- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Best Topical Dutasteride: 8 Choices for 2026
-
10 Best Shampoos for Thinning Hair Men
-
Does Semaglutide Cause Hair Loss?
-
Does Dandruff Cause Hair Loss?
-
Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
-
Ulo Review: #1 Customized Hair Loss Treatment in 2026
-
10 Best Hair Loss Treatments for Men
-
Best Minoxidil for Men: Top Picks for 2026
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
Articles10 Best Hair Loss Treatments for Men
First Published May 8 2026Last Updated Apr 5 2026Pharmaceutical Researched & Written By:Sophie Grice, PhD
Researched & Written By:Sophie Grice, PhD Reviewed By:Rob English, Medical Editor
Reviewed By:Rob English, Medical Editor
Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
If you are at the start of your hair-loss journey, figuring out where to begin can feel overwhelming. There is no shortage of products and treatments that promise results, but not all of them are worth your time and money. In this article, we break down 10 of the best hair loss treatments for men, in no particular order. We explore the clinical evidence supporting them and explain the situations where each treatment tends to work best.
Full Article
When starting a hair growth journey, it’s easy to feel overwhelmed. A quick search online brings up hundreds of treatments. From prescription medications and over-the-counter solutions to devices, supplements, and medical procedures, all of which claim to regrow hair. With so many options, it’s hard to know which treatments are actually backed by scientific evidence and are worth spending your time and money on.
In reality, there is no single best hair loss treatment that works for everyone. That’s because male pattern hair loss does not look the same for everyone. It can vary widely in pattern, progression, and the underlying biology. Meaning some treatments tend to work better in certain situations than in others. Some are more effective for diffuse thinning, while others are better for localized recession. Some treatments are better for convenience, while others need consistency and long-term application to see results.
In this article, we break down 10 of the best hair loss treatments for men. We have not ranked them from best to worst because each works best in specific circumstances. We look into how each treatment works, the clinical evidence backing it, safety considerations, and who it’s most likely to work best for.
Oral Finasteride
Ideal For:
- Men looking for an FDA-approved treatment
- Men who prefer the convenience of taking one pill per day versus a topical application regimen.
- Men with diffuse thinning rather than localized thinning. With diffuse thinning, applying topical treatments evenly every day can be time-consuming and difficult compared to targeting a small area.
Finasteride is a prescription medication that treats androgenic alopecia (AGA) by inhibiting the enzyme 5ɑ-reductase (5AR), which converts testosterone into dihydrotestosterone (DHT). In people with AGA, DHT is elevated and binds to androgen receptors, which act as landing pads for DHT to have its effect. The binding of DHT to androgen receptors in the scalp leads to follicular miniaturization and leads to hair loss.[1]Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/
Finasteride works by selectively inhibiting type II 5AR. By blocking this enzyme, finasteride reduces the conversion of testosterone to DHT. DHT levels drop, and the hormonal process that causes pattern hair loss is disrupted, helping to maintain existing hair and induce hair regrowth.
Figure 1. Structure of finasteride.[2]Wikipedia, (2009), Finasteride. Available at: https://en.wikipedia.org/wiki/Finasteride#/media/File:Finasteride.svg (Accessed: 24 February 2026) Image in the public domain.
Clinical Evidence for Oral Finasteride
Oral finasteride is one of the most well-studied hair loss treatments and is one of two FDA-approved treatments for AGA. Over the years, there have been many clinical studies that demonstrate oral finasteride is able to slow hair loss and promote new hair growth in many patients. Large randomized placebo-controlled trials consistently show that daily oral finasteride leads to improvements in hair count, hair density, and participant satisfaction.
A notable trial involved 1,500 men with AGA. Of those treated with oral finasteride (1 mg), 48% were rated as improved based on standardized photographs after 12 months, compared to 7% in the placebo-treated group.[3]Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride … Continue reading Also, hair counts in the oral finasteride group increased or stabilized, whereas those in the placebo group experienced progressive hair loss.
The typical dose of oral finasteride for AGA is 1 mg. Increasing the dose to 5 mg does not tend to improve hair regrowth or further suppress scalp DHT in a meaningful way. For example, studies found scalp DHT reductions of 64% with 1 mg and 69% with 5 mg, which is a negligible difference.[4]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., … Continue reading
Lowering the dose to 0.2 mg daily shows promising results. In one 48-week trial, 54% of men who took 0.2 mg oral finasteride showed improvement, compared to 58% on 1 mg, suggesting only a marginal difference. That said, the relative percentage change in terminal hair counts was higher in the 1 mg versus 0.2 mg group.[5]Fertig, R.M., Caresse, G.A., Darwin, E., Gaudi, S. (2017). Sexual Side Effects Of 5-ɑ-Reductase Inhibitors Finasteride And Dutasteride: A Comprehensive Review. Dermatology Online Journal. 23(11). … Continue reading
If you’re looking for more information on how to take oral finasteride, read our article here.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Safe is Oral Finasteride?
Oral finasteride is generally well tolerated. However, as oral finasteride is ingested and reaches the systemic circulation, some people may experience sexual side effects, including reduced libido and erectile difficulties. Clinical trials suggest the risk is around 15% of users; however, these effects usually resolve after stopping medication.[6]Estill, M.C., Ford, A., Omeira, R., Rodman, M. (2023). Finasteride And Dutasteride For The Treatment Of Male Androgenetic Alopecia: A Review Of Efficacy And Reproductive Adverse Effects. Georgetown … Continue reading Oral finasteride may temporarily reduce sperm count or semen volume, but this is typically reversible.[7]Amory, J.K., Wang, C., Swerdloff, R.S., Anawalt, B.D., Matsumoto, A.M., Bremner, W.J., Walker, S.E., Haberer, L.J., Clark, R.V. (2007). The Effect Of 5ɑ-Reductase Inhibition With Dutasteride And … Continue reading Other potential side effects include breast tenderness and mood changes.
As with any medication, if you are considering finasteride, you should discuss potential risk factors and fertility plans with a clinician before beginning treatment and ensure you consult with a clinician if you experience any side effects.
If you’re a member and would like to find out more about oral finasteride, read our ultimate guide here.
Topical Minoxidil
Ideal For:
- Men who are looking for an over-the-counter, FDA-approved treatment for localized thinning.
- Men who are looking for a standard 5% minoxidil formulation or higher-strength, compounded formulations (e.g., 7%) if they have not responded to standard formulations.
- Men who are willing to consider combination therapies with tretinoin or microneedling.
Topical minoxidil at standard 2% and 5% concentrations is an over-the-counter, FDA-approved liquid or foam treatment. It can also be custom-compounded at higher concentrations from 7-15%, but this is not FDA-approved.
Topical minoxidil is a prodrug and is converted to its active form (minoxidil sulfate) by scalp enzymes called sulfotransferases, primarily the SULT1A1 enzyme. The activity of this enzyme can differ between people and in different regions of the scalp.
Minoxidil works by altering the hair cycle by shortening the resting (telogen) phase and promoting earlier entry into the growth (anagen) phase. This increases the proportion of follicles that actively produce hair. Minoxidil can also increase the size of miniaturized hair follicles, resulting in thicker and longer hair shafts.[8]Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: … Continue reading
Minoxidil can also increase local blood flow, activate the Wnt/β-catenin signaling pathway involved in follicular cell proliferation and differentiation, and has some cytoprotective and anti-inflammatory effects, which may increase prostaglandin E2 production.
Figure 2. Structure of minoxidil.[9]Wikipedia, (2015), Minoxidil Structure Svg. Available at: https://en.wikipedia.org/wiki/Minoxidil#/media/File:Minoxidil_structure.svg (Accessed: 20 February 2026) Image in the public domain.
Clinical Evidence for Topical Minoxidil
Topical minoxidil has consistently been shown in many randomized controlled trials to increase hair count, prolong the anagen phase, and improve short-term patient satisfaction, especially at a 5% concentration.[10]Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189
That said, despite strong early efficacy, after one year or longer, response rates tend to drop or plateau, particularly when used without adjunctive therapies like microneedling or anti-androgens.
