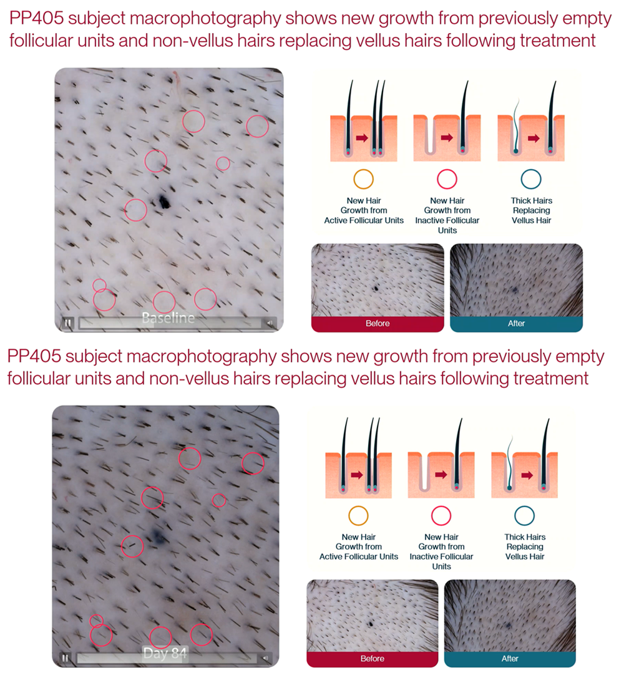
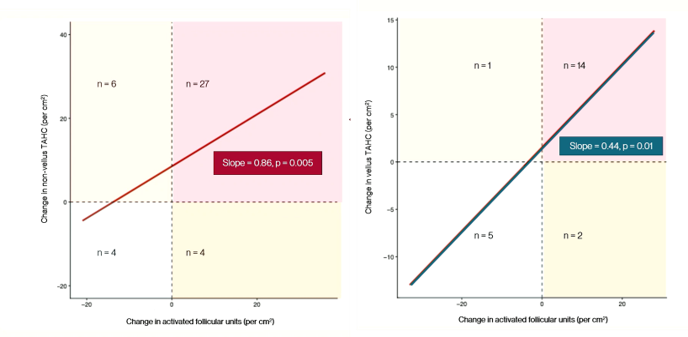
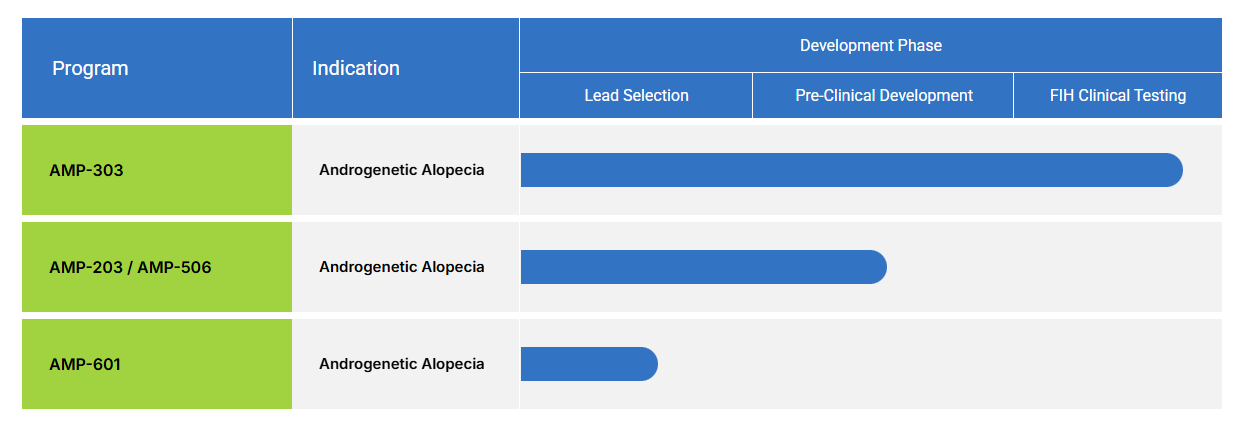
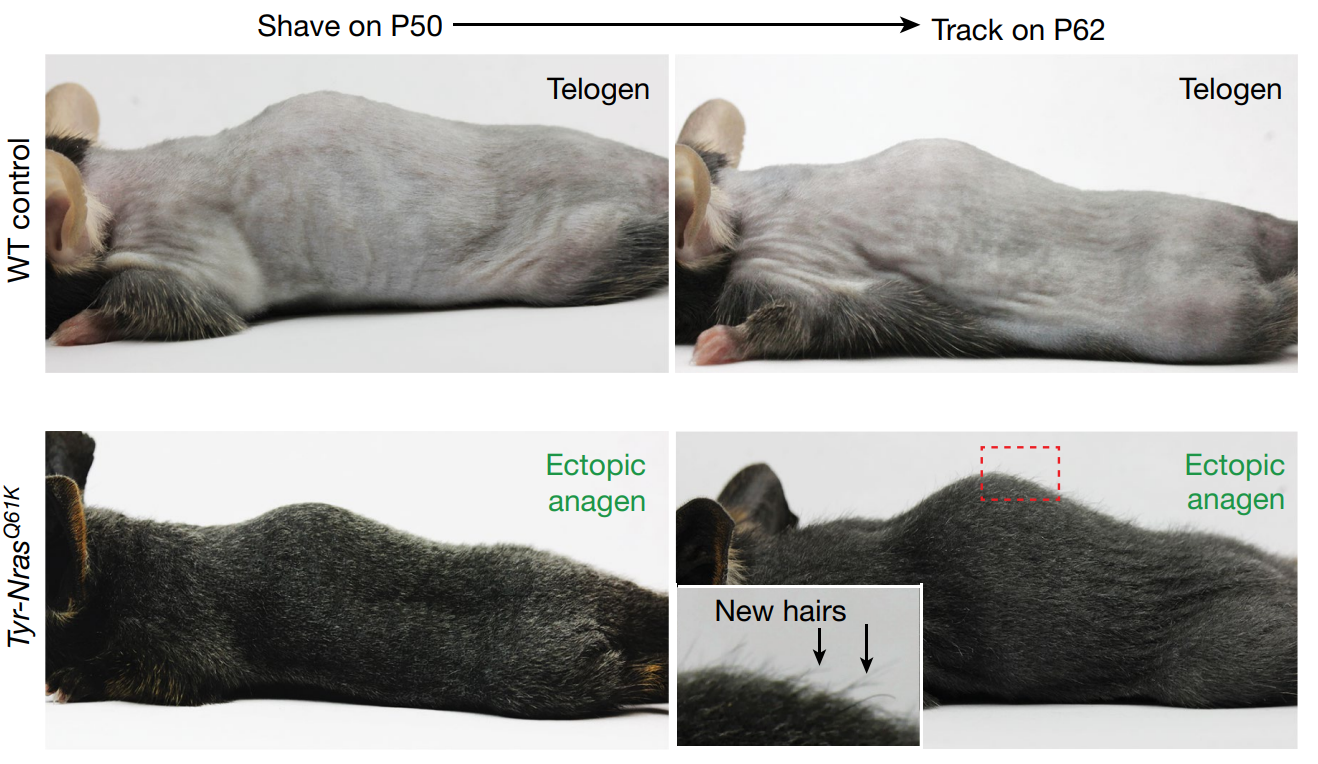
- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
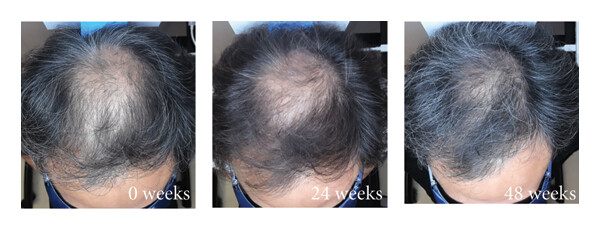
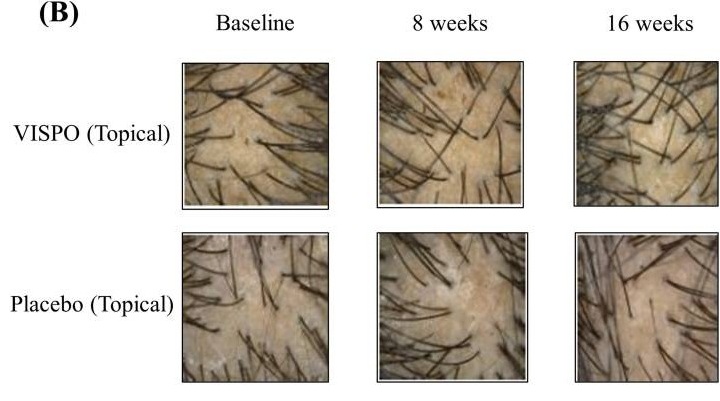
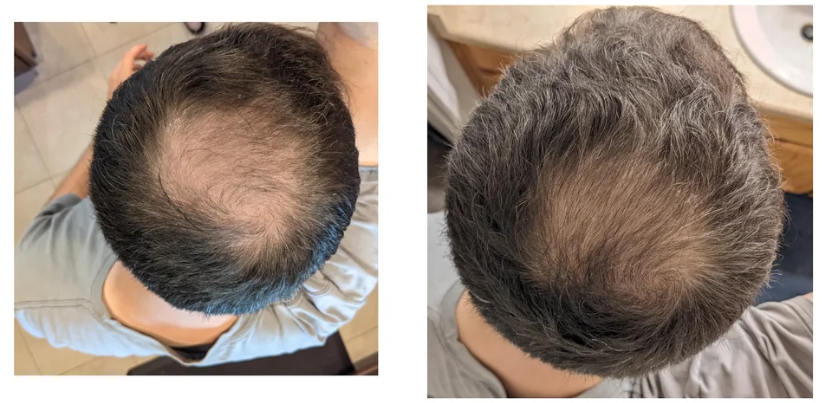
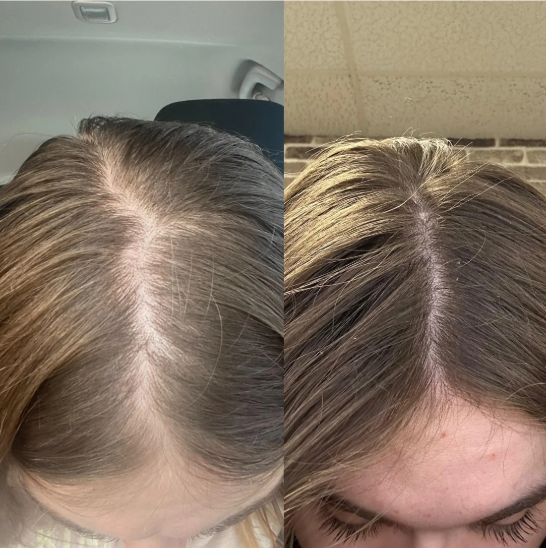
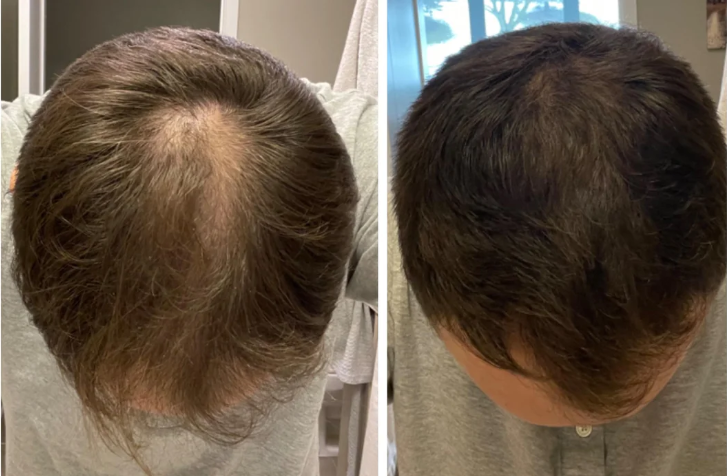
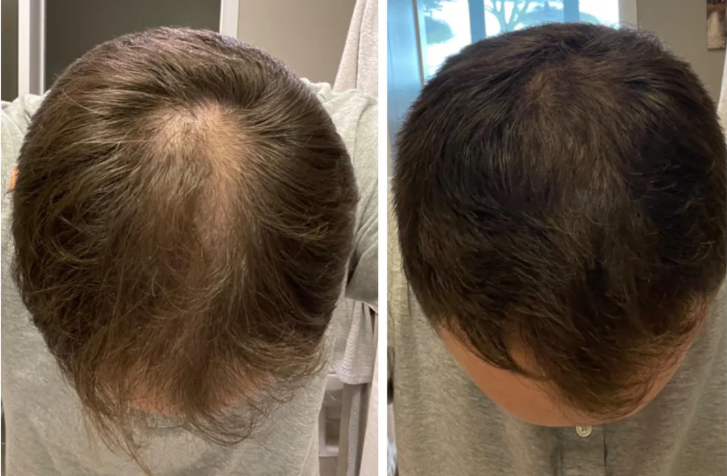
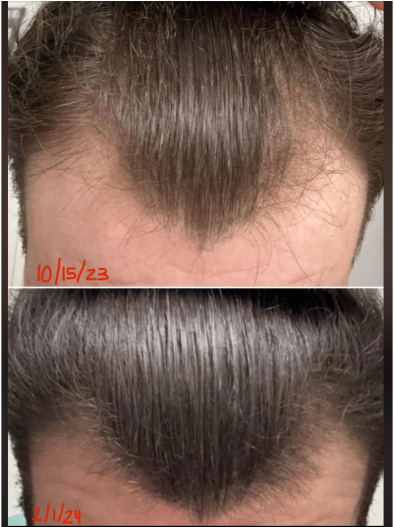
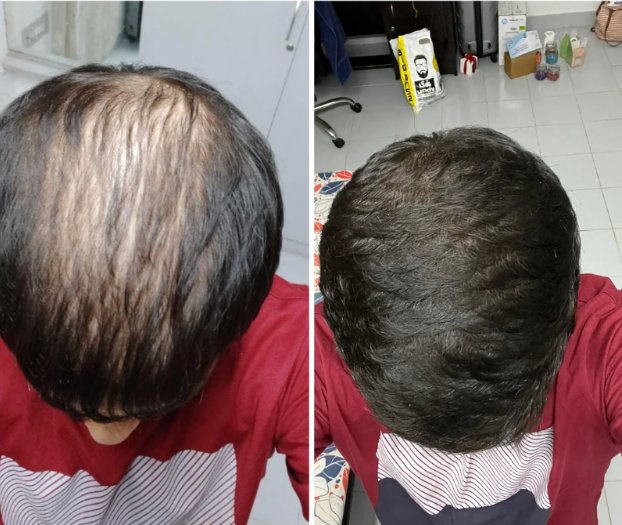
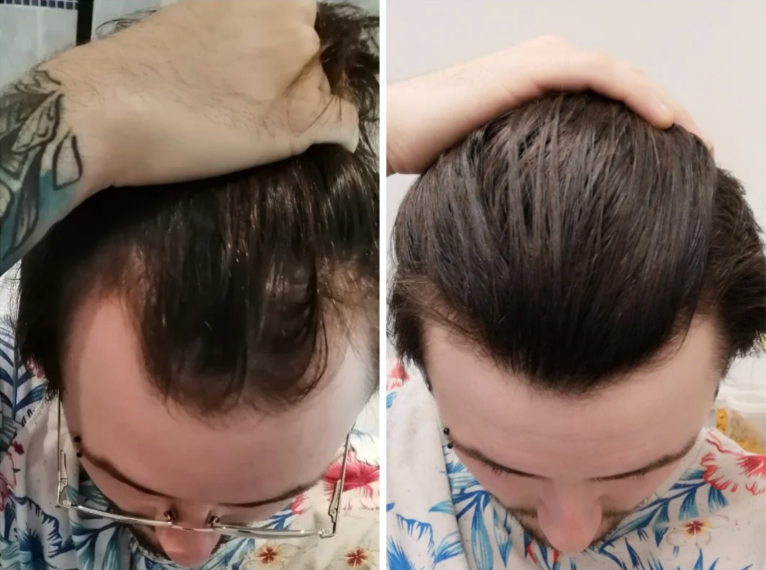
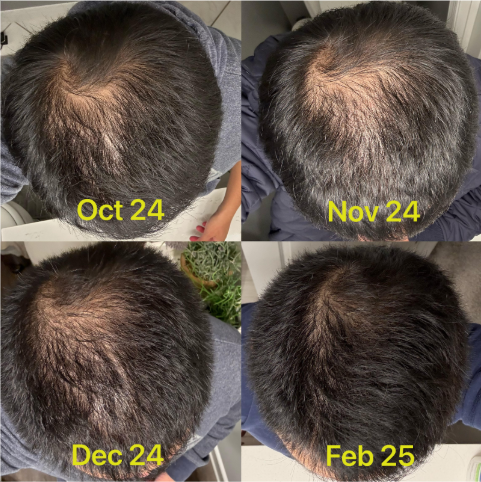
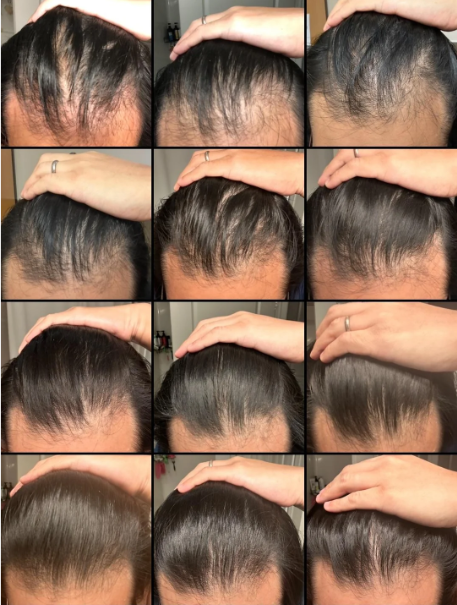
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
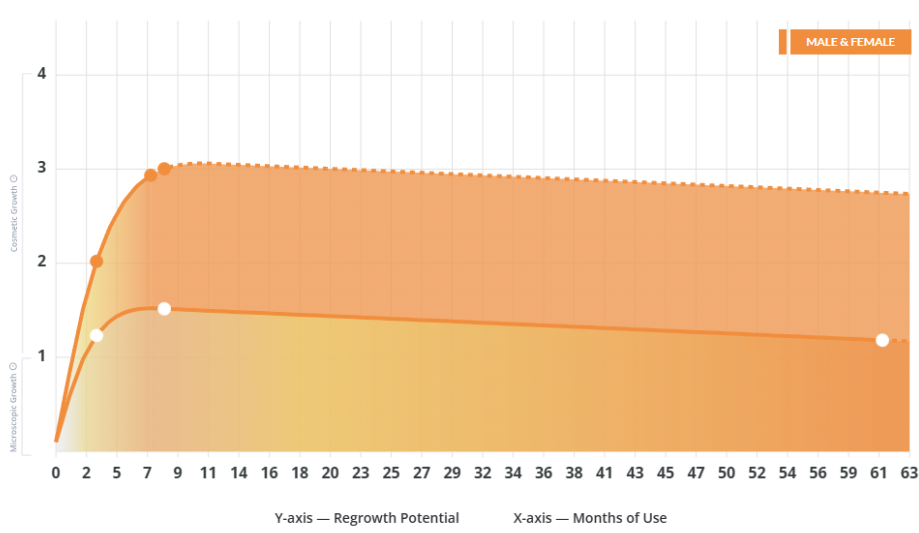
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Does Dandruff Cause Hair Loss?
-
Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
-
Ulo Review: #1 Customized Hair Loss Treatment in 2026
-
10 Best Hair Loss Treatments for Men
-
Best Minoxidil for Men: Top Picks for 2026
-
Does Ozempic Cause Hair Loss?
-
Does Masturbation Cause Hair Loss?
-
6 Best Hair Loss Treatments for Women
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
Articles
Dandruff is an extremely common scalp condition that affects up to half of adults worldwide. It’s often dismissed as a minor cosmetic issue causing flakes on dark clothing, an itchy scalp, or the need for medicated shampoo. But many people with persistent dandruff also notice increased hair shedding, raising an important question: can dandruff actually contribute to hair loss?
The relationship between dandruff and hair loss is more complex than it first appears. “Dandruff” is not a single diagnosis but an umbrella term covering several flaking scalp conditions, some of which involve inflammation. This distinction matters because inflammation can disrupt the normal hair growth cycle and interfere with the effectiveness of hair loss treatments. In some cases, dandruff-related inflammation may push hairs prematurely into the shedding phase (telogen effluvium) or exacerbate existing pattern hair loss.
That said, dandruff alone does not guarantee hair loss. Many people experience flaking without any lasting effect on hair density. The key lies in understanding the underlying cause, which could be microbial overgrowth, nutrient imbalance, or inflammatory skin disease. In this article, we’ll explore what dandruff really is, the causes, what current research says about its potential role in hair shedding and hair loss, and how to treat it effectively so that you can optimize your hair regrowth.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What Is Dandruff?
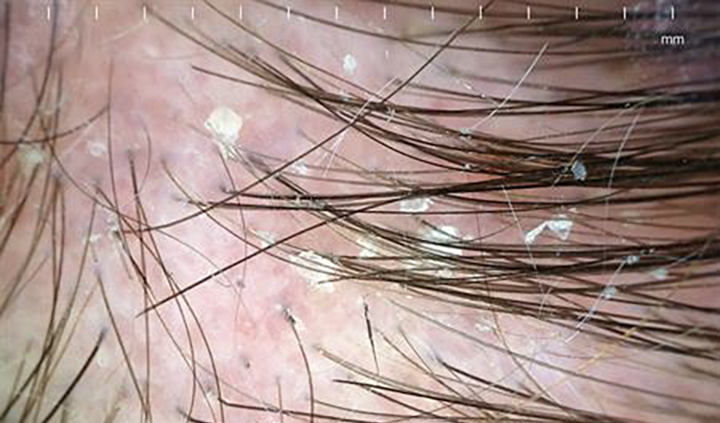
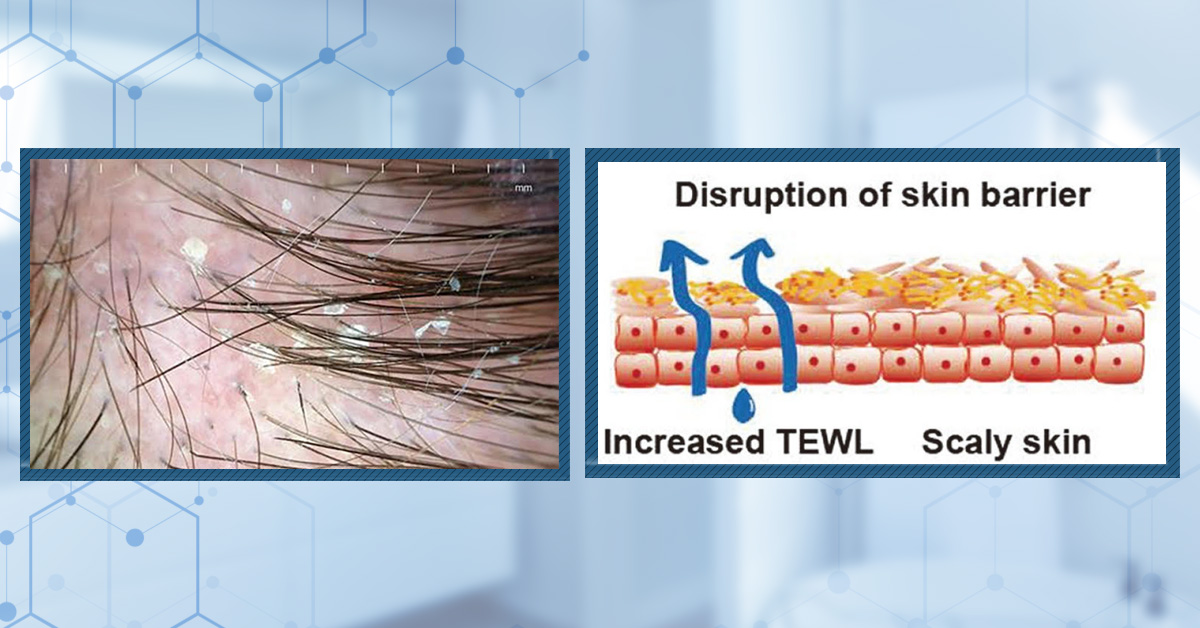
Dandruff is defined by visible scalp flaking caused by increased shedding of outer skin cells. This can cause white, yellow, or grey flakes on the scalp.
Dandruff is typically used as an umbrella term to encompass several conditions that can cause flakiness on the scalp. The types of dandruff and dandruff-like conditions include:
- Dry dandruff: Small, white, or grayish flakes that fall easily from the scalp
- Pityriasis steatoides: Thick, yellowish, greasy flakes that stick to the hair and scalp. The scalp may be itchy, but not red or inflamed.
- Seborrheic dermatitis: Thick, yellowish, greasy flakes and scales. The scalp is inflamed, causing red, greasy, and scaly patches.[1]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. J Clin Investig Dermatol. 3(2). Available at: https://doi.org/10.13188/2373-1044.1000019
- Scalp psoriasis: Itchy, dry, red, and silvery-white plaques on the scalp caused by autoimmune conditions
Because of this, other symptoms may also accompany dandruff. Those with dandruff may experience itchiness, dryness, redness, inflammation, or trichodynia (hair root pain).
Dandruff progresses along a typical timeline that starts at puberty. Peak severity is reached roughly at the age of 20, and dandruff may persist until the age of 50. Beyond 50, the prevalence of dandruff typically declines.[2]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. J Clin Investig Dermatol. 3(2). Available at: https://doi.org/10.13188/2373-1044.1000019
How Common Is It?
Dandruff is an extremely common condition, affecting roughly half of all adults worldwide and at least 50 million Americans.
A study examining populations in the United States and China found that dandruff is most common in people of African American descent, where it affects 81% to 95% of the population. The incidence of dandruff in caucasians is 66% to 82%, while only 30% to 42% of people of Chinese descent are affected by dandruff.[3]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. J Clin Investig Dermatol. 3(2). Available at: https://doi.org/10.13188/2373-1044.1000019
Dandruff vs. Dandruff-Like Conditions
Dandruff is typically used to describe cases where there is flaking on the scalp but no visible inflammation. This is “true” dandruff, an example being pityriasis steatoides.
Seborrheic dermatitis and scalp psoriasis are where inflammation comes into the picture. These conditions are not considered “true” dandruff, but are often described as severe forms of dandruff because they produce dandruff-like symptoms alongside more noticeable irritation.
What Causes Dandruff?
Dandruff may be common, but the cause of dandruff is not well understood. For example, there are some aspects of dandruff susceptibility that we still don’t understand.[4]Del Rosso, J.Q. (2011). Adult Seborrheic Dermatitis: A Status Report On Practical Topical Management. J Clin Aesthet Dermatol. 4(5). Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3100109/
Because dandruff is an umbrella term that can encompass many different conditions, studies have identified several main reasons why dandruff might happen. These studies have shown that flaking occurs alongside microorganism overgrowth, nutrient deficiencies, and autoimmune conditions, potential culprits in causing dandruff.
We’ll go through each of these potential causes of dandruff and explain what the science says about them.
Microorganism Overgrowth
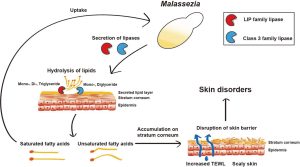
So far, the overgrowth of the fungi Malassezia is one of the best-established factors in causing dandruff.
Malassezia is a yeast naturally found on the skin and is not problematic for some scalps. However, some species of Malassezia release enzymes that break down skin sebum (oil), leaving behind toxic molecules like oleic acid (a fatty acid) and reactive oxygen species (unstable, highly reactive oxygen-containing molecules).[5]Jourdain, R., Moga, A., Magiatis, P., Fontanié, M., Velegraki, A., Papadimou, C., Rahoul, V., Guéniche, A., Chopra, T., Gaitanis, G. (2023). Malassezia Restricta-Mediated Lipoperoxidation: A Novel … Continue reading,[6]Park, M., Park, S., Jung, W.H. (2021). Skin Commensal Fungus Malassezia And Its Lipases. J Microbiol Biotechnol. 31(5). 637-644. Available at: https://doi.org/10.4014/jmb.2012.12048
Both of these molecules can irritate the skin:
- Oleic acid: Disrupts the order of skin lipids, making the skin more porous and permeable. This weakens the skin barrier, promoting infiltration of irritants and microorganisms that can lead to infection.[7]Kováčik, A., Kopečná, M., Hrdinová, I., Opálka, L., Boncheva Bettex, M., Vávrová, K. (2023). Time-Dependent Differences In The Effects Of Oleic Acid And Oleyl Alcohol On The Human Skin … Continue reading, [8]Tanojo, H., Boelsma, E., Junginger, H.E., Ponec, M., Boddé, H.E. (1999). In Vivo Human Skin Permeability Enhancement By Oleic Acid: A Laser Doppler Velocimetry Study. J Control Release. 58(1). … Continue reading
- Reactive oxygen species: React with the lipids of skin cells, causing pores that increase skin permeability and weaken the skin barrier.[9]Liu, H.M., Cheng, M.Y., Xun, M.H., Zhao, Z.W., Zhang, Y., Tang, W., Cheng, J., Ni, J., Wang, W. (2023). Possible Mechanisms Of Oxidative Stress-Induced Skin Cellular Senescence, Inflammation, And … Continue reading They can also disrupt the structure and function of skin cells, and trigger inflammation.[10]Sikder, M.M., Li, X., Akumwami, S., Labony, S.A. (2025). Reactive Oxygen Species: Role In Pathophysiology, And Mechanism Of Endogenous And Dietary Antioxidants During Oxidative Stress. Chonnam Med J. … Continue reading,[11]Kim, M.J., Woo, S.W., Kim, M.S., Park, J.E., Hwang, J.K. (2014). Anti-Photoaging Effect Of Aaptamine In UVB-Irradiated Human Dermal Fibroblasts And Epidermal Keratinocytes. Journal Of Asian Natural … Continue reading
The actions of these molecules can therefore encourage irritation, skin dryness, flakiness, and itchiness. With this in mind, it’s by no coincidence that higher levels of Malassezia have been found in people with more severe dandruff.[12]Rudramurthy, S.M., Honnavar, P., Dogra, S., Yegneswaran, P.P., Handa, S., Chakrabarti, A. (2014). Association Of Malassezia Species With Dandruff. Indian J Med Res. 139(3). 431-437. Available at: … Continue reading,[13]Clavaud, C., Jourdain, R., Bar-Hen, A., Tichit, M., Bouchier, C., Pouradier, F., El Rawadi, C., Guillot, J., Ménard-Szczebara, F., Breton, L., Latgé, J.P., Mouyna, I. (2013). Dandruff Is Associated … Continue reading
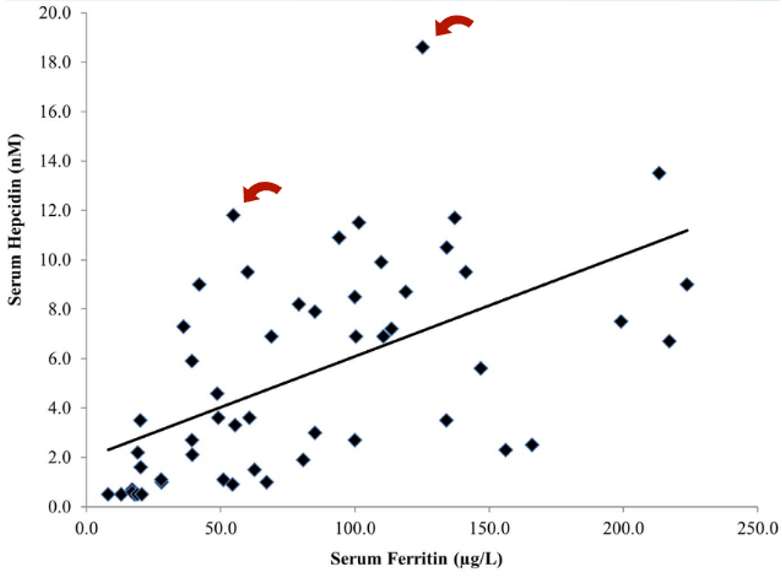
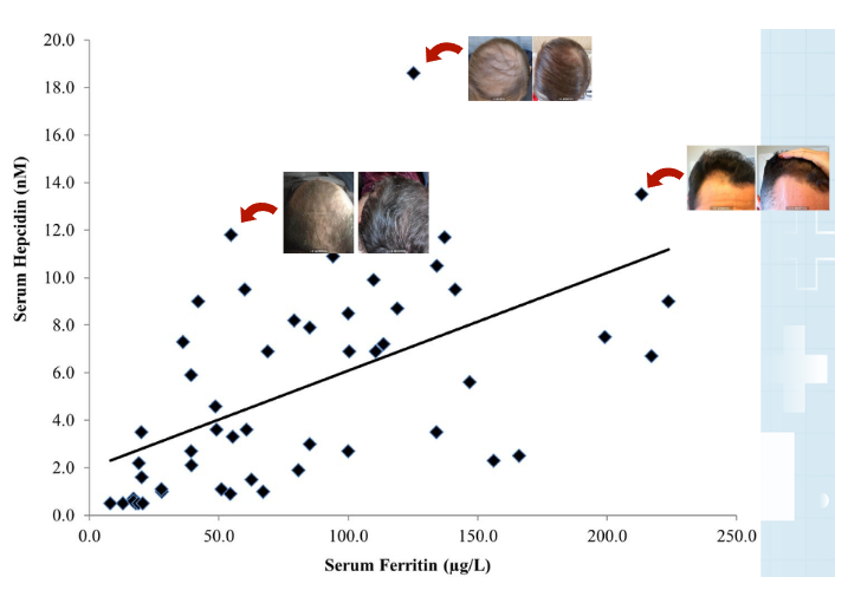
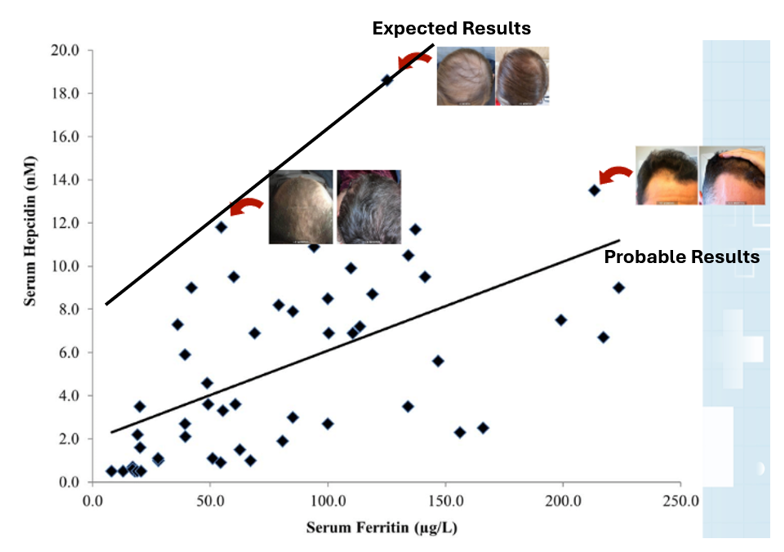
Figure 2: The role of Malassezia in promoting skin disorders (like dandruff). Adapted from Figure 1.[14]Park, M., Park, S., Jung, W.H. (2021). Skin Commensal Fungus Malassezia And Its Lipases. J Microbiol Biotechnol. 31(5). 637-644. Available at: https://doi.org/10.4014/jmb.2012.12048 Image used under the Creative Commons License.
However, it’s not really about the total Malassezia count. It’s about the type of Malassezia species present. For example, counts of Malassezia furfur were found to be the same between people with dandruff and those with healthy scalps.[15]Ashbee, H.R., Ingham, E., Holland, K.T., Cunliffe, W.J. (1993). The Carriage Of Malassezia Furfur Serovars A, B And C In Patients With Pityriasis Versicolor, Seborrhoeic Dermatitis And Controls. … Continue reading In contrast, the counts of Malassezia globosa and Malassezia restricta have been found to be higher on scalps with dandruff.[16]Dawson, T.L. Jr. (2007). Malassezia Globosa And Restricta: Breakthrough Understanding Of The Etiology And Treatment Of Dandruff And Seborrheic Dermatitis Through Whole-Genome Analysis. J Investig … Continue reading
The concept of Malassezia as the primary cause of dandruff is supported by the fact that antifungals often work as a treatment, and improvements in dandruff often correlate with lower Malassezia counts.[17]Piérard-Franchimont, C., Piérard, G.E., Arrese, J.E., De Doncker, P. (2001). Effect Of Ketoconazole 1% And 2% Shampoos On Severe Dandruff And Seborrhoeic Dermatitis: Clinical, Squamometric And … Continue reading
So why do some people have these overgrowths of certain Malassezia species, while others don’t? In truth, we are not entirely sure. It could be that:
- Lowered zinc and vitamin D3 levels, which have been correlated with seborrheic dermatitis, prevent healthier Malassezia diversity.[18]Aktaş Karabay, E., Aksu Çerman, A. (2019). Serum Zinc Levels In Seborrheic Dermatitis: A Case-Control Study. Turk J Med Sci. 49(5). 1503-1508. Available at: https://doi.org/10.3906/sag-1906-72,[19]Rahimi, S., Nemati, N., Shafaei-Tonekaboni, S.S. (2021). Serum Levels Of 25-Hydroxyvitamin D In Patients With Seborrheic Dermatitis: A Case-Control Study. Dermatol Res Pract. 2021. 6623271. Available … Continue reading
- Genetic predispositions leave some with a weakened scalp barrier naturally, opening up the possibility of Malassezia overgrowth.[20]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. J Clin Investig Dermatol. 3(2). Available at: https://doi.org/10.13188/2373-1044.1000019
However, the research so far is limited.
Nutrient Deficiencies
Studies show that deficiencies in vitamin B2, vitamin B3, vitamin B6, and vitamin B7 can cause changes to the skin similar to seborrheic dermatitis and general dermatitis in animals and humans.[21]Jen, M., Yan, A.C. (2010). Syndromes Associated With Nutritional Deficiency And Excess. Clinics In Dermatology. 28(6). 669-685. Available at: https://doi.org/10.1016/j.clindermatol.2010.03.029,[22]Stone, O.J. (1989). Pyridoxine Deficiency And Antagonism Produce Increased Ground Substance Viscosity With Resulting Seborrheic Dermatitis And Increased Tumor Resistance. Med Hypotheses. 30(4). … Continue reading,[23]Redzic, S., Hashmi, M.F., Gupta, V. (2023). Niacin Deficiency. StatPearls [Internet]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK557728/,[24]Balnave, D. (1977). Clinical Symptoms Of Biotin Deficiency In Animals. The American Journal Of Clinical Nutrition. 30(9). 1408-1413. Available at: https://doi.org/10.1093/ajcn/30.9.1408
We have also established that lowered zinc and vitamin D3 levels have been found in those with seborrheic dermatitis. But could nutrient deficiency be a cause of dandruff, or a consequence of Malassezia overgrowth?
Both of these possibilities are plausible, and, unfortunately, we don’t know the answer to this question either.
A cause? Zinc, for example, has anti-inflammatory properties that can reduce sebum production, improve immune response, and initiate wound repair.[25]Demetree, J.W., Safer, L.F., Artis, W.M. (1980). The Effect Of Zinc On The Sebum Secretion Rate. Acta Derm Venereol. 60(2). 166-169. Available at: https://pubmed.ncbi.nlm.nih.gov/6155029/,[26]Lin, P.H., Sermersheim, M., Li, H., Lee, P.H.U., Steinberg, S.M., Ma, J. (2017). Zinc In Wound Healing Modulation. Nutrients. 10(1). 16. Available at: https://doi.org/10.3390/nu10010016,[27]Arribas Lopez, E., Zand, N., Ojo, O., Kochhar, T. (2025). Systematic Review And Meta-Analysis Of The Effect Of Zinc On Wound Healing. BMJ Nutr Prev Health. 8(1). e000952. Available at: … Continue reading,[28]Haase, H., Schomburg, L. (2019). You’d Better Zinc-Trace Element Homeostasis In Infection And Inflammation. Nutrients. 11(9). 2078. Available at: https://doi.org/10.3390/nu11092078 Less sebum means less oleic acid and reactive oxygen species produced by Malassezia. A better immune response means less risk of dandruff-causing infections and a more effective response to irritants.
A consequence? Pathogenic organisms (like Malassezia) are known to compete for nutrients with their host and deplete nutrients at the tissue sites where they grow.[29]Bachman, M.A., Weiser, J.N. (2015). Competition For Metals Among Microbes And Their Host. Trace Metals And Infectious Diseases [Internet]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK569680/,[30]Potrykus, J., Ballou, E.R., Childers, D.S., Brown, A.J.P. (2014). Conflicting Interests In The Pathogen–Host Tug Of War: Fungal Micronutrient Scavenging Versus Mammalian Nutritional Immunity. PLoS … Continue reading The inflammation caused by overgrowth can also increase nutritional demand (to strengthen the immune response).[31]Gupta, S., Read, S.A., Shackel, N.A., Hebbard, L., George, J., Ahlenstiel, G. (2019). The Role Of Micronutrients In The Infection And Subsequent Response To Hepatitis C Virus. Cells. 8(6). 603. … Continue reading,[32]Roth-Walter, F., Berni Canani, R., O’Mahony, L., Peroni, D., Sokolowska, M., Vassilopoulou, E., Venter, C. (2024). Nutrition In Chronic Inflammatory Conditions: Bypassing The Mucosal Block For … Continue reading This creates a situation where pathogenic organisms cause nutrient deficiencies, while also increasing the requirement for more nutrients.
All-Natural Hair Supplement + Topical
The top natural ingredients for hair growth, all in one serum & supplement.
Take the next step in your hair growth journey with a world-class natural serum & supplement. Ingredients, doses, & concentrations built by science.

Autoimmunity
Autoimmunity is when your body’s defense system mistakenly attacks your own healthy cells, causing inflammation. Since inflammation can produce irritation, this helps explain why autoimmune scalp conditions can sometimes be responsible for dandruff-like symptoms.
Several autoimmune or immune-related conditions can produce dandruff-like conditions:
- Scalp eczema: Known to cause dry, itchy, and irritated skin at the scalp, and is often triggered by genetics, environmental factors (i.e., skin barrier dysfunctions, hypersensitivity to irritants, allergens), or microbial imbalances.[33]Magnifico, I., Petronio Petronio, G., Venditti, N., Cutuli, M.A., Pietrangelo, L., Vergalito, F., Mangano, K., Zella, D., Di Marco, R. (2020). Atopic Dermatitis As A Multifactorial Skin Disorder. Can … Continue reading
- Scalp psoriasis: A condition characterized by thick, scaly, itchy, or painful patches of skin. It is often triggered by genetics, infections, stress, or injury.[34]Armstrong, A.W., Read, C. (2020). Pathophysiology, Clinical Presentation, And Treatment Of Psoriasis: A Review. JAMA. 323(19). 1945-1960. Available at: https://doi.org/10.1001/jama.2020.4006
- Scarring alopecia: A disease where the immune system destroys hair follicles, replacing them with scar tissue, causing permanent hair loss often accompanied by severe dandruff-like scales, itching, burning, and pain.[35]Lepe, K., Nassereddin, A., Syed, H.A., et al. (2024). Lichen Planopilaris. StatPearls [Internet]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK470325/
- Seborrheic dermatitis: This is also a condition that isn’t classed as autoimmune. But, studies show that seborrheic dermatitis can produce specific immune dysregulations, which exaggerate inflammation.[36]Navarro Triviño, F.J., Velasco Amador, J.P., Rivera Ruiz, I. (2025). Seborrheic Dermatitis Revisited: Pathophysiology, Diagnosis, And Emerging Therapies-A Narrative Review. Biomedicines. 13(10). … Continue reading
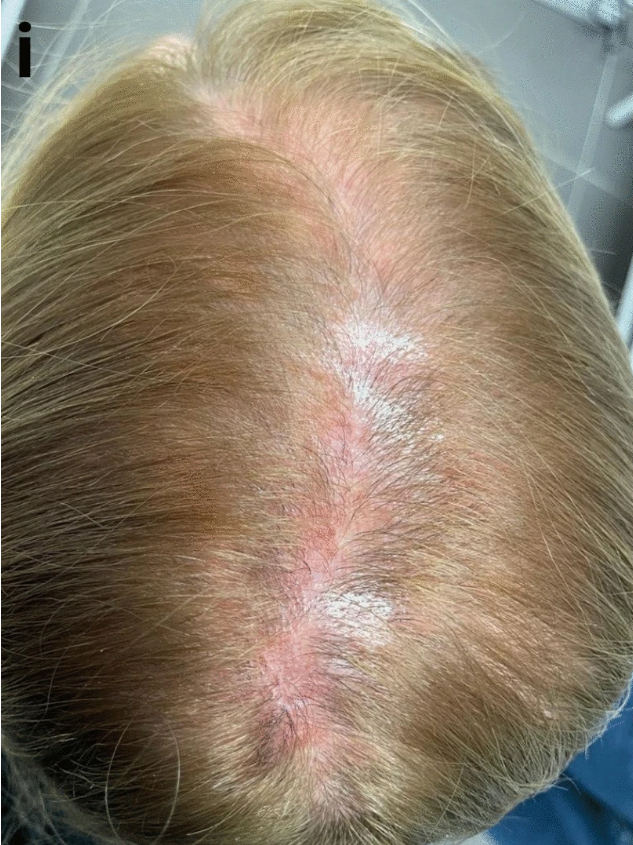
Figure 3: Patient with lichen planopilaris, the most frequent primary scarring alopecia. Adapted from Figure 9.[37]Umar, S., Kan, P., Carter, M.J. (2022). Lichen Planopilaris Responsive To A Novel Phytoactive Botanical Treatment: A Case Series. Dermatology And Therapy. 12. 1697-1710. Available at: … Continue reading Image used under the Creative Commons License.
