can “kickstart” a new anagen (growth) phase of hairs affected by androgenic alopecia (AGA). This can lead us to shed any hairs that were already primed to fall out soon anyway – specifically, catagen or telogen hairs – thereby giving the illusion of thinner hair in the first 1-2 months of treatment. However, as these hairs grow back, they’re usually much thicker, and thereby improve hair density. Long-story short: in most cases, hair shedding from minoxidil isn’t long-lived.
So, for this exercise, let’s discount increased body/facial hair growth and increased hair shedding as temporary and/or non-problematic side effects. Instead, let’s re-run our analysis and only consider serious side effects – edema, EKG alterations, etc.
Within this context, are all dosages of oral minoxidil as scary?
No. In fact, it seems like there’s a “sweet spot” for oral minoxidil where we can maximize hair regrowth while minimizing our risk of bad side effects: at 2.5mg daily.
Keep in mind the following chart is based on preliminary data on low-dose oral minoxidil, and that this article reflects the clinical studies available on oral minoxidil for androgenic alopecia as of 2020. As better-designed studies are published, these numbers will evolve:
Moreover, it’s really only at dosages of 5mg that we see an appreciable increase in concerning side effects – namely, edema (water retention / swelling) and cardiac alterations (i.e., lower heart rates).
So, 2.5 mg daily of oral minoxidil might be the “sweet spot” for most male pattern hair loss sufferers.
#2: Oral minoxidil’s efficacy may vary depending on hair loss severity
Most clinical trials on androgenic alopecia will select study participants who all have similar severities of hair loss. This is known as standardization. And for most trials, investigators usually prefer men who have medium-severity androgenic alopecia (i.e., Norwood 3-4). This is usually because men with Norwood 3-4 level hair loss (1) are representative of the population of hair loss sufferers who may later opt for this treatment, and (2) have enough hair follicle miniaturization and hair loss to effectively evaluate cosmetic improvements to hair thinning.
Having said that, most studies on oral minoxidil aren’t standardized to Norwood 3-4 participants. So, it’s a bit disingenuous to make comparisons across studies for response rates and side effects. In other words, please take our above analysis with a grain of salt.
With that said, with different age and/or hair loss severity across studies, we can get more granular data on who tends to respond well to oral minoxidil.
Based on the above studies (and others we looked into for our analysis), the trend aligned with intuition: if you don’t have severe hair loss, you can get away with lower dosages of oral minoxidil. If you do have severe hair loss, you’ll need a higher dose.
In other words:
- If you have mild male pattern baldness, 0.25mg daily might work fine as a starting point
- A more moderate case of hair loss might require a dose of 1-2.5mg daily
- If you have severe male pattern baldness, you’re probably going to need closer to 5mg daily
- If other treatments haven’t worked for you (resistant hair loss), you may also need a higher daily dose (2.5-5mg) or combination treatments with finasteride
Then again, higher doses come with more side effects. This is where dosing gets hyper-specific.
#3: If you’re going to try oral minoxidil, you’ll need to work with a doctor to figure out your unique risk profile and the right dose
Oral minoxidil is an antihypertensive drug (lowers blood pressure). It can also cause fluid retention. Therefore, if your health history indicates problems surrounding low blood pressure, fainting spells, or edema (swelling), you may be at a higher risk of complications from taking the drug.
Moreover, oral minoxidil can stimulate hair growth everywhere… not just on the scalp. For some men, this may be a bonus. For others, it might be a drawback. If this is a drawback for you, then it’s worth noting that reports of increased body / facial hair even occurred at lower dosages (0.25 mg) of oral minoxidil. So, if you’re concerned about this, maybe oral minoxidil isn’t right for you.
Long-tory short: for the safest and most effective use of oral minoxidil, discuss your medical history and preferences with your doctor. Then convince him or her to prescribe you oral minoxidil.
Note: a dermatologist specializing in hair loss is much more likely to write you a prescription. So, if you don’t want to waste any time, make a list of dermatologists in your area, call them to see if they’re open to prescribing oral minoxidil, and then only visit the ones who prescribe the drug.
#4: Like topical minoxidil, oral minoxidil likely works better alongside other treatments
When it comes to treating pattern hair loss, combination treatments tend to almost always outperform mono-treatments.
In some cases, combination therapies allow us to use the lowest dose of a drug possible without sacrificing results. Some studies suggest this is the case for women with pattern hair loss who take 0.25mg of oral minoxidil + 25mg of spironolactone: they minimize the risk of side effects of either drug while getting hair regrowth that often exceeds that of high dosages of either drug.
In other cases, combination therapies can actually enhance the efficacy of drugs. This tends to be true of men taking topical minoxidil, and who then add in once-weekly microneedling, thereby making topical minoxidil 400% more effective (according to some investigation groups). [5]English RS Jr, Ruiz S, DoAmaral P. Microneedling and Its Use in Hair Loss Disorders: A Systematic Review. Dermatol Ther (Heidelb). 2022 Jan;12(1):41-60. doi: 10.1007/s13555-021-00653-2. Epub 2021 Dec … Continue reading
While there aren’t many studies that exhaustively explore this relationship for oral minoxidil, the odds are that this medication also works better as a combination therapy. So, if you’re going to commit to oral minoxidil, consider stacking it with other therapies.
Again, research here is limited, but there are a host of things you can try in combination with oral minoxidil that might increase results.
- DHT reducers (finasteride, saw palmetto, etc.)
- Microneedling
- Massage
- Ketoconazole shampoo
- Low-level laser therapy (LLLT)
- Platelet-rich plasma therapy (PRP)
…and more.
The bottom line
When it comes to oral minoxidil, the best daily dosage for men with pattern hair loss may vary depending on(1) your tolerance for certain side effects, and (2) your severity of hair loss. Consider these recommendations a mere starting point until more research emerges:
- Men with mild AGA, men at a higher risk of complication, or men who don’t want extra hair growth on their body/face: 0.25-1mg.
- Men with moderate AGA: 1-2.5mg.
- Men with resistant AGA (you’ve tried other treatments and they haven’t worked) or severe AGA: 2.5-5mg.
Although these guidelines are a rough ballpark, chances are you fit into one of these categories and, with the help of a doctor, can find the best oral minoxidil dosage for you.
Questions? Comments? Please reach out in the comments section.
References[+]
References ↑1, ↑3 Jimenez-Cauhe, Juan et al. Effectiveness and safety of low-dose oral minoxidil in male androgenetic alopecia. Journal of the American Academy of Dermatology, Volume 81, Issue 2, 648 – 649 ↑2 Pirmez, Rodrigo et al. Very-low-dose oral minoxidil in male androgenetic alopecia: A study with quantitative trichoscopic documentation. Journal of the American Academy of Dermatology, Volume 82, Issue 1, e21 – e22 ↑4 Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Journal of the American Academy of Dermatology, Volume 72, Issue 5, AB113 ↑5 English RS Jr, Ruiz S, DoAmaral P. Microneedling and Its Use in Hair Loss Disorders: A Systematic Review. Dermatol Ther (Heidelb). 2022 Jan;12(1):41-60. doi: 10.1007/s13555-021-00653-2. Epub 2021 Dec 1. PMID: 34854067; PMCID: PMC8776974. Vitamin B1 – also known as thiamin (or thiamine) – is part of the B-vitamin complex. Marketers claim that vitamin B1 can help support healthy hair growth, reduce hair shedding, and even prevent hair loss. Then again, marketers also make the same claims about B-vitamins like biotin, niacin, and vitamin B12. And typically, the claims are just plain wrong.
So, is vitamin B1 any different? In this article, we’ll dive into the evidence (and answers).
First, we’ll uncover why some people that vitamin B1 helps support hair growth. Then, we’ll dive into the evidence on the vitamin B1 / thiamin-hair loss connection. Finally, we’ll dive into evidence that might implicate vitamin B1 as an accelerator of hair loss… and steps to take if you suspect you’re deficient.
By the end, you’ll have a better idea of whether vitamin B1 is a worthy investment for your hair, or just another marketing gimmick. If you have any questions or comments, please post them below!
All-Natural Hair Supplement
The top natural ingredients for hair growth, all in one supplement.
Take the next step in your hair growth journey with a world-class natural supplement. Ingredients, doses, & concentrations built by science.

What is Vitamin B1?
Vitamin B1 (also thiamin or thiamine), is one of the many members of the B vitamin family.
Vitamin B1/thiamine
Researchers first identified this essential micronutrient through the study of beri beri, a serious disease of the nervous system that was common in South East Asia prior to the 1900s.
Unlike most diseases in that time, beri beri was much more common among wealthy citizens than it was among poorer citizens. Upon further investigation, researchers found the reason for the perplexing discrepancy was actually attributed to the differences in rice consumption.
While poorer individuals tended to consume brown rice, richer individuals tended to consume milled white rice — devoid of the husks, bran, and germ.
Through experimentation with this brown rice, a Polish biochemist, Casmir Funk, was able to isolate the compound that prevented beri beri. This compound? He termed it thiamine, meaning sulfur-containing amine — what we now know as vitamin B1.
Fast forward to today: we now know that vitamin B1 plays a crucial role in mitochondrial health, metabolism of macronutrients, and energy production — processes that are essential for the normal functioning of almost every cell in the body (1).
But, beyond the prevention of serious neurological conditions, does vitamin B1 have any benefit to our hair?
Let’s explore the evidence.
Vitamin B1: might it help support hair growth?
There’s no doubt that vitamin B1 is critical for important processes, like (1):
- The production of ATP, the energy molecule that fuels cellular activity
- The synthesis of amino acids
- The creation of NADPH, a co-factor necessary for steroid hormone production, fatty acid synthesis, and more.
And similar to other B-complex vitamins, vitamin B1’s role in these processes often form the basis of the claim that vitamin B1 can influence hair loss. After all, if you can’t produce amino acids — the raw material our hair is actually made of — how can you grow hair?
But, is this actually true? Are there any other ways that B1 might influence our hair? And what does this mean in the context of diet in the developed world?
Let’s explore the evidence.
Does a thiamine / vitamin B1 deficiency cause hair loss?
Maybe. But this isn’t the right question to ask. Rather, we need to ask this question in two parts:
- Does a vitamin B1 deficiency cause hair loss?
- Does a vitamin B1 deficiency within realistic parameters cause hair loss?
Why would we do this? Because at the extremes, almost anything causes hair loss. For instance, a “water deficiency” can cause hair loss. If we don’t drink water, we die. If we’re dead, we can’t grow hair. But that doesn’t mean that drinking water will regrow our hair. It also doesn’t mean we should warn people that hair loss is a side effect of a water deficiency.
The truth is that these types of logic leaps are what marketers use to claim that deficiencies in selenium, vitamin E, and iodine can all cause hair loss. Yes, this is true – but only if our scope of deficiency includes the endpoints: malnourished poverty-stricken children, people with genetic disorders who can’t absorb these nutrients, and people with certain chronic conditions that make nutrient assimilation nearly impossible.
All this is to say that we should ask if a thiamine deficiency, in the absolutes, causes hair loss. But the better question is: does a thiamine / vitamin B1 deficiency within realistic parameters cause hair loss, too?
Let’s take these one-by-one.
1. Does a thiamine deficiency, at the extremes, cause hair loss?
Maybe (in rodent models).
This 1968 study (2) sought to determine what happens in mice fed a diet that rapidly induces a thiamine deficiency. After two and a half weeks, some mice began to experience rapid weight loss, followed by abnormal hair shedding. Soon thereafter, neurological function began to decline. After four weeks, the mice were confused and could barely walk – symptoms similar to those seen in humans with beri beri.
However, it was unclear if the hair loss was caused by the vitamin B1 deficiency or the rapid weight loss.
2. Does a thiamine deficiency, within realistic parameters, cause hair loss?
Probably not.
For starters, people with beri beri rarely reported hair shedding (even despite their rapid weight loss). Moreover, when we expand our scope to human studies, we haven’t found any hard evidence that causally links a vitamin B1 deficiency to hair loss.
We could close the case right there, and say the article is done. At the same time, the absence of evidence doesn’t always imply evidence of absence.
For instance, there’s always the possibility that vitamin B1 might exacerbate certain chronic conditions linked to hair loss, or certain disease states associated with shedding disorders.
In fact, we could assert that since a vitamin B1 deficiency can lead to rapid neurological decline and thereby weight loss, and because rapid weight loss can trigger excessive (but temporary) hair shedding, then vitamin B1 deficiencies might be indirectly related to hair loss. The deficiency causes the weight loss; the weight loss causes the hair loss.
But again, if you’re so deficient in vitamin B1 that you start losing weight, you’ve got bigger things to worry about than your hair (like rapid impending neurological deterioration).
So, do we see any other circumstances where vitamin B1 is indirectly linked to hair shedding or hair loss?
Potentially. We can find them by look at the role of vitamin B1 in the body, and then comparing this to how different types of hair loss actually develop.
Vitamin B1 deficiency may exacerbate autoimmunity
At present time, there have been several animal studies conducted to investigate a link between vitamin B1 and autoimmunity. In general, these studies have suggested that vitamin B1 deficiencies may exacerbate autoimmunity in certain autoimmune disorders – specifically, multiple sclerosis (3, 4). It stands to reason that improving vitamin B1 deficiency may also improve these autoimmune conditions.
So, how could these effects translate to hair loss?
There are several forms of hair loss that seem to be mediated by autoimmune processes. These include alopecia areata as well as some forms of scarring alopecia.
If thiamine deficiencies happened to exacerbate the autoimmune processes involved in these hair loss disorders, it’s possible that restoring thiamine levels could improve these conditions.
Again, there’s no evidence that vitamin B1 deficiency is related to these hair-related autoimmune conditions. And while all autoimmune conditions involve autoimmune processes, not all autoimmune conditions develop in the same way. Thus, we can’t necessarily extrapolate the results from the animal studies, which primarily looked at multiple sclerosis, to the autoimmune conditions that lead to hair loss.
There’s also another point to consider: the effects of certain compounds reflected in animal studies are also traditionally very difficult to extrapolate to humans. Take our rodent study from earlier: thiamin-deficient rodents developed weight loss, neurological decline, and hair loss; whereas humans with beri beri – a sign of a thiamin deficiency – typically only develop weight loss and neurological decline.
That leaves us with one last piece of evidence to consider when it comes to linking autoimmune hair loss to vitamin B1: a case series on three human patients with autoimmune thyroid conditions (5).
Vitamin B1, autoimmune thyroid disorders, and hair shedding: a possible connection?
Autoimmune conditions that affect the thyroid – like Graves’ disease and Hashimoto’s thyroiditis – can have a domino effect on hair growth which is, in part, controlled by thyroid hormones. When these hormones get too high (i.e., Graves’ disease) or too low (i.e., Hashimoto’s thyroiditis), it can cause telogen effluvium – a form of diffuse hair shedding.