Multiple studies report rates below 30%, and even lower in five-year follow-up populations, with only about 20-30% of users satisfied with their results.[11]Olsen, E.A., Weiner, M.S., Amara, I.A., DeLong, E.R. (1990). Five-Year Follow-Up Of Men With Androgenetic Alopecia Treated With Topical Minoxidil. Journal Of The American Academy Of Dermatology. … Continue reading In one trial, men using 5% minoxidil twice daily saw a nearly 60% increase in hair “weight” (total mass of regrown/thickened hair) at 6 months. By 96 weeks, gains had diminished to just 25% above the baseline, a significant drop from the initial response.[12]Price, V.H., Menefee, E., Strauss, P.C. (1999). Changes In Hair Weight And Hair Count In Men With Androgenetic Alopecia, After Application Of 5% And 2% Topical Minoxidil, Placebo, Or No Treatment. … Continue reading
In men with AGA, once-daily application of 5% topical minoxidil achieves hair count improvements comparable to twice-daily use of a 2% solution.[13]Olsen, E.A., Dunlap, F.E., Funicella, T., Koperski, J.A., Swinehart, J.M., Tschen, E.H., Trancik, R.J. (2002). A Randomized Clinical Trial Of 5% Topical Minoxidil Versus 2% Topical Minoxidil And … Continue reading Higher concentrations (7%) may offer incremental benefit in some non-responders. With 7% formulations showing modest additional improvement in some studies, but results are mixed, and overall evidence remains limited.[14]Singh, S., Patil, A., Kianfar, N., Waskiel-Burnat, A., Rudnicka, L., Sinclair, R., Goldust, M. (2022). Does Topical Minoxidil At Concentrations Higher Than 5% Provide Additional Clinical Benefit? … Continue reading
Data on 10-15% topical minoxidil are conflicting. Some studies report no meaningful advantage over 5%, while others report small gains in specific subgroups. However, adverse effects tend to increase at these higher concentrations.[15]Vasantha, K.L. (2025). Efficiency And Safety Of 10% Minoxidil In The Treatment Of Alopecia Areata: A Randomised Controlled Trial. Journal Of Population Therapeutics And Clinical Pharmacology. 32(4). … Continue reading,[16]Ghonemy, S., Alarawi, A., Bessar, H. (2021). Efficacy And Safety Of A New 10% Topical Minoxidil Versus 5% Topical Minoxidil And Placebo In The Treatment Of Male Androgenetic Alopecia: A Trichoscopic … Continue reading
Adjunctive strategies aimed at enhancing minoxidil activation and penetration have shown promising results. Anecdotal evidence from our PHH members suggests that prescription concentration of topical minoxidil (7%) in combination with enhancers like tretinoin often improves outcomes.
One landmark study reported a nearly fourfold greater increase in hair count (approximately a 40% gain) compared to minoxidil alone at 12 weeks, with participants experiencing actual visible improvements in density.[17]Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I.A., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating The Efficacy And Safety Of Combined … Continue reading
Topical retinoic acid (such as tretinoin) can also be combined with minoxidil to upregulate SULT1A1 activity. Thereby enhancing the local activation of minoxidil in the hair follicle. Peer-reviewed research indicates that applying topical tretinoin upregulates follicle sulfotransferase enzymes, thereby enhancing the response to minoxidil.[18]Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvan, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin Enhances Minoxidil Response In … Continue reading
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Safe is Topical Minoxidil?
Topical minoxidil is generally safe. As it is applied directly to the scalp, it has a low risk of systemic side effects because it is minimally absorbed into the bloodstream. The most common adverse reactions are local, such as scalp irritation, itching, and flaking. These effects are usually due to the presence of propylene glycol in liquid formulations, which is known to cause scalp irritation.[19]Patel, K., Palmer, A., Nixon, R. (2023). Allergic Contact Dermatitis From Propylene Glycol: A Case Series From Australia. Contact Dermatitis. 89(2). 79-84. Available at: … Continue reading Foam formulations tend to be propylene glycol-free and fast-drying.
More severe side effects, like hypertrichosis (unwanted hair growth outside the scalp) or cardiovascular symptoms, are rare and tend to be linked to misuse or highly sensitive individuals. Topical minoxidil is highly toxic to cats, so it should be handled with care by pet owners.
Learn more about topical minoxidil in our article here.
Topical Finasteride
Ideal For:
- Men with localized AGA who want the DHT-lowering benefits of finasteride but prefer a topical, scalp-focused approach.
- Men who have concerns about systemic side effects with oral finasteride or have previously experienced unwanted effects on oral therapy.
- Men seeking a more potent topical option than minoxidil alone, particularly in DHT-driven thinning at the frontal scalp or crown.
Topical finasteride has emerged as a potential alternative to oral finasteride for AGA. It works by the same mechanism as oral finasteride, blocking type II 5AR to prevent the conversion of testosterone to DHT. But the difference is that topical finasteride is applied directly to the scalp rather than ingested.
By delivering finasteride directly locally to the hair follicles, the aim is to reduce scalp DHT whilst limiting systemic absorption. The goal of this approach is to maintain the benefits for hair regrowth while lowering the risk of whole-body side effects.
You can find more information on how topical finasteride works in our article here.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Topical Finasteride
Clinical studies suggest that topical finasteride can improve hair parameters in men with AGA. One study reported that 1% topical finasteride showed comparable efficacy to 1 mg oral finasteride in terms of hair thickness, hair counts, and the size of the bald area.[20]Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of … Continue reading
In a randomized controlled trial carried out in men with AGA over 24 weeks, they found that topical finasteride (0.25%) significantly improved target area hair count compared to placebo.[21]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2021). Efficacy And Safety Of Topical Finasteride Spray … Continue reading
How Safe is Topical Finasteride?
Topical finasteride has a different safety profile than oral finasteride. Studies show that while some systemic absorption still occurs, circulating finasteride and DHT changes are substantially smaller than with oral finasteride.[22]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2022). Efficacy And Safety Of Topical Finasteride Spray … Continue reading
But systemic effects are still possible. A 2025 FDA pharmacovigilance review of adverse event reports submitted between 2019 and 2024 identified 32 cases involving topical finasteride formulations. Most of these cases involved the use of compounded products. Reported symptoms included erectile dysfunction, anxiety, brain fog, depression, fatigue, insomnia, decreased libido, and testicular pain.[23]US Food and Drug Administration. (2025). FDA alerts health care providers, compounders and consumers of potential risks associated with compounded topical finasteride products. FDA. Available at: … Continue reading
That said, the most common side effects associated with topical finasteride are local skin reactions. A 2018 systematic review noted reports of erythema, contact dermatitis, and scalp irritation in several studies; but serious cutaneous adverse events were absent.[24]Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading In many cases, scalp irritation is thought to be related to the formulation itself, particularly ingredients like propylene glycol.
If you’re a member, you can find out more about topical finasteride in our ultimate guide here.
Oral Dutasteride
Ideal For:
- Men who are willing to consider a stronger systemic therapy with more extensive or diffuse AGA.
- Men with progressive thinning across the scalp rather than isolated areas.
- Men who are looking for the next step if they have not achieved their desired results, or whose results have plateaued with finasteride.
So we know that DHT drives follicle miniaturization, and testosterone is converted to DHT by 5AR. But there are two types of 5AR: type I and type II.
This is where dutasteride and finasteride differ. Finasteride only blocks type II 5AR, whereas dutasteride can block both type I and type II 5AR. Because dutasteride blocks both types of 5AR, it suppresses DHT more strongly than finasteride. Oral dutasteride is FDA-approved for benign prostatic hyperplasia, but is commonly prescribed off-label for hair loss.
Figure 3. Structure of dutasteride.[25]Wikipedia, (2009), Dutasteride. Available at: https://en.wikipedia.org/wiki/Dutasteride#/media/File:Dutasteride.svg (Accessed: 24 February 2026) Image in the public domain.
Clinical Evidence for Oral Dutasteride
Across multiple studies, dutasteride has been shown to consistently outperform finasteride on hair parameters. In one notable study carried out in men with AGA over 24 weeks, 0.5 mg dutasteride significantly increased total hair count and decreased thin hair count per cm2 compared to 1 mg finasteride.[26]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading
Notably, another study carried out in men with AGA over 24 weeks, showed that 0.5 mg dutasteride significantly increased terminal hair count and hair width compared to 1 mg finasteride.[27]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading
Additionally, across studies, investigators were more likely to rate dutasteride users as “improved” or “markedly improved” compared to those receiving finasteride.[28]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Safe is Oral Dutasteride?
Oral dutasteride is generally well-tolerated by many users. But some people may experience the following side effects:
- Sexual side effects: Reduced libido, erectile dysfunction, or ejaculatory disorders (reduced volume or delayed ejaculation) may occur. These effects are often dependent on the dose and usually improve after stopping treatment.[29]Choi, G.S., Sim, W.Y., Kang, H., Huh, C.H., Lee, Y.W., Shantakumar, S., Ho, Y.F., Oh, E.J., Duh, M.S., Cheng, W.Y., Bobbili, P., Thompson-Leduc, P., Ong, G. (2022). Long-Term Effectiveness And Safety … Continue reading
- Mood changes: Low mood or depressive symptoms, increased anxiety, or lack of motivation. These are uncommon, but any mood changes should be taken seriously if they do occur.[30]Hirshburg, J.M., Kelsey, P.A., Therrien, C.A., Gavino, A.C., Reichenberg, J.S. (2016). Adverse Effects And Safety Of 5-Alpha Reductase Inhibitors (Finasteride, Dutasteride): A Systematic Review. … Continue reading
- Breast changes: Hormonal shifts may lead to breast tenderness or enlargement (gynecomastia), which warrants medical review.[31]Marihart, S., Harik, M., Djavan, B. (2005). Dutasteride: A Review Of Current Data On A Novel Dual Inhibitor Of 5alpha Reductase. Reviews In Urology. 7(4). 203-210
- Prostate-specific antigen (PSA): Dutasteride may lower PSA, which could mask early signs of prostate cancer.[32]Marihart, S., Harik, M., Djavan, B. (2005). Dutasteride: A Review Of Current Data On A Novel Dual Inhibitor Of 5alpha Reductase. Reviews In Urology. 7(4). 203-210
Check out our article here on oral dutasteride to find out more about side effects, dosing, and how it compares to finasteride.