Dandruff-like symptoms caused by autoimmune conditions tend to be more persistent and treatment-resistant than other causes of dandruff. Why? Because the root cause of the problem is an internal, chronic issue. Compared to an external overgrowth of Malassezia, this makes it a lot harder to treat.
Autoimmune diseases that cause dandruff-like symptoms usually arise in genetically susceptible people after a combination of environmental exposures. Contributing factors may include diet, lifestyle (i.e., activity level), or certain environmental triggers (i.e., allergies, an irritating beauty product).
The typical Western diet of processed foods, sugar, and unhealthy fats, in addition to smoking, are two well‑studied examples that can negatively impact the gut microbiome and keep the body in a low‑grade inflammatory state.[38]Manzel, A., Muller, D.N., Hafler, D.A., Erdman, S.E., Linker, R.A., Kleinewietfeld, M. (2014). Role Of “Western Diet” In Inflammatory Autoimmune Diseases. Curr Allergy Asthma Rep. 14(1). … Continue reading,[39]Perricone, C., Versini, M., Ben-Ami, D., Gertel, S., Watad, A., Segel, M.J., Ceccarelli, F., Conti, F., Cantarini, L., Bogdanos, D.P., Antonelli, A., Amital, H., Valesini, G., Shoenfeld, Y. (2016). … Continue reading This makes it easier for the immune system to start attacking the body’s own tissues in vulnerable people.
Does Dandruff Cause Hair Loss?
This is the big question. The short answer is: we’re not sure. Researchers have not yet identified the exact mechanism by which dandruff connects to hair loss.
Having dandruff does not guarantee hair loss, but inflammation associated with dandruff can increase shedding. We do know that there are two hair loss conditions commonly tied to dandruff:
- Telogen effluvium
- Pattern hair loss
Let’s look deeper into the science behind this.
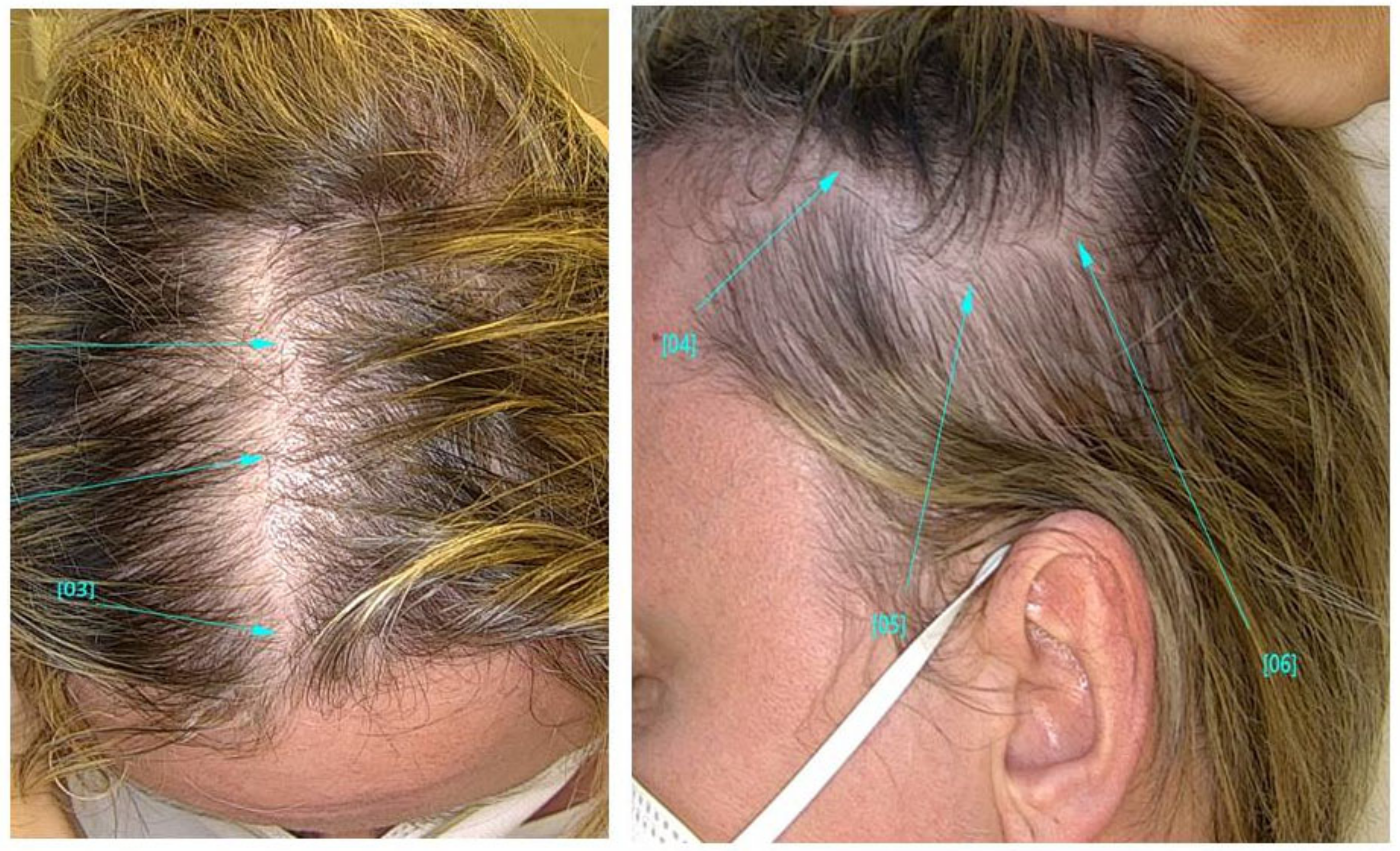
Dandruff and Telogen Effluvium
Telogen effluvium is a condition where there are too many hairs in the telogen phase, causing premature hair shedding. This usually happens after a “negative event”, like high fever, illness, childbirth (postpartum), severe surgical trauma, sudden weight loss, thyroid issues, iron deficiencies, and starting or stopping medication.[40]Hussain, N., Agarwala, P., Iqbal, K., et al. (2022). A Systematic Review Of Acute Telogen Effluvium, A Harrowing Post-COVID-19 Manifestation. J Med Virol. 94. 1391-1401. Available at: … Continue reading,[41]Bin Dayel, S., Hussein, R.S., Atia, T., Abahussein, O., Al Yahya, R.S., Elsayed, S.H. (2024). Is Thyroid Dysfunction A Common Cause Of Telogen Effluvium?: A Retrospective Study. Medicine. 103(1). … Continue reading,[42]Kang, D.H., Kwon, S.H., Sim, W.Y., Lew, B.L. (2024). Telogen Effluvium Associated With Weight Loss: A Single Center Retrospective Study. Ann Dermatol. 36(6). 384-388. Available at: … Continue reading
Figure 4: A patient with telogen effluvium following COVID infection. Adapted from Figure 2.[43]Monari, P., Gualdi, G., Bettoni, G., Costa, R., Ragni, G., Zani, F., Bianchi, G., Casella, S., Casella, E., Crippa, M., Calzavara Pinton, P., Di Nicola, M., Porreca, A., Amerio, P., Guizzi, P. … Continue reading Image used under the Creative Commons License.
Hair growth and shedding are a cycle, and by causing premature shedding, telogen effluvium disrupts this cycle, leaving a gap between when hairs fall out and grow in. The good news is that telogen effluvium is temporary, and when the cause is identified, it can be treated appropriately. Usually, hair shedding stops within 3 to 6 months of removing the trigger, and regrowth will occur over the following 3 to 6 months.[44]Malkud, S. (2015). Telogen Effluvium: A Review. J Clin Diagn Res. 9(9). WE01-3. Available at: https://doi.org/10.7860/JCDR/2015/15219.6492
Could dandruff be a “negative event”? Studies show that excess dandruff can, in fact, disrupt the hair cycle. This suggests dandruff may be able to trigger telogen effluvium.
The process from dandruff to hair loss could occur like this:
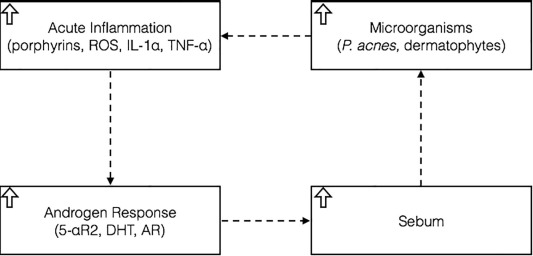
- Microbes like Malassezia and Propionibacterium acnes colonize the scalp and feed on sebum.
- The toxic by-products (like porphyrins) produced by sebum feeding by Malassezia and P. acnes react with UV radiation and increase free radical activity, causing oxidative stress and inflammation at the scalp.
- The body responds to this stress by producing more sebum and accelerating turnover of the scalps outer skin layer, creating more dead skin cells.
- The additional sebum acts as more food for Malassezia and P. acnes, increasing their numbers.
- The cycle repeats as microbial numbers grow, and inflammation increases.
- Eventually, this chronic inflammation causes hairs to shed before completion of the normal growth cycle (telogen effluvium).
Research shows that Malassezia is associated with hair shedding.[45]Nematian, J., Ravaghi, M., Gholamrezanezhad, A., Nematian, E. (2006). Increased Hair Shedding May Be Associated With The Presence Of Pityrosporum Ovale. Am J Clin Dermatol. 7(4). 263-266. Available … Continue reading
Figure 5: A feedback loop triggering excessive hair shedding. Adapted from Figure 2.[46]English, R.S. (2018). A Hypothetical Pathogenesis Model For Androgenic Alopecia: Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors. Medical Hypotheses. 111. 73-81. … Continue reading Image used under the Creative Commons License.
Dandruff and Pattern Hair Loss
Pattern hair loss is not temporary. Known as androgenic alopecia, this is a chronic, progressive hair loss condition that is triggered by genetics, androgen levels, and the scalp environment.[47]Chen, S., Li, L., Ding, W., Zhu, Y., Zhou, N. (2025). Androgenetic Alopecia: An Update On Pathogenesis And Pharmacological Treatment. Drug Des Devel Ther. 19. 7349-7363. Available at: … Continue reading
Pattern hair loss progresses through cycles of hair growth (the anagen phase) and hair rest (the telogen phase), followed by shedding (the exogen phase). However, in each cycle, the anagen phase becomes shorter, and the telogen phase becomes longer. Over time, a progressively finer hair shaft is produced (follicle miniaturization). Hair follicles need to complete shedding before they miniaturize. So in essence, if there is no hair shedding, there is no follicle miniaturization, and androgenic alopecia will not progress.
We’ve discussed how dandruff could trigger telogen effluvium and excess shedding. It’s reasonable, then, to speculate that this excess shedding could also accelerate hair follicle miniaturization in those with androgenic alopecia. More shedding means more hair follicle miniaturization. This hasn’t been demonstrated in research, but it is a plausible scenario.
For diffuse alopecia conditions (like telogen effluvium), studies show that there is a positive correlation between dandruff severity and the number of hairs in the telogen phase.[48]Piérard-Franchimont, C., Xhauflaire-Uhoda, E., Loussouarn, G., Saint Léger, D., Piérard, G.E. (2006). Dandruff-Associated Smouldering Alopecia: A Chronobiological Assessment Over 5 Years. Clin Exp … Continue reading Perhaps dandruff could also exacerbate telogen phase lengthening caused by androgenic alopecia as well.
So while dandruff does not cause androgenic alopecia, it could exaggerate hair loss caused by this condition, and so addressing any dandruff or inflammation is crucial to support better regrowth outcomes when treating this condition.
Why Resolving Scalp Inflammation Matters for Hair Growth Treatments
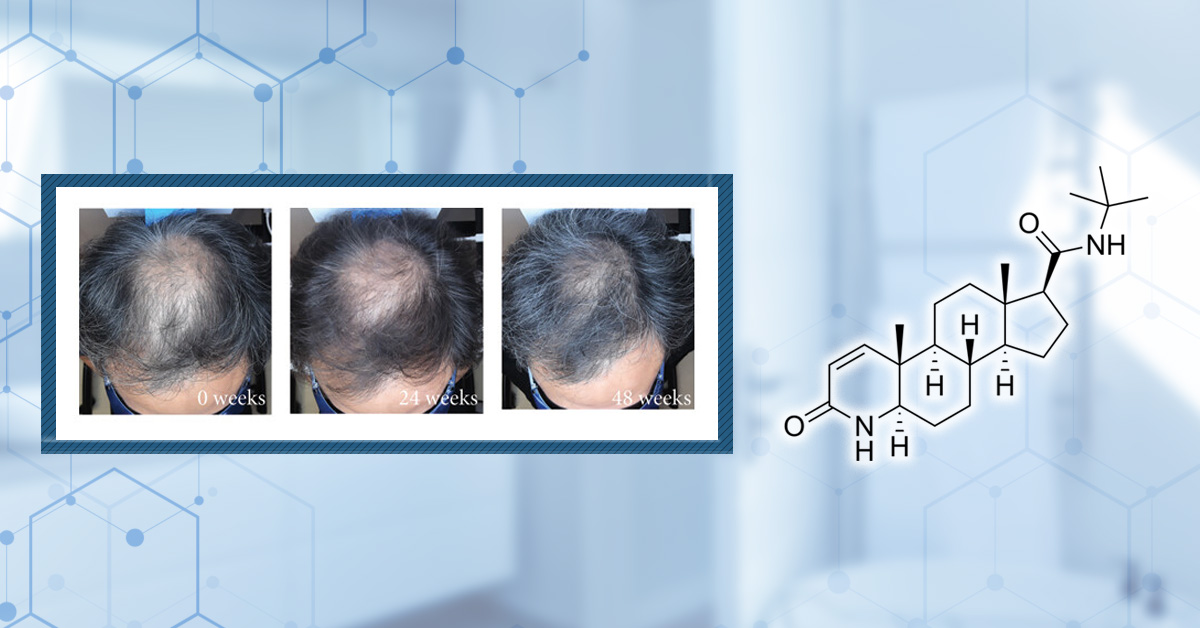
There are several FDA-approved treatments for androgenic alopecia. However, inflammation at the scalp could stop these treatments from working effectively.
In a study of 64 men with androgenic alopecia, low-light laser therapy (LLLT) was found to reduce inflammatory molecules at the scalp. Subsequent use of LLLT before 2% minoxidil was found to boost hair regrowth similar to use of 5% minoxidil.[49]Mahe, Y.F., Cheniti, A., Tacheau, C., Antonelli, R., Planard-Luong, L., de Bernard, S., Buffat, L., Barbarat, P., Kanoun-Copy, L. (2021). Low-Level Light Therapy Downregulates Scalp Inflammatory … Continue reading The result was just a correlation (i.e., other factors could have boosted hair regrowth other than reduced inflammation), but suggests a link between lowered inflammation markers and improved hair regrowth outcomes.
This evidence suggests that if you are looking for hair loss treatments and you have dandruff or an inflamed scalp, don’t ignore it! It’s important to treat dandruff and inflammation first, or alongside regrowth regimens.
Identify Your Dandruff Driver
Knowing the cause of your dandruff is the first step in choosing the right treatment.
Do you experience:
- Small, white, powdery, and dry flakes?
- A scalp that feels tight, itchy, and dry?
Likely cause: Dehydrated scalp
Do you experience:
- Large, yellow or white flakes that feel greasy or waxy?
- A scalp feels greasy quickly, and oily hair?
- Intense itching, redness, or scaly patches?
Likely cause: Too much sebum and microbial overgrowth
Do you experience:
- Persistent redness on the scalp?
- Persistent pain in the scalp area?
- Persistent “dandruff” that doesn’t resolve with the use of shampoos?
Likely cause: An underlying autoimmune condition
But you shouldn’t self-diagnose. Speak with a dermatologist if you are experiencing dandruff or dandruff-like symptoms. A healthcare professional can often diagnose dandruff simply by looking at your hair and scalp, and may be able to advise on possible causes.
Treatments That Could Work
Once you’ve identified the likely culprit of your dandruff, you can now seek out the most appropriate treatments. We’ll walk through the different types of treatments available for each cause of dandruff.
For Microorganism-Driven Dandruff
When dandruff is driven by the overgrowth of microorganisms, the aim is to lower the levels of these organisms on the scalp and make it a less hospitable place for them to grow. There are several products available to support this mission.
Zinc pyrithione 1% shampoo
Zinc pyrithione is a potent antimicrobial and antifungal agent. It is a very common ingredient in shampoos targeted at dandruff, like Head & Shoulders products.
Zinc pyrithione acts in three key ways to disrupt microbial growth:
- It increases the zinc levels and copper levels within individual cells of microbes, like Malassezia, which can damage the structure and formation of iron-sulphur clusters.[50]Park, M., Cho, Y.J., Lee, Y.W., Jung, W.H. (2018). Understanding The Mechanism Of Action Of The Anti-Dandruff Agent Zinc Pyrithione Against Malassezia Restricta. Sci Rep. 8(1). 12086. Available at: … Continue reading,[51]Reeder, N.L., Xu, J., Youngquist, R.S., Schwartz, J.R., Rust, R.C., Saunders, C.W. (2011). The Antifungal Mechanism Of Action Of Zinc Pyrithione. Br J Dermatol. 165 Suppl 2. 9-12. Available at: … Continue reading
- In addition to damaging iron-sulphur clusters, it disrupts the synthesis process for iron-sulphur clusters.[52]Park, M., Cho, Y.J., Lee, Y.W., Jung, W.H. (2018). Understanding The Mechanism Of Action Of The Anti-Dandruff Agent Zinc Pyrithione Against Malassezia Restricta. Sci Rep. 8(1). 12086. Available at: … Continue reading
- It disrupts the production of lipid-eating enzymes (like those needed to “eat” sebum).[53]Park, M., Cho, Y.J., Lee, Y.W., Jung, W.H. (2018). Understanding The Mechanism Of Action Of The Anti-Dandruff Agent Zinc Pyrithione Against Malassezia Restricta. Sci Rep. 8(1). 12086. Available at: … Continue reading
Why is this important? Iron-sulphur clusters are contained within mitochondria, the “powerhouse of the cell”. Iron-sulphur clusters, alongside other cellular components, are needed to produce energy from mitochondria. Without energy, the microbes can not grow or survive.
The clinical evidence shows that zinc pyrithione is effective against dandruff and may even support hair regrowth, possibly by supporting a healthier scalp environment.
Study Subjects and Treatment Outcome Notes Marks et al. 1985.[54]Marks, R., Pearse, A.D., Walker, A.P. (1985). The Effects Of A Shampoo Containing Zinc Pyrithione On The Control Of Dandruff. Br J Dermatol. 112(4). 415-422. Available at: … Continue reading 32 subjects with dandruff, one half of the head was washed with 1% zinc pyrithione and the other half without. Progressive reduction in dandruff on the side of the head washed with zinc pyrithione, and the difference between the placebo side was statistically significant. Small subject number, but results suggest a positive influence that may be generalizable to larger groups. Berger et al. 2003.[55]Berger, R.S., Fu, J.L., Smiles, K.A., Turner, C.B., Schnell, B.M., Werchowski, K.M., Lammers, K.M. (2003). The Effects Of Minoxidil, 1% Pyrithione Zinc And A Combination Of Both On Hair Density: A … Continue reading 200 men with Hamilton-Norwood type III vertex or type IV baldness, treated with either 1% zinc pyrithione, 5% minoxidil, or a combination of both. Minoxidil produced the most substantial outcome, but all treatments resulted in a significant increase in hair counts. The benefit of the 1% pyrithione zinc shampoo used alone tended to be apparent only to the investigator, users did not notice a cosmetic change. Schwartz et al. 2013.[56]Schwartz, J.R., Bacon, R.A., Shah, R., Mizoguchi, H., Tosti, A. (2013). Therapeutic Efficacy Of Anti-Dandruff Shampoos: A Randomized Clinical Trial Comparing Products Based On Potentiated Zinc … Continue reading 620 subjects with scalp flaking, treated with either a “potentiated” zinc pyrithione (a formula that maximizes delivery of zinc pyrithione at the scalp), or a dual-active shampoo containing zinc pyrithione and another antifungal, climbazole Potentiated zinc pyrithione was more effective than the dual-active shampoo at reducing flaking. The treatment was used for 4 weeks, so only short-term benefits can be seen here. Zinc pyrithione shampoos are widely available. You should be able to purchase this shampoo easily over-the-counter at any pharmacy or supermarket.
Ketoconazole 1% or 2% shampoo
Ketoconazole is another antifungal medication. Ketoconazole shampoo is often recommended in conjunction with hair loss products, such as minoxidil or finasteride. This is because it doesn’t just help reduce dandruff, it can also reduce androgens at the scalp, which drive androgenic alopecia.
Ketoconazole fights pathogenic scalp microbes in two key ways:
- It blocks the production of a lipid needed to maintain the outer structure (cell wall) of fungi cells, which ultimately damages them.[57]Tynes, B.E., Johnson, C.D., Vaish, M.H., Abbott, B., Vučenović, J., Varrassi, G., Potharaju, P., Lopez Torres, Y., Lee, Z., Ahmadzadeh, S., Shekoohi, S., Kaye, A.D. (2024). Ketoconazole Shampoo For … Continue reading
- It changes the lipids present on the scalp, and reduces sebum production, both of which block the “food” available for Malassezia overgrowth and promote the growth of more friendly microbes.[58]Goularte-Silva, V., Paulino, L.C. (2022). Ketoconazole Beyond Antifungal Activity: Bioinformatics-Based Hypothesis On Lipid Metabolism In Dandruff And Seborrheic Dermatitis. Exp Dermatol. 31. … Continue reading
Like zinc pyrithione, the clinical evidence for ketoconazole shows that it is an effective treatment against dandruff, with 2% ketoconazole having the greatest impact.
Study Subjects and Treatment Outcome Notes Piérard-Franchimont et al. 2001.[59]Piérard-Franchimont, C., Piérard, G.E., Arrese, J.E., De Doncker, P. (2001). Effect Of Ketoconazole 1% And 2% Shampoos On Severe Dandruff And Seborrhoeic Dermatitis: Clinical, Squamometric And … Continue reading 66 subjects with severe dandruff or seborrhoeic dermatitis, treated with either 1% ketoconazole or 2% ketoconazole Both resulted in clear benefits, but flakiness, Malassezia count, and dandruff score were reduced more with 2% ketoconazo le compared to 1% ketoconazole.
Treatment lasted only 4 weeks. Longer trials are needed to determine whether this is a long-term solution. Also, the results may not be relevant to milder cases. Piérard-Franchimont et al. 2002.[60]Piérard-Franchimont, C., Goffin, V., Decroix, J., Piérard, G.E. (2002). A Multicenter Randomized Trial Of Ketoconazole 2% And Zinc Pyrithione 1% Shampoos In Severe Dandruff And Seborrheic … Continue reading 331 subjects with severe dandruff or seborrhoeic dermatitis, treated with either 1% ketoconazole or 2% ketoconazole Both treatments resulted in clear benefits, but the benefits were greater with 2% ketoconazole. There was a 73% improvement in total dandruff severity score with 2% ketoconazole, and instances of disease reccurrence was fewer. Treatment lasted only 4 weeks. Longer trials are needed to determine whether this is a long-term solution. Also, the results may not be relevant to milder cases. Lorette and Ermosilla 2006.[61]Lorette, G., Ermosilla, V. (2006). Clinical Efficacy Of A New Ciclopiroxolamine/Zinc Pyrithione Shampoo In Scalp Seborrheic Dermatitis Treatment. Eur J Dermatol. 16(5). Available at: … Continue reading 189 subjects with seborrheic dermatitis, treated with either 1.5% ciclopiroxolamine and 1% zinc pyrithione, or 2% ketoconazole All treatments reduced itching, redness, and inflammation, and it was found that both treatments were efficient in treating seborrheic dermatitis. Whether the results are relevant to “true” dandruff cases and cases of milder dandruff is unclear. Ketoconazole at 1% is available over-the-counter, but 2% requires a prescription. If you have mild to moderate dandruff, try 1% zinc pyrithione or 1% ketoconazole shampoo first. If you don’t see results or have more severe dandruff, contact your physician to see whether you can escalate to 2% ketoconzole.
Tea tree oil 5% shampoo
For those who seek out more natural remedies, tea tree oil is a widely used ingredient for treating dandruff naturally. It can be found in shampoos, conditioners, scalp oils, hair masks, lotions, and many other products of this sort.
Tea tree oil contains a compound known as terpinen-4-ol. This compound can disrupt the cell membrane of fungal cells, which disrupts their growth and ability to replicate.[62]Yu, D., Wang, J., Shao, X., Xu, F., Wang, H. (2015). Antifungal Modes Of Action Of Tea Tree Oil And Its Two Characteristic Components Against Botrytis Cinerea. J Appl Microbiol. 119(5). 1253-1262. … Continue reading
Unlike zinc pyrithione or ketoconazole, there is only one study supporting the use of tea tree oil against dandruff. However, it’s a well-designed study, so the validity of the conclusions are strong.
In this randomized, single-blind, and controlled study, 126 subjects with mild to moderate dandruff were treated either with 5% tree tea oil shampoo or a placebo. After 4 weeks, areas of the scalp showed a 41% improvement in severity score with 5% tea tree oil, while just a 11% improvement in the placebo group. Itchiness and greasiness were also significantly reduced with the 5% tea tree oil shampoo.[63]Satchell, A.C., Saurajen, A., Bell, C., Barnetson, R.S. (2002). Treatment Of Dandruff With 5% Tea Tree Oil Shampoo. J Am Acad Dermatol. 47(6). 852-855. Available at: … Continue reading
If you’re interested in trying tea tree oil as a natural solution to dandruff, you can find it at many pharmacies or online, but just make sure the concentration (5%) is clearly stated on the label!
Black seed oil 0.5% topical
Black cumin seed oil, also known as Nigella sativa, is a “trending” ingredient that you may see touted for its anti-aging properties. For dandruff, black cumin seed oil is not known to inhibit the growth of Malassezia like the other products in this list. Instead, it may:
- Reduce inflammation through its active component, thymoquinone.[64]Amin, B., Hosseinzadeh, H. (2016). Black Cumin (Nigella sativa) and Its Active Constituent, Thymoquinone: An Overview on the Analgesic and Anti-inflammatory Effects. Planta Medica 82(01/02):8-16. … Continue reading,[65]Arshad, M.T., Maqsood, S., Ikram, A., Abdullahi, M.A. (2025). Functional, Nutraceutical, And Pharmacological Properties Of Black Seed. Food Science & Nutrition. 13(8). e70725. Available at: … Continue reading
- Support a stronger skin barrier by delivering linoleic acid to the scalp, which stimulates ceramide synthesis (a key skin barrier lipid), thus reducing the impact of inflammatory molecules and microbes.[66]Rahim, M.A., Shoukat, A., Khalid, W., Ejaz, A., Itrat, N., Majeed, I., Koraqi, H., Imran, M., Nisa, M.U., Nazir, A., Alansari, W.S., Eskandrani, A.A., Shamlan, G., Al-Farga, A. (2022). A Narrative … Continue reading,[67]Coderch, L., López, O., de la Maza, A., Parra, J.L. (2003). Ceramides And Skin Function. Am J Clin Dermatol. 4(2). 107-129. Available at: https://doi.org/10.2165/00128071-200304020-00004
These effects counteract the adverse actions of Malassezia.
Unfortunately, there haven’t been any clinical trials testing the effect of black seed oil on subjects with dandruff. But, a randomized, double-blinded pilot study of 20 people with telogen effluvium suggested that this oil could reduce inflammation and hair shedding.[68]Rossi, A., Priolo, L., Iorio, A., Vescarelli, E., Gerardi, M., Campo, D., Nunno, D.D., Ceccarelli, S., Calvieri, S., Angeloni, A., Marchese, C. (2013). Evaluation Of A Therapeutic Alternative For … Continue reading
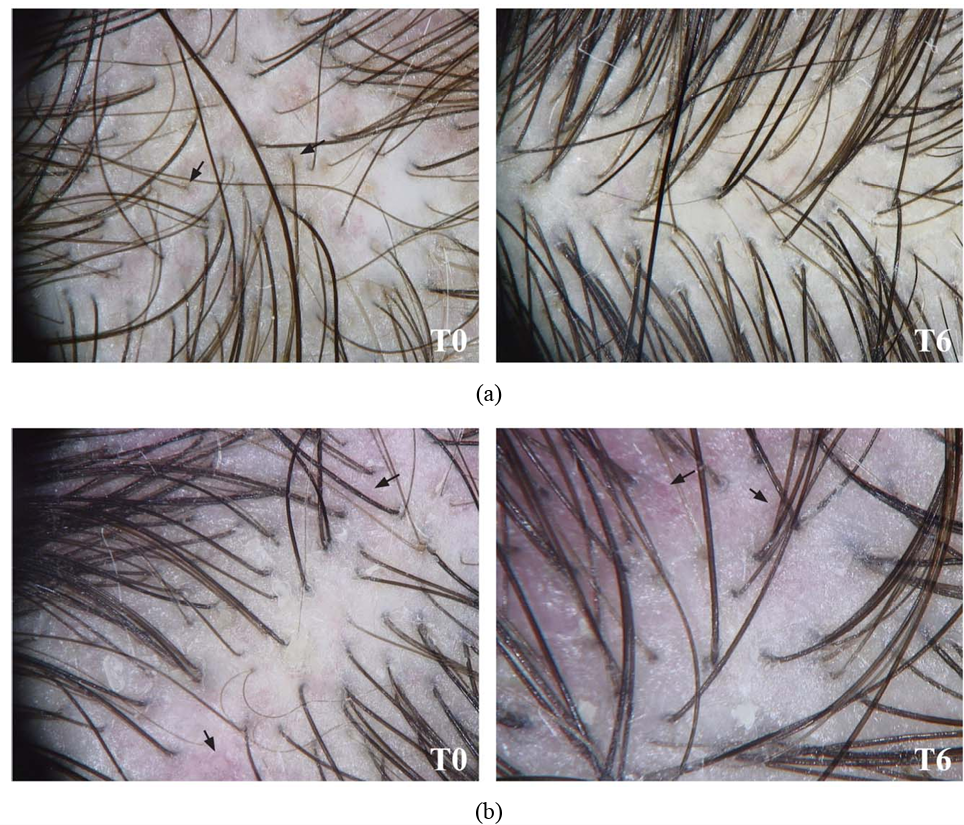
In the study, subjects were treated either with a topical product containing 0.5% black seed oil and 0.4% lavender oil, or a placebo with only 0.4% lavender oil. After 6 months, the following results were found:
- 90% of subjects using the 0.5% black seed oil and 0.4% lavender oil topical saw an increase in the number of hairs, while 60% of those using the placebo continued to lose hair
- Hair density significantly increased in 70% of subjects using the 0.5% black seed oil and 0.4% lavender oil topical, but only 30% in those using the placebo
- Subjects with scalp inflammation using the 0.5% black seed oil and 0.4% lavender oil topical were found to have total regression of their scalp inflammation, while none of the subjects with inflammation using the placebo saw any resolution to their inflammation
Figure 7: Scalp inflammation at baseline (T0) and after 6 months of treatment (T6) with either black seed oil (a) or a placebo (b). Patients treated with the placebo showed persistent scalp inflammation. Adapted from Figure 2.[69]Rossi, A., Priolo, L., Iorio, A., Vescarelli, E., Gerardi, M., Campo, D., Nunno, D.D., Ceccarelli, S., Calvieri, S., Angeloni, A., Marchese, C. (2013). Evaluation Of A Therapeutic Alternative For … Continue reading Image used under the Creative Commons License.
We know that Malassezia causes inflammation, and that inflammation is related to hair loss, so by counteracting these effects, black seed oil could be an effective addition to any anti-dandruff regimen, or even any anti-hair shedding regimen.
Because the active ingredient is thymoquinone, using black seed oil for dandruff and hair loss is less about the concentration of the black seed oil and more about the overall thymoquinone content in the formula. Since black seed oil is well-tolerated, you could probably go higher than 0.5% and make your own formula at a concentration of 2-5%. This will increase the likelihood that you’re getting enough of this anti-inflammatory compound.
For Nutrient-Linked Dandruff
If your dandruff is due to nutrient deficiencies, the obvious answer is to supplement your diet with foods that contain these missing vitamins, or alternatively look towards taking daily supplements.
We know that those with seborrheic dermatitis have been found with lowered levels of B vitamins, as well as vitamin D3 and zinc.[70]Aktaş Karabay, E., Aksu Çerman, A. (2019). Serum Zinc Levels In Seborrheic Dermatitis: A Case-Control Study. Turk J Med Sci. 49(5). 1503-1508. Available at: https://doi.org/10.3906/sag-1906-72,[71]Rahimi, S., Nemati, N., Shafaei-Tonekaboni, S.S. (2021). Serum Levels Of 25-Hydroxyvitamin D In Patients With Seborrheic Dermatitis: A Case-Control Study. Dermatol Res Pract. 2021. 6623271. Available … Continue reading,[72]Jen, M., Yan, A.C. (2010). Syndromes Associated With Nutritional Deficiency And Excess. Clinics In Dermatology. 28(6). 669-685. Available at: https://doi.org/10.1016/j.clindermatol.2010.03.029,[73]Stone, O.J. (1989). Pyridoxine Deficiency And Antagonism Produce Increased Ground Substance Viscosity With Resulting Seborrheic Dermatitis And Increased Tumor Resistance. Med Hypotheses. 30(4). … Continue reading,[74]Redzic, S., Hashmi, M.F., Gupta, V. (2023). Niacin Deficiency. StatPearls [Internet]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK557728/,[75]Balnave, D. (1977). Clinical Symptoms Of Biotin Deficiency In Animals. The American Journal Of Clinical Nutrition. 30(9). 1408-1413. Available at: https://doi.org/10.1093/ajcn/30.9.1408
There is no clinical evidence to show that supplementation will treat dandruff in those who are nutrient-deficient. However, there is evidence to show that nutrient supplementation can reverse adverse effects in those who are nutrient-deficient.
For example, biotin deficiency is a cause of hair thinning in women who are biotin-deficient. The supplementation of biotin to biotin-deficient individuals has been found to restore hair growth.[76]Patel, D.P., Swink, S.M., Castelo-Soccio, L. (2017). A Review Of The Use Of Biotin For Hair Loss. Skin Appendage Disord. 3(3). 166-169. Available at: https://doi.org/10.1159/000462981 So, it stands to reason that vitamin supplementation may also support the reduction or elimination of dandruff if this is the cause.
If you’re interested in learning more about the connection between nutrient deficiencies and hair loss, read our article here.
For Autoimmune and Inflammatory Dandruff
Unfortunately, if you think you might have dandruff caused by an autoimmune disorder, this is a lot harder to treat. The problem is internal, not just at the surface of the scalp, and the cause of an autoimmune disorder can be both complex and very personal.
Autoimmune disorders are often treated with immunosuppressive drugs. But you can also look at altering your diet, lifestyle, or environment to support a journey back to normalcy.
- Diet: Maintain a diet high in nutrients, and minimize exposure to any food allergens.[77]Mazzucca, C.B., Raineri, D., Cappellano, G., Chiocchetti, A. (2021). How To Tackle The Relationship Between Autoimmune Diseases And Diet: Well Begun Is Half-Done. Nutrients. 13(11). 3956. Available … Continue reading An elimination diet can work well to see which foods may be causing dandruff for you – eliminate a food from your diet for a few weeks and record the results. Blood tests can reveal nutrients that you might be deficient in, but a visit to your doctor may be needed to rule out malabsorption or other disease states causing nutrient deficiency.
- Lifestyle: Incorporate low-grade physical activity into your daily routine, ensure you are having regular sunlight exposure (for vitamin D!), and try to have consistent, high-quality sleep.[78]Blank, M., Israeli, D., Shoenfeld, Y. (2024). Exercise, Autoimmune Diseases And T-Regulatory Cells. Journal Of Autoimmunity. 149. 103317. Available at: https://doi.org/10.1016/j.jaut.2024.103317,[79]Yang, C.Y., Leung, P.S.C., Adamopoulos, I.E. (2013). The Implication Of Vitamin D And Autoimmunity: A Comprehensive Review. Clinical Reviews In Allergy And Immunology. 45. 217-226. Available at: … Continue reading,[80]Hsiao, Y.H., Chen, Y.T., Tseng, C.M., Wu, L.A., Lin, W.C., Su, V.Y.F., Perng, D.W., Chang, S.C., Chen, Y.M., Chen, T.J., Lee, Y.C., Chou, K.T. (2015). Sleep Disorders And Increased Risk Of Autoimmune … Continue reading
- Environment: Reduce your exposure to environmental toxins, like heavy metals, plastic chemicals, and persistent organic pollutants like dioxins and polychlorinated biphenyls, triclosan, and food colorings. Foreign substances like this can increase susceptibility to autoimmune diseases.[81]Pollard, K.M., Christy, J.M., Cauvi, D.M., Kono, D.H. (2018). Environmental Xenobiotic Exposure And Autoimmunity. Current Opinion In Toxicology. 10. 15-22. Available at: … Continue reading You can test for “toxic overload” using blood tests, and can even test your hair for heavy metals to assess your current toxin exposure.
Autoimmune disorders can affect your quality of life. If you think you might have an autoimmune or inflammatory disorder, speak with your physician to discuss suitable treatment routes.
What to Avoid If You Have Dandruff
#1 – Oleic acid
Organisms like Malassezia and P. acnes can break down sebum and produce oleic acid, which we have established can be harmful to the skin barrier. This lipid is not only harmful but can also encourage the growth of microorganisms like these, so it follows that those with dandruff should avoid any topical products containing oleic acid.[82]Trompezinski, S., Weber, S., Cadars, B., Larue, F., Ardiet, N., Chavagnac-Bonneville, M., Sayag, M., Jourdan, E. (2016). Assessment Of A New Biological Complex Efficacy On Dysseborrhea, Inflammation, … Continue reading
Oleic acid can be found naturally in vegetable oils (i.e., olive oil, corn oil, canola oil, avocado oil) and animal fats (i.e., tallow and lard), but you don’t have to avoid eating oleic acid if you have dandruff. There is no evidence to suggest eating oleic acid causes more oleic acid at the scalp, and in fact studies show that eating oleic acid can reduce inflammation and reduce the activity of stearoyl-CoA desaturase (the enzyme that produces oleic acid in the body).[83]Wang, Q., Liu, R., Chang, M., Zhang, H., Jin, Q., Wang, X. (2022). Dietary Oleic Acid Supplementation And Blood Inflammatory Markers: A Systematic Review And Meta-Analysis Of Randomized Controlled … Continue reading,[84]Dobrzyn, P., Pyrkowska, A., Jazurek, M., Dobrzyn, A. (2012). Increased Availability Of Endogenous And Dietary Oleic Acid Contributes To The Upregulation Of Cardiac Fatty Acid Oxidation. … Continue reading
#2 – Allergenic foods
For autoimmune-prone individuals, there may be a particular source driving seborrheic dermatitis, eczema, or scalp psoriasis. In these cases, it may be worthwhile to review your dietary choices and try to determine any potential causes. Anecdotally, several people we have worked with have reported that dietary overhauls (including the elimination of gluten, dairy, and seed oils) significantly improved their dandruff problems.
What If My Dandruff Is Resistant to Treatment?
PHH members with treatment-resistant dandruff have reported success when rotating shampoos with different active ingredients.
An example rotation may look like this:
2% ketoconazole (2-3 days) → selenium sulfide (2-3 days) → coal tar (2-3 days) → zinc pyrithione (2-3 days) → 2% ketoconazole (2-3 days)…
Rotating treatments like these can be effective because each active ingredient targets dandruff in a distinct way, creating a more comprehensive approach to addressing its underlying causes:
- Ketoconazole: Fights pathogenic scalp microbes by inhibiting fungal cell wall synthesis.[85]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. Journal Of Clinical And Investigative Dermatology. 3(2). 10.13188/2373-1044.1000019. Available at: … Continue reading,[86]Koc, E., Arca, E., Kose, O., Akar, A. (2009). An Open, Randomized, Prospective, Comparative Study Of Topical Pimecrolimus 1% Cream And Topical Ketoconazole 2% Cream In The Treatment Of Seborrheic … Continue reading
- Selenium sulfide: Stops the growth and proliferation of cells, and is also keratolytic – it exfoliates the outer layer of skin by breaking down keratin.[87]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. Journal Of Clinical And Investigative Dermatology. 3(2). 10.13188/2373-1044.1000019. Available at: … Continue reading,[88]Danby, F.W., Maddin, W.S., Margesson, L.J., Rosenthal, D. (1993). A Randomized, Double-Blind, Placebo-Controlled Trial Of Ketoconazole 2% Shampoo Versus Selenium Sulfide 2.5% Shampoo In The Treatment … Continue reading
- Coal tar: This is an anti-fungal, anti-inflammatory, and keratolytic compound that can also reduce sebum production.[89]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. Journal Of Clinical And Investigative Dermatology. 3(2). 10.13188/2373-1044.1000019. Available at: … Continue reading,[90]Davies, D., Boorman, G.C., Shuttleworth, D. (1999). Comparative Efficacy Of Shampoos Containing Coal Tar (4.0% W/W; TarmedTM), Coal Tar (4.0% W/W) Plus Ciclopirox Olamine (1.0% W/W; TarmedTM AF) And … Continue reading
- Zinc pyrithione: Interferes with iron-sulfur proteins and energy metabolism in microbes.[91]Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. Journal Of Clinical And Investigative Dermatology. 3(2). 10.13188/2373-1044.1000019. Available at: … Continue reading,[92]Reeder, N.L., Xu, J., Youngquist, R.S., Schwartz, J.R., Rust, R.C., Saunders, C.W. (2011). The Antifungal Mechanism Of Action Of Zinc Pyrithione. Br J Dermatol. 165 Suppl 2. 9-12. Available at: … Continue reading
Final Remarks
Dandruff is a multifactorial condition influenced by microbes, the skin barrier, nutrition, and immune activity. Although it does not directly cause permanent hair loss, the inflammation and scalp imbalance associated with dandruff can increase shedding and potentially accelerate pattern hair loss in susceptible individuals. Encouragingly, addressing dandruff often improves overall scalp health and may enhance the effectiveness of hair growth treatments. The most successful strategy is to first identify the underlying driver of dandruff, whether it be microbial, nutritional, or inflammatory, and then select the most appropriate treatment. By doing this, you can build a healthier scalp environment that can support normal hair cycling, and hopefully stronger hair regrowth as well, over time.