Interestingly, vitamin B1 might have relevance here, especially in the context of Hashimoto’s thyroiditis.
In one case series, doctors administered vitamin B1 to three patients with Hashimoto’s thyroiditis. They hypothesized that vitamin B1 could help relieve one of the hallmark symptoms of low thyroid hormone: fatigue.
Amazingly, that’s exactly what vitamin B1 did. In just a few hours to a few days, administration of B1 drastically improved the patients’ fatigue.
But, this wasn’t because of a subsequent improvement in the underlying autoimmune condition.
Instead, the authors hypothesized that the autoimmune processes involved in Hashimoto’s thyroiditis may have resulted in a vitamin B1 deficiency, subsequently leading to a reduction in energy production and, thus, fatigue.
In other words, the vitamin B1 deficiency likely isn’t a contributor to the development of Hashimoto’s thyroiditis. Instead, vitamin B1 deficiency, in this case, is a consequence of the condition.
As such, we can’t expect vitamin B1 to actually improve Hashimoto’s thyroiditis or the hair loss that occurs as a result. Instead, vitamin B1 can only improve symptoms related to a vitamin B1 deficiency that may occur alongside the condition.
So, we’ve established that vitamin B1 may or may not improve autoimmune forms of hair loss. We’ve also ruled out vitamin B1 as a means to improve Hashimoto’s thyroiditis and the hair shedding that ensues as a result.
But, are there any other pathways by which B1 might influence hair loss? Maybe… and that leads us to point number two.
Vitamin B1 deficiency may impair glutathione synthesis
Glutathione is a sulfur-containing compound with powerful antioxidant activity. Unlike antioxidants we consume in our diet (like polyphenols in green tea, berries, and other health-promoting foods), glutathione is manufactured by our own cells from amino acids like cysteine, glycine, and glutamic acid. This process requires NADPH, which requires vitamin B1 (along with various other B vitamins) (6).
So, how does this relate to hair loss?
Glutathione deficiency is associated with many conditions including diabetes, cardiovascular disease, as well as autoimmune diseases (7). Some studies also show low glutathione is associated with androgenic alopecia (AGA) (8).
As an anti-inflammatory agent, it’s possible that glutathione deficiency could exacerbate the microinflammation in AGA follicles — a process that drives the hair loss observed in AGA (7, 9).
In this context, it’s possible that through a possible increase in glutathione, vitamin B1 could reduce inflammation in AGA and, thus, improve AGA.
But there’s a difference between possible and plausible. Yes, if we stretch our imagination, it’s possible that a vitamin B1 deficiency might decrease glutathione production, and that if enough of this occurs in balding hair follicle sites, this decrease might exacerbate inflammation in AGA.
Possible, yes. But plausible?
Probably not.
While B1 deficiency seems to be related to low glutathione levels, vitamin B1 deficiency is extremely rare amongst most of the population. So, in this context, vitamin B1 is probably not something that most AGA patients need to worry about.
In these cases, glutathione deficiency is more likely to be related to co-morbidities seen in AGA: conditions like diabetes, obesity, and cardiovascular disease (all of which we know can deplete glutathione) (10).
So, while vitamin B1 deficiency could, technically, lead to or exacerbate a glutathione deficiency, it’s safe to say this is probably not the case for most AGA patients with low glutathione.
A quick recap
So far, we’ve established that B1 is an essential micronutrient. We’ve also established that in its complete absence, it can cause hair loss in rodents. Aside from that, it doesn’t appear that a vitamin B1 deficiency is a major driver of hair loss in humans.
The reason for this is three-fold:
- Vitamin B1 deficiency is extremely uncommon in the general hair loss population.
- In the rare cases where vitamin B1 levels are depleted and associated with hair loss, improving vitamin B1 levels only improves symptoms associated with vitamin B1 depletion. In other words, vitamin B1 improves nervous system abnormalities like fatigue but not the underlying conditions that lead to hair loss.
- When looking at the relationship between vitamin B1, glutathione, and AGA, it’s unlikely that vitamin B1 deficiency contributes to low glutathione levels that may exacerbate hair loss. Rather, low glutathione levels observed in some patients appear to be a consequence of some common co-morbidities associated with AGA.
So, that leads us to this conclusion: in the overwhelming majority of cases, vitamin B1 is not likely to confer any benefit in hair loss.
This leaves us with one last question worth asking before we close the books on the vitamin B1-hair health connection…
Could increasing vitamin B1 levels beyond normal levels have any negative effect on hair loss?
Maybe, maybe not. Let’s look at the research.
Why vitamin B1 might be bad for hair: the NADPH-DHT connection
Vitamin B1 is an essential cofactor in the production of the molecule, NADPH.
NADPH – or nicotinamide adenine dinucleotide phosphate – is a cofactor for enzymatic reactions. In other words, it’s a molecule that helps kickstart processes in the body. Earlier we established that NADPH was crucial for glutathione production. But that’s not all that NADPH does. NADPH is also essential for the production of steroid hormones.
Specifically, NADPH is a key molecule of the enzymatic process that converts testosterone into dihydrotestosterone, or DHT. Without NADPH, the enzyme that performs this conversion cannot function (11).
So, what does this mean for hair?
If you’ve done any research into androgenic alopecia (AGA), you probably already know that DHT is a significant contributor to the development of pattern hair loss (12). As such, any increase in DHT conversion could potentially worsen or speed up the balding process.
But, this would require NADPH to increase beyond what’s considered “physiological” — or what’s considered normal. So, does vitamin B1 do this?
Probably not. But we just don’t know.
What we do know is that, oftentimes, enzymes that produce molecules like NADPH have negative feedback mechanisms in place. This means that when their end-products increase, the body automatically reduces the activity of the enzymes that produce them. The net product is no increase in production.
However, this isn’t always the case. In some cases, these negative feedback loops are dysfunctional.
So, it’s possible that vitamin B1 doesn’t increase NADPH beyond what’s considered normal. As such, it’s also possible that vitamin B1 has no impact on DHT levels. At the same time, it’s also possible that it could.
In either case, the solution is the same: leverage diet to ensure vitamin B1 sufficiency and address a vitamin B1 deficiency if it’s present — but don’t go overboard.
If we’re deficient in B1, what should we do?
It’s important to note that an overwhelming majority of individuals likely aren’t deficient in vitamin B1. But, that doesn’t mean it’s impossible. Of the small pool of individuals B1 deficiency seems to affect in the modern world, risk factors appear to be:
- Hashimoto’s thyroiditis (5)
- Gastric bypass surgery (13)
- Anorexia (13)
- Severe crash dieting (13)
- Marked impairments in nutrient absorption, as in severe inflammatory bowel disease (13)
- Alcoholism (13)
- Septic shock (14)
But, again, it’s important to underscore that, even in these cases, we shouldn’t expect vitamin B1 to regrow our hair. This is because a vitamin B1 insufficiency is highly likely to present alongside other contributors to hair loss – like low thyroid hormone, zinc deficiency, iron deficiency, and severe calorie deficit. So, an improvement in B1 levels alone isn’t going to override these other factors. If anything, the thiamine deficiency-hair loss connection is more association than it is causation.
In any case, maintaining sufficient vitamin B1 levels is essential for overall health. So, you should aim to hit the recommended daily intake (RDI) everyday.
The good news is that you’re probably already doing this without even thinking about it – especially with all of the B-complex fortified foods out there. And if you find yourself falling into any of the risk categories, it’s not hard to find a supplement containing thiamine; nearly every multivitamin includes it as an ingredient.
At the same time, there could be a small minority of B1 deficiencies that go undetected, as was the case with the case series of Hashimoto’s thyroiditis patients. So, if you find yourself with fatigue that isn’t responding to standard treatment for Hashimoto’s thyroiditis, it may be worth discussing the possibility of vitamin B1 supplementation with your doctor.
Summary
Vitamin B1 is an essential vitamin. It ensures our body can effectively produce amino acids, glutathione, and other cofactors that are crucial for cellular function.
Deficiencies in vitamin B1 have been linked to weight loss, neurological decline, and hair loss in rodents. In humans, the evidence points more so toward neurological decline and weight loss than it does hair shedding. Having said that, a vitamin B1 deficiency might indirectly exacerbate hair loss through:
- Rapid weight loss
- Autoimmunity
- Glutathione impairment
But the bottom line is this: if your thiamine / vitamin B1 levels are low enough to associate with hair loss, you’ve got bigger problems than hair… like mental debilitation, neurological deterioration, and impending death.
Needless to say, 99.9% of us probably don’t need to be supplementing with vitamin B1 as a hair loss preventive. In fact, given vitamin B1’s relationship to NADPH production, and NADPH’s relationship to DHT, we could make a similar logic-leap argument that too much vitamin B1 may exacerbate hair loss.
This leaves us with one firm conclusion: maintain sufficient vitamin B1 levels by consuming the recommended daily intake. In the developed world, nearly every single diet will do this for you… provided you don’t have any of the hallmarks that increase your risk of a B1 deficiency (i.e., alcoholism, gastric bypass surgery, anorexia, etc.).
In the rare case that you are deficient, work with a doctor to address why your levels might be decreased in the first place and, if needed, to increase your levels with supplementation.
Have any questions about Vitamin B1 and hair health? Please leave them below in the comments!
References
- Davis, R. E., & Icke, G. C. (1983). Clinical chemistry of thiamin. Advances in clinical chemistry, 23, 93–140. https://doi.org/10.1016/s0065-2423(08)60399-6
- McCandless, D. W., Schenker, S., & Cook, M. (1968). Encephalopathy of thiamine deficieny: studies of intracerebral mechanisms. The Journal of clinical investigation, 47(10), 2268–2280. https://doi.org/10.1172/JCI105912
- Ji, Z., Fan, Z., Zhang, Y., Yu, R., Yang, H., Zhou, C., Luo, J., & Ke, Z. J. (2014). Thiamine deficiency promotes T cell infiltration in experimental autoimmune encephalomyelitis: the involvement of CCL2. Journal of immunology (Baltimore, Md. : 1950), 193(5), 2157–2167. https://doi.org/10.4049/jimmunol.1302702
- Natalie Nemazannikova, Kathleen Mikkelsen, Lily Stojanovska, Gregory L. Blatch and Vasso Apostolopoulos*, “Is there a Link between Vitamin B and Multiple Sclerosis?”, Medicinal Chemistry (2018) 14: 170. https://doi.org/10.2174/1573406413666170906123857
- Costantini, A., & Pala, M. I. (2014). Thiamine and Hashimoto's thyroiditis: a report of three cases. Journal of alternative and complementary medicine (New York, N.Y.), 20(3), 208–211. https://doi.org/10.1089/acm.2012.0612
- Martel JL, Kerndt CC, Franklin DS. Vitamin B1 (Thiamine) [Updated 2020 May 24]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482360/
- Ballatori, N., Krance, S. M., Notenboom, S., Shi, S., Tieu, K., & Hammond, C. L. (2009). Glutathione dysregulation and the etiology and progression of human diseases. Biological chemistry, 390(3), 191–214. https://doi.org/10.1515/BC.2009.033
- Prie, B. E., Iosif, L., Tivig, I., Stoian, I., & Giurcaneanu, C. (2016). Oxidative stress in androgenetic alopecia. Journal of medicine and life, 9(1), 79–83.
- Mahé, Y. F., Michelet, J. F., Billoni, N., Jarrousse, F., Buan, B., Commo, S., Saint-Léger, D., & Bernard, B. A. (2000). Androgenetic alopecia and microinflammation. International journal of dermatology, 39(8), 576–584. https://doi.org/10.1046/j.1365-4362.2000.00612.x
- Su, L. H., & Chen, T. H. (2010). Association of androgenetic alopecia with metabolic syndrome in men: a community-based survey. The British journal of dermatology, 163(2), 371–377. https://doi.org/10.1111/j.1365-2133.2010.09816.x
- Azzouni, F., Godoy, A., Li, Y., & Mohler, J. (2012). The 5 alpha-reductase isozyme family: a review of basic biology and their role in human diseases. Advances in urology, 2012, 530121. https://doi.org/10.1155/2012/530121
- Ustuner E. T. (2013). Cause of androgenic alopecia: crux of the matter. Plastic and reconstructive surgery. Global open, 1(7), e64. https://doi.org/10.1097/GOX.0000000000000005
- Wiley KD, Gupta M. Vitamin B1 Thiamine Deficiency. [Updated 2020 Jun 22]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537204/
- Moskowitz, A., & Donnino, M. W. (2020). Thiamine (vitamin B1) in septic shock: a targeted therapy. Journal of thoracic disease, 12(Suppl 1), S78–S83. https://doi.org/10.21037/jtd.2019.12.82
Subscription services have gained popularity in recent years for all types of products, from home cleaning supplies to underwear, razors, and even toothpaste. These services make the most sense for products used consistently, saving the consumer time and money. Hair regrowth treatments certainly meet the criteria. For the most part, they only work as long as you keep using them. So, it was only a matter of time before startups put men’s hair care on autopay.
Of the telemedicine providers focusing on men’s health, Hims, Keeps and Roman have risen to the top of this competitive market. But which one is best? We offer an objective side-by-side comparison of these 3 hair loss subscription companies. In the process, you’ll learn about the following:
- About the Products Sold
- What Differentiates the Providers
- Why Choose One Over The Other
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

About the Products
Hims, Keeps, and Roman all offer the same hair regrowth products, namely Finasteride and Minoxidil. These are the only two medications approved by the FDA to treat androgenic alopecia (AGA), or male pattern baldness.
Finasteride
Finasteride, also known by the brand name Propecia, is one of the most well studied, and most powerful, drug for AGA. The daily pill stops the progression of hair loss in 80-90% of men and, on average, leads to a 10% increase in hair count over two years.[1]https://www.sciencedirect.com/science/article/pii/S0022202X15529357
Oral finasteride is an FDA-approved treatment for AGA that inhibits the enzyme type II 5-alpha reductase, and thereby reduces dihydrotestosterone (DHT) levels in the body. While it’s effective in slowing, stopping, and partially reversing AGA’s progression, some men can experience side effects associated with the systemic reduction in DHT levels.
Topical Finasteride aims to avoid such side effects by localizing the drug’s effects to the scalp. Research on topical finasteride is still in its infancy, but the drug does appear to be effective for lowering DHT in the scalp. Real world results vary widely in part because method of delivery, dilution ratios and carrier agents (non-active ingredients) used to deliver the drug, also vary widely.
Minoxidil
Minoxidil, also known by the brand name Rogain, is the other FDA-approved drug for the treatment of AGA. Although available orally, it’s most popular as a topical. Over half of those who try topical minoxidil respond to the drug within 3-6 months. Of those, research reports hair count increases of up to 12% over 48 weeks.[2]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6691938/
Among the downsides of Minoxidil is that the treatment’s effectiveness does wane over time. When that happens, those who quit tend to lose any and all gains they had from the drug, ending up back where they started before ever having used it.
