- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
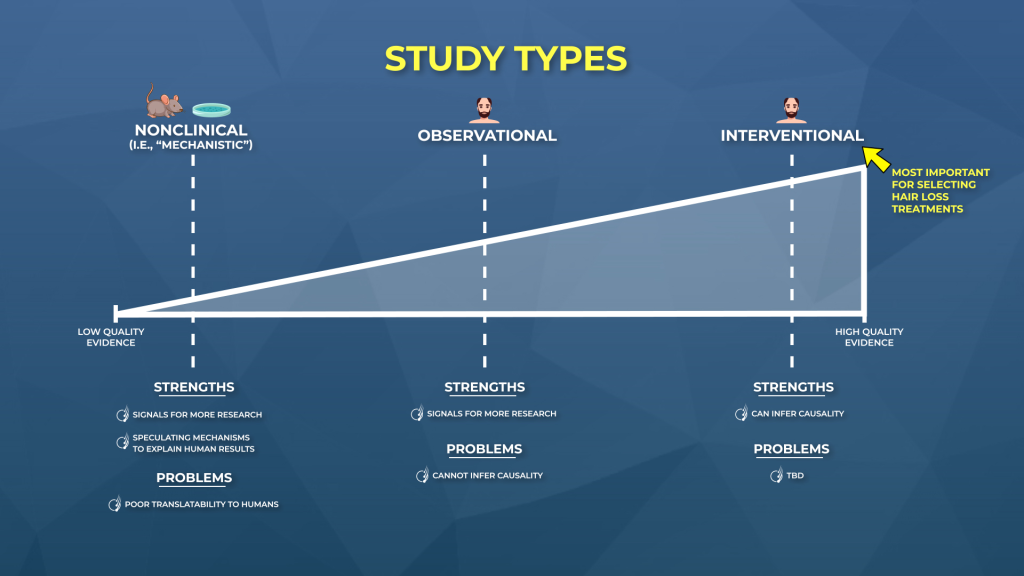
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Musely Reviews for Hair Loss: Are They Trustworthy?
-
Best Minoxidil for Women: Top 6 Brands of 2026
-
Best Topical Finasteride: 5 Top Products of 2026
-
Best Topical Dutasteride: 8 Choices for 2026
-
10 Best Shampoos for Thinning Hair Men
-
Does Semaglutide Cause Hair Loss?
-
Does Dandruff Cause Hair Loss?
-
Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
Articles6 Best Hair Loss Treatments for Women
First Published Apr 24 2026Last Updated Apr 5 2026Pharmaceutical Researched & Written By:Cassie Hopton, PhD
Researched & Written By:Cassie Hopton, PhD Reviewed By:Rob English, Medical Editor
Reviewed By:Rob English, Medical Editor
Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
Female pattern hair loss treatments vary widely in effectiveness, safety, and suitability across life stages. In this evidence-based guide, we review six of the best options for women, from minoxidil to low-level laser therapy, explaining how they work, the clinical evidence behind them, key safety considerations, and who they’re best for.
Full Article
With so many hair loss treatments on the market, finding the best one to treat female pattern hair loss (androgenic alopecia) can feel overwhelming. But what really is “best”? The best hair loss treatment is one that is effective, safe, and suitable for long-term use, but these standards can vary from person to person. This is particularly true for women, where menopause and family planning intentions play a part in treatment safety and efficacy.
In this article, we evaluate hair loss treatments for female pattern hair loss based on their quality of evidence, effectiveness, and broad suitability. We list the 6 best hair loss treatments for women, guiding you through how each treatment works, the clinical evidence for it, who the ideal candidates are, and who it’s safe for, so that you can decide on the most suitable treatment going forward.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Defining What Is “Best”
“Best” is hard to define. So many different treatments can be the best in different ways: safest, most effective, most affordable, or most tolerated.
What is best is highly personal and depends on each person’s circumstances, such as the severity of hair loss, lifestyle, age, and family-planning intentions. All of these factors affect the degree of regrowth, effectiveness, and safety of each treatment.
At Perfect Hair Health, we evaluate treatments using three metrics: evidence quality, regrowth potential, and long-term viability.
- Evidence quality: Assesses the quality and totality of evidence. Studies that use humans in randomized, blinded, and controlled clinical trials are ranked highest for evidence quality, while anecdotal evidence ranks lowest.
- Regrowth potential: Assesses how much hair regrowth can be expected based on hair count (particularly terminal hairs) and hair thickness improvements demonstrated in human clinical trials.
- Long-term viability: Assesses treatment adherence over time, i.e., do people keep up with daily administration of the treatment?
For this article, we define “best” by considering 6 treatments that have scored highest in evidence quality and regrowth potential.
This is not a best-to-worst list. Each treatment may be more or less suitable depending on personal circumstances, and please note that many of these treatments are not suitable for women who are already pregnant or breastfeeding.
6 Best Hair Loss Treatments for Women
#1 Topical Minoxidil
Evidence quality score – 97/100
Topical minoxidil is currently the only FDA-approved treatment for female pattern hair loss. It is considered the gold-standard treatment, and typically comes as a topical solution or foam at doses of 2% or 5%. Higher doses are also available, but these are not FDA-approved.
Ideal for:
- Women looking for an over-the-counter solution
- Women looking for an FDA-approved solution
- Women who can commit to daily topical application
- Women with localized thinning
Safe for:
- Women of reproductive age, as long as they are not pregnant, trying to conceive, or breastfeeding, and have effective contraception in place
- Perimenopausal women
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women who are pregnant, trying to conceive, or breastfeeding
- Women with scalp hypersensitivity
- Women with damaged or inflamed scalps
- Women who own cats
How Does It Work?
The action of minoxidil against hair loss is not clear-cut. It is known to work through a variety of biological mechanisms.
- Local vasodilation: Minoxidil can promote increased blood flow.[1]Sica, D.A. (2004). Minoxidil: An Underused Vasodilator For Resistant Or Severe Hypertension. J Clin Hypertens (Greenwich). 6(5). 283-287. Available at: https://doi.org/10.1111/j.1524-6175.2004.03585.x When applied topically, this may enhance conditions favorable for growth at hair follicles.
- VEGF up-regulation: Vascular endothelial growth factor (VEGF) mediates angiogenesis (the formation of new blood vessels), which may support hair growth.[2]Zeltzer, A.A., Keren, A., Paus, R., Gilhar, A. (2024). Topical Minoxidil Rejuvenates Hair Follicles From Men With Androgenetic Alopecia In Vivo. Acta Dermato Venereologica. 104(24213). Available at: … Continue reading
- Anagen induction and telogen shortening: Minoxidil can induce the anagen phase (growth phase) of hair, and shorten the resting phase (telogen phase).[3]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study … Continue reading
- Wnt/β-catenin activation: The signalling pathway Wnt/β-catenin is essential for hair follicle development, regeneration, and maintenance.[4]Kwack, M.H., Kang, B.M., Kim, M.K., Kim, J.C., Sung, Y.K. (2011). Minoxidil Activates Beta-Catenin Pathway In Human Dermal Papilla Cells: A Possible Explanation For Its Anagen Prolongation Effect. … Continue reading
- Prostaglandin E2 elevation: Prostaglandin E2 is associated with hair growth, possibly by protecting the follicle environment from inflammation.[5]Michelet, J.F., Commo, S., Billoni, N., Mahe, Y.F., Bernard, B.A. (1997). Activation Of Cytoprotective Prostaglandin Synthase-1 By Minoxidil As A Possible Explanation For Its Hair Growth-Stimulating … Continue reading
The Clinical Evidence
There is an extensive number of high-quality clinical trials showing that minoxidil between 2% and 5% can promote hair regrowth.[6]Olsen, E.A., DeLong, E.R., Weiner, M.S. (1987). Long-Term Follow-Up of Men with Male Pattern Baldness Treated with Topical Minoxidil. Journal of the American Academy of Dermatology. 16(3). 688–695. … Continue reading,[7]Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the … Continue reading,[8]Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta … Continue reading[9]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study … Continue reading It’s important to note here that dosing and application matter – for women with female pattern hair loss, clinical trials show that once-daily application of 5% minoxidil can be just as effective as twice-daily use of 2% minoxidil in improving hair regrowth and hair counts.[10]Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., Bartels, N.G. (2011). A Randomized, Single-Blind Trial Of 5% Minoxidil Foam Once Daily Versus 2% Minoxidil Solution Twice Daily In The … Continue reading
For most users of topical minoxidil, substantial improvements in hair growth can be seen between 3 and 6 months. But after more than a year, regrowth rates decline.[11]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study … Continue reading
Why does this decline happen? It’s because minoxidil does not target the androgen hormone dihydrotestosterone (DHT). DHT impacts hair growth, and high levels can cause miniaturization of the hair follicles that ultimately leads to weaker, finer, and non-growing hair. DHT is not only the primary cause of androgenic alopecia, but the levels of this hormone are often higher in those with this condition.[12]Ustuner, E. T. (2013). Cause of Androgenic Alopecia: Crux of the Matter. Plast Reconstr Surg Glob Open. 1(7). e64. Available at: https://doi.org/10.1097/GOX.0000000000000005,[13]Sekhavat, H., Bar Yehuda, S., Asotra, S. (2025). Using the Mechanisms of Action Involved in the Pathogenesis of Androgenetic Alopecia to Treat Hair Loss. Int J Mol Sci. 26(21). 10712. Available at: … Continue reading
Because minoxidil does not target DHT, it does not block the effects of DHT, and therefore hair shafts still become thinner over time. However, that doesn’t mean you have to stop using minoxidil. Minoxidil can be supplemented with other therapies, like tretinoin and microneedling, that enhance penetration and revitalize regrowth.[14]Shin, H.S., Won, C.H., Lee, S.H., Kwon, O.S., Kim, K.H., Eun, H.C. (2007). Efficacy of 5% Minoxidil Versus Combined 5% Minoxidil and 0.01% Tretinoin for Male Pattern Hair Loss. American Journal of … Continue reading,[15]Zhang, Y., Sheng, Y., Zeng, Y., Hu, R., Zhao, J., Wang, W., Yang, Q. (2022). Randomized Trial Of Microneedling Combined With 2% Minoxidil Topical Solution For The Treatment Of Female Pattern Hair … Continue reading
Interested in how to enhance results with minoxidil? Read our article.