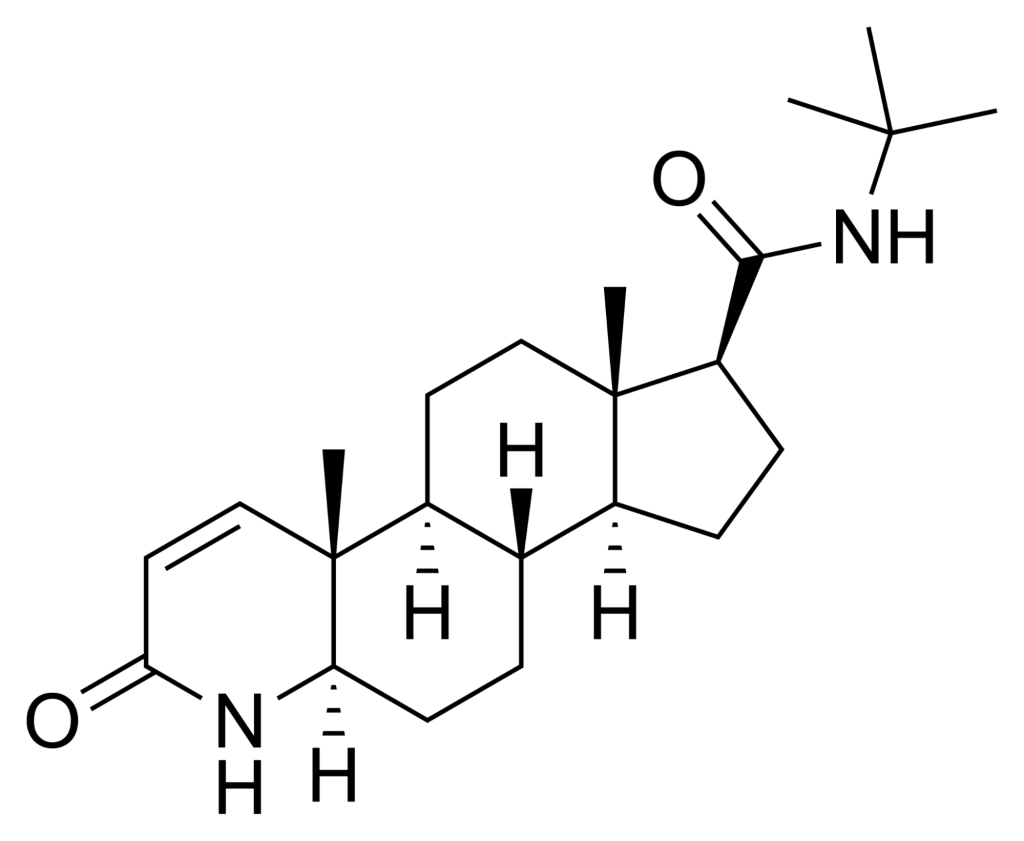
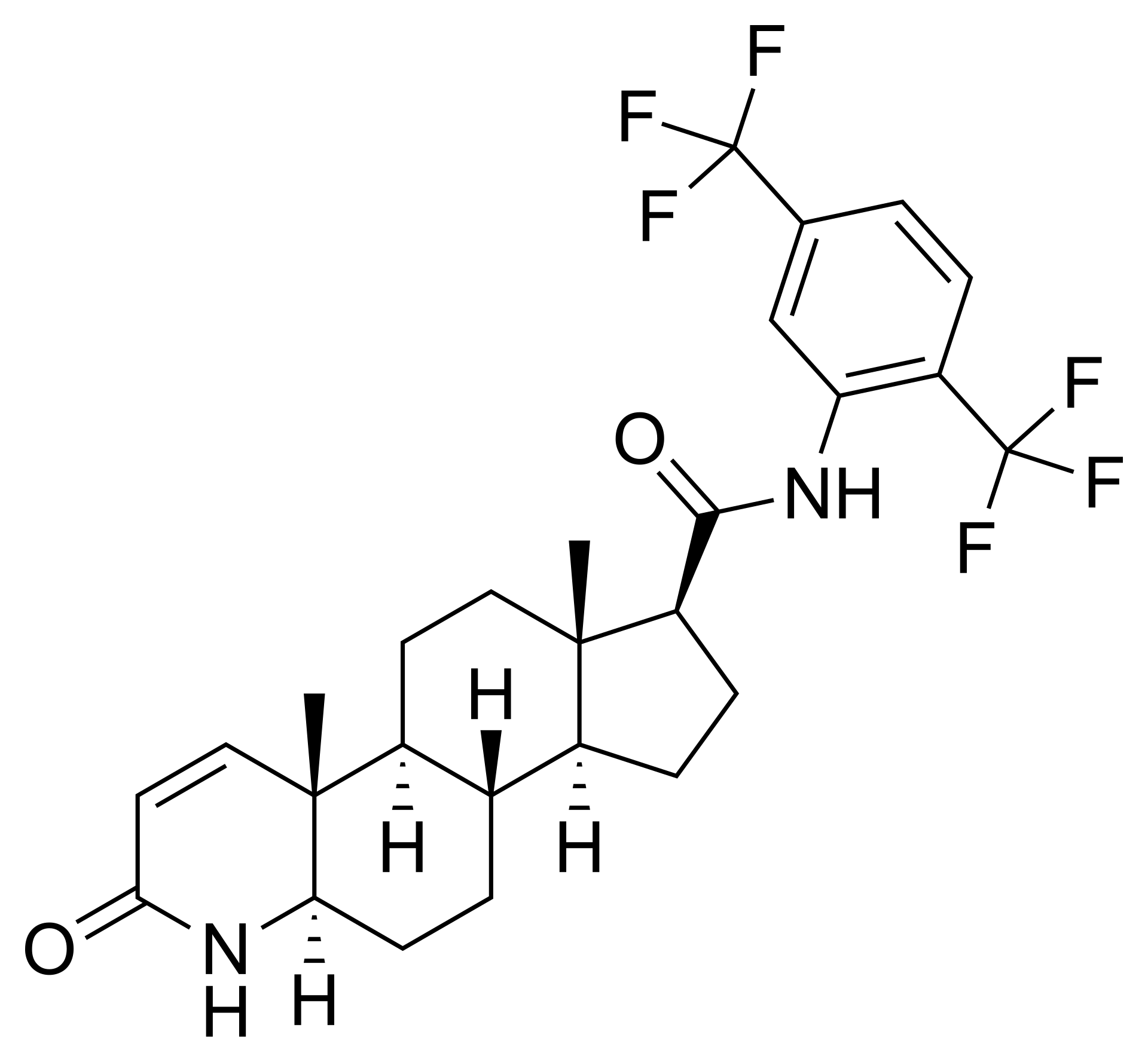
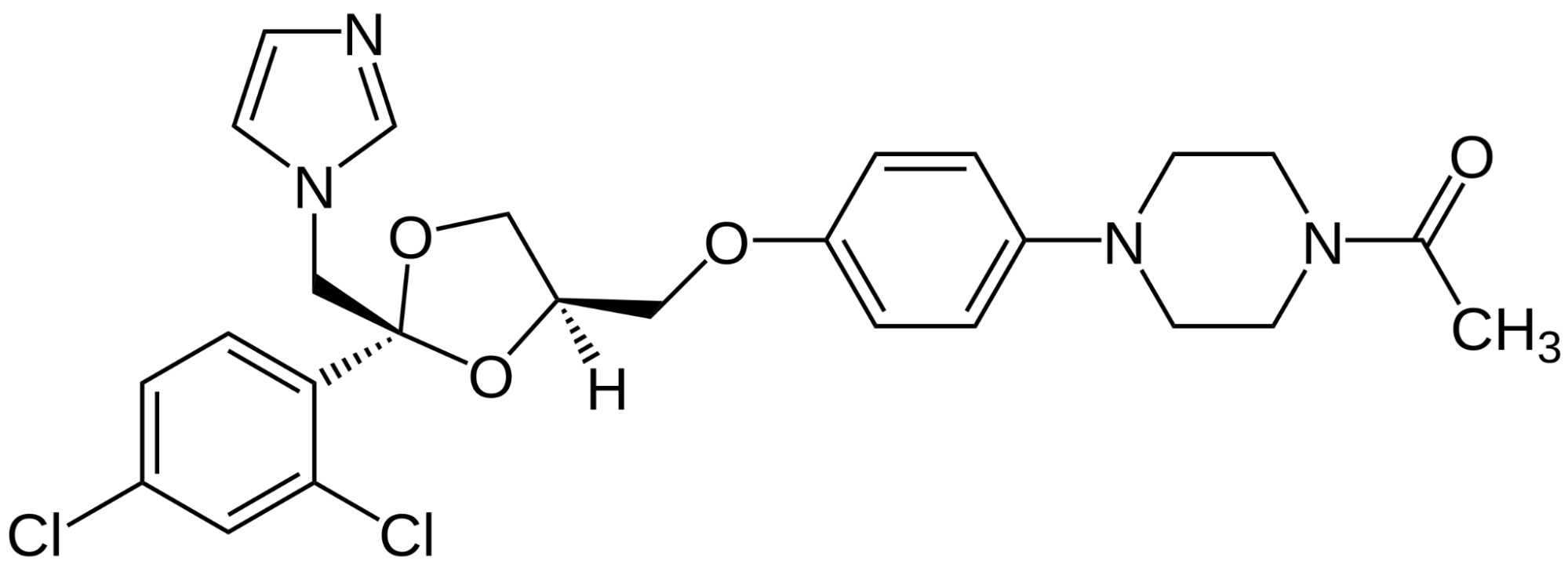
Topical Dutasteride
Ideal For:
- Men with AGA and localized thinning (e.g., the crown or frontal scalp).