References[+]
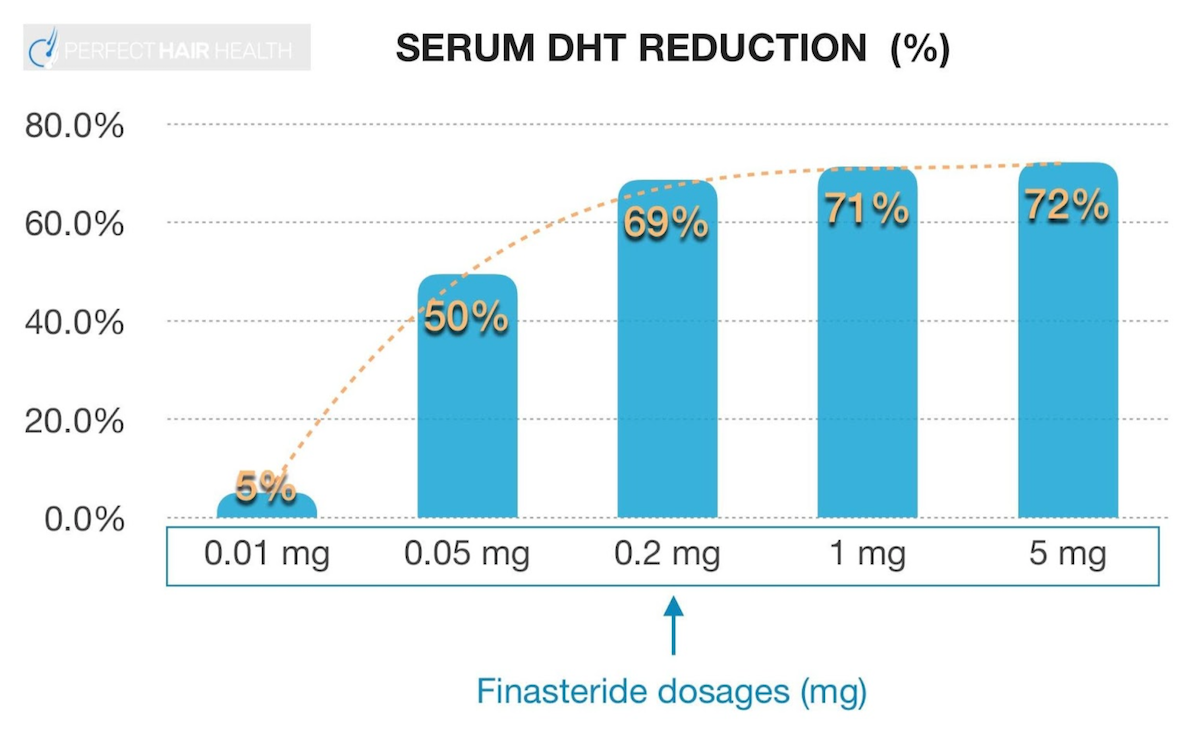
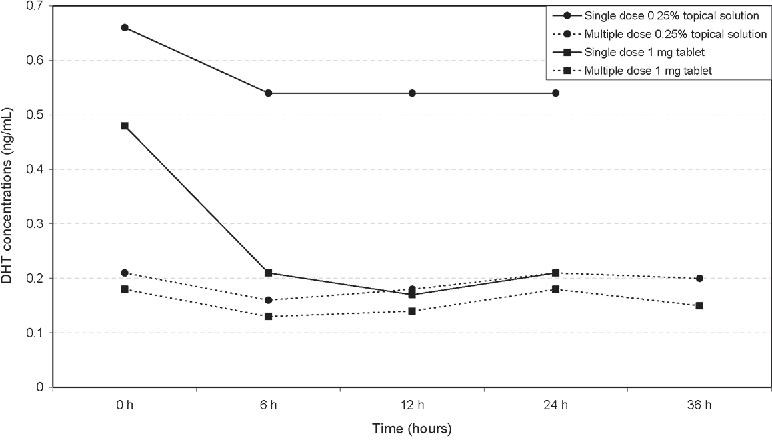
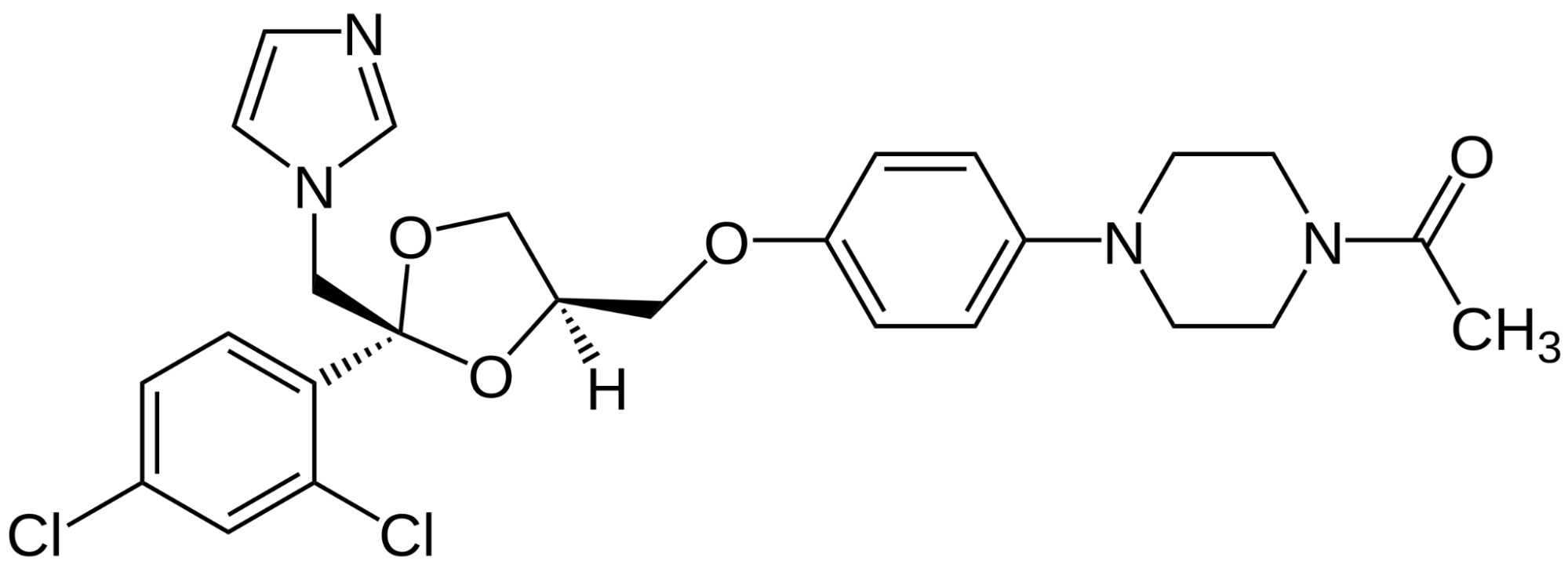
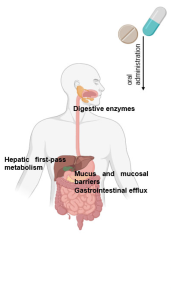
References ↑1, ↑2, ↑3, ↑20 Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. J Clin Investig Dermatol. 3(2). Available at: https://doi.org/10.13188/2373-1044.1000019 ↑4 Del Rosso, J.Q. (2011). Adult Seborrheic Dermatitis: A Status Report On Practical Topical Management. J Clin Aesthet Dermatol. 4(5). Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3100109/ ↑5 Jourdain, R., Moga, A., Magiatis, P., Fontanié, M., Velegraki, A., Papadimou, C., Rahoul, V., Guéniche, A., Chopra, T., Gaitanis, G. (2023). Malassezia Restricta-Mediated Lipoperoxidation: A Novel Trigger In Dandruff. Acta Derm Venereol. 103. adv00868. Available at: https://doi.org/10.2340/actadv.v103.4808 ↑6, ↑14 Park, M., Park, S., Jung, W.H. (2021). Skin Commensal Fungus Malassezia And Its Lipases. J Microbiol Biotechnol. 31(5). 637-644. Available at: https://doi.org/10.4014/jmb.2012.12048 ↑7 Kováčik, A., Kopečná, M., Hrdinová, I., Opálka, L., Boncheva Bettex, M., Vávrová, K. (2023). Time-Dependent Differences In The Effects Of Oleic Acid And Oleyl Alcohol On The Human Skin Barrier. Mol Pharm. 20(12). 6237-6245. Available at: https://doi.org/10.1021/acs.molpharmaceut.3c00648 ↑8 Tanojo, H., Boelsma, E., Junginger, H.E., Ponec, M., Boddé, H.E. (1999). In Vivo Human Skin Permeability Enhancement By Oleic Acid: A Laser Doppler Velocimetry Study. J Control Release. 58(1). 97-104. Available at: https://doi.org/10.1016/s0168-3659(98)00144-8 ↑9 Liu, H.M., Cheng, M.Y., Xun, M.H., Zhao, Z.W., Zhang, Y., Tang, W., Cheng, J., Ni, J., Wang, W. (2023). Possible Mechanisms Of Oxidative Stress-Induced Skin Cellular Senescence, Inflammation, And Cancer And The Therapeutic Potential Of Plant Polyphenols. Int J Mol Sci. 24(4). Available at: https://doi.org/10.3390/ijms24043755 ↑10 Sikder, M.M., Li, X., Akumwami, S., Labony, S.A. (2025). Reactive Oxygen Species: Role In Pathophysiology, And Mechanism Of Endogenous And Dietary Antioxidants During Oxidative Stress. Chonnam Med J. 61(1). 32-45. Available at: https://doi.org/10.4068/cmj.2025.61.1.32 ↑11 Kim, M.J., Woo, S.W., Kim, M.S., Park, J.E., Hwang, J.K. (2014). Anti-Photoaging Effect Of Aaptamine In UVB-Irradiated Human Dermal Fibroblasts And Epidermal Keratinocytes. Journal Of Asian Natural Products Research. 16(12). 1139-1147. Available at: https://doi.org/10.1080/10286020.2014.983092 ↑12 Rudramurthy, S.M., Honnavar, P., Dogra, S., Yegneswaran, P.P., Handa, S., Chakrabarti, A. (2014). Association Of Malassezia Species With Dandruff. Indian J Med Res. 139(3). 431-437. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4069738/ ↑13 Clavaud, C., Jourdain, R., Bar-Hen, A., Tichit, M., Bouchier, C., Pouradier, F., El Rawadi, C., Guillot, J., Ménard-Szczebara, F., Breton, L., Latgé, J.P., Mouyna, I. (2013). Dandruff Is Associated With Disequilibrium In The Proportion Of The Major Bacterial And Fungal Populations Colonizing The Scalp. PLoS One. 8(3). e58203. Available at: https://doi.org/10.1371/journal.pone.0058203 ↑15 Ashbee, H.R., Ingham, E., Holland, K.T., Cunliffe, W.J. (1993). The Carriage Of Malassezia Furfur Serovars A, B And C In Patients With Pityriasis Versicolor, Seborrhoeic Dermatitis And Controls. British Journal Of Dermatology. 129(5). 533-540. Available at: https://doi.org/10.1111/j.1365-2133.1993.tb00480.x ↑16 Dawson, T.L. Jr. (2007). Malassezia Globosa And Restricta: Breakthrough Understanding Of The Etiology And Treatment Of Dandruff And Seborrheic Dermatitis Through Whole-Genome Analysis. J Investig Dermatol Symp Proc. 12(2). 15-19. Available at: https://doi.org/10.1038/sj.jidsymp.5650049 ↑17, ↑59 Piérard-Franchimont, C., Piérard, G.E., Arrese, J.E., De Doncker, P. (2001). Effect Of Ketoconazole 1% And 2% Shampoos On Severe Dandruff And Seborrhoeic Dermatitis: Clinical, Squamometric And Mycological Assessments. Dermatology. 202(2). 171-176. Available at: https://doi.org/10.1159/000051628 ↑18, ↑70 Aktaş Karabay, E., Aksu Çerman, A. (2019). Serum Zinc Levels In Seborrheic Dermatitis: A Case-Control Study. Turk J Med Sci. 49(5). 1503-1508. Available at: https://doi.org/10.3906/sag-1906-72 ↑19, ↑71 Rahimi, S., Nemati, N., Shafaei-Tonekaboni, S.S. (2021). Serum Levels Of 25-Hydroxyvitamin D In Patients With Seborrheic Dermatitis: A Case-Control Study. Dermatol Res Pract. 2021. 6623271. Available at: https://doi.org/10.1155/2021/6623271 ↑21, ↑72 Jen, M., Yan, A.C. (2010). Syndromes Associated With Nutritional Deficiency And Excess. Clinics In Dermatology. 28(6). 669-685. Available at: https://doi.org/10.1016/j.clindermatol.2010.03.029 ↑22, ↑73 Stone, O.J. (1989). Pyridoxine Deficiency And Antagonism Produce Increased Ground Substance Viscosity With Resulting Seborrheic Dermatitis And Increased Tumor Resistance. Med Hypotheses. 30(4). 277-280. Available at: https://doi.org/10.1016/0306-9877(89)90037-6 ↑23, ↑74 Redzic, S., Hashmi, M.F., Gupta, V. (2023). Niacin Deficiency. StatPearls [Internet]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK557728/ ↑24, ↑75 Balnave, D. (1977). Clinical Symptoms Of Biotin Deficiency In Animals. The American Journal Of Clinical Nutrition. 30(9). 1408-1413. Available at: https://doi.org/10.1093/ajcn/30.9.1408 ↑25 Demetree, J.W., Safer, L.F., Artis, W.M. (1980). The Effect Of Zinc On The Sebum Secretion Rate. Acta Derm Venereol. 60(2). 166-169. Available at: https://pubmed.ncbi.nlm.nih.gov/6155029/ ↑26 Lin, P.H., Sermersheim, M., Li, H., Lee, P.H.U., Steinberg, S.M., Ma, J. (2017). Zinc In Wound Healing Modulation. Nutrients. 10(1). 16. Available at: https://doi.org/10.3390/nu10010016 ↑27 Arribas Lopez, E., Zand, N., Ojo, O., Kochhar, T. (2025). Systematic Review And Meta-Analysis Of The Effect Of Zinc On Wound Healing. BMJ Nutr Prev Health. 8(1). e000952. Available at: https://doi.org/10.1136/bmjnph-2024-000952 ↑28 Haase, H., Schomburg, L. (2019). You’d Better Zinc-Trace Element Homeostasis In Infection And Inflammation. Nutrients. 11(9). 2078. Available at: https://doi.org/10.3390/nu11092078 ↑29 Bachman, M.A., Weiser, J.N. (2015). Competition For Metals Among Microbes And Their Host. Trace Metals And Infectious Diseases [Internet]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK569680/ ↑30 Potrykus, J., Ballou, E.R., Childers, D.S., Brown, A.J.P. (2014). Conflicting Interests In The Pathogen–Host Tug Of War: Fungal Micronutrient Scavenging Versus Mammalian Nutritional Immunity. PLoS Pathogens. Available at: https://doi.org/10.1371/journal.ppat.1003910 ↑31 Gupta, S., Read, S.A., Shackel, N.A., Hebbard, L., George, J., Ahlenstiel, G. (2019). The Role Of Micronutrients In The Infection And Subsequent Response To Hepatitis C Virus. Cells. 8(6). 603. Available at: https://doi.org/10.3390/cells8060603 ↑32 Roth-Walter, F., Berni Canani, R., O’Mahony, L., Peroni, D., Sokolowska, M., Vassilopoulou, E., Venter, C. (2024). Nutrition In Chronic Inflammatory Conditions: Bypassing The Mucosal Block For Micronutrients. Allergy. 79(2). 353-383. Available at: https://doi.org/10.1111/all.15972 ↑33 Magnifico, I., Petronio Petronio, G., Venditti, N., Cutuli, M.A., Pietrangelo, L., Vergalito, F., Mangano, K., Zella, D., Di Marco, R. (2020). Atopic Dermatitis As A Multifactorial Skin Disorder. Can The Analysis Of Pathophysiological Targets Represent The Winning Therapeutic Strategy? Pharmaceuticals (Basel). 13(11). 411. Available at: https://doi.org/10.3390/ph13110411 ↑34 Armstrong, A.W., Read, C. (2020). Pathophysiology, Clinical Presentation, And Treatment Of Psoriasis: A Review. JAMA. 323(19). 1945-1960. Available at: https://doi.org/10.1001/jama.2020.4006 ↑35 Lepe, K., Nassereddin, A., Syed, H.A., et al. (2024). Lichen Planopilaris. StatPearls [Internet]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK470325/ ↑36 Navarro Triviño, F.J., Velasco Amador, J.P., Rivera Ruiz, I. (2025). Seborrheic Dermatitis Revisited: Pathophysiology, Diagnosis, And Emerging Therapies-A Narrative Review. Biomedicines. 13(10). 2458. Available at: https://doi.org/10.3390/biomedicines13102458 ↑37 Umar, S., Kan, P., Carter, M.J. (2022). Lichen Planopilaris Responsive To A Novel Phytoactive Botanical Treatment: A Case Series. Dermatology And Therapy. 12. 1697-1710. Available at: https://doi.org/10.1007/s13555-022-00749-3 ↑38 Manzel, A., Muller, D.N., Hafler, D.A., Erdman, S.E., Linker, R.A., Kleinewietfeld, M. (2014). Role Of “Western Diet” In Inflammatory Autoimmune Diseases. Curr Allergy Asthma Rep. 14(1). 404. Available at: https://doi.org/10.1007/s11882-013-0404-6 ↑39 Perricone, C., Versini, M., Ben-Ami, D., Gertel, S., Watad, A., Segel, M.J., Ceccarelli, F., Conti, F., Cantarini, L., Bogdanos, D.P., Antonelli, A., Amital, H., Valesini, G., Shoenfeld, Y. (2016). Smoke And Autoimmunity: The Fire Behind The Disease. Autoimmunity Reviews. 15(4). 354-374. Available at: https://doi.org/10.1016/j.autrev.2016.01.001 ↑40 Hussain, N., Agarwala, P., Iqbal, K., et al. (2022). A Systematic Review Of Acute Telogen Effluvium, A Harrowing Post-COVID-19 Manifestation. J Med Virol. 94. 1391-1401. Available at: https://doi.org/10.1002/jmv.27534 ↑41 Bin Dayel, S., Hussein, R.S., Atia, T., Abahussein, O., Al Yahya, R.S., Elsayed, S.H. (2024). Is Thyroid Dysfunction A Common Cause Of Telogen Effluvium?: A Retrospective Study. Medicine. 103(1). e36803. Available at: https://doi.org/10.1097/MD.0000000000036803 ↑42 Kang, D.H., Kwon, S.H., Sim, W.Y., Lew, B.L. (2024). Telogen Effluvium Associated With Weight Loss: A Single Center Retrospective Study. Ann Dermatol. 36(6). 384-388. Available at: https://doi.org/10.5021/ad.24.043 ↑43 Monari, P., Gualdi, G., Bettoni, G., Costa, R., Ragni, G., Zani, F., Bianchi, G., Casella, S., Casella, E., Crippa, M., Calzavara Pinton, P., Di Nicola, M., Porreca, A., Amerio, P., Guizzi, P. (2022). Post-SARS-CoV-2 Acute Telogen Effluvium: An Expected Complication. Journal Of Clinical Medicine. 11(5). 1234. Available at: https://doi.org/10.3390/jcm11051234 ↑44 Malkud, S. (2015). Telogen Effluvium: A Review. J Clin Diagn Res. 9(9). WE01-3. Available at: https://doi.org/10.7860/JCDR/2015/15219.6492 ↑45 Nematian, J., Ravaghi, M., Gholamrezanezhad, A., Nematian, E. (2006). Increased Hair Shedding May Be Associated With The Presence Of Pityrosporum Ovale. Am J Clin Dermatol. 7(4). 263-266. Available at: https://doi.org/10.2165/00128071-200607040-00008 ↑46 English, R.S. (2018). A Hypothetical Pathogenesis Model For Androgenic Alopecia: Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors. Medical Hypotheses. 111. 73-81. Available at: https://doi.org/10.1016/j.mehy.2017.12.027 ↑47 Chen, S., Li, L., Ding, W., Zhu, Y., Zhou, N. (2025). Androgenetic Alopecia: An Update On Pathogenesis And Pharmacological Treatment. Drug Des Devel Ther. 19. 7349-7363. Available at: https://doi.org/10.2147/DDDT.S542000 ↑48 Piérard-Franchimont, C., Xhauflaire-Uhoda, E., Loussouarn, G., Saint Léger, D., Piérard, G.E. (2006). Dandruff-Associated Smouldering Alopecia: A Chronobiological Assessment Over 5 Years. Clin Exp Dermatol. 31(1). 23-26. Available at: https://doi.org/10.1111/j.1365-2230.2005.02026.x ↑49 Mahe, Y.F., Cheniti, A., Tacheau, C., Antonelli, R., Planard-Luong, L., de Bernard, S., Buffat, L., Barbarat, P., Kanoun-Copy, L. (2021). Low-Level Light Therapy Downregulates Scalp Inflammatory Biomarkers In Men With Androgenetic Alopecia And Boosts Minoxidil 2% To Bring A Sustainable Hair Regrowth Activity. Lasers Surg Med. 53. 1208-1219. Available at: https://doi.org/10.1002/lsm.23398 ↑50, ↑52, ↑53 Park, M., Cho, Y.J., Lee, Y.W., Jung, W.H. (2018). Understanding The Mechanism Of Action Of The Anti-Dandruff Agent Zinc Pyrithione Against Malassezia Restricta. Sci Rep. 8(1). 12086. Available at: https://doi.org/10.1038/s41598-018-30588-2 ↑51, ↑92 Reeder, N.L., Xu, J., Youngquist, R.S., Schwartz, J.R., Rust, R.C., Saunders, C.W. (2011). The Antifungal Mechanism Of Action Of Zinc Pyrithione. Br J Dermatol. 165 Suppl 2. 9-12. Available at: https://doi.org/10.1111/j.1365-2133.2011.10571.x ↑54 Marks, R., Pearse, A.D., Walker, A.P. (1985). The Effects Of A Shampoo Containing Zinc Pyrithione On The Control Of Dandruff. Br J Dermatol. 112(4). 415-422. Available at: https://doi.org/10.1111/j.1365-2133.1985.tb02314.x ↑55 Berger, R.S., Fu, J.L., Smiles, K.A., Turner, C.B., Schnell, B.M., Werchowski, K.M., Lammers, K.M. (2003). The Effects Of Minoxidil, 1% Pyrithione Zinc And A Combination Of Both On Hair Density: A Randomized Controlled Trial. Br J Dermatol. 149(2). 354-362. Available at: https://doi.org/10.1046/j.1365-2133.2003.05435.x ↑56 Schwartz, J.R., Bacon, R.A., Shah, R., Mizoguchi, H., Tosti, A. (2013). Therapeutic Efficacy Of Anti-Dandruff Shampoos: A Randomized Clinical Trial Comparing Products Based On Potentiated Zinc Pyrithione And Zinc Pyrithione/Climbazole. Int J Cosmet Sci. 35(4). 381-387. Available at: https://doi.org/10.1111/ics.12055 ↑57 Tynes, B.E., Johnson, C.D., Vaish, M.H., Abbott, B., Vučenović, J., Varrassi, G., Potharaju, P., Lopez Torres, Y., Lee, Z., Ahmadzadeh, S., Shekoohi, S., Kaye, A.D. (2024). Ketoconazole Shampoo For Seborrheic Dermatitis Of The Scalp: A Narrative Review. Cureus. 16(8). e67532. Available at: https://doi.org/10.7759/cureus.67532 ↑58 Goularte-Silva, V., Paulino, L.C. (2022). Ketoconazole Beyond Antifungal Activity: Bioinformatics-Based Hypothesis On Lipid Metabolism In Dandruff And Seborrheic Dermatitis. Exp Dermatol. 31. 821-822. Available at: https://doi.org/10.1111/exd.14505 ↑60 Piérard-Franchimont, C., Goffin, V., Decroix, J., Piérard, G.E. (2002). A Multicenter Randomized Trial Of Ketoconazole 2% And Zinc Pyrithione 1% Shampoos In Severe Dandruff And Seborrheic Dermatitis. Skin Pharmacol Appl Skin Physiol. 15(6). 434-441. Available at: https://doi.org/10.1159/000066452 ↑61 Lorette, G., Ermosilla, V. (2006). Clinical Efficacy Of A New Ciclopiroxolamine/Zinc Pyrithione Shampoo In Scalp Seborrheic Dermatitis Treatment. Eur J Dermatol. 16(5). Available at: https://pubmed.ncbi.nlm.nih.gov/17101479/ ↑62 Yu, D., Wang, J., Shao, X., Xu, F., Wang, H. (2015). Antifungal Modes Of Action Of Tea Tree Oil And Its Two Characteristic Components Against Botrytis Cinerea. J Appl Microbiol. 119(5). 1253-1262. Available at: https://doi.org/10.1111/jam.12939 ↑63 Satchell, A.C., Saurajen, A., Bell, C., Barnetson, R.S. (2002). Treatment Of Dandruff With 5% Tea Tree Oil Shampoo. J Am Acad Dermatol. 47(6). 852-855. Available at: https://doi.org/10.1067/mjd.2002.122734 ↑64 Amin, B., Hosseinzadeh, H. (2016). Black Cumin (Nigella sativa) and Its Active Constituent, Thymoquinone: An Overview on the Analgesic and Anti-inflammatory Effects. Planta Medica 82(01/02):8-16. Available at: 10.1055/s-0035-1557838 ↑65 Arshad, M.T., Maqsood, S., Ikram, A., Abdullahi, M.A. (2025). Functional, Nutraceutical, And Pharmacological Properties Of Black Seed. Food Science & Nutrition. 13(8). e70725. Available at: https://doi.org/10.1002/fsn3.70725 ↑66 Rahim, M.A., Shoukat, A., Khalid, W., Ejaz, A., Itrat, N., Majeed, I., Koraqi, H., Imran, M., Nisa, M.U., Nazir, A., Alansari, W.S., Eskandrani, A.A., Shamlan, G., Al-Farga, A. (2022). A Narrative Review On Various Oil Extraction Methods, Encapsulation Processes, Fatty Acid Profiles, Oxidative Stability, And Medicinal Properties Of Black Seed (Nigella Sativa). Foods. 11(18). 2826. Available at: https://doi.org/10.3390/foods11182826 ↑67 Coderch, L., López, O., de la Maza, A., Parra, J.L. (2003). Ceramides And Skin Function. Am J Clin Dermatol. 4(2). 107-129. Available at: https://doi.org/10.2165/00128071-200304020-00004 ↑68 Rossi, A., Priolo, L., Iorio, A., Vescarelli, E., Gerardi, M., Campo, D., Nunno, D.D., Ceccarelli, S., Calvieri, S., Angeloni, A., Marchese, C. (2013). Evaluation Of A Therapeutic Alternative For Telogen Effluvium: A Pilot Study. Journal Of Cosmetics, Dermatological Sciences And Applications. 2013. 9-16. Available at: https://file.scirp.org/pdf/JCDSA_2013081513583870.pdf ↑69 Rossi, A., Priolo, L., Iorio, A., Vescarelli, E., Gerardi, M., Campo, D., Nunno, D.D., Ceccarelli, S., Calvieri, S., Angeloni, A., Marchese, C. (2013). Evaluation Of A Therapeutic Alternative For Telogen Effluvium: A Pilot Study. Journal Of Cosmetics, Dermatological Sciences And Applications. 2013. 9-16. Available at:https://file.scirp.org/pdf/JCDSA_2013081513583870.pdf ↑76 Patel, D.P., Swink, S.M., Castelo-Soccio, L. (2017). A Review Of The Use Of Biotin For Hair Loss. Skin Appendage Disord. 3(3). 166-169. Available at: https://doi.org/10.1159/000462981 ↑77 Mazzucca, C.B., Raineri, D., Cappellano, G., Chiocchetti, A. (2021). How To Tackle The Relationship Between Autoimmune Diseases And Diet: Well Begun Is Half-Done. Nutrients. 13(11). 3956. Available at: https://doi.org/10.3390/nu13113956 ↑78 Blank, M., Israeli, D., Shoenfeld, Y. (2024). Exercise, Autoimmune Diseases And T-Regulatory Cells. Journal Of Autoimmunity. 149. 103317. Available at: https://doi.org/10.1016/j.jaut.2024.103317 ↑79 Yang, C.Y., Leung, P.S.C., Adamopoulos, I.E. (2013). The Implication Of Vitamin D And Autoimmunity: A Comprehensive Review. Clinical Reviews In Allergy And Immunology. 45. 217-226. Available at: https://doi.org/10.1007/s12016-013-8361-3 ↑80 Hsiao, Y.H., Chen, Y.T., Tseng, C.M., Wu, L.A., Lin, W.C., Su, V.Y.F., Perng, D.W., Chang, S.C., Chen, Y.M., Chen, T.J., Lee, Y.C., Chou, K.T. (2015). Sleep Disorders And Increased Risk Of Autoimmune Diseases In Individuals Without Sleep Apnea. Sleep. 38(4). 581-586. Available at: https://doi.org/10.5665/sleep.4574 ↑81 Pollard, K.M., Christy, J.M., Cauvi, D.M., Kono, D.H. (2018). Environmental Xenobiotic Exposure And Autoimmunity. Current Opinion In Toxicology. 10. 15-22. Available at: https://doi.org/10.1016/j.cotox.2017.11.009 ↑82 Trompezinski, S., Weber, S., Cadars, B., Larue, F., Ardiet, N., Chavagnac-Bonneville, M., Sayag, M., Jourdan, E. (2016). Assessment Of A New Biological Complex Efficacy On Dysseborrhea, Inflammation, And Propionibacterium Acnes Proliferation. Clinical, Cosmetic And Investigational Dermatology. 9. 233-239. Available at: https://doi.org/10.2147/CCID.S110655 ↑83 Wang, Q., Liu, R., Chang, M., Zhang, H., Jin, Q., Wang, X. (2022). Dietary Oleic Acid Supplementation And Blood Inflammatory Markers: A Systematic Review And Meta-Analysis Of Randomized Controlled Trials. Critical Reviews In Food Science And Nutrition. 62(9). 2508-2525. Available at: https://doi.org/10.1080/10408398.2020.1854673 ↑84 Dobrzyn, P., Pyrkowska, A., Jazurek, M., Dobrzyn, A. (2012). Increased Availability Of Endogenous And Dietary Oleic Acid Contributes To The Upregulation Of Cardiac Fatty Acid Oxidation. Mitochondrion. 12(1). 132-137. Available at: https://doi.org/10.1016/j.mito.2011.05.007 ↑85, ↑87, ↑89, ↑91 Borda, L.J., Wikramanayake, T.C. (2015). Seborrheic Dermatitis And Dandruff: A Comprehensive Review. Journal Of Clinical And Investigative Dermatology. 3(2). 10.13188/2373-1044.1000019. Available at: https://doi.org/10.13188/2373-1044.1000019 ↑86 Koc, E., Arca, E., Kose, O., Akar, A. (2009). An Open, Randomized, Prospective, Comparative Study Of Topical Pimecrolimus 1% Cream And Topical Ketoconazole 2% Cream In The Treatment Of Seborrheic Dermatitis. Journal Of Dermatological Treatment. 20(1). 4-9. Available at: https://doi.org/10.1080/09546630802286993 ↑88 Danby, F.W., Maddin, W.S., Margesson, L.J., Rosenthal, D. (1993). A Randomized, Double-Blind, Placebo-Controlled Trial Of Ketoconazole 2% Shampoo Versus Selenium Sulfide 2.5% Shampoo In The Treatment Of Moderate To Severe Dandruff. Journal Of The American Academy Of Dermatology. 29(6). 1008-1012. Available at: https://doi.org/10.1016/0190-9622(93)70282-X ↑90 Davies, D., Boorman, G.C., Shuttleworth, D. (1999). Comparative Efficacy Of Shampoos Containing Coal Tar (4.0% W/W; TarmedTM), Coal Tar (4.0% W/W) Plus Ciclopirox Olamine (1.0% W/W; TarmedTM AF) And Ketoconazole (2.0% W/W; NizoralTM) For The Treatment Of Dandruff/Seborrhoeic Dermatitis. Journal Of Dermatological Treatment. 10(3). 177-183. Available at: https://doi.org/10.3109/09546639909056025 Finasteride is among the most effective drugs for androgenic alopecia. And while side effects are often overstated, it can lead to reduced libido or lowered sperm counts in some men. For this reason, many choose topical versus oral finasteride, hoping to limit the drug’s DHT-reducing effects to the scalp. But at certain doses, even topical finasteride can become systemic. So, to minimize side effects, which finasteride dosage, formula and application is best?
In this article, we’ll review
- About oral versus topical finasteride
- Why topical finasteride can still go systemic
- Why choosing the lowest percentage solution isn’t the only answer
- How to maximize finasteride gains, while minimizing side effects
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

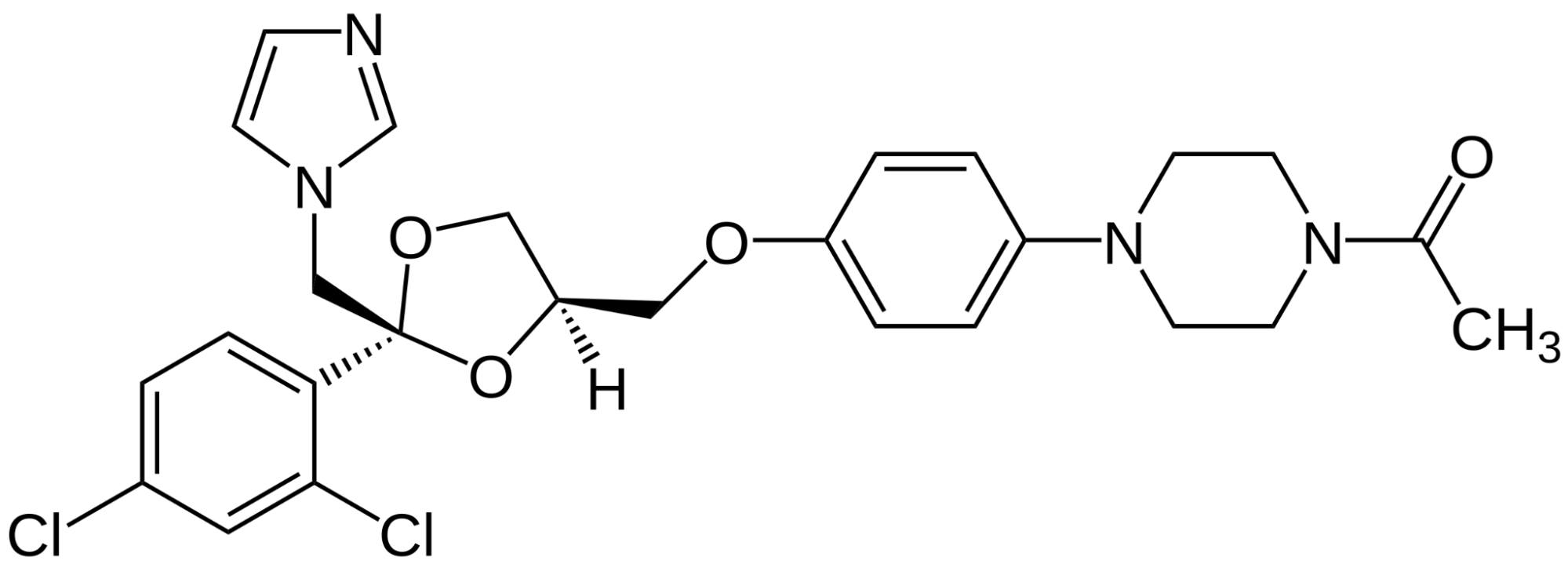
About Finasteride
Finasteride, also known under the brand name Propecia or Proscar, is a prescription medication approved by the FDA for the treatment of androgenic alopecia (AGA). The anti-androgen works by reducing the production of Dihydrotestosterone (DHT), a hormone linked to pattern hair loss. In use since 1992, Finasteride is among the most powerful and well-studied drugs for hair loss.
Oral Finasteride
Oral finasteride stops AGA progression in 80-90% of men and, on average, leads to a 10% increase in hair count over two years.[1]https://www.sciencedirect.com/science/article/pii/S0022202X15529357 For men wanting a “hands-off” approach to hair maintenance, oral finasteride can be an excellent option. With a once-daily pill, it’s expected that hair loss will stop at approximately 6 months, and thereafter, improve.
However, oral finasteride isn’t for everyone. While the risk of side effects are often overstated online, the drug appears to reduce libido in a certain percentage of men. Oral finasteride can also temporarily lower sperm counts, which might make conception more difficult during its first six months of use. In some men, the use of finasteride appears to increase anxiety and/or depression. While the true incidence and magnitude of these reports are hard to discern, it’s understandable that many are weary of taking this once daily pill.
Fortunately, recent improvements in finasteride’s delivery may mean we no longer need to throw the proverbial baby out with the bathwater.
Topical Finasteride
The biggest reason people seek out topical finasteride (instead of oral finasteride) is because they want to minimize systemic exposure to the drug, and in doing so, localize finasteride’s effects to the scalp.
While research is still in the early stages, evidence suggests that topical finasteride can produce similar levels of hair regrowth compared to oral finasteride, with a significantly reduced incidence of side effects.
Topical finasteride works identically to its oral counterpart: by inhibiting the type II 5-alpha reductase enzyme to reduce DHT. It is designed to target scalp DHT instead of systemic DHT levels.
However, limiting finasteride’s reach to scalp, not serum, DHT is harder than perhaps anticipated. To understand why, let’s take a closer look at scalp versus serum DHT.
Serum vs Scalp DHT
There’s a lot of evidence that the hormone DHT is directly implicated in androgenic alopecia. In fact, research directly links DHT to all three of AGA’s defining characteristics:
- Increased telogen:anagen ratio. DHT’s influence on DKK-1 expression increases hair shedding
- Increased anagen cycling. DHT’s effects on signaling pathways limit the growth phase of hairs
- Hair follicle miniaturization. DHT’s effects on dermal papilla sizing thins hair with each cycle
There are a wide variety of DHT reducers available, including prescription drugs, over-the-counter products, intradermal injections, and even herbal supplements. Finasteride reduces DHT in two ways.
- Oral finasteride reduces DHT everywhere in the body. Beyond just the scalp, it reduces DHT in the blood, brain, and prostate.
- Topical finasteride attempts to reduce DHT only where it is applied – in the scalp – with minimal effects on DHT levels elsewhere.
However, it’s far easier than most realize for topical finasteride to go systemic. And when it does, it potentially reduces DHT everywhere – leading to the very side effects it’s meant to prevent.
Topical Finasteride Can Still Go Systemic
Why? When it comes to lowering DHT (the goal of the drug), finasteride has a highly-sensitive, dose-dependent response curve. This means that while 0.01 mg of finasteride barely reduces any DHT at all, 0.1 mg reduces almost as much DHT as 5 mg, a much larger dose.
This implies that when applying topical finasteride, only a tiny fraction of it needs to go systemic in order to produce the same DHT-lowering effects as oral finasteride. If this happens, the purpose of using the topical formulation is completely defeated.
Research suggests a 1% topical finasteride formulation, applied twice daily, is ‘non-inferior,’ meaning equivalent, to 1 mg oral finasteride tablets.[2]https://www.ncbi.nlm.nih.gov/pubmed/19172031 And while that’s a positive in terms of hair growth, using this amount topically twice daily pretty much guarantees systemic absorption. In other words, if 1% topical is the equivalent of 1 mg oral, we can expect it to reduce serum DHT levels by 71%.
So what if we choose a formula with even lower percentages of the active drug? A study on 0.25% topical finasteride showed just a 35% reduction in serum DHT levels versus 55% for the oral medication, and yet similar outcomes for hair regrowth in both oral and topical groups.[3]https://onlinelibrary.wiley.com/doi/10.1111/jdv.17738
So what if we go even lower? A study on 0.005% alcohol-based topical finasteride instructed participants to use the formulation twice daily, with 1 mL applied per session.[4]https://dx.doi.org/10.3109%2F09546639709160517 This led to no appreciable changes in serum DHT levels… meaning that for this ultra-low topical formulation, there was little-to-no systemic absorption (and presumably, few-to-no side effects). The good news? This group still experienced great hair growth outcomes.
So Just Reach for the Lowest Percentage, Yes?
It’s tempting from the above to infer we’ve reached the end of our story. As long as we reach for the lowest percentage of topical finasteride, we can be free from worries about the drug’s systemic effects. But, reality is a bit more complicated than that.
Systemic absorption isn’t just about dilution percentages. Topical finasteride’s systemic absorption actually depends on (at least) for variables:
- The drug’s formulation. Common carrier agents include alcohol and propylene glycol.
- The amount applied per use. The amount of topical finasteride applied to the scalp greatly varies.
- The application frequency. Applying the drug twice per day can more than double its effects on serum DHT.
- The days of application. Topical finasteride can take many days to “leak” into the blood before its final effects on serum DHT are realized.
For an example, see this figure from a 2014 study measuring how one versus two applications of 1 mL of 0.25% topical finasteride affected serum DHT levels.[5]https://www.ncbi.nlm.nih.gov/pubmed/25074865
Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen levels in healthy male volunteers. International journal of clinical pharmacology and therapeutics, 52(10), 842–849.
Over a 24-hour period, what were the findings?
- One application (i.e., 1 mL of 0.25% topical finasteride) lowered blood DHT levels by ~20%
- Two applications (i.e., 2 mL total) lowered blood DHT levels by ~70%
As such, we need to factor in these variables if we plan on sticking with topical finasteride for the long-term. Otherwise, we risk lowering our DHT levels to the same degree as 1 mg of oral finasteride, which would defeat the purpose of using the topical in the first place.
Topical Finasteride and Serum DHT: What Do The Clinical Studies Say?
Let’s take a closer look at some of the studies mentioned above. In addition to the percentage of active drug in the formula, there are a few more variables to understand. How much of the topical was applied, and which carrier agents were used?
The table below shows how even a .25% formula, for example, can reduce DHT in the serum by 24-70%
Based on this table, our total mg of daily finasteride exposure is probably the biggest factor in determining systemic leakage. And our total daily exposure (in mg) is a function of topical finasteride dilution (%) and the amount (mL) applied daily.
Knowing this, we can turn this table into a chart and sort it by mg of daily exposure. In doing so, we see a clear trend:
The good news? At both extremes of this chart – 0.091 mg and 2.275 mg daily – topical finasteride was shown to produce clinical results in improving hair parameters. Based on this, if we’re going for topical finasteride, we probably want to be prescribed topical finasteride solutions that net us daily exposure volumes of 0.228 mg and lower. After all, at 0.091 mg of topical finasteride daily, no systemic effects on DHT were observed.[6]https://www.tandfonline.com/doi/abs/10.3109/09546639709160517
In other words, low dilutions (i.e., 1-2 mL of 0.005% to 0.02% of topical finasteride) confer significant benefits but at reduced risks of side effects due to lower systemic absorption – provided that guidelines for daily amounts (in mL) are also followed.
So What’s the Perfect Amount and Formula?
Our analyses from member-submitted lab tests and the clinical data suggest that 10-15% of topical finasteride will enter the bloodstream, at least when it’s formulated with alcohol and/or propylene glycol as carrier ingredients (as most compounding pharmacies do). So, if we apply 1-2 mL of 0.005% to 0.02% topical finasteride, this might equate to just 0.01-0.03 mg of systemic drug exposure.
For the overwhelming majority of people, that’s not enough systemic leakage to significantly lower DHT levels in the body. But it is enough to produce great hair loss outcomes.Our partial analysis estimating systemic leakage of topical finasteride from the study: Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial (2021)
Join our Membership Program to get the full analysis.
Long-story short: stick to 1-2 mL of 0.005% to 0.02% topical finasteride solutions. This equates to roughly 0.1-0.2mg of daily finasteride exposure to the scalp. Any more than that, and there’s risk of significant systemic leakage, which defeats the purpose of using topical finasteride altogether.
How to Maximize Gains and Minimize Side Effects
Maximizing gains while minimizing the side effects of finasteride can be done, but it’s not a perfect science. We’ve outlined several tips below that we think might help. Due to individual variance, the most important step is to always start with testing.
1. Get Lab Tests for DHT
The goal with topical finasteride is to reduce the risk of side effects. To do this, we must minimize the amount of finasteride that leaks into the bloodstream. The best way to do this isn’t to rely on estimated metrics from clinical studies, but to collect personal data.
Get a serum DHT test before using the topical to establish a baseline. Then, DHT levels should be retested at one month to gauge just how much is going systemic. Testing is easier than most expect, and in our experience, the expenses are worth the peace of mind.
If future lab tests deviate from baseline, it’s an indication of just how much topical finasteride is going systemic. According to the clinical data, 30 days of application is more than enough time for finasteride to saturate at its maximum levels in the scalp and serum.
As such, measuring DHT levels after one month of finasteride use offers a great reference point to see if topical finasteride is impacting serum DHT levels. If needed, changes can be made to the application or use frequency depending on any changes to your blood levels of DHT.