Having Trouble Separating Follicle Facts from Fiction?
One reason why Minoxidil’s effectiveness might taper is that it doesn’t address hair follicle miniaturization. So, while it does increase hair counts by kicking hair back into the growth cycle, hair continues to thin over time.
When combined with additional therapies, however, most if not all of these downsides can be mitigated. Minoxidil may work better when combined with scalp exfoliators such as retinol or retinoic acid. Research also finds minoxidil is up to four times more effective when combined with microneedling.[3]http://www.ijtrichology.com/article.asp?issn=0974-7753;year=2013;volume=5;issue=1;spage=6;epage=11;aulast=Dhurat
The effectiveness of Minoxidil may be best when used with Finasteride. These two treatment options can be combined.
- Minoxidil Response rate: 30-40% alone; 70+% if combined with microneedling and/or finasteride
- Minoxidil Regrowth rate: ~10% alone; 25-40% if combined with microneedling and/or finasteride
Both Finasteride and Minoxidil require ongoing, daily application for results maintenance. Hence, the appeal of subscription services. So let’s take a closer look at the three biggest players offering Finasteride and Minoxidil online.
Hims vs Keeps vs Roman
Hims, Keeps and Roman are telemedicine providers who, after a phone or video consultation, provide prescriptions for hair loss treatments. Products are shipped discreetly to the buyer’s home, with savings available for ongoing, subscription-based delivery.
Hims
Hims brands themselves as ‘all about personal wellness.’ The company offers products not only for hair loss, but also for men’s sexual health. They sell both prescription-based and over-the-counter solutions.
Roman
Roman is the men’s health wing of Ro.co, a consumer healthcare company with a mission to make healthcare accessible and convenient. The digital healthcare provider wants to make it easier for more men to seek preventative care. Of these 3 companies, they treat the widest range of men’s health conditions.
Keeps
Keeps focuses only on treating hair loss, versus general men’s health. Founded in 2018, the company aims to ‘help more men keep more hair.’ In addition to selling prescription treatments and hair care products, Keeps will link clients to hair restoration surgeons.
Each of the above companies sells generic Finasteride and Minoxidil, but the products differ slightly between sites. Below is a closer look at what’s on offer.
Subscription-Based Hair Loss Products
Hims Products
Of the 3 providers, Hims sells the greatest variety of products and hair treatment packages. The Hair Power Pack includes a combination of oral finasteride and 5% Minoxidil, plus Biotin gummies and a thickening shampoo.
Hims is also the only of the three companies to offer a topical Finasteride. The topical combines 0.3% finasteride with 6% Minoxidil. Active ingredients are diluted in alcohol (ethanol), propylene glycol and citric acid and delivered via spray bottle.
Propylene glycol as a carrier does lead to skin irritation in up to 6% of users, although this is considered a very mild side effect.[4]https://pubchem.ncbi.nlm.nih.gov/compound/Propylene-glycol
Keeps Products
Keeps does not offer a topical finasteride, but does include options for either foam or serum-based Minoxidil just as Hims does. They are also the only provider to sell a 2% ketoconazole shampoo. Alongside finasteride and minoxidil, ketoconazole is considered one of the “big three” treatment options for pattern hair loss.
Despite its popularity, however, ketoconazole is not actually FDA-approved for pattern hair loss, nor is it as well-researched as minoxidil or finasteride. However, the low-cost, low-effort treatment (used 2-3 times per week) seems to carry little (if any) risk of side effects.
Roman Products
Roman is the biggest generalist of the companies we’re comparing here. Hair loss is not necessarily their primary focus and they have the least number of hair-related products on offer. Roman offers oral finasteride, a Propecia generic made by Ascend in India, as well as a solution-based Minoxidil made by Pure Source, LLC. They do not offer a foam-based Minoxidil as Hims and Keeps do.
The Subscription & Cost
Topical minoxidil is an over-the-counter product and can be purchased directly without a telemedicine consult. Finasteride, whether oral or topical, and 2% ketoconazole do, however, require a prescription. How this process works differs slightly between Roman, Hims and Keeps.
The Hims Consultation Process
Hims begins with a free online consultation that includes a few personal questions, and offers quick access to a licensed medical provider, in all 50 states. Subscribed medications are then filled at a licensed pharmacy, and shipped to you directly.
Shipping info and billing frequency??
The Keeps Process
Keeps also begins with a free online consultation. After answering a few questions online, Keeps requires an upload of photos, which are then reviewed by a licensed provider. Treatments are shipped every 3, 6 or 12 months. Continued on-demand access to a live medical professional is available via their app for $10/month.
The Roman Process
Like the rest, Roman’s process begins with a free online visit. Depending on the state, a phone or video chat with a doctor or nurse practitioner may be required. Prescriptions are filled via the Ro Pharmacy Network, and shipped directly in discreet packaging. Roman offers free, unlimited follow-ups if needed. Prescriptions are available in monthly or quarterly auto-shipments.
Cost Comparison
Pros and Cons
Whether Roman, Hims, or Keeps is best is a personal decision that depends on the treatment sought, any extra over-the-counter shampoos, vitamins or other add-ons desired, and what state the patient lives in.
Hims Pros & Cons
Of the three companies in our comparison, Hims offers the most treatment options and is the only one to offer topical finasteride. Topical finasteride is also available from the following, lesser known companies:
- Happy Head: 0.25% Finasteride + 8% Minoxidil 8% + 0.01% Retinoic Acid + 1% Hydrocortisone. The formula is customizable and available for $79+/month
- Strut: Strut offers a customizable gel or solution-based topical with the following options, Finasteride 0.1-0.25%, Minoxidil 0.0%-7.5%, Tretinoin 0.0%-0.0125%, available starting at $59/month
On the other hand, Hims also ranks as the most expensive for either Finasteride or Minoxidil.
Keeps Pros & Cons
Keeps is the only provider on our list to offer Ketoconazole shampoo. So for those interested in trying the ‘Big 3’ protocol for hair loss, Keeps is a good bet. It’s also the least expensive provider of Finasteride and Minoxidil.
The main drawback of Keeps is that it’s not yet available in every state. They can, however, fill prescriptions in every state. So if you have a prescription from another doctor, Keeps may be able to fill it for a lower price.
Roman Pros & Cons
Of these 3 digital medicine providers, Roman is doing its best to position itself as a one-stop-shop for all things health-related. Contact with a licensed healthcare professional is available in every state, and you can go to Roman for nearly all things health-related.
The downside is that Roman is not focused on hair loss prevention nor hair regrowth alone, and they offer the most limited treatments options out of the 3 on our list.
Summary
Roman, Hims, and Keeps are all telemedicine providers who have risen to the top of the competitive men’s wellness market in part due to their focus on hair loss prevention and treatment.
Each company focuses primarily on oral finasteride and topical (OTC) minoxidil and will prescribe the former after a brief online consultation.
Subscription services and products vary. Which is best may depend on the state the patient lives in, and exactly which treatments are being sought.
References[+]
References ↑1 https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑2 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6691938/ ↑3 http://www.ijtrichology.com/article.asp?issn=0974-7753;year=2013;volume=5;issue=1;spage=6;epage=11;aulast=Dhurat ↑4 https://pubchem.ncbi.nlm.nih.gov/compound/Propylene-glycol If you’re experiencing hair loss, and you’re searching for a solution to help regrow hair, you’ve likely read several articles that reference different parts of the scalp. But what would you say if you were asked where on your scalp you’re losing hair? And how well do you understand the various parts of the scalp that lay beneath the skin, as well as their roles in hair loss?
In this article, we’ll give a quick introduction to the scalp. After that, we’ll take a tour of the scalp, starting with the different regions on the surface; then moving beneath the skin through the various layers of scalp tissue; then exploring the bones that lay beneath the tissue; then discussing the arteries, veins, nerves, and lymph vessels than run through those layers of tissue; and then examining the muscles that pull on the scalp. After touring the different areas of the scalp, we’ll discuss some ways that scalp mechanics may impact hair growth. Finally, we’ll look at some clinical evidence that has opened up the possibility of new treatments for androgenic alopecia.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Introduction to the Scalp
The scalp consists of layers of skin and subcutaneous tissue that cover the human cranium. It extends from the supraorbital foramina (i.e. two openings in the bone just above the eye sockets and just beneath the eyebrows) to the superior nuchal line (i.e. one of four curved ridges on the exterior surface of the occipital bone, which lies at the back of the head). It is bordered on the front by the face and on the sides and back by the neck.
The scalp acts as a physical barrier to protect the cranium against physical trauma and potential pathogens. It is also the area where human hair typically grows in order to 1) aid in heat conservation, and 2) play a role in aesthetic appearance and sexual signaling.
A Tour of Your Head: Regions of the Scalp
If someone were to ask you where on your scalp you’re losing hair, would you know how to answer them? If you’re unsure how to answer that question, check out the descriptions below.
The surface of your head has seven main areas, or regions, which are described below:
- Temples: The temples are located on either side of the head, behind the eye and between the forehead and ear. Each of the two temples is a latch where four skull bones – the frontal, parietal, temporal, and sphenoid bones – fuse together. Male pattern baldness typically starts at the temples, then continues to recede into the scalp, resulting in an M-shaped hairline.
- Frontal Region: If you were to draw an imaginary vertical line through your head, with the line positioned just in front of your ears, that line would demarcate the border between the frontal and mid-scalp regions. The part of the scalp that lies in front of that line is the frontal region. Also called the forelock, this region includes the hairline and the hair above the temples. If you’re losing hair due to male pattern baldness, this is often one of the first places you can expect to see hair loss.
- Mid-Scalp Region: If you were to draw two imaginary vertical lines through your head, with the first line positioned just in front of your ears and the second line positioned just behind them, the relatively flat area between those two lines would be the mid-scalp region. Unless you’re at an advanced stage of male pattern baldness, you probably won’t experience much hair loss in the mid-scalp region.
- Vertex Transition Zone: Also called the vertex transition point, this area is located between the mid-scalp region and the vertex. The vertex transition zone is the area where the scalp goes from a horizontal to a vertical plane posteriorly. For this reason, it is often referred to as the posterior hairline. Although this area is technically part of the vertex (see next bullet), it is often listed as a separate region.
- Vertex (Crown): Also called the crown, the vertex is the highest point on your scalp (the word “vertex” literally means “highest point”), which is located towards the back of your head. On non-balding heads, the vertex is typically identified by a swirl pattern of hair. In addition to losing hair around the temples and in the frontal region, many folks see noticeable hair loss on the crowns of their heads.
- Sides (Temporal Regions): Also known as the temporal regions (because they sit over the temporal bones and muscles), these are the areas on either side of the head that are positioned just above and behind the ears. Because the sides of the head have a different subcutaneous anatomy than the rest of the scalp, they are sometimes treated as separate from the scalp. However, the temporal regions generally grow hair, so we’ve chosen to include them in this tour of the scalp. Hair loss is much less common in this region.
- Back (Occipital Region): Also known as the occipital region (because it sits over the occipital bone and muscle), this area covers the back of the head, underneath the crown. As with the sides of the head, hair loss is much less common in this region.
Digging into the Anatomy: Layers of the Scalp
While the previous section provides a good overview of the various areas on the surface of the scalp, there is a lot more going on beneath the surface.
The scalp consists of the following five layers:Tajran J, Gosman AA. Anatomy, Head and Neck, Scalp. [Updated 2021 Jul 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-.
- Skin: The first layer of the scalp is the skin, which acts as a protective barrier to the rest of the scalp, helps prevent the loss of moisture, and helps regulate body temperature, among other things. This layer is thick and contains numerous hair follicles and sebaceous glands (i.e. small, oil-producing glands).
- Dense Connective Tissue: The second layer is the dense connective tissue layer, which connects the skin to the galea aponeurotica (described below). In addition to connective tissue, this layer contains arteries, veins, nerves, and lymphatic vessels. Hair follicles can also extend from the skin down into this layer.
- Galea Aponeurotica: Also known as the epicranial aponeurosis, the galea aponeurotica is a layer of dense, fibrous connective tissue that runs along the top of the cranium, connecting to the frontalis muscle at the front of the head and the occipitalis muscle at the back of the head.
The first three layers of the scalp are firmly attached to each other and move as a unified structure.
- Loose Areolar Connective Tissue: This layer forms a flexible connection to both the galea aponeurotica and the pericranium (described below), allowing the top three layers of the scalp to slide over the lower pericranium. The loose areolar connective tissue also contains numerous blood vessels.
- Pericranium: Composed of dense connective tissue, the pericranium is the deepest layer of the scalp, which tightly adheres to the calvaria (i.e. the bones that form the top of the skull). It also contains the vascular supply that’s essential to supporting the calvaria.
The Scalp’s Foundation: The Calvaria and Other Cranial Bones
As noted above, the pericranium is firmly attached to the calvaria bones, which form the top of the human skull. In addition to the calvaria, there are some other cranial bones that play a role in scalp function. We discuss these bones below.
Minonzio, Claudio. (2019). A step towards aberration corrections for transcranial ultrasound – Estimation of skull thickness and speed of sound.
The neurocranium, which is pictured in the image above, consists of the upper part of the skull that surrounds the cranial cavity and contains the brain. There are eight bones that make up the neurocranium:
- The frontal bone forms a skeletal roof above the eye sockets and nasal cavity, and forms the main part of the forehead.
- The left and right parietal bones sit behind the frontal bone. These bones form the top and sides of the cranium.
- The occipital bone forms the back and base of the skull. It has a large opening, called the foramen magnum, where the spinal cord passes into the skull.
- The left and right temporal bones help form the sides and base of the skull, where they protect the temporal lobe of the brain and surround the ear canal.
- The sphenoid bone is situated in the middle of the skull towards the front. It helps form the base and sides of the skull, and has a shape that resembles a butterfly.
- The ethmoid bone is a cube-shaped bone located in the center of the skull between the eyes. It helps form the walls of the eye sockets, as well as the roof, sides, and interior of the nasal cavity.
Although these are eight individual bones, they are joined together by various fibrous sutures.
The neurocranium can be further divided into an upper and lower part:
- The upper part, known as the calvaria, consists of the flat bones of the skull that form a dome-like roof. This includes the frontal bone, parietal bones, and occipital bone.
- The lower part, known as the cranial base, includes the lower parts of the frontal bone, the sphenoid bone, ethmoid bone, temporal bones, and the lower parts of the occipital bone.
Because the calvaria makes up the top part of the skull, these bones are more significant when discussing hair growth/loss. However, the muscles that attach to the temporal and sphenoid bones may also play a role in hair loss (more on that in a bit).
