- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Does Dandruff Cause Hair Loss?
-
Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
-
Ulo Review: #1 Customized Hair Loss Treatment in 2026
-
10 Best Hair Loss Treatments for Men
-
Best Minoxidil for Men: Top Picks for 2026
-
Does Ozempic Cause Hair Loss?
-
Does Masturbation Cause Hair Loss?
-
6 Best Hair Loss Treatments for Women
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
ArticlesDoes Ozempic Cause Hair Loss?
First Published May 1 2026Last Updated Apr 5 2026PharmaceuticalResearched & Written By:Catherine Kennedy, PhDReviewed By:Rob English, Medical EditorWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
GLP-1 medications like Ozempic are transforming weight-loss treatment. But viral posts and patient reports have raised concerns about unexpected hair shedding, and emerging research hints there may be something to it. But what does the science really show? We unpack the latest evidence to examine whether a true link exists, why hair loss might occur, and who should be concerned.
Full Article
Ozempic and other GLP-1 agonists have rapidly gained the status of “cultural phenomenon” over the past few years. A 2024 survey found that around 12% of adults in the US have used a GLP-1 agonist drug, with 6% currently taking the drug.[1]Montero, A., Sparks, G., Presiado, M., Hamel, L., (2024), KFF Health Tracking Poll May 2024: The Public’s Use And Views Of GLP-1 Drugs. Available at: … Continue reading This equates to roughly 30 million people, and numbers are only rising.
While the use of GLP-1 agonists was originally limited to the treatment of diabetes, they are now facing huge demand for their weight loss actions. Amongst these GLP-1 agonists, Ozempic is the most popular, making up approximately 37.8% of the GLP-1 agonist market with sales of around $22.3 billion in 2025.[2]Future Market Insights Inc., (no date), Ozempic Market Share & Size 2025-2035. Available at: https://www.futuremarketinsights.com/reports/ozempic-market (Accessed: 27 February 2026),[3]Research And Markets, (2024), Global GLP-1 Receptor Agonist (Ozempic, Rybelsus, Trulicity, Saxenda, Wegovy, Victoza, Mounjaro, Zepbound) Market Analysis Report 2024-2030 Featuring Eli Lilly And C, … Continue reading
GLP-1 agonists have not been in the public sphere for very long, meaning that understanding of long-term side effects is limited. As more and more people are using the drugs, potential side effects are beginning to emerge.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

One such side effect receiving growing concern on various social media and forums is hair loss, with reports including increased shedding and thinning of hair. But how accurate are these reports? Are they based on clinical evidence, or are they purely anecdotal?
To find out, we have searched through clinical literature to see whether the science supports these claims. In this article, we will review what Ozempic is and how it works, the available evidence supporting hair loss claims, and the mechanism through which Ozempic may be causing these issues.
What is Ozempic?
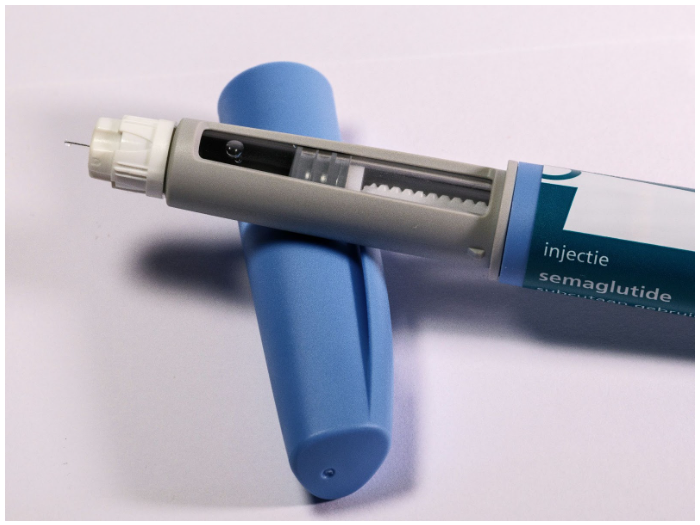
Ozempic is the brand name for the drug semaglutide, which is a glucagon-like peptide-1 (GLP-1) agonist. It is an FDA-approved drug originally developed to improve glycemic control in adults with type 2 diabetes, and now commonly used off-label as a weight-loss treatment.[4]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
While conducting early clinical trials for the use of semaglutide in diabetes, researchers noticed that patients taking semaglutide consistently lost more weight than those taking the placebo.[5]Sorli, C., Harashima, S.I., Tsoukas, G.M. (2017). Efficacy And Safety Of Once-Weekly Semaglutide Monotherapy Versus Placebo In Patients With Type 2 Diabetes (SUSTAIN 1): A Double-Blind, Randomised, … Continue reading These results prompted researchers to investigate semaglutide specifically as a weight-loss drug, where they demonstrated a ~15% reduction in weight in those treated with a weekly injection of semaglutide.[6]Garvey, W.T., Batterham, R.L., Bhatta, M. (2022). Two-Year Effects Of Semaglutide In Adults With Overweight Or Obesity: The STEP 5 Trial. Nature Medicine. 28(10). 2083-2091. Available at: … Continue reading
Semaglutide is available in three main forms:[7]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
Ozempic: once-weekly injection; FDA-approved for the treatment of type 2 diabetes.
Rybelsus: once daily tablets; FDA-approved for the treatment of type 2 diabetes.
Wegovy: once weekly injection; FDA-approved for weight loss.Other related GLP-1 agonists include Mounjaro and Liraglutide; these do not contain semaglutide as their active ingredient and work through slightly different mechanisms.
How Does Ozempic Work?
GLP-1 is a hormone that is released into the bloodstream in response to food consumption. It acts in the pancreas, gastrointestinal tract, and central nervous system. Its main effects are:[8]Holst, J.J. (2007). The Physiology Of Glucagon-Like Peptide 1. Physiological Reviews. 87(4). 1409-1439. Available at: https://doi.org/10.1152/physrev.00034.2006,[9]Drucker, D.J. (2022). GLP-1 Physiology Informs The Pharmacotherapy Of Obesity. Molecular Metabolism. 57. 101351. Available at: https://doi.org/10.1016/j.molmet.2021.101351
- Stimulating insulin secretion after food consumption to improve blood glucose levels post-prandial (after eating).
- Inhibiting glucagon secretion to limit glucose in the blood and help to reduce spikes in blood sugar.
- Slowing the emptying of the stomach, which further limits blood sugar spikes and helps people to feel fuller for longer.
- Activating GLP-1 receptors in the brain (responsible for appetite regulation), which results in increased satiety and reduced food cravings.
Ozempic is a synthetic analogue of natural GLP-1, essentially mimicking GLP-1 in the body to activate GLP-1 receptors. Through this, it activates the same pathways within the body as natural GLP-1, thereby acting to reduce blood sugar and food cravings.[10]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
Is There Any Association Between Ozempic and Hair Loss?
While Ozempic has been hailed by many as a miracle drug for the growing obesity crisis, it is not without risks. Many people experience gastrointestinal-linked side effects, including nausea, diarrhoea, stomach pain, and reflux.[11]Kommu, S., Berg, R.L. (2024). Efficacy And Safety Of Once-Weekly Subcutaneous Semaglutide On Weight Loss In Patients With Overweight Or Obesity Without Diabetes Mellitus-A Systematic Review And … Continue reading A small subset of people may experience more serious side effects, such as pancreatitis, issues with the gallbladder, or even kidney injury.[12]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
These side effects have been demonstrated in clinical trials and are relatively well-established. However, other side effects are seemingly coming to light on social media and forums, such as Reddit, with hair loss appearing to be a significant cause for concern. But is this true?
We will investigate the clinical evidence supporting this link between Ozempic and hair loss and discuss what the science says so far.
Clinical Evidence
The existing clinical data demonstrating hair loss with Ozempic treatment are limited. During the process of getting a product to the market, drugs like Ozempic go through extensive clinical trials to assess how effective they are, in addition to their safety. Any side effects or adverse events are recorded, enabling researchers to assess whether the benefits of the drug outweigh potential harms.
The initial clinical trials with Ozempic did not record alopecia or hair loss as a side effect. However, once the drug hit the market, reports of hair loss began to appear. There were sufficient reports that the FDA issued a warning in 2023 that GLP-1 receptor agonists (including Ozempic) may increase the risk of alopecia.[13]Center For Drug Evaluation And Research, (2024), July – September 2023 | Potential Signals Of Serious Risks/New Safety Information Identified By The FDA Adverse Event Reporting System (FAERS). … Continue reading
Case Reports
Case reports are short, published articles describing rare or unexpected clinical scenarios, such as adverse drug reactions. They typically describe one patient, and therefore do not demonstrate comprehensive proof of a drug effect for all users, only a one-off situation where this has occurred.
With the introduction of Ozempic to the masses, clinical case reports have begun to emerge demonstrating rare incidences of alopecia in those taking Ozempic.
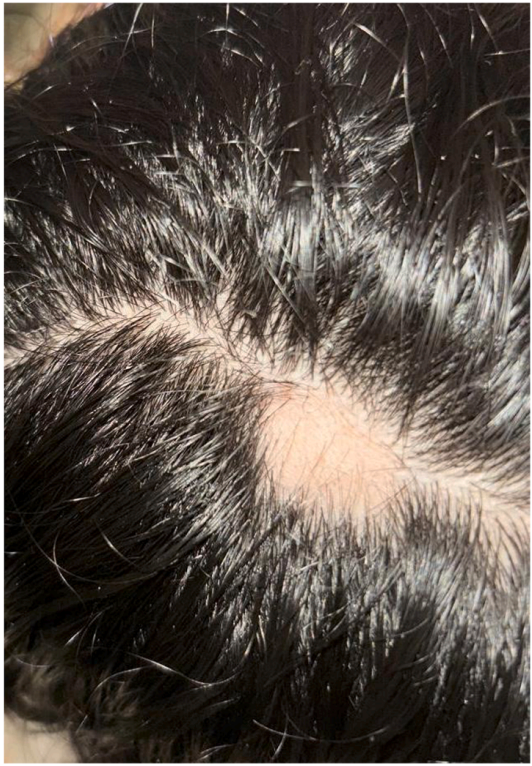
In one such study, an obese female in her early twenties who had been taking semaglutide for weight loss for four months started to develop patches of hair loss after three months (on an increase in dose from 0.25 to 0.5 mg weekly). After cessation of semaglutide and the initiation of alopecia management (consisting of intralesional corticosteroid injections, ketoconazole 2% shampoo, and minoxidil 5% topical solution), hair regrowth occurred.[14]Alzahrani, W.S., Bahkali, S.A., Alharthy, R.F., Alsabban, A.S. (2025). Alopecia Areata Following Semaglutide Treatment For Weight Loss: A Case Report. JAAD Case Reports. 63. 44-46. Available at: … Continue reading
Figure 2. Patch of hair loss following semaglutide treatment. Adapted from Figure 1.[15]Alzahrani, W.S., Bahkali, S.A., Alharthy, R.F., Alsabban, A.S. (2025). Alopecia Areata Following Semaglutide Treatment For Weight Loss: A Case Report. JAAD Case Reports. 63. 44-46. Available at: … Continue reading Image used under Creative Commons License.
An additional case study similarly showed a patch of hair loss after semaglutide treatment (2.4 mg weekly). An initial small patch occurred after only two days of treatment and had increased to 5 cm2 after 6 weeks. The alopecia had improved within two months of stopping semaglutide treatment.[16]Cheng, J.R., Zheng, J., Li, Y. (2022). Weight Loss-Associated Alopecia Areata. American Journal Of Therapeutics. Available at: https://doi.org/10.1097/MJT.0000000000001851
Study #1
The first study investigating the link between Ozempic and alopecia was published in 2025. They investigated cases of alopecia associated with semaglutide and tirzepatide (Mounjaro) reported via FAERS, the FDA Averse Event Reporting System, which collects reports of drug adverse events from both consumers (i.e., the general public) and healthcare professionals.[17]Godfrey, H., Leibovit-Reiben, Z., Jedlowski, P., Thiede, R. (2025). Alopecia Associated With The Use Of Semaglutide And Tirzepatide: A Disproportionality Analysis Using The FDA Adverse Event … Continue reading
Their search covered cases reported from 2022 to 2023. Within this time, there were 199 reports of alopecia for semaglutide, 179 reports for tirzepatide, and a lower number for other GLP-1 receptor agonists (GLP-1 RAs).
The researchers then carried out a disproportionality analysis, a type of statistical test that assesses whether side effects are reported more often in people using a drug than would occur in the general population. They demonstrated increased reporting odds, meaning that alopecia was reported more frequently in those taking semaglutide (or tirzepatide) than would be expected in the general population.
While this study highlights a potential association between semaglutide and alopecia, the methodology and type of analysis carried out cannot prove a direct, causal relationship.
In addition, the study was limited by the nature of FAERS, where the reporting of adverse events is voluntary. As such, the data can be influenced by heightened awareness of side effects, such as through media attention or exposure to reports on social media.
The researchers reported that 84% of the alopecia reports made to FAERS were from consumers and only 16% were from healthcare professionals. This makes it likely that a significant proportion of reports were made by people with no clinician-confirmed diagnosis of alopecia. Moreover, the type of hair loss was not reported in many cases, further limiting the conclusions that can be drawn from this study.
In summary, although providing evidence for a possible association between semaglutide and alopecia, further research is essential to determine whether semaglutide is directly affecting hair loss or whether other factors (such as weight loss, nutrient deficiencies, or underlying health conditions) are primarily at fault.
Study #2
A second study, also published in 2025, used retrospective analysis to investigate the link between GLP-1 RAs and alopecia in a real-world setting.[18]Burke, O., Sa, B., Alvarez Cespedes, D., Sechi, A., & Tosti, A. (2025). Glucagon-like peptide-1 receptor agonist medications and hair loss: a retrospective cohort study. Journal of the American … Continue reading They analyzed the medical records of 283 patients who were taking GLP-1 RAs and who had visited their dermatology department between 2021 and 2023.
Of these 283 patients, the majority did not experience any hair loss (84.1%). Three patients (1.2%) with no previous reports of hair loss experienced new-onset hair loss. This is approximately the same rate as would be expected in the general population over two years, and so does not indicate any major effects of GLP-1 RA treatment.
However, for those who had preexisting hair loss, the story is different. 32 people within the study had preexisting hair loss (approximately 13%), and of these people, over 90% reported that their hair loss had worsened since taking GLP-1 RAs. While this sounds conclusive and concerning, it should be noted that male pattern hair loss (androgenic alopecia; AGA) is a progressive condition where hair loss worsens over time. As such, it is difficult to separate normal hair loss over time from medication-induced hair loss.
On performing statistical analyses, no significant association was found between GLP-1 RAs and AGA; however, semaglutide did show a possible trend towards increased hair loss.
This study provides more supporting evidence towards a link between semaglutide and AGA; however, researchers conceded several limitations with their study. Similarly to the first study described above, this work was retrospective and lacked a control group. As such, it is not possible to prove a cause-and-effect relationship due to there being no way to compare the normal progression of AGA hair loss over time with drug-induced hair loss.
In addition, the duration of use of the GLP-1 RA medication was not recorded, nor was the reason for starting the medication. The study also did not use objective measures to clinically assess hair loss, such as hair counts, but relied on self-reported data from patients.
As with the previous study, this work provided some evidence supporting a link between semaglutide and hair loss, but larger, controlled clinical trials with objective hair measurements are essential to conclusively prove whether the drug is affecting hair growth.
Study #3
A third study demonstrated a similar conclusion to the first two studies. They carried out a retrospective analysis evaluating exposure to GLP-1 RAs and the risk of hair loss.[19]Akiska, Y.M., Vidal, S.I., Menta, N. (2025). Increased Incidence And Risk Of Hair Loss With Glucagon-Like Peptide 1 Receptor Agonists: A Real-World Multicentre Cohort Study. EMJ. 13(1). 52-54. … Continue reading They used the TriNetX US Collaborative Network, a real-world data network of healthcare organizations in the U.S. that pools electronic health record data. They matched cohorts of over 500,000 adults and demonstrated that GLP-1 RA exposure was significantly associated with an increased risk of AGA.
Again, this study was limited by its retrospective analysis and inability to prove causation, but it does provide further evidence linking GLP-1 RAs to hair loss.
How Might Ozempic Cause Hair Loss?
Data supporting a potential link between Ozempic and hair loss is growing, but what do scientists suggest is causing this link?
Rapid Weight Loss and Telogen Effluvium
Ozempic causes rapid weight loss due to appetite suppression, reduced food cravings, and an overall calorie reduction. When the body experiences a prolonged calorie deficit, it goes under a form of metabolic stress.
Metabolic stress caused by rapid weight loss is a well-documented trigger for a condition known as telogen effluvium. Hair goes through a growth cycle consisting of multiple phases, including the anagen phase (growth) and the telogen phase (resting). In a healthy scalp, approximately 85% of hair is in the anagen phase and 15% in the telogen phase.[20]Hughes, E.C., Syed, H.A., Saleh, D., (2025), Telogen Effluvium. Available at: http://www.ncbi.nlm.nih.gov/books/NBK430848/ (Accessed: 02 March 2026)
Telogen effluvium is a condition where a physiological stress (such as rapid weight loss) causes hairs to abruptly switch from the anagen to the telogen phase, resulting in increased hair shedding. The affected hairs are often diffuse throughout the head, resulting in hair thinning over the whole scalp, as opposed to patches of hair loss. The shedding often occurs several months after the initial stressor.
Rapid weight loss leading to telogen efluvium is a known phenomenon, having been identified in many weight loss situations, including crash dieting and bariatric surgery.[21]Kang, D.H., Kwon, S.H., Sim, W.Y., Lew, B.L. (2024). Telogen Effluvium Associated With Weight Loss: A Single Center Retrospective Study. Annals Of Dermatology. 36(6). 384-388. Available at: … Continue reading,[22]Zhang, W., Fan, M., Wang, C. (2021). Hair Loss After Metabolic And Bariatric Surgery: A Systematic Review And Meta-Analysis. Obesity Surgery. 31(6). 2649-2659. Available at: … Continue reading
Telogen effluvium is generally a temporary and reversible condition, where the removal of the trigger (i.e., sudden weight loss) causes a slowing in shedding and a gradual return of hair density.
Nutritional Deficiencies
Ozempic reduces appetite and drives the decrease in food eaten. People on Ozempic must be careful with their diet to ensure that they are still eating a balanced diet containing all of the necessary vitamins and minerals to remain healthy. Studies have shown that GLP-1 RA users often do not eat adequate amounts of protein and have insufficient intake of multiple key nutrients.[23]Johnson, B., Milstead, M., Thomas, O. (2025). Investigating Nutrient Intake During Use Of Glucagon-Like Peptide-1 Receptor Agonist: A Cross-Sectional Study. Frontiers In Nutrition. 12. 1566498. … Continue reading
Certain nutrients, such as protein, iron, and zinc, are essential for normal hair cycles and hair growth. Deficiencies in these important nutrients may increase the risk of hair loss.
More research is needed in this field to definitively determine whether nutritional deficiencies are responsible for Ozempic-induced hair loss.
Direct Effect on the Hair Growth Cycle
The majority of suggested mechanisms for Ozempic-induced hair loss are indirect effects, such as metabolic stress or nutritional deficiencies. However, it may be possible that Ozempic is directly influencing the hair growth cycle.
Research in animals has found that GLP-1 receptors can be found in the skin, localized around hair follicles. One study found that GLP-1 can activate a specific signalling pathway in skin cells, promoting cell proliferation. It is possible that this could affect hair growth, but this was not proven in the study.[24]List, J.F., He, H., Habener, J.F. (2006). Glucagon-Like Peptide-1 Receptor And Proglucagon Expression In Mouse Skin. Regulatory Peptides. 134(2). 149-157. Available at: … Continue reading
The evidence for GLP-1 receptor expression in human skin is severely limited, and there is no evidence that Ozempic directly affects the hair follicle and subsequent hair growth.
It may also be possible that Ozempic disrupts metabolic and hormonal signalling pathways, such as the insulin/IGF-1 pathway. Disturbance in pathways such as these may accelerate the onset or progression of follicle shrinkage and AGA in individuals who are already predisposed to AGA. However, this mechanism is purely speculative and has not been proven.
Distinguishing AGA From Telogen Effluvium
Hair loss of any type can be distressing. In the case of potential Ozempic-induced hair loss, the main types appear to be AGA and telogen effluvium (although there is limited evidence for either). But how can you tell the difference between the two?
AGA and telogen effluvium can have overlapping symptoms, but they differ in several key areas:
- Onset: Telogen effluvium is a sudden increase in shedding occurring approximately 2-3 months after a trigger (such as starting Ozempic), whereas AGA occurs gradually over years.
- Pattern: Telogen effluvium generally results in a diffuse reduction in hair density across the whole scalp, preserving the hairline. AGA occurs in a specific pattern, with bitemporal recession (receding hairline) and vertex thinning (a bald patch at the crown).
- Hair shaft thickness: The hairs shed in telogen effluvium mostly appear normal, with a regular shaft diameter. In AGA, the miniaturization of the hair follicle often results in thinner, vellus-like hairs.
It has been suggested that Ozempic and other GLP-1 RAs may reveal an underlying propensity to AGA and accelerate the hair loss progression. In addition, rapid weight loss has been shown to trigger temporary shedding (telogen effluvium). Ozempic users may experience either AGA or telogen effluvium, both concurrently, or neither.
Should You Be Concerned?
If you have no hair loss: probably not.
The current evidence suggests that those who have not experienced any hair loss in the past are at a low risk of experiencing hair loss on taking Ozempic. The induction of new-onset hair loss appears to be uncommon in the studies carried out on this phenomenon so far. As a result, the absolute risk of hair loss is low.
If you have AGA: potentially.
The evidence so far points towards people with existing pattern hair loss experiencing a worsening of the condition on taking GLP-1 RA medications, such as Ozempic. The exact mechanisms behind this are unclear, and a lack of placebo-controlled clinical trials limits our knowledge of whether any effects seen are simply the natural progression of AGA over time. Rapid weight loss and metabolic stress could also reveal previously unnoticed hair thinning.
If you have early or unnoticed AGA: potentially.
People with a genetic predisposition to male pattern hair loss may appear to experience a sudden, dramatic loss of hair. This may occur due to the combination of underlying AGA progression with telogen effluvium-induced shedding, making previous hair loss much more noticeable.
Practical Considerations
There are many good reasons to start taking Ozempic; however, the decision should be made with the full knowledge of potential side effects, including the possibility of hair loss.
Individuals starting Ozempic who are worried about hair loss should consider:
- Avoiding extreme calorie restriction.
- Ensuring that their diet still contains an adequate amount of protein, vitamins, and minerals, including iron and zinc.
- Carrying out a baseline scalp evaluation before starting Ozempic so that any hair loss can be identified.
- Monitoring shedding.
- Starting hair loss treatments, particularly if predisposed to AGA.
Managing hair loss at the same time as completing weight-loss treatments can help to reduce visible hair loss and maintain hair density. Hair loss treatments, such as topical minoxidil, are considered safe for both men and women and may be used alongside Ozempic without fear of drug interactions.
Other hair loss treatments, such as finasteride and dutasteride, may also be able to be used alongside Ozempic, although the additional safety concerns associated with these products may discourage use.
FAQs
Q: Does semaglutide cause hair loss?
A: Ozempic is the brand name for the drug semaglutide. The data discussed in this article applies to both.
Q: Does Wegovy cause hair loss?
A: Wegovy and Ozempic both contain semaglutide, but at different dosing ranges and in different formulations. The data discussed in this article can be applied to both.
Q: What should I do if I notice hair thinning while taking Ozempic?
A: Monitor how much you are shedding and speak to your doctor. Addressing underlying hair loss early may help, as you can start using hair loss treatments alongside Ozempic.
Q: Will losing weight quickly result in hair loss?
A: Rapid weight loss is known to result in telogen effluvium for some people. The faster the weight loss, the more likely the body is to go into metabolic stress, and the higher the chance of temporary hair shedding.
Final Thoughts
So, does Ozempic cause hair loss? In summary, we’re not sure, but potentially for some people.
Reports of hair loss associated with Ozempic are increasing, but the current scientific evidence does not provide any direct evidence supporting Ozempic-induced hair loss. The available data suggest a possible association, particularly in individuals genetically predisposed to AGA or who have already started to experience hair loss prior to taking Ozempic. However, the studies so far have all been retrospective, lacked a control group, and relied on self-reported outcomes, and as a result, have been unable to demonstrate causation.
For many users, any shedding experienced is likely linked to rapid weight loss, nutritional deficiencies, or the natural progression of existing AGA, not directly as a result of Ozempic use. If you already experience AGA, it may be a good idea to monitor hair loss after starting Ozempic and to consider preventative treatment options.
As the popularity of GLP-1 RAs continues to rise, more robust, controlled clinical trials will be needed to establish the true relationship between Ozempic and hair loss. In the meantime, awareness of the potential side effects and ongoing monitoring, rather than alarm, is the correct response.
References[+]
References ↑1 Montero, A., Sparks, G., Presiado, M., Hamel, L., (2024), KFF Health Tracking Poll May 2024: The Public’s Use And Views Of GLP-1 Drugs. Available at: https://www.kff.org/health-costs/kff-health-tracking-poll-may-2024-the-publics-use-and-views-of-glp-1-drugs/ (Accessed: 27 February 2026) ↑2 Future Market Insights Inc., (no date), Ozempic Market Share & Size 2025-2035. Available at: https://www.futuremarketinsights.com/reports/ozempic-market (Accessed: 27 February 2026) ↑3 Research And Markets, (2024), Global GLP-1 Receptor Agonist (Ozempic, Rybelsus, Trulicity, Saxenda, Wegovy, Victoza, Mounjaro, Zepbound) Market Analysis Report 2024-2030 Featuring Eli Lilly And C, Sanofi, Novo Nordisk, AstraZeneca. Available at: https://www.globenewswire.com/news-release/2024/07/17/2914462/28124/en/Global-GLP-1-Receptor-Agonist-Ozempic-Rybelsus-Trulicity-Saxenda-Wegovy-Victoza-Mounjaro-Zepbound-Market-Analysis-Report-2024-2030-Featuring-Eli-Lilly-and-C-Sanofi-Novo-Nordisk-Ast.html (Accessed: 27 February 2026) ↑4, ↑7, ↑10, ↑12 Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026) ↑5 Sorli, C., Harashima, S.I., Tsoukas, G.M. (2017). Efficacy And Safety Of Once-Weekly Semaglutide Monotherapy Versus Placebo In Patients With Type 2 Diabetes (SUSTAIN 1): A Double-Blind, Randomised, Placebo-Controlled, Parallel-Group, Multinational, Multicentre Phase 3a Trial. Lancet Diabetes And Endocrinology. 5(4). 251-260. Available at: https://doi.org/10.1016/S2213-8587(17)30013-X ↑6 Garvey, W.T., Batterham, R.L., Bhatta, M. (2022). Two-Year Effects Of Semaglutide In Adults With Overweight Or Obesity: The STEP 5 Trial. Nature Medicine. 28(10). 2083-2091. Available at: https://doi.org/10.1038/s41591-022-02026-4 ↑8 Holst, J.J. (2007). The Physiology Of Glucagon-Like Peptide 1. Physiological Reviews. 87(4). 1409-1439. Available at: https://doi.org/10.1152/physrev.00034.2006 ↑9 Drucker, D.J. (2022). GLP-1 Physiology Informs The Pharmacotherapy Of Obesity. Molecular Metabolism. 57. 101351. Available at: https://doi.org/10.1016/j.molmet.2021.101351 ↑11 Kommu, S., Berg, R.L. (2024). Efficacy And Safety Of Once-Weekly Subcutaneous Semaglutide On Weight Loss In Patients With Overweight Or Obesity Without Diabetes Mellitus-A Systematic Review And Meta-Analysis Of Randomized Controlled Trials. Obesity Reviews. 25(9). e13792. Available at: https://doi.org/10.1111/obr.13792 ↑13 Center For Drug Evaluation And Research, (2024), July – September 2023 | Potential Signals Of Serious Risks/New Safety Information Identified By The FDA Adverse Event Reporting System (FAERS). Available at: https://www.fda.gov/drugs/fdas-adverse-event-reporting-system-faers/july-september-2023-potential-signals-serious-risksnew-safety-information-identified-fda-adverse (Accessed: 02 March 2026) ↑14, ↑15 Alzahrani, W.S., Bahkali, S.A., Alharthy, R.F., Alsabban, A.S. (2025). Alopecia Areata Following Semaglutide Treatment For Weight Loss: A Case Report. JAAD Case Reports. 63. 44-46. Available at: https://doi.org/10.1016/j.jdcr.2025.06.012 ↑16 Cheng, J.R., Zheng, J., Li, Y. (2022). Weight Loss-Associated Alopecia Areata. American Journal Of Therapeutics. Available at: https://doi.org/10.1097/MJT.0000000000001851 ↑17 Godfrey, H., Leibovit-Reiben, Z., Jedlowski, P., Thiede, R. (2025). Alopecia Associated With The Use Of Semaglutide And Tirzepatide: A Disproportionality Analysis Using The FDA Adverse Event Reporting System (FAERS) From 2022 To 2023. Journal Of The European Academy Of Dermatology And Venereology. 39(2). e153. Available at: https://doi.org/10.1111/jdv.20197 ↑18 Burke, O., Sa, B., Alvarez Cespedes, D., Sechi, A., & Tosti, A. (2025). Glucagon-like peptide-1 receptor agonist medications and hair loss: a retrospective cohort study. Journal of the American Academy of Dermatology. 92(5). 1141–1143. Available at: https://doi.org/10.1016/j.jaad.2025.01.046 ↑19 Akiska, Y.M., Vidal, S.I., Menta, N. (2025). Increased Incidence And Risk Of Hair Loss With Glucagon-Like Peptide 1 Receptor Agonists: A Real-World Multicentre Cohort Study. EMJ. 13(1). 52-54. Available at: https://doi.org/10.33590/emjdermatol/TYEW1122 ↑20 Hughes, E.C., Syed, H.A., Saleh, D., (2025), Telogen Effluvium. Available at: http://www.ncbi.nlm.nih.gov/books/NBK430848/ (Accessed: 02 March 2026) ↑21 Kang, D.H., Kwon, S.H., Sim, W.Y., Lew, B.L. (2024). Telogen Effluvium Associated With Weight Loss: A Single Center Retrospective Study. Annals Of Dermatology. 36(6). 384-388. Available at: https://doi.org/10.5021/ad.24.043 ↑22 Zhang, W., Fan, M., Wang, C. (2021). Hair Loss After Metabolic And Bariatric Surgery: A Systematic Review And Meta-Analysis. Obesity Surgery. 31(6). 2649-2659. Available at: https://doi.org/10.1007/s11695-021-05311-2 ↑23 Johnson, B., Milstead, M., Thomas, O. (2025). Investigating Nutrient Intake During Use Of Glucagon-Like Peptide-1 Receptor Agonist: A Cross-Sectional Study. Frontiers In Nutrition. 12. 1566498. Available at: https://doi.org/10.3389/fnut.2025.1566498 ↑24 List, J.F., He, H., Habener, J.F. (2006). Glucagon-Like Peptide-1 Receptor And Proglucagon Expression In Mouse Skin. Regulatory Peptides. 134(2). 149-157. Available at: https://doi.org/10.1016/j.regpep.2006.02.007 Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreCatherine Kennedy, PhD
Catherine is a scientist and researcher with a background in biosciences and clinical neuroscience. She completed an integrated master’s degree in Biosciences (MBiol) before earning a PhD in Clinical Neurosciences at the University of Cambridge. Throughout her career, she has contributed to the scientific literature through the publication of numerous peer-reviewed research papers.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down