2. Understand That DHT Levels Fluctuate Diurnally
Keep in mind, some fluctuation in DHT levels is normal. DHT levels fluctuate throughout the day and across seasons. As such, 15-20% differences across tests are normal and expected. Anything beyond 20% suggests that topical finasteride might be having slight systemic effects.
Because of this fluctuation, however, it’s important to get blood draws done at the same time of day – preferably in the morning and while in a fasted state. Also, try not to make drastic changes to diet, lifestyle, or environment prior to testing. Heavy drinking, deviations from a typical daily diet, the introduction of creatine powders, and/or sleep deprivation can all influence DHT levels and muddy test results. In the 3-5 days prior to the second test, try to keep things as they were when you first went in for testing.
3. Avoid supplementing with Quercetin and/or Creatine
Maintaining systemic DHT levels, while maximizing the effects of finasteride, isn’t just about the drug. There are other activities that could potentially affect DHT levels. Supplementing with quercetin and/or creatine is one common mistake that could impact results.
Studies on mice suggest that quercetin can inhibit the DHT-reducing effects of finasteride.[7]https://joe.bioscientifica.com/view/journals/joe/181/3/493.xmlWhile the translatability to humans has not yet been studied, the dosages used in these mouse models were comparable to what humans typically consume from quercetin supplements. As such, it may be best for those using finasteride to avoid this supplement.
When it comes to creatine, one study found that in training athletes, creatine supplementation increased serum DHT levels by over 70%.[8]https://pubmed.ncbi.nlm.nih.gov/19741313/ While the study was small, we have to reconcile these findings with the reality that for the overwhelming majority of training athletes, creatine is unnecessary. Bodybuilders can still look great without using it, and so can the every-day gym goer. Competitive bodybuilders are, however, a different story.
4. Set Realistic Expectations and Track Progress
Finasteride is the best-studied intervention for androgenic alopecia. In order to appreciate its full effects, consider the following:
- Know the timeline for results. Clinical studies show that finasteride improves pattern hair loss outcomes in 80-90% of men. Having said that, cosmetic degrees of hair regrowth don’t often occur until after 10-12 months of treatment. Moreover, the drug’s full effects typically take two years to manifest, with the biggest degrees of cosmetic regrowth occurring between year one and year two. As such, don’t expect any miracles in the first six months, it may take the full two years to see an effect.
- Track progress. It’s hard to know if something is improving without objective measurements. While not all of us have access to trichoscopic hair-counting equipment, most of us do have a smartphone, which can easily be used to take photos to track progress (particularly when the photosets are standardized). This can be done relatively effortlessly.
Following the tips above allows for the best possible shot at measurable hair growth results, while minimizing the potential effects of systemic DHT reduction.
Summary
Oral finasteride works great for male pattern hair loss, but by significantly reducing serum DHT, it can cause unwanted side effects.
Topical finasteride was developed as a solution, with a goal to reduce DHT in the scalp only. But when applied in high percentages or large amounts, it too, can go systemic and reduce serum DHT levels.
To minimize the side effects of finasteride without missing out on the benefits, stick to 1-2 mL of 0.005% to 0.02% topical finasteride solutions. This equates to roughly 0.1-0.2mg of daily finasteride exposure to the scalp.
The best way to ensure a reduced risk for side effects is to track serum DHT levels. Establish a baseline by taking one test before using finasteride, then test again one month later.
References[+]
References ↑1 https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑2 https://www.ncbi.nlm.nih.gov/pubmed/19172031 ↑3 https://onlinelibrary.wiley.com/doi/10.1111/jdv.17738 ↑4 https://dx.doi.org/10.3109%2F09546639709160517 ↑5 https://www.ncbi.nlm.nih.gov/pubmed/25074865 ↑6 https://www.tandfonline.com/doi/abs/10.3109/09546639709160517 ↑7 https://joe.bioscientifica.com/view/journals/joe/181/3/493.xml ↑8 https://pubmed.ncbi.nlm.nih.gov/19741313/ The range of treatments available for hair loss can seem bewildering. Over-the-counter options range from clinically proven pharmaceuticals to ineffective fads and overpriced shampoos. Prescription options can provide robust results for many, but come with a long list of potential side effects, drawbacks, and tradeoffs.
We started Perfect Hair Health to provide clarity and evidence-based information in a space that is too often characterized by exaggerated claims and misinformation. Unfortunately, the more we learned about the hair loss industry, the more stark the reality of the problems became. While telehealth companies have made effective treatments more accessible than ever, too often brands rely on poorly formulated pharmaceuticals that present unnecessary risks to users and ignore clinical data.
We co-founded Ulo in response to these issues to provide evidence-backed hair loss treatments that can be truly customized to work for you. In this Ulo review, we’ll take a deeper look at the issues in the industry and how Ulo does things differently. We’ll review the clinical evidence supporting Ulo’s choice of pharmaceuticals and ingredients, and see how its approach translates into real-world outcomes for safety, tolerability, and hair regrowth.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What is Ulo?
After many years of consumer advocacy and watching hair loss brands provide suboptimal service and promote ineffective treatments, we co-founded Ulo.
Ulo is a telehealth provider focused on hair loss treatments. It’s committed to evidence-based treatments, real personalization, and customer safety. At its core, Ulo is built around three principles: evidence, personalization, and consumer safety. These principles are applied to ingredient selection, dosing strategies, and formulation design.
Problems in the Hair Loss Industry
There is a range of options available for hair loss that can produce real results. Best-selling pharmaceuticals like minoxidil and finasteride are backed by many years of clinical research, as well as the personal experiences of millions of users. So do you just need to pick the treatment that offers the most powerful regrowth?
Unfortunately, it’s not so simple. Finding the best treatment for you depends on finding the right dose and the most appropriate delivery method, as well as understanding potential side effects and whether combinations of treatments might optimize your results. That’s why customization is so essential for producing the best results.
However, instead of providing clear routes to effective treatments, telehealth companies sometimes compound these issues by introducing a whole new set of problems.
Problem #1: Unproven Treatments and Ingredients
The hair loss telehealth industry is a crowded one, and it’s sometimes hard to stand out. Because of this, many brands turn to unique ingredients and trends to attract new customers. Unfortunately, many of these treatments are not backed up by robust evidence.
It’s important to differentiate between strong evidence, which typically comes from multiple clinical trials conducted in humans, and weaker, limited evidence. Many drugs, compounds, and natural ingredients show some promise in petri dishes in the lab. Many even appear to regrow hair in animal models, typically mice. But this rarely translates directly into measurable outcomes in real people.
Unfortunately, some brands use this limited evidence as a basis to include these ingredients in treatments, even using them as unique selling points. If you’ve been watching progress in the hair loss industry for as long as we have, you’ll have seen these trends come and go.
Today, it’s peptides. Peptides have exploded in popularity and are touted as the solution to a wide range of health issues. You’re most likely to come across copper peptides in lists of ingredients for over-the-counter hair loss products, as well as prescription formulations.
One copper peptide, called AHK-Cu, has been shown to enhance the growth of human hair follicle cells in the lab, while another, GHK-Cu, has been shown to have anti-inflammatory properties.[1]Gruchlik, A., Jurzak, M., Chodurek, E. W. A., & Dzierzewicz, Z. (2012). Effect of Gly-Gly-His, Gly-His-Lys and their copper complexes on TNF-alpha-dependent IL-6 secretion in normal human dermal … Continue reading These experiments on cells in a petri dish are an important step in finding new drugs and understanding the molecular mechanisms behind hair loss. Unfortunately, they don’t tell us much about what will happen in actual humans.
For that, we need clinical data, preferably from placebo-controlled clinical trials. The only clinical trial we’re aware of that tested copper peptides in humans found no statistical difference in hair length or thickness after 6 months of treatment with GHK-Cu.[2]Lee, W. J., Sim, H. B., Jang, Y. H., Lee, S. J., Kim, D. W., & Yim, S. H. (2016). Efficacy of a complex of 5-aminolevulinic acid and glycyl-histidyl-lysine peptide on hair growth. *Annals of … Continue reading
That is not to say that copper peptides might not be effective at the right dose for the right person. But without clinical evidence, there’s no reason for you to expect improved outcomes, or for telehealth and hair loss brands to market them for hair loss. Copper peptides are only one example of a vast range of unproven ingredients that are regularly used in expensive hair loss treatments.
You can read more about understanding evidence in the hair loss industry in our article.
Problem #2: Irritants and Potentially Dangerous Ingredients
Ineffective ingredients can be an expensive, but ultimately harmless, addition to formulations. But what if ingredients went beyond useless, and were actually actively harmful? Unfortunately, that is potentially the case in some treatments currently on the market.
Propylene Glycol
On the low end of the scale are irritants, like propylene glycol. Propylene glycol is used in hair loss treatments as a carrier, meaning it’s a vehicle and helper for the active ingredients. Propylene glycol helps moisturize the skin and has been shown to improve the penetration of drugs into the skin in some models.[3]Carrer, V., Alonso, C., Pont, M., Zanuy, M., Córdoba, M., Espinosa, S., Barba, C., Oliver, M. A., Martí, M., & Coderch, L. (2020). Effect of propylene glycol on the skin penetration of drugs. … Continue reading,[4]Lessmann, H., Schnuch, A., Geier, J., Uter, W. (2005). Skin-sensitizing and irritant properties of propylene glycol. Contact Dermatitis. 53(5). 247-259. Available at: … Continue reading
Unfortunately, it’s also a known irritant and can cause contact dermatitis in a small proportion of people.[5]Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelb). 13(5). 1157-1169. Available at: … Continue reading. This might be a fair trade-off if it were an essential ingredient. However, it’s perfectly possible to produce effective topicals for hair loss without propylene glycol.
Ingredients like propylene glycol can cause inflammation and irritation, which can make hair loss worse and stop treatments from working. Some brands use additional ingredients to dampen this inflammation, rather than stopping it at the source by removing potentially inflammatory, inessential components.
Corticosteroids
Corticosteroids, for example, help lower scalp inflammation and may provide additional benefits for people with alopecia areata or scarring alopecia.[6]Alsantali, A. (2011). Alopecia areata: a new treatment plan. *Clinical, Cosmetic and Investigational Dermatology.* 4. 107–115. Available at: https://doi.org/10.2147/CCID.S22767 However, they are not designed for long-term use, and applying steroids like fluocinolone to your skin every day for a year can cause conditions like skin thinning and spider veins.[7]Torok, H. M., Jones, T., Rich, P., Smith, S., & Tschen, E. (2005). Hydroquinone 4%, tretinoin 0.05%, fluocinolone acetonide 0.01%: a safe and efficacious 12-month treatment for melasma. *Cutis.* … Continue reading What’s more, hair loss treatments are typically taken daily for many years, even decades, further increasing the likelihood of complications.
You can read more about the safety concerns behind corticosteroids in our full-length article.
Latanoprost
You might also spot hair loss formulations that include a drug called latanoprost. After initial promise in studies, latanoprost has had very mixed results from a number of clinical trials. Regardless, it was branded as new and exciting, becoming a popular treatment option.[8]Blume-Peytavi, U., Lönnfors, S., Hillmann, K., & Garcia Bartels, N. (2012). A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by … Continue reading,[9]Bloch, L. D., Escudeiro, C. C., Sarruf, F. D., & Sakai Valente, N. Y. (2018). Latanoprost and minoxidil: comparative double-blind, placebo-controlled study for the treatment of hair loss. … Continue reading
However, latanoprost was initially developed as a glaucoma treatment, and studies have shown that it can potentially change iris color in anywhere between 10% and 70% of patients.[10]Teus, M. A., Arranz-Marquez, E., & Lucea-Suescun, P. (2002). Incidence of iris colour change in latanoprost treated eyes. *British Journal of Ophthalmology.* 86(10). 1085–1088. Available at: … Continue reading This risk of permanent side effects might be reasonable for glaucoma patients, but it’s difficult to justify for those with hair loss who might not see any benefits at all.
Problem #3: Mega-Dosed Topicals
Finasteride is an effective treatment for hair loss that works by lowering the level of dihydrotestosterone (DHT) in the scalp. DHT drives changes in the follicles that lead to androgenic alopecia (AGA). However, finasteride can also lower DHT in other parts of the body, leading to side effects.[11]Zito, P. M., Bistas, K. G., Patel, P., & Syed, K. (2024). Finasteride. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: … Continue reading
Topical treatments are often presented as a way to localize drug activity to the scalp while minimizing systemic exposure. However, the extent to which drugs stay localized is heavily dependent on dosing.
Importantly, the relationship between the dose of finasteride and DHT reduction is not linear. When taken orally, even small amounts of the drug can significantly suppress serum DHT levels, reflecting systemic absorption of the drug into the blood. This means that for topical formulations applied to the scalp, even small amounts of absorption into the bloodstream can have an effect.
Data we’ve compiled from multiple clinical trials shows that only at doses of 0.005% is there no significant decrease in serum DHT. Despite this, many telehealth brands now offer high-strength topical finasteride formulations, often in the range of 0.25% to 0.3%, up to 60 times higher. These concentrations significantly increase the chances of side effects caused by systemic DHT reduction, while offering diminishing returns in terms of regrowth potential.
Figure 2. Data collected from multiple studies shows only application of very low (0.005%) concentrations of finasteride does not lower serum DHT levels.
If the aim of topical formulations is to reduce systemic absorption of DHT and minimize side effects, then mega-dose topicals run counter to this basic premise.
You can read more about finasteride dosing in our in-depth article.
Problem #4: No Customization
Hair loss is not a one-size-fits-all condition. The rate of hair loss, response to treatment, and incidence of side effects can vary significantly from person to person. Despite this, many telehealth platforms rely on highly standardized formulations.
This is a major issue when users experience side effects. The absence of options to decrease doses or look for alternatives can lead people to struggle through their treatment, or to abandon it entirely.
Conversely, if you find that your hair growth has plateaued or hair loss is increasing, there is no route to increase dosing or add additional treatments to your plan.
Problem #5 Improper Dutasteride Compounding
Dutasteride is a highly effective drug for reducing DHT, but its effectiveness depends heavily on its formulation. Unlike finasteride or minoxidil, dutasteride is poorly soluble in water and requires a lipid-based delivery system to be properly absorbed. That’s why it’s typically formulated as a soft-gel capsule
However, some providers are now compounding dutasteride into powder form in order to combine it with other ingredients, such as minoxidil or vitamins, in a single capsule. This can then be marketed as a more convenient, all-in-one solution.
Unfortunately, this may significantly reduce the drug’s bioavailability. To test this, we performed a small test. One participant took 3 pills of 0.5 mg powdered dutasteride (i.e., 1.5 mg total) and then checked changes in their blood DHT levels 12 hours later. Based on pharmacological data, we’d expect to see around an 80-90% reduction in DHT. However, we saw a reduction of only 13.6%.
Compounding of dutasteride without proper emulsifiers may be leading to lower DHT reduction, and therefore worse outcomes for hair regrowth.
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Does Ulo Solve These Problems?
Rather than searching for the newest trends and gimmicks, Ulo only uses pharmaceuticals and treatments backed by robust clinical evidence. This means clinical trials performed in humans, not only preliminary data from cell-based models or animal studies.
When it comes to incorporating these clinically validated ingredients into effective treatments, Ulo prioritizes customizability rather than relying on high-strength, one-size-fits-all topical formulations. Ulo offers both low-dose and full-strength versions of topical finasteride and dutasteride, with optional additions such as higher-strength minoxidil, tretinoin, caffeine, melatonin, or cetirizine, depending on individual needs and tolerability.
What Does Ulo Offer?
Ulo offers a range of proven hair loss treatments at a range of concentrations. Here, we’ll explore the catalog in detail and look at some of the evidence supporting it.
Finasteride
As we’ve noted above, finasteride is backed by many years of clinical evidence and the experience of hundreds of thousands of users across the globe. [12]Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride … Continue reading Oral finasteride, typically taken as a 1 mg tablet daily, is one of only two FDA-approved treatments for hair loss. Ulo offers oral finasteride at the FDA-approved concentration of 1mg.
As we’ve already discussed, androgenic alopecia (AGA) can be driven by increases in DHT. Finasteride works by blocking an enzyme called 5ɑ-reductase (5AR), which converts testosterone into DHT. Therefore, by blocking this conversion, the drug can decrease DHT in the scalp, and therefore inhibit its effects on hair.[13]Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/
However, there are some downsides to oral finasteride. Because taking oral finasteride decreases DHT throughout the body, it can cause secondary side effects. This inlcludes decreased libido, erectile dysfunction, and ejaculation disorders, which affect a small proportion of users. [14]Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic … Continue reading, [15]Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has … Continue reading
Due to these side effects, topical formulations of finasteride have been developed to localize the drug to the scalp. As we’ve already seen, how localized the drug stays depends on the dose and volume of finasteride applied. Ulo offers finasteride at a very low dose of 0.005% and at 0.2%.
Very low concentrations are designed to minimize systemic exposure as much as possible. One small clinical study showed that 0.005% topical finasteride found no significant changes in serum DHT levels, suggesting minimal systemic absorption. Despite this, participants still experienced meaningful hair growth.[16]Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia: preliminary evaluations after a … Continue reading
In line with Ulo’s commitment to customization, a higher dose is available for those looking for a more powerful solution for growth. Finding the right balance of strength vs side effects is tricky. One study showed that 0.1% topical finasteride showed comparable efficacy to 1 mg oral finasteride, while a more recent trial found that 0.25% topical significantly improved target area hair count compared to placebo.[17]Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of … Continue reading
Increasing topical concentrations above 0.2% appears to provide diminishing returns for hair growth while increasing the risk of side effects. As such, this is the highest concentration available from Ulo.
Importantly, Ulo’s topical formulations are fully customizable, including the addition of growth stimulants and other add-ons to help you better find the right combination for you.
Minoxidil
Minoxidil is one such growth stimulant, which can work alone or in combination with treatments. Topical minoxidil is an FDA-approved treatment for hair loss at 5% concentrations, and it works through different mechanisms to finasteride. It alters the hair cycle, promoting entry of hair follicles into the growth phase, while shortening the resting phase. It can also increase blood flow around follicles and activate pro-growth molecular signaling pathways.[18]Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: … Continue reading
Multiple clinical trials have demonstrated the efficacy of minoxidil for increasing hair growth. [19]Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189 Because minoxidil works via different mechanisms, using the drug in combination with DHT-blockers like finasteride can promote growth by multiple routes.
Ulo offers minoxidil at 7%, higher than the over-the-counter concentration of 5%. Topical minoxidil is typically safe, with the most common side effects including scalp irritation and itching. Many incidences of itching and irritation are likely due to the inclusion of propylene glycol, which is entirely absent from all of Ulo’s formulations.
Because the risk of adverse effects is relatively low, this higher concentration can improve results without incurring significant risk. Perfect hair health members have consistently reported strong results using 7% minoxidil, particularly when combined with enhancers like tretinoin that can help the drug to penetrate into the skin.
DHT-blockers like finasteride are not typically recommended for women who may become pregnant, due to significant danger during pregnancy. As such, minoxidil may be a better solution for pre-menopausal women, or anyone concerned about side effects.
Ulo also offers oral minoxidil in a 2.5mg tablet. This may be a good solution for users who struggle with the appearance of their hair after using topical formulations, or who find it hard to maintain consistent, daily application. Treating hair loss is a long game, and often the best solution is one that you can apply consistently every day over many years.
Oral minoxidil does come with some increased risk of side effects that users should be aware of. These include hypertrichosis (excessive hair growth outside of the scalp), mild fluid retention or ankle swelling, and lightheadedness. Cardiac issues are a more serious, but less commonly reported, issue.[20]Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair … Continue reading
Again, customization is key. Minoxidil can be effective on its own, or in combination with other therapies. Ulo offers a range of options to suit your needs and priorities.
Dutasteride
Like finasteride, dutasteride lowers levels of DHT by blocking 5AR. However, dutasteride blocks two forms of the enzyme, while finasteride only blocks one, which can lead to even greater DHT reduction. Clinical evidence consistently supports this, and across multiple studies, 0.5 mg oral dutasteride has been shown to outperform 1 mg finasteride in increasing total hair count, improving hair thickness, and reversing follicle miniaturization.[21]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading
However, this mechanism of action can also result in side effects, as we saw with finasteride. For this reason, topical formulations were developed. A 2025 randomized controlled trial comparing topical dutasteride (0.01-0.05%) to oral finasteride reported greater improvements in hair density and global photographic assessments in the topical dutasteride group.[22]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading
Ulo offers dutasteride at concentrations of 0.02% to 0.2%, again allowing users to balance tolerability with the strength of treatment.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Tretinoin (Retinoic Acid)
Perfect Hair Health’s many years of consumer advocacy and the insights from our community have highlighted the importance of add-ons to well-established pharmaceutical options.
Tretonoin is a derivative of retinoic acid and has been shown to have a modest impact on hair growth when used alone.[23]Bazzano, G. S., Terezakis, N., & Galen, W. (1986). Topical tretinoin for hair growth promotion. *Journal of the American Academy of Dermatology.* 15(4). 880–893. Available at: … Continue reading However, it’s most effective when used alongside minoxidil.
For minoxidil to work, it first has to be activated in the scalp by an enzyme called SULT1A1. As a result, the efficacy of minoxidil is highly dependent on levels of SULT1A1. Tretinoin has been shown to increase SULT1A1 in the outer root sheath, enhancing the local activation of minoxidil. [24]Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvin, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin enhances minoxidil response in … Continue reading
Ulo allows you to add tretinoin to your formulations to optimize their efficacy and drive better results, and may be particularly beneficial for patients who have responded poorly to minoxidil or have low levels of SULT1A1.
Melatonin
Another available add-on is melatonin. While it doesn’t provide the same kind of results as pharmaceutical options, topical melatonin has been shown to reduce rates of active hair shedding and, in some cases, improve hair density and counts, particularly in individuals with early-stage AGA.[25]Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., & Elsner, P. (2012). Topical melatonin for treatment of androgenetic alopecia. *International Journal of Trichology.* 4(4). 236–245. … Continue reading
Ulo offers 0.01% melatonin to add to topical hair loss formulations. Melatonin alone is unlikely to drive significant results, but as an adjunct to drugs like finasteride and minoxidil, it can help reduce shedding and is recommended for those with early signs of AGA.
Caffeine
Caffeine is commonly found in shampoos and conditioners, though it’s unlikely to have any significant impact on hair growth in these wash-off treatments. There is some evidence to suggest that topical solutions of 0.2% can help boost growth, though caffeine hasn’t been as thoroughly investigated as the other ingredients we see here.[26]Dhurat, R., Chitallia, J., May, T. W., Jayaraaman, A. M., Madhukara, J., Anandan, S., Vaidya, P., & Klenk, A. (2018). An open-label randomized multicenter study assessing the noninferiority of a … Continue reading
Instead of acting as the primary active ingredient, caffeine is also best used as an add-on. Clinical evidence suggests that 5% minoxidil with 1% caffeine and 1.5% azelaic acid can be more effective than minoxidil alone.[27]Pazoki-Toroudi, H., Moghadam, R. M., Ajami, M., Nassiri-Kashani, M., Ehsani, A., Tabatabaie, H., Khani, S., Abbasi, Z., Shizarpour, M., Firooz, A., & Dowlati, Y. (2013). The efficacy and safety … Continue reading
Caffeine, therefore, is probably best understood as a relatively low-risk addition that may provide modest benefits when used alongside better-supported therapies. For this reason, Ulo offers 0.2% caffeine as an add-on to prescription therapies.
Cetirizine
Cetirizine is an antihistamine best known as a treatment for allergies. There is, however, encouraging clinical evidence suggesting it can help treat hair loss. In one placebo-controlled trial, 43.3% of participants using topical cetirizine showed hair regrowth, compared to none in the placebo group. In another trial, cetirizine improved hair density, although minoxidil performed better overall.[[Zaky, M. S., Abo Khodeir, H., Ahmed, H. A., & Elsaie, M. L. (2021). Therapeutic implications of topical cetirizine 1% in treatment of male androgenetic alopecia: a case‐controlled study. Journal of Cosmetic Dermatology. 20(4). 1154-1159. Available at: https://doi.org/10.1111/jocd.13940}}
It’s unclear whether cetirizine will be effective as a stand-alone treatment. Following this evidence, Ulo offers it as an add-on to better evidenced treatments to take advantage of its hair growth potential. What’s more, because it’s so widely used in allergy medications, the safety profile of cetirizine is very well understood.
Is Ulo the #1 Customized Hair Loss Treatment in 2026?
There is a range of telehealth brands that offer great solutions for many people. Some companies have made amazing progress in improving access to hair loss treatments, offering combinations of oral and topical therapies, simplified prescription processes, and greater flexibility in delivery.
However, these solutions are often still built around standardized formulations and rigid ingredient lists. For many people, these platforms can still deliver solid results, particularly for those who respond well to first-line therapies and don’t require fine-tuning.
Ulo builds on the strengths of these platforms, including accessibility, convenience, and clinically validated treatments, but expands them by offering genuine flexibility in treatment design. Users can choose between oral and topical treatments, low-dose and full-strength formulations, and can add evidence-supported adjuncts like tretinoin, melatonin, caffeine, or cetirizine depending on their goals, tolerability, and stage of hair loss.
Available Ulo before and afters and reviews suggest this approach is providing meaningful results for customers, with treatment on their terms. Some users need to start cautiously to minimize side effects, while others need stronger interventions or add-ons to push beyond a plateau. Ulo’s range of oral and topical finasteride and dutasteride, higher-strength minoxidil, and customizable adjuncts allows treatment to be adjusted around the individual.
Ulo distinguishes itself by combining a foundation of evidence-based therapies with customization, formulation control, and evidence-based decision-making that are still largely absent elsewhere.
Looking to the Future of Ulo
Ulo’s approach is not limited to its current product line. A core part of its long-term strategy is continued investment in research and development, with the goal of improving both the effectiveness and safety of hair loss treatments over time. This includes work on new proprietary base formulations designed to enhance localization, helping active ingredients remain targeted to the scalp while minimizing systemic exposure.
Future treatments also aim to provide more options for individuals with sensitivities, as well as a broader range of concentrations, formulations, and ingredient combinations tailored to different stages of hair loss and treatment goals.
Final Thoughts
At Perfect Hair Health, our goal has never been to criticize the industry without cause. However, we continue to highlight issues because, as they persist, they continue to provide a less effective, less safe treatment space for consumers.
Ultimately, we would prefer a world where these issues no longer exist, where treatments are consistently evidence-based, properly formulated, and tailored to the individual. That would be better for consumers, and it would mean there’s less need to constantly question the quality of what’s being offered.
Ulo is our attempt to move in that direction. It reflects the standards we’ve been advocating for over the past decade: better formulations, better dosing strategies, and a more thoughtful approach to customization.
References[+]
References ↑1 Gruchlik, A., Jurzak, M., Chodurek, E. W. A., & Dzierzewicz, Z. (2012). Effect of Gly-Gly-His, Gly-His-Lys and their copper complexes on TNF-alpha-dependent IL-6 secretion in normal human dermal fibroblasts. *Acta Poloniae Pharmaceutica.* 69(6). 1303–1306. Available at: https://pubmed.ncbi.nlm.nih.gov/23285694/ ↑2 Lee, W. J., Sim, H. B., Jang, Y. H., Lee, S. J., Kim, D. W., & Yim, S. H. (2016). Efficacy of a complex of 5-aminolevulinic acid and glycyl-histidyl-lysine peptide on hair growth. *Annals of Dermatology.* 28(4). 438–443. Available at: https://doi.org/10.5021/ad.2016.28.4.438 ↑3 Carrer, V., Alonso, C., Pont, M., Zanuy, M., Córdoba, M., Espinosa, S., Barba, C., Oliver, M. A., Martí, M., & Coderch, L. (2020). Effect of propylene glycol on the skin penetration of drugs. *Archives of Dermatological Research.* 312(5). 337–352. Available at: https://doi.org/10.1007/s00403-019-02017-5 ↑4 Lessmann, H., Schnuch, A., Geier, J., Uter, W. (2005). Skin-sensitizing and irritant properties of propylene glycol. Contact Dermatitis. 53(5). 247-259. Available at: https://doi.org/10.1111/j.0105-1873.2005.00693.x. ↑5 Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelb). 13(5). 1157-1169. Available at: https://doi.org/10.1007/s13555-023-00919-x ↑6 Alsantali, A. (2011). Alopecia areata: a new treatment plan. *Clinical, Cosmetic and Investigational Dermatology.* 4. 107–115. Available at: https://doi.org/10.2147/CCID.S22767 ↑7 Torok, H. M., Jones, T., Rich, P., Smith, S., & Tschen, E. (2005). Hydroquinone 4%, tretinoin 0.05%, fluocinolone acetonide 0.01%: a safe and efficacious 12-month treatment for melasma. *Cutis.* 75(1). 57–62. Available at: https://pubmed.ncbi.nlm.nih.gov/15732437/ ↑8 Blume-Peytavi, U., Lönnfors, S., Hillmann, K., & Garcia Bartels, N. (2012). A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by latanoprost 0.1% on hair growth and pigmentation in healthy volunteers with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 66(5). 794–800. Available at: https://doi.org/10.1016/j.jaad.2011.05.026 ↑9 Bloch, L. D., Escudeiro, C. C., Sarruf, F. D., & Sakai Valente, N. Y. (2018). Latanoprost and minoxidil: comparative double-blind, placebo-controlled study for the treatment of hair loss. *Surgical & Cosmetic Dermatology.* 10(1). 39–43. Available at: https://doi.org/10.5935/scd1984-8773.20181011015 ↑10 Teus, M. A., Arranz-Marquez, E., & Lucea-Suescun, P. (2002). Incidence of iris colour change in latanoprost treated eyes. *British Journal of Ophthalmology.* 86(10). 1085–1088. Available at: https://doi.org/10.1136/bjo.86.10.1085 ↑11 Zito, P. M., Bistas, K. G., Patel, P., & Syed, K. (2024). Finasteride. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK513329/ (Accessed: October 2025) ↑12 Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride In The Treatment Of Men With Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 39(4 Pt 1). 578-589. Available at: https://doi.org/10.1016/s0190-9622(98)70007-6 ↑13 Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ ↑14 Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic review. *The Journal of Clinical and Aesthetic Dermatology.* 9(7). 56–62. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC5023004/ ↑15 Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has comparable urinary symptom efficacy and prostate volume reduction, but less sexual side effects and breast complications than dutasteride. *International Journal of Clinical Practice.* 66(11). 1052–1055. Available at: https://doi.org/10.1111/j.1742-1241.2012.03010.x ↑16 Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia: preliminary evaluations after a 16-month therapy course. *Journal of Dermatological Treatment.* 8(3). 189–192. Available at: https://doi.org/10.3109/09546639709160517 ↑17 Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of Dermatology, Venereology and Leprology.* 75. 47. Available at: https://doi.org/10.4103/0378-6323.45220 ↑18 Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑19 Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189 ↑20 Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair loss: a multicenter study of 1404 patients. Journal of the American Academy of Dermatology. 84(6). 1644–1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑21 Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. *Indian Journal of Dermatology, Venereology and Leprology.* 83. 47. Available at: https://doi.org/10.4103/0378-6323.188652 ↑22 Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑23 Bazzano, G. S., Terezakis, N., & Galen, W. (1986). Topical tretinoin for hair growth promotion. *Journal of the American Academy of Dermatology.* 15(4). 880–893. Available at: https://doi.org/10.1016/S0190-9622(86)80024-X ↑24 Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvin, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin enhances minoxidil response in androgenetic alopecia patients by upregulating follicular sulfotransferase enzymes. Dermatologic Therapy. 32(3). 12915. Available at: https://doi.org/10.1111/dth.12915 ↑25 Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., & Elsner, P. (2012). Topical melatonin for treatment of androgenetic alopecia. *International Journal of Trichology.* 4(4). 236–245. Available at: https://doi.org/10.4103/0974-7753.111199 ↑26 Dhurat, R., Chitallia, J., May, T. W., Jayaraaman, A. M., Madhukara, J., Anandan, S., Vaidya, P., & Klenk, A. (2018). An open-label randomized multicenter study assessing the noninferiority of a caffeine-based topical liquid 0.2% versus minoxidil 5% solution in male androgenetic alopecia. *Skin Pharmacology and Physiology.* 30(6). 298–305. Available at: https://doi.org/10.1159/000481141 ↑27 Pazoki-Toroudi, H., Moghadam, R. M., Ajami, M., Nassiri-Kashani, M., Ehsani, A., Tabatabaie, H., Khani, S., Abbasi, Z., Shizarpour, M., Firooz, A., & Dowlati, Y. (2013). The efficacy and safety of minoxidil 5% combination with azelaic acid 1/5% and caffeine 1% solution on male pattern hair loss. *Journal of Investigative Dermatology.* 133. S84–S84. Available at: https://doi.org/10.1038/jid.2013.97 When starting a hair growth journey, it’s easy to feel overwhelmed. A quick search online brings up hundreds of treatments. From prescription medications and over-the-counter solutions to devices, supplements, and medical procedures, all of which claim to regrow hair. With so many options, it’s hard to know which treatments are actually backed by scientific evidence and are worth spending your time and money on.
In reality, there is no single best hair loss treatment that works for everyone. That’s because male pattern hair loss does not look the same for everyone. It can vary widely in pattern, progression, and the underlying biology. Meaning some treatments tend to work better in certain situations than in others. Some are more effective for diffuse thinning, while others are better for localized recession. Some treatments are better for convenience, while others need consistency and long-term application to see results.
In this article, we break down 10 of the best hair loss treatments for men. We have not ranked them from best to worst because each works best in specific circumstances. We look into how each treatment works, the clinical evidence backing it, safety considerations, and who it’s most likely to work best for.
Oral Finasteride
Ideal For:
- Men looking for an FDA-approved treatment
- Men who prefer the convenience of taking one pill per day versus a topical application regimen.
- Men with diffuse thinning rather than localized thinning. With diffuse thinning, applying topical treatments evenly every day can be time-consuming and difficult compared to targeting a small area.
Finasteride is a prescription medication that treats androgenic alopecia (AGA) by inhibiting the enzyme 5ɑ-reductase (5AR), which converts testosterone into dihydrotestosterone (DHT). In people with AGA, DHT is elevated and binds to androgen receptors, which act as landing pads for DHT to have its effect. The binding of DHT to androgen receptors in the scalp leads to follicular miniaturization and leads to hair loss.[1]Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/
Finasteride works by selectively inhibiting type II 5AR. By blocking this enzyme, finasteride reduces the conversion of testosterone to DHT. DHT levels drop, and the hormonal process that causes pattern hair loss is disrupted, helping to maintain existing hair and induce hair regrowth.
Figure 1. Structure of finasteride.[2]Wikipedia, (2009), Finasteride. Available at: https://en.wikipedia.org/wiki/Finasteride#/media/File:Finasteride.svg (Accessed: 24 February 2026) Image in the public domain.
Clinical Evidence for Oral Finasteride
Oral finasteride is one of the most well-studied hair loss treatments and is one of two FDA-approved treatments for AGA. Over the years, there have been many clinical studies that demonstrate oral finasteride is able to slow hair loss and promote new hair growth in many patients. Large randomized placebo-controlled trials consistently show that daily oral finasteride leads to improvements in hair count, hair density, and participant satisfaction.
A notable trial involved 1,500 men with AGA. Of those treated with oral finasteride (1 mg), 48% were rated as improved based on standardized photographs after 12 months, compared to 7% in the placebo-treated group.[3]Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride … Continue reading Also, hair counts in the oral finasteride group increased or stabilized, whereas those in the placebo group experienced progressive hair loss.
The typical dose of oral finasteride for AGA is 1 mg. Increasing the dose to 5 mg does not tend to improve hair regrowth or further suppress scalp DHT in a meaningful way. For example, studies found scalp DHT reductions of 64% with 1 mg and 69% with 5 mg, which is a negligible difference.[4]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., … Continue reading
Lowering the dose to 0.2 mg daily shows promising results. In one 48-week trial, 54% of men who took 0.2 mg oral finasteride showed improvement, compared to 58% on 1 mg, suggesting only a marginal difference. That said, the relative percentage change in terminal hair counts was higher in the 1 mg versus 0.2 mg group.[5]Fertig, R.M., Caresse, G.A., Darwin, E., Gaudi, S. (2017). Sexual Side Effects Of 5-ɑ-Reductase Inhibitors Finasteride And Dutasteride: A Comprehensive Review. Dermatology Online Journal. 23(11). … Continue reading
If you’re looking for more information on how to take oral finasteride, read our article here.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Safe is Oral Finasteride?
Oral finasteride is generally well tolerated. However, as oral finasteride is ingested and reaches the systemic circulation, some people may experience sexual side effects, including reduced libido and erectile difficulties. Clinical trials suggest the risk is around 15% of users; however, these effects usually resolve after stopping medication.[6]Estill, M.C., Ford, A., Omeira, R., Rodman, M. (2023). Finasteride And Dutasteride For The Treatment Of Male Androgenetic Alopecia: A Review Of Efficacy And Reproductive Adverse Effects. Georgetown … Continue reading Oral finasteride may temporarily reduce sperm count or semen volume, but this is typically reversible.[7]Amory, J.K., Wang, C., Swerdloff, R.S., Anawalt, B.D., Matsumoto, A.M., Bremner, W.J., Walker, S.E., Haberer, L.J., Clark, R.V. (2007). The Effect Of 5ɑ-Reductase Inhibition With Dutasteride And … Continue reading Other potential side effects include breast tenderness and mood changes.
As with any medication, if you are considering finasteride, you should discuss potential risk factors and fertility plans with a clinician before beginning treatment and ensure you consult with a clinician if you experience any side effects.
If you’re a member and would like to find out more about oral finasteride, read our ultimate guide here.
Topical Minoxidil
Ideal For:
- Men who are looking for an over-the-counter, FDA-approved treatment for localized thinning.
- Men who are looking for a standard 5% minoxidil formulation or higher-strength, compounded formulations (e.g., 7%) if they have not responded to standard formulations.
- Men who are willing to consider combination therapies with tretinoin or microneedling.
Topical minoxidil at standard 2% and 5% concentrations is an over-the-counter, FDA-approved liquid or foam treatment. It can also be custom-compounded at higher concentrations from 7-15%, but this is not FDA-approved.
Topical minoxidil is a prodrug and is converted to its active form (minoxidil sulfate) by scalp enzymes called sulfotransferases, primarily the SULT1A1 enzyme. The activity of this enzyme can differ between people and in different regions of the scalp.
Minoxidil works by altering the hair cycle by shortening the resting (telogen) phase and promoting earlier entry into the growth (anagen) phase. This increases the proportion of follicles that actively produce hair. Minoxidil can also increase the size of miniaturized hair follicles, resulting in thicker and longer hair shafts.[8]Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: … Continue reading
Minoxidil can also increase local blood flow, activate the Wnt/β-catenin signaling pathway involved in follicular cell proliferation and differentiation, and has some cytoprotective and anti-inflammatory effects, which may increase prostaglandin E2 production.
Figure 2. Structure of minoxidil.[9]Wikipedia, (2015), Minoxidil Structure Svg. Available at: https://en.wikipedia.org/wiki/Minoxidil#/media/File:Minoxidil_structure.svg (Accessed: 20 February 2026) Image in the public domain.
Clinical Evidence for Topical Minoxidil
Topical minoxidil has consistently been shown in many randomized controlled trials to increase hair count, prolong the anagen phase, and improve short-term patient satisfaction, especially at a 5% concentration.[10]Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189
That said, despite strong early efficacy, after one year or longer, response rates tend to drop or plateau, particularly when used without adjunctive therapies like microneedling or anti-androgens.
Multiple studies report rates below 30%, and even lower in five-year follow-up populations, with only about 20-30% of users satisfied with their results.[11]Olsen, E.A., Weiner, M.S., Amara, I.A., DeLong, E.R. (1990). Five-Year Follow-Up Of Men With Androgenetic Alopecia Treated With Topical Minoxidil. Journal Of The American Academy Of Dermatology. … Continue reading In one trial, men using 5% minoxidil twice daily saw a nearly 60% increase in hair “weight” (total mass of regrown/thickened hair) at 6 months. By 96 weeks, gains had diminished to just 25% above the baseline, a significant drop from the initial response.[12]Price, V.H., Menefee, E., Strauss, P.C. (1999). Changes In Hair Weight And Hair Count In Men With Androgenetic Alopecia, After Application Of 5% And 2% Topical Minoxidil, Placebo, Or No Treatment. … Continue reading
In men with AGA, once-daily application of 5% topical minoxidil achieves hair count improvements comparable to twice-daily use of a 2% solution.[13]Olsen, E.A., Dunlap, F.E., Funicella, T., Koperski, J.A., Swinehart, J.M., Tschen, E.H., Trancik, R.J. (2002). A Randomized Clinical Trial Of 5% Topical Minoxidil Versus 2% Topical Minoxidil And … Continue reading Higher concentrations (7%) may offer incremental benefit in some non-responders. With 7% formulations showing modest additional improvement in some studies, but results are mixed, and overall evidence remains limited.[14]Singh, S., Patil, A., Kianfar, N., Waskiel-Burnat, A., Rudnicka, L., Sinclair, R., Goldust, M. (2022). Does Topical Minoxidil At Concentrations Higher Than 5% Provide Additional Clinical Benefit? … Continue reading
Data on 10-15% topical minoxidil are conflicting. Some studies report no meaningful advantage over 5%, while others report small gains in specific subgroups. However, adverse effects tend to increase at these higher concentrations.[15]Vasantha, K.L. (2025). Efficiency And Safety Of 10% Minoxidil In The Treatment Of Alopecia Areata: A Randomised Controlled Trial. Journal Of Population Therapeutics And Clinical Pharmacology. 32(4). … Continue reading,[16]Ghonemy, S., Alarawi, A., Bessar, H. (2021). Efficacy And Safety Of A New 10% Topical Minoxidil Versus 5% Topical Minoxidil And Placebo In The Treatment Of Male Androgenetic Alopecia: A Trichoscopic … Continue reading
Adjunctive strategies aimed at enhancing minoxidil activation and penetration have shown promising results. Anecdotal evidence from our PHH members suggests that prescription concentration of topical minoxidil (7%) in combination with enhancers like tretinoin often improves outcomes.
One landmark study reported a nearly fourfold greater increase in hair count (approximately a 40% gain) compared to minoxidil alone at 12 weeks, with participants experiencing actual visible improvements in density.[17]Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I.A., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating The Efficacy And Safety Of Combined … Continue reading
Topical retinoic acid (such as tretinoin) can also be combined with minoxidil to upregulate SULT1A1 activity. Thereby enhancing the local activation of minoxidil in the hair follicle. Peer-reviewed research indicates that applying topical tretinoin upregulates follicle sulfotransferase enzymes, thereby enhancing the response to minoxidil.[18]Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvan, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin Enhances Minoxidil Response In … Continue reading
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Safe is Topical Minoxidil?
Topical minoxidil is generally safe. As it is applied directly to the scalp, it has a low risk of systemic side effects because it is minimally absorbed into the bloodstream. The most common adverse reactions are local, such as scalp irritation, itching, and flaking. These effects are usually due to the presence of propylene glycol in liquid formulations, which is known to cause scalp irritation.[19]Patel, K., Palmer, A., Nixon, R. (2023). Allergic Contact Dermatitis From Propylene Glycol: A Case Series From Australia. Contact Dermatitis. 89(2). 79-84. Available at: … Continue reading Foam formulations tend to be propylene glycol-free and fast-drying.
More severe side effects, like hypertrichosis (unwanted hair growth outside the scalp) or cardiovascular symptoms, are rare and tend to be linked to misuse or highly sensitive individuals. Topical minoxidil is highly toxic to cats, so it should be handled with care by pet owners.
Learn more about topical minoxidil in our article here.
Topical Finasteride
Ideal For:
- Men with localized AGA who want the DHT-lowering benefits of finasteride but prefer a topical, scalp-focused approach.
- Men who have concerns about systemic side effects with oral finasteride or have previously experienced unwanted effects on oral therapy.
- Men seeking a more potent topical option than minoxidil alone, particularly in DHT-driven thinning at the frontal scalp or crown.
Topical finasteride has emerged as a potential alternative to oral finasteride for AGA. It works by the same mechanism as oral finasteride, blocking type II 5AR to prevent the conversion of testosterone to DHT. But the difference is that topical finasteride is applied directly to the scalp rather than ingested.
By delivering finasteride directly locally to the hair follicles, the aim is to reduce scalp DHT whilst limiting systemic absorption. The goal of this approach is to maintain the benefits for hair regrowth while lowering the risk of whole-body side effects.