The Vertical Layer: Arteries, Veins, Nerves, and Lymphatic Vessels
We previously discussed the five layers of the scalp. There are also a number of arteries, veins, nerves, and lymphatic vessels that run through those layers. We discuss each of these below.
Arteries of the Scalp
The common carotid arteries, a pair of arteries on either side of the neck, provide the main supply of blood to the scalp. While still in the neck, each of the common carotid arteries splits into an internal and external carotid artery. Each of these branches supplies blood to different areas of the scalp.
Internal Carotid Artery
Wikipedia contributors. Ophthalmic artery. Wikipedia, The Free Encyclopedia. January 5, 2022, 11:55 UTC.
The internal carotid artery (on either side of the skull) gives rise to an ophthalmic artery, which further branches into a supratrochlear artery and a supraorbital artery. Both the supratrochlear and supraorbital arteries rise through the supraorbital foramen (i.e. an opening in the bone just above the eye socket and just beneath the eyebrow) and connect with their counterparts on the opposite side of the skull, as well as with the superficial temporal artery (also on either side of the skull), which provides most of the blood supply to the front of the scalp.
External Carotid Artery
Hacking, C., Bell, D. Scalp. Reference article, Radiopaedia.org. (accessed on 12 May 2022)
The external carotid artery (on either side of the skull) branches into the following arteries:
- The superficial temporal artery passes over the back of the cheekbone, where it splits again into a frontal and parietal branch. The frontal branch runs across the temple, supplying blood to the frontal and temporal regions of the scalp. The parietal branch continues over the top of the skull, supplying blood to the parietal region of the scalp.
- The posterior auricular artery splits away from the external carotid artery just above/behind the jaw and just below the ear. The posterior auricular artery then passes underneath the ear and travels to the deep tissues that converge between the mastoid process (i.e. the protruding, cone-shaped bone found just below and behind the ears on either side of the head) and the cartilage of the ear. This artery supplies blood to the parts of the scalp located above and behind the ears.
- The occipital artery splits from the external carotid artery at the back of the neck, passing under the ear and in front of the mastoid process. This artery supplies blood to the back of the scalp.
Veins of the Scalp
The venous drainage of the scalp can be split into superficial and deep components.
Superficial Veins
The superficial veins follow their respective arteries. The supraorbital and supratrochlear veins (on either side of the skull) drain blood from the top of the scalp through the front of the face, while the superficial temporal, occipital, and posterior auricular veins (also on either side of the skull) drain blood from the scalp through the back of the head. Also, just like its arterial counterparts, the superficial temporal vein has a frontal and parietal branch.Tajran J, Gosman AA. Anatomy, Head and Neck, Scalp. 2021 Jul 26. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan–. PMID: 31855392.
Deep Veins
The deeper regions of the scalp drain into the pterygoid venous plexus (on either side of the skull), which is a large plexus (i.e. a bundle of intersecting blood vessels, nerves, or lymphatic vessels) of veins situated between the temporalis (i.e. thin, fan-shaped muscles on either side of the skull) and lateral pterygoid (i.e. one of the muscles in the jaw that helps us chew) muscles. From there, the venous blood passes into the maxillary vein (on either side of the skull), then into the retromandibular vein (on either side of the skull), and then into the external jugular vein (on either side of the skull), which is the venous counterpart to the external carotid artery.Rivard AB, Kortz MW, Burns B. Anatomy, Head and Neck, Internal Jugular Vein. 2021 Jul 26. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan–. PMID: 30020630.
The Connection Between Superficial and Deep Veins
It’s also worth noting that there is a drainage connection between the superficial and deep venous systems, whereby parietal emissary veins located in the loose areolar connective tissue layer of the scalp connect to diploic veins that penetrate the skull, eventually flowing into the dural venous sinuses (e.g. superior sagittal sinus) that drain venous blood from inside the cranial cavity.Germann AM, Jamal Z, Al Khalili Y. Anatomy, Head and Neck, Scalp Veins. 2021 Dec 15. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan–. PMID: 31082005.
Nerves of the Scalp
The front and sides of the scalp are innervated by trigeminal nerves, while the back of the scalp is innervated by cervical nerves.
Trigeminal Nerves
Wikipedia contributors. Trigeminal nerve. Wikipedia, The Free Encyclopedia. March 31, 2022, 01:23 UTC.
There are two trigeminal nerves – one on each side of the skull. Each trigeminal nerve has three major branches:
- The ophthalmic nerve branches into the frontal nerve, which eventually splits into the following two branches:
- The supratrochlear nerve innervates the front, middle section of the forehead.
- The supraorbital nerve innervates a large portion of the scalp including the top and sides of the forehead, as well as the vertex.
- The maxillary nerve branches into the zygomaticotemporal nerve, which innervates the temple.
- The mandibular nerve branches into the auriculotemporal nerve, which innervates the skin in front of and above the ear.
Cervical Nerves
Cervical nerves emerge from the spinal cord to innervate the rest of the body. There are four cervical nerves that innervate the scalp:Roesch ZK, Tadi P. Anatomy, Head and Neck, Neck. 2021 Jul 26. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan–. PMID: 31194453.
- The lesser occipital nerve (on either side of the skull) arises from the anterior ramus (i.e. front-facing branch) of the second cervical nerve and innervates the skin above and behind the ear.
- The greater occipital nerve (on either side of the skull) arises from the posterior ramus (i.e. rear-facing branch) of the second cervical nerve and innervates the skin at the upper back of the scalp.
- The great auricular nerve (on either side of the skull) arises from the anterior rami (i.e. front-facing branches) of the second and third cervical nerves and innervates the skin below and behind the ear.
- The occipital nerve (on either side of the skull) arises from the posterior ramus (i.e. rear-facing branch) of the third cervical nerve and innervates the skin at the lower back of the scalp.
The Scalp’s Lymphatic Vessels
The lymphatic vessels of the scalp are located in the dense connective tissue layer and follow the venous drainage. In general, the front portions of the scalp drain lymphatic fluid through the parotid nodes (i.e. lymph nodes found in front of and just below each ear), which continue to drain through the submandibular lymph nodes (i.e. lymph nodes located just beneath the jaw bone) and deep cervical lymph nodes (i.e. lymph nodes located in the neck).Rivard AB, Kortz MW, Burns B. Anatomy, Head and Neck, Internal Jugular Vein. 2021 Jul 26. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan–.
The rear portions of the scalp drain lymphatic fluid through the posterior auricular lymph nodes (i.e. a small group of lymph nodes located just beneath the ear) and occipital lymph nodes (i.e. lymph nodes located at the back of the head, near the occipital bone). The posterior auricular lymph nodes drain the area of the scalp located directly behind the ear and flow into the occipital lymph nodes, while the occipital lymph nodes drain lymphatic fluid from the remaining regions in the back of the scalp.
Muscles that Act on the Scalp
There are a handful of muscles that act on the scalp.
The largest and most important muscle in the scalp is the occipitofrontalis muscle. The occipitofrontalis muscle is actually two sections of muscle, known as bellies, that connect to the front and back of the galea aponeurotica. The frontalis belly connects to the galea aponeurotica at the front of the head and inserts into the superior orbicularis oculi (i.e. the muscle that closes the eyelids) near the eyebrow. The occipitalis belly connects to the galea aponeurotica at the back of the head and attaches to the superior nuchal line (i.e. one of four curved ridges on the exterior surface of the occipital bone) and mastoid processes (i.e. the protruding, cone-shaped bones found just below and behind the ears on either side of the head) at the bottom of the scalp. These muscles, or bellies, work in conjunction to pull the scalp back and elevate the eyebrows (the contraction of the frontalis belly creates wrinkles on the forehead).Roesch ZK, Tadi P. Anatomy, Head and Neck, Neck. 2021 Jul 26. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan–. PMID: 31194453.
The auricular muscles are a group of muscles of the auricle (i.e. the visible portion of the ear). Although there are nine total auricular muscles on either side of the skull, six of those muscles are intrinsic muscles located deep within the skull, while the other three are extrinsic muscles that are located more superficially. It is the three extrinsic muscles that we’re concerned with for this article.
- The auricularis superior, the largest of the three extrinsic auricular muscles, is a thin, fan-shaped muscle located above the ear. It helps to elevate the ear, attaching to the galea aponeurotica at the top and a tendon just above the ear at the bottom.
- The auricularis anterior, the smallest of the three extrinsic auricular muscles, is a thin, fan-shaped muscle located in front of the ear. It helps draw the ear upward and forward, attaching to the lateral edge of the galea aponeurotica at one end and cartilage at the top, front of the ear at the other.
- The auricularis posterior is a muscle located behind the ear. It helps pull the ear backward, attaching to the mastoid process (i.e. the protruding, cone-shaped bone found just below and behind the ears on either side of the head) at one end and the lower part of the cranial surface at the back of the ear at the other end.
Although the auricular muscles play a minor role in fixing the position of the ear, they are considered vestigial in humans. Nevertheless, these muscles may play a role in restricting blood flow to the scalp (more on this in a bit).
The temporoparietalis muscles are a pair of muscles located just above and in front of the auricularis superior muscles on either side of the head. These muscles lie over both the temporal and occipital bones, connecting to the fascia (i.e. a thin sheath of fibrous tissue enclosing a muscle or other organ) just above the ear on one end and the galea aponeurotica on the other. The temporoparietalis muscles help to fix the galea aponeurotic and elevate the ears.
The temporalis muscles are broad, fan-shaped muscles located on either side of the head. These muscles cover most of the temporal bones and help produce movements of the mandible, or jawbone, when chewing. Although these muscles are more associated with the mandible than they are the scalp, the contraction of these muscles may restrict blood flow to the scalp, which could have an effect on hair growth/loss (more on this below).
Ways Scalp Mechanics May Impact Hair Growth
Now that you have a better understanding of scalp anatomy, we can examine ways that scalp mechanics may impact hair growth. In particular, the galea aponeurotica and the surrounding musculature may play a role in restricting blood flow (and oxygen) to the scalp.
We know from previous studies that balding scalps tend to have 2.6 x less subcutaneous blood supply compared to non-badling controls. Moreover, several blood-pressure-lowering medications – such as minoxidil, diaoxide, and pinacidil – have been shown to improve androgenic alopecia – one of the world’s most common hair loss disorders. These findings implicate reduced blood supply as a potential contributor to pattern hair loss. And while it’s still debated just how much the reductions to blood flow are a cause or consequence of androgenic alopecia, there may be a therapeutic benefit to improving blood, oxygen, and nutrient levels to balding hair follicles.
Below, we examine the impact of scalp musculature on arterial branches, as well as the impact of the galea aponeurotica and surrounding musculature on microcirculation (i.e. capillaries in the scalp).
The Impact of Musculature on Arterial Blood Flow
Dermatologists report that roughly 80% of men with androgenic alopecia tend to have “tight” scalps, suggesting that the muscles around the perimeter of the scalp have involuntarily contracted and are pinching the arterial branches, causing a reduction in blood supply. This is also supported by research measuring scalp hardness in balding versus non-balding men across a variety of scalp regions.[1]https://www.researchgate.net/publication/338624064_Androgenetic_alopecia_is_associated_with_increased_scalp_hardness
So, do the muscles surrounding the scalp perimeter and anchored to the galea aponeurotica have anything to do with these phenomena? Some evidence suggests yes.
As noted previously, the supraorbital and supratrochlear arteries are two of the three arterial branches that supply the majority of blood to the frontal region of the scalp. These arteries pass through the cranial bone at the eyebrows, run underneath the frontalis muscle, then pierce through the frontalis muscle before running upward toward the hair line. When the frontalis muscle flexes, this flexing can compress the supraorbital and supratrochlear arteries, restricting blood flow to the scalp.[2]https://academic.oup.com/asj/article-abstract/41/11/NP1599/6206455
In addition, the deep temporal artery resides between the cranium and the temporalis muscles. When these muscles contract, they can compress the deep temporal artery against the skull and constrict blood supply.
Moreover, some branches of the auricularis anterior and posterior arteries weave between the auricular muscles and their supporting tendons. It is possible that the contraction of the auricular muscles may restrict blood flow in the same manner.
Finally, while many of the scalp’s arteries reside in the fascia that overlies the scalp muscles, the contraction of the muscles underlying these arterial branches can still affect them. We know this because studies have shown that when the temporalis muscle contracts, the artery overlying it (the superficial temporal artery) constricts by 50%, thus leading to a ~75% decrease in blood supply in that arterial branch.[3]https://n.neurology.org/content/11/11/935
But that’s not the whole story.
The Impact of the Galea Aponeurotica on Microcirculation
Despite the possible restriction of arterial blood flow described in the previous section, it is generally believed that reductions to blood flow in androgenic alopecia are mainly due to the loss/restriction of the microcapillary networks (i.e., the small blood vessels supporting the hair follicles themselves), rather than reductions from the carotid arterial branches supporting those microcapillary networks.
The microcapillary networks may constrict or compress as a result of mechanical stretch across the galea aponeurotica (mediated by the contraction of the scalp’s perimeter muscles). Because the top of the scalp is a fixed area, stretching of the galea aponeurotica would generate compression of the underlying microcapillary networks supplying hair follicles.
How Relevant Is Blood Flow to Androgenic Alopecia?
We see hypoxia (i.e. insufficient supply of oxygen) as a trigger of hair loss in mouse models, but is it relevant to humans with androgenic alopecia? Unfortunately, we still don’t know (since there are also reductions to blood supply resulting from the hair follicle miniaturization that follows each re-entry into the anagen stage of the hair cycle).
Interestingly, there used to be a surgery called “scalp reduction,” where surgeons would place a balloon underneath the galea aponeurotica, slowly inflate it over a series of weeks, and then give patients a surgery to “remove” bald regions and pull the hair-bearing stretched skin over to create the appearance of a fuller head of hair. In the 1990s, these surgeries were quietly abandoned after surgeons started voicing concerns of accelerated hair loss in patients following the procedure. Was this accelerated hair loss caused by skin tension? Did this tension lead to microcapillary compression? We still don’t know the answers, but the questions certainly are interesting.
Clinical Evidence Suggests Possible New Hair Loss Treatment Targets
Despite the lack of certainty regarding the impact of blood flow (and oxygen) on androgenic alopecia, the results from a number of Botox studies show that when the muscles surrounding the scalp are forcibly relaxed in patients with androgenic alopecia, hair growth/loss improves.
Botulinum toxin, which is commonly referred to by the brand name Botox, is an injectable neuro modifier that’s used as a therapeutic treatment for many clinical and cosmetic concerns. Specifically, Botox is used to help relax muscles and reduce certain inflammatory signaling proteins.
Over the last decade, there have been five clinical studies published on the hair-promoting effects of Botox on men with androgenic alopecia. Four of those studies tested intramuscular Botox injections (i.e. injections directly into the scalp’s perimeter muscles), while the fifth study tested intradermal Botox injections (i.e. injections directly into balding regions of the scalp). We examine the results from both types of studies below.