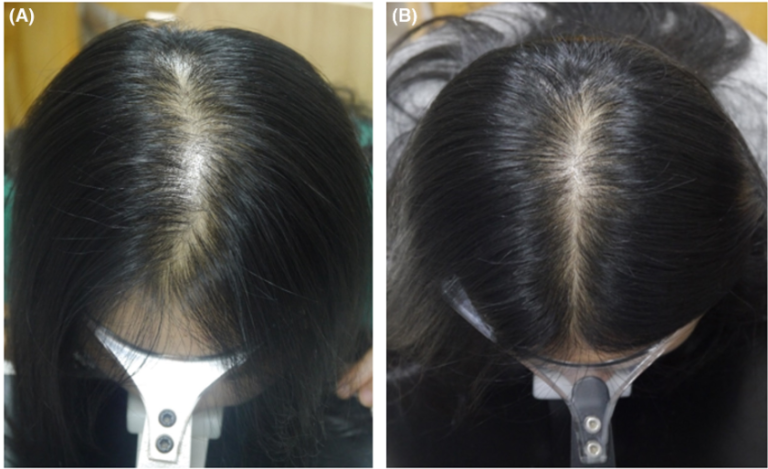
Figure 3: The scalp of a female patient at baseline (A) and after being treated with 2% minoxidil and microneeding for 24 weeks (B). Adapted from Figure 1.[16]Zhang, Y., Sheng, Y., Zeng, Y., Hu, R., Zhao, J., Wang, W., Yang, Q. (2022). Randomized Trial Of Microneedling Combined With 2% Minoxidil Topical Solution For The Treatment Of Female Pattern Hair … Continue reading Image used under the Creative Commons License.
Anecdotal evidence from our PHH members shows that 7% minoxidil with enhancers like tretinoin often improves hair regrowth outcomes.
Safety & Side Effects
Scalp Irritation
Topical minoxidil can cause dermatitis in 2-6% of users. But this is mainly due to propylene glycol, a harmful additive common in many topical minoxidil products. It’s added to enhance the dissolution and penetration of minoxidil, but it can cause itching, redness, and sometimes swelling.[17]Jadeed, H.B., Almudimeegh, A.M., Alomran, S.A., Alshathry, A.H. (2021). A Case Of Contact Allergic Dermatitis To Topical Minoxidil. Cureus. 13(1). E12510. Available at: … Continue reading
Topical minoxidil formulations may also include alcohols, which can again irritate the scalp and cause dryness and flaking.
Systemic absorption
As with any topical treatment, systemic absorption (whole body absorption) is possible, and this can cause side effects seen in those taking oral minoxidil. We’ll get to this next.
Pregnancy
There is no direct evidence linking topical minoxidil to reduced fertility or conception. However, there are a few cases where adverse events during pregnancy have occurred, and there isn’t enough evidence to confidently show that it is safe.[18]Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., Moroni, F. (2003). Topically Applied Minoxidil May Cause Fetal Malformation: A Case Report. Birth Defects Research Part A: Clinical And … Continue reading,[19]Differences In Reproductive Toxicology Between Alopecia Drugs: An Analysis On Adverse Events Among Female And Male Cases. (no year). Oncotarget. 7(50). 82074-82084. Available at: … Continue reading So, it is generally not recommended for women who are planning to become pregnant, are pregnant, or are breastfeeding. You can read more about this here.
Pets
In addition to human side effects, minoxidil is highly toxic to cats, so best avoided for cat lovers.[20]DeClementi, C., Bailey, K.L., Goldstein, S.C., Orser, M.S. (2004). Suspected Toxicosis After Topical Administration Of Minoxidil In 2 Cats. Veterinary Emergency & Critical Care. 14(4). 287-292. … Continue reading
#2 Oral Minoxidil
Evidence quality score – 63/100
Oral minoxidil is just that – an oral form of minoxidil that is often taken in doses between 0.25 mg and 5 mg daily. It is an off-label treatment for female pattern hair loss.
Ideal for:
- Women who do not respond well to topical minoxidil or find adherence difficult
- Women looking for ease of use
- Women who are comfortable with unwanted hair growth outside of the scalp
- Women with diffuse thinning
Safe for:
- Women of reproductive age, as long as they are not pregnant, trying to conceive, or breastfeeding, and have effective contraception in place
- Perimenopausal women
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women who are pregnant, trying to conceive, or breastfeeding
- Women with pre-existing heart, liver, or kidney issues
Figure 4: Ulo oral minoxidil.
How Does It Work?
This form of minoxidil works in the same way as topical minoxidil, but the effects are not localized to the scalp since oral minoxidil is systemically absorbed.
Users may consider oral minoxidil if they are not seeing benefits from topical minoxidil. The effectiveness of topical minoxidil depends on scalp penetration, i.e., whether it actually reaches the hair follicle. Studies show that only 1.4% of applied minoxidil is absorbed through skin.[21]Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2022). Minoxidil: A Comprehensive Review. Journal Of Dermatological Treatment. 33(4). 1896-1906. Available at: … Continue reading
Absorption can be influenced by the delivery vehicle (e.g., whether it is a foam, alcohol-based solution, etc.), the integrity of the skin barrier, and the presence of certain biological components (specifically the sulfotransferase enzyme SULT1A1) that transforms minoxidil into minoxidil sulfate, its active form.[22]Sattur, S., Talathi, A., Shetty, G., Arsiwala, S., Pereira, R., Dhoot, D. (2023). Comparative Clinical Study Evaluating The Efficacy And Safety Of Topical 5% Cetosomal Minoxidil And Topical 5% … Continue reading Since the activity of SULT1A1 at the scalp varies between individuals, factors like this can also impact treatment effectiveness.[23]Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A … Continue reading
So, for those who didn’t respond to topical minoxidil either due to inadequate penetration or SULT1A1 activity limitations, oral minoxidil offers a solution as it does not rely on penetration and can be activated in the liver, where SULT1A1 is abundant.
The Clinical Evidence
Even though it is not an FDA-approved treatment for female pattern hair loss, oral minoxidil at 1 mg taken daily has been found to be just as effective as 5% topical minoxidil in enhancing terminal hair counts after 24 weeks (i.e., hairs that contribute to the appearance of a fuller head of hair).[24]Ramos, M.P., Sinclair, R.D., Kasprzak, M., Miot, H.A. (2020). Minoxidil 1 Mg Oral Versus Minoxidil 5% Topical Solution For The Treatment Of Female-Pattern Hair Loss. Journal Of The American Academy … Continue reading
One retrospective study, which analysed the results of many cases of men with androgenic alopecia being treated with oral minoxidil, showed that approximately 90% of men showed substantial regrowth at 6-12 months after taking oral minoxidil between 2.5 mg and 5 mg.[25]Jiminiez-Couche, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral … Continue reading We can therefore presume these results would apply to women as well.
Overall, oral minoxidil is considered an effective medication for androgenic alopecia, and there is a plethora of clinical evidence to back this up (you can check out our other free articles on minoxidil to find out more information).[26]Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: … Continue reading,[27]Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global … Continue reading,[28]Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 … Continue reading,[29]Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). … Continue reading
Safety & Side Effects
The concern with oral minoxidil is not effectiveness, but safety. We mentioned previously that there is a risk of systemic absorption with topical minoxidil – this is something that happens with oral minoxidil, and can lead to a variety of safety concerns and side effects.
Cardiovascular Risk
Some people taking low-dose oral minoxidil have developed pericardial effusion (fluid around the heart) and pericarditis (inflammation around the heart).[30]Dlova, N.C., Jacobs, T., Singh, S. (2022). Pericardial, Pleural Effusion And Anasarca: A Rare Complication Of Low-Dose Oral Minoxidil For Hair Loss. JAAD Case Reports. 11(28). 94-96. Available at: … Continue reading,[31]Bentivegna, K., Zhou, A.E., Adalsteinsson, J.A., Sloan, B. (2022). Letter In Reply: Pericarditis And Peripheral Edema In A Healthy Man On Low-Dose Oral Minoxidil Therapy. JAAD Case Reports. 20(29). … Continue reading. But this is a risk, not an inevitability.
At higher doses, minoxidil may also change the electrical activity of the heart. This has been recorded in 60% to 90% of users, but often resolves with continued use or dose reduction.[32]Hall, D., Charocopos, F., Froer, K.L., Rudolph, W. (1979). ECG Changes During Long-Term Minoxidil Therapy For Severe Hypertension. Archives Of Internal Medicine. 139. 790-794. Available at: … Continue reading
Hypertrichosis
Hypertrichosis is excessive hair growth on unwanted areas of the body. One study reported that users of 1 mg minoxidil have reported facial and body hypertrichosis in 32% and 47% of cases.[33]Bloch, D.L., Carlos, R.M.D. (2025). Side Effects’ Frequency Assessment Of Low Dose Oral Minoxidil In Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: … Continue reading
You can read more about how to avoid hypertrichosis when using minoxidil here.
Edema
In one study, lower leg edema (swelling) has been reported in 75% of cases in those taking 1 mg minoxidil, while periorbital (eye) edema has been reported in 50% of cases of those taking 1 mg and 50% of cases in those moving from 2 mg to 2.5 mg.[34]Bloch, D.L., Carlos, R.M.D. (2025). Side Effects’ Frequency Assessment Of Low Dose Oral Minoxidil In Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: … Continue reading
Managing Risk
If oral minoxidil feels like the right option for you but is causing some unwanted side effects, you can try a lower dose (and gradually work to higher doses), splitting the daily dose between morning and evening, or taking the medication sublingually (letting it dissolve under your tongue).
Interested in Oral Minoxidil?
Low-dose oral minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

#3 Oral Spironolactone
Evidence quality score – 49/100
Spironolactone is another off-label treatment for female pattern hair loss. It was originally designed to treat hypertension, heart failure, and edema, but research shows oral spironolactone between 25 mg and 200 mg per day could also provide hair regrowth benefits.