- Men who have experienced side effects with oral dutasteride or are concerned about systemic effects.
- Men who are looking for a potentially stronger topical DHT-targeting option than finasteride.
- Men who are comfortable using a newer treatment that has relatively limited clinical evidence.
Topical dutasteride is another emerging therapy that works by the same mechanism as oral dutasteride, inhibiting both type I and type II 5AR to prevent the conversion of testosterone to DHT.
The difference is that topical dutasteride is applied directly to the scalp, with the goal of limiting systemic side effects. Topical dutasteride is not FDA-approved and is usually obtained through compounding pharmacies.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Topical Dutasteride
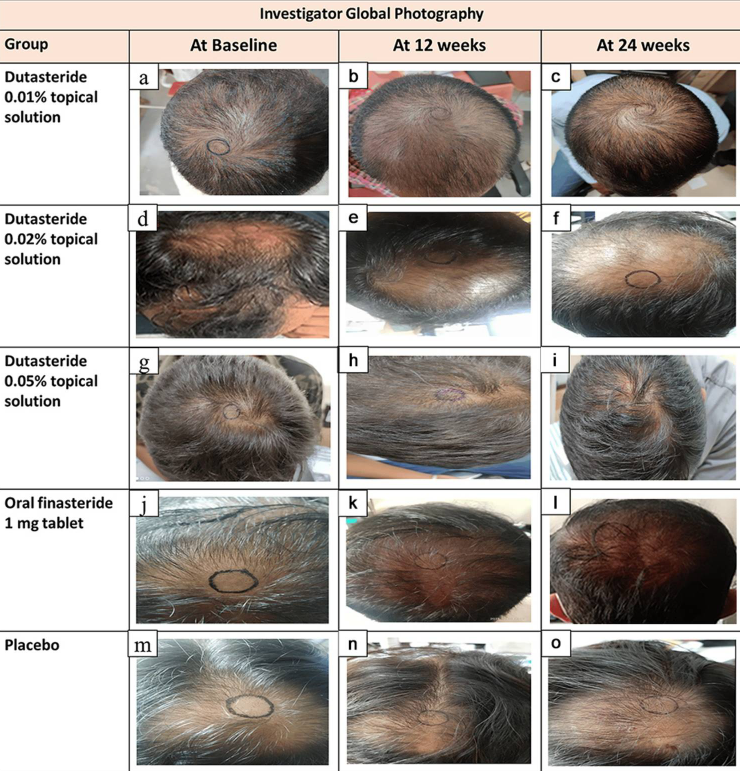
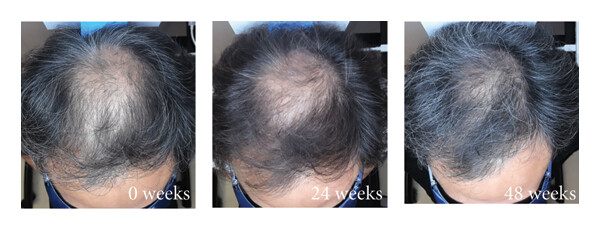
Clinical evidence for topical dutasteride is still limited compared with oral therapies. But, at the end of 2025, the first RCT involving 135 men with AGA comparing 1 mg oral finasteride to (0.01-0.05%) topical dutasteride over 24 weeks was published.[33]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading
In the study, several hair parameters appeared to improve more in the topical dutasteride (0.05%) group than in those receiving oral finasteride (1 mg). The authors reported an average increase of 75 hairs per cm² with topical dutasteride compared to 41 hairs per cm² with oral finasteride.
Additionally, 69% of participants using topical dutasteride were rated as “improved” on global photographic assessment, compared with 21% in the oral finasteride group.
But it’s important to note that we are a little skeptical of the results from this study. This is because the baseline hair counts are suspicious, and the measurement circle clearly moves in location, size, and shape.
Figure 4. Representative hair growth images. Adapted from Figure 2.[34]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading Image used under Creative Commons License
If you’d like to learn more about our take on this study, read our article here.
How Safe is Topical Dutasteride?
In this same study, systemic hormone changes with topical dutasteride were minimal. Plasma dutasteride levels were mostly near the quantification limit, but some samples spiked to 2,555 pg/mL, and dutasteride remained detectable at day 168 while participants were still receiving treatment. This suggests that average exposure was low, but absorption may be unpredictable in some people.
Because topical dutasteride is applied directly to the scalp, it may have fewer side effects than oral dutasteride. The most common side effects are local scalp reactions, including redness, itching, flaking, or contact dermatitis.
This is usually related to the carrier solution rather than the dutasteride itself. Switching carriers or adjusting how often it is applied often helps these symptoms.[35]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading
If you’re interested in finding out more about how topical dutasteride works, have a read of our article here.
Oral Minoxidil
Ideal For:
- Men who have experienced side effects from finasteride or dutasteride.
- Men who prefer the convenience of a once-daily pill over topical application.
- Men who have struggled with adherence to topical treatments due to cosmetic residue, scalp irritation, or lifestyle factors.
- Men who have not responded well to topical minoxidil, including those with poor scalp absorption or SULT1A1 activity.
Oral minoxidil is a prescription medication that was originally used to treat high blood pressure. At lower doses (0.25-5 mg daily), it’s prescribed off-label to treat hair loss, especially in people who don’t respond well to topical minoxidil or struggle with applying topical treatments consistently.
Oral minoxidil supports regrowth by improving blood flow around follicles, shifting more hairs into the growth (anagen) phase, and activating follicular growth pathways.
Some people prefer oral minoxidil over topical due to the key differences in how each one is activated and delivered to the hair follicles. Topical minoxidil is a pro-drug that needs conversion to its active form by SULT1A1 enzymes located in scalp hair follicles.[36]Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A … Continue reading
The activity of this enzyme varies significantly between people. Also, topical minoxidil faces a penetration problem where only about 1.4% of the drug applied is actually absorbed into the scalp skin under normal conditions.[37]Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil And Its Use In Hair Disorders: A Review. Drug Design, Development And Therapy. 13. 2777-2786. Available at: … Continue reading
Oral minoxidil helps to bypass these limitations. Oral minoxidil is absorbed through the gastrointestinal tract and converted to its active form in the liver, where sulfotransferase activity is abundant.
This ensures that nearly all minoxidil that is ingested is activated and delivered systemically to the hair follicles throughout the scalp and body, regardless of individual differences in scalp enzyme activity. As a result, oral minoxidil can be effective even for people who have not responded to topical minoxidil due to low scalp sulfotransferase activity or poor drug penetration.[38]Beach, R.A. (2018). Case Series Of Oral Minoxidil For Androgenetic And Traction Alopecia: Tolerability & The Five C’s Of Oral Therapy. Dermatologic Therapy. 31(6). E12707. Available at: … Continue reading
Interested in Oral Minoxidil?
Low-dose oral minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Oral Minoxidil
One study in men with AGA reported a 100% response rate with 5 mg daily oral minoxidil, alongside an average 19% increase in hair count at 24 weeks.[39]Lueangarun, S., Panchaprateep, R., Tempark, T., Noppakun, N. (2015). Efficacy And Safety Of Oral Minoxidil 5 Mg Daily During 24-Week Treatment In Male Androgenetic Alopecia. Journal Of The American … Continue reading Additionally, a retrospective study showed a 90% response rate at 6-12 months for men taking 2.5-5 mg of oral minoxidil daily. Notably, around 60% of these men had tried other treatments for AGA, but had previously quit due to side effects or lack of efficacy.[40]Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral … Continue reading
Lower doses of oral minoxidil may still be effective in stabilizing progression. In one study, 0.25 mg minoxidil achieved a response rate of 60% in men with AGA. That’s lower than what is typically seen at 2.5-5 mg concentrations, but it is favourable nevertheless.[41]Pirmez, R., Salas-Callo, C.I. (2020). Very-Low-Dose Oral Minoxidil In Male Androgenetic Alopecia: A Study With Quantitative Trichoscopic Documentation. Journal Of The American Academy Of Dermatology. … Continue reading
How Safe is Oral Minoxidil?