The Results of Studies Testing Intramuscular Botox Injections
Across the four studies testing intramuscular Botox injections, 75-80% of participants responded favorably (i.e. hair growth) with an average hair count increase of 18-21% after 6 to 10 months. Injections into the scalp perimeter muscles were done once every 4-6 months, with results becoming cosmetically significant after the second round of injections.
In addition to testing the use of Botox injections on their own, one of the four studies also examined using Botox injections alongside oral finasteride (i.e. the active ingredient in Propecia). This combined therapeutic approach led to improved response rates and hair count increases that average nearly 35%.
Researchers suspect that two mechanisms contributed to these results:
- First, intramuscular Botox injections might relax involuntarily contracted muscles around the perimeter of the scalp. As noted previously, arterial branches run through those perimeter muscles in order to supply the scalp with blood. When perimeter muscles are contracted, they may pinch those arteries, restricting the supply of blood to the scalp. Researchers theorize that injecting those perimeter muscles with Botox helps to unclamp the pinched arterial branches, increasing the supply of blood (and oxygen).
- Second, by relaxing those perimeter muscles, researchers also speculate that Botox injections might help reduce tension across the top of the scalp, and in doing so, potentially reduce tension-mediated inflammation and/or lower dihydrotestosterone (DHT) in the scalp.
Despite researcher’s suspicions, it’s important to note that these suggested mechanisms are only speculative at this point. We will need more data in order to corroborate these suspicions.
The Results of the Study Testing Intradermal Botox Injections
In the one study that tested intradermal Botox injections, 60-80% of participants responded favorably (i.e. hair growth) with an average hair count increase of 5% after 6 months. Injections into balding regions were done every four weeks.
Although this study’s response rate is similar to the intramuscular studies above, the hair count increases were much lower. It’s worth noting that these results might improve with higher-dose injections. This study used just 30 units of Botox, spread across the entire scalp. When another investigator increased the amount of Botox injected from 30 units to 100 units, and also doubled the frequency of injections, they observed more significant hair growth.[4]https://www.dovepress.com/getfile.php?fileID=73853
While conducting the study of intradermal Botox injections, researchers also conducted a cell culture on human hair follicles. They found that Botox appears to decrease the expression of transforming growth factor beta 1 (TGFB-1), a signaling protein that acts as a negative regulator of the hair cycle. TGFB-1 also appears to be intimately tied to hair follicle miniaturization, causing researchers to speculate that intradermal Botox injections might be down regulating TGFB-1 in human hair follicle sites, and in doing so, allowing for hair loss improvements.
It’s important to note that the reduction of TGFB-1 happens in a dose- and time-dependent manner. Given this fact, it makes sense that increasing both the volume and frequency of intradermal injections evokes a more robust improvement in hair count.
As with the intramuscular studies above, it’s important to note that the suspected mechanism (i.e. the down regulating of TGFB-1) identified in the intradermal study is only speculative. In order to corroborate this suspicion, more data will need to be collected.
How Reliable Are the Findings from the Botox Studies?
Although the results from the Botox studies look promising, the amount of evidence on Botox as a treatment for androgenic alopecia is still rather small. And while several research groups have found consistent results, these studies tend to score lower on the hierarchy of evidence. So, we should interpret those results with caution.
There are at least three significant issues with the Botox studies:
- The Botox Studies Had a Small Number of Participants: To date, fewer than 200 participants have tested Botox to treat androgenic alopecia (in a clinical, peer-reviewed setting). For comparison, thousands of participants have tested the use of finasteride for treating androgenic alopecia.
- None of the Botox Studies Used a Control Group: At this point, no Botox studies have used control groups (i.e. patients who receive a placebo treatment). Using placebo groups is important because they help researchers control for hair count improvements (and losses) due to seasonal hair shedding.
- None of the Botox Studies Assessed Hair Diameter: While the Botox studies assessed hair count, they did not examine changes to hair diameter. Androgenic alopecia progresses through the process of hair follicle miniaturization, and we can effectively stop androgenic alopecia by reversing this process. Because we don’t know whether Botox has an effect on hair follicle miniaturization, we also don’t know whether Botox is an effective long-term solution for androgenic alopecia.
In addition to the above three issues, it’s also worth noting that 1) there are currently no established best practices for the use of Botox to treat androgenic alopecia, and 2) Botox treatment can be rather expensive, with intramuscular injections costing $2,400 to $4,000 per year and intradermal injections running $4,800 to $12,000 per year.
This isn’t to say we should dismiss the evidence in favor of using Botox to treat androgenic alopecia. At the same time, it’s important not to jump to any conclusions regarding the efficacy of using Botox versus other treatment options.
Final Thoughts
In this article, we’ve shown how the scalp has a rather complex anatomy – including seven regions on the surface of the scalp; five tissue layers; several bones that the scalp attaches to; a number of arteries, veins, nerves, and lymphatic vessels running through the scalp; and a collection of muscles that pull on the scalp. We’ve also discussed ways that scalp mechanics may impact hair growth, and reviewed five studies that suggest Botox injections may improve hair loss by relaxing the muscles around the perimeter of the scalp.
Although the findings from Botox studies are still preliminary, they have opened up the possibility of new treatment targets for androgenic alopecia. Any relevant treatment targets would need to relax the muscles around the scalp so that 1) arterial branches can deliver an adequate supply of blood (and oxygen) to the scalp, and 2) tension in the galea aponeurotica doesn’t restrict blood flow in the microcapillary networks that feed hair follicles.
In addition to the results from the Botox studies, there’s mechanistic research to suggest that skin tension in balding regions (generated from the contraction of muscles around the scalp) might have something to do with the arrival of dihydrotestosterone (DHT) and the balding process itself.[5]https://www.sciencedirect.com/science/article/pii/S0306987717310411
Finally, it’s worth noting that Botox injections are not the only way to relax the muscles around the scalp. Scalp massages may offer a viable, less-expensive method for treating androgenic alopecia that targets the same mechanisms as Botox injections.
References[+]
References ↑1 https://www.researchgate.net/publication/338624064_Androgenetic_alopecia_is_associated_with_increased_scalp_hardness ↑2 https://academic.oup.com/asj/article-abstract/41/11/NP1599/6206455 ↑3 https://n.neurology.org/content/11/11/935 ↑4 https://www.dovepress.com/getfile.php?fileID=73853 ↑5 https://www.sciencedirect.com/science/article/pii/S0306987717310411 Recent years have witnessed a surge of interest in topical dutasteride for hair loss, especially among those who have either not tolerated or seen insufficient results with conventional oral therapies. This growing popularity is fueled by multiple online anecdotes and user testimonials that describe marked improvements in hair density, shedding reduction, and scalp health.
However, these topical dutasteride before and after photos typically show the very extremes of success or failure, which can give a misleading impression of what average outcomes truly are. This tends to create an environment where typical results are overshadowed by outliers, making it harder to gauge what most people should realistically expect.
Where does topical dutasteride truly fit in the landscape of hair loss treatment? How can one reliably distinguish between what’s achievable and what’s most likely? What’s the best way to interpret anecdotal stories versus the outcomes most users may expect? And, crucially, what kind of regrowth pattern could unfold after years on topical therapy?
This article tackles these questions head-on. Clear expectations will be set for topical dutasteride use, including practical guidance on interpreting online results, examples of standout before-and-after transformations, and insights into what the average treatment journey actually looks like.
We will also introduce a framework for evaluating topical therapies, helping readers compare dutasteride’s regrowth potential, evidence strength, and long-term safety against other hair loss options.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What is Topical Dutasteride?
Topical dutasteride is a compounded medication for androgenic alopecia (AGA) that works as a dual 5-alpha reductase inhibitor, reducing dihydrotestosterone (DHT) locally at the scalp. The main distinction from oral dutasteride is that topical delivery is intended to minimize systemic absorption, though some absorption into the bloodstream still occurs, and complete elimination of systemic exposure is not guaranteed. DHT suppression with topical forms is less studied compared to oral; oral dutasteride can lower systemic DHT by 90% or more, but topical formulations do not consistently reach those levels in serum.
How Topical Dutasteride Works
Dutasteride inhibits both type 1 and type II 5-alpha reductase, the enzymes responsible for converting testosterone to DHT.[1]Frye, S.V. (2006). Discovery and clinical development of dutasteride, a potent dual 5alpha-reductase inhibitor. Current topics in medicinal chemistry. 6(5). 405-421. Available at: … Continue reading By inhibiting these enzymes, topical dutasteride targets DHT production where hair loss is occurring at the scalp, rather than throughout the body.
Studies show that topical application leads to significantly lower systemic absorption compared with oral forms, which should result in fewer side effects. Absorption data from Franz cell and skin retention studies indicate that only a small fraction of the topically applied dose reaches the circulation.[2]Noor, N.M., Sheikh, K., Somavarapu, S., Taylor, K.M.G. (2017). Preparation and characterization of dutasteride-loaded nanostructured lipid carriers coated with stearic acid-chitosan oligomer for … Continue reading
DHT reduction with topical dutasteride is robust in scalp tissue but generally milder in serum compared to oral therapy.
Common compounded strengths vary widely: for example, Ulo offers concentrations ranging from 0.02% to 0.2%.
No topical dutasteride product is FDA-approved; all are compounded by pharmacies for individual patients based on prescriptions, with dosing customized to tolerance and response.
We could find three studies that report significant local efficacy:
Study 1: Nada et al., 2018 (Prospective Randomized; 30 Men; 24 Weeks)
- Design/groups: Compared minoxidil plus topical dutasteride (0.02%) versus minoxidil alone.[3]Nada, E.A., El-Dawla, R.E., El Maged, W.M.A., Emagd, A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). Available at: … Continue reading
- Outcomes: The dutasteride group showed a significant increase in hair density, hair shaft width, and terminal to vellus hair ratio relative to minoxidil-only controls. Patient-reported satisfaction was higher for the topical dutasteride cohort.
- Systemic/Serum: A reduction in serum DHT was observed in the topical dutasteride group, confirming some degree of systemic impact but still substantially less than typical oral dosing.
Study 2: Obeid et al, 2024 (Double-Blind RCT; 24 men, 6 women; 16 Weeks)
- Design/groups: Compared minoxidil plus topical dutasteride (0.01%) to minoxidil alone.[4]Obeid, M.N.A., Fattah, N.S.A., Elfangary, M.M., Husseni, R.M.A. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 … Continue reading
- Outcomes: The combination group achieved superior overall improvements in hair thickness and density compared to minoxidil monotherapy. Patient satisfaction and photographic evidence suggested enhanced regrowth when dutasteride was added.
- Systemic/Serum: Systemic DHT or hormone changes were not evaluated or reported in this study.
Study 3: Panuganti et al., 2025 (Phase II Double-Blind; 135 Men; 24 Weeks)
- Design/Groups: Compared three concentrations of topical dutasteride (0.01%, 0.02%, 0.05% w/v), oral finasteride (1 mg), and placebo.[5]Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., KSSVV, S.R., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate … Continue reading
- Outcomes: Dose-dependent improvements in target area hair count (TAHC) were observed. Dutasteride 0.05% was superior at 24 weeks to both topical finasteride and placebo. Mean TAHC increase: 0.015 (12.78), 0.02% (20.03), 0.05% (34.30) vs oral finasteride (12.57). More patients reached global photographic assessment thresholds in the 0.05% group.
- Systemic/Serum: Topical dutasteride produced only modest changes in serum testosterone and DHT compared to moderate suppression seen with oral finasteride. The highest topical strength exhibited a better pharmacokinetic profile (lower systemic absorption) than oral finasteride.
These data provide evidence that topical dutasteride can offer clinically relevant improvement in hair quantity and quality, while keeping systemic risk modest and tolerability high in well-compounded, patient-tailored regimens.
Why Expectation Setting Matters
Setting realistic expectations is important for anyone considering topical dutasteride or other emerging therapies for AGA. Online testimonials and dramatic before-and-after photos can mislead, often emphasizing rare “hyper responders” or downplaying variables like lighting and styling.
In truth, most users experience gradual, modest improvements, not dramatic transformations, and disappointment is common when hopes are too high. Conversely, setting expectations too low may discourage trials of potentially worthwhile treatments that could deliver meaningful benefits.
For better outcomes with topical dutasteride:
- Expect gradual change rather than remarkable regrowth.
- Rely on professional advice and published clinical data, not social media images for guidance.
Remember: staying grounded in evidence and expert recommendations will help identify therapies truly worth pursuing on the hair regrowth journey.
Principle #1: What’s Possible ≠ What’s Probable
When looking at treatment outcomes, it’s essential to distinguish between what can happen and what usually happens. Look at the scatterplot below: individual data points are scattered widely, showing what’s possible for one person, while the trendline running through the middle represents the average, or what’s probable.
Figure 1: A graph showing possible and probable results. The data points show possible results, and the trendline shows probable results.[6]Badenhorst CE, Dawson B, Goodman C, Sim M, Cox GR, Gore CJ, Tjalsma H, Swinkels DW, Peeling P. Influence of post-exercise hypoxic exposure on hepcidin response in athletes. Eur J Appl Physiol. 2014 … Continue reading
Online forums tend to showcase the most eye-catching success stories with topical dutasteride, cases where dramatic regrowth happens.
Figure 2: Anecdotal results are often way more dramatic than the typical.
These anecdotes, while genuine, sit far above the trendline. The problem comes when patients see only these high points and assume the same results are likely for themselves. When their own progress looks slower or less dramatic, they may quit early or wrongly conclude that the treatment doesn’t work.
Figure 3: Expected results are often far more dramatic than probable results.
The key is avoiding unrealistic benchmarks based on outliers. Our Regrowth Potential framework is designed to ground expectations in the probable but not the possible. For most patients, topical dutasteride leads to stabilization plus modest regrowth, with rare cases of strong “hyper responses” that shouldn’t be seen as the norm.
Most people using topical dutasteride experience stabilization of hair loss first, with the earliest cosmetic regrowth occurring at around 3 months. Typical regrowth usually brings hair growth back to where it was between 1-3 years ago, rather than to pre-hair loss density.
Several factors influence where someone falls on the response spectrum with topical dutasteride. Starting treatment earlier generally leads to better outcomes, as newer thinning areas respond more readily while long-standing bald spots are far less likely to improve. The specific therapy or combination of treatments used also matters, since some approaches are more effective depending on the stage or pattern of hair loss. Consistency is critical; missed applications or stopping too soon can quickly lower results below the expected average.
Principle #2: Poorer Evidence = Greater Variability
Predictability depends heavily on evidence quality. Treatments with a large body of research product outcomes that cluster tightly around the average, while those with fewer studies show more variability between patients.