Ideal for:
- Women who have struggled with the side effects of oral minoxidil
- Women looking to supplement topical minoxidil
- Women looking for ease of use
- Women who are comfortable with the potential for irregular periods
Safe for:
- Women of reproductive age, as long as they are not pregnant, trying to conceive, or breastfeeding, and have effective contraception in place
- Perimenopausal women with effective contraception in place
- Menopausal women who are not at risk of hyperkalemia
- Postmenopausal women who are not at risk of hyperkalemia
Not recommended for:
- Women who are pregnant or trying to conceive
- Perimenopausal women without effective contraception in place
- Women at any stage of life who are at risk of hyperkalemia
How Does It Work?
Spironolactone targets DHT, the primary cause of female pattern hair loss. It does this in two ways:
- It binds and blocks androgen receptors.[35]Corvol, P., Michaud, A., Menard, J., Freifeld, M., Mahoudeau, J. (1975). Antiandrogenic Effect Of Spirolactones: Mechanism Of Action. Endocrinology. 97(1). 52-58. Available at: … Continue reading
- It interferes with the production of androgen hormones.[36]Menard, R.H., Stripp, B., Gillette, J.R. (1974). Spironolactone And Testicular Cytochrome P-450: Decreased Testosterone Formation In Several Species And Changes In Hepatic Drug Metabolism. … Continue reading,[37]Kossor, D.C., Kominami, S., Takemori, S., Colby, H.D. (1992). Destruction Of Testicular Cytochrome P-450 By 7α-Thiospironolactone Is Catalyzed By The 17α-Hydroxylase. The Journal Of Steroid … Continue reading
The binding of DHT to androgen receptors activates the process of hair loss. So by blocking DHT binding, spironolactone helps reduce follicle miniaturization.
Spironolactone has been shown to reduce the production of androgen hormones like testosterone. It hasn’t been shown to reduce the production of DHT, but DHT is derived from testosterone, so it’s possible (but not proven) that spironolactone could limit the conversion of testosterone to DHT.
The Clinical Evidence
So far, there is clinical evidence for oral spironolactone, but quality studies are lacking. The overall conclusions from the use of 25-50 mg/day spironolactone for female pattern hair loss suggest that daily intake could reduce hair loss severity.[38]Sinclair, R.D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. International Journal of Dermatology. 57(1). … Continue reading,[39]Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: … Continue reading However, the methodologies of these studies have several caveats:
- There were no placebo controls.
- Hair density was determined visually.
- Hair shedding was self-reported in some cases.
- Only one of these studies is a clinical trial, the other is a retrospective study.
So, whether oral spironolactone at this level is actually effective for treating hair loss is not shown by robust research yet. There is, however, a greater number of studies showing the use of spironolactone between 100 mg and 200 mg per day, particularly in conjunction with minoxidil.[40]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading,[41]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading,[42]Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: … Continue reading,[43]Hoedemaker, C., Van Egmond, S., Sinclair, R. (2007). Treatment of female pattern hair loss with a combination of spironolactone and minoxidil. Australasian Journal of Dermatology. 48(1). 43–45. … Continue reading
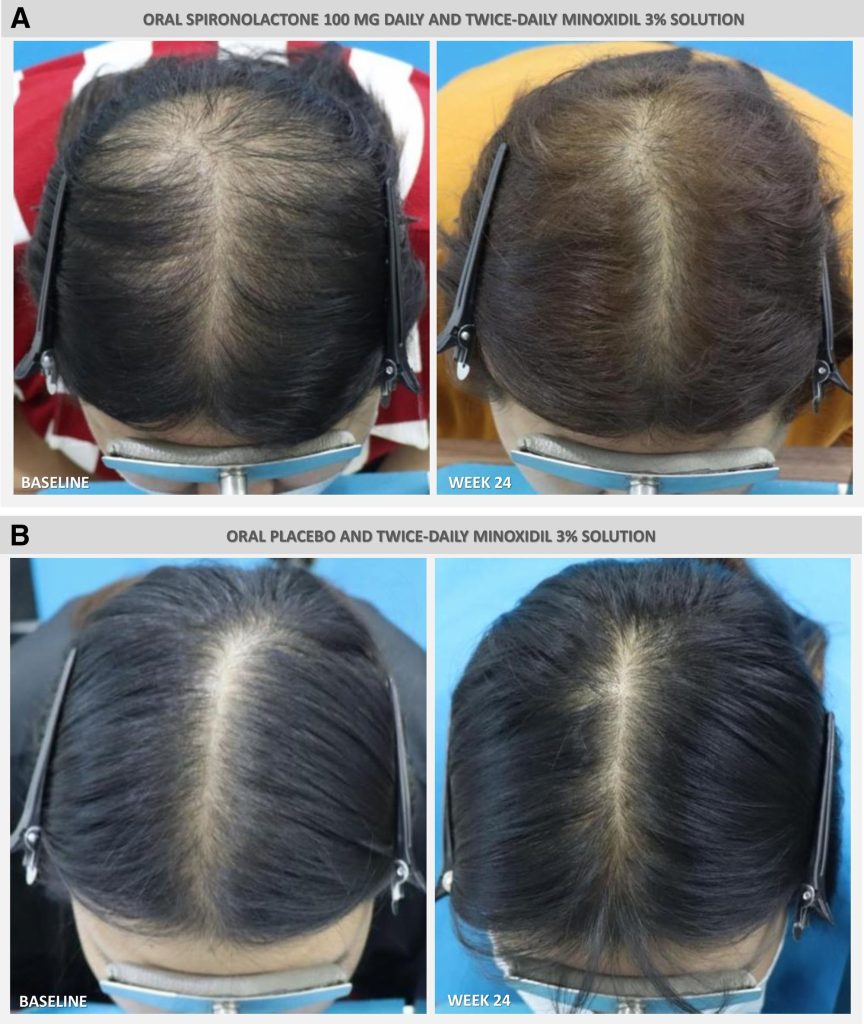
Figure 5: Images of hair density at baseline and following 24 weeks of daily treatment with either 100 mg spironolactone and 3% topical minoxidil or a placebo and topical 3% minoxidil. Adapted from Figure 5.[44]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading Image used under the Creative Commons License.
These studies are not without limitations themselves. Some of them do not count terminal hair, some of them show hair changes that are not statistically significant, some have small participant numbers, and some are case studies, not clinical trials. But overall, most report positive outcomes for the use of oral spironolactone with topical minoxidil, showing that this combination could improve the following more effectively than topical minoxidil alone:
- Terminal hair count
- Total hair count
- Hair diameter
This suggests that oral spironolactone could be a promising treatment option for those looking to enhance hair regrowth with minoxidil.
Safety & Side Effects
Oral spironolactone is not without its side effects, but they don’t always occur, as doses of 200 mg/day have been safely administered with no adverse effects reported.[45]Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: … Continue reading However, there is a common set of side effects reported that are worth considering before starting this medication.
Menstrual and Fertility Problems
Irregular menstruation has been reported in 15-37% of women taking oral spironolactone.[46]Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English … Continue reading,[47]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading It may also cause temporary fertility. However, menstruation and fertility should return to normal after 2 months of stopping.[48]NHS, (no date), Pregnancy, Breastfeeding And Fertility While Taking Spironolactone. Available at: … Continue reading
Hyperkalemia
Hyperkalemia is when there is too much potassium in the blood. Those taking medium doses of oral spironolactone have reported hyperkalemia, which can impact nerve and muscle function.[49]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading,[50]Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: … Continue reading,[51]Collins, M.S., Ali, S., Wiss, I.P., Senna, M.M. (2023). Retrospective Analysis Of The Risk Of Hyperkalaemia In Women Older Than 65 Years Of Age Prescribed Spironolactone For Female-Pattern Hair Loss. … Continue reading Hyperkalemia risk increases with age.[52]Sevamontree, C., Jintajirapan, S., Phakdeekitcharoen, P., Phakdeekitcharoen, B. (2024). The Prevalence And Risk Factors Of Hyperkalemia In The Outpatient Setting. Int J Nephrol. 2024. 5694131. … Continue reading
Edema
This may seem paradoxical since spironolactone treats edema, but spironolactone can also cause edema because it promotes shifts in fluids from the bloodstream to the tissues.[53]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading
Pregnancy
Because oral spironolactone interferes with DHT everywhere in the body, it is not recommended for those who are planning on pregnancy or are pregnant. DHT is a hormone required for male development, so those taking spironolactone are at risk of causing harm to male fetuses. Talk to your doctor if you’re considering becoming pregnant while taking spironolactone.
Additional Side Effects
Fewer than 5% of women report breast tenderness, reduced libido, nausea, headache, and fatigue when taking oral spironolactone.[54]Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English … Continue reading,[55]Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: … Continue reading
#4 Topical Finasteride
Evidence quality score – 69/100
Oral finasteride is FDA-approved for male pattern hair loss, but it is not recommended for women due to its potential side effects and harm during pregnancy. Topical finasteride provides a solution to those who do not tolerate, or who can not take, oral finasteride.
Ideal for:
- Women who have not seen results from topical or oral minoxidil
- Women who can commit to daily topical application
- Women with localized thinning
- Women more willing to risk side effects
Safe for:
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women of reproductive age, who are pregnant, trying to conceive, or breastfeeding
- Perimenopausal women
- Women with scalp hypersensitivity
- Women with damaged or inflamed scalps
How Does It Work?
Like spironolactone, finasteride blocks DHT. But it doesn’t block DHT in the same way. Topical finasteride inhibits the type II version of the enzyme that forms DHT, known as 5-ɑ-reductase type II, to reduce DHT production at the scalp.[56]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading,[57]Shapiro, J., & Kaufman, K. D. (2003). Use of finasteride in the treatment of men with androgenetic alopecia (male pattern hair loss). *Journal of Investigative Dermatology Symposium Proceedings.* … Continue reading.