At low doses, oral minoxidil is generally well tolerated, but side effects are more common than with topical minoxidil because the drug circulates systemically.[42]Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair … Continue reading
The most common side effects include hypertrichosis, mild fluid retention or ankle swelling, and lightheadedness, particularly when starting or increasing the dose.
Less commonly, oral minoxidil can affect heart rate or cardiac function. Rare but serious events (e.g., pericardial effusion) have been reported, particularly in people with underlying cardiovascular or renal issues. Most side effects are dependent on the dose and often improve with dose reduction or discontinuation.
If you’re considering starting oral minoxidil treatment, start with a lower dose and only increase gradually if it is needed. Splitting the dose between morning and evening can improve tolerability for some. If results plateau, oral minoxidil is often combined with other therapies such as microneedling or topical agents rather than increasing the dose alone.
Oral minoxidil should be used under medical supervision, especially in anyone with cardiovascular, renal, or hepatic concerns.
If you are a member and would like to learn more about oral minoxidil, read our ultimate guide here.
Low-Level Laser Therapy (LLLT)
Ideal For:
- Men with early AGA who are looking to avoid pharmaceutical treatments.
- Men who are willing to be consistent with long-term device use.
- Men willing to pay the costs for device use, either at home or through visits to a clinic.
- Men who are willing to try more experimental methods.
- Men who are willing to try combination treatments, as LLLT will most likely have minimal effects as a standalone solution.
LLLT is therapeutic exposure to red or near-infrared radiation. For hair loss, this involves placing the scalp under infrared-emitting laser diodes. A typical laser therapy session lasts from 20 to 60 minutes, and you can complete them at a doctor’s office, with a take-home laser helmet, or even with a laser brush.
Laser diodes for hair loss emit red light at wavelengths of 630-670 nanometers. The sun emits wavelengths from 250 to 2,500 nanometers. LLLT is essentially exposure to one part of the sun’s spectrum for a controlled period of time. It turns out that some wavelengths show benefits to human physiology, from wound healing to inflammation resolution and even hair recovery.
Currently, there is still speculation about how LLLT actually works for hair regrowth. Potential explanations include:
- The laser’s heat activates heat shock proteins.[43]Wikramanayake, T.C., Rodriguez, R., Choudhary, S., Mauro, L.M., Nouri, K., Schachner, L.A., Jimenez, J.J. (2012). Effects of the Lexington LaserComb on Hair Regrowth in the C3H/HeJ Mouse Model of … Continue reading
- The laser’s light increases tissue oxygenation.[44]Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts and Bolts of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available … Continue reading
- The laser’s light increases blood flow.[45]Makihara, E., Masumi, S. (2008). Blood Flow Changes of a Superficial Temporal Artery Before and After Low-Level Laser Irradiation Applied to the Temporomandibular Joint Area. Nihon Hotetsu Shika … Continue reading
- The laser’s light generates acute tissue inflammation.[46]Bouzari, N., Firooz, A.R. (2006). Lasers May Induce Terminal Hair Growth. Dermatologic Surgery. 32(3). 460. Available at: https://doi.org/10.1111/j.1524-4725.2006.32092.x
- The laser’s light can inhibit 5AR.
Clinical Evidence for Low-Level Laser Therapy
Clinical trials of LLLT devices for AGA generally report modest improvements in hair density versus placebo (sham) devices. A systematic review and meta-analysis of seven randomized controlled trials using FDA-approved home-use LLLT devices found significantly greater increases in hair density in treated participants compared with sham controls in both men and women, particularly with helmet and comb-type devices.[47]Lueangarun, S., Visutjindaporn, P., Parcharoen, Y., Jamparuang, P., Tempark, T. (2021). A Systematic Review and Meta-Analysis of Randomized Controlled Trials of United States Food and Drug … Continue reading
In a recently published study, they investigated the effects of using a helmet-type LLLT device (646-675 nm) for 20 mins, 3 times per week, on AGA in both men and women (51 men, 17 women) over 48 weeks.
They found that hair density significantly increased from baseline from 99.2 to 124.2 hairs/cm2, and mean hair shaft thickness increased from 65.1 to 74.9 μm. Also, 59% of participants were rated as improved based on global photographs, and over 85% expressed satisfaction with treatment.[48]Shin, J.W., Paik, K., Na, J.I., Lew, B.L., Huh, C.H. (2026). Long-Term Efficacy and Safety of Low-Level Laser Therapy for Androgenetic Alopecia: A 12-Month Prospective Trial. Dermatologic Therapy. … Continue reading
Figure 5. Investigator-assessed clinical improvement over time after use of an LLLT device. Adapted from Figure 3a.[49]Shin, J.W., Paik, K., Na, J.I., Lew, B.L., Huh, C.H. (2026). Long-Term Efficacy and Safety of Low-Level Laser Therapy for Androgenetic Alopecia: A 12-Month Prospective Trial. Dermatologic Therapy. … Continue reading Used under Creative Commons license.
However, based on anecdotal evidence from our members, we have not seen a single case where LLLT has worked, so take this information with a pinch of salt.
There is also a broader methodological issue with LLLT device trials that is worth noting. Human hair growth shows seasonal variation, with increased telogen shedding often happening in late summer and early autumn in the Northern Hemisphere.
Because many LLLT devices emit wavelengths that overlap with natural sunlight exposure, trial timing across this seasonal window could theoretically amplify any differences seen between LLLT devices and sham devices if light exposure modifies the dynamics of seasonal shedding.
This does not invalidate the clinical evidence, but it highlights how environmental biology can also interact with LLLT trials in ways that may exaggerate benefits.
If you’re a member and would like more info on this topic, watch our video here.
How Safe is LLLT?
LLLT is generally safe and well-tolerated for most people. Most effects are mild and localized to the scalp, including scalp warmth, redness, or itching during or after use.
Find out more about LLLT in our article here.
Microneedling
Ideal For:
- Men who are willing to be consistent with use.
- Men who are looking to boost the impact of topical treatment like minoxidil, especially if they’ve seen plateauing results or suspect poor topical response.
Microneedling, also known as percutaneous wound induction, is the act of deliberately wounding skin tissues for therapeutic benefit. This is generally done with hundreds of tiny, medical-grade needles ranging in length from 0.1 to 5.0mm.
There are a variety of microneedling devices, from hand-held rollers to automated pens to stamps. But the ones people are most familiar with (in the hair loss world) are dermarollers. When rolled against the skin, they puncture its top layers to evoke acute inflammation. And yes, being rolled with essentially a tiny lint roller covered in spikes does hurt.
There are multiple possible explanations as to how microneedling may improve AGA:
- Micro-wounding may increase proteins and growth factors linked to hair growth.
- Micro-wounding might downregulate proteins linked to scarring.
- Micro-wounding might promote angiogenesis (new blood vessel formation).
- Microneedling may improve the absorption/efficacy of other hair loss treatments.
Clinical Evidence for Microneedling
There are many before-and-after photos online from microneedling regimens. If you’d like to see these photos, a user at HairLossTalk has done a great job accumulating them, which you can view here.[50]HairLossTalk, (2019), Microneedling Photo Results Summary. Available at: https://www.hairlosstalk.com/interact/threads/microneedling-photo-results-summary.121072/ (Accessed: 24 February 2026)
But the more useful question is what the clinical literature shows. Overall, microneedling appears to improve outcomes when combined with other therapies, such as topical minoxidil, finasteride, or platelet-rich plasma (PRP).
A systematic review and meta-analysis of 13 randomized controlled trials showed that microneedling in combination with other treatments resulted in greater improvements in hair density and diameter compared to treatments used alone.[51]Pei, D., Zeng, L., Huang, X., Wang, B., Liu, L., Zhang, G. (2024). Efficacy and Safety of Combined Microneedling Therapy for Androgenic Alopecia: A Systematic Review and Meta-Analysis of Randomized … Continue reading
Notably, over 12 weeks, once-weekly microneedling plus twice-daily minoxidil increased hair counts by 4X compared with minoxidil alone.[52]Dhurat, R., Sukesh, M., Avhad, G., Dandale, A., Pal, A., Pund, P. (2013). A Randomized Evaluator Blinded Study of Effect of Microneedling in Androgenetic Alopecia: A Pilot Study. International … Continue reading Also over 6 months, once-weekly microneedling alongside daily finasteride and 5% minoxidil resulted in a +2 or +3 improvement in hair loss gradient for non-responders to both finasteride and minoxidil.[53]Dhurat, R., Mathapati, S. (2015). Response to Microneedling Treatment in Men with Androgenetic Alopecia Who Failed to Respond to Conventional Therapy. Indian Journal of Dermatology. 60(3). 260-263. … Continue reading
There is also evidence that microneedling may have a standalone benefit. In one 24-week study, microneedling carried out every 14 days produced greater increases in hair density than topical minoxidil alone (23.4 vs 18.8 hairs/cm² from baseline).[54]Bao, L., Gong, L., Guo, M., Liu, T., Shi, A., Zong, H. (2020). Randomized Trial of Electrodynamic Microneedle Combined with 5% Minoxidil Topical Solution for the Treatment of Chinese Male … Continue reading
Ultimately, microneedling tends to work best as an adjunct (especially with minoxidil), but it may still offer meaningful benefit as a standalone option for some people.