Figure 4: Bell curves are a great way to show the typical outcomes depending on evidence. For finasteride, a narrow bell curve shows predictable outcomes. For topical dutasteride, a wide, shallow curve represents a much wider spread of outcomes.
Finasteride is a good comparison point: with dozens of randomised controlled trials, we can be confident that most users fall close to the mean response, represented by a narrow bell curve. Topical dutasteride, however, is newer and much less studied. While emerging data and user reports suggest potential benefits, even in some cases matching or exceeding oral outcomes, the evidence base is still limited.
In short, the more research that exists, the stronger our confidence in expected results. For topical dutasteride, the smaller body of evidence means higher variability. Some users may beat the average, but others will fall below it. Recognizing this can help you temper expectations and make better-informed decisions.
Evidence quality is the rubric we can use to determine whether the results will be predictable or variable.
10 Topical Dutasteride User Transformations
Now that we’ve covered the mechanisms of topical dutasteride while setting reasonable expectations, let’s look at what the results might actually look like in practice. Below, we’ve curated 10 Reddit case studies of users who saw clear improvement with topical dutasteride.
The examples below help see what’s possible and do not represent what every user should expect. They are purely anecdotal and not independently verified by us or any qualified third party.
User Profile #1: 14–15 Months on Topical Dutasteride 0.1% (once weekly; early 1%)
Source: u/inferismetal via r/tressless.
This user started topical dutasteride monotherapy, initially at 1% for the first 3 weeks, then at 0.1%, which he typically applied once weekly at 2 mL. He also used a low-level laser therapy (LLLT) device 3x/week in 15-minute sessions.
He reported noticeable thickening at month 3 and significant visible improvement with complete recovery of the crown by month 4.5. He also reported that results were maintained at months 14-15 and that gains were stabilized as of a later post. He observed that shedding had reduced to 5 hairs/day from the peak of 100/day. He did not report any systemic side effects on topical dutasteride 0.1%.
User Profile #2: 6 Months on Topical Dutasteride 2%
Source: u/Asleep_Ad_8009 via r/tressless.
This user chose topical dutasteride 2% twice per week for 6 months, increasing the frequency to daily application on the hairline only. He previously tried topical minoxidil and weekly microneedling but considered himself a non-responder.
He reported a heavy shedding phase starting from month 2 and lasting for 1-2 months. At the 6-month mark, he reported that shedding is still above pre-treatment levels but is lower than the peak shedding phase. He also reported no regrowth at month 6 and stated that his hair was in worse condition than at baseline. He reported no side effects overall from topical dutasteride.
User Profile #3: 8 Months on Topical Dutasteride 0.1% Monotherapy
Source: u/colter108 via r/tressless.
This male chose to use topical dutasteride monoetherapy at the 0.1% concentration with an application frequency of once weekly at months 0-4 and twice weekly at months 5-8.
At the 8-month mark, the photos showed subtle improvement with the user perceiving a slightly improved appearance. The user stated that his alopecia was aggressive and that a twice-weekly application may still be insufficient. He reported no side effects when asked.
User Profile #4: 4 Months on Topical Dutasteride 0.1% (with Minoxidil/Tret Mix in Vehicle)
Source: u/DaBoa70 via r/tressless
This user started applying topical dutasteride 0.1% every other day, eventually switching to once daily. His compounded lotion also contained minoxidil, plus tretinoin, melatonin, silicon, and biotin. He used a separate AM minoxidil and tretinoin when at home, in addition to once-weekly derma rolling sessions.
After 4 months on the topical dutasteride-based regimen, he reported the emergence of some baby hairs and some definite regrowth. He exhibited a noticeably better hairline compared to the pre-minoxidil baseline. He reported no side effects.
User Profile #5: 2 Months on Topical Dutasteride (w/ Minoxidil)
Source: u/Methibosheth via r/tressless.
This user applied both a topical dutasteride 0.5% solution and topical minoxidil twice daily over the course of 2 months. The user stated that his shedding had dropped noticeably after adding dutasteride to his minoxidil regimen. He reported no side effects with topical dutasteride and acknowledged that twice-daily application at 0.5% may be overkill.
It should be noted that it is really quite difficult to see improvements in cases where users take photos with different lighting and at different distances. If you are tracking your progress, try to keep as many variables the same as possible so that you can give yourself as accurate a comparison as possible.
User Profile #6: 6 Months on Topical Dutasteride 0.05% + Dutasteride Mesotherapy 0.01%
Source: u/Zealousideal-Ice4996 via r/tressless.
This user presented with diffuse thinning and was previously on finasteride with no side effects. He is motivated and willing to use medical interventions to achieve recovery. He applied topical dutasteride 0.05% twice weekly and dutasteride mesotherapy 0.01% once weekly via a 0.5 mm dermapen for a total duration of 6 months.
At the 6-month follow-up, he showed visible thickening, although he acknowledged that the results have not been dramatic. He stated in replies that he attained enough stabilization to later proceed with a crown transplant. The user reported no side effects.
User Profile #7: Male (53), 10 Weeks on Topical Dutasteride 0.3% + Minoxidil 8% (twice daily)
Source: u/Ok-Carrot-9987 via r/tressless.
This 53-year-old male applied a topical solution of 0.3% dutasteride and 8% minoxidil twice daily over 10 weeks. He later reduced the frequency to once daily and added intermittent microneedling, rosemary oil, and onion juice for gray hairs.
He noted a shedding phase starting in week 2, with visible thickening and density improvement by week 10, when he posted the before/after photos. In a later update, the user affirmed his continued improvement with reduced application frequency (once daily) and maintenance of results. He reported no side effects and expressed satisfaction with the early response.
User Profile #8: 4 Months on Topical Dutasteride + Topical Minoxidil
Source: u/Weird_Year2254 via r/tressless.
This Redditor’s intervention of choice was topical dutasteride (Anagenica) once daily, topical minoxidil, and dermastamp once weekly. At the 4-month mark, he reported a 90% reduction in hair shedding, the emergence of baby hairs at the crown and temples, and early vellus activation at the crown, which remained slower to respond.
He reported no side effects to date and did not mention any scalp irritation despite weekly stamping sessions. Notably, he did not state the exact dutasteride concentration in the topical solution, nor whether his dutasteride and minoxidil were contained in separate bottles or were pre-mixed in a single solution.
User Profile #9: Post-Hair Transplant Patient, 5.5 Months on Topical Dutasteride (Avodart capsules mixed into topical Minoxidil)
Source: u/grandtheftpixel via r/tressless.
This user began using a self-prepared solution by crushing Avodart (dutasteride) capsules and mixing them into a minoxidil PG/alcohol bottle, which he applied twice daily for 5.5 months. He did not report the dutasteride concentration in his homemade solution. He observed significant shedding and thinning, especially at the vulnerable right-sided native zone. After the first month, he also observed many thin, white vellus hairs appearing on top. He reported no systemic side effects.
User Profile #10: Male (NW7) with 18 Months Total Therapy Prior to Full-Dose Oral Dut
Source: u/Oxi_Dat_Ion via r/tressless.
This man used topical dutasteride for an unspecified period after 12+ months on minoxidil, before escalating to oral dutasteride 0.5 mg every two weeks, then to twice daily. Additionally, he underwent microneedling monthly at 0.75 mm and used ketoconazole shampoo 1 to 2 times weekly.
The patient showed visible improvement despite starting therapy from an advanced stage of hair loss (NW7). He reported no significant side effects, but has decreased his microneedling frequency due to scalp soreness.
Since he did not specify the duration of his topical dutasteride use and given that he has used several other formulations at varying doses over the 18 months, it is not possible to reliably estimate the degree to which topical dutasteride is responsible for the regrowth.
Probable Regrowth Timeline for Topical Dutasteride
We have laid out a likely timeline for topical dutasteride, but this is based on three studies we could find using it.
Months 0-3: Early cosmetic improvements
- Most users won’t see regrowth yet.
- Shedding may occur in the first few weeks (especially if combined with microneedling).
- Early pharmacokinetic studies show scalp DHT reduction with minimal systemic absorption at appropriate doses.
Months 3-6: First cosmetic improvements
- This is the case for both low (0.025%) and higher concentrations (0.1-0.3%) of topical dutasteride.
- Clinical studies found measurable increases in hair density when topical dutasteride was paired with microneedling.
- Improvements are usually more visible in the crown and mid-scalp than at long-standing bald spots.
Months 6-9: Noticeable regrowth in responders
- Hair density and thickness continue to improve.
- Studies reported ~10-15% increases in hair accounts and 15-20% increases in density, especially with microneedling.
- Cosmetic changes (less scalp show-through, fuller appearance) often emerge in this window.
- Data is limited after ~7 months.
Months 9+
- Likely that many responders achieve their most visible results by 9-12 months.
- Long-term clinical data are sparse; by 36 months, most hair gains will have occurred.
So, what about all the success stories we see online? This is typically called survivorship bias.
Figure 5: Survivorship bias skews perception of hair loss treatments. Online accounts often highlight extreme outcomes, either exceptional success or very poor results, while the typical, average outcomes experienced by most remain underrepresented.
Combining Treatments
If you want to increase the effects of topical dutasteride, combining it with additional therapies can have a beneficial effect.
One study compared topical minoxidil 5% alone vs. in combination with topical dutasteride, showing both approaches to be effective and safe for AGA in men and women.[7]Obeid, M.N.A., Fattah, N.S.A., Elfangary, M.M., Husseni, R.M.A. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 … Continue reading
Another trial evaluated “minoxidil-dutasteride tattooing”, a combined delivery, which led to notable clinical improvement in hair growth for patients with AGA.[8]Ragi, S.D., Ghanian, S., Rogers, N., Peterson, D.M., Johnson, L.S., Wambier, C.G. (2023). Evaluation of hair regrowth after minoxidil and dutasteride tattooing in men with androgenetic alopecia. JAAD … Continue reading
Microneedling is increasingly used to enhance drug penetration for hair loss therapies. Clinical protocols recommend microneedling (often with 1.5-2.5 mm rollers)before topical dutasteride application for improved efficacy, with studies confirming significant increases in hair density and shaft thickness.[9]Sanchez-Meza.E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical … Continue reading
Recent reviews discuss rotational therapy for partial responders, switching between topical and oral 5ɑ-reductase inhibitors to individualize regimens and maximize hair regrowth while managing side effects.[10]Mysore, V., Kumaresan, M., Dashore, S., Venkatram, A. (2023). Combination and Rotational Therapy in Androgenetic Alopecia. Journal of Cutaneous and Aesthetic Surgery. 16(2). 71-80. Available at: … Continue reading
How Can I Maintain Realistic Expectations?
To keep realistic expectations with topical dutasteride, it’s important to remember that most users will see noticeable improvement and stabilization, though individual results can vary. Realistic does not mean pessimistic; most people benefit, but baseline expectations should be guided by typical clinical results rather than extreme cases.
Topical dutasteride may “rewind” hair loss by several months to a couple of years, but full restoration to pre-hair loss density is uncommon. The most likely outcome is gradual improvement in hair density and a slowing of hair loss, rather than dramatic, multi-year regrowth or complete reversal.
Practical Takeaways
- Topical dutasteride is promising, but still considered to be experimental. Early clinical studies show significant efficacy, especially in comparison to finasteride, but more long-term research is needed to confirm its effectiveness and safety in larger populations.
- Systemic side effects appear reduced compared to oral use, but topical dutasteride is not entirely risk-free. Phase II trials report little to no local irritation and only modest changes in serum hormone levels with topical formulations, suggesting an improved systemic safety profile, but ongoing monitoring is recommended.
- Topical dutasteride is best used as part of a multi-therapy regimen. Combination approaches (for example, with minoxidil or microneedling) tend to enhance results and individualize treatment, maximizing regrowth while managing side effects.
Final Thoughts
When evaluating topical dutasteride, it’s essential to apply the framework of Regrowth Potential and Evidence Quality. While initial research and clinical experience suggest topical dutasteride is promising, the clinical data are lacking. Setting realistic expectations is important; focusing on typical results rather than extreme anecdotes helps prevent disappointment and treatment abandonment, encouraging patients to make informed decisions based on both scientific evidence and personal goals.
References[+]
References ↑1 Frye, S.V. (2006). Discovery and clinical development of dutasteride, a potent dual 5alpha-reductase inhibitor. Current topics in medicinal chemistry. 6(5). 405-421. Available at: https://doi.org/10.2174/156802606776743101 ↑2 Noor, N.M., Sheikh, K., Somavarapu, S., Taylor, K.M.G. (2017). Preparation and characterization of dutasteride-loaded nanostructured lipid carriers coated with stearic acid-chitosan oligomer for topical delivery. European journal of pharmaceutics and biopharmaceutics. 117. 372-384. Available at: https://doi.org/10.1016/j.ejpb.2017.04.012 ↑3 Nada, E.A., El-Dawla, R.E., El Maged, W.M.A., Emagd, A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). Available at: https://smj.journals.ekb.eg/article_42083_24b61cbba4be9982db23c318414034c0.pdf ↑4, ↑7 Obeid, M.N.A., Fattah, N.S.A., Elfangary, M.M., Husseni, R.M.A. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 mg) in Treatment of Androgenetic Alopecia. QJM: An International Journal of Medicine. 117(2). Available at: https://doi.org/10.1093/qjmed/hcae175.207 ↑5 Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., KSSVV, S.R., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects with Androgenetic Alopecia. Cureus. 17(8). E89309. Available at: https://doi.org/10.7759/cureus.89309 ↑6 Badenhorst CE, Dawson B, Goodman C, Sim M, Cox GR, Gore CJ, Tjalsma H, Swinkels DW, Peeling P. Influence of post-exercise hypoxic exposure on hepcidin response in athletes. Eur J Appl Physiol. 2014 May;114(5):951-9. doi: 10.1007/s00421-014-2829-6. Epub 2014 Feb 1. PMID: 24487960. ↑8 Ragi, S.D., Ghanian, S., Rogers, N., Peterson, D.M., Johnson, L.S., Wambier, C.G. (2023). Evaluation of hair regrowth after minoxidil and dutasteride tattooing in men with androgenetic alopecia. JAAD International. 12. 103-104. Available at: PMID 37404245 ↑9 Sanchez-Meza.E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical dutasteride solution for androgenetic alopecia: a randomized placebo-controlled study. Journal of the European Academy of Dermatology and Venereology. 36(10). E806-e808. Available at: https://doi.org/10.1111/jdv.18285 ↑10 Mysore, V., Kumaresan, M., Dashore, S., Venkatram, A. (2023). Combination and Rotational Therapy in Androgenetic Alopecia. Journal of Cutaneous and Aesthetic Surgery. 16(2). 71-80. Available at: https://doi.org/10.4103/JCAS.JCAS_212_22 Oral finasteride has long been the mainstay for androgenic alopecia (AGA), but many patients and clinicians have turned to dutasteride, a more potent 5ɑ-reductase inhibitor, in pursuit of superior hair growth.
Oral dutasteride (0.5 mg daily) can reduce dihydrotestosterone (DHT) by 90% or more (compared to ~70% with finasteride), and clinical studies show it grows more hair than finasteride. In fact, a 24-week trial found that oral dutasteride 0.5 mg significantly outperformed finasteride 1 mg in increasing hair counts.[1]Shanshanwal, S.J.S., Dhurat, R.S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: A randomized controlled … Continue reading
However, dutasteride’s very potency raises concerns: will it also increase side effects like decreased libido or hormonal disturbances? Interestingly, research so far suggests that dutasteride has a similar tolerability profile to finasteride with no clear increase in sexual side effects despite its stronger DHT suppression.[2]Almudimeegh, A., Almutairi, H., AlTassan, F., AlQuraishi, Y., Nagshabandi, K.N. (2024). Comparison between dutasteride and finasteride in hair regrowth and reversal of miniaturization in male and … Continue reading
Even so, oral dutasteride’s reputation (it’s approved for prostate enlargement and used off-label for hair loss) makes some men uneasy, especially those who experienced side effects on finasteride. This has led to rising interest in topical dutasteride formulations. By delivering dutasteride directly to the scalp, the goal is to concentrate its DHT-blocking action where it’s needed (hair follicles) while limiting how much enters the bloodstream. The questions we need to answer are:
- Does topical dutasteride work?
- Does it match or even exceed the efficacy of oral treatments?
- Can it maintain these benefits with a more favorable side effect profile?
Ulo offers dutasteride options that range from low to high dose dutasteride – allowing you to be flexible in your treatment choices.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Oral vs. Topical Dutasteride
Early evidence indicates that topical dutasteride can indeed improve hair growth, in some cases achieving results comparable to oral therapy but with lower systemic DHT reduction.
Study One
One study tested dutasteride solutions of 0.01%, 0.02%, and 0.05% against a placebo (and an active control) over 24 weeks. All dutasteride groups showed significant increases in hair count versus placebo, and the 0.05% topical solution actually showed more efficacy than oral finasteride (1 mg/day) in promoting regrowth.[3]Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammed, J., Rao, S., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the … Continue reading
Figure 1: Representative images of hair growth in male-pattern androgenetic alopecia after treatment with 0.01% dutasteride topical solution at 12 week (b) and 24 week (c) vs baseline (a); 0.02% dutasteride topical solution at 12 week (e) and 24 week (f) vs baseline (d); 0.05% dutasteride topical solution at 12 week (h) and 24 week (i) vs baseline (g); oral finasteride 1 mg tablets at week 12 (k) and week 24 (l) vs baseline (j); placebo at week 12 (n) and week 24 (o) vs baseline (m).[4]Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammed, J., Rao, S., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the … Continue reading Image obtained in line with the PMC Copyright License.
Study Two
A recent randomized controlled study compared oral dutasteride (0.5 mg daily) to topical dutasteride (0.02% administered via monthly microneedling), both combined with daily topical minoxidil 5%. The trial found that all groups (including minoxidil alone) saw improvement, but the topical dutasteride via microneedling group demonstrated results comparable to the oral dutasteride group in terms of increased hair density and width, with the added advantage of fewer systemic side effects.[5]Obeid, M.N.A., Fatteh, N.S.A., Elfangary, M.M., Husseni, R.M.A. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 … Continue reading
Patient satisfaction and physician assessment favored the microneedling + topical dutasteride group, suggesting that this method may offer similar efficacy to systemic dutasteride, but with reduced risk of systemic adverse effects. The study included both men and women with AGA.
How Does Dutasteride Work?
Dutasteride is a potent inhibitor of the enzyme 5ɑ-reductase, which exists in multiple isoforms in the body. Unlike finasteride (which selectively targets the type II isoenzyme), dutasteride blocks both type I and type II 5ɑ-reductase.[6]Botto, H., Lan, O., Poulain, J-E., Comenducci, A. (2005). Effect of dutasteride on reduction of plasma DHT following finasteride therapy in patients with benign prostatic hyperplasia. Progrés en … Continue reading
Type II is abundant in hair follicles and the prostate, while Type I is found in skin, sebaceous glands, and liver. By inhibiting both, dutasteride more completely prevents the conversion of testosterone to DHT.
At a standard oral dose of 0.5 mg/day, dutasteride can drive serum and scalp DHT levels down by ~90% or more.[7]Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology. (5-6). 833-843. Available at: … Continue reading This is a more extensive suppression than finasteride achieves at typical doses. This dramatic drop in DHT removes the androgenic stimulus that causes susceptible hair follicles to shrink and enter shorter growth phases, thereby slowing hair loss and allowing follicles to recover over time.
Topical dutasteride aims to harness this same mechanism locally. When applied on the scalp, dutasteride penetrates into the skin and hair follicle, binding to 5ɑ-reductase enzymes in the dermis and around the follicle bulb. Inhibiting local DHT production creates a scalp environment more conducive to hair growth.
Pharmacokinetic studies of dutasteride demonstrate parallel linear and nonlinear elimination, with nonlinear (saturable) pathways dominating at low doses. These pathways quickly become saturated, so even small concentrations can lead to substantial enzyme inhibition. As drug concentrations rise and saturate the target enzyme, higher doses yield diminishing increases in effect.[8]Gisleskog, P.O., Hermann, D., Hammarlund-Udenaes, M., Karlsson, M.O. (1999). The pharmacokinetic modelling of GI198745 (dutasteride), a compound with parallel linear and nonlinear elimination. … Continue reading
Therefore, topical dutasteride at 0.01-0.05% may achieve a large fraction of the DHT reduction that an oral dose would, as long as it reaches the target tissue. This is why even leakage of a small dose into circulation can suppress serum DHT measurably.
Another factor is dutasteride’s long half-life, about 4-5 weeks.[9]Azzouni, F., Godoy, A., Li, Y., Mohler, J. (2011). The 5 Alpha-Reductase Isozyme Family: A Review of Basic Biology and Their Role in Human Diseases. Advances in Urology. 2012(530121). 1-18. Available … Continue reading This prolonged half-life means that any dutasteride absorbed systemically will accumulate with continued use. From a mechanism standpoint, this is a double-edged sword:
- It ensures continuous enzyme inhibition (dutasteride remains bound to 5ɑ-reductase).
- But it also means systemic exposure is amplified over time if absorption isn’t minimized.
What the Science Says
Research on topical dutasteride is not as extensive as that on topical finasteride, but a growing number of trials and case series shed light on its efficacy. Below, we summarize key findings across different concentrations and use cases:
Study Concentration & Vehicle Hair Growth-Outcomes Systemic/Serum Findings Nada et al, 2018, prospective randomized study, 30 men, 24 weeks. Group 1: Minoxidil + topical dutasteride (0.02%). Group 2: Minoxidil only.
Group 1 showed a significant increase in hair density, width, and terminal/vellus ratio compared to Group 2. Patient assessment showed higher satisfaction in Group 1 than in Group 2. Reduction in serum DHT observed. Sanchez-Meta et al, 2022, double-blind RCT, 34 men, 16 weeks. Group 1: Minoxidil and topical dutasteride (0.01%). Group 2: Minoxidil only.
Minoxidil and dutasteride produced a superior overall change in hair thickness and density compared to minoxidil alone. Not evaluated. Panuganti et al, 2025, phase II randomized, double-blind study, 135 men, 24 weeks. Group 1: Dutasteride 0.01% w/v topical solution. Group 2: Dutasteride 0.02% w/v. Group 3: Dutasteride 0.05% w/v. Group 4: Oral finasteride 1 mg. Group 5: Placebo. Dose-dependent increase in target area hair count (TAHC) vs placebo. Dutasteride 0.05% superior to finasteride at week 24. Mean TAHC increase: 0.01% (12.78), 0.02% (20.03), 0.05% (34.30) vs finasteride (12.57). More patients achieved investigator global photography assessment ≥+2 with 0.05% vs finasteride. Dutasteride caused modest changes in serum testosterone/DHT, while finasteride caused moderate changes. 0.05% dutasteride showed a better pharmacokinetic profile with reduced systemic absorption compared to oral finasteride. Overall, while there is a lack of peer-reviewed scientific literature, it does support the idea that topical dutasteride is an effective treatment for AGA. Where topical dutasteride does shine is in offering these benefits with reduced systemic involvement, making it a potential option for those who need dutasteride’s effects but are wary of its systemic effects.
Why Formulation and Dose Matter
Not all topical dutasteride products are created equal. Two main factors determine how well topical dutasteride works (and how “safe” it is systemically): formulation (the vehicle and additives) and dosage regimen (concentration and frequency).
- Vehicle and Penetration: Dutasteride is a large lipophilic molecule with poor water solubility.[10]Kim, N.A., Choi, D.H., Kim, J.Y., Kim, K.H., Lim, D.G., Lee, E., Park, E-S., Jeong, S.H. (2014). Investigation of polymeric excipients for dutasteride solid dispersion and its physicochemical … Continue reading This makes formulation challenging; you need solvents or carriers that keep it stable and help it traverse the skin barrier. Newer vehicles like lipid carriers or cyclodextrin inclusion complexes might improve scalp targeting while limiting diffusion into blood.[11]Kim, M-S., Ha, E-S., Choo, G-H., Baek, I-H. (2015). Preparation and in vivo evaluation of a dutasteride-loaded solid-supersaturable self-microemulsifying drug delivery system. International Journal … Continue reading,[12]Min, M-H., Park, J-H., Choi, M-R., Hur, J-H., Ahn, B-N., Kim, D-D. (2018). Formulation of a film-coated dutasteride tablet bioequivalent to a soft gelatin capsule (Avodart): Effect of γ-cyclodextrin … Continue reading
- Applied Dose vs. Concentration: Users often focus on the percentage (e.g., 0.1% vs 0.02%), but the total amount of drug applied per session is just as important. For example, using 2 mL of a 0.1% solution (which contains 2 mg dutasteride) will deliver four times the dutasteride to your scalp as 1 mL of the same solution. For example, one study using a 0.25% topical used at 4 sprays (around 400 μL) caused a much larger drop in serum DHT than the same solution used at 1-2 sprays (100-200 μL).[13]Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology. 240(5-6). 833-843. Available at: … Continue reading
- Furthermore, volume matters: a seemingly “low” concentration could cause systemic effects if applied in large quantities. Conversely, high concentrations used sparingly might localize better.
- Dosing Frequency: Dutasteride’s long half-life means you may not need daily application to maintain its effect on the scalp. Ultimately, the goal is to find a regimen that keeps follicles saturated with dutasteride whilst avoiding leakage into the bloodstream.
Minimizing Systemic Absorption
As we have mentioned above, one of the primary motivations for using topical dutasteride is to limit the risk of side effects that come with systemic DHT suppression. There are several strategies you can use to achieve this.
Vehicle & Penetration Enhancers
As discussed, formulation plays a huge role. Nanoemulsion-based gels and nanoemulgels significantly increase skin permeation and deposition of dutasteride compared to conventional formulations, with optimized nanoemulsions showing up to 1.5-fold enhancement in permeation.[14]Ali, M.S., Alam, M.S., Alam, N., Siddiqui, M.R. (2014). Preparation, Characterization and Stability Study of Dutasteride-Loaded Nanoemulsion for Treatment of Benign Prostatic Hypertrophy. Iranian … Continue reading If systemic absorption is a concern, you could opt for a slightly less efficient formulation.
Time On Scalp
The longer the product stays on your scalp, the more the drug can penetrate, increasing efficacy (and potentially systemic absorption). Washing your hair too soon can rinse away some of the medication, potentially reducing efficacy. Many protocols recommend allowing at least 4 hours of contact time (and preferably 6-8 hours) before washing off a topical.
Application Frequency
Frequency can be adjusted to manage systemic load. Because dutasteride binds 5ɑ-reductase for so long, daily application might not be necessary for full benefit. If you find that daily use lowers your serum DHT more than desired, you could try every other day or even twice-weekly dosing.
Scalp Condition and Individual Variation
Your skin’s permeability is unique to you..Differences in skin thickness can affect drug permeability (and therefore efficacy).[15]Noor, N.M., Abudl-Aziz, A., Sheikh, K., Somavarapu, S., Taylor, K.M.G. (2020). In vitro Performance of Dutasteride-Nanostructured Lipid Carriers Coated with Lauric Acid-Chitosan Oligomer for Dermal … Continue reading However, you should be mindful that if you have a scalp condition (like seborrheic dermatitis, etc), you should avoid using any topical hair loss treatments until it has resolved.
Application Area
The larger the area of scalp you treat, the more drug can be absorbed. Treating just the crown vs. the entire scalp could proportionally change systemic exposure. If you have diffuse thinning and apply broadly, you might consider using a lower concentration to compensate for the larger area.
To minimize systemic absorption without compromising results, a practical approach can be:
- Use the lowest effective concentration.
- Use the smallest effective volume.
- Use the least frequent schedule.
This will often require trial and error as you find out what works for you. If you notice side effects like decreased libido, brain fog, or breast tenderness, these could be signs of systemic absorption. In such cases, dial back the frequency or concentration.
How to Maximize Gains and Minimize Risk
If you decide to use topical dutasteride, here are strategies to get the best hair growth results while keeping your risk low:
- Establish a Baseline (Lab Tests): Before starting, it’s wise to get bloodwork for your hormone levels – especially serum DHT and testosterone. This baseline will tell you where you started. Some people also get a full hormone panel (if advised to do so by their doctor). You can then re-test after 4-6 weeks to monitor any significant changes.
- Standardize Your Application Routine: Consistency is key for results. Apply the treatment to a dry scalp. If using a dropper or spray, try to target thinning areas evenly and rub it in gently. Pick a time of day you can stick with. Many prefer nighttime so it can absorb overnight; just be cautious about transferring to pillows (and thereby to partners). If you apply it in the morning, ensure it dries before any activity that might cause sweat (sweating could increase absorption or cause the product).
- Add Proven Adjuncts (like minoxidil or microneedling): To maximize regrowth, consider combining therapies. Minoxidil can synergize with dutasteride by stimulating follicles directly, while dutasteride tackles the hormonal aspect. As seen above, studies show that dutasteride and minoxidil yield better density than either alone. Microneedling is another option. Weekly or monthly sessions of dermarolling have been shown to boost topical drug delivery and induce growth factors.[16]Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating the efficacy and safety of combined … Continue reading
- Be Patient and Track Progress: Hair growth is a slow process. Do not expect major changes in the first weeks. In fact, you might experience some shedding initially. This can happen with any effective treatment and usually subsides. Give the treatment at least 6 months to a year to judge its effect. To accurately gauge progress, use objective tracking, such as standardized photos, hair counting, or a dermascope.
- Lifestyle and Supplements: While on treatment, avoid introducing supplements that might complicate treatment. For instance, creatine may increase DHT, and quercetin may decrease it.[17]van der Merwe, J., Brooke, N.E., Myburgh, K.H. (2009). Three weeks of creatine monohydrate supplementation affects dihydrotestosterone to testosterone ratio in college-aged rugby players. Clinical … Continue reading,[18]Ma, Z., Nguyen, T.H., Huynh, T.H., Do, P.T., Huynh, H. (2004). Reduction of rat prostate weight by combined quercetin-finasteride treatment is associated with cell cycle deregulation. Journal of … Continue reading
- Monitor Side Effects: If you notice any difference, then keep a note and let your healthcare provider know. You may be able to adjust the dosage to maintain hair gains and reduce side effects.
Combination Therapies: Enhancing Outcomes
Combination therapy can amplify the benefits of topical dutasteride. We’ve summarized a few of these below:
Dutasteride and Fractional CO2 Laser
One study was conducted in which 30 male AGA patients were randomized to either fractional CO2 laser plus topical dutasteride (0.002%) applied after each session.[19]Galal, S.A., Ali, M.S., HafizHala, H.S.A. (2025). Comparative study between fractional CO2 laser alone versus fractional CO2 laser combined with topical dutasteride in treatment of male androgenic … Continue reading Each group received 3 sessions, one month apart. The combination group had significantly superior increases in terminal hair count and reductions in vellus hair and hair diameter diversity versus laser alone. Higher patient satisfaction was also reported in the combination group.
Figure 1: Combination treatment results: (A) Clinical photos before treatment (B) showing moderate improvement after treatment. (C) dermoscopic photos before treatment, counting vellus hair (16.8%), terminal hair (83.2%), and calculating hair diameter diversity (52.1%) per field. (D) dermoscopy after treatment with fractional CO2 laser combined with topical dutasteride; vellus hair (13.5%), terminal hair (86.5%), and hair diameter diversity (86.5%) per field. Vellus hair; blue arrow, terminal hair; black arrow, single pilosebaceous unit; green arrow, and yellow dot; yellow arrow.[20]Galal, S.A., Ali, M.S., HafizHala, H.S.A. (2025). Comparative study between fractional CO2 laser alone versus fractional CO2 laser combined with topical dutasteride in treatment of male androgenic … Continue reading Image obtained in line with the Creative Commons License.
Triple Topical Therapy: Dutasteride, Finasteride, and Minoxidil
Fifteen male AGA patients, including both atopic and non-atopic subjects, used a topical compound containing finasteride, dutasteride, and minoxidil (“NuH Hair”), with optional additions of oral finasteride, topical minoxidil foam, and ketoconazole shampoo.[21]Rafi, A.W., Katz, R.M. (2011). Pilot Study of 15 Patients Receiving a New Treatment Regimen for Androgenic Alopecia: The Effects of Atopy on AGA. ISRN Dermatology. 2011(241953). Available at: … Continue reading
All patients experienced significant new hair growth. Among those using all four components, significant regrowth was seen as early as 30 days, with “major” growth in three patients by day 30 and in all by day 60. In those using only the triple topical, significant regrowth was observed in all by 3 months, despite some using it less frequently than recommended. No systemic or local adverse effects were reported.
Topical Dutasteride + Microneedling
A 20-week, randomized, double-blind, placebo-controlled trial of microneedling plus topical 0.01% dutasteride solution (MNSD) vs. microneedling plus saline. The combination group demonstrated significant improvement in hair growth and density compared to the control. No systemic androgen suppression was detected, indicating a favorable safety profile.[22]Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical … Continue reading
The overarching theme of combinations like the above is synergy. By attacking hair loss from multiple angles, you often get faster, fuller, and more sustained regrowth.
Who Should (or Shouldn’t) Use Topical Dutasteride
Good Candidates
- Men with AGA who need a stronger option.
- Those who experienced side effects from oral finasteride/dutasteride.
Bad Candidates
- Anyone trying to conceive, or with infants, toddlers, or pregnant partners.
- People with non-AGA hair loss.
- Those with severe persistent side effects from 5ɑ-reductase inhibitors.
- People who aren’t able to commit to a consistent routine.
Final Verdict
So, does topical dutasteride work? In short, yes. Clinical trials and real-world use show that topical dutasteride can significantly improve hair growth and reduce shedding in men with androgenetic alopecia. When formulated correctly, it offers comparable scalp DHT suppression and regrowth potential to oral finasteride, and in some cases, it even approaches the efficacy of oral dutasteride, but with lower systemic exposure and fewer side effects.
The key is proper use. Results depend heavily on the right concentration, vehicle, dosage, and frequency. A well-balanced regimen, often guided by a medical provider, offers the best chance of success.
References[+]
References ↑1 Shanshanwal, S.J.S., Dhurat, R.S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: A randomized controlled open-label, evaluator-blinded study. 83(1). 47-54. Available at: https://doi.org/10.4103/0378-6323.188652 ↑2 Almudimeegh, A., Almutairi, H., AlTassan, F., AlQuraishi, Y., Nagshabandi, K.N. (2024). Comparison between dutasteride and finasteride in hair regrowth and reversal of miniaturization in male and female androgenetic alopecia: a systematic review. Dermatology Reports. 16(4). 9909. Available at: https://doi.org/10.4081/dr.2024.9909 ↑3 Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammed, J., Rao, S., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects with Androgenetic Alopecia. Cureus. 17(8). E89309. Available at: https://doi.org/10.7759/cureus.89309 ↑4 Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammed, J., Rao, S., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects with Androgenetic Alopecia. Cureus. 17(8). E89309. Available at: https://doi.org/10.7759/cureus.89309 ↑5 Obeid, M.N.A., Fatteh, N.S.A., Elfangary, M.M., Husseni, R.M.A. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 mg) in Treatment of Androgenetic Alopecia. An International Journal of Medicine. 117(2). Available at: https://doi.org/10.1093/qjmed/hcae175.207 ↑6 Botto, H., Lan, O., Poulain, J-E., Comenducci, A. (2005). Effect of dutasteride on reduction of plasma DHT following finasteride therapy in patients with benign prostatic hyperplasia. Progrés en Urologie. 15(6). 1090-1095. Available at: PMID: 16429658 ↑7 Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology. (5-6). 833-843. Available at: https://doi.org/10.1159/000541395 ↑8 Gisleskog, P.O., Hermann, D., Hammarlund-Udenaes, M., Karlsson, M.O. (1999). The pharmacokinetic modelling of GI198745 (dutasteride), a compound with parallel linear and nonlinear elimination. British Journal of Clinical Pharmacology. 47(1). 53-58. Available at: https://doi.org/10.1046/j.1365-2125.1999.00843.x ↑9 Azzouni, F., Godoy, A., Li, Y., Mohler, J. (2011). The 5 Alpha-Reductase Isozyme Family: A Review of Basic Biology and Their Role in Human Diseases. Advances in Urology. 2012(530121). 1-18. Available at: https://doi.org/10.1155/2012/530121 ↑10 Kim, N.A., Choi, D.H., Kim, J.Y., Kim, K.H., Lim, D.G., Lee, E., Park, E-S., Jeong, S.H. (2014). Investigation of polymeric excipients for dutasteride solid dispersion and its physicochemical characterization. Archives of Pharmacal Research. 37(2). 214-224. Available at: https://doi.org/10.1007/s12272-013-0180-9 ↑11 Kim, M-S., Ha, E-S., Choo, G-H., Baek, I-H. (2015). Preparation and in vivo evaluation of a dutasteride-loaded solid-supersaturable self-microemulsifying drug delivery system. International Journal of Molecular Sciences. 16(5). 10821-10833. Available at: https://doi.org/10.3390/ijms160510821 ↑12 Min, M-H., Park, J-H., Choi, M-R., Hur, J-H., Ahn, B-N., Kim, D-D. (2018). Formulation of a film-coated dutasteride tablet bioequivalent to a soft gelatin capsule (Avodart): Effect of γ-cyclodextrin and solubilizers. Asian Journal of Pharmaceutical Sciences. 14(3). 313-320. Available at: https://doi.org/10.1016/j.alps.2018.08.007 ↑13 Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology. 240(5-6). 833-843. Available at: https://doi.org/10.1159/000541395 ↑14 Ali, M.S., Alam, M.S., Alam, N., Siddiqui, M.R. (2014). Preparation, Characterization and Stability Study of Dutasteride-Loaded Nanoemulsion for Treatment of Benign Prostatic Hypertrophy. Iranian Journal of Pharmaceutical Research. 13(4). 1125-1140. Available at: PMID: 25587300 ↑15 Noor, N.M., Abudl-Aziz, A., Sheikh, K., Somavarapu, S., Taylor, K.M.G. (2020). In vitro Performance of Dutasteride-Nanostructured Lipid Carriers Coated with Lauric Acid-Chitosan Oligomer for Dermal Delivery. Pharmaceutics. 12(10). 994. Available at: https://doi.org/10.3390/pharmaceutics12100994 ↑16 Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating the efficacy and safety of combined microneedling therapy versus topical Minoxidil in androgenetic alopecia: a systematic review and meta-analysis. Archives of Dermatological Research. 317(1). 528. Available at: https://doi.org/10.1007/s00403-025-04032-1 ↑17 van der Merwe, J., Brooke, N.E., Myburgh, K.H. (2009). Three weeks of creatine monohydrate supplementation affects dihydrotestosterone to testosterone ratio in college-aged rugby players. Clinical Journal of Sports Medicine. 19(5). 399-404. Available at: https://doi.org/10.1097/JSM.0b013e3181b8b52f. ↑18 Ma, Z., Nguyen, T.H., Huynh, T.H., Do, P.T., Huynh, H. (2004). Reduction of rat prostate weight by combined quercetin-finasteride treatment is associated with cell cycle deregulation. Journal of Endocrinology. 181(3). 493-507. Available at: https://doi.org/10.1677/joe.0.1810493 ↑19 Galal, S.A., Ali, M.S., HafizHala, H.S.A. (2025). Comparative study between fractional CO2 laser alone versus fractional CO2 laser combined with topical dutasteride in treatment of male androgenic alopecia. 40(1). 16. Available at: https://doi.org/10.1007/s10103-024-04269-8 ↑20 Galal, S.A., Ali, M.S., HafizHala, H.S.A. (2025). Comparative study between fractional CO2 laser alone versus fractional CO2 laser combined with topical dutasteride in treatment of male androgenic alopecia. 40(1). 16. Available at: https://doi.org/10.1007/s10103-024-04269-8 ↑21 Rafi, A.W., Katz, R.M. (2011). Pilot Study of 15 Patients Receiving a New Treatment Regimen for Androgenic Alopecia: The Effects of Atopy on AGA. ISRN Dermatology. 2011(241953). Available at: https://doi.org/10.5402/2011/241953 ↑22 Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical dutasteride solution for androgenetic alopecia: a randomized placebo-controlled study. JEADV. 36(10). Available at: https://doi.org/10.1111/jdv.18285 IngredientsPharmaceuticalsNatural RemediesCompany ReviewsBy Perfect Hair Health TeamDec 16, 2025Microneedling For Hair Loss: 5 Strategies to Reduce Pain
Microneedling is an experimental therapy for androgenic alopecia and other hair loss disorders. One of its downsides is that it is painful to administer. However, there are simple and effective strategies to reduce pain levels while microneedling across the scalp:(1) opting for devices that ensure n...By Perfect Hair Health TeamDec 16, 2025Saw Palmetto: Is It As Effective As Finasteride?
Saw palmetto is less effective than finasteride for treating pattern hair loss – with clinical studies showing lower response rates and regrowth rates in men taking 320mg of saw palmetto versus 1mg of finasteride daily. However, saw palmetto is also associated with fewer sexual side effects. This me...By Perfect Hair Health TeamDec 16, 2025Platelet-Rich Plasma (PRP): Does It Regrow Hair? Maybe, Maybe Not.
PRP can increase hair counts by 25-40% for those with androgenic alopecia. However, it’s expensive and requires ongoing injections to maintain results. It works best as an adjunct treatment, and it might be less effective for women. Before you invest in PRP, make sure to vet your dermatologist with ...By Perfect Hair Health TeamDec 16, 2025The Misleading Results Of The Pumpkin Seed Oil-Hair Loss Study
People looking for natural solutions to hair loss often stumble upon pumpkin seed oil. After all, many articles claim that a 2016 study showed this supplement can regrow hair. But these articles are missing a key detail: that 2016 study wasn’t on pumpkin seed oil; it was on an herbal blend. Wh...By Perfect Hair Health TeamDec 15, 2025Topical Latanoprost For Hair Loss: Results, Doses, & Side Effect Concerns
For androgenic alopecia, clinical studies suggest that ~0.1 mg daily of topical latanoprost can improve hair counts, with visual improvements occurring in 30-50% of hair loss patients over 6-8 months. Telehealth brands are prescribin doses 10-20 times higher than what was clinically studied, thus po...By Richard Clayton, PhDDec 8, 2025Finasteride For Women: What’s The Perfect Dose?
Oral finasteride is an effective treatment for male pattern hair loss, also known as androgenic alopecia. However, the drug’s benefits to female pattern hair loss are less clear. Some studies show that females taking between 1.0 to 5.0 mg of finasteride daily improve their hair loss, while oth...By Sarah King, PhDDec 4, 2025Topical Finasteride Side Effects: What to Know
Topical finasteride is marketed as a “safer, localized” alternative to the oral pill, but how true is that? In this article, we break down what the science actually shows about systemic absorption, sexual and mood-related side effects, real-world user reports, and why responses vary so widely. Learn...By Sarah King, PhDDec 3, 2025Low-Dose Topical Dutasteride: Better Than Oral Finasteride? (New Study & Photos)
A new clinical trial claims that low-dose topical dutasteride outperforms oral finasteride for hair regrowth, a result so surprising that it contradicts nearly everything we’ve seen in 5+ years of real-world member data. In this deep dive, we break down what we previously knew about topical dutaster...By Perfect Hair Health TeamNov 29, 2025Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
Since earning FDA approval in 1997, Finasteride has been one of the most widely used hair loss drugs. To avoid potential sexual side effects, many opt for topical finasteride in hopes of isolating the drug’s DHT-reducing effects on the scalp. However, at certain doses, topical finasteride can enter ...By Michael Williams, PhDNov 27, 2025Blueprint Haircare Stack: Product Review
Bryan Johnson’s Blueprint Haircare Stack promises hair longevity rooted in science, but how much does that translate into real-world results? In this review, we unpack the ingredients, scrutinize the evidence, compare it to proven alternatives, and ask whether the Haircare Stack offers meaningful be... - Mission Statement
 Scroll Down
Scroll Down