You can find more information on how topical finasteride works in our article here.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Topical Finasteride
Clinical studies suggest that topical finasteride can improve hair parameters in men with AGA. One study reported that 1% topical finasteride showed comparable efficacy to 1 mg oral finasteride in terms of hair thickness, hair counts, and the size of the bald area.[20]Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of … Continue reading
In a randomized controlled trial carried out in men with AGA over 24 weeks, they found that topical finasteride (0.25%) significantly improved target area hair count compared to placebo.[21]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2021). Efficacy And Safety Of Topical Finasteride Spray … Continue reading
How Safe is Topical Finasteride?
Topical finasteride has a different safety profile than oral finasteride. Studies show that while some systemic absorption still occurs, circulating finasteride and DHT changes are substantially smaller than with oral finasteride.[22]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2022). Efficacy And Safety Of Topical Finasteride Spray … Continue reading
But systemic effects are still possible. A 2025 FDA pharmacovigilance review of adverse event reports submitted between 2019 and 2024 identified 32 cases involving topical finasteride formulations. Most of these cases involved the use of compounded products. Reported symptoms included erectile dysfunction, anxiety, brain fog, depression, fatigue, insomnia, decreased libido, and testicular pain.[23]US Food and Drug Administration. (2025). FDA alerts health care providers, compounders and consumers of potential risks associated with compounded topical finasteride products. FDA. Available at: … Continue reading
That said, the most common side effects associated with topical finasteride are local skin reactions. A 2018 systematic review noted reports of erythema, contact dermatitis, and scalp irritation in several studies; but serious cutaneous adverse events were absent.[24]Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading In many cases, scalp irritation is thought to be related to the formulation itself, particularly ingredients like propylene glycol.
If you’re a member, you can find out more about topical finasteride in our ultimate guide here.
Oral Dutasteride
Ideal For:
- Men who are willing to consider a stronger systemic therapy with more extensive or diffuse AGA.
- Men with progressive thinning across the scalp rather than isolated areas.
- Men who are looking for the next step if they have not achieved their desired results, or whose results have plateaued with finasteride.
So we know that DHT drives follicle miniaturization, and testosterone is converted to DHT by 5AR. But there are two types of 5AR: type I and type II.
This is where dutasteride and finasteride differ. Finasteride only blocks type II 5AR, whereas dutasteride can block both type I and type II 5AR. Because dutasteride blocks both types of 5AR, it suppresses DHT more strongly than finasteride. Oral dutasteride is FDA-approved for benign prostatic hyperplasia, but is commonly prescribed off-label for hair loss.
Figure 3. Structure of dutasteride.[25]Wikipedia, (2009), Dutasteride. Available at: https://en.wikipedia.org/wiki/Dutasteride#/media/File:Dutasteride.svg (Accessed: 24 February 2026) Image in the public domain.
Clinical Evidence for Oral Dutasteride
Across multiple studies, dutasteride has been shown to consistently outperform finasteride on hair parameters. In one notable study carried out in men with AGA over 24 weeks, 0.5 mg dutasteride significantly increased total hair count and decreased thin hair count per cm2 compared to 1 mg finasteride.[26]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading
Notably, another study carried out in men with AGA over 24 weeks, showed that 0.5 mg dutasteride significantly increased terminal hair count and hair width compared to 1 mg finasteride.[27]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading
Additionally, across studies, investigators were more likely to rate dutasteride users as “improved” or “markedly improved” compared to those receiving finasteride.[28]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Safe is Oral Dutasteride?
Oral dutasteride is generally well-tolerated by many users. But some people may experience the following side effects:
- Sexual side effects: Reduced libido, erectile dysfunction, or ejaculatory disorders (reduced volume or delayed ejaculation) may occur. These effects are often dependent on the dose and usually improve after stopping treatment.[29]Choi, G.S., Sim, W.Y., Kang, H., Huh, C.H., Lee, Y.W., Shantakumar, S., Ho, Y.F., Oh, E.J., Duh, M.S., Cheng, W.Y., Bobbili, P., Thompson-Leduc, P., Ong, G. (2022). Long-Term Effectiveness And Safety … Continue reading
- Mood changes: Low mood or depressive symptoms, increased anxiety, or lack of motivation. These are uncommon, but any mood changes should be taken seriously if they do occur.[30]Hirshburg, J.M., Kelsey, P.A., Therrien, C.A., Gavino, A.C., Reichenberg, J.S. (2016). Adverse Effects And Safety Of 5-Alpha Reductase Inhibitors (Finasteride, Dutasteride): A Systematic Review. … Continue reading
- Breast changes: Hormonal shifts may lead to breast tenderness or enlargement (gynecomastia), which warrants medical review.[31]Marihart, S., Harik, M., Djavan, B. (2005). Dutasteride: A Review Of Current Data On A Novel Dual Inhibitor Of 5alpha Reductase. Reviews In Urology. 7(4). 203-210
- Prostate-specific antigen (PSA): Dutasteride may lower PSA, which could mask early signs of prostate cancer.[32]Marihart, S., Harik, M., Djavan, B. (2005). Dutasteride: A Review Of Current Data On A Novel Dual Inhibitor Of 5alpha Reductase. Reviews In Urology. 7(4). 203-210
Check out our article here on oral dutasteride to find out more about side effects, dosing, and how it compares to finasteride.
Topical Dutasteride
Ideal For:
- Men with AGA and localized thinning (e.g., the crown or frontal scalp).
- Men who have experienced side effects with oral dutasteride or are concerned about systemic effects.
- Men who are looking for a potentially stronger topical DHT-targeting option than finasteride.
- Men who are comfortable using a newer treatment that has relatively limited clinical evidence.
Topical dutasteride is another emerging therapy that works by the same mechanism as oral dutasteride, inhibiting both type I and type II 5AR to prevent the conversion of testosterone to DHT.
The difference is that topical dutasteride is applied directly to the scalp, with the goal of limiting systemic side effects. Topical dutasteride is not FDA-approved and is usually obtained through compounding pharmacies.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Topical Dutasteride
Clinical evidence for topical dutasteride is still limited compared with oral therapies. But, at the end of 2025, the first RCT involving 135 men with AGA comparing 1 mg oral finasteride to (0.01-0.05%) topical dutasteride over 24 weeks was published.[33]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading
In the study, several hair parameters appeared to improve more in the topical dutasteride (0.05%) group than in those receiving oral finasteride (1 mg). The authors reported an average increase of 75 hairs per cm² with topical dutasteride compared to 41 hairs per cm² with oral finasteride.
Additionally, 69% of participants using topical dutasteride were rated as “improved” on global photographic assessment, compared with 21% in the oral finasteride group.
But it’s important to note that we are a little skeptical of the results from this study. This is because the baseline hair counts are suspicious, and the measurement circle clearly moves in location, size, and shape.
Figure 4. Representative hair growth images. Adapted from Figure 2.[34]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading Image used under Creative Commons License
If you’d like to learn more about our take on this study, read our article here.
How Safe is Topical Dutasteride?
In this same study, systemic hormone changes with topical dutasteride were minimal. Plasma dutasteride levels were mostly near the quantification limit, but some samples spiked to 2,555 pg/mL, and dutasteride remained detectable at day 168 while participants were still receiving treatment. This suggests that average exposure was low, but absorption may be unpredictable in some people.
Because topical dutasteride is applied directly to the scalp, it may have fewer side effects than oral dutasteride. The most common side effects are local scalp reactions, including redness, itching, flaking, or contact dermatitis.
This is usually related to the carrier solution rather than the dutasteride itself. Switching carriers or adjusting how often it is applied often helps these symptoms.[35]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading
If you’re interested in finding out more about how topical dutasteride works, have a read of our article here.
Oral Minoxidil
Ideal For:
- Men who have experienced side effects from finasteride or dutasteride.
- Men who prefer the convenience of a once-daily pill over topical application.
- Men who have struggled with adherence to topical treatments due to cosmetic residue, scalp irritation, or lifestyle factors.
- Men who have not responded well to topical minoxidil, including those with poor scalp absorption or SULT1A1 activity.
Oral minoxidil is a prescription medication that was originally used to treat high blood pressure. At lower doses (0.25-5 mg daily), it’s prescribed off-label to treat hair loss, especially in people who don’t respond well to topical minoxidil or struggle with applying topical treatments consistently.
Oral minoxidil supports regrowth by improving blood flow around follicles, shifting more hairs into the growth (anagen) phase, and activating follicular growth pathways.
Some people prefer oral minoxidil over topical due to the key differences in how each one is activated and delivered to the hair follicles. Topical minoxidil is a pro-drug that needs conversion to its active form by SULT1A1 enzymes located in scalp hair follicles.[36]Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A … Continue reading
The activity of this enzyme varies significantly between people. Also, topical minoxidil faces a penetration problem where only about 1.4% of the drug applied is actually absorbed into the scalp skin under normal conditions.[37]Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil And Its Use In Hair Disorders: A Review. Drug Design, Development And Therapy. 13. 2777-2786. Available at: … Continue reading
Oral minoxidil helps to bypass these limitations. Oral minoxidil is absorbed through the gastrointestinal tract and converted to its active form in the liver, where sulfotransferase activity is abundant.
This ensures that nearly all minoxidil that is ingested is activated and delivered systemically to the hair follicles throughout the scalp and body, regardless of individual differences in scalp enzyme activity. As a result, oral minoxidil can be effective even for people who have not responded to topical minoxidil due to low scalp sulfotransferase activity or poor drug penetration.[38]Beach, R.A. (2018). Case Series Of Oral Minoxidil For Androgenetic And Traction Alopecia: Tolerability & The Five C’s Of Oral Therapy. Dermatologic Therapy. 31(6). E12707. Available at: … Continue reading
Interested in Oral Minoxidil?
Low-dose oral minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Oral Minoxidil
One study in men with AGA reported a 100% response rate with 5 mg daily oral minoxidil, alongside an average 19% increase in hair count at 24 weeks.[39]Lueangarun, S., Panchaprateep, R., Tempark, T., Noppakun, N. (2015). Efficacy And Safety Of Oral Minoxidil 5 Mg Daily During 24-Week Treatment In Male Androgenetic Alopecia. Journal Of The American … Continue reading Additionally, a retrospective study showed a 90% response rate at 6-12 months for men taking 2.5-5 mg of oral minoxidil daily. Notably, around 60% of these men had tried other treatments for AGA, but had previously quit due to side effects or lack of efficacy.[40]Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral … Continue reading
Lower doses of oral minoxidil may still be effective in stabilizing progression. In one study, 0.25 mg minoxidil achieved a response rate of 60% in men with AGA. That’s lower than what is typically seen at 2.5-5 mg concentrations, but it is favourable nevertheless.[41]Pirmez, R., Salas-Callo, C.I. (2020). Very-Low-Dose Oral Minoxidil In Male Androgenetic Alopecia: A Study With Quantitative Trichoscopic Documentation. Journal Of The American Academy Of Dermatology. … Continue reading
How Safe is Oral Minoxidil?
At low doses, oral minoxidil is generally well tolerated, but side effects are more common than with topical minoxidil because the drug circulates systemically.[42]Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair … Continue reading
The most common side effects include hypertrichosis, mild fluid retention or ankle swelling, and lightheadedness, particularly when starting or increasing the dose.
Less commonly, oral minoxidil can affect heart rate or cardiac function. Rare but serious events (e.g., pericardial effusion) have been reported, particularly in people with underlying cardiovascular or renal issues. Most side effects are dependent on the dose and often improve with dose reduction or discontinuation.
If you’re considering starting oral minoxidil treatment, start with a lower dose and only increase gradually if it is needed. Splitting the dose between morning and evening can improve tolerability for some. If results plateau, oral minoxidil is often combined with other therapies such as microneedling or topical agents rather than increasing the dose alone.
Oral minoxidil should be used under medical supervision, especially in anyone with cardiovascular, renal, or hepatic concerns.
If you are a member and would like to learn more about oral minoxidil, read our ultimate guide here.
Low-Level Laser Therapy (LLLT)
Ideal For:
- Men with early AGA who are looking to avoid pharmaceutical treatments.
- Men who are willing to be consistent with long-term device use.
- Men willing to pay the costs for device use, either at home or through visits to a clinic.
- Men who are willing to try more experimental methods.
- Men who are willing to try combination treatments, as LLLT will most likely have minimal effects as a standalone solution.
LLLT is therapeutic exposure to red or near-infrared radiation. For hair loss, this involves placing the scalp under infrared-emitting laser diodes. A typical laser therapy session lasts from 20 to 60 minutes, and you can complete them at a doctor’s office, with a take-home laser helmet, or even with a laser brush.
Laser diodes for hair loss emit red light at wavelengths of 630-670 nanometers. The sun emits wavelengths from 250 to 2,500 nanometers. LLLT is essentially exposure to one part of the sun’s spectrum for a controlled period of time. It turns out that some wavelengths show benefits to human physiology, from wound healing to inflammation resolution and even hair recovery.
Currently, there is still speculation about how LLLT actually works for hair regrowth. Potential explanations include:
- The laser’s heat activates heat shock proteins.[43]Wikramanayake, T.C., Rodriguez, R., Choudhary, S., Mauro, L.M., Nouri, K., Schachner, L.A., Jimenez, J.J. (2012). Effects of the Lexington LaserComb on Hair Regrowth in the C3H/HeJ Mouse Model of … Continue reading
- The laser’s light increases tissue oxygenation.[44]Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts and Bolts of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available … Continue reading
- The laser’s light increases blood flow.[45]Makihara, E., Masumi, S. (2008). Blood Flow Changes of a Superficial Temporal Artery Before and After Low-Level Laser Irradiation Applied to the Temporomandibular Joint Area. Nihon Hotetsu Shika … Continue reading
- The laser’s light generates acute tissue inflammation.[46]Bouzari, N., Firooz, A.R. (2006). Lasers May Induce Terminal Hair Growth. Dermatologic Surgery. 32(3). 460. Available at: https://doi.org/10.1111/j.1524-4725.2006.32092.x
- The laser’s light can inhibit 5AR.
Clinical Evidence for Low-Level Laser Therapy
Clinical trials of LLLT devices for AGA generally report modest improvements in hair density versus placebo (sham) devices. A systematic review and meta-analysis of seven randomized controlled trials using FDA-approved home-use LLLT devices found significantly greater increases in hair density in treated participants compared with sham controls in both men and women, particularly with helmet and comb-type devices.[47]Lueangarun, S., Visutjindaporn, P., Parcharoen, Y., Jamparuang, P., Tempark, T. (2021). A Systematic Review and Meta-Analysis of Randomized Controlled Trials of United States Food and Drug … Continue reading
In a recently published study, they investigated the effects of using a helmet-type LLLT device (646-675 nm) for 20 mins, 3 times per week, on AGA in both men and women (51 men, 17 women) over 48 weeks.
They found that hair density significantly increased from baseline from 99.2 to 124.2 hairs/cm2, and mean hair shaft thickness increased from 65.1 to 74.9 μm. Also, 59% of participants were rated as improved based on global photographs, and over 85% expressed satisfaction with treatment.[48]Shin, J.W., Paik, K., Na, J.I., Lew, B.L., Huh, C.H. (2026). Long-Term Efficacy and Safety of Low-Level Laser Therapy for Androgenetic Alopecia: A 12-Month Prospective Trial. Dermatologic Therapy. … Continue reading
Figure 5. Investigator-assessed clinical improvement over time after use of an LLLT device. Adapted from Figure 3a.[49]Shin, J.W., Paik, K., Na, J.I., Lew, B.L., Huh, C.H. (2026). Long-Term Efficacy and Safety of Low-Level Laser Therapy for Androgenetic Alopecia: A 12-Month Prospective Trial. Dermatologic Therapy. … Continue reading Used under Creative Commons license.
However, based on anecdotal evidence from our members, we have not seen a single case where LLLT has worked, so take this information with a pinch of salt.
There is also a broader methodological issue with LLLT device trials that is worth noting. Human hair growth shows seasonal variation, with increased telogen shedding often happening in late summer and early autumn in the Northern Hemisphere.
Because many LLLT devices emit wavelengths that overlap with natural sunlight exposure, trial timing across this seasonal window could theoretically amplify any differences seen between LLLT devices and sham devices if light exposure modifies the dynamics of seasonal shedding.
This does not invalidate the clinical evidence, but it highlights how environmental biology can also interact with LLLT trials in ways that may exaggerate benefits.
If you’re a member and would like more info on this topic, watch our video here.
How Safe is LLLT?
LLLT is generally safe and well-tolerated for most people. Most effects are mild and localized to the scalp, including scalp warmth, redness, or itching during or after use.
Find out more about LLLT in our article here.
Microneedling
Ideal For:
- Men who are willing to be consistent with use.
- Men who are looking to boost the impact of topical treatment like minoxidil, especially if they’ve seen plateauing results or suspect poor topical response.
Microneedling, also known as percutaneous wound induction, is the act of deliberately wounding skin tissues for therapeutic benefit. This is generally done with hundreds of tiny, medical-grade needles ranging in length from 0.1 to 5.0mm.
There are a variety of microneedling devices, from hand-held rollers to automated pens to stamps. But the ones people are most familiar with (in the hair loss world) are dermarollers. When rolled against the skin, they puncture its top layers to evoke acute inflammation. And yes, being rolled with essentially a tiny lint roller covered in spikes does hurt.
There are multiple possible explanations as to how microneedling may improve AGA:
- Micro-wounding may increase proteins and growth factors linked to hair growth.
- Micro-wounding might downregulate proteins linked to scarring.
- Micro-wounding might promote angiogenesis (new blood vessel formation).
- Microneedling may improve the absorption/efficacy of other hair loss treatments.
Clinical Evidence for Microneedling
There are many before-and-after photos online from microneedling regimens. If you’d like to see these photos, a user at HairLossTalk has done a great job accumulating them, which you can view here.[50]HairLossTalk, (2019), Microneedling Photo Results Summary. Available at: https://www.hairlosstalk.com/interact/threads/microneedling-photo-results-summary.121072/ (Accessed: 24 February 2026)
But the more useful question is what the clinical literature shows. Overall, microneedling appears to improve outcomes when combined with other therapies, such as topical minoxidil, finasteride, or platelet-rich plasma (PRP).
A systematic review and meta-analysis of 13 randomized controlled trials showed that microneedling in combination with other treatments resulted in greater improvements in hair density and diameter compared to treatments used alone.[51]Pei, D., Zeng, L., Huang, X., Wang, B., Liu, L., Zhang, G. (2024). Efficacy and Safety of Combined Microneedling Therapy for Androgenic Alopecia: A Systematic Review and Meta-Analysis of Randomized … Continue reading
Notably, over 12 weeks, once-weekly microneedling plus twice-daily minoxidil increased hair counts by 4X compared with minoxidil alone.[52]Dhurat, R., Sukesh, M., Avhad, G., Dandale, A., Pal, A., Pund, P. (2013). A Randomized Evaluator Blinded Study of Effect of Microneedling in Androgenetic Alopecia: A Pilot Study. International … Continue reading Also over 6 months, once-weekly microneedling alongside daily finasteride and 5% minoxidil resulted in a +2 or +3 improvement in hair loss gradient for non-responders to both finasteride and minoxidil.[53]Dhurat, R., Mathapati, S. (2015). Response to Microneedling Treatment in Men with Androgenetic Alopecia Who Failed to Respond to Conventional Therapy. Indian Journal of Dermatology. 60(3). 260-263. … Continue reading
There is also evidence that microneedling may have a standalone benefit. In one 24-week study, microneedling carried out every 14 days produced greater increases in hair density than topical minoxidil alone (23.4 vs 18.8 hairs/cm² from baseline).[54]Bao, L., Gong, L., Guo, M., Liu, T., Shi, A., Zong, H. (2020). Randomized Trial of Electrodynamic Microneedle Combined with 5% Minoxidil Topical Solution for the Treatment of Chinese Male … Continue reading
Ultimately, microneedling tends to work best as an adjunct (especially with minoxidil), but it may still offer meaningful benefit as a standalone option for some people.
The choice of needle length also matters. In general, we currently recommend sticking with 0.6-0.8mm needle lengths for automated pens, and 1.0-1.5mm needle lengths for rollers, needling once weekly, or once every two weeks.
To find out more about the best microneedling length, read our article here.
How Safe is Microneedling?
In short-term studies, microneedling is generally safe. The most common side effects are mild and localized, including redness, swelling, itching, pinpoint bleeding, and discomfort during treatment.
Some people also notice temporary flaking in the days after treatment. Scarring is rare when users use a sensible needle depth and avoid overdoing it. But anyone prone to keloid scarring should be cautious.
You can find more information on microneedling and scarring in our article here.
The risk of infection is low with basic hygiene (e.g., clean scalp, clean hands, and proper device sterilization). As a general safety rule, sticking to needle lengths of 2.0 mm or less and avoiding excessive session frequency reduces risk.
If you’re a member, you can learn more about microneedling in our ultimate guide here.
Ketoconazole
Ideal For:
- Men with AGA who also have dandruff, seborrheic dermatitis, oily scalp, or scalp inflammation, as these factors can worsen follicle miniaturization.
- Men with AGA alone, or AGA plus seborrheic dermatitis or telogen effluvium.
- Men with hair shedding disorders where microbial overgrowth or scalp inflammation contributes to shedding may particularly benefit from ketoconazole due to its antifungal and anti-inflammatory effects.
You might have heard about ketoconazole under the brand name Nizoral®, and you might know about it as a shampoo. Ketoconazole is an antifungal medication used to help improve fungal-related conditions such as dandruff, fungal infections, certain hormone-linked diseases, and even hair loss from fungal and non-fungal causes.
Figure 7. Structure of ketoconazole. Adapted from Wikimedia.[55]Wikimedia Commons, (2025), Ketoconazole Chemical Structure (SVG). Available at: https://commons.wikimedia.org/wiki/Category:Ketoconazole#/media/File:(2S,4R)-Ketoconazol.svg (Accessed: 24 February … Continue reading Image in the public domain.
Ketoconazole may improve telogen effluvium by killing scalp pathogens, thereby normalizing excessive hair shedding triggered by microorganism-mediated inflammation. It may also prolong the anagen phase of the hair cycle.
Ketoconazole may improve AGA via three key mechanisms:
- Killing pathogens in the scalp, therefore normalizing excessive hair shedding triggered by microorganism-mediated inflammation.
- Reducing scalp DHT levels by inhibiting 5-alpha reductase (a key enzyme that converts testosterone to DHT) and blocking androgen receptors (“landing pads” for DHT to exert its effects).
- Prolonging the anagen phase of the hair cycle.
You can buy 1% ketoconazole shampoo over the counter, typically at around $16 for 200 mL. Whereas, 2% ketoconazole shampoo usually requires a prescription.
Figure 8. Image of Nizoral shampoo, containing 1% ketoconazole. Available at Walmart.
Clinical Evidence for Ketoconazole
Clinical studies on ketoconazole shampoo, especially 2% strength, provide evidence that ketoconazole can improve hair parameters in men with AGA, including hair density, shaft diameter, and the proportion of hairs in the anagen phase.
In one study involving men with AGA comparing 2% ketoconazole shampoo with a non-medicated shampoo over 21 months, ketoconazole showed progressive increases in a composite hair growth index (anagen percentage × shaft diameter). Those using standard shampoo experienced a gradual decline.[56]Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: … Continue reading
Also, a pilot study showed that 2% ketoconazole may produce hair shaft diameter improvements similar to 2% minoxidil, with reductions in sebaceous gland size, which was not seen in the minoxidil-only group.[57]Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: … Continue reading
Evidence for 1% ketoconazole is weaker but still supportive. In men with AGA associated with dandruff or telogen effluvium, 1% ketoconazole used 2 to 3 times per week reduced hair shedding and increased anagen hair proportion and shaft diameter over 6 months.[58]Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine … Continue reading
Ketoconazole also tends to work well when used in combination with other treatments. In one study, finasteride plus ketoconazole demonstrated improvements in self- and physician-assessments.[59]Khandpur, S., Suman, M., Reddy, B.S. (2002). Comparative Efficacy Of Various Treatment Regimens For Androgenetic Alopecia In Men. J Dermatol. 29(8). 489-98. Available at: … Continue reading
Based on the available clinical evidence, there is stronger support for the use of 2% ketoconazole shampoo for hair growth than for 1%. But, as there are currently no direct head-to-head studies comparing the effectiveness of 1% ketoconazole shampoo versus 2% for hair regrowth, this is purely speculative.
How Safe is Ketoconazole?
Ketoconazole shampoo is generally safe and well-tolerated when used as directed. Because it is applied briefly to the scalp and then rinsed off, systemic absorption is minimal, and side effects are usually limited to mild, local scalp irritation or dryness.
Leave-on topical ketoconazole is available in some countries, but this increases the risk of systemic exposure. These non-shampoo formulations have not been studied for hair loss in humans. So even though in theory topical ketoconazole may seem more effective, it’s sensible to stick with ketoconazole shampoo, which has science-backed evidence.
Oral ketoconazole has significant systemic risks, but these concerns do not apply to shampoo use due to the much lower exposure. Overall, 2% ketoconazole shampoo has a favourable safety profile when used a few times per week.
If you’re a member, you can find out more information on ketoconazole in our ultimate guide here.
Saw Palmetto
Ideal For:
- Men with early to mid-stage AGA.
- Men who want to avoid pharmaceutical options.
- Men who are willing to try combinational treatments.
Saw palmetto (Serenoa repens) is a palm plant native to the Southeastern United States. It grows up to 10 feet tall and can live over 700 years. It’s a botanical extract used for hair loss because it can reduce 5AR activity and may lower DHT signalling.
Saw palmetto is different from finasteride and dutasteride as it is not a standardized pharmaceutical. So its strength can vary across products depending on how it is harvested and extracted.
Saw palmetto is available in both oral and topical formulations (serums/lotions/shampoos).
All-Natural Hair Topical
The top natural ingredients for hair growth, all in one serum.
Take the next step in your hair growth journey with a world-class natural serum. Ingredients, doses, & concentrations built by science.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Clinical Evidence for Saw Palmetto
Overall, the clinical evidence for saw palmetto is mixed and tends to be of lower quality than the evidence that is available for pharmaceutical treatments like finasteride, minoxidil, or dutasteride.
Nevertheless, there is some clinical evidence to back this treatment. In one 16-week randomized controlled trial involving adults with AGA, both oral and topical standardized saw palmetto oil (named VISPO™) significantly reduced hair shedding compared to placebo and produced small increases in hair density (around 5-7%). Oral saw palmetto also reduced serum DHT, whereas topical did not.[60]Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral And Topical Administration Of A Standardized Saw Palmetto Oil Reduces Hair Fall And Improves The … Continue reading
Also, in one study, oral saw palmetto (320 mg/day) was compared to finasteride (1 mg/day) in 100 men with mild to moderate AGA over 24 months. Hair regrowth assessed by global photographic assessment was reported 38% of the saw palmetto group compared to 68% in the finasteride group.[61]Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparitive Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A … Continue reading
What people often miss from this study is that of those saw palmetto users, 45/50 men saw a stop in hair loss. In fact, that number was the same in the finasteride group (45/50). In other words, saw palmetto stopped the progression of hair loss in 90% of men. And even better, the saw palmetto group also reported zero side effects.[62]Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparitive Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A … Continue reading
In terms of topical saw palmetto, this formulation tends to look more effective for hair regrowth when combined with other hair growth-supporting ingredients.[63]Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., Bunnag, T., Pimonrat, T., Muangdang, N., Pichai, P. (2016). Treatment of Male Androgenetic Alopecia with Topical Products Containing … Continue reading But it’s difficult to see how much contribution saw palmetto made because it has been studied in multi-ingredient blends.
There is one 24-week pilot study published on topical saw palmetto, but they reported a very large increase in terminal-hair count of 74.1%, and the study lacked a placebo control, so we recommend taking these results with a pinch of salt.[64]Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. … Continue reading
Figure 9: Images showing changes in scalp hair following 16-week treatment of VISPO™ oil or a placebo oil. Adapted from Figure 4.[65]Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the … Continue reading Image obtained in line with the Creative Commons License.
How Safe is Saw Palmetto?
Saw palmetto is generally safe and well-tolerated. The most common adverse effects of oral saw palmetto include stomach upset, headache, or fatigue. Sexual side effects are uncommon compared to pharmaceutical 5AR inhibitors (e.g., finasteride and dutasteride).
For topical saw palmetto, most of the effects are localized to the scalp, including inflammation, itching, dryness, and flaking.
Because saw palmetto supplements are not tightly regulated, the quality and strength of the product can vary across brands, which can affect the safety and tolerability.
Find out more about saw palmetto in our article here.
General Guidelines for Use
When starting hair loss treatment, it can feel daunting and difficult to know where to begin. While each treatment has its own considerations around dosing, frequency, and application, some general tips and recommendations could help when starting any new hair loss treatment:
- Speak to a clinician first: This is especially important for any prescription treatments or if you have any underlying health concerns, risk factors, or fertility plans.
- Give treatments enough time: Most evidence-based hair loss treatments take at least 3 to 6 months before meaningful changes are visible.
- Track your progress: Take consistent photographs in the same lighting and at the same angle. This makes it much easier to assess whether something is working or isn’t.
- Monitor any side effects: If you do notice any unwanted effects, it’s important to discuss any adjustments or alternatives with a licensed clinician rather than pushing through discomfort.
- Prioritize consistency: The best treatment is one you can use consistently over time. If you’re not able to stick with it, even a strong medication won’t work as well as it should.
- Adjust when needed: Hair loss changes over time, and treatment plans must be able to evolve with it.
Final Thoughts
Ultimately, there is no single best hair loss treatment for men. The right choice depends on your specific pattern of hair loss, your goals, and your preferences. Whether that’s a prescription medication, an over-the-counter option, a natural approach like saw palmetto, or a technique like microneedling.
What matters is choosing a hair loss treatment you can tolerate and stick with consistently over time. Hair loss treatment is rarely about finding the strongest option. It’s more about finding one that you can realistically fit into your routine and that you are comfortable maintaining.
With the right expectations and a tailored plan, many men are able to stabilize their hair loss and maintain hair growth improvements over time.
References[+]
References ↑1 Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ ↑2 Wikipedia, (2009), Finasteride. Available at: https://en.wikipedia.org/wiki/Finasteride#/media/File:Finasteride.svg (Accessed: 24 February 2026) ↑3 Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride In The Treatment Of Men With Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 39(4 Pt 1). 578-589. Available at: https://doi.org/10.1016/s0190-9622(98)70007-6 ↑4 Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., Carrington, P., Gencheff, C., Cole, G.W., Pariser, D.M., Epstein, E.S., Tanaka, W., Dallob, A., Vandormael, K., Geissler, L., Waldstreicher, J. (1999). The Effects Of Finasteride On Scalp Skin And Serum Androgen Levels In Men With Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 41(4). 550-554 ↑5 Fertig, R.M., Caresse, G.A., Darwin, E., Gaudi, S. (2017). Sexual Side Effects Of 5-ɑ-Reductase Inhibitors Finasteride And Dutasteride: A Comprehensive Review. Dermatology Online Journal. 23(11). 1-19. Available at: https://doi.org/10.5070/D32311037240 ↑6 Estill, M.C., Ford, A., Omeira, R., Rodman, M. (2023). Finasteride And Dutasteride For The Treatment Of Male Androgenetic Alopecia: A Review Of Efficacy And Reproductive Adverse Effects. Georgetown Medical Review. 7(1). Available at: https://doi.org/10.52504/001c.88531 ↑7 Amory, J.K., Wang, C., Swerdloff, R.S., Anawalt, B.D., Matsumoto, A.M., Bremner, W.J., Walker, S.E., Haberer, L.J., Clark, R.V. (2007). The Effect Of 5ɑ-Reductase Inhibition With Dutasteride And Finasteride On Semen Parameters And Serum Hormones In Healthy Men. The Journal Of Clinical Endocrinology & Metabolism. 92(5). 1659-1665. Available at: https://doi.org/10.1210/jc.2006-2203 ↑8 Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑9 Wikipedia, (2015), Minoxidil Structure Svg. Available at: https://en.wikipedia.org/wiki/Minoxidil#/media/File:Minoxidil_structure.svg (Accessed: 20 February 2026) ↑10 Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189 ↑11 Olsen, E.A., Weiner, M.S., Amara, I.A., DeLong, E.R. (1990). Five-Year Follow-Up Of Men With Androgenetic Alopecia Treated With Topical Minoxidil. Journal Of The American Academy Of Dermatology. 22(4). 643-646. Available at: https://doi.org/10.1016/0190-9622(90)70089-z ↑12 Price, V.H., Menefee, E., Strauss, P.C. (1999). Changes In Hair Weight And Hair Count In Men With Androgenetic Alopecia, After Application Of 5% And 2% Topical Minoxidil, Placebo, Or No Treatment. Journal Of The American Academy Of Dermatology. 41(5 Pt 1). 717-721. Available at: https://doi.org/10.1016/s0190-9622(99)70006-x ↑13 Olsen, E.A., Dunlap, F.E., Funicella, T., Koperski, J.A., Swinehart, J.M., Tschen, E.H., Trancik, R.J. (2002). A Randomized Clinical Trial Of 5% Topical Minoxidil Versus 2% Topical Minoxidil And Placebo In The Treatment Of Androgenetic Alopecia In Men. Journal Of The American Academy Of Dermatology. 47(3). 377-385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑14 Singh, S., Patil, A., Kianfar, N., Waskiel-Burnat, A., Rudnicka, L., Sinclair, R., Goldust, M. (2022). Does Topical Minoxidil At Concentrations Higher Than 5% Provide Additional Clinical Benefit? Clinical And Experimental Dermatology. 47(11). 1951-1955. Available at: https://doi.org/10.1111/ced.15338 ↑15 Vasantha, K.L. (2025). Efficiency And Safety Of 10% Minoxidil In The Treatment Of Alopecia Areata: A Randomised Controlled Trial. Journal Of Population Therapeutics And Clinical Pharmacology. 32(4). 724-730. Available at: https://doi.org/10.53555/56ggpq88 ↑16 Ghonemy, S., Alarawi, A., Bessar, H. (2021). Efficacy And Safety Of A New 10% Topical Minoxidil Versus 5% Topical Minoxidil And Placebo In The Treatment Of Male Androgenetic Alopecia: A Trichoscopic Evaluation. Journal Of Dermatological Treatment. 32(2). 236-241. Available at: https://doi.org/10.1080/09546634.2019.1654070 ↑17 Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I.A., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating The Efficacy And Safety Of Combined Microneedling Therapy Versus Topical Minoxidil In Androgenetic Alopecia: A Systematic Review And Meta-Analysis. Archives Of Dermatological Research. 317(1). 528. Available at: https://doi.org/10.1007/s00403-025-04032-1 ↑18 Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvan, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin Enhances Minoxidil Response In Androgenetic Alopecia Patients By Upregulating Follicular Sulfotransferase Enzymes. Dermatologic Therapy. 32(3). E12915. Available at: https://doi.org/10.1111/dth.12915 ↑19 Patel, K., Palmer, A., Nixon, R. (2023). Allergic Contact Dermatitis From Propylene Glycol: A Case Series From Australia. Contact Dermatitis. 89(2). 79-84. Available at: https://doi.org/10.1111/cod.14325 ↑20 Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of Dermatology, Venereology and Leprology.* 75. 47. Available at: https://doi.org/10.4103/0378-6323.45220 ↑21 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2021). Efficacy And Safety Of Topical Finasteride Spray Solution For Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical Trial. Journal Of The European Academy Of Dermatology And Venereology. 36(2). 286–294. Available at: https://doi.org/10.1111/jdv.17738 ↑22 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E. (2022). Efficacy And Safety Of Topical Finasteride Spray Solution For Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical Trial. Journal Of The European Academy Of Dermatology And Venereology. 36(2). 286–294. Available at: https://doi.org/10.1111/jdv.17738 ↑23 US Food and Drug Administration. (2025). FDA alerts health care providers, compounders and consumers of potential risks associated with compounded topical finasteride products. FDA. Available at: https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-consumers-potential-risks-associated-compounded (Accessed: December 2025) ↑24 Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in Dermatology. 17(4). 457-463. Available at: PMID: 29601622 ↑25 Wikipedia, (2009), Dutasteride. Available at: https://en.wikipedia.org/wiki/Dutasteride#/media/File:Dutasteride.svg (Accessed: 24 February 2026) ↑26 Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. *Indian Journal of Dermatology, Venereology and Leprology.* 83. 47. Available at: https://doi.org/10.4103/0378-6323.188652 ↑27 Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 70(3). 489–498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑28 Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male androgenic alopecia in South Korea: a multicentre chart review study. *Annals of Dermatology.* 34(5). 349. Available at: https://doi.org/10.5021/ad.22.027 ↑29 Choi, G.S., Sim, W.Y., Kang, H., Huh, C.H., Lee, Y.W., Shantakumar, S., Ho, Y.F., Oh, E.J., Duh, M.S., Cheng, W.Y., Bobbili, P., Thompson-Leduc, P., Ong, G. (2022). Long-Term Effectiveness And Safety Of Dutasteride Versus Finasteride In Patients With Male Androgenic Alopecia In South Korea: A Multicentre Chart Review Study. Annals Of Dermatology. 34(5). 349-359. Available at: https://doi.org/10.5021/ad.22.027 ↑30 Hirshburg, J.M., Kelsey, P.A., Therrien, C.A., Gavino, A.C., Reichenberg, J.S. (2016). Adverse Effects And Safety Of 5-Alpha Reductase Inhibitors (Finasteride, Dutasteride): A Systematic Review. Journal Of Clinical And Aesthetic Dermatology. 9(7). 56-62 ↑31, ↑32 Marihart, S., Harik, M., Djavan, B. (2005). Dutasteride: A Review Of Current Data On A Novel Dual Inhibitor Of 5alpha Reductase. Reviews In Urology. 7(4). 203-210 ↑33, ↑34, ↑35 Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑36 Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A Review. Advances In Dermatology And Allergology. 39(3). 472-478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑37 Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil And Its Use In Hair Disorders: A Review. Drug Design, Development And Therapy. 13. 2777-2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑38 Beach, R.A. (2018). Case Series Of Oral Minoxidil For Androgenetic And Traction Alopecia: Tolerability & The Five C’s Of Oral Therapy. Dermatologic Therapy. 31(6). E12707. Available at: https://doi.org/10.1111/dth.12707 ↑39 Lueangarun, S., Panchaprateep, R., Tempark, T., Noppakun, N. (2015). Efficacy And Safety Of Oral Minoxidil 5 Mg Daily During 24-Week Treatment In Male Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 72(5). AB113. Available at: https://doi.org/10.1016/j.jaad.2015.02.466 ↑40 Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral Minoxidil In Male Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 81(2). 648-649. Available at: https://doi.org/10.1016/j.jaad.2019.04.054 ↑41 Pirmez, R., Salas-Callo, C.I. (2020). Very-Low-Dose Oral Minoxidil In Male Androgenetic Alopecia: A Study With Quantitative Trichoscopic Documentation. Journal Of The American Academy Of Dermatology. 82(1). E21-E22. Available at: https://doi.org/10.1016/j.jaad.2019.08.084 ↑42 Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair loss: a multicenter study of 1404 patients. Journal of the American Academy of Dermatology. 84(6). 1644–1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑43 Wikramanayake, T.C., Rodriguez, R., Choudhary, S., Mauro, L.M., Nouri, K., Schachner, L.A., Jimenez, J.J. (2012). Effects of the Lexington LaserComb on Hair Regrowth in the C3H/HeJ Mouse Model of Alopecia Areata. Lasers in Medical Science. 27(2). 431-436. Available at: https://doi.org/10.1007/s10103-011-0953-7 ↑44 Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts and Bolts of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available at: https://doi.org/10.1007/s10439-011-0454-7 ↑45 Makihara, E., Masumi, S. (2008). Blood Flow Changes of a Superficial Temporal Artery Before and After Low-Level Laser Irradiation Applied to the Temporomandibular Joint Area. Nihon Hotetsu Shika Gakkai Zasshi. 52(2). 167-170. Available at: https://doi.org/10.2186/jjps.52.167 ↑46 Bouzari, N., Firooz, A.R. (2006). Lasers May Induce Terminal Hair Growth. Dermatologic Surgery. 32(3). 460. Available at: https://doi.org/10.1111/j.1524-4725.2006.32092.x ↑47 Lueangarun, S., Visutjindaporn, P., Parcharoen, Y., Jamparuang, P., Tempark, T. (2021). A Systematic Review and Meta-Analysis of Randomized Controlled Trials of United States Food and Drug Administration-Approved, Home-Use, Low-Level Light/Laser Therapy Devices for Pattern Hair Loss: Device Design and Technology. Journal of Clinical and Aesthetic Dermatology. 14(11). E64-E75 ↑48, ↑49 Shin, J.W., Paik, K., Na, J.I., Lew, B.L., Huh, C.H. (2026). Long-Term Efficacy and Safety of Low-Level Laser Therapy for Androgenetic Alopecia: A 12-Month Prospective Trial. Dermatologic Therapy. Available at: https://doi.org/10.1155/dth/6621458 ↑50 HairLossTalk, (2019), Microneedling Photo Results Summary. Available at: https://www.hairlosstalk.com/interact/threads/microneedling-photo-results-summary.121072/ (Accessed: 24 February 2026) ↑51 Pei, D., Zeng, L., Huang, X., Wang, B., Liu, L., Zhang, G. (2024). Efficacy and Safety of Combined Microneedling Therapy for Androgenic Alopecia: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Journal of Cosmetic Dermatology. Available at: https://doi.org/10.1111/jocd.16186 ↑52 Dhurat, R., Sukesh, M., Avhad, G., Dandale, A., Pal, A., Pund, P. (2013). A Randomized Evaluator Blinded Study of Effect of Microneedling in Androgenetic Alopecia: A Pilot Study. International Journal of Trichology. 5(1). 6-11. Available at: https://doi.org/10.4103/0974-7753.114700 ↑53 Dhurat, R., Mathapati, S. (2015). Response to Microneedling Treatment in Men with Androgenetic Alopecia Who Failed to Respond to Conventional Therapy. Indian Journal of Dermatology. 60(3). 260-263. Available at: https://doi.org/10.4103/0019-5154.156361 ↑54 Bao, L., Gong, L., Guo, M., Liu, T., Shi, A., Zong, H. (2020). Randomized Trial of Electrodynamic Microneedle Combined with 5% Minoxidil Topical Solution for the Treatment of Chinese Male Androgenetic Alopecia. Journal of Dermatological Treatment. Available at: https://doi.org/10.1080/14764172.2017.1376094 ↑55 Wikimedia Commons, (2025), Ketoconazole Chemical Structure (SVG). Available at: https://commons.wikimedia.org/wiki/Category:Ketoconazole#/media/File:(2S,4R)-Ketoconazol.svg (Accessed: 24 February 2026) ↑56, ↑57 Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: https://doi.org/10.1159/000017954 ↑58 Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine And 1% Zinc Pyrithione Formulations. International Journal Of Cosmetic Science. 24(5). 249-256. Available at: https://doi.org/10.1046/j.1467-2494.2002.00145.x ↑59 Khandpur, S., Suman, M., Reddy, B.S. (2002). Comparative Efficacy Of Various Treatment Regimens For Androgenetic Alopecia In Men. J Dermatol. 29(8). 489-98. Available at: https://doi.org/10.1111/j.1346-8138.2002.tb00314.x ↑60 Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral And Topical Administration Of A Standardized Saw Palmetto Oil Reduces Hair Fall And Improves The Hair Growth In Androgenetic Alopecia Subjects – A 16-Week Randomized, Placebo-Controlled Study. Clinical, Cosmetic And Investigational Dermatology. 16. 3251–3266. Available at: https://doi.org/10.2147/CCID.S435795 ↑61, ↑62 Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparitive Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A Two-Year Study. International Journal of Immunopathology and Pharmacology. 25(4). 1167-1173. Available at: https://doi.org/10.1177/039463201202500435 ↑63 Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., Bunnag, T., Pimonrat, T., Muangdang, N., Pichai, P. (2016). Treatment of Male Androgenetic Alopecia with Topical Products Containing Serenoa Repens Extract. Australasian Journal of Dermatology. 57(3). e76-e82. Available at: https://doi.org/10.1111/ajd.12352 ↑64 Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. 57(3), e76-e82. Available at: https://doi.org/10.1111/ajd.12352 ↑65 Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the Hair Growth in Androgenetic Alopecia Subjects – A 16-Week Randomized, Placebo-Controlled Study. Clin Cosmet Investig Dermatol. 16, 3251-3266. Available at: https://doi.org/10.2147/CCID.S435795 Men’s hair loss is a booming multi-billion-dollar industry, yet the vast majority of over-the-counter products fall short when measured against rigorous clinical evidence. This landscape is cluttered with quick-fix promises, leaving consumers sifting through marketing claims with little scientific support. Among all topical hair regrowth options, minoxidil is one of the most extensively studied. It is also one of two FDA-approved treatments for male pattern hair loss (in its topical form).