The choice of needle length also matters. In general, we currently recommend sticking with 0.6-0.8mm needle lengths for automated pens, and 1.0-1.5mm needle lengths for rollers, needling once weekly, or once every two weeks.
To find out more about the best microneedling length, read our article here.
How Safe is Microneedling?
In short-term studies, microneedling is generally safe. The most common side effects are mild and localized, including redness, swelling, itching, pinpoint bleeding, and discomfort during treatment.
Some people also notice temporary flaking in the days after treatment. Scarring is rare when users use a sensible needle depth and avoid overdoing it. But anyone prone to keloid scarring should be cautious.
You can find more information on microneedling and scarring in our article here.
The risk of infection is low with basic hygiene (e.g., clean scalp, clean hands, and proper device sterilization). As a general safety rule, sticking to needle lengths of 2.0 mm or less and avoiding excessive session frequency reduces risk.
If you’re a member, you can learn more about microneedling in our ultimate guide here.
Ketoconazole
Ideal For:
- Men with AGA who also have dandruff, seborrheic dermatitis, oily scalp, or scalp inflammation, as these factors can worsen follicle miniaturization.
- Men with AGA alone, or AGA plus seborrheic dermatitis or telogen effluvium.
- Men with hair shedding disorders where microbial overgrowth or scalp inflammation contributes to shedding may particularly benefit from ketoconazole due to its antifungal and anti-inflammatory effects.
You might have heard about ketoconazole under the brand name Nizoral®, and you might know about it as a shampoo. Ketoconazole is an antifungal medication used to help improve fungal-related conditions such as dandruff, fungal infections, certain hormone-linked diseases, and even hair loss from fungal and non-fungal causes.
Figure 7. Structure of ketoconazole. Adapted from Wikimedia.[55]Wikimedia Commons, (2025), Ketoconazole Chemical Structure (SVG). Available at: https://commons.wikimedia.org/wiki/Category:Ketoconazole#/media/File:(2S,4R)-Ketoconazol.svg (Accessed: 24 February … Continue reading Image in the public domain.
Ketoconazole may improve telogen effluvium by killing scalp pathogens, thereby normalizing excessive hair shedding triggered by microorganism-mediated inflammation. It may also prolong the anagen phase of the hair cycle.
Ketoconazole may improve AGA via three key mechanisms:
- Killing pathogens in the scalp, therefore normalizing excessive hair shedding triggered by microorganism-mediated inflammation.
- Reducing scalp DHT levels by inhibiting 5-alpha reductase (a key enzyme that converts testosterone to DHT) and blocking androgen receptors (“landing pads” for DHT to exert its effects).
- Prolonging the anagen phase of the hair cycle.
You can buy 1% ketoconazole shampoo over the counter, typically at around $16 for 200 mL. Whereas, 2% ketoconazole shampoo usually requires a prescription.
Figure 8. Image of Nizoral shampoo, containing 1% ketoconazole. Available at Walmart.
Clinical Evidence for Ketoconazole
Clinical studies on ketoconazole shampoo, especially 2% strength, provide evidence that ketoconazole can improve hair parameters in men with AGA, including hair density, shaft diameter, and the proportion of hairs in the anagen phase.
In one study involving men with AGA comparing 2% ketoconazole shampoo with a non-medicated shampoo over 21 months, ketoconazole showed progressive increases in a composite hair growth index (anagen percentage × shaft diameter). Those using standard shampoo experienced a gradual decline.[56]Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: … Continue reading
Also, a pilot study showed that 2% ketoconazole may produce hair shaft diameter improvements similar to 2% minoxidil, with reductions in sebaceous gland size, which was not seen in the minoxidil-only group.[57]Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: … Continue reading
Evidence for 1% ketoconazole is weaker but still supportive. In men with AGA associated with dandruff or telogen effluvium, 1% ketoconazole used 2 to 3 times per week reduced hair shedding and increased anagen hair proportion and shaft diameter over 6 months.[58]Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine … Continue reading
Ketoconazole also tends to work well when used in combination with other treatments. In one study, finasteride plus ketoconazole demonstrated improvements in self- and physician-assessments.[59]Khandpur, S., Suman, M., Reddy, B.S. (2002). Comparative Efficacy Of Various Treatment Regimens For Androgenetic Alopecia In Men. J Dermatol. 29(8). 489-98. Available at: … Continue reading
Based on the available clinical evidence, there is stronger support for the use of 2% ketoconazole shampoo for hair growth than for 1%. But, as there are currently no direct head-to-head studies comparing the effectiveness of 1% ketoconazole shampoo versus 2% for hair regrowth, this is purely speculative.
How Safe is Ketoconazole?
Ketoconazole shampoo is generally safe and well-tolerated when used as directed. Because it is applied briefly to the scalp and then rinsed off, systemic absorption is minimal, and side effects are usually limited to mild, local scalp irritation or dryness.
Leave-on topical ketoconazole is available in some countries, but this increases the risk of systemic exposure. These non-shampoo formulations have not been studied for hair loss in humans. So even though in theory topical ketoconazole may seem more effective, it’s sensible to stick with ketoconazole shampoo, which has science-backed evidence.
Oral ketoconazole has significant systemic risks, but these concerns do not apply to shampoo use due to the much lower exposure. Overall, 2% ketoconazole shampoo has a favourable safety profile when used a few times per week.
If you’re a member, you can find out more information on ketoconazole in our ultimate guide here.
Saw Palmetto
Ideal For:
- Men with early to mid-stage AGA.
- Men who want to avoid pharmaceutical options.
- Men who are willing to try combinational treatments.
Saw palmetto (Serenoa repens) is a palm plant native to the Southeastern United States. It grows up to 10 feet tall and can live over 700 years. It’s a botanical extract used for hair loss because it can reduce 5AR activity and may lower DHT signalling.
Saw palmetto is different from finasteride and dutasteride as it is not a standardized pharmaceutical. So its strength can vary across products depending on how it is harvested and extracted.
Saw palmetto is available in both oral and topical formulations (serums/lotions/shampoos).
All-Natural Hair Topical
The top natural ingredients for hair growth, all in one serum.
Take the next step in your hair growth journey with a world-class natural serum. Ingredients, doses, & concentrations built by science.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Saw Palmetto
Overall, the clinical evidence for saw palmetto is mixed and tends to be of lower quality than the evidence that is available for pharmaceutical treatments like finasteride, minoxidil, or dutasteride.
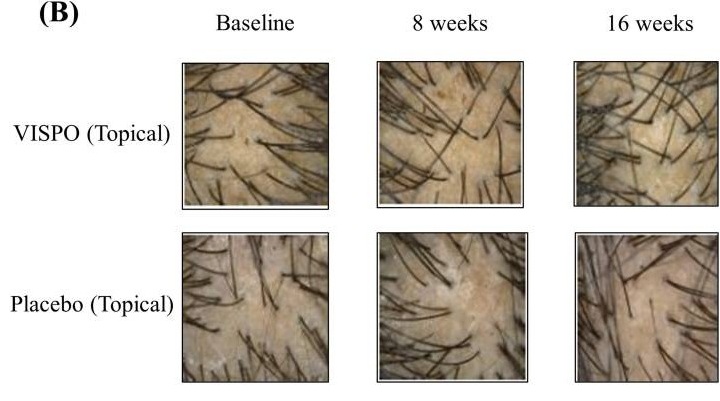
Nevertheless, there is some clinical evidence to back this treatment. In one 16-week randomized controlled trial involving adults with AGA, both oral and topical standardized saw palmetto oil (named VISPO™) significantly reduced hair shedding compared to placebo and produced small increases in hair density (around 5-7%). Oral saw palmetto also reduced serum DHT, whereas topical did not.[60]Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral And Topical Administration Of A Standardized Saw Palmetto Oil Reduces Hair Fall And Improves The … Continue reading
Also, in one study, oral saw palmetto (320 mg/day) was compared to finasteride (1 mg/day) in 100 men with mild to moderate AGA over 24 months. Hair regrowth assessed by global photographic assessment was reported 38% of the saw palmetto group compared to 68% in the finasteride group.[61]Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparitive Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A … Continue reading
What people often miss from this study is that of those saw palmetto users, 45/50 men saw a stop in hair loss. In fact, that number was the same in the finasteride group (45/50). In other words, saw palmetto stopped the progression of hair loss in 90% of men. And even better, the saw palmetto group also reported zero side effects.[62]Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparitive Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A … Continue reading
In terms of topical saw palmetto, this formulation tends to look more effective for hair regrowth when combined with other hair growth-supporting ingredients.[63]Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., Bunnag, T., Pimonrat, T., Muangdang, N., Pichai, P. (2016). Treatment of Male Androgenetic Alopecia with Topical Products Containing … Continue reading But it’s difficult to see how much contribution saw palmetto made because it has been studied in multi-ingredient blends.
There is one 24-week pilot study published on topical saw palmetto, but they reported a very large increase in terminal-hair count of 74.1%, and the study lacked a placebo control, so we recommend taking these results with a pinch of salt.[64]Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. … Continue reading
Figure 9: Images showing changes in scalp hair following 16-week treatment of VISPO™ oil or a placebo oil. Adapted from Figure 4.[65]Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the … Continue reading Image obtained in line with the Creative Commons License.
How Safe is Saw Palmetto?
Saw palmetto is generally safe and well-tolerated. The most common adverse effects of oral saw palmetto include stomach upset, headache, or fatigue. Sexual side effects are uncommon compared to pharmaceutical 5AR inhibitors (e.g., finasteride and dutasteride).
For topical saw palmetto, most of the effects are localized to the scalp, including inflammation, itching, dryness, and flaking.
Because saw palmetto supplements are not tightly regulated, the quality and strength of the product can vary across brands, which can affect the safety and tolerability.
Find out more about saw palmetto in our article here.
General Guidelines for Use
When starting hair loss treatment, it can feel daunting and difficult to know where to begin. While each treatment has its own considerations around dosing, frequency, and application, some general tips and recommendations could help when starting any new hair loss treatment:
- Speak to a clinician first: This is especially important for any prescription treatments or if you have any underlying health concerns, risk factors, or fertility plans.
- Give treatments enough time: Most evidence-based hair loss treatments take at least 3 to 6 months before meaningful changes are visible.
- Track your progress: Take consistent photographs in the same lighting and at the same angle. This makes it much easier to assess whether something is working or isn’t.
- Monitor any side effects: If you do notice any unwanted effects, it’s important to discuss any adjustments or alternatives with a licensed clinician rather than pushing through discomfort.
- Prioritize consistency: The best treatment is one you can use consistently over time. If you’re not able to stick with it, even a strong medication won’t work as well as it should.
- Adjust when needed: Hair loss changes over time, and treatment plans must be able to evolve with it.
Final Thoughts
Ultimately, there is no single best hair loss treatment for men. The right choice depends on your specific pattern of hair loss, your goals, and your preferences. Whether that’s a prescription medication, an over-the-counter option, a natural approach like saw palmetto, or a technique like microneedling.
What matters is choosing a hair loss treatment you can tolerate and stick with consistently over time. Hair loss treatment is rarely about finding the strongest option. It’s more about finding one that you can realistically fit into your routine and that you are comfortable maintaining.
With the right expectations and a tailored plan, many men are able to stabilize their hair loss and maintain hair growth improvements over time.
References[+]
References ↑1 Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ ↑2 Wikipedia, (2009), Finasteride. Available at: https://en.wikipedia.org/wiki/Finasteride#/media/File:Finasteride.svg (Accessed: 24 February 2026) ↑3 Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride In The Treatment Of Men With Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 39(4 Pt 1). 578-589. Available at: https://doi.org/10.1016/s0190-9622(98)70007-6 ↑4 Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., Carrington, P., Gencheff, C., Cole, G.W., Pariser, D.M., Epstein, E.S., Tanaka, W., Dallob, A., Vandormael, K., Geissler, L., Waldstreicher, J. (1999). The Effects Of Finasteride On Scalp Skin And Serum Androgen Levels In Men With Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 41(4). 550-554 ↑5 Fertig, R.M., Caresse, G.A., Darwin, E., Gaudi, S. (2017). Sexual Side Effects Of 5-ɑ-Reductase Inhibitors Finasteride And Dutasteride: A Comprehensive Review. Dermatology Online Journal. 23(11). 1-19. Available at: https://doi.org/10.5070/D32311037240 ↑6 Estill, M.C., Ford, A., Omeira, R., Rodman, M. (2023). Finasteride And Dutasteride For The Treatment Of Male Androgenetic Alopecia: A Review Of Efficacy And Reproductive Adverse Effects. Georgetown Medical Review. 7(1). Available at: https://doi.org/10.52504/001c.88531 ↑7 Amory, J.K., Wang, C., Swerdloff, R.S., Anawalt, B.D., Matsumoto, A.M., Bremner, W.J., Walker, S.E., Haberer, L.J., Clark, R.V. (2007). The Effect Of 5ɑ-Reductase Inhibition With Dutasteride And Finasteride On Semen Parameters And Serum Hormones In Healthy Men. The Journal Of Clinical Endocrinology & Metabolism. 92(5). 1659-1665. Available at: https://doi.org/10.1210/jc.2006-2203 ↑8 Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑9 Wikipedia, (2015), Minoxidil Structure Svg. Available at: https://en.wikipedia.org/wiki/Minoxidil#/media/File:Minoxidil_structure.svg (Accessed: 20 February 2026) ↑10 Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189 ↑11 Olsen, E.A., Weiner, M.S., Amara, I.A., DeLong, E.R. (1990). Five-Year Follow-Up Of Men With Androgenetic Alopecia Treated With Topical Minoxidil. Journal Of The American Academy Of Dermatology. 22(4). 643-646. Available at: https://doi.org/10.1016/0190-9622(90)70089-z ↑12 Price, V.H., Menefee, E., Strauss, P.C. (1999). Changes In Hair Weight And Hair Count In Men With Androgenetic Alopecia, After Application Of 5% And 2% Topical Minoxidil, Placebo, Or No Treatment. Journal Of The American Academy Of Dermatology. 41(5 Pt 1). 717-721. Available at: https://doi.org/10.1016/s0190-9622(99)70006-x ↑13 Olsen, E.A., Dunlap, F.E., Funicella, T., Koperski, J.A., Swinehart, J.M., Tschen, E.H., Trancik, R.J. (2002). A Randomized Clinical Trial Of 5% Topical Minoxidil Versus 2% Topical Minoxidil And Placebo In The Treatment Of Androgenetic Alopecia In Men. Journal Of The American Academy Of Dermatology. 47(3). 377-385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑14 Singh, S., Patil, A., Kianfar, N., Waskiel-Burnat, A., Rudnicka, L., Sinclair, R., Goldust, M. (2022). Does Topical Minoxidil At Concentrations Higher Than 5% Provide Additional Clinical Benefit? Clinical And Experimental Dermatology. 47(11). 1951-1955. Available at: https://doi.org/10.1111/ced.15338 ↑15 Vasantha, K.L. (2025). Efficiency And Safety Of 10% Minoxidil In The Treatment Of Alopecia Areata: A Randomised Controlled Trial. Journal Of Population Therapeutics And Clinical Pharmacology. 32(4). 724-730. Available at: https://doi.org/10.53555/56ggpq88 ↑16 Ghonemy, S., Alarawi, A., Bessar, H. (2021). Efficacy And Safety Of A New 10% Topical Minoxidil Versus 5% Topical Minoxidil And Placebo In The Treatment Of Male Androgenetic Alopecia: A Trichoscopic Evaluation. Journal Of Dermatological Treatment. 32(2). 236-241. Available at: https://doi.org/10.1080/09546634.2019.1654070 ↑17 Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I.A., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating The Efficacy And Safety Of Combined Microneedling Therapy Versus Topical Minoxidil In Androgenetic Alopecia: A Systematic Review And Meta-Analysis. Archives Of Dermatological Research. 317(1). 528. Available at: https://doi.org/10.1007/s00403-025-04032-1 ↑18 Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvan, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin Enhances Minoxidil Response In Androgenetic Alopecia Patients By Upregulating Follicular Sulfotransferase Enzymes. Dermatologic Therapy. 32(3). E12915. Available at: https://doi.org/10.1111/dth.12915 ↑19 Patel, K., Palmer, A., Nixon, R. (2023). Allergic Contact Dermatitis From Propylene Glycol: A Case Series From Australia. Contact Dermatitis. 89(2). 79-84. Available at: https://doi.org/10.1111/cod.14325 ↑20 Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of Dermatology, Venereology and Leprology.* 75. 47. Available at: https://doi.org/10.4103/0378-6323.45220 ↑21 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2021). Efficacy And Safety Of Topical Finasteride Spray Solution For Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical Trial. Journal Of The European Academy Of Dermatology And Venereology. 36(2). 286–294. Available at: https://doi.org/10.1111/jdv.17738 ↑22 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2022). Efficacy And Safety Of Topical Finasteride Spray Solution For Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical Trial. Journal Of The European Academy Of Dermatology And Venereology. 36(2). 