The Clinical Evidence
Research for topical finasteride is still in the early stages, and most of the data is from men. But if we consider that the data may be generalizable to women, the research shows that topical finasteride can produce similar hair regrowth when compared to oral finasteride.
- Medium doses: Topical finasteride at 0.25% has shown to be effective for the treatment of androgenic alopecia, supporting the steady improvement of hair thickness, hair counts, and an overall reduction in hair loss comparable to oral finasteride.[58]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading,[59]Gupta, A.K., Talukder, M. (2022). Topical Finasteride For Male And Female Pattern Hair Loss: Is It A Safe And Effective Alternative? Journal of Cosmetic Dermatolology. 21(5). 1841-1848. Available at: … Continue reading
- High doses: Doses of 1% can provide improvements in hair count equivalent to oral finasteride.[60]Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing The Therapeutic Effects Of Finasteride Gel And Tablet In Treatment Of The Androgenetic Alopecia. Indian J Dermatol Venereol … Continue reading
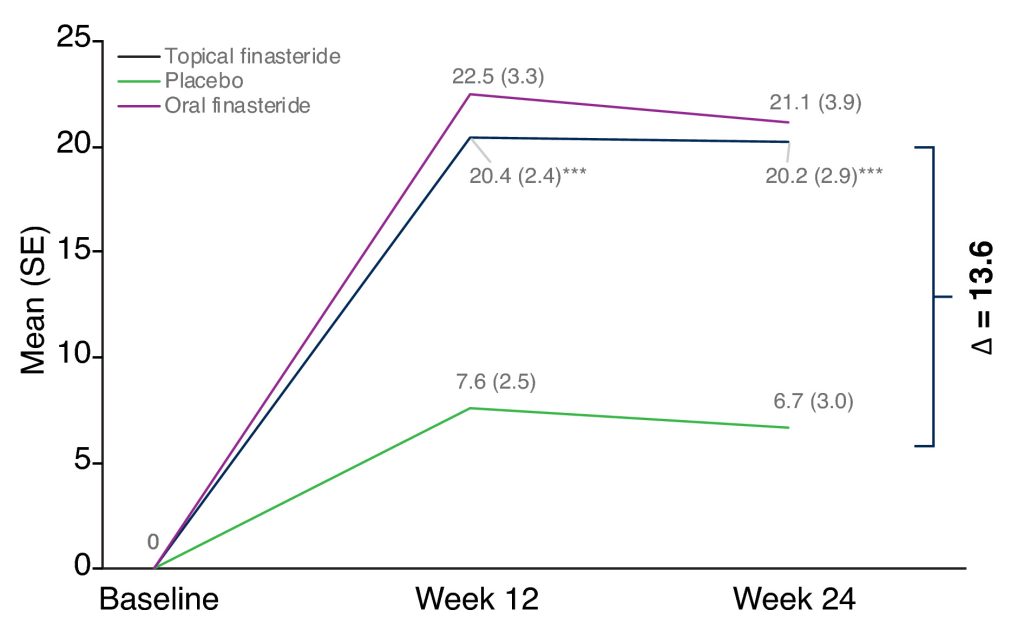
Mean change in hair count from baseline to week 24 of treatment with 0.25% topical finasteride, 1 mg oral finasteride, or a placebo. Adapted from Figure 3.[61]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading Image used under the Creative Commons License.
Outside of direct comparison between delivery methods, low doses of finasteride at 0.005% to 0.1% have also been shown to improve hair growth outcomes, particularly when combined with other hair loss treatments.[62]Mazzarella, G., Loconsole, G., Cammisa, G., Mastrolonardo, G., & Vena, G. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy … Continue reading,[63]Tanglertsampan C. Efficacy and safety of 3% minoxidil versus combined 3% minoxidil / 0.1% finasteride in male pattern hair loss: a randomized, double-blind, comparative study. J Med Assoc Thai. 2012 … Continue reading
Doses of 0.5% finasteride along with 2% minoxidil have also been shown to improve female pattern hair loss after 18 months of treatment, although this was based on visual analysis. While visual analysis can show visible improvements in hair growth, it does not provide objective data and is therefore less robust.[64]Rossi, A., Magri, F., D’Arino, A., Pigliacelli, F., Muscianese, M., Leoncini, P., Caro, G., Federico, A., Fortuna, M.C., Carlesimo, M. (2020). Efficacy Of Topical Finasteride 0.5% Vs … Continue reading Nonetheless, the clinical promise of topical finasteride is evident, but requires more randomized, blinded, and controlled clinical trials.
Safety & Side Effects
Oral finasteride may be effective, but it comes with several serious side effects. Unfortunately, those using topical finasteride are not exempt from these effects, since finasteride can be absorbed systemically even when applied topically.
Systemic Absorption
Clinical trials with topical finasteride at 0.25% show that, despite localized application to the scalp, serum (blood) DHT levels could fall by 24% to 34.5%.[65]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading,[66]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading This shows that topical finasteride can be systemically absorbed. Systemically absorbed finasteride is associated with depression, sexual dysfunction, and breast changes.
In 2025, the FDA released a warning that no FDA-approved topical finasteride exists, and that the FDA has not evaluated their safety, effectiveness, or quality prior to marketing. 32 cases linked to topical finasteride were reported between 2019 and 2024, where reports describe adverse events that are consistent with those reported with oral finasteride. In other words, these cases involved adverse events associated with systemically absorbed topical finasteride.
Pregnancy
There is a reason that topical finasteride is FDA-approved for male pattern baldness, but not female pattern baldness. The risk of systemic absorption makes this treatment unsuitable for women who could become pregnant, because the DHT-blocking effects of finasteride can cause toxicity to male fetuses. The magnitude of serum DHT lowering is much higher for finasteride than spironolactone, despite the fact that both are DHT blockers. So, unlike spironolactone, this treatment is usually only recommended for menopausal and postmenopausal women where there is no risk of pregnancy.
Scalp Irritation
Trials with 0.25% finasteride show that topical application can cause pruritus (irritating sensations) and erythema (inflamed skin) in around 2% of users, but this is only a small amount.[67]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading,[68]Gupta, A.K., Talukder, M. (2022). Topical Finasteride For Male And Female Pattern Hair Loss: Is It A Safe And Effective Alternative? Journal of Cosmetic Dermatolology. 21(5). 1841-1848. Available at: … Continue reading
Mitigating Risks
Application of 0.005% finasteride has been found to promote hair growth, but not impact serum DHT levels.[69]Mazzarella, G.F., Loconsole, G.F., Cammisa, G.A., Mastrolonardo, G.M. and Vena, G., 1997. Topical Finasteride in the Treatment of Androgenic Alopecia: Preliminary Evaluations After a 16-Month Therapy … Continue reading So, lowering the dosage of finasteride used is one option to mitigate risk. But systemic absorption is not just about dosage; it’s also about the treatment formulations, volume, frequency, and length of application.
For example, a study comparing once-daily application of 0.25% finasteride and twice-daily application of 0.25% finasteride found that once-daily application lowered serum DHT levels by 20%, while twice-daily application lowered levels by 70%![70]Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., Palmieri, R. (2014). A Novel Finasteride 0.25% Topical Solution For Androgenetic Alopecia: Pharmacokinetics And Effects On Plasma Androgen … Continue reading
Our analysis of member-submitted lab tests and clinical data suggests that 1-2 mL of topical finasteride at 0.005% to 0.02% is the best combination to minimize systemic absorption while maintaining positive hair growth outcomes.
You can read more about maximizing regrowth while minimizing side effects from finasteride here.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

#5 Topical Dutasteride
Evidence quality score – 55/100
Dutasteride is an FDA-approved medication for benign prostate hyperplasia but is also an off-label medication for male androgenic alopecia. It is typically used in doses from 0.02% to 0.2%
Ideal for:
- Women who have not seen results from minoxidil or finasteride
- Women who can commit to daily topical application
- Women with localized thinning
- Women who are willing to take treatments with less clinical evidence but high potential
Safe for:
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women of reproductive age, or who are pregnant, trying to conceive, or breastfeeding
- Perimenopausal women
- Women with scalp hypersensitivity
- Women with damaged or inflamed scalps
How Does It Work?
Dutasteride is another DHT blocker. It inhibits both type I and type II 5-ɑ-reductase.[71]Arif, T., Dorjay, K., Adil, M., Sami, M., (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). Available at: 10.2174/157488471266617031011112 Dutasteride, applied topically, could therefore help to combat hair loss by reducing DHT levels on the scalp, and has the potential to be more effective than finasteride due to blocking more than one type of 5-ɑ-reductase.
The Clinical Evidence
While the mechanism of action of dutasteride points to a promising treatment for androgenic alopecia, the research is still in its early stages. The potential effectiveness of dutasteride as a hair loss treatment started emerging around 2020. So, there isn’t a lot of evidence.
But it’s not just a lack of evidence; we’re limited in quality evidence. If you’re interested in taking a deep dive into the flaws behind a key dutasteride study, read our article here. But to summarize, studies so far have used:
- Inconsistent methods – For example, inconsistent timings in product application, or changes in the size, shape, and location of areas chosen to measure regrowth, which make results less valid
- Dual therapies – Some of the studies only used topical dutasteride in combination with microneedling, which does not help us isolate the true effect of dutasteride.
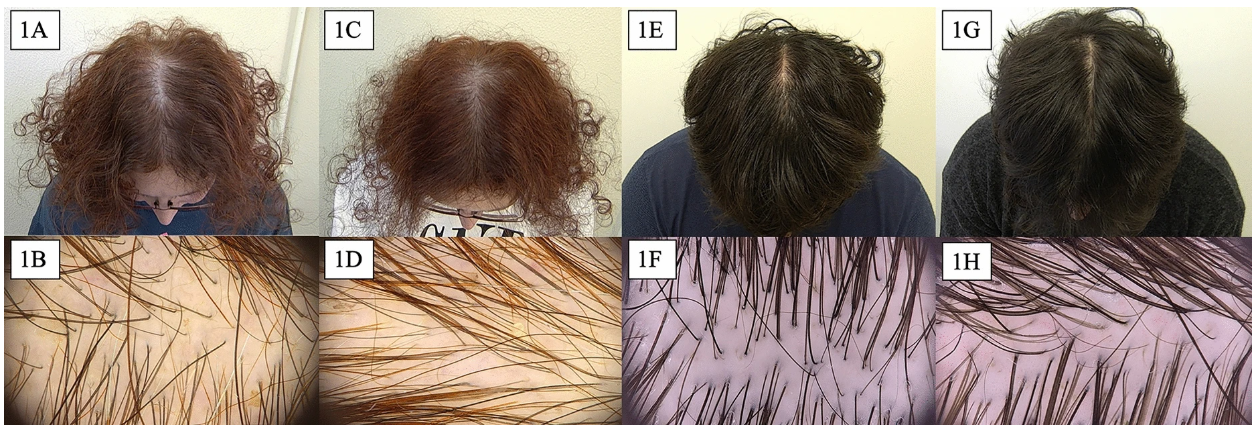
However, in all studies, the conclusions from the use of 0.01% to 0.05% dutasteride are positive. These studies report that dutasteride localizes well to the scalp, minimally affects serum DHT, and effectively slows or stops hair loss.[72]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled … Continue reading,[73]Nada, E.A., Sharkawy, R.E., Maged, W.M., & Elmagd, M.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Southern Medical Journal, 22, 387-400,[74]Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martínez-Moreno, A., Ocampo-Garza, S.S., (2022). Microneedling Plus Topical … Continue reading,[75]Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M., (2025). Efficacy of Skin Patting and Iontophoresis With Dutasteride Gel in Male and … Continue reading In support of this, our members report that use of 0.01 to 0.02% dutasteride does not appear to regrow hair, but it does minimize the hair loss process.
Figure 7: Scalp images at Baseline (1A, 1B, 1E, 1F) and after 8 weeks treatment (1C, 1D, 1G, 1H) with a 6% dutasteride gel. Adapted from Figure 1.[76]Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M., (2025). Efficacy of Skin Patting and Iontophoresis With Dutasteride Gel in Male and … Continue reading Image used under the Creative Commons License.
Safety & Side Effects
As a DHT blocker and topical formula, many of the risks posed by dutasteride are similar to those of topical finasteride.
Systemic Absorption
Topical dutasteride application appears to only modestly reduce serum DHT levels – in other words, topical application has limited systemic absorption when used appropriately.[77]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled … Continue reading,[78]Nada, E.A., Sharkawy, R.E., Maged, W.M., & Elmagd, M.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Southern Medical Journal, 22, 387-400 Systemic absorption is associated with several side effects similar to finasteride, such as sexual dysfunction and breast changes, so minimizing this is a great benefit.
Pregnancy
While dutasteride may only modestly affect serum DHT, any change in serum DHT levels is considered inappropriate for use in women who hope to or can conceive. Again, this is due to the toxicity risk to male fetuses associated with blocking DHT.
Scalp Irritation
Scalp irritation has been reported with the use of topical dutasteride in a few cases.[79]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled … Continue reading
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

#6 Low-Level Laser Therapy (LLLT)
Evidence quality score – not evaluated
LLLT is a therapy using light. For women who aren’t suitable for traditional treatments, this therapy could potentially work as an alternative.
Ideal for:
- Women wanting to avoid pharmaceutical solutions
- Women willing to adhere to the usage of LLLT over a long period of time
- Women willing to pay to use LLLT over a long period of time
- Women willing to try more experimental methods
Safe for:
- Women of reproductive age
- Perimenopausal women
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women who are pregnant or breastfeeding
How Does It Work?
These devices emit infrared and near-infrared light, which are naturally emitted by the sun. They often come in the form of a cap or headpiece that can be worn for long periods of time. Infrared and near-infrared light tend to elongate hair growth cycles and improve hair counts.
We’re not entirely sure of why this happens, but the proposed mechanism is that light waves in the infrared and near-infrared range stimulate molecules like cytochrome c oxidase (which drives energy production for hair growth and regulates th in the mitochondria of cells.
You may remember from school that mitochondria generate the energy required by a cell to function. It is thought that the stimulation by LLLT increases energy production and triggers genetic changes leading to enhanced cell growth and repair.[80]Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts And Bolts Of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available … Continue reading,[81]Lyu, K., Liu, X., Jiang, L., Chen, Y., Lu, J., Zhu, B., Liu, X., Li, Y., Wang, D., Li, S. (2022). The Functions And Mechanisms Of Low-Level Laser Therapy In Tendon Repair (Review). Frontiers in … Continue reading
So, in essence, LLLT directed at the scalp could promote hair follicle cells to grow.
The Clinical Evidence
So far, the clinical evidence for the use of LLLT technology on participants with androgenic alopecia has been reported as overwhelmingly positive.
- LLLT combs have been found to increase terminal hair counts, total hair counts, and hair strength compared to a control product.[82]Satino, J.L., Markou, M. (2003). Hair Regrowth And Increased Hair Tensile Strength Using The HairMax LaserComb For Low-Level Laser Therapy. International Journal of Cosmetic Surgery and Aesthetic … Continue reading,[83]Leavitt, M., Charles, G., Heyman, E. (2009). HairMax LaserComb® Laser Phototherapy Device In The Treatment Of Male Androgenetic Alopecia. Clinical Drug Investigation. 29(5). 283-292. Available at: … Continue reading
- LLLT hats have increased terminal and total hair counts as well as hair density compared to users of a control product or treatment.[84]Lanzafame, R.J., Blanche, R.R., Bodian, A.B., Chiacchierini, R.P., Fernandez-Obregon, A., Kazmirek, E.R. (2013). The Growth Of Human Scalp Hair Mediated By Visible Red Light Laser And LED Sources In … Continue reading,[85]Kim, H., Choi, J.W., Kim, J.Y., Shin, J.W., Lee, S.J., Huh, C.H. (2013). Low-Level Light Therapy For Androgenetic Alopecia: A 24-Week, Randomized, Double-Blind, Sham Device-Controlled Multicenter … Continue reading,[86]Friedman, S., Schnoor, P. (2017). Novel Approach To Treating Androgenetic Alopecia In Females With Photobiomodulation (Low-Level Laser Therapy). Dermatologic Surgery. 43(6). 856-867. Available at: … Continue reading
- A retrospective analysis of many studies concluded that LLLT is both safe and effective for the treatment of androgenic alopecia.[87]Avci, P., Gupta, G.K., Clark, J., Wikonkal, N., Hamblin, M.R. (2014). Low-Level Laser (Light) Therapy (LLLT) For Treatment Of Hair Loss. Lasers in Surgery and Medicine. 46(2). 144-151. Available at: … Continue reading
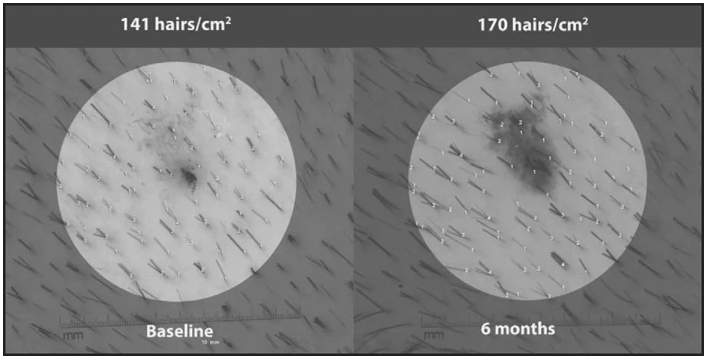
Figure 9: Non-vellus hair density macroimages at baseline and 6 months in one patient in the HairMax LaserComb® group. Adapted from Figure 1.[88]Leavitt, M., Charles, G., Heyman, E. (2009). HairMax LaserComb® Laser Phototherapy Device In The Treatment Of Male Androgenetic Alopecia. Clinical Drug Investigation. 29(5). 283-292. Available at: … Continue reading Image used under the Creative Commons License.
This has led to companies selling items like LLLT caps at extremely high prices – in the range of $200 to $3,000!
But what’s the truth? Let’s look deeper into the science.
Firstly, not all studies report positive results. A study comparing the use of LLLT and 5% minoxidil with a group using 5% minoxidil alone found that there was no difference in improvement between the groups.[89]Sondagar, D.M., Mehta, H.H., Agharia, R.S., Jhavar, M.K. (2023). Efficacy Of Low-Level Laser Therapy In Androgenetic Alopecia – A Randomized Controlled Trial. International Journal of … Continue reading
Secondly, let’s consider the timing of clinical trials. For most people, regardless of hair condition, hair shedding typically peaks from late summer to early autumn (August to September). This is because the number of hairs in the telogen phase is highest by September. Following the telogen phase, hair reaches the exogen phase, the phase where hair sheds and falls out.[90]Courtois, M., Loussouarn, G., Hourseau, S., Grollier, J.F. (1996). Periodicity In The Growth And Shedding Of Hair. British Journal of Dermatology. 134(1). 47-54,[91]Kunz, M., Seifert, B., Trüeb, R.M. (2009). Seasonality Of Hair Shedding In Healthy Women Complaining Of Hair Loss. Dermatology. 219(2). 105-110. Available at: https://doi.org/10.1159/000216832,[92]Liu, C., Yang, J., Qu, L., Gu, M., Liu, Y., Gao, J., Collaudin, C., Loussouarn, G. (2014). Changes In Chinese Hair Growth Along A Full Year. International Journal of Cosmetic Science. 36(6). 531-536. … Continue reading The reason this happens is likely a combination of factors – light-driven patterns, summer stressors such as ultraviolet and heat, and more frequent washing.
So what would happen if we designed a clinical study where participants with androgenic alopecia were split into two groups: group 1 that had to use an LLLT cap, and group 2 assigned a placebo cap with no lights. Let’s say we started the trial in July and finished in October. Remember – natural hair shedding will occur from August to September. We would likely see that:
- The LLLT therapy minimizes the effect of typical seasonal shedding in September.
- Group 2 would undergo the typical hair shedding in September.
So when we look at the results in October, there will likely be a big divide between the number of hairs in Group 1 and the number of hairs in Group 2. This divide would be bigger than if we designed the experiment mid-summer, for example.
We’re not saying that LLLT isn’t effective, but we are saying that the time a clinical trial happens could affect whether a result is statistically significant.
And even when results are statistically significant, there isn’t enough comparative data to confidently say whether hair regrowth with LLLT is as substantial as regrowth with the use of minoxidil, finasteride, or dutasteride.[93]Sondagar, D.M., Mehta, H.H., Agharia, R.S., Jhavar, M.K. (2023). Efficacy Of Low-Level Laser Therapy In Androgenetic Alopecia – A Randomized Controlled Trial. International Journal of … Continue reading,[94]Esmat, S.M., Hegazy, R.A., Gawdat, H.I., Abdel Hay, R.M., Allam, R.S., El Naggar, R., Moneib, H. (2017). Low Level Light-Minoxidil 5% Combination Versus Either Therapeutic Modality Alone In … Continue reading For now, LLLT is not a treatment that should be used in isolation; it should be used (for those that can afford it) with other medications.[95]Munck, A., Gavazzoni, M.F., Trüeb, R.M. (2014). Use Of Low-Level Laser Therapy As Monotherapy Or Concomitant Therapy For Male And Female Androgenetic Alopecia. International Journal Of Trichology. … Continue reading
These reasons may explain why effects from using LLLT in real-life often conflict with the clinical study outcomes we see advertised. However, for women where options are more limited, LLLT could provide another treatment route.
Safety & Side Effects
Scalp Irritation
Some users report erythema, pruritus, dry scalp, and acne or eczema after use of a LLLT therapy device.[96]Hamblin, M.R. (2019). Photobiomodulation For The Management Of Alopecia: Mechanisms Of Action, Patient Selection And Perspectives. Clinical, Cosmetic And Investigational Dermatology. 12. 669-678. … Continue reading,[97]Egger, A., Resnik, S.R., Aickara, D., Maranda, E., Kaiser, M., Wikramanayake, T.C., Jimenez, J.J. (2020). Examining The Safety And Efficacy Of Low-Level Laser Therapy For Male And Female Pattern Hair … Continue reading
Pregnancy
LLLT should be safe for most women, but there is a lack of safety data to confidently prove that this therapy is safe for pregnant or breastfeeding women.
Honorable Mentions
Some hair loss ingredients have a decent evidence score behind them, but lower regrowth and long-term viability scores. These ingredients generally have less clinical evidence and lower efficacy, so are most effective when used in conjunction with one of the “best” treatments, but they are largely safe for most women at any stage of their life. So, we think they deserve an honorable mention.
Topical Ingredient Evidence Quality Score Evidence Melatonin 66/100 Leave-on, topical melatonin has not only been found to be safe, but a synthesis of five clinical studies shows that it can increase hair density, reduce hair loss, and increase hair strength in those with androgenic alopecia.[98]Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., Elsner, P. (2012). Topical Melatonin for Treatment of Androgenetic Alopecia. Int J Trichology. 4(4). 236-245. Available at: … Continue reading Cetirizine 57/100 Topical 1% cetirizine can improve hair growth, hair density, and hair thickness in patients with androgenic alopecia. However, the evidence so far is limited, with no evidence of efficacy beyond 6 months of use.[99]Zaky, M.S., Abo Khodeir, H., Ahmed, H.-A., Elsaie, M.L., (2021). Therapeutic Implications of Topical Cetirizine 1% in Treatment of Male Androgenetic Alopecia: A Case-Controlled Study. Journal of … Continue reading,[100]Hossein Mostafa, D., Samadi, A., Niknam, S., Nasrollahi, S.A., Guishard, A., Firooz, A., (2021). Efficacy of Cetirizine 1% Versus Minoxidil 5% Topical Solution in the Treatment of Male Alopecia: A … Continue reading,[101]Bassiouny, E.A., El-Samanoudy, S.I., Abbassi, M.M., Nada, H.R., Farid, S.F., (2023). Comparison Between Topical Cetirizine With Minoxidil Versus Topical Placebo With Minoxidil in Female Androgenetic … Continue reading Saw palmetto 54/100 Saw palmetto may strengthen hair and could improve thickness, although more robust evidence with longer trials is needed.[102]Morganti, P., Fabrizi, G., James, B., Bruno, C. (1998). Effect of gelatin-cystine and serenoa repens extract on free radicals level and hair growth. Journal of Applied Cosmetology. 16, 57-64. … Continue reading,[103]Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. … Continue reading,[104]Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the … Continue reading Ketoconzole 50/100 A review summarising seven studies using topical ketoconazole concluded that ketoconazole appears to increase hair diameter and result in clinical improvement in those with androgenic alopecia.[105]Fields, J. R., Vonu, P. M., Monir, R. L., Schoch, J. J. (2020). Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review. Dermatol Ther. 33(1). e13202. Available at: … Continue reading Guidelines for Starting a New Treatment
Starting a new treatment is not a journey you should take on your own. When considering a new treatment, always:
- Speak to a doctor
- Track your progress
- Track side effects
- Look to switch if side effects occur
And remember that consistency matters most in hair loss treatments! Only regular, long-term use keeps hair follicles in the growth phase and prevents ongoing shedding. So, choosing the right method of delivery to suit your lifestyle and routines is key.
Final Thoughts
Choosing a hair loss treatment is deeply personal. Factors like age, pregnancy plans, menopause stage, health history, and treatment tolerance all shape what may be effective and safe for you. While some options have stronger evidence or broader suitability, there is no single “best” treatment for every woman. Instead, the goal is to find the approach that aligns with your stage in life, lifestyle, and long-term needs. Discuss options with a qualified clinician, monitor results, and adjust over time to maximize hair regrowth along with safety.
References[+]
References ↑1 Sica, D.A. (2004). Minoxidil: An Underused Vasodilator For Resistant Or Severe Hypertension. J Clin Hypertens (Greenwich). 6(5). 283-287. Available at: https://doi.org/10.1111/j.1524-6175.2004.03585.x ↑2 Zeltzer, A.A., Keren, A., Paus, R., Gilhar, A. (2024). Topical Minoxidil Rejuvenates Hair Follicles From Men With Androgenetic Alopecia In Vivo. Acta Dermato Venereologica. 104(24213). Available at: https://doi.org/10.2340/actadv.v104.24213 ↑3 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study Protocol. Skin Research And Technology. 26(4). 542-557. Available at: https://doi.org/10.1111/srt.12827 ↑4 Kwack, M.H., Kang, B.M., Kim, M.K., Kim, J.C., Sung, Y.K. (2011). Minoxidil Activates Beta-Catenin Pathway In Human Dermal Papilla Cells: A Possible Explanation For Its Anagen Prolongation Effect. Journal Of Dermatological Science. 154-159. Available at: https://doi.org/10.1016/j.jdermsci.2011.01.013 ↑5 Michelet, J.F., Commo, S., Billoni, N., Mahe, Y.F., Bernard, B.A. (1997). Activation Of Cytoprotective Prostaglandin Synthase-1 By Minoxidil As A Possible Explanation For Its Hair Growth-Stimulating Effect. Journal Of Investigative Dermatology. 108(2). 205-209. Available at: https://doi.org/10.1111/1523-1747.ep12334249 ↑6 Olsen, E.A., DeLong, E.R., Weiner, M.S. (1987). Long-Term Follow-Up of Men with Male Pattern Baldness Treated with Topical Minoxidil. Journal of the American Academy of Dermatology. 16(3). 688–695. Available at: https://doi.org/10.1016/S0190-9622(87)70089-9 ↑7 Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the Treatment of Androgenetic Alopecia in Men. Journal of the American Academy of Dermatology. 57(5). 767–774. Available at: https://doi.org/10.1016/j.jaad.2007.04.012 ↑8 Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta Dermatovenerologica Alpina, Pannonica et Adriatica. 25(3). 41–44. Available at: https://doi.org/10.15570/actaapa.2016.12 ↑9 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study Protocol. Skin Research and Technology. 26(4). 542–557. Available at: https://doi.org/10.1111/srt.12827 ↑10 Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., Bartels, N.G. (2011). A Randomized, Single-Blind Trial Of 5% Minoxidil Foam Once Daily Versus 2% Minoxidil Solution Twice Daily In The Treatment Of Androgenetic Alopecia In Women. Journal Of The American Academy Of Dermatology. 65(6). 1126-1134. Available at: https://doi.org/10.1016/j.jaad.2010.09.724 ↑11 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study Protocol. Skin Research & Technology. 26(4). 542-557. Available at: https://doi.org/10.1111/srt.12827 ↑12 Ustuner, E. T. (2013). Cause of Androgenic Alopecia: Crux of the Matter. Plast Reconstr Surg Glob Open. 1(7). e64. Available at: https://doi.org/10.1097/GOX.0000000000000005 ↑13 Sekhavat, H., Bar Yehuda, S., Asotra, S. (2025). Using the Mechanisms of Action Involved in the Pathogenesis of Androgenetic Alopecia to Treat Hair Loss. Int J Mol Sci. 26(21). 10712. Available at: https://doi.org/10.3390/ijms262110712 ↑14 Shin, H.S., Won, C.H., Lee, S.H., Kwon, O.S., Kim, K.H., Eun, H.C. (2007). Efficacy of 5% Minoxidil Versus Combined 5% Minoxidil and 0.01% Tretinoin for Male Pattern Hair Loss. American Journal of Clinical Dermatology. 8(5). 285–290. Available at: https://doi.org/10.2165/00128071-200708050-00003 ↑15, ↑16 Zhang, Y., Sheng, Y., Zeng, Y., Hu, R., Zhao, J., Wang, W., Yang, Q. (2022). Randomized Trial Of Microneedling Combined With 2% Minoxidil Topical Solution For The Treatment Of Female Pattern Hair Loss In A Chinese Population. Journal Of Cosmetic Dermatology. 21(12). 6985-6991. Available at: https://doi.org/10.1111/jocd.15424 ↑17 Jadeed, H.B., Almudimeegh, A.M., Alomran, S.A., Alshathry, A.H. (2021). A Case Of Contact Allergic Dermatitis To Topical Minoxidil. Cureus. 13(1). E12510. Available at: https://doi.org/10.7759/cureus.12510 ↑18 Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., Moroni, F. (2003). Topically Applied Minoxidil May Cause Fetal Malformation: A Case Report. Birth Defects Research Part A: Clinical And Molecular Teratology. 67(12). 997-1001. Available at: https://doi.org/10.1002/bdra.10095 ↑19 Differences In Reproductive Toxicology Between Alopecia Drugs: An Analysis On Adverse Events Among Female And Male Cases. (no year). Oncotarget. 7(50). 82074-82084. Available at: https://doi.org/10.18632/oncotarget.12617 ↑20 DeClementi, C., Bailey, K.L., Goldstein, S.C., Orser, M.S. (2004). Suspected Toxicosis After Topical Administration Of Minoxidil In 2 Cats. Veterinary Emergency & Critical Care. 14(4). 287-292. Available at: https://doi.org/10.1111/j.1476-4431.2004.04014.x ↑21 Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2022). Minoxidil: A Comprehensive Review. Journal Of Dermatological Treatment. 33(4). 1896-1906. Available at: https://doi.org/10.1080/09546634.2021.1945527 ↑22 Sattur, S., Talathi, A., Shetty, G., Arsiwala, S., Pereira, R., Dhoot, D. (2023). Comparative Clinical Study Evaluating The Efficacy And Safety Of Topical 5% Cetosomal Minoxidil And Topical 5% Alcohol-Based Minoxidil Solutions For The Treatment Of Androgenetic Alopecia In Indian Men. Cureus. 15(10). E46568. Available at: https://doi.org/10.7759/cureus.46568 ↑23 Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A Review. Advances In Dermatology And Allergology. 39(3). 472-478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑24 Ramos, M.P., Sinclair, R.D., Kasprzak, M., Miot, H.A. (2020). Minoxidil 1 Mg Oral Versus Minoxidil 5% Topical Solution For The Treatment Of Female-Pattern Hair Loss. Journal Of The American Academy Of Dermatology. 82(1). 252-253. Available at: https://www.jaad.org/article/S0190-9622(19)32666-0/fulltext ↑25 Jiminiez-Couche, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral Minoxidil In Male Androgenetic Alopecia. 81(2). 648-649. Available at: https://doi.org/10.1016/j.jaad.2019.04.054 ↑26 Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: https://www.researchgate.net/profile/Suparuj-Lueangarun/publication/319006716_Efficacy_and_safety_of_oral_minoxidil_5_mg_daily_during_24-week_treatment_in_male_androgenetic_alopecia/links/5b46f1690f7e9b4637cde38b/Efficacy-and-safety-of-oral-minoxidil-5-mg-daily-during-24-week-treatment-in-male-androgenetic-alopecia.pdf ↑27 Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global photographic assessment. *Dermatology and Therapy.* 10(6). 1345–1357. Available at: https://doi.org/10.1007/s13555-020-00448-x ↑28 Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 mg versus 1 mg. 396–399. Available at: https://doi.org/10.1016/j.jaad.2022.01.017 ↑29 Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). 104–109. Available at: https://doi.org/10.1111/ijd.13838 ↑30 Dlova, N.C., Jacobs, T., Singh, S. (2022). Pericardial, Pleural Effusion And Anasarca: A Rare Complication Of Low-Dose Oral Minoxidil For Hair Loss. JAAD Case Reports. 11(28). 94-96. Available at: https://doi.org/10.1016/j.jdcr.2022.07.044 ↑31 Bentivegna, K., Zhou, A.E., Adalsteinsson, J.A., Sloan, B. (2022). Letter In Reply: Pericarditis And Peripheral Edema In A Healthy Man On Low-Dose Oral Minoxidil Therapy. JAAD Case Reports. 20(29). 110-111. Available at: https://doi.org/10.1016/j.jdcr.2022.08.057 ↑32 Hall, D., Charocopos, F., Froer, K.L., Rudolph, W. (1979). ECG Changes During Long-Term Minoxidil Therapy For Severe Hypertension. Archives Of Internal Medicine. 139. 790-794. Available at: https://doi.org/10.1001/archinte.1979.03630440052018 ↑33, ↑34 Bloch, D.L., Carlos, R.M.D. (2025). Side Effects’ Frequency Assessment Of Low Dose Oral Minoxidil In Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: https://doi.org/10.1159/000539969 ↑35 Corvol, P., Michaud, A., Menard, J., Freifeld, M., Mahoudeau, J. (1975). Antiandrogenic Effect Of Spirolactones: Mechanism Of Action. Endocrinology. 97(1). 52-58. Available at: https://doi.org/10.1210/endo-97-1-52 ↑36 Menard, R.H., Stripp, B., Gillette, J.R. (1974). Spironolactone And Testicular Cytochrome P-450: Decreased Testosterone Formation In Several Species And Changes In Hepatic Drug Metabolism. Endocrinology. 94(6). 1628-1636. Available at: https://doi.org/10.1210/endo-94-6-1628 ↑37 Kossor, D.C., Kominami, S., Takemori, S., Colby, H.D. (1992). Destruction Of Testicular Cytochrome P-450 By 7α-Thiospironolactone Is Catalyzed By The 17α-Hydroxylase. The Journal Of Steroid Biochemistry And Molecular Biology. 42(3–4). 421-424. Available at: https://doi.org/10.1016/0960-0760(92)90147-B ↑38 Sinclair, R.D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. International Journal of Dermatology. 57(1). 104–109. Available at: https://doi.org/10.1111/ijd.13838 ↑39, ↑50, ↑55 Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: https://jddonline.com/?post_type=article&p=15021 ↑40, ↑44, ↑47 Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, double-blind, placebo-controlled, parallel-group pilot study. International Journal of Women’s Dermatology. 11(3). e227. Available at: https://doi.org/10.1097/JW9.0000000000000227 ↑41, ↑49, ↑53 Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A Prospective, Single-Center, Parallel-Group, Evaluator Blinded, Randomized Trial. Frontiers in Medicine. 9. Article 905140. Available at: https://doi.org/10.3389/fmed.2022.905140 ↑42, ↑45 Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: https://doi.org/10.1046/j.1365-2265.1997.3761162.x ↑43 Hoedemaker, C., Van Egmond, S., Sinclair, R. (2007). Treatment of female pattern hair loss with a combination of spironolactone and minoxidil. Australasian Journal of Dermatology. 48(1). 43–45. Available at: https://doi.org/10.1111/j.1440-0960.2007.00332.x ↑46, ↑54 Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English Edition). 111(8). 639–649. Available at: https://doi.org/10.1016/j.adengl.2020.03.015 ↑48 NHS, (no date), Pregnancy, Breastfeeding And Fertility While Taking Spironolactone. Available at: https://www.nhs.uk/medicines/spironolactone/pregnancy-breastfeeding-and-fertility-while-taking-spironolactone/ (Accessed: 20 February 2026) ↑51 Collins, M.S., Ali, S., Wiss, I.P., Senna, M.M. (2023). Retrospective Analysis Of The Risk Of Hyperkalaemia In Women Older Than 65 Years Of Age Prescribed Spironolactone For Female-Pattern Hair Loss. British Journal Of Dermatology. 188(3). 429-447. Available at: https://doi.org/10.1093/bjd/ljac068 ↑52 Sevamontree, C., Jintajirapan, S., Phakdeekitcharoen, P., Phakdeekitcharoen, B. (2024). The Prevalence And Risk Factors Of Hyperkalemia In The Outpatient Setting. Int J Nephrol. 2024. 5694131. Available at: https://doi.org/10.1155/2024/5694131 ↑56 Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. Journal of the American Academy of Dermatology. 55(6). 1014–1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007 ↑57 Shapiro, J., & Kaufman, K. D. (2003). Use of finasteride in the treatment of men with androgenetic alopecia (male pattern hair loss). *Journal of Investigative Dermatology Symposium Proceedings.* 8(1). 20–23. Available at: https://doi.org/10.1046/j.1523-1747.2003.12167.x ↑58, ↑61, ↑65, ↑67 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology and Venereology. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑59, ↑68 Gupta, A.K., Talukder, M. (2022). Topical Finasteride For Male And Female Pattern Hair Loss: Is It A Safe And Effective Alternative? Journal of Cosmetic Dermatolology. 21(5). 1841-1848. Available at: https://doi.org/10.1111/jocd.14895 ↑60 Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing The Therapeutic Effects Of Finasteride Gel And Tablet In Treatment Of The Androgenetic Alopecia. Indian J Dermatol Venereol Leprol. 75(1). 47-51. Available at: https://doi.org/10.4103/0378-6323.45220 ↑62 Mazzarella, G., Loconsole, G., Cammisa, G., Mastrolonardo, G., & Vena, G. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy course. Journal of Dermatological Treatment, 8(3), 189–192. https://doi.org/10.3109/09546639709160517 ↑63 Tanglertsampan C. Efficacy and safety of 3% minoxidil versus combined 3% minoxidil / 0.1% finasteride in male pattern hair loss: a randomized, double-blind, comparative study. J Med Assoc Thai. 2012 Oct;95(10):1312-6. Available at: http://www.jmatonline.com/PDF/1312-1316-PB-95-10.pdf ↑64 Rossi, A., Magri, F., D’Arino, A., Pigliacelli, F., Muscianese, M., Leoncini, P., Caro, G., Federico, A., Fortuna, M.C., Carlesimo, M. (2020). Efficacy Of Topical Finasteride 0.5% Vs 17α-Estradiol 0.05% In The Treatment Of Postmenopausal Female Pattern Hair Loss: A Retrospective, Single-Blind Study Of 119 Patients. Dermatol Pract Concept. 10(2). e2020039. Available at: https://doi.org/10.5826/dpc.1002a39 ↑66 Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. International Journal of Clinical Pharmacology and Therapeutics. 54(1). 19-27. Available at: https://doi.org/10.5414/CP202467 ↑69 Mazzarella, G.F., Loconsole, G.F., Cammisa, G.A., Mastrolonardo, G.M. and Vena, G., 1997. Topical Finasteride in the Treatment of Androgenic Alopecia: Preliminary Evaluations After a 16-Month Therapy Course. Journal Of Dermatological Treatment, 8(3), pp.189–192. Available at: https://doi.org/10.3109/09546639709160517 ↑70 Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., Palmieri, R. (2014). A Novel Finasteride 0.25% Topical Solution For Androgenetic Alopecia: Pharmacokinetics And Effects On Plasma Androgen Levels In Healthy Male Volunteers. Int J Clin Pharmacol Ther. 52(10). 842-849. Available at: https://doi.org/10.5414/CP202119 ↑71 Arif, T., Dorjay, K., Adil, M., Sami, M., (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). Available at: 10.2174/157488471266617031011112 ↑72, ↑77, ↑79 Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled Phase II Study To Evaluate The Safety And Efficacy Of Novel Dutasteride Topical Solution (0.01%, 0.02%, And 0.05% W/V) In Male Subjects With Androgenetic Alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑73, ↑78 Nada, E.A., Sharkawy, R.E., Maged, W.M., & Elmagd, M.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Southern Medical Journal, 22, 387-400 ↑74 Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martínez-Moreno, A., Ocampo-Garza, S.S., (2022). Microneedling Plus Topical Dutasteride Solution for Androgenetic Alopecia: A Randomized Placebo-Controlled Study. Journal of the European Academy of Dermatology and Venereology. 36(10). e806–e808. Available at: https://doi.org/10.1111/jdv.18285 ↑75, ↑76 Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M., (2025). Efficacy of Skin Patting and Iontophoresis With Dutasteride Gel in Male and Menopausal Female Androgenetic Alopecia: A Pilot Study. Dermatology and Therapy (Heidelberg). 15(11). 3419–3424. Available at: https://doi.org/10.1007/s13555-025-01532-w ↑80 Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts And Bolts Of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available at: https://doi.org/10.1007/s10439-011-0454-7 ↑81 Lyu, K., Liu, X., Jiang, L., Chen, Y., Lu, J., Zhu, B., Liu, X., Li, Y., Wang, D., Li, S. (2022). The Functions And Mechanisms Of Low-Level Laser Therapy In Tendon Repair (Review). Frontiers in Physiology. 13. 808374. Available at: https://doi.org/10.3389/fphys.2022.808374 ↑82 Satino, J.L., Markou, M. (2003). Hair Regrowth And Increased Hair Tensile Strength Using The HairMax LaserComb For Low-Level Laser Therapy. International Journal of Cosmetic Surgery and Aesthetic Dermatology. 5(2). 113-117. Available at: https://doi.org/10.1089/153082003769591209 ↑83, ↑88 Leavitt, M., Charles, G., Heyman, E. (2009). HairMax LaserComb® Laser Phototherapy Device In The Treatment Of Male Androgenetic Alopecia. Clinical Drug Investigation. 29(5). 283-292. Available at: https://doi.org/10.2165/00044011-200929050-00001 ↑84 Lanzafame, R.J., Blanche, R.R., Bodian, A.B., Chiacchierini, R.P., Fernandez-Obregon, A., Kazmirek, E.R. (2013). The Growth Of Human Scalp Hair Mediated By Visible Red Light Laser And LED Sources In Males. Lasers in Surgery and Medicine. 45(8). 487-495. Available at: https://doi.org/10.1002/lsm.22173 ↑85 Kim, H., Choi, J.W., Kim, J.Y., Shin, J.W., Lee, S.J., Huh, C.H. (2013). Low-Level Light Therapy For Androgenetic Alopecia: A 24-Week, Randomized, Double-Blind, Sham Device-Controlled Multicenter Trial. Dermatologic Surgery. 39(8). 1177-1183. Available at: https://doi.org/10.1111/dsu.12200 ↑86 Friedman, S., Schnoor, P. (2017). Novel Approach To Treating Androgenetic Alopecia In Females With Photobiomodulation (Low-Level Laser Therapy). Dermatologic Surgery. 43(6). 856-867. Available at: https://doi.org/10.1097/DSS.0000000000001114 ↑87 Avci, P., Gupta, G.K., Clark, J., Wikonkal, N., Hamblin, M.R. (2014). Low-Level Laser (Light) Therapy (LLLT) For Treatment Of Hair Loss. Lasers in Surgery and Medicine. 46(2). 144-151. Available at: https://doi.org/10.1002/lsm.22170 ↑89, ↑93 Sondagar, D.M., Mehta, H.H., Agharia, R.S., Jhavar, M.K. (2023). Efficacy Of Low-Level Laser Therapy In Androgenetic Alopecia – A Randomized Controlled Trial. International Journal of Trichology. 15(1). 25-32. Available at: https://doi.org/10.4103/ijt.ijt_5_22 ↑90 Courtois, M., Loussouarn, G., Hourseau, S., Grollier, J.F. (1996). Periodicity In The Growth And Shedding Of Hair. British Journal of Dermatology. 134(1). 47-54 ↑91 Kunz, M., Seifert, B., Trüeb, R.M. (2009). Seasonality Of Hair Shedding In Healthy Women Complaining Of Hair Loss. Dermatology. 219(2). 105-110. Available at: https://doi.org/10.1159/000216832 ↑92 Liu, C., Yang, J., Qu, L., Gu, M., Liu, Y., Gao, J., Collaudin, C., Loussouarn, G. (2014). Changes In Chinese Hair Growth Along A Full Year. International Journal of Cosmetic Science. 36(6). 531-536. Available at: https://doi.org/10.1111/ics.12151 ↑94 Esmat, S.M., Hegazy, R.A., Gawdat, H.I., Abdel Hay, R.M., Allam, R.S., El Naggar, R., Moneib, H. (2017). Low Level Light-Minoxidil 5% Combination Versus Either Therapeutic Modality Alone In Management Of Female Patterned Hair Loss: A Randomized Controlled Study. Lasers In Surgery And Medicine. 49. 835-843. Available at: https://doi.org/10.1002/lsm.22684 ↑95 Munck, A., Gavazzoni, M.F., Trüeb, R.M. (2014). Use Of Low-Level Laser Therapy As Monotherapy Or Concomitant Therapy For Male And Female Androgenetic Alopecia. International Journal Of Trichology. 6(2). 45-49. Available at: https://doi.org/10.4103/0974-7753.138584 ↑96 Hamblin, M.R. (2019). Photobiomodulation For The Management Of Alopecia: Mechanisms Of Action, Patient Selection And Perspectives. Clinical, Cosmetic And Investigational Dermatology. 12. 669-678. Available at: https://doi.org/10.2147/CCID.S184979 ↑97 Egger, A., Resnik, S.R., Aickara, D., Maranda, E., Kaiser, M., Wikramanayake, T.C., Jimenez, J.J. (2020). Examining The Safety And Efficacy Of Low-Level Laser Therapy For Male And Female Pattern Hair Loss: A Review Of The Literature. Skin Appendage Disorders. 6(5). 259-267. Available at: https://doi.org/10.1159/000509001 ↑98 Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., Elsner, P. (2012). Topical Melatonin for Treatment of Androgenetic Alopecia. Int J Trichology. 4(4). 236-245. Available at: https://doi.org/10.4103/0974-7753.111199 ↑99 Zaky, M.S., Abo Khodeir, H., Ahmed, H.-A., Elsaie, M.L., (2021). Therapeutic Implications of Topical Cetirizine 1% in Treatment of Male Androgenetic Alopecia: A Case-Controlled Study. Journal of Cosmetic Dermatology. 20. 1154–1159. Available at: https://doi.org/10.1111/jocd.13940 ↑100 Hossein Mostafa, D., Samadi, A., Niknam, S., Nasrollahi, S.A., Guishard, A., Firooz, A., (2021). Efficacy of Cetirizine 1% Versus Minoxidil 5% Topical Solution in the Treatment of Male Alopecia: A Randomized, Single-blind Controlled Study. Journal of Pharmacy & Pharmaceutical Sciences. 24. 191–199. Available at: https://doi.org/10.18433/jpps31456 ↑101 Bassiouny, E.A., El-Samanoudy, S.I., Abbassi, M.M., Nada, H.R., Farid, S.F., (2023). Comparison Between Topical Cetirizine With Minoxidil Versus Topical Placebo With Minoxidil in Female Androgenetic Alopecia: A Randomized, Double-Blind, Placebo-Controlled Study. Archives of Dermatological Research. 315. 1293–1304. Available at: https://doi.org/10.1007/s00403-022-02512-2 ↑102 Morganti, P., Fabrizi, G., James, B., Bruno, C. (1998). Effect of gelatin-cystine and serenoa repens extract on free radicals level and hair growth. Journal of Applied Cosmetology. 16, 57-64. Available at: https://www.researchgate.net/publication/294672324_Effect_of_gelatin-cystine_and_serenoa_repens_extract_on_free_radicals_level_and_hair_growth ↑103 Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. 57(3), e76-e82. Available at: https://doi.org/10.1111/ajd.12352 ↑104 Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the Hair Growth in Androgenetic Alopecia Subjects – A 16-Week Randomized, Placebo-Controlled Study. Clin Cosmet Investig Dermatol. 16, 3251-3266. Available at: https://doi.org/10.2147/CCID.S435795 ↑105 Fields, J. R., Vonu, P. M., Monir, R. L., Schoch, J. J. (2020). Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review. Dermatol Ther. 33(1). e13202. Available at: https://doi.org/10.1111/dth.13202 Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn More
Cassie Hopton, PhD
Cassie is a multidisciplinary researcher with expertise spanning biochemistry, genetics, and microbiology. She completed a BSc in Biochemistry, including a year-long placement in cancer research, followed by an MScR focused on cyanobacterial genetics. She then earned a PhD in Microbiology at the University of Edinburgh, where her research examined the limits of bacterial survival in ammonia-rich environments.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down