In this comprehensive guide, we rank our top minoxidil picks of 2026 across Best Overall, Best Value, and other categories to help you navigate the crowded hair loss market with more clarity and confidence.
Quick Look: Best Minoxidil for Men in 2026
Product Strength Format Customization Price Best: Ulo 7% Solution High $41.65 Overall Kirkland Minoxidil 5% Solution None $17.99 Value Rogaine 5% Solution/Foam None $49.97 Sensitive Scalp Keeps 5% Solution/Foam/Spray Low $16.67 Subscription Convenience Happy Head 5-8% Solution/Gel High $59 Strength What is Minoxidil and How Does it Work?
Minoxidil was originally developed to treat high blood pressure and is now widely used in both topical and oral forms as a first-line therapy for androgenic alopecia (AGA).
What is Minoxidil?
Minoxidil was first introduced in the 1970s as a powerful oral vasodilator for resistant hypertension. Clinicians soon noticed a frequent side effect, generalized increased hair growth (hypertrichosis).[1]Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2021). Minoxidil: a comprehensive review. Journal of Dermatological Treatment. 33(4). 1896-1906. Available at: … Continue reading This led to the development of topical formulations specifically for AGA. Today, 2-5% topical minoxidil is FDA-approved for both male and female pattern hair loss and is used off-label for other alopecias.
How Does Minoxidil Work?
Minoxidil is a potassium-channel opener that causes vasodilation and improves microcirculation around hair follicles.[2]Hussein, R.S., Dayel, S.B., Abahussein, O., El-Sherbiny, A.A. (2024). Applications and efficacy of minoxidil in dermatology. Skin Health and Disease. 4(6). E472. Available at: … Continue reading It also acts as a prodrug; sulfotransferase converts it to minoxidil sulfate, which prolongs the anagen (growth) phase and shortens the telogen (resting) phase, shifting more hair follicles into active growth.[3]Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: a … Continue reading Additional data suggests that induction of key growth factors like vascular endothelial growth factor (VEGF) and activation of Wnt/ꞵ-catenin signaling further support hair regrowth.[4]Gupta, A.K., Talukder, M., Shemer, A., Piraccini, B.M., Tosti, A. (2023). Low-Dose Oral Minoxidil for Alopecia: A Comprehensive Review. Skin Appendage Disorders. 9(6). 423-437. Available at: … Continue reading
Although minoxidil doesn’t target dihydrotestosterone, it can improve the effectiveness of other treatments by targeting multiple other targets. When combined with anti-DHT drugs like finasteride, which reduce the hormonal trigger of AGA, minoxidil provides a complementary, non-hormonal pathway. The two treatments have been shown to provide synergistic improvement in hair density in men.[5]Asad, N., Naseer, M., Ghafoor, R. (2024). Efficacy of Topical Finasteride 0.25% with Minoxidil 5% versus Topical Minoxidil 5% Alone in Treatment of Male Pattern Androgenic Alopecia. Journal of Drugs … Continue reading
OTC vs Prescription-Strength
Over-the-counter (OTC) minoxidil solutions (2% and 5%) can be effective for many men, but absorption and vehicle limits mean “stronger” drug percentages above 5% often add irritation rather than results unless the entire formulation is engineered and supervised medically.
Early minoxidil development work showed a clear dose-response between low strengths (1-2%) and mid-strength (5%) solutions, with 5% yielding larger gains in hair counts and shaft diameter in AGA.[6]Olsen, E.A., Dunlap, F.E., Funicella, T., Koperski, J.A., Swinehart, J.M., Tschen, E.H., Trancik, R.J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical minoxidil and … Continue reading
Other companies offer concentrations of 10% or over; however, recent head-to-head studies have shown that 5% topical minoxidil is moderately superior to the higher concentration.[7]Ghonemy, S., Alarawi, A., Bessar, H. (2021). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic … Continue reading Additionally, the 10% minoxidil was associated with more marked irritation.
So, with that in mind, let’s take a look at some of our top minoxidil picks of 2026.
Best Minoxidil Picks for Men
#1 Best Overall: Ulo Minoxidil Rx
Pros: Cons: ✓ Clinically-backed combinations ✗Prescription products are only available in the USA ✓ Ongoing medical monitoring and a user-friendly platform ✓ Quality-tested ingredients free of irritants Ulo’s Minoxidil Rx takes the top spot for men who want a data-driven, medically supervised approach rather than a one-size-fits-all bottle. Custom strengths and evidence-based optional add-ons allow precise targeting for hair regrowth.
Their high-strength concentration can be combined with tretinoin 0.01% to enhance absorption and efficacy, as well as cetirizine 1%, melatonin 0.01% for antioxidant protection, and caffeine 0.2% to boost circulation, which further improves the product’s effectiveness.
This treatment begins at $49 per month, increasing to $54 if you choose to have all of the add-ons.
Bottom Line: Ulo’s customizable, science-driven minoxidil prescription offers one of the most advanced, value-conscious options for men who are serious about long-term hair regrowth.
#2 Best Value – Kirkland Signature Minoxidil 5%
Pros: Cons: ✓ Lowest cost we could find for 6 months’ worth. ✗Contains propylene glycol, which can cause irritation or dryness in some users. ✓ Same core 5% minoxidil strength. ✗ No customization possible for strength or add-on ingredients. ✓ Widely available at multiple stores, including Amazon, Costco, etc. ✗ No medical consultation or tailoring of treatment included. ✓ Simple formula. Kirkland Signature Minoxidil 5% is the classic value choice for me who want an over-the-counter treatment without paying for branding or bundled medical services. You get a standard 5% solution similar in strength to more expensive competitors, making it ideal for cost-conscious users who are happy to follow a simple twice-daily routine and do not need personalized compounding.
Bottom line: If you want the most affordable entry point into 5% minoxidil, Kirkland’s no-frills formula is hard to beat for sheer value per effective dose.
#3 Best for Sensitive Scalps: Rogaine 5% Foam
Pros: Cons: ✓ Clinically proven 5% minoxidil strength ✗On the higher end in terms of price. ✓ Foam vehicle is propylene glycol-free, so it is usually better tolerated on sensitive or irritated scalps. ✗ Fixed strength for men with no customization or add-on actives. ✓ Quick drying, less greasy texture ✗ No medical consultation or tailoring of treatment included. ✓ Widely available OTC Rogaine 5% is a strong pick for men who need minoxidil but find traditional liquid formulas too irritating or cosmetically heavy. The foam format avoids common irritants found in many solutions and tends to be more comfortable for daily use, which can improve adherence and long-term outcomes.
Bottom Line: For sensitive scalps that still need full-strength 5% minoxidil, Rogaine Foam provides a gentler, easy-to-use option that balances proven efficacy with better tolerability.
#4 Best Subscription Convenience: Keeps Minoxidil
Pros: Cons: ✓ Automatic subscription refills so you rarely run out or miss doses. ✗More expensive than buying generic minoxidil in bulk. ✓ Offers both 5% foam and 5% solution to suit different scalp and styling preferences. ✗ Limited to preset strengths and formulas with no true custom compounding. ✓ Option to bundle with finasteride and other treatments for a complete regimen. ✓ Online prescribing and follow-up. Keeps Minoxidil is designed for men who value a set-and-forget routine, trading a price increase ($16.67 for 3 months compared to Kirkland’s $17.99 for 6 months) for automatic deliveries and integrated telehealth support. The ability to pair minoxidil with finasteride and keep everything managed through a single platform could make adherence simpler for busy users.
Bottom Line: If you want 5% minoxidil with minimal hassle, Keeps’ subscription model is a strong fit for convenience-focused men who prefer not to manage refills and prescriptions on their own.
#5 Best High-Strength Formula: Happy Head
Pros: Cons: ✓ Offers high-strength minoxidil concentrations beyond standard 5%. ✗Higher strengths may carry a greater risk of irritation or systemic absorption. ✓ Can be combined with topical finasteride and other actives in a single solution. ✗ More expensive than generic 5% minoxidil and some standard telehealth offerings. ✓ Telehealth model with online consultation and prescription management. ✗Corticosteroids used to offset irritation from propylene glycol. ✓ Multiple formula options aimed at men who have plateaued on basic OTC products. Happy Head is built for men who want to move past entry-level minoxidil and explore higher-strength, prescription-only blends under medical oversight. By pairing elevated minoxidil concentrations (8%) with agents like tretinoin and topical finasteride, Happy Head targets users seeking a more comprehensive protocol. With that said, some ingredients included, like the carrier agent propylene glycol, which is used in their base formula, can cause irritation. To offset this, Happy Head uses corticosteroids; however, this may cause skin thinning long-term.
You can read more about corticosteroid usage in hair loss treatments here.
Bottom Line: Happy Head delivers a good jump in strength for minoxidil users who need to increase efficacy.
Timeline: What Results to Expect from Minoxidil
It’s important that realistic expectations are set for regrowth when starting out on your minoxidil usage journey.
Timeframe What to Expect Clinical Evidence Months 0-3 This is the initial shedding phase, which usually resolves in the first 4-8 weeks. [8]Nohria, A., Desai, D., Sikora, M., Mandal, S., Shapiro, J., Lo Sicco, K. (2024). Combating “dread shed”: The impact of overlapping topical and oral minoxidil on temporary hair shedding during … Continue reading Months 3-6 Early visible improvements, like the appearance of new hairs, can be expected. [9]Amit, K., Mansukh, G., Satyaprakash, M., Dhiraj, D., Hanmant, B. (2023). Real-World Effectiveness, Safety, and Tolerability of Cetosomal Minoxidil 5% Alone and a Fixed Drug Combination of Cetosomal … Continue reading Months 6-12 Cosmetic changes peak, with stronger hair, increased density, and most coverage is seen. [10]Katz, H.I., Hien, N.T., Prawer, S.E., Goldman, S.J. (1987). Long-term efficacy of topical minoxidil in male pattern baldness. Journal of the American Academy of Dermatology. 16(3). 711-718. Available … Continue reading Months 12+ Further gains plateau, continued use needed to maintain improvements. [11]Katz, H.I., Hien, N.T., Prawer, S.E., Goldman, S.J. (1987). Long-term efficacy of topical minoxidil in male pattern baldness. Journal of the American Academy of Dermatology. 16(3). 711-718. Available … Continue reading Consistency is Everything
Minoxidil works by prolonging the anagen phase and shortening the telogen phase. Regular dosing maintains these effects at the follicle level; repeatedly missing applications reduces the cumulative time that hair follicles spend in a pro-growth, anagen-biased state.[12]Messenger, A.G., Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 2(1). 186-194. Available at: … Continue reading
Because human scalp follicles cycle slowly (anagen can last years), many follicles need months of uninterrupted exposure before visible density gains can be seen.[13]Hoover E, Alhajj M, Flores JL. Physiology, Hair. [Updated 2023 Jul 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: … Continue reading This is why most guidance recommends at least 6 months of continuous use before judging the response.
Why Stopping Reverses Progress
Minoxidil is not disease-modifying for AGA, meaning that it does not correct androgen-driven miniaturization but counteracts it while present by improving hair cycle dynamics and follicle size. When treatment is stopped, these pharmacologic effects wane, and follicles gradually revert toward their genetically determined, androgen-sensitive pattern.
Clinical and patient information sources consistently note that any gain from minoxidil is maintained only while therapy continues. After discontinuation, increased shedding and loss of density over months are typical, ultimately leading to a reversal of gains to the original starting point.
What Minoxidil Formulations are Available?
You can buy minoxidil in several different vehicles that differ mainly in tolerability, practicality, and how well they fit a given scalp and routine. Choosing the “best” format is less about raw efficacy and more about matching the base to skin sensitivity and your preference of application.
- Foam – Best for Sensitive Skin: Propylene glycol-free foams were developed to reduce contact dermatitis and dryness seen with classic liquid solutions. Reviews note lower rates of irritation and quicker drying, which makes them better for patients with reactive or dry scalps.[14]Purnak, T., Senel, E., Sahin, C. (2011). Liquid formulation of minoxidil versus its foam formulation. Indian Journal of Dermatology. 56(4). 462. Available at: https://doi.org/10.4103/0019-5154.84714
- Liquid – Best for Cost-Effectiveness: Traditional solutions with alcohol and propylene glycol are widely available, inexpensive, and easy to use, but they may cause increased itching, flaking, and stinging. This formulation may suit users with non-sensitive skin who are price-conscious.
- Spray – Easiest for Coverage: Spray tops or aerosolized formats can make it faster to hit broad thinning areas, especially in longer hair. However, the product may end up more on the hair shafts or adjacent skin.
- Gel – Targeted and Controlled: Gel or cream-gel vehicles (often from compounders) sit where you place them, minimize dripping, and are useful for localized areas such as temples or post-transplant zones.
- Liposomal Delivery: Newer liposomal or phospholipid bases aim to enhance follicular penetration while limiting systemic absorption and irritation. This is usually marketed as an “advanced delivery” method.
Side Effects and Safety
Minoxidil is generally well tolerated, but like any active drug, it can cause local irritation, a transient shedding phase, and very rare systemic effects, especially in people with underlying skin or cardiovascular issues.
Common local side effects include:
- Itching
- Flaking
- Redness
- Burning
- Temporary shedding
Systemic and heart-related side effects are very rare with correctly used topical minoxidil, but case reports and product information advise seeking medical help if symptoms like chest pain, palpitations, dizziness, or unexplained swelling occur, as these could reflect systemic absorption or accidental ingestion. Risk is higher with oral minoxidil or overdose, where hypotension, tachycardia, and even heart failure have been documented.[15]Tripathee, S., Benyovszky, A., Devbhandari, R., Quiza, K., Boris, J. (2024). A Very Bad Hair Day: Minoxidil Ingestion Causing Shock and Heart Failure. Cureus. 16(8). E66039. Available at: … Continue reading
To minimize problems, many clinicians suggest starting once daily and titrating up, choosing foam or gentler vehicles to reduce dermatitis, and using custom compounded formulas (for example, lower alcohol, lower strength, or liposomal bases) in those with sensitive or disease-prone scalps.
People with chronic inflammatory scalp conditions such as active psoriasis, eczema, or severe seborrheic dermatitis are typically advised to get the primary condition under control before adding topical minoxidil or other actives, as applying them to an inflamed barrier increases irritation, may worsen the disease, and can unpredictably alter absorption.[16]Junge, A., Jic-Hoesli, S.R., Bossart, S., Simon, D., de Viragh, P., Hunger, R.E., Heidemeye, K., Seyed Jafari, S.M. (2025). Contact Dermatitis Caused by Topical Minoxidil: Allergy or Just Irritation. … Continue reading
Final Verdict – Best Minoxidil for Men in 2026
In 2026, several minoxidil-based options stand out for different priorities, from cost to customization. All of them rely on the same core ingredient, but vehicle, strength, and add-ons make a real-world difference in adherence and results.
Whichever route you choose, there is strong evidence that consistent minoxidil use can stabilize loss and improve density for many men, and modern platforms make it easier than ever to find a formulation that fits your scalp, schedule, and risk tolerance.
References[+]
References ↑1 Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2021). Minoxidil: a comprehensive review. Journal of Dermatological Treatment. 33(4). 1896-1906. Available at: https://doi.org/10.1080/09546634.2021.1945527 ↑2 Hussein, R.S., Dayel, S.B., Abahussein, O., El-Sherbiny, A.A. (2024). Applications and efficacy of minoxidil in dermatology. Skin Health and Disease. 4(6). E472. Available at: https://doi.org/10.1002/ski2.472 ↑3 Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: a review. Advances in Dermatology and Allergology. 39(3). 472-478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑4 Gupta, A.K., Talukder, M., Shemer, A., Piraccini, B.M., Tosti, A. (2023). Low-Dose Oral Minoxidil for Alopecia: A Comprehensive Review. Skin Appendage Disorders. 9(6). 423-437. Available at: https://doi.org/10.1159/0000531890 ↑5 Asad, N., Naseer, M., Ghafoor, R. (2024). Efficacy of Topical Finasteride 0.25% with Minoxidil 5% versus Topical Minoxidil 5% Alone in Treatment of Male Pattern Androgenic Alopecia. Journal of Drugs in Dermatology. 23(11). 1003-1008. Available at: https://doi.org/10.36849/JDD.7826 ↑6 Olsen, E.A., Dunlap, F.E., Funicella, T., Koperski, J.A., Swinehart, J.M., Tschen, E.H., Trancik, R.J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical minoxidil and placebo in the treatment of androgenetic alopecia in men. Journal of the American Academy of Dermatology. 47(3). 377-385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑7 Ghonemy, S., Alarawi, A., Bessar, H. (2021). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic evaluation. Journal of Dermatological Treatment. 32(2). 236-241. Available at: https://doi.org/10.1080/09546634.2019.1654070 ↑8 Nohria, A., Desai, D., Sikora, M., Mandal, S., Shapiro, J., Lo Sicco, K. (2024). Combating “dread shed”: The impact of overlapping topical and oral minoxidil on temporary hair shedding during oral minoxidil initiation. JAAD International. 15. 220-224. Available at: https://doi.org/10.1016/j.jdin.2024.03.005 ↑9 Amit, K., Mansukh, G., Satyaprakash, M., Dhiraj, D., Hanmant, B. (2023). Real-World Effectiveness, Safety, and Tolerability of Cetosomal Minoxidil 5% Alone and a Fixed Drug Combination of Cetosomal Minoxidil 5% With Finasteride 0.1% in the Management of Androgenetic Alopecia (Inbilt Study). Cureus. 15(7). E41681. Available at: https://doi.org/10.7759/cureus.41681 ↑10, ↑11 Katz, H.I., Hien, N.T., Prawer, S.E., Goldman, S.J. (1987). Long-term efficacy of topical minoxidil in male pattern baldness. Journal of the American Academy of Dermatology. 16(3). 711-718. Available at: https://doi.org/10.1016/s0190-9622(87)70092-9 ↑12 Messenger, A.G., Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 2(1). 186-194. Available at: https://doi.org/https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑13 Hoover E, Alhajj M, Flores JL. Physiology, Hair. [Updated 2023 Jul 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499948/ (Accessed: November 2025) ↑14 Purnak, T., Senel, E., Sahin, C. (2011). Liquid formulation of minoxidil versus its foam formulation. Indian Journal of Dermatology. 56(4). 462. Available at: https://doi.org/10.4103/0019-5154.84714 ↑15 Tripathee, S., Benyovszky, A., Devbhandari, R., Quiza, K., Boris, J. (2024). A Very Bad Hair Day: Minoxidil Ingestion Causing Shock and Heart Failure. Cureus. 16(8). E66039. Available at: https://doi.org/10.7759/cureus.66039 ↑16 Junge, A., Jic-Hoesli, S.R., Bossart, S., Simon, D., de Viragh, P., Hunger, R.E., Heidemeye, K., Seyed Jafari, S.M. (2025). Contact Dermatitis Caused by Topical Minoxidil: Allergy or Just Irritation. Acta Dermato-Venereologica. 105. 42401. Available at: https://doi.org/10.2340/actadv.v105.42401 Ozempic and other GLP-1 agonists have rapidly gained the status of “cultural phenomenon” over the past few years. A 2024 survey found that around 12% of adults in the US have used a GLP-1 agonist drug, with 6% currently taking the drug.[1]Montero, A., Sparks, G., Presiado, M., Hamel, L., (2024), KFF Health Tracking Poll May 2024: The Public’s Use And Views Of GLP-1 Drugs. Available at: … Continue reading This equates to roughly 30 million people, and numbers are only rising.
While the use of GLP-1 agonists was originally limited to the treatment of diabetes, they are now facing huge demand for their weight loss actions. Amongst these GLP-1 agonists, Ozempic is the most popular, making up approximately 37.8% of the GLP-1 agonist market with sales of around $22.3 billion in 2025.[2]Future Market Insights Inc., (no date), Ozempic Market Share & Size 2025-2035. Available at: https://www.futuremarketinsights.com/reports/ozempic-market (Accessed: 27 February 2026),[3]Research And Markets, (2024), Global GLP-1 Receptor Agonist (Ozempic, Rybelsus, Trulicity, Saxenda, Wegovy, Victoza, Mounjaro, Zepbound) Market Analysis Report 2024-2030 Featuring Eli Lilly And C, … Continue reading
GLP-1 agonists have not been in the public sphere for very long, meaning that understanding of long-term side effects is limited. As more and more people are using the drugs, potential side effects are beginning to emerge.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

One such side effect receiving growing concern on various social media and forums is hair loss, with reports including increased shedding and thinning of hair. But how accurate are these reports? Are they based on clinical evidence, or are they purely anecdotal?
To find out, we have searched through clinical literature to see whether the science supports these claims. In this article, we will review what Ozempic is and how it works, the available evidence supporting hair loss claims, and the mechanism through which Ozempic may be causing these issues.
What is Ozempic?
Ozempic is the brand name for the drug semaglutide, which is a glucagon-like peptide-1 (GLP-1) agonist. It is an FDA-approved drug originally developed to improve glycemic control in adults with type 2 diabetes, and now commonly used off-label as a weight-loss treatment.[4]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
While conducting early clinical trials for the use of semaglutide in diabetes, researchers noticed that patients taking semaglutide consistently lost more weight than those taking the placebo.[5]Sorli, C., Harashima, S.I., Tsoukas, G.M. (2017). Efficacy And Safety Of Once-Weekly Semaglutide Monotherapy Versus Placebo In Patients With Type 2 Diabetes (SUSTAIN 1): A Double-Blind, Randomised, … Continue reading These results prompted researchers to investigate semaglutide specifically as a weight-loss drug, where they demonstrated a ~15% reduction in weight in those treated with a weekly injection of semaglutide.[6]Garvey, W.T., Batterham, R.L., Bhatta, M. (2022). Two-Year Effects Of Semaglutide In Adults With Overweight Or Obesity: The STEP 5 Trial. Nature Medicine. 28(10). 2083-2091. Available at: … Continue reading
Semaglutide is available in three main forms:[7]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
Ozempic: once-weekly injection; FDA-approved for the treatment of type 2 diabetes.
Rybelsus: once daily tablets; FDA-approved for the treatment of type 2 diabetes.
Wegovy: once weekly injection; FDA-approved for weight loss.Other related GLP-1 agonists include Mounjaro and Liraglutide; these do not contain semaglutide as their active ingredient and work through slightly different mechanisms.
How Does Ozempic Work?
GLP-1 is a hormone that is released into the bloodstream in response to food consumption. It acts in the pancreas, gastrointestinal tract, and central nervous system. Its main effects are:[8]Holst, J.J. (2007). The Physiology Of Glucagon-Like Peptide 1. Physiological Reviews. 87(4). 1409-1439. Available at: https://doi.org/10.1152/physrev.00034.2006,[9]Drucker, D.J. (2022). GLP-1 Physiology Informs The Pharmacotherapy Of Obesity. Molecular Metabolism. 57. 101351. Available at: https://doi.org/10.1016/j.molmet.2021.101351
- Stimulating insulin secretion after food consumption to improve blood glucose levels post-prandial (after eating).
- Inhibiting glucagon secretion to limit glucose in the blood and help to reduce spikes in blood sugar.
- Slowing the emptying of the stomach, which further limits blood sugar spikes and helps people to feel fuller for longer.
- Activating GLP-1 receptors in the brain (responsible for appetite regulation), which results in increased satiety and reduced food cravings.
Ozempic is a synthetic analogue of natural GLP-1, essentially mimicking GLP-1 in the body to activate GLP-1 receptors. Through this, it activates the same pathways within the body as natural GLP-1, thereby acting to reduce blood sugar and food cravings.[10]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
Is There Any Association Between Ozempic and Hair Loss?
While Ozempic has been hailed by many as a miracle drug for the growing obesity crisis, it is not without risks. Many people experience gastrointestinal-linked side effects, including nausea, diarrhoea, stomach pain, and reflux.[11]Kommu, S., Berg, R.L. (2024). Efficacy And Safety Of Once-Weekly Subcutaneous Semaglutide On Weight Loss In Patients With Overweight Or Obesity Without Diabetes Mellitus-A Systematic Review And … Continue reading A small subset of people may experience more serious side effects, such as pancreatitis, issues with the gallbladder, or even kidney injury.[12]Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026)
These side effects have been demonstrated in clinical trials and are relatively well-established. However, other side effects are seemingly coming to light on social media and forums, such as Reddit, with hair loss appearing to be a significant cause for concern. But is this true?
We will investigate the clinical evidence supporting this link between Ozempic and hair loss and discuss what the science says so far.
Clinical Evidence
The existing clinical data demonstrating hair loss with Ozempic treatment are limited. During the process of getting a product to the market, drugs like Ozempic go through extensive clinical trials to assess how effective they are, in addition to their safety. Any side effects or adverse events are recorded, enabling researchers to assess whether the benefits of the drug outweigh potential harms.
The initial clinical trials with Ozempic did not record alopecia or hair loss as a side effect. However, once the drug hit the market, reports of hair loss began to appear. There were sufficient reports that the FDA issued a warning in 2023 that GLP-1 receptor agonists (including Ozempic) may increase the risk of alopecia.[13]Center For Drug Evaluation And Research, (2024), July – September 2023 | Potential Signals Of Serious Risks/New Safety Information Identified By The FDA Adverse Event Reporting System (FAERS). … Continue reading
Case Reports
Case reports are short, published articles describing rare or unexpected clinical scenarios, such as adverse drug reactions. They typically describe one patient, and therefore do not demonstrate comprehensive proof of a drug effect for all users, only a one-off situation where this has occurred.
With the introduction of Ozempic to the masses, clinical case reports have begun to emerge demonstrating rare incidences of alopecia in those taking Ozempic.
In one such study, an obese female in her early twenties who had been taking semaglutide for weight loss for four months started to develop patches of hair loss after three months (on an increase in dose from 0.25 to 0.5 mg weekly). After cessation of semaglutide and the initiation of alopecia management (consisting of intralesional corticosteroid injections, ketoconazole 2% shampoo, and minoxidil 5% topical solution), hair regrowth occurred.[14]Alzahrani, W.S., Bahkali, S.A., Alharthy, R.F., Alsabban, A.S. (2025). Alopecia Areata Following Semaglutide Treatment For Weight Loss: A Case Report. JAAD Case Reports. 63. 44-46. Available at: … Continue reading
Figure 2. Patch of hair loss following semaglutide treatment. Adapted from Figure 1.[15]Alzahrani, W.S., Bahkali, S.A., Alharthy, R.F., Alsabban, A.S. (2025). Alopecia Areata Following Semaglutide Treatment For Weight Loss: A Case Report. JAAD Case Reports. 63. 44-46. Available at: … Continue reading Image used under Creative Commons License.
An additional case study similarly showed a patch of hair loss after semaglutide treatment (2.4 mg weekly). An initial small patch occurred after only two days of treatment and had increased to 5 cm2 after 6 weeks. The alopecia had improved within two months of stopping semaglutide treatment.[16]Cheng, J.R., Zheng, J., Li, Y. (2022). Weight Loss-Associated Alopecia Areata. American Journal Of Therapeutics. Available at: https://doi.org/10.1097/MJT.0000000000001851
Study #1
The first study investigating the link between Ozempic and alopecia was published in 2025. They investigated cases of alopecia associated with semaglutide and tirzepatide (Mounjaro) reported via FAERS, the FDA Averse Event Reporting System, which collects reports of drug adverse events from both consumers (i.e., the general public) and healthcare professionals.[17]Godfrey, H., Leibovit-Reiben, Z., Jedlowski, P., Thiede, R. (2025). Alopecia Associated With The Use Of Semaglutide And Tirzepatide: A Disproportionality Analysis Using The FDA Adverse Event … Continue reading
Their search covered cases reported from 2022 to 2023. Within this time, there were 199 reports of alopecia for semaglutide, 179 reports for tirzepatide, and a lower number for other GLP-1 receptor agonists (GLP-1 RAs).
The researchers then carried out a disproportionality analysis, a type of statistical test that assesses whether side effects are reported more often in people using a drug than would occur in the general population. They demonstrated increased reporting odds, meaning that alopecia was reported more frequently in those taking semaglutide (or tirzepatide) than would be expected in the general population.
While this study highlights a potential association between semaglutide and alopecia, the methodology and type of analysis carried out cannot prove a direct, causal relationship.
In addition, the study was limited by the nature of FAERS, where the reporting of adverse events is voluntary. As such, the data can be influenced by heightened awareness of side effects, such as through media attention or exposure to reports on social media.
The researchers reported that 84% of the alopecia reports made to FAERS were from consumers and only 16% were from healthcare professionals. This makes it likely that a significant proportion of reports were made by people with no clinician-confirmed diagnosis of alopecia. Moreover, the type of hair loss was not reported in many cases, further limiting the conclusions that can be drawn from this study.
In summary, although providing evidence for a possible association between semaglutide and alopecia, further research is essential to determine whether semaglutide is directly affecting hair loss or whether other factors (such as weight loss, nutrient deficiencies, or underlying health conditions) are primarily at fault.
Study #2
A second study, also published in 2025, used retrospective analysis to investigate the link between GLP-1 RAs and alopecia in a real-world setting.[18]Burke, O., Sa, B., Alvarez Cespedes, D., Sechi, A., & Tosti, A. (2025). Glucagon-like peptide-1 receptor agonist medications and hair loss: a retrospective cohort study. Journal of the American … Continue reading They analyzed the medical records of 283 patients who were taking GLP-1 RAs and who had visited their dermatology department between 2021 and 2023.
Of these 283 patients, the majority did not experience any hair loss (84.1%). Three patients (1.2%) with no previous reports of hair loss experienced new-onset hair loss. This is approximately the same rate as would be expected in the general population over two years, and so does not indicate any major effects of GLP-1 RA treatment.
However, for those who had preexisting hair loss, the story is different. 32 people within the study had preexisting hair loss (approximately 13%), and of these people, over 90% reported that their hair loss had worsened since taking GLP-1 RAs. While this sounds conclusive and concerning, it should be noted that male pattern hair loss (androgenic alopecia; AGA) is a progressive condition where hair loss worsens over time. As such, it is difficult to separate normal hair loss over time from medication-induced hair loss.
On performing statistical analyses, no significant association was found between GLP-1 RAs and AGA; however, semaglutide did show a possible trend towards increased hair loss.
This study provides more supporting evidence towards a link between semaglutide and AGA; however, researchers conceded several limitations with their study. Similarly to the first study described above, this work was retrospective and lacked a control group. As such, it is not possible to prove a cause-and-effect relationship due to there being no way to compare the normal progression of AGA hair loss over time with drug-induced hair loss.
In addition, the duration of use of the GLP-1 RA medication was not recorded, nor was the reason for starting the medication. The study also did not use objective measures to clinically assess hair loss, such as hair counts, but relied on self-reported data from patients.
As with the previous study, this work provided some evidence supporting a link between semaglutide and hair loss, but larger, controlled clinical trials with objective hair measurements are essential to conclusively prove whether the drug is affecting hair growth.
Study #3
A third study demonstrated a similar conclusion to the first two studies. They carried out a retrospective analysis evaluating exposure to GLP-1 RAs and the risk of hair loss.[19]Akiska, Y.M., Vidal, S.I., Menta, N. (2025). Increased Incidence And Risk Of Hair Loss With Glucagon-Like Peptide 1 Receptor Agonists: A Real-World Multicentre Cohort Study. EMJ. 13(1). 52-54. … Continue reading They used the TriNetX US Collaborative Network, a real-world data network of healthcare organizations in the U.S. that pools electronic health record data. They matched cohorts of over 500,000 adults and demonstrated that GLP-1 RA exposure was significantly associated with an increased risk of AGA.
Again, this study was limited by its retrospective analysis and inability to prove causation, but it does provide further evidence linking GLP-1 RAs to hair loss.
How Might Ozempic Cause Hair Loss?
Data supporting a potential link between Ozempic and hair loss is growing, but what do scientists suggest is causing this link?
Rapid Weight Loss and Telogen Effluvium
Ozempic causes rapid weight loss due to appetite suppression, reduced food cravings, and an overall calorie reduction. When the body experiences a prolonged calorie deficit, it goes under a form of metabolic stress.
Metabolic stress caused by rapid weight loss is a well-documented trigger for a condition known as telogen effluvium. Hair goes through a growth cycle consisting of multiple phases, including the anagen phase (growth) and the telogen phase (resting). In a healthy scalp, approximately 85% of hair is in the anagen phase and 15% in the telogen phase.[20]Hughes, E.C., Syed, H.A., Saleh, D., (2025), Telogen Effluvium. Available at: http://www.ncbi.nlm.nih.gov/books/NBK430848/ (Accessed: 02 March 2026)
Telogen effluvium is a condition where a physiological stress (such as rapid weight loss) causes hairs to abruptly switch from the anagen to the telogen phase, resulting in increased hair shedding. The affected hairs are often diffuse throughout the head, resulting in hair thinning over the whole scalp, as opposed to patches of hair loss. The shedding often occurs several months after the initial stressor.
Rapid weight loss leading to telogen efluvium is a known phenomenon, having been identified in many weight loss situations, including crash dieting and bariatric surgery.[21]Kang, D.H., Kwon, S.H., Sim, W.Y., Lew, B.L. (2024). Telogen Effluvium Associated With Weight Loss: A Single Center Retrospective Study. Annals Of Dermatology. 36(6). 384-388. Available at: … Continue reading,[22]Zhang, W., Fan, M., Wang, C. (2021). Hair Loss After Metabolic And Bariatric Surgery: A Systematic Review And Meta-Analysis. Obesity Surgery. 31(6). 2649-2659. Available at: … Continue reading
Telogen effluvium is generally a temporary and reversible condition, where the removal of the trigger (i.e., sudden weight loss) causes a slowing in shedding and a gradual return of hair density.
Nutritional Deficiencies
Ozempic reduces appetite and drives the decrease in food eaten. People on Ozempic must be careful with their diet to ensure that they are still eating a balanced diet containing all of the necessary vitamins and minerals to remain healthy. Studies have shown that GLP-1 RA users often do not eat adequate amounts of protein and have insufficient intake of multiple key nutrients.[23]Johnson, B., Milstead, M., Thomas, O. (2025). Investigating Nutrient Intake During Use Of Glucagon-Like Peptide-1 Receptor Agonist: A Cross-Sectional Study. Frontiers In Nutrition. 12. 1566498. … Continue reading
Certain nutrients, such as protein, iron, and zinc, are essential for normal hair cycles and hair growth. Deficiencies in these important nutrients may increase the risk of hair loss.
More research is needed in this field to definitively determine whether nutritional deficiencies are responsible for Ozempic-induced hair loss.
Direct Effect on the Hair Growth Cycle
The majority of suggested mechanisms for Ozempic-induced hair loss are indirect effects, such as metabolic stress or nutritional deficiencies. However, it may be possible that Ozempic is directly influencing the hair growth cycle.
Research in animals has found that GLP-1 receptors can be found in the skin, localized around hair follicles. One study found that GLP-1 can activate a specific signalling pathway in skin cells, promoting cell proliferation. It is possible that this could affect hair growth, but this was not proven in the study.[24]List, J.F., He, H., Habener, J.F. (2006). Glucagon-Like Peptide-1 Receptor And Proglucagon Expression In Mouse Skin. Regulatory Peptides. 134(2). 149-157. Available at: … Continue reading
The evidence for GLP-1 receptor expression in human skin is severely limited, and there is no evidence that Ozempic directly affects the hair follicle and subsequent hair growth.
It may also be possible that Ozempic disrupts metabolic and hormonal signalling pathways, such as the insulin/IGF-1 pathway. Disturbance in pathways such as these may accelerate the onset or progression of follicle shrinkage and AGA in individuals who are already predisposed to AGA. However, this mechanism is purely speculative and has not been proven.
Distinguishing AGA From Telogen Effluvium
Hair loss of any type can be distressing. In the case of potential Ozempic-induced hair loss, the main types appear to be AGA and telogen effluvium (although there is limited evidence for either). But how can you tell the difference between the two?
AGA and telogen effluvium can have overlapping symptoms, but they differ in several key areas:
- Onset: Telogen effluvium is a sudden increase in shedding occurring approximately 2-3 months after a trigger (such as starting Ozempic), whereas AGA occurs gradually over years.
- Pattern: Telogen effluvium generally results in a diffuse reduction in hair density across the whole scalp, preserving the hairline. AGA occurs in a specific pattern, with bitemporal recession (receding hairline) and vertex thinning (a bald patch at the crown).
- Hair shaft thickness: The hairs shed in telogen effluvium mostly appear normal, with a regular shaft diameter. In AGA, the miniaturization of the hair follicle often results in thinner, vellus-like hairs.
It has been suggested that Ozempic and other GLP-1 RAs may reveal an underlying propensity to AGA and accelerate the hair loss progression. In addition, rapid weight loss has been shown to trigger temporary shedding (telogen effluvium). Ozempic users may experience either AGA or telogen effluvium, both concurrently, or neither.
Should You Be Concerned?
If you have no hair loss: probably not.
The current evidence suggests that those who have not experienced any hair loss in the past are at a low risk of experiencing hair loss on taking Ozempic. The induction of new-onset hair loss appears to be uncommon in the studies carried out on this phenomenon so far. As a result, the absolute risk of hair loss is low.
If you have AGA: potentially.
The evidence so far points towards people with existing pattern hair loss experiencing a worsening of the condition on taking GLP-1 RA medications, such as Ozempic. The exact mechanisms behind this are unclear, and a lack of placebo-controlled clinical trials limits our knowledge of whether any effects seen are simply the natural progression of AGA over time. Rapid weight loss and metabolic stress could also reveal previously unnoticed hair thinning.
If you have early or unnoticed AGA: potentially.
People with a genetic predisposition to male pattern hair loss may appear to experience a sudden, dramatic loss of hair. This may occur due to the combination of underlying AGA progression with telogen effluvium-induced shedding, making previous hair loss much more noticeable.
Practical Considerations
There are many good reasons to start taking Ozempic; however, the decision should be made with the full knowledge of potential side effects, including the possibility of hair loss.
Individuals starting Ozempic who are worried about hair loss should consider:
- Avoiding extreme calorie restriction.
- Ensuring that their diet still contains an adequate amount of protein, vitamins, and minerals, including iron and zinc.
- Carrying out a baseline scalp evaluation before starting Ozempic so that any hair loss can be identified.
- Monitoring shedding.
- Starting hair loss treatments, particularly if predisposed to AGA.
Managing hair loss at the same time as completing weight-loss treatments can help to reduce visible hair loss and maintain hair density. Hair loss treatments, such as topical minoxidil, are considered safe for both men and women and may be used alongside Ozempic without fear of drug interactions.
Other hair loss treatments, such as finasteride and dutasteride, may also be able to be used alongside Ozempic, although the additional safety concerns associated with these products may discourage use.
FAQs
Q: Does semaglutide cause hair loss?
A: Ozempic is the brand name for the drug semaglutide. The data discussed in this article applies to both.
Q: Does Wegovy cause hair loss?
A: Wegovy and Ozempic both contain semaglutide, but at different dosing ranges and in different formulations. The data discussed in this article can be applied to both.
Q: What should I do if I notice hair thinning while taking Ozempic?
A: Monitor how much you are shedding and speak to your doctor. Addressing underlying hair loss early may help, as you can start using hair loss treatments alongside Ozempic.
Q: Will losing weight quickly result in hair loss?
A: Rapid weight loss is known to result in telogen effluvium for some people. The faster the weight loss, the more likely the body is to go into metabolic stress, and the higher the chance of temporary hair shedding.
Final Thoughts
So, does Ozempic cause hair loss? In summary, we’re not sure, but potentially for some people.
Reports of hair loss associated with Ozempic are increasing, but the current scientific evidence does not provide any direct evidence supporting Ozempic-induced hair loss. The available data suggest a possible association, particularly in individuals genetically predisposed to AGA or who have already started to experience hair loss prior to taking Ozempic. However, the studies so far have all been retrospective, lacked a control group, and relied on self-reported outcomes, and as a result, have been unable to demonstrate causation.
For many users, any shedding experienced is likely linked to rapid weight loss, nutritional deficiencies, or the natural progression of existing AGA, not directly as a result of Ozempic use. If you already experience AGA, it may be a good idea to monitor hair loss after starting Ozempic and to consider preventative treatment options.
As the popularity of GLP-1 RAs continues to rise, more robust, controlled clinical trials will be needed to establish the true relationship between Ozempic and hair loss. In the meantime, awareness of the potential side effects and ongoing monitoring, rather than alarm, is the correct response.
References[+]
References ↑1 Montero, A., Sparks, G., Presiado, M., Hamel, L., (2024), KFF Health Tracking Poll May 2024: The Public’s Use And Views Of GLP-1 Drugs. Available at: https://www.kff.org/health-costs/kff-health-tracking-poll-may-2024-the-publics-use-and-views-of-glp-1-drugs/ (Accessed: 27 February 2026) ↑2 Future Market Insights Inc., (no date), Ozempic Market Share & Size 2025-2035. Available at: https://www.futuremarketinsights.com/reports/ozempic-market (Accessed: 27 February 2026) ↑3 Research And Markets, (2024), Global GLP-1 Receptor Agonist (Ozempic, Rybelsus, Trulicity, Saxenda, Wegovy, Victoza, Mounjaro, Zepbound) Market Analysis Report 2024-2030 Featuring Eli Lilly And C, Sanofi, Novo Nordisk, AstraZeneca. Available at: https://www.globenewswire.com/news-release/2024/07/17/2914462/28124/en/Global-GLP-1-Receptor-Agonist-Ozempic-Rybelsus-Trulicity-Saxenda-Wegovy-Victoza-Mounjaro-Zepbound-Market-Analysis-Report-2024-2030-Featuring-Eli-Lilly-and-C-Sanofi-Novo-Nordisk-Ast.html (Accessed: 27 February 2026) ↑4, ↑7, ↑10, ↑12 Kommu, S., Whitfield, P., (2025), Semaglutide. Available at: http://www.ncbi.nlm.nih.gov/books/NBK603723/ (Accessed: 27 February 2026) ↑5 Sorli, C., Harashima, S.I., Tsoukas, G.M. (2017). Efficacy And Safety Of Once-Weekly Semaglutide Monotherapy Versus Placebo In Patients With Type 2 Diabetes (SUSTAIN 1): A Double-Blind, Randomised, Placebo-Controlled, Parallel-Group, Multinational, Multicentre Phase 3a Trial. Lancet Diabetes And Endocrinology. 5(4). 251-260. Available at: https://doi.org/10.1016/S2213-8587(17)30013-X ↑6 Garvey, W.T., Batterham, R.L., Bhatta, M. (2022). Two-Year Effects Of Semaglutide In Adults With Overweight Or Obesity: The STEP 5 Trial. Nature Medicine. 28(10). 2083-2091. Available at: https://doi.org/10.1038/s41591-022-02026-4 ↑8 Holst, J.J. (2007). The Physiology Of Glucagon-Like Peptide 1. Physiological Reviews. 87(4). 1409-1439. Available at: https://doi.org/10.1152/physrev.00034.2006 ↑9 Drucker, D.J. (2022). GLP-1 Physiology Informs The Pharmacotherapy Of Obesity. Molecular Metabolism. 57. 101351. Available at: https://doi.org/10.1016/j.molmet.2021.101351 ↑11 Kommu, S., Berg, R.L. (2024). Efficacy And Safety Of Once-Weekly Subcutaneous Semaglutide On Weight Loss In Patients With Overweight Or Obesity Without Diabetes Mellitus-A Systematic Review And Meta-Analysis Of Randomized Controlled Trials. Obesity Reviews. 25(9). e13792. Available at: https://doi.org/10.1111/obr.13792 ↑13 Center For Drug Evaluation And Research, (2024), July – September 2023 | Potential Signals Of Serious Risks/New Safety Information Identified By The FDA Adverse Event Reporting System (FAERS). Available at: https://www.fda.gov/drugs/fdas-adverse-event-reporting-system-faers/july-september-2023-potential-signals-serious-risksnew-safety-information-identified-fda-adverse (Accessed: 02 March 2026) ↑14, ↑15 Alzahrani, W.S., Bahkali, S.A., Alharthy, R.F., Alsabban, A.S. (2025). Alopecia Areata Following Semaglutide Treatment For Weight Loss: A Case Report. JAAD Case Reports. 63. 44-46. Available at: https://doi.org/10.1016/j.jdcr.2025.06.012 ↑16 Cheng, J.R., Zheng, J., Li, Y. (2022). Weight Loss-Associated Alopecia Areata. American Journal Of Therapeutics. Available at: https://doi.org/10.1097/MJT.0000000000001851 ↑17 Godfrey, H., Leibovit-Reiben, Z., Jedlowski, P., Thiede, R. (2025). Alopecia Associated With The Use Of Semaglutide And Tirzepatide: A Disproportionality Analysis Using The FDA Adverse Event Reporting System (FAERS) From 2022 To 2023. Journal Of The European Academy Of Dermatology And Venereology. 39(2). e153. Available at: https://doi.org/10.1111/jdv.20197 ↑18 Burke, O., Sa, B., Alvarez Cespedes, D., Sechi, A., & Tosti, A. (2025). Glucagon-like peptide-1 receptor agonist medications and hair loss: a retrospective cohort study. Journal of the American Academy of Dermatology. 92(5). 1141–1143. Available at: https://doi.org/10.1016/j.jaad.2025.01.046 ↑19 Akiska, Y.M., Vidal, S.I., Menta, N. (2025). Increased Incidence And Risk Of Hair Loss With Glucagon-Like Peptide 1 Receptor Agonists: A Real-World Multicentre Cohort Study. EMJ. 13(1). 52-54. Available at: https://doi.org/10.33590/emjdermatol/TYEW1122 ↑20 Hughes, E.C., Syed, H.A., Saleh, D., (2025), Telogen Effluvium. Available at: http://www.ncbi.nlm.nih.gov/books/NBK430848/ (Accessed: 02 March 2026) ↑21 Kang, D.H., Kwon, S.H., Sim, W.Y., Lew, B.L. (2024). Telogen Effluvium Associated With Weight Loss: A Single Center Retrospective Study. Annals Of Dermatology. 36(6). 384-388. Available at: https://doi.org/10.5021/ad.24.043 ↑22 Zhang, W., Fan, M., Wang, C. (2021). Hair Loss After Metabolic And Bariatric Surgery: A Systematic Review And Meta-Analysis. Obesity Surgery. 31(6). 2649-2659. Available at: https://doi.org/10.1007/s11695-021-05311-2 ↑23 Johnson, B., Milstead, M., Thomas, O. (2025). Investigating Nutrient Intake During Use Of Glucagon-Like Peptide-1 Receptor Agonist: A Cross-Sectional Study. Frontiers In Nutrition. 12. 1566498. Available at: https://doi.org/10.3389/fnut.2025.1566498 ↑24 List, J.F., He, H., Habener, J.F. (2006). Glucagon-Like Peptide-1 Receptor And Proglucagon Expression In Mouse Skin. Regulatory Peptides. 134(2). 149-157. Available at: https://doi.org/10.1016/j.regpep.2006.02.007 With so many hair loss treatments on the market, finding the best one to treat female pattern hair loss (androgenic alopecia) can feel overwhelming. But what really is “best”? The best hair loss treatment is one that is effective, safe, and suitable for long-term use, but these standards can vary from person to person. This is particularly true for women, where menopause and family planning intentions play a part in treatment safety and efficacy.