286–294. Available at: https://doi.org/10.1111/jdv.17738 ↑23 US Food and Drug Administration. (2025). FDA alerts health care providers, compounders and consumers of potential risks associated with compounded topical finasteride products. FDA. Available at: https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-consumers-potential-risks-associated-compounded (Accessed: December 2025) ↑24 Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in Dermatology. 17(4). 457-463. Available at: PMID: 29601622 ↑25 Wikipedia, (2009), Dutasteride. Available at: https://en.wikipedia.org/wiki/Dutasteride#/media/File:Dutasteride.svg (Accessed: 24 February 2026) ↑26 Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. *Indian Journal of Dermatology, Venereology and Leprology.* 83. 47. Available at: https://doi.org/10.4103/0378-6323.188652 ↑27 Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 70(3). 489–498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑28 Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male androgenic alopecia in South Korea: a multicentre chart review study. *Annals of Dermatology.* 34(5). 349. Available at: https://doi.org/10.5021/ad.22.027 ↑29 Choi, G.S., Sim, W.Y., Kang, H., Huh, C.H., Lee, Y.W., Shantakumar, S., Ho, Y.F., Oh, E.J., Duh, M.S., Cheng, W.Y., Bobbili, P., Thompson-Leduc, P., Ong, G. (2022). Long-Term Effectiveness And Safety Of Dutasteride Versus Finasteride In Patients With Male Androgenic Alopecia In South Korea: A Multicentre Chart Review Study. Annals Of Dermatology. 34(5). 349-359. Available at: https://doi.org/10.5021/ad.22.027 ↑30 Hirshburg, J.M., Kelsey, P.A., Therrien, C.A., Gavino, A.C., Reichenberg, J.S. (2016). Adverse Effects And Safety Of 5-Alpha Reductase Inhibitors (Finasteride, Dutasteride): A Systematic Review. Journal Of Clinical And Aesthetic Dermatology. 9(7). 56-62 ↑31, ↑32 Marihart, S., Harik, M., Djavan, B. (2005). Dutasteride: A Review Of Current Data On A Novel Dual Inhibitor Of 5alpha Reductase. Reviews In Urology. 7(4). 203-210 ↑33, ↑34, ↑35 Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑36 Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A Review. Advances In Dermatology And Allergology. 39(3). 472-478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑37 Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil And Its Use In Hair Disorders: A Review. Drug Design, Development And Therapy. 13. 2777-2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑38 Beach, R.A. (2018). Case Series Of Oral Minoxidil For Androgenetic And Traction Alopecia: Tolerability & The Five C’s Of Oral Therapy. Dermatologic Therapy. 31(6). E12707. Available at: https://doi.org/10.1111/dth.12707 ↑39 Lueangarun, S., Panchaprateep, R., Tempark, T., Noppakun, N. (2015). Efficacy And Safety Of Oral Minoxidil 5 Mg Daily During 24-Week Treatment In Male Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 72(5). AB113. Available at: https://doi.org/10.1016/j.jaad.2015.02.466 ↑40 Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral Minoxidil In Male Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 81(2). 648-649. Available at: https://doi.org/10.1016/j.jaad.2019.04.054 ↑41 Pirmez, R., Salas-Callo, C.I. (2020). Very-Low-Dose Oral Minoxidil In Male Androgenetic Alopecia: A Study With Quantitative Trichoscopic Documentation. Journal Of The American Academy Of Dermatology. 82(1). E21-E22. Available at: https://doi.org/10.1016/j.jaad.2019.08.084 ↑42 Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair loss: a multicenter study of 1404 patients. Journal of the American Academy of Dermatology. 84(6). 1644–1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑43 Wikramanayake, T.C., Rodriguez, R., Choudhary, S., Mauro, L.M., Nouri, K., Schachner, L.A., Jimenez, J.J. (2012). Effects of the Lexington LaserComb on Hair Regrowth in the C3H/HeJ Mouse Model of Alopecia Areata. Lasers in Medical Science. 27(2). 431-436. Available at: https://doi.org/10.1007/s10103-011-0953-7 ↑44 Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts and Bolts of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available at: https://doi.org/10.1007/s10439-011-0454-7 ↑45 Makihara, E., Masumi, S. (2008). Blood Flow Changes of a Superficial Temporal Artery Before and After Low-Level Laser Irradiation Applied to the Temporomandibular Joint Area. Nihon Hotetsu Shika Gakkai Zasshi. 52(2). 167-170. Available at: https://doi.org/10.2186/jjps.52.167 ↑46 Bouzari, N., Firooz, A.R. (2006). Lasers May Induce Terminal Hair Growth. Dermatologic Surgery. 32(3). 460. Available at: https://doi.org/10.1111/j.1524-4725.2006.32092.x ↑47 Lueangarun, S., Visutjindaporn, P., Parcharoen, Y., Jamparuang, P., Tempark, T. (2021). A Systematic Review and Meta-Analysis of Randomized Controlled Trials of United States Food and Drug Administration-Approved, Home-Use, Low-Level Light/Laser Therapy Devices for Pattern Hair Loss: Device Design and Technology. Journal of Clinical and Aesthetic Dermatology. 14(11). E64-E75 ↑48, ↑49 Shin, J.W., Paik, K., Na, J.I., Lew, B.L., Huh, C.H. (2026). Long-Term Efficacy and Safety of Low-Level Laser Therapy for Androgenetic Alopecia: A 12-Month Prospective Trial. Dermatologic Therapy. Available at: https://doi.org/10.1155/dth/6621458 ↑50 HairLossTalk, (2019), Microneedling Photo Results Summary. Available at: https://www.hairlosstalk.com/interact/threads/microneedling-photo-results-summary.121072/ (Accessed: 24 February 2026) ↑51 Pei, D., Zeng, L., Huang, X., Wang, B., Liu, L., Zhang, G. (2024). Efficacy and Safety of Combined Microneedling Therapy for Androgenic Alopecia: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Journal of Cosmetic Dermatology. Available at: https://doi.org/10.1111/jocd.16186 ↑52 Dhurat, R., Sukesh, M., Avhad, G., Dandale, A., Pal, A., Pund, P. (2013). A Randomized Evaluator Blinded Study of Effect of Microneedling in Androgenetic Alopecia: A Pilot Study. International Journal of Trichology. 5(1). 6-11. Available at: https://doi.org/10.4103/0974-7753.114700 ↑53 Dhurat, R., Mathapati, S. (2015). Response to Microneedling Treatment in Men with Androgenetic Alopecia Who Failed to Respond to Conventional Therapy. Indian Journal of Dermatology. 60(3). 260-263. Available at: https://doi.org/10.4103/0019-5154.156361 ↑54 Bao, L., Gong, L., Guo, M., Liu, T., Shi, A., Zong, H. (2020). Randomized Trial of Electrodynamic Microneedle Combined with 5% Minoxidil Topical Solution for the Treatment of Chinese Male Androgenetic Alopecia. Journal of Dermatological Treatment. Available at: https://doi.org/10.1080/14764172.2017.1376094 ↑55 Wikimedia Commons, (2025), Ketoconazole Chemical Structure (SVG). Available at: https://commons.wikimedia.org/wiki/Category:Ketoconazole#/media/File:(2S,4R)-Ketoconazol.svg (Accessed: 24 February 2026) ↑56, ↑57 Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: https://doi.org/10.1159/000017954 ↑58 Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine And 1% Zinc Pyrithione Formulations. International Journal Of Cosmetic Science. 24(5). 249-256. Available at: https://doi.org/10.1046/j.1467-2494.2002.00145.x ↑59 Khandpur, S., Suman, M., Reddy, B.S. (2002). Comparative Efficacy Of Various Treatment Regimens For Androgenetic Alopecia In Men. J Dermatol. 29(8). 489-98. Available at: https://doi.org/10.1111/j.1346-8138.2002.tb00314.x ↑60 Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral And Topical Administration Of A Standardized Saw Palmetto Oil Reduces Hair Fall And Improves The Hair Growth In Androgenetic Alopecia Subjects – A 16-Week Randomized, Placebo-Controlled Study. Clinical, Cosmetic And Investigational Dermatology. 16. 3251–3266. Available at: https://doi.org/10.2147/CCID.S435795 ↑61, ↑62 Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparitive Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A Two-Year Study. International Journal of Immunopathology and Pharmacology. 25(4). 1167-1173. Available at: https://doi.org/10.1177/039463201202500435 ↑63 Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., Bunnag, T., Pimonrat, T., Muangdang, N., Pichai, P. (2016). Treatment of Male Androgenetic Alopecia with Topical Products Containing Serenoa Repens Extract. Australasian Journal of Dermatology. 57(3). e76-e82. Available at: https://doi.org/10.1111/ajd.12352 ↑64 Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. 57(3), e76-e82. Available at: https://doi.org/10.1111/ajd.12352 ↑65 Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the Hair Growth in Androgenetic Alopecia Subjects – A 16-Week Randomized, Placebo-Controlled Study. Clin Cosmet Investig Dermatol. 16, 3251-3266. Available at: https://doi.org/10.2147/CCID.S435795 Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn More
Sophie Grice, PhD
Sophie completed a BSc in Pharmacology before earning a PhD in Immunopharmacology at the University of Liverpool. Her doctoral research examined drug hypersensitivity reactions in patients treated with immune checkpoint inhibitors. She later pursued postdoctoral research focused on T cell mediated immune responses, with an emphasis on the immunogenicity of gene therapies.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down