In this article, we evaluate hair loss treatments for female pattern hair loss based on their quality of evidence, effectiveness, and broad suitability. We list the 6 best hair loss treatments for women, guiding you through how each treatment works, the clinical evidence for it, who the ideal candidates are, and who it’s safe for, so that you can decide on the most suitable treatment going forward.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Defining What Is “Best”
“Best” is hard to define. So many different treatments can be the best in different ways: safest, most effective, most affordable, or most tolerated.
What is best is highly personal and depends on each person’s circumstances, such as the severity of hair loss, lifestyle, age, and family-planning intentions. All of these factors affect the degree of regrowth, effectiveness, and safety of each treatment.
At Perfect Hair Health, we evaluate treatments using three metrics: evidence quality, regrowth potential, and long-term viability.
- Evidence quality: Assesses the quality and totality of evidence. Studies that use humans in randomized, blinded, and controlled clinical trials are ranked highest for evidence quality, while anecdotal evidence ranks lowest.
- Regrowth potential: Assesses how much hair regrowth can be expected based on hair count (particularly terminal hairs) and hair thickness improvements demonstrated in human clinical trials.
- Long-term viability: Assesses treatment adherence over time, i.e., do people keep up with daily administration of the treatment?
For this article, we define “best” by considering 6 treatments that have scored highest in evidence quality and regrowth potential.
This is not a best-to-worst list. Each treatment may be more or less suitable depending on personal circumstances, and please note that many of these treatments are not suitable for women who are already pregnant or breastfeeding.
6 Best Hair Loss Treatments for Women
#1 Topical Minoxidil
Evidence quality score – 97/100
Topical minoxidil is currently the only FDA-approved treatment for female pattern hair loss. It is considered the gold-standard treatment, and typically comes as a topical solution or foam at doses of 2% or 5%. Higher doses are also available, but these are not FDA-approved.
Ideal for:
- Women looking for an over-the-counter solution
- Women looking for an FDA-approved solution
- Women who can commit to daily topical application
- Women with localized thinning
Safe for:
- Women of reproductive age, as long as they are not pregnant, trying to conceive, or breastfeeding, and have effective contraception in place
- Perimenopausal women
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women who are pregnant, trying to conceive, or breastfeeding
- Women with scalp hypersensitivity
- Women with damaged or inflamed scalps
- Women who own cats
How Does It Work?
The action of minoxidil against hair loss is not clear-cut. It is known to work through a variety of biological mechanisms.
- Local vasodilation: Minoxidil can promote increased blood flow.[1]Sica, D.A. (2004). Minoxidil: An Underused Vasodilator For Resistant Or Severe Hypertension. J Clin Hypertens (Greenwich). 6(5). 283-287. Available at: https://doi.org/10.1111/j.1524-6175.2004.03585.x When applied topically, this may enhance conditions favorable for growth at hair follicles.
- VEGF up-regulation: Vascular endothelial growth factor (VEGF) mediates angiogenesis (the formation of new blood vessels), which may support hair growth.[2]Zeltzer, A.A., Keren, A., Paus, R., Gilhar, A. (2024). Topical Minoxidil Rejuvenates Hair Follicles From Men With Androgenetic Alopecia In Vivo. Acta Dermato Venereologica. 104(24213). Available at: … Continue reading
- Anagen induction and telogen shortening: Minoxidil can induce the anagen phase (growth phase) of hair, and shorten the resting phase (telogen phase).[3]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study … Continue reading
- Wnt/β-catenin activation: The signalling pathway Wnt/β-catenin is essential for hair follicle development, regeneration, and maintenance.[4]Kwack, M.H., Kang, B.M., Kim, M.K., Kim, J.C., Sung, Y.K. (2011). Minoxidil Activates Beta-Catenin Pathway In Human Dermal Papilla Cells: A Possible Explanation For Its Anagen Prolongation Effect. … Continue reading
- Prostaglandin E2 elevation: Prostaglandin E2 is associated with hair growth, possibly by protecting the follicle environment from inflammation.[5]Michelet, J.F., Commo, S., Billoni, N., Mahe, Y.F., Bernard, B.A. (1997). Activation Of Cytoprotective Prostaglandin Synthase-1 By Minoxidil As A Possible Explanation For Its Hair Growth-Stimulating … Continue reading
The Clinical Evidence
There is an extensive number of high-quality clinical trials showing that minoxidil between 2% and 5% can promote hair regrowth.[6]Olsen, E.A., DeLong, E.R., Weiner, M.S. (1987). Long-Term Follow-Up of Men with Male Pattern Baldness Treated with Topical Minoxidil. Journal of the American Academy of Dermatology. 16(3). 688–695. … Continue reading,[7]Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the … Continue reading,[8]Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta … Continue reading[9]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study … Continue reading It’s important to note here that dosing and application matter – for women with female pattern hair loss, clinical trials show that once-daily application of 5% minoxidil can be just as effective as twice-daily use of 2% minoxidil in improving hair regrowth and hair counts.[10]Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., Bartels, N.G. (2011). A Randomized, Single-Blind Trial Of 5% Minoxidil Foam Once Daily Versus 2% Minoxidil Solution Twice Daily In The … Continue reading
For most users of topical minoxidil, substantial improvements in hair growth can be seen between 3 and 6 months. But after more than a year, regrowth rates decline.[11]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study … Continue reading
Why does this decline happen? It’s because minoxidil does not target the androgen hormone dihydrotestosterone (DHT). DHT impacts hair growth, and high levels can cause miniaturization of the hair follicles that ultimately leads to weaker, finer, and non-growing hair. DHT is not only the primary cause of androgenic alopecia, but the levels of this hormone are often higher in those with this condition.[12]Ustuner, E. T. (2013). Cause of Androgenic Alopecia: Crux of the Matter. Plast Reconstr Surg Glob Open. 1(7). e64. Available at: https://doi.org/10.1097/GOX.0000000000000005,[13]Sekhavat, H., Bar Yehuda, S., Asotra, S. (2025). Using the Mechanisms of Action Involved in the Pathogenesis of Androgenetic Alopecia to Treat Hair Loss. Int J Mol Sci. 26(21). 10712. Available at: … Continue reading
Because minoxidil does not target DHT, it does not block the effects of DHT, and therefore hair shafts still become thinner over time. However, that doesn’t mean you have to stop using minoxidil. Minoxidil can be supplemented with other therapies, like tretinoin and microneedling, that enhance penetration and revitalize regrowth.[14]Shin, H.S., Won, C.H., Lee, S.H., Kwon, O.S., Kim, K.H., Eun, H.C. (2007). Efficacy of 5% Minoxidil Versus Combined 5% Minoxidil and 0.01% Tretinoin for Male Pattern Hair Loss. American Journal of … Continue reading,[15]Zhang, Y., Sheng, Y., Zeng, Y., Hu, R., Zhao, J., Wang, W., Yang, Q. (2022). Randomized Trial Of Microneedling Combined With 2% Minoxidil Topical Solution For The Treatment Of Female Pattern Hair … Continue reading
Interested in how to enhance results with minoxidil? Read our article.
Figure 3: The scalp of a female patient at baseline (A) and after being treated with 2% minoxidil and microneeding for 24 weeks (B). Adapted from Figure 1.[16]Zhang, Y., Sheng, Y., Zeng, Y., Hu, R., Zhao, J., Wang, W., Yang, Q. (2022). Randomized Trial Of Microneedling Combined With 2% Minoxidil Topical Solution For The Treatment Of Female Pattern Hair … Continue reading Image used under the Creative Commons License.
Anecdotal evidence from our PHH members shows that 7% minoxidil with enhancers like tretinoin often improves hair regrowth outcomes.
Safety & Side Effects
Scalp Irritation
Topical minoxidil can cause dermatitis in 2-6% of users. But this is mainly due to propylene glycol, a harmful additive common in many topical minoxidil products. It’s added to enhance the dissolution and penetration of minoxidil, but it can cause itching, redness, and sometimes swelling.[17]Jadeed, H.B., Almudimeegh, A.M., Alomran, S.A., Alshathry, A.H. (2021). A Case Of Contact Allergic Dermatitis To Topical Minoxidil. Cureus. 13(1). E12510. Available at: … Continue reading
Topical minoxidil formulations may also include alcohols, which can again irritate the scalp and cause dryness and flaking.
Systemic absorption
As with any topical treatment, systemic absorption (whole body absorption) is possible, and this can cause side effects seen in those taking oral minoxidil. We’ll get to this next.
Pregnancy
There is no direct evidence linking topical minoxidil to reduced fertility or conception. However, there are a few cases where adverse events during pregnancy have occurred, and there isn’t enough evidence to confidently show that it is safe.[18]Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., Moroni, F. (2003). Topically Applied Minoxidil May Cause Fetal Malformation: A Case Report. Birth Defects Research Part A: Clinical And … Continue reading,[19]Differences In Reproductive Toxicology Between Alopecia Drugs: An Analysis On Adverse Events Among Female And Male Cases. (no year). Oncotarget. 7(50). 82074-82084. Available at: … Continue reading So, it is generally not recommended for women who are planning to become pregnant, are pregnant, or are breastfeeding. You can read more about this here.
Pets
In addition to human side effects, minoxidil is highly toxic to cats, so best avoided for cat lovers.[20]DeClementi, C., Bailey, K.L., Goldstein, S.C., Orser, M.S. (2004). Suspected Toxicosis After Topical Administration Of Minoxidil In 2 Cats. Veterinary Emergency & Critical Care. 14(4). 287-292. … Continue reading
#2 Oral Minoxidil
Evidence quality score – 63/100
Oral minoxidil is just that – an oral form of minoxidil that is often taken in doses between 0.25 mg and 5 mg daily. It is an off-label treatment for female pattern hair loss.
Ideal for:
- Women who do not respond well to topical minoxidil or find adherence difficult
- Women looking for ease of use
- Women who are comfortable with unwanted hair growth outside of the scalp
- Women with diffuse thinning
Safe for:
- Women of reproductive age, as long as they are not pregnant, trying to conceive, or breastfeeding, and have effective contraception in place
- Perimenopausal women
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women who are pregnant, trying to conceive, or breastfeeding
- Women with pre-existing heart, liver, or kidney issues
Figure 4: Ulo oral minoxidil.
How Does It Work?
This form of minoxidil works in the same way as topical minoxidil, but the effects are not localized to the scalp since oral minoxidil is systemically absorbed.
Users may consider oral minoxidil if they are not seeing benefits from topical minoxidil. The effectiveness of topical minoxidil depends on scalp penetration, i.e., whether it actually reaches the hair follicle. Studies show that only 1.4% of applied minoxidil is absorbed through skin.[21]Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2022). Minoxidil: A Comprehensive Review. Journal Of Dermatological Treatment. 33(4). 1896-1906. Available at: … Continue reading
Absorption can be influenced by the delivery vehicle (e.g., whether it is a foam, alcohol-based solution, etc.), the integrity of the skin barrier, and the presence of certain biological components (specifically the sulfotransferase enzyme SULT1A1) that transforms minoxidil into minoxidil sulfate, its active form.[22]Sattur, S., Talathi, A., Shetty, G., Arsiwala, S., Pereira, R., Dhoot, D. (2023). Comparative Clinical Study Evaluating The Efficacy And Safety Of Topical 5% Cetosomal Minoxidil And Topical 5% … Continue reading Since the activity of SULT1A1 at the scalp varies between individuals, factors like this can also impact treatment effectiveness.[23]Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A … Continue reading
So, for those who didn’t respond to topical minoxidil either due to inadequate penetration or SULT1A1 activity limitations, oral minoxidil offers a solution as it does not rely on penetration and can be activated in the liver, where SULT1A1 is abundant.
The Clinical Evidence
Even though it is not an FDA-approved treatment for female pattern hair loss, oral minoxidil at 1 mg taken daily has been found to be just as effective as 5% topical minoxidil in enhancing terminal hair counts after 24 weeks (i.e., hairs that contribute to the appearance of a fuller head of hair).[24]Ramos, M.P., Sinclair, R.D., Kasprzak, M., Miot, H.A. (2020). Minoxidil 1 Mg Oral Versus Minoxidil 5% Topical Solution For The Treatment Of Female-Pattern Hair Loss. Journal Of The American Academy … Continue reading
One retrospective study, which analysed the results of many cases of men with androgenic alopecia being treated with oral minoxidil, showed that approximately 90% of men showed substantial regrowth at 6-12 months after taking oral minoxidil between 2.5 mg and 5 mg.[25]Jiminiez-Couche, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral … Continue reading We can therefore presume these results would apply to women as well.
Overall, oral minoxidil is considered an effective medication for androgenic alopecia, and there is a plethora of clinical evidence to back this up (you can check out our other free articles on minoxidil to find out more information).[26]Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: … Continue reading,[27]Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global … Continue reading,[28]Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 … Continue reading,[29]Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). … Continue reading
Safety & Side Effects
The concern with oral minoxidil is not effectiveness, but safety. We mentioned previously that there is a risk of systemic absorption with topical minoxidil – this is something that happens with oral minoxidil, and can lead to a variety of safety concerns and side effects.
Cardiovascular Risk
Some people taking low-dose oral minoxidil have developed pericardial effusion (fluid around the heart) and pericarditis (inflammation around the heart).[30]Dlova, N.C., Jacobs, T., Singh, S. (2022). Pericardial, Pleural Effusion And Anasarca: A Rare Complication Of Low-Dose Oral Minoxidil For Hair Loss. JAAD Case Reports. 11(28). 94-96. Available at: … Continue reading,[31]Bentivegna, K., Zhou, A.E., Adalsteinsson, J.A., Sloan, B. (2022). Letter In Reply: Pericarditis And Peripheral Edema In A Healthy Man On Low-Dose Oral Minoxidil Therapy. JAAD Case Reports. 20(29). … Continue reading. But this is a risk, not an inevitability.
At higher doses, minoxidil may also change the electrical activity of the heart. This has been recorded in 60% to 90% of users, but often resolves with continued use or dose reduction.[32]Hall, D., Charocopos, F., Froer, K.L., Rudolph, W. (1979). ECG Changes During Long-Term Minoxidil Therapy For Severe Hypertension. Archives Of Internal Medicine. 139. 790-794. Available at: … Continue reading
Hypertrichosis
Hypertrichosis is excessive hair growth on unwanted areas of the body. One study reported that users of 1 mg minoxidil have reported facial and body hypertrichosis in 32% and 47% of cases.[33]Bloch, D.L., Carlos, R.M.D. (2025). Side Effects’ Frequency Assessment Of Low Dose Oral Minoxidil In Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: … Continue reading
You can read more about how to avoid hypertrichosis when using minoxidil here.
Edema
In one study, lower leg edema (swelling) has been reported in 75% of cases in those taking 1 mg minoxidil, while periorbital (eye) edema has been reported in 50% of cases of those taking 1 mg and 50% of cases in those moving from 2 mg to 2.5 mg.[34]Bloch, D.L., Carlos, R.M.D. (2025). Side Effects’ Frequency Assessment Of Low Dose Oral Minoxidil In Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: … Continue reading
Managing Risk
If oral minoxidil feels like the right option for you but is causing some unwanted side effects, you can try a lower dose (and gradually work to higher doses), splitting the daily dose between morning and evening, or taking the medication sublingually (letting it dissolve under your tongue).
Interested in Oral Minoxidil?
Low-dose oral minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

#3 Oral Spironolactone
Evidence quality score – 49/100
Spironolactone is another off-label treatment for female pattern hair loss. It was originally designed to treat hypertension, heart failure, and edema, but research shows oral spironolactone between 25 mg and 200 mg per day could also provide hair regrowth benefits.
Ideal for:
- Women who have struggled with the side effects of oral minoxidil
- Women looking to supplement topical minoxidil
- Women looking for ease of use
- Women who are comfortable with the potential for irregular periods
Safe for:
- Women of reproductive age, as long as they are not pregnant, trying to conceive, or breastfeeding, and have effective contraception in place
- Perimenopausal women with effective contraception in place
- Menopausal women who are not at risk of hyperkalemia
- Postmenopausal women who are not at risk of hyperkalemia
Not recommended for:
- Women who are pregnant or trying to conceive
- Perimenopausal women without effective contraception in place
- Women at any stage of life who are at risk of hyperkalemia
How Does It Work?
Spironolactone targets DHT, the primary cause of female pattern hair loss. It does this in two ways:
- It binds and blocks androgen receptors.[35]Corvol, P., Michaud, A., Menard, J., Freifeld, M., Mahoudeau, J. (1975). Antiandrogenic Effect Of Spirolactones: Mechanism Of Action. Endocrinology. 97(1). 52-58. Available at: … Continue reading
- It interferes with the production of androgen hormones.[36]Menard, R.H., Stripp, B., Gillette, J.R. (1974). Spironolactone And Testicular Cytochrome P-450: Decreased Testosterone Formation In Several Species And Changes In Hepatic Drug Metabolism. … Continue reading,[37]Kossor, D.C., Kominami, S., Takemori, S., Colby, H.D. (1992). Destruction Of Testicular Cytochrome P-450 By 7α-Thiospironolactone Is Catalyzed By The 17α-Hydroxylase. The Journal Of Steroid … Continue reading
The binding of DHT to androgen receptors activates the process of hair loss. So by blocking DHT binding, spironolactone helps reduce follicle miniaturization.
Spironolactone has been shown to reduce the production of androgen hormones like testosterone. It hasn’t been shown to reduce the production of DHT, but DHT is derived from testosterone, so it’s possible (but not proven) that spironolactone could limit the conversion of testosterone to DHT.
The Clinical Evidence
So far, there is clinical evidence for oral spironolactone, but quality studies are lacking. The overall conclusions from the use of 25-50 mg/day spironolactone for female pattern hair loss suggest that daily intake could reduce hair loss severity.[38]Sinclair, R.D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. International Journal of Dermatology. 57(1). … Continue reading,[39]Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: … Continue reading However, the methodologies of these studies have several caveats:
- There were no placebo controls.
- Hair density was determined visually.
- Hair shedding was self-reported in some cases.
- Only one of these studies is a clinical trial, the other is a retrospective study.
So, whether oral spironolactone at this level is actually effective for treating hair loss is not shown by robust research yet. There is, however, a greater number of studies showing the use of spironolactone between 100 mg and 200 mg per day, particularly in conjunction with minoxidil.[40]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading,[41]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading,[42]Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: … Continue reading,[43]Hoedemaker, C., Van Egmond, S., Sinclair, R. (2007). Treatment of female pattern hair loss with a combination of spironolactone and minoxidil. Australasian Journal of Dermatology. 48(1). 43–45. … Continue reading
Figure 5: Images of hair density at baseline and following 24 weeks of daily treatment with either 100 mg spironolactone and 3% topical minoxidil or a placebo and topical 3% minoxidil. Adapted from Figure 5.[44]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading Image used under the Creative Commons License.
These studies are not without limitations themselves. Some of them do not count terminal hair, some of them show hair changes that are not statistically significant, some have small participant numbers, and some are case studies, not clinical trials. But overall, most report positive outcomes for the use of oral spironolactone with topical minoxidil, showing that this combination could improve the following more effectively than topical minoxidil alone:
- Terminal hair count
- Total hair count
- Hair diameter
This suggests that oral spironolactone could be a promising treatment option for those looking to enhance hair regrowth with minoxidil.
Safety & Side Effects
Oral spironolactone is not without its side effects, but they don’t always occur, as doses of 200 mg/day have been safely administered with no adverse effects reported.[45]Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: … Continue reading However, there is a common set of side effects reported that are worth considering before starting this medication.
Menstrual and Fertility Problems
Irregular menstruation has been reported in 15-37% of women taking oral spironolactone.[46]Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English … Continue reading,[47]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading It may also cause temporary fertility. However, menstruation and fertility should return to normal after 2 months of stopping.[48]NHS, (no date), Pregnancy, Breastfeeding And Fertility While Taking Spironolactone. Available at: … Continue reading
Hyperkalemia
Hyperkalemia is when there is too much potassium in the blood. Those taking medium doses of oral spironolactone have reported hyperkalemia, which can impact nerve and muscle function.[49]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading,[50]Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: … Continue reading,[51]Collins, M.S., Ali, S., Wiss, I.P., Senna, M.M. (2023). Retrospective Analysis Of The Risk Of Hyperkalaemia In Women Older Than 65 Years Of Age Prescribed Spironolactone For Female-Pattern Hair Loss. … Continue reading Hyperkalemia risk increases with age.[52]Sevamontree, C., Jintajirapan, S., Phakdeekitcharoen, P., Phakdeekitcharoen, B. (2024). The Prevalence And Risk Factors Of Hyperkalemia In The Outpatient Setting. Int J Nephrol. 2024. 5694131. … Continue reading
Edema
This may seem paradoxical since spironolactone treats edema, but spironolactone can also cause edema because it promotes shifts in fluids from the bloodstream to the tissues.[53]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading
Pregnancy
Because oral spironolactone interferes with DHT everywhere in the body, it is not recommended for those who are planning on pregnancy or are pregnant. DHT is a hormone required for male development, so those taking spironolactone are at risk of causing harm to male fetuses. Talk to your doctor if you’re considering becoming pregnant while taking spironolactone.
Additional Side Effects
Fewer than 5% of women report breast tenderness, reduced libido, nausea, headache, and fatigue when taking oral spironolactone.[54]Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English … Continue reading,[55]Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: … Continue reading
#4 Topical Finasteride
Evidence quality score – 69/100
Oral finasteride is FDA-approved for male pattern hair loss, but it is not recommended for women due to its potential side effects and harm during pregnancy. Topical finasteride provides a solution to those who do not tolerate, or who can not take, oral finasteride.
Ideal for:
- Women who have not seen results from topical or oral minoxidil
- Women who can commit to daily topical application
- Women with localized thinning
- Women more willing to risk side effects
Safe for:
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women of reproductive age, who are pregnant, trying to conceive, or breastfeeding
- Perimenopausal women
- Women with scalp hypersensitivity
- Women with damaged or inflamed scalps
How Does It Work?
Like spironolactone, finasteride blocks DHT. But it doesn’t block DHT in the same way. Topical finasteride inhibits the type II version of the enzyme that forms DHT, known as 5-ɑ-reductase type II, to reduce DHT production at the scalp.[56]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading,[57]Shapiro, J., & Kaufman, K. D. (2003). Use of finasteride in the treatment of men with androgenetic alopecia (male pattern hair loss). *Journal of Investigative Dermatology Symposium Proceedings.* … Continue reading.
The Clinical Evidence
Research for topical finasteride is still in the early stages, and most of the data is from men. But if we consider that the data may be generalizable to women, the research shows that topical finasteride can produce similar hair regrowth when compared to oral finasteride.
- Medium doses: Topical finasteride at 0.25% has shown to be effective for the treatment of androgenic alopecia, supporting the steady improvement of hair thickness, hair counts, and an overall reduction in hair loss comparable to oral finasteride.[58]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading,[59]Gupta, A.K., Talukder, M. (2022). Topical Finasteride For Male And Female Pattern Hair Loss: Is It A Safe And Effective Alternative? Journal of Cosmetic Dermatolology. 21(5). 1841-1848. Available at: … Continue reading
- High doses: Doses of 1% can provide improvements in hair count equivalent to oral finasteride.[60]Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing The Therapeutic Effects Of Finasteride Gel And Tablet In Treatment Of The Androgenetic Alopecia. Indian J Dermatol Venereol … Continue reading
Mean change in hair count from baseline to week 24 of treatment with 0.25% topical finasteride, 1 mg oral finasteride, or a placebo. Adapted from Figure 3.[61]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading Image used under the Creative Commons License.
Outside of direct comparison between delivery methods, low doses of finasteride at 0.005% to 0.1% have also been shown to improve hair growth outcomes, particularly when combined with other hair loss treatments.[62]Mazzarella, G., Loconsole, G., Cammisa, G., Mastrolonardo, G., & Vena, G. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy … Continue reading,[63]Tanglertsampan C. Efficacy and safety of 3% minoxidil versus combined 3% minoxidil / 0.1% finasteride in male pattern hair loss: a randomized, double-blind, comparative study. J Med Assoc Thai. 2012 … Continue reading
Doses of 0.5% finasteride along with 2% minoxidil have also been shown to improve female pattern hair loss after 18 months of treatment, although this was based on visual analysis. While visual analysis can show visible improvements in hair growth, it does not provide objective data and is therefore less robust.[64]Rossi, A., Magri, F., D’Arino, A., Pigliacelli, F., Muscianese, M., Leoncini, P., Caro, G., Federico, A., Fortuna, M.C., Carlesimo, M. (2020). Efficacy Of Topical Finasteride 0.5% Vs … Continue reading Nonetheless, the clinical promise of topical finasteride is evident, but requires more randomized, blinded, and controlled clinical trials.
Safety & Side Effects
Oral finasteride may be effective, but it comes with several serious side effects. Unfortunately, those using topical finasteride are not exempt from these effects, since finasteride can be absorbed systemically even when applied topically.
Systemic Absorption
Clinical trials with topical finasteride at 0.25% show that, despite localized application to the scalp, serum (blood) DHT levels could fall by 24% to 34.5%.[65]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading,[66]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading This shows that topical finasteride can be systemically absorbed. Systemically absorbed finasteride is associated with depression, sexual dysfunction, and breast changes.
In 2025, the FDA released a warning that no FDA-approved topical finasteride exists, and that the FDA has not evaluated their safety, effectiveness, or quality prior to marketing. 32 cases linked to topical finasteride were reported between 2019 and 2024, where reports describe adverse events that are consistent with those reported with oral finasteride. In other words, these cases involved adverse events associated with systemically absorbed topical finasteride.
Pregnancy
There is a reason that topical finasteride is FDA-approved for male pattern baldness, but not female pattern baldness. The risk of systemic absorption makes this treatment unsuitable for women who could become pregnant, because the DHT-blocking effects of finasteride can cause toxicity to male fetuses. The magnitude of serum DHT lowering is much higher for finasteride than spironolactone, despite the fact that both are DHT blockers. So, unlike spironolactone, this treatment is usually only recommended for menopausal and postmenopausal women where there is no risk of pregnancy.
Scalp Irritation
Trials with 0.25% finasteride show that topical application can cause pruritus (irritating sensations) and erythema (inflamed skin) in around 2% of users, but this is only a small amount.[67]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading,[68]Gupta, A.K., Talukder, M. (2022). Topical Finasteride For Male And Female Pattern Hair Loss: Is It A Safe And Effective Alternative? Journal of Cosmetic Dermatolology. 21(5). 1841-1848. Available at: … Continue reading
Mitigating Risks
Application of 0.005% finasteride has been found to promote hair growth, but not impact serum DHT levels.[69]Mazzarella, G.F., Loconsole, G.F., Cammisa, G.A., Mastrolonardo, G.M. and Vena, G., 1997. Topical Finasteride in the Treatment of Androgenic Alopecia: Preliminary Evaluations After a 16-Month Therapy … Continue reading So, lowering the dosage of finasteride used is one option to mitigate risk. But systemic absorption is not just about dosage; it’s also about the treatment formulations, volume, frequency, and length of application.
For example, a study comparing once-daily application of 0.25% finasteride and twice-daily application of 0.25% finasteride found that once-daily application lowered serum DHT levels by 20%, while twice-daily application lowered levels by 70%![70]Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., Palmieri, R. (2014). A Novel Finasteride 0.25% Topical Solution For Androgenetic Alopecia: Pharmacokinetics And Effects On Plasma Androgen … Continue reading
Our analysis of member-submitted lab tests and clinical data suggests that 1-2 mL of topical finasteride at 0.005% to 0.02% is the best combination to minimize systemic absorption while maintaining positive hair growth outcomes.
You can read more about maximizing regrowth while minimizing side effects from finasteride here.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

#5 Topical Dutasteride
Evidence quality score – 55/100
Dutasteride is an FDA-approved medication for benign prostate hyperplasia but is also an off-label medication for male androgenic alopecia. It is typically used in doses from 0.02% to 0.2%
Ideal for:
- Women who have not seen results from minoxidil or finasteride
- Women who can commit to daily topical application
- Women with localized thinning
- Women who are willing to take treatments with less clinical evidence but high potential
Safe for:
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women of reproductive age, or who are pregnant, trying to conceive, or breastfeeding
- Perimenopausal women
- Women with scalp hypersensitivity
- Women with damaged or inflamed scalps
How Does It Work?
Dutasteride is another DHT blocker. It inhibits both type I and type II 5-ɑ-reductase.[71]Arif, T., Dorjay, K., Adil, M., Sami, M., (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). Available at: 10.2174/157488471266617031011112 Dutasteride, applied topically, could therefore help to combat hair loss by reducing DHT levels on the scalp, and has the potential to be more effective than finasteride due to blocking more than one type of 5-ɑ-reductase.
The Clinical Evidence
While the mechanism of action of dutasteride points to a promising treatment for androgenic alopecia, the research is still in its early stages. The potential effectiveness of dutasteride as a hair loss treatment started emerging around 2020. So, there isn’t a lot of evidence.
But it’s not just a lack of evidence; we’re limited in quality evidence. If you’re interested in taking a deep dive into the flaws behind a key dutasteride study, read our article here. But to summarize, studies so far have used:
- Inconsistent methods – For example, inconsistent timings in product application, or changes in the size, shape, and location of areas chosen to measure regrowth, which make results less valid
- Dual therapies – Some of the studies only used topical dutasteride in combination with microneedling, which does not help us isolate the true effect of dutasteride.
However, in all studies, the conclusions from the use of 0.01% to 0.05% dutasteride are positive. These studies report that dutasteride localizes well to the scalp, minimally affects serum DHT, and effectively slows or stops hair loss.[72]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled … Continue reading,[73]Nada, E.A., Sharkawy, R.E., Maged, W.M., & Elmagd, M.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Southern Medical Journal, 22, 387-400,[74]Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martínez-Moreno, A., Ocampo-Garza, S.S., (2022). Microneedling Plus Topical … Continue reading,[75]Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M., (2025). Efficacy of Skin Patting and Iontophoresis With Dutasteride Gel in Male and … Continue reading In support of this, our members report that use of 0.01 to 0.02% dutasteride does not appear to regrow hair, but it does minimize the hair loss process.
Figure 7: Scalp images at Baseline (1A, 1B, 1E, 1F) and after 8 weeks treatment (1C, 1D, 1G, 1H) with a 6% dutasteride gel. Adapted from Figure 1.[76]Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M., (2025). Efficacy of Skin Patting and Iontophoresis With Dutasteride Gel in Male and … Continue reading Image used under the Creative Commons License.
Safety & Side Effects
As a DHT blocker and topical formula, many of the risks posed by dutasteride are similar to those of topical finasteride.
Systemic Absorption
Topical dutasteride application appears to only modestly reduce serum DHT levels – in other words, topical application has limited systemic absorption when used appropriately.[77]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled … Continue reading,[78]Nada, E.A., Sharkawy, R.E., Maged, W.M., & Elmagd, M.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Southern Medical Journal, 22, 387-400 Systemic absorption is associated with several side effects similar to finasteride, such as sexual dysfunction and breast changes, so minimizing this is a great benefit.
Pregnancy
While dutasteride may only modestly affect serum DHT, any change in serum DHT levels is considered inappropriate for use in women who hope to or can conceive. Again, this is due to the toxicity risk to male fetuses associated with blocking DHT.
Scalp Irritation
Scalp irritation has been reported with the use of topical dutasteride in a few cases.[79]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled … Continue reading
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

#6 Low-Level Laser Therapy (LLLT)
Evidence quality score – not evaluated
LLLT is a therapy using light. For women who aren’t suitable for traditional treatments, this therapy could potentially work as an alternative.
Ideal for:
- Women wanting to avoid pharmaceutical solutions
- Women willing to adhere to the usage of LLLT over a long period of time
- Women willing to pay to use LLLT over a long period of time
- Women willing to try more experimental methods
Safe for:
- Women of reproductive age
- Perimenopausal women
- Menopausal women
- Postmenopausal women
Not recommended for:
- Women who are pregnant or breastfeeding
How Does It Work?
These devices emit infrared and near-infrared light, which are naturally emitted by the sun. They often come in the form of a cap or headpiece that can be worn for long periods of time. Infrared and near-infrared light tend to elongate hair growth cycles and improve hair counts.
We’re not entirely sure of why this happens, but the proposed mechanism is that light waves in the infrared and near-infrared range stimulate molecules like cytochrome c oxidase (which drives energy production for hair growth and regulates th in the mitochondria of cells.
You may remember from school that mitochondria generate the energy required by a cell to function. It is thought that the stimulation by LLLT increases energy production and triggers genetic changes leading to enhanced cell growth and repair.[80]Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts And Bolts Of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available … Continue reading,[81]Lyu, K., Liu, X., Jiang, L., Chen, Y., Lu, J., Zhu, B., Liu, X., Li, Y., Wang, D., Li, S. (2022). The Functions And Mechanisms Of Low-Level Laser Therapy In Tendon Repair (Review). Frontiers in … Continue reading
So, in essence, LLLT directed at the scalp could promote hair follicle cells to grow.
The Clinical Evidence
So far, the clinical evidence for the use of LLLT technology on participants with androgenic alopecia has been reported as overwhelmingly positive.
- LLLT combs have been found to increase terminal hair counts, total hair counts, and hair strength compared to a control product.[82]Satino, J.L., Markou, M. (2003). Hair Regrowth And Increased Hair Tensile Strength Using The HairMax LaserComb For Low-Level Laser Therapy. International Journal of Cosmetic Surgery and Aesthetic … Continue reading,[83]Leavitt, M., Charles, G., Heyman, E. (2009). HairMax LaserComb® Laser Phototherapy Device In The Treatment Of Male Androgenetic Alopecia. Clinical Drug Investigation. 29(5). 283-292. Available at: … Continue reading
- LLLT hats have increased terminal and total hair counts as well as hair density compared to users of a control product or treatment.[84]Lanzafame, R.J., Blanche, R.R., Bodian, A.B., Chiacchierini, R.P., Fernandez-Obregon, A., Kazmirek, E.R. (2013). The Growth Of Human Scalp Hair Mediated By Visible Red Light Laser And LED Sources In … Continue reading,[85]Kim, H., Choi, J.W., Kim, J.Y., Shin, J.W., Lee, S.J., Huh, C.H. (2013). Low-Level Light Therapy For Androgenetic Alopecia: A 24-Week, Randomized, Double-Blind, Sham Device-Controlled Multicenter … Continue reading,[86]Friedman, S., Schnoor, P. (2017). Novel Approach To Treating Androgenetic Alopecia In Females With Photobiomodulation (Low-Level Laser Therapy). Dermatologic Surgery. 43(6). 856-867. Available at: … Continue reading
- A retrospective analysis of many studies concluded that LLLT is both safe and effective for the treatment of androgenic alopecia.[87]Avci, P., Gupta, G.K., Clark, J., Wikonkal, N., Hamblin, M.R. (2014). Low-Level Laser (Light) Therapy (LLLT) For Treatment Of Hair Loss. Lasers in Surgery and Medicine. 46(2). 144-151. Available at: … Continue reading
Figure 9: Non-vellus hair density macroimages at baseline and 6 months in one patient in the HairMax LaserComb® group. Adapted from Figure 1.[88]Leavitt, M., Charles, G., Heyman, E. (2009). HairMax LaserComb® Laser Phototherapy Device In The Treatment Of Male Androgenetic Alopecia. Clinical Drug Investigation. 29(5). 283-292. Available at: … Continue reading Image used under the Creative Commons License.
This has led to companies selling items like LLLT caps at extremely high prices – in the range of $200 to $3,000!
But what’s the truth? Let’s look deeper into the science.
Firstly, not all studies report positive results. A study comparing the use of LLLT and 5% minoxidil with a group using 5% minoxidil alone found that there was no difference in improvement between the groups.[89]Sondagar, D.M., Mehta, H.H., Agharia, R.S., Jhavar, M.K. (2023). Efficacy Of Low-Level Laser Therapy In Androgenetic Alopecia – A Randomized Controlled Trial. International Journal of … Continue reading
Secondly, let’s consider the timing of clinical trials. For most people, regardless of hair condition, hair shedding typically peaks from late summer to early autumn (August to September). This is because the number of hairs in the telogen phase is highest by September. Following the telogen phase, hair reaches the exogen phase, the phase where hair sheds and falls out.[90]Courtois, M., Loussouarn, G., Hourseau, S., Grollier, J.F. (1996). Periodicity In The Growth And Shedding Of Hair. British Journal of Dermatology. 134(1). 47-54,[91]Kunz, M., Seifert, B., Trüeb, R.M. (2009). Seasonality Of Hair Shedding In Healthy Women Complaining Of Hair Loss. Dermatology. 219(2). 105-110. Available at: https://doi.org/10.1159/000216832,[92]Liu, C., Yang, J., Qu, L., Gu, M., Liu, Y., Gao, J., Collaudin, C., Loussouarn, G. (2014). Changes In Chinese Hair Growth Along A Full Year. International Journal of Cosmetic Science. 36(6). 531-536. … Continue reading The reason this happens is likely a combination of factors – light-driven patterns, summer stressors such as ultraviolet and heat, and more frequent washing.
So what would happen if we designed a clinical study where participants with androgenic alopecia were split into two groups: group 1 that had to use an LLLT cap, and group 2 assigned a placebo cap with no lights. Let’s say we started the trial in July and finished in October. Remember – natural hair shedding will occur from August to September. We would likely see that:
- The LLLT therapy minimizes the effect of typical seasonal shedding in September.
- Group 2 would undergo the typical hair shedding in September.
So when we look at the results in October, there will likely be a big divide between the number of hairs in Group 1 and the number of hairs in Group 2. This divide would be bigger than if we designed the experiment mid-summer, for example.
We’re not saying that LLLT isn’t effective, but we are saying that the time a clinical trial happens could affect whether a result is statistically significant.
And even when results are statistically significant, there isn’t enough comparative data to confidently say whether hair regrowth with LLLT is as substantial as regrowth with the use of minoxidil, finasteride, or dutasteride.[93]Sondagar, D.M., Mehta, H.H., Agharia, R.S., Jhavar, M.K. (2023). Efficacy Of Low-Level Laser Therapy In Androgenetic Alopecia – A Randomized Controlled Trial. International Journal of … Continue reading,[94]Esmat, S.M., Hegazy, R.A., Gawdat, H.I., Abdel Hay, R.M., Allam, R.S., El Naggar, R., Moneib, H. (2017). Low Level Light-Minoxidil 5% Combination Versus Either Therapeutic Modality Alone In … Continue reading For now, LLLT is not a treatment that should be used in isolation; it should be used (for those that can afford it) with other medications.[95]Munck, A., Gavazzoni, M.F., Trüeb, R.M. (2014). Use Of Low-Level Laser Therapy As Monotherapy Or Concomitant Therapy For Male And Female Androgenetic Alopecia. International Journal Of Trichology. … Continue reading
These reasons may explain why effects from using LLLT in real-life often conflict with the clinical study outcomes we see advertised. However, for women where options are more limited, LLLT could provide another treatment route.
Safety & Side Effects
Scalp Irritation
Some users report erythema, pruritus, dry scalp, and acne or eczema after use of a LLLT therapy device.[96]Hamblin, M.R. (2019). Photobiomodulation For The Management Of Alopecia: Mechanisms Of Action, Patient Selection And Perspectives. Clinical, Cosmetic And Investigational Dermatology. 12. 669-678. … Continue reading,[97]Egger, A., Resnik, S.R., Aickara, D., Maranda, E., Kaiser, M., Wikramanayake, T.C., Jimenez, J.J. (2020). Examining The Safety And Efficacy Of Low-Level Laser Therapy For Male And Female Pattern Hair … Continue reading
Pregnancy
LLLT should be safe for most women, but there is a lack of safety data to confidently prove that this therapy is safe for pregnant or breastfeeding women.
Honorable Mentions
Some hair loss ingredients have a decent evidence score behind them, but lower regrowth and long-term viability scores. These ingredients generally have less clinical evidence and lower efficacy, so are most effective when used in conjunction with one of the “best” treatments, but they are largely safe for most women at any stage of their life. So, we think they deserve an honorable mention.
Topical Ingredient Evidence Quality Score Evidence Melatonin 66/100 Leave-on, topical melatonin has not only been found to be safe, but a synthesis of five clinical studies shows that it can increase hair density, reduce hair loss, and increase hair strength in those with androgenic alopecia.[98]Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., Elsner, P. (2012). Topical Melatonin for Treatment of Androgenetic Alopecia. Int J Trichology. 4(4). 236-245. Available at: … Continue reading Cetirizine 57/100 Topical 1% cetirizine can improve hair growth, hair density, and hair thickness in patients with androgenic alopecia. However, the evidence so far is limited, with no evidence of efficacy beyond 6 months of use.[99]Zaky, M.S., Abo Khodeir, H., Ahmed, H.-A., Elsaie, M.L., (2021). Therapeutic Implications of Topical Cetirizine 1% in Treatment of Male Androgenetic Alopecia: A Case-Controlled Study. Journal of … Continue reading,[100]Hossein Mostafa, D., Samadi, A., Niknam, S., Nasrollahi, S.A., Guishard, A., Firooz, A., (2021). Efficacy of Cetirizine 1% Versus Minoxidil 5% Topical Solution in the Treatment of Male Alopecia: A … Continue reading,[101]Bassiouny, E.A., El-Samanoudy, S.I., Abbassi, M.M., Nada, H.R., Farid, S.F., (2023). Comparison Between Topical Cetirizine With Minoxidil Versus Topical Placebo With Minoxidil in Female Androgenetic … Continue reading Saw palmetto 54/100 Saw palmetto may strengthen hair and could improve thickness, although more robust evidence with longer trials is needed.[102]Morganti, P., Fabrizi, G., James, B., Bruno, C. (1998). Effect of gelatin-cystine and serenoa repens extract on free radicals level and hair growth. Journal of Applied Cosmetology. 16, 57-64. … Continue reading,[103]Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. … Continue reading,[104]Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the … Continue reading Ketoconzole 50/100 A review summarising seven studies using topical ketoconazole concluded that ketoconazole appears to increase hair diameter and result in clinical improvement in those with androgenic alopecia.[105]Fields, J. R., Vonu, P. M., Monir, R. L., Schoch, J. J. (2020). Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review. Dermatol Ther. 33(1). e13202. Available at: … Continue reading Guidelines for Starting a New Treatment
Starting a new treatment is not a journey you should take on your own. When considering a new treatment, always:
- Speak to a doctor
- Track your progress
- Track side effects
- Look to switch if side effects occur
And remember that consistency matters most in hair loss treatments! Only regular, long-term use keeps hair follicles in the growth phase and prevents ongoing shedding. So, choosing the right method of delivery to suit your lifestyle and routines is key.
Final Thoughts
Choosing a hair loss treatment is deeply personal. Factors like age, pregnancy plans, menopause stage, health history, and treatment tolerance all shape what may be effective and safe for you. While some options have stronger evidence or broader suitability, there is no single “best” treatment for every woman. Instead, the goal is to find the approach that aligns with your stage in life, lifestyle, and long-term needs. Discuss options with a qualified clinician, monitor results, and adjust over time to maximize hair regrowth along with safety.
References[+]
References ↑1 Sica, D.A. (2004). Minoxidil: An Underused Vasodilator For Resistant Or Severe Hypertension. J Clin Hypertens (Greenwich). 6(5). 283-287. Available at: https://doi.org/10.1111/j.1524-6175.2004.03585.x ↑2 Zeltzer, A.A., Keren, A., Paus, R., Gilhar, A. (2024). Topical Minoxidil Rejuvenates Hair Follicles From Men With Androgenetic Alopecia In Vivo. Acta Dermato Venereologica. 104(24213). Available at: https://doi.org/10.2340/actadv.v104.24213 ↑3 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study Protocol. Skin Research And Technology. 26(4). 542-557. Available at: https://doi.org/10.1111/srt.12827 ↑4 Kwack, M.H., Kang, B.M., Kim, M.K., Kim, J.C., Sung, Y.K. (2011). Minoxidil Activates Beta-Catenin Pathway In Human Dermal Papilla Cells: A Possible Explanation For Its Anagen Prolongation Effect. Journal Of Dermatological Science. 154-159. Available at: https://doi.org/10.1016/j.jdermsci.2011.01.013 ↑5 Michelet, J.F., Commo, S., Billoni, N., Mahe, Y.F., Bernard, B.A. (1997). Activation Of Cytoprotective Prostaglandin Synthase-1 By Minoxidil As A Possible Explanation For Its Hair Growth-Stimulating Effect. Journal Of Investigative Dermatology. 108(2). 205-209. Available at: https://doi.org/10.1111/1523-1747.ep12334249 ↑6 Olsen, E.A., DeLong, E.R., Weiner, M.S. (1987). Long-Term Follow-Up of Men with Male Pattern Baldness Treated with Topical Minoxidil. Journal of the American Academy of Dermatology. 16(3). 688–695. Available at: https://doi.org/10.1016/S0190-9622(87)70089-9 ↑7 Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the Treatment of Androgenetic Alopecia in Men. Journal of the American Academy of Dermatology. 57(5). 767–774. Available at: https://doi.org/10.1016/j.jaad.2007.04.012 ↑8 Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta Dermatovenerologica Alpina, Pannonica et Adriatica. 25(3). 41–44. Available at: https://doi.org/10.15570/actaapa.2016.12 ↑9 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study Protocol. Skin Research and Technology. 26(4). 542–557. Available at: https://doi.org/10.1111/srt.12827 ↑10 Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., Bartels, N.G. (2011). A Randomized, Single-Blind Trial Of 5% Minoxidil Foam Once Daily Versus 2% Minoxidil Solution Twice Daily In The Treatment Of Androgenetic Alopecia In Women. Journal Of The American Academy Of Dermatology. 65(6). 1126-1134. Available at: https://doi.org/10.1016/j.jaad.2010.09.724 ↑11 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies With Topical Minoxidil 2% Or 5% In Male-Patterned Hair Loss Treated With Oral Finasteride Employing An Analytical And Exhaustive Study Protocol. Skin Research & Technology. 26(4). 542-557. Available at: https://doi.org/10.1111/srt.12827 ↑12 Ustuner, E. T. (2013). Cause of Androgenic Alopecia: Crux of the Matter. Plast Reconstr Surg Glob Open. 1(7). e64. Available at: https://doi.org/10.1097/GOX.0000000000000005 ↑13 Sekhavat, H., Bar Yehuda, S., Asotra, S. (2025). Using the Mechanisms of Action Involved in the Pathogenesis of Androgenetic Alopecia to Treat Hair Loss. Int J Mol Sci. 26(21). 10712. Available at: https://doi.org/10.3390/ijms262110712 ↑14 Shin, H.S., Won, C.H., Lee, S.H., Kwon, O.S., Kim, K.H., Eun, H.C. (2007). Efficacy of 5% Minoxidil Versus Combined 5% Minoxidil and 0.01% Tretinoin for Male Pattern Hair Loss. American Journal of Clinical Dermatology. 8(5). 285–290. Available at: https://doi.org/10.2165/00128071-200708050-00003 ↑15, ↑16 Zhang, Y., Sheng, Y., Zeng, Y., Hu, R., Zhao, J., Wang, W., Yang, Q. (2022). Randomized Trial Of Microneedling Combined With 2% Minoxidil Topical Solution For The Treatment Of Female Pattern Hair Loss In A Chinese Population. Journal Of Cosmetic Dermatology. 21(12). 6985-6991. Available at: https://doi.org/10.1111/jocd.15424 ↑17 Jadeed, H.B., Almudimeegh, A.M., Alomran, S.A., Alshathry, A.H. (2021). A Case Of Contact Allergic Dermatitis To Topical Minoxidil. Cureus. 13(1). E12510. Available at: https://doi.org/10.7759/cureus.12510 ↑18 Smorlesi, C., Caldarella, A., Caramelli, L., Di Lollo, S., Moroni, F. (2003). Topically Applied Minoxidil May Cause Fetal Malformation: A Case Report. Birth Defects Research Part A: Clinical And Molecular Teratology. 67(12). 997-1001. Available at: https://doi.org/10.1002/bdra.10095 ↑19 Differences In Reproductive Toxicology Between Alopecia Drugs: An Analysis On Adverse Events Among Female And Male Cases. (no year). Oncotarget. 7(50). 82074-82084. Available at: https://doi.org/10.18632/oncotarget.12617 ↑20 DeClementi, C., Bailey, K.L., Goldstein, S.C., Orser, M.S. (2004). Suspected Toxicosis After Topical Administration Of Minoxidil In 2 Cats. Veterinary Emergency & Critical Care. 14(4). 287-292. Available at: https://doi.org/10.1111/j.1476-4431.2004.04014.x ↑21 Gupta, A.K., Talukder, M., Venkataraman, M., Bamimore, M.A. (2022). Minoxidil: A Comprehensive Review. Journal Of Dermatological Treatment. 33(4). 1896-1906. Available at: https://doi.org/10.1080/09546634.2021.1945527 ↑22 Sattur, S., Talathi, A., Shetty, G., Arsiwala, S., Pereira, R., Dhoot, D. (2023). Comparative Clinical Study Evaluating The Efficacy And Safety Of Topical 5% Cetosomal Minoxidil And Topical 5% Alcohol-Based Minoxidil Solutions For The Treatment Of Androgenetic Alopecia In Indian Men. Cureus. 15(10). E46568. Available at: https://doi.org/10.7759/cureus.46568 ↑23 Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 Activity In Hair Follicle, A Prognostic Marker Of Response To The Minoxidil Treatment In Patients With Androgenetic Alopecia: A Review. Advances In Dermatology And Allergology. 39(3). 472-478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑24 Ramos, M.P., Sinclair, R.D., Kasprzak, M., Miot, H.A. (2020). Minoxidil 1 Mg Oral Versus Minoxidil 5% Topical Solution For The Treatment Of Female-Pattern Hair Loss. Journal Of The American Academy Of Dermatology. 82(1). 252-253. Available at: https://www.jaad.org/article/S0190-9622(19)32666-0/fulltext ↑25 Jiminiez-Couche, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Fernandez-Nieto, D., Vano-Galvan, S. (2019). Effectiveness And Safety Of Low-Dose Oral Minoxidil In Male Androgenetic Alopecia. 81(2). 648-649. Available at: https://doi.org/10.1016/j.jaad.2019.04.054 ↑26 Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: https://www.researchgate.net/profile/Suparuj-Lueangarun/publication/319006716_Efficacy_and_safety_of_oral_minoxidil_5_mg_daily_during_24-week_treatment_in_male_androgenetic_alopecia/links/5b46f1690f7e9b4637cde38b/Efficacy-and-safety-of-oral-minoxidil-5-mg-daily-during-24-week-treatment-in-male-androgenetic-alopecia.pdf ↑27 Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global photographic assessment. *Dermatology and Therapy.* 10(6). 1345–1357. Available at: https://doi.org/10.1007/s13555-020-00448-x ↑28 Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 mg versus 1 mg. 396–399. Available at: https://doi.org/10.1016/j.jaad.2022.01.017 ↑29 Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). 104–109. Available at: https://doi.org/10.1111/ijd.13838 ↑30 Dlova, N.C., Jacobs, T., Singh, S. (2022). Pericardial, Pleural Effusion And Anasarca: A Rare Complication Of Low-Dose Oral Minoxidil For Hair Loss. JAAD Case Reports. 11(28). 94-96. Available at: https://doi.org/10.1016/j.jdcr.2022.07.044 ↑31 Bentivegna, K., Zhou, A.E., Adalsteinsson, J.A., Sloan, B. (2022). Letter In Reply: Pericarditis And Peripheral Edema In A Healthy Man On Low-Dose Oral Minoxidil Therapy. JAAD Case Reports. 20(29). 110-111. Available at: https://doi.org/10.1016/j.jdcr.2022.08.057 ↑32 Hall, D., Charocopos, F., Froer, K.L., Rudolph, W. (1979). ECG Changes During Long-Term Minoxidil Therapy For Severe Hypertension. Archives Of Internal Medicine. 139. 790-794. Available at: https://doi.org/10.1001/archinte.1979.03630440052018 ↑33, ↑34 Bloch, D.L., Carlos, R.M.D. (2025). Side Effects’ Frequency Assessment Of Low Dose Oral Minoxidil In Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: https://doi.org/10.1159/000539969 ↑35 Corvol, P., Michaud, A., Menard, J., Freifeld, M., Mahoudeau, J. (1975). Antiandrogenic Effect Of Spirolactones: Mechanism Of Action. Endocrinology. 97(1). 52-58. Available at: https://doi.org/10.1210/endo-97-1-52 ↑36 Menard, R.H., Stripp, B., Gillette, J.R. (1974). Spironolactone And Testicular Cytochrome P-450: Decreased Testosterone Formation In Several Species And Changes In Hepatic Drug Metabolism. Endocrinology. 94(6). 1628-1636. Available at: https://doi.org/10.1210/endo-94-6-1628 ↑37 Kossor, D.C., Kominami, S., Takemori, S., Colby, H.D. (1992). Destruction Of Testicular Cytochrome P-450 By 7α-Thiospironolactone Is Catalyzed By The 17α-Hydroxylase. The Journal Of Steroid Biochemistry And Molecular Biology. 42(3–4). 421-424. Available at: https://doi.org/10.1016/0960-0760(92)90147-B ↑38 Sinclair, R.D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. International Journal of Dermatology. 57(1). 104–109. Available at: https://doi.org/10.1111/ijd.13838 ↑39, ↑50, ↑55 Devjani, S., Ezemma, O., Jothishankar, B., Saberi, S., Kelley, K.J., Senna, M.M. (2024). Efficacy Of Low-Dose Spironolactone For Hair Loss In Women. 23(3). E91. Available at: https://jddonline.com/?post_type=article&p=15021 ↑40, ↑44, ↑47 Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, double-blind, placebo-controlled, parallel-group pilot study. International Journal of Women’s Dermatology. 11(3). e227. Available at: https://doi.org/10.1097/JW9.0000000000000227 ↑41, ↑49, ↑53 Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A Prospective, Single-Center, Parallel-Group, Evaluator Blinded, Randomized Trial. Frontiers in Medicine. 9. Article 905140. Available at: https://doi.org/10.3389/fmed.2022.905140 ↑42, ↑45 Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: https://doi.org/10.1046/j.1365-2265.1997.3761162.x ↑43 Hoedemaker, C., Van Egmond, S., Sinclair, R. (2007). Treatment of female pattern hair loss with a combination of spironolactone and minoxidil. Australasian Journal of Dermatology. 48(1). 43–45. Available at: https://doi.org/10.1111/j.1440-0960.2007.00332.x ↑46, ↑54 Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English Edition). 111(8). 639–649. Available at: https://doi.org/10.1016/j.adengl.2020.03.015 ↑48 NHS, (no date), Pregnancy, Breastfeeding And Fertility While Taking Spironolactone. Available at: https://www.nhs.uk/medicines/spironolactone/pregnancy-breastfeeding-and-fertility-while-taking-spironolactone/ (Accessed: 20 February 2026) ↑51 Collins, M.S., Ali, S., Wiss, I.P., Senna, M.M. (2023). Retrospective Analysis Of The Risk Of Hyperkalaemia In Women Older Than 65 Years Of Age Prescribed Spironolactone For Female-Pattern Hair Loss. British Journal Of Dermatology. 188(3). 429-447. Available at: https://doi.org/10.1093/bjd/ljac068 ↑52 Sevamontree, C., Jintajirapan, S., Phakdeekitcharoen, P., Phakdeekitcharoen, B. (2024). The Prevalence And Risk Factors Of Hyperkalemia In The Outpatient Setting. Int J Nephrol. 2024. 5694131. Available at: https://doi.org/10.1155/2024/5694131 ↑56 Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. Journal of the American Academy of Dermatology. 55(6). 1014–1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007 ↑57 Shapiro, J., & Kaufman, K. D. (2003). Use of finasteride in the treatment of men with androgenetic alopecia (male pattern hair loss). *Journal of Investigative Dermatology Symposium Proceedings.* 8(1). 20–23. Available at: https://doi.org/10.1046/j.1523-1747.2003.12167.x ↑58, ↑61, ↑65, ↑67 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology and Venereology. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑59, ↑68 Gupta, A.K., Talukder, M. (2022). Topical Finasteride For Male And Female Pattern Hair Loss: Is It A Safe And Effective Alternative? Journal of Cosmetic Dermatolology. 21(5). 1841-1848. Available at: https://doi.org/10.1111/jocd.14895 ↑60 Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing The Therapeutic Effects Of Finasteride Gel And Tablet In Treatment Of The Androgenetic Alopecia. Indian J Dermatol Venereol Leprol. 75(1). 47-51. Available at: https://doi.org/10.4103/0378-6323.45220 ↑62 Mazzarella, G., Loconsole, G., Cammisa, G., Mastrolonardo, G., & Vena, G. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy course. Journal of Dermatological Treatment, 8(3), 189–192. https://doi.org/10.3109/09546639709160517 ↑63 Tanglertsampan C. Efficacy and safety of 3% minoxidil versus combined 3% minoxidil / 0.1% finasteride in male pattern hair loss: a randomized, double-blind, comparative study. J Med Assoc Thai. 2012 Oct;95(10):1312-6. Available at: http://www.jmatonline.com/PDF/1312-1316-PB-95-10.pdf ↑64 Rossi, A., Magri, F., D’Arino, A., Pigliacelli, F., Muscianese, M., Leoncini, P., Caro, G., Federico, A., Fortuna, M.C., Carlesimo, M. (2020). Efficacy Of Topical Finasteride 0.5% Vs 17α-Estradiol 0.05% In The Treatment Of Postmenopausal Female Pattern Hair Loss: A Retrospective, Single-Blind Study Of 119 Patients. Dermatol Pract Concept. 10(2). e2020039. Available at: https://doi.org/10.5826/dpc.1002a39 ↑66 Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. International Journal of Clinical Pharmacology and Therapeutics. 54(1). 19-27. Available at: https://doi.org/10.5414/CP202467 ↑69 Mazzarella, G.F., Loconsole, G.F., Cammisa, G.A., Mastrolonardo, G.M. and Vena, G., 1997. Topical Finasteride in the Treatment of Androgenic Alopecia: Preliminary Evaluations After a 16-Month Therapy Course. Journal Of Dermatological Treatment, 8(3), pp.189–192. Available at: https://doi.org/10.3109/09546639709160517 ↑70 Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., Palmieri, R. (2014). A Novel Finasteride 0.25% Topical Solution For Androgenetic Alopecia: Pharmacokinetics And Effects On Plasma Androgen Levels In Healthy Male Volunteers. Int J Clin Pharmacol Ther. 52(10). 842-849. Available at: https://doi.org/10.5414/CP202119 ↑71 Arif, T., Dorjay, K., Adil, M., Sami, M., (2017). Dutasteride in Androgenetic Alopecia: An Update. Current Clinical Pharmacology. 12(1). Available at: 10.2174/157488471266617031011112 ↑72, ↑77, ↑79 Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M. (2025). A Randomized, Double-Blind, Placebo And Active Controlled Phase II Study To Evaluate The Safety And Efficacy Of Novel Dutasteride Topical Solution (0.01%, 0.02%, And 0.05% W/V) In Male Subjects With Androgenetic Alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑73, ↑78 Nada, E.A., Sharkawy, R.E., Maged, W.M., & Elmagd, M.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Southern Medical Journal, 22, 387-400 ↑74 Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martínez-Moreno, A., Ocampo-Garza, S.S., (2022). Microneedling Plus Topical Dutasteride Solution for Androgenetic Alopecia: A Randomized Placebo-Controlled Study. Journal of the European Academy of Dermatology and Venereology. 36(10). e806–e808. Available at: https://doi.org/10.1111/jdv.18285 ↑75, ↑76 Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M., (2025). Efficacy of Skin Patting and Iontophoresis With Dutasteride Gel in Male and Menopausal Female Androgenetic Alopecia: A Pilot Study. Dermatology and Therapy (Heidelberg). 15(11). 3419–3424. Available at: https://doi.org/10.1007/s13555-025-01532-w ↑80 Chung, H., Dai, T., Sharma, S.K., Huang, Y.Y., Carroll, J.D., Hamblin, M.R. (2012). The Nuts And Bolts Of Low-Level Laser (Light) Therapy. Annals of Biomedical Engineering. 40(2). 516-533. Available at: https://doi.org/10.1007/s10439-011-0454-7 ↑81 Lyu, K., Liu, X., Jiang, L., Chen, Y., Lu, J., Zhu, B., Liu, X., Li, Y., Wang, D., Li, S. (2022). The Functions And Mechanisms Of Low-Level Laser Therapy In Tendon Repair (Review). Frontiers in Physiology. 13. 808374. Available at: https://doi.org/10.3389/fphys.2022.808374 ↑82 Satino, J.L., Markou, M. (2003). Hair Regrowth And Increased Hair Tensile Strength Using The HairMax LaserComb For Low-Level Laser Therapy. International Journal of Cosmetic Surgery and Aesthetic Dermatology. 5(2). 113-117. Available at: https://doi.org/10.1089/153082003769591209 ↑83, ↑88 Leavitt, M., Charles, G., Heyman, E. (2009). HairMax LaserComb® Laser Phototherapy Device In The Treatment Of Male Androgenetic Alopecia. Clinical Drug Investigation. 29(5). 283-292. Available at: https://doi.org/10.2165/00044011-200929050-00001 ↑84 Lanzafame, R.J., Blanche, R.R., Bodian, A.B., Chiacchierini, R.P., Fernandez-Obregon, A., Kazmirek, E.R. (2013). The Growth Of Human Scalp Hair Mediated By Visible Red Light Laser And LED Sources In Males. Lasers in Surgery and Medicine. 45(8). 487-495. Available at: https://doi.org/10.1002/lsm.22173 ↑85 Kim, H., Choi, J.W., Kim, J.Y., Shin, J.W., Lee, S.J., Huh, C.H. (2013). Low-Level Light Therapy For Androgenetic Alopecia: A 24-Week, Randomized, Double-Blind, Sham Device-Controlled Multicenter Trial. Dermatologic Surgery. 39(8). 1177-1183. Available at: https://doi.org/10.1111/dsu.12200 ↑86 Friedman, S., Schnoor, P. (2017). Novel Approach To Treating Androgenetic Alopecia In Females With Photobiomodulation (Low-Level Laser Therapy). Dermatologic Surgery. 43(6). 856-867. Available at: https://doi.org/10.1097/DSS.0000000000001114 ↑87 Avci, P., Gupta, G.K., Clark, J., Wikonkal, N., Hamblin, M.R. (2014). Low-Level Laser (Light) Therapy (LLLT) For Treatment Of Hair Loss. Lasers in Surgery and Medicine. 46(2). 144-151. Available at: https://doi.org/10.1002/lsm.22170 ↑89, ↑93 Sondagar, D.M., Mehta, H.H., Agharia, R.S., Jhavar, M.K. (2023). Efficacy Of Low-Level Laser Therapy In Androgenetic Alopecia – A Randomized Controlled Trial. International Journal of Trichology. 15(1). 25-32. Available at: https://doi.org/10.4103/ijt.ijt_5_22 ↑90 Courtois, M., Loussouarn, G., Hourseau, S., Grollier, J.F. (1996). Periodicity In The Growth And Shedding Of Hair. British Journal of Dermatology. 134(1). 47-54 ↑91 Kunz, M., Seifert, B., Trüeb, R.M. (2009). Seasonality Of Hair Shedding In Healthy Women Complaining Of Hair Loss. Dermatology. 219(2). 105-110. Available at: https://doi.org/10.1159/000216832 ↑92 Liu, C., Yang, J., Qu, L., Gu, M., Liu, Y., Gao, J., Collaudin, C., Loussouarn, G. (2014). Changes In Chinese Hair Growth Along A Full Year. International Journal of Cosmetic Science. 36(6). 531-536. Available at: https://doi.org/10.1111/ics.12151 ↑94 Esmat, S.M., Hegazy, R.A., Gawdat, H.I., Abdel Hay, R.M., Allam, R.S., El Naggar, R., Moneib, H. (2017). Low Level Light-Minoxidil 5% Combination Versus Either Therapeutic Modality Alone In Management Of Female Patterned Hair Loss: A Randomized Controlled Study. Lasers In Surgery And Medicine. 49. 835-843. Available at: https://doi.org/10.1002/lsm.22684 ↑95 Munck, A., Gavazzoni, M.F., Trüeb, R.M. (2014). Use Of Low-Level Laser Therapy As Monotherapy Or Concomitant Therapy For Male And Female Androgenetic Alopecia. International Journal Of Trichology. 6(2). 45-49. Available at: https://doi.org/10.4103/0974-7753.138584 ↑96 Hamblin, M.R. (2019). Photobiomodulation For The Management Of Alopecia: Mechanisms Of Action, Patient Selection And Perspectives. Clinical, Cosmetic And Investigational Dermatology. 12. 669-678. Available at: https://doi.org/10.2147/CCID.S184979 ↑97 Egger, A., Resnik, S.R., Aickara, D., Maranda, E., Kaiser, M., Wikramanayake, T.C., Jimenez, J.J. (2020). Examining The Safety And Efficacy Of Low-Level Laser Therapy For Male And Female Pattern Hair Loss: A Review Of The Literature. Skin Appendage Disorders. 6(5). 259-267. Available at: https://doi.org/10.1159/000509001 ↑98 Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., Elsner, P. (2012). Topical Melatonin for Treatment of Androgenetic Alopecia. Int J Trichology. 4(4). 236-245. Available at: https://doi.org/10.4103/0974-7753.111199 ↑99 Zaky, M.S., Abo Khodeir, H., Ahmed, H.-A., Elsaie, M.L., (2021). Therapeutic Implications of Topical Cetirizine 1% in Treatment of Male Androgenetic Alopecia: A Case-Controlled Study. Journal of Cosmetic Dermatology. 20. 1154–1159. Available at: https://doi.org/10.1111/jocd.13940 ↑100 Hossein Mostafa, D., Samadi, A., Niknam, S., Nasrollahi, S.A., Guishard, A., Firooz, A., (2021). Efficacy of Cetirizine 1% Versus Minoxidil 5% Topical Solution in the Treatment of Male Alopecia: A Randomized, Single-blind Controlled Study. Journal of Pharmacy & Pharmaceutical Sciences. 24. 191–199. Available at: https://doi.org/10.18433/jpps31456 ↑101 Bassiouny, E.A., El-Samanoudy, S.I., Abbassi, M.M., Nada, H.R., Farid, S.F., (2023). Comparison Between Topical Cetirizine With Minoxidil Versus Topical Placebo With Minoxidil in Female Androgenetic Alopecia: A Randomized, Double-Blind, Placebo-Controlled Study. Archives of Dermatological Research. 315. 1293–1304. Available at: https://doi.org/10.1007/s00403-022-02512-2 ↑102 Morganti, P., Fabrizi, G., James, B., Bruno, C. (1998). Effect of gelatin-cystine and serenoa repens extract on free radicals level and hair growth. Journal of Applied Cosmetology. 16, 57-64. Available at: https://www.researchgate.net/publication/294672324_Effect_of_gelatin-cystine_and_serenoa_repens_extract_on_free_radicals_level_and_hair_growth ↑103 Wessagowit, V., Tangjaturonrusamee, C., Kootiratrakarn, T., et al. (2016). Treatment of male androgenetic alopecia with topical products containing Serenoa repens extract. Australas J Dermatol. 57(3), e76-e82. Available at: https://doi.org/10.1111/ajd.12352 ↑104 Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the Hair Growth in Androgenetic Alopecia Subjects – A 16-Week Randomized, Placebo-Controlled Study. Clin Cosmet Investig Dermatol. 16, 3251-3266. Available at: https://doi.org/10.2147/CCID.S435795 ↑105 Fields, J. R., Vonu, P. M., Monir, R. L., Schoch, J. J. (2020). Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review. Dermatol Ther. 33(1). e13202. Available at: https://doi.org/10.1111/dth.13202 Key Takeaways
- Branding: PP-405 is positioned as a next-generation regenerative hair loss therapy, focusing on reactivating dormant hair follicles rather than simply slowing hair loss.
- Unique Selling Point: Targets hair follicle metabolism, aiming to activate dormant hair follicle stem cells and potentially regenerate new hair growth.
- Clinical Support: Early Phase 1/2a data (n=78) suggest some efficacy, with ~31% of treated participants achieving >20% increases in hair density vs 0% in placebo. However, results are based on company-released data and conference presentations, not peer-reviewed publications.
- Concerns: There is currently a lack of peer-reviewed clinical data, and the proposed mechanism, reactivating dormant hair follicles, has not been convincingly demonstrated. Notably, the placebo group also showed signs of “new growth,” which is likely explained by natural hair cycling (kenogen) rather than a true treatment effect.
- Evidence Quality: scored 10/100 for evidence quality, by our metrics.
- Recommendations to Pelage: Pelage should publish full clinical trial data in peer-reviewed journals and provide clear mechanistic evidence, such as biopsies confirming stem cell activation. The company should also conduct larger, longer-term randomized controlled trials using validated endpoints, and release detailed safety data from all phases of testing.
What is PP-405?
PP-405 is a drug that inhibits the mitochondrial pyruvate carrier (MPC). MPC is an important regulator of metabolism, and it has been suggested that inhibiting MPC can ‘wake up’ hair follicle stem cells from a dormant state and force them into an active state. This may lead to hair growth in areas where it had previously stopped[1]Flores, A., Schell, J., Krall, A. S., Jelinek, D., Miranda, M., Grigorian, M., Braas, D., et al. (2017). Lactate dehydrogenase activity drives hair follicle stem cell activation. *Nature Cell … Continue reading The drug is being developed by a company called Pelage Pharmaceuticals, founded by researchers from the University of California, Los Angeles (UCLA) who originally discovered the importance of MPC in hair follicles.
PP-405 is currently undergoing clinical trials. Phase 1/2a clinical trials have been completed, and Pelage has released some promising results.[2]ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: … Continue reading,[3]Pelage Pharmaceuticals. (2026). Pelage Pharmaceuticals’ PP405 and its impact on follicular regeneration to be presented at the American Academy of Dermatology (AAD) Annual Meeting 2026. *Pelage … Continue reading However, these are yet to be published in a peer-reviewed scientific journal. Phase 3 clinical trials are believed to be currently ongoing.
Who is the Target Population?
Clinical trials to assess PP-405 have included men and women with androgenic alopecia. The company’s website highlights multiple target conditions: androgenic alopecia, telogen effluvium, and drug-induced alopecia.[4]Pelage Pharmaceuticals. (n.d.). Regenerative medicine treatments for hair loss. *Pelage Pharma.* Available at: https://pelagepharma.com/
What PP-405 Products are on Offer?
PP-405 is currently undergoing clinical trials, and is therefore not approved for sale. However, given the interest and excitement generated by early experiments, some companies are selling similar molecules, or their own compounded versions of PP-405. Given that this drug has not been approved, and there is very limited safety data available, it is not recommended to buy any of these products.
How is PP-405 Unique?
PP-405 is the only drug that targets metabolism in hair follicle stem cells to promote hair growth that is currently undergoing clinical trials. Pelage Pharmaceutical claims that the drug could go beyond slowing hair loss and “directly drive hair follicle regeneration”.[5]Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: … Continue reading
Product Science: Deep Dive
PP405 was discovered through research investigating important cells in the hair follicle known as stem cells.
Stem cells act like factories that can produce different types of cells in the body. In the hair follicle, the production of new hairs depends on the activity of stem cells, which are activated at the start of the growth phase of the hair cycle. Hair follicle stem cells (HFSCs) can give rise to all the different types of cells in the hair follicle and are essential for the growth of new hair.[6]Hsu, Y. C., Pasolli, H. A., Fuchs, E., et al. (2011). Dynamics between stem cells, niche, and progeny in the hair follicle. *Cell.* 144. 92–105. Available at: … Continue reading
Because the hair follicle produces hairs in cycles, HFSCs go through periods of activation and a dormant period known as quiescence. Researchers at UCLA were interested in how the metabolic activity of HFSCs changes when they switch from a quiescent state to an active one.
When HFSCs are quiescent, they show relatively low metabolic activity. The researchers found that HFSCs generate energy through a process called glycolysis more than most other cells do. Glycolysis is a form of anaerobic respiration, meaning it doesn’t use oxygen. You’re most likely to experience anaerobic respiration when exercising: if your body can’t get enough oxygen to your muscles fast enough, they revert to glycolysis to produce energy.
HFSCs, however, seem to use glycolysis at a high rate even when there is plenty of oxygen available. The researchers also found that activating metabolic pathways involved in anaerobic respiration is essential for activating HFSCs. One enzyme involved in these pathways is called lactate dehydrogenase.[7]Flores, A., Schell, J., Krall, A. S., Jelinek, D., Miranda, M., Grigorian, M., Braas, D., et al. (2017). Lactate dehydrogenase activity drives hair follicle stem cell activation. *Nature Cell … Continue reading
Using mouse models, the researchers showed that stopping lactate dehydrogenase from working can prevent HFSC activation, meaning they stay in a quiescent state. Conversely, forcing HSFCs into anaerobic respiration can promote hair growth. A molecule called the mitochondrial pyruvate carrier (MPC) is responsible for “deciding” whether cells prefer aerobic (with oxygen) or anaerobic respiration (without oxygen). If you block MPC, then you can guide HFSCs towards anaerobic respiration, causing them to ‘wake up’ from their dormant state.
The researchers identified a number of molecules that are capable of blocking MPC.[8]Liu, X., Flores, A. A., Situ, L., Gu, W., Ding, H., Christofk, H. R., Lowry, W. E., & Jung, M. E. (2021). Development of novel mitochondrial pyruvate carrier inhibitors to treat hair loss. … Continue reading Some of these UCLA researchers, led by William Lowry, went on to start Pelage Pharmaceuticals, and one of the MPC inhibitor molecules they later produced was PP-405.
It seems they were confident that activating hair growth by blocking MPC in HFSCs would translate well into a treatment for hair loss in humans.
Clinical Data
Following this promising experimental data and the founding of Pelage Pharmaceuticals, clinical trials began enrolling in 2024.[9]ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: … Continue reading Clinical trials registration information states that phase 1/2a trials were completed in July 2025, and the next day, Pelage announced “positive results”.[10]Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: … Continue reading
Their press release provides some details of the study. 78 men and women with androgenic alopecia were enrolled and randomized into either the treatment group or placebo group. The treatment group applied PP-405 to their scalp once daily for 4 weeks, with follow-up at 12 weeks.
While this phase of clinical trials is intended to test the safety of a treatment and generally operates as a ‘proof-of-concept’ study, Pelage did release some efficacy information. They state that 4 weeks after treatment was completed, 31% of participants in the treatment group exhibited a greater than 20% increase in hair density, compared to 0% of patients in the placebo group.
Unfortunately, these results have yet to be published in a peer-reviewed journal, which would allow us to better scrutinize their data. However, the company did present some results from the Phase 2a clinical trial at the American Academy of Dermatology (AAD) Annual Meeting in March 2026.
As of April 2026, the presentation is available on Vimeo (starting at around 1:21).[11]Vimeo. (2026). Pelage PP405 presentation at American Academy of Dermatology (AAD) Annual Meeting 2026. *Vimeo.* Available at: https://vimeo.com/1177843989/a90148385b
In their presentation, Pelage highlights the importance of follicular units: bundles of 1 to 4 hair follicles, along with associated glands, nerves, and muscles, that are the tissue structure used in hair transplantation.
You can see an example of the images presented in Figure 1. The researchers took pictures of patients’ scalps prior to treatment (using micro tattooing to confirm they were observing the same area), and again at week 12. The claim to ‘track’ the follicular units in the images over time, and suggest that there is new growth visible where previously there was none. They also claim that some follicular units that were producing vellus hairs are now producing terminal (visible) hairs, and some follicular units are producing multiple hairs where previously they were producing only one.
Figure 1. Images taken from Pelage Pharmaceuticals’ presentation at the AAD Annual Meeting in March 2026. They claim red circles highlight new growth after treatment (bottom), where previously hair follicles were dormant (top).
Pelage also presented some summary data from the trial, which you can see in Figure 2. They suggest that new growth in the treated group represents terminal (non-vellus) hairs, which will contribute to the appearance of hair, while new growth in the placebo group represents vellus hairs, which are not visible to the naked eye.
Figure 2. Summary data taken from Pelage Pharmaceuticals’ presentation at the AAD Annual Meeting in March 2026. Change in “activated follicular units” was related to an increase in non-vellus hairs in PP-405-treated patients (left), while it was related to an increase in vellus hairs in placebo patients (right).
They support this claim by showing a relationship between “activated follicular units” and the number of non-vellus hairs in the treatment group, while there is a relationship between “activated follicular units” and vellus hairs in the placebo group. I.e., while there is new growth in the placebo group, this is less likely to contribute to the appearance of hair.
Problems with the Data
While these data represent only a snapshot of the trial results, they could be very promising. Unfortunately, the data presented thus far don’t back up the claims made by Pelage.
In their presentation, the researchers consistently refer to “activated follicular units”. Based on the mechanism of action that they identified in laboratory models, they claim that these represent dormant follicles that have been reawakened via activation of their stem cells.
However, hair follicles go through natural cycles of shedding and regrowth. Hair follicles can spend long periods of time “empty” following a shedding event, where they are stuck in kenogen, the “resting” stage of the follicular cycle, where no fiber is produced. In fact, in a healthy scalp, about 10% of hair follicles may be in this state at any given time, which typically lasts about 2 months.[12]Guarrera, M., & Rebora, A. (2019). The higher number and longer duration of kenogen hairs are the main cause of the hair rarefaction in androgenetic alopecia. *Skin Appendage Disorders.* 5(3). … Continue reading
As such, there is no evidence to demonstrate that apparent new growth in PP-405 treated patients is not simply hairs exiting kenogen.
This is demonstrated by Pelage’s own data, where new growth from apparently empty areas is visible in participants who received placebo. If we look more closely at the x axes of the summary data, we can see that while ~76% (31 out of 41) patients in the treated group displayed a positive change in activated follicular units, ~73% (16 out of 21) in the placebo group also showed a positive change.
This makes the claim that PP-405 is activating dormant follicles less robust.
The data does still seem to suggest an increase in terminal hairs, which may translate to more visible hair. When Pelage publishes its data in full, and we see further evidence from larger trials, it will be possible to begin assessing the drug’s efficacy.
Safety Data
Pelage has not released the safety data from their early phase clinical trials, though they do claim that there are “no concerns” regarding safety based on these studies.[13]Vimeo. (2026). Pelage PP405 presentation at American Academy of Dermatology (AAD) Annual Meeting 2026. *Vimeo.* Available at: https://vimeo.com/1177843989/a90148385b It is important to wait until safety data from larger studies is released before making any firm decisions about the safety of the drug.
What is the Regulatory Status of PP-405?
PP-405 is still undergoing clinical trials and has not yet been approved in any jurisdiction. Phase 3 clinical trials are expected to finish in 2026, and the drug won’t be available until 2027/2028 at the earliest.
Should I Use PP-405?
It is not currently possible to assess the safety or efficacy of PP-405, or any similar MPC inhibitors. While some non-FDA-approved treatments might be available online, these represent untested and potentially unsafe treatments.
Recommendations to Pelage
The small snapshots of data we have seen from PP-405 could be promising. However, information is currently limited, and there are some steps we’d like to see Pelage take:
- Publish all safety and efficacy data from clinical trials in a peer-reviewed journal.
- Perform experiments to test the hypothesis that PP-405 is activating dormant hair follicles. This could include biopsies or other techniques to explore what is happening at the follicular level. Currently available data showing new hair growth do not demonstrate their proposed mechanism of action.
- Perform larger, randomized, placebo-controlled trials with validated endpoints to explore whether PP-405 increases hair growth over a long period of time.
Final Thoughts
The laboratory experiments that led to the development of PP-405 are robust and genuinely groundbreaking. The researchers highlighted a previously unexplored aspect of hair follicle physiology and identified a potential route to regrow hair.
There are also some promising results in terms of increases in non-vellus hairs. However, it is not currently possible to independently assess these data, as they have not been published in a peer-reviewed journal. Furthermore, the data we have seen so far do not sufficiently support Pelage’s claims regarding the mechanism of action of PP-405.
Still, PP-405 remains “one to watch”, and we look forward to more complete data being released in the near future.
References[+]
References ↑1, ↑7 Flores, A., Schell, J., Krall, A. S., Jelinek, D., Miranda, M., Grigorian, M., Braas, D., et al. (2017). Lactate dehydrogenase activity drives hair follicle stem cell activation. *Nature Cell Biology.* 19(9). 1017–1026. Available at: https://doi.org/10.1038/ncb3575 ↑2 ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: https://clinicaltrials.gov/study/NCT06393452 ↑3 Pelage Pharmaceuticals. (2026). Pelage Pharmaceuticals’ PP405 and its impact on follicular regeneration to be presented at the American Academy of Dermatology (AAD) Annual Meeting 2026. *Pelage Pharma.* Available at: https://pelagepharma.com/press-releases/pelage-pp405-aad-2026/ ↑4 Pelage Pharmaceuticals. (n.d.). Regenerative medicine treatments for hair loss. *Pelage Pharma.* Available at: https://pelagepharma.com/ ↑5 Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: https://www.businesswire.com/news/home/20250617338859/en/Pelage-Pharmaceuticals-Announces-Positive-Phase-2a-Clinical-Trial-Results-for-PP405-in-Regenerative-Hair-Loss-Therapy ↑6 Hsu, Y. C., Pasolli, H. A., Fuchs, E., et al. (2011). Dynamics between stem cells, niche, and progeny in the hair follicle. *Cell.* 144. 92–105. Available at: https://doi.org/10.1016/j.cell.2010.11.049 ↑8 Liu, X., Flores, A. A., Situ, L., Gu, W., Ding, H., Christofk, H. R., Lowry, W. E., & Jung, M. E. (2021). Development of novel mitochondrial pyruvate carrier inhibitors to treat hair loss. *Journal of Medicinal Chemistry.* 64(4). 2046–2063. Available at: https://doi.org/10.1021/acs.jmedchem.0c01570 ↑9 ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: https://clinicaltrials.gov/study/NCT06393452 ↑10 Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: https://www.businesswire.com/news/home/20250617338859/en/Pelage-Pharmaceuticals-Announces-Positive-Phase-2a-Clinical-Trial-Results-for-PP405-in-Regenerative-Hair-Loss-Therapy ↑11, ↑13 Vimeo. (2026). Pelage PP405 presentation at American Academy of Dermatology (AAD) Annual Meeting 2026. *Vimeo.* Available at: https://vimeo.com/1177843989/a90148385b ↑12 Guarrera, M., & Rebora, A. (2019). The higher number and longer duration of kenogen hairs are the main cause of the hair rarefaction in androgenetic alopecia. *Skin Appendage Disorders.* 5(3). 152–154. Available at: https://doi.org/10.1159/000493587 Key Takeaways
- Branding: AMP-303 is a proprietary injectable treatment for androgenic alopecia (AGA), developed by Amplifica Holdings Group Inc.
- Unique Selling Point: Amplifica claims that AMP-303 works by delivering “unique biological signaling molecules” intradermally to reactivate dormant hair follicles and stimulate the conversion of vellus hairs into terminal hairs – a process known as hair terminalization.
- Clinical Support: One first-in-human, randomized, double-blind, placebo-controlled, multicenter Phase I trial has been completed, with results announced in September 2024 and presented at the Society for Investigative Dermatology Annual Meeting in May 2025. Male participants aged 18-45 with AGA were stratified by duration of hair loss (3-5 years vs. ≥10 years) and received AMP-303 on one side of the scalp and placebo on the other. A statistically significant percentage of participants showed >15% increase in non-vellus hair count at 60 days post-treatment (p<0.001) compared to placebo, with >10% increase still observed at 150 days post-treatment. Safety was described as favourable, with no severe adverse events.
- Concerns:
- Results for the long-standing hair loss group (Group 2) have not been separately disclosed, raising questions about whether AMP-303 is effective in this population.
- No raw data or clinical photographs have been published; all information currently derives from press releases and a single conference abstract. It is therefore impossible to assess the visible clinical impact of the treatment.
- There are no data beyond 150 days, so the durability of effect, need for repeat dosing, and long-term safety are all unknown.
- The precise active ingredient and mechanism of action have not been published in a peer-reviewed journal.
- Evidence Quality: AMP-303 scored 31/100 for evidence quality, by our metrics.
- Recommendations to Amplifica: We recommend that Amplifica publish the full Phase I trial dataset in a peer-reviewed journal, including subgroup data separated by hair loss duration, responder versus non-responder analyses, and objective mean hair count data for the full treated population. Phase II/III trials with adequate sample sizes, enrolment of both sexes, a wider age range, active comparator arms, and follow-up of at least 12 months are needed before the clinical place of AMP-303 can be properly assessed. Full disclosure of the mechanism of action would substantially increase confidence in the product.
What is AMP-303?
AMP-303 is an intradermal injectable treatment for androgenic alopecia (AGA), currently in first-in-human trials. It was developed by Amplifica Holdings Group Inc, a clinical-stage biopharmaceutical company co-founded by Dr Maksim Plikus (Chief Scientific Officer), Dr William Rassman (Chief Medical Officer), and Frank Fazio (President and CEO).[1]Amplifica, (no date). About Us. Available at: https://amplificabio.com/about-us/ (Accessed: April 2026) AMP-303 is described as a polysaccharide-based formulation designed to be delivered into the deep dermis via intradermal microinjection in the frontotemporal scalp.
Who is the Target Population?
While Amplifica’s first-in-human trial has only enrolled men aged 18-45, the company has stated that it expects AMP-303 to be effective in both men and women. Future research is expected to include women.
What Amplifica Products are Offered?
It should be noted that none of Amplifica’s products are currently available to use; however, Amplifica currently has three hair loss treatments in the pipeline.
Its lead candidate, currently in first-in-human trials, is AMP-303. Additional compounds include AMP-203/506, an osteopontin-based compound, and AMP-601, a SCUBE-3-based compound.[2]Amplifica. Pipeline. Developing novel injectable treatments to address androgenetic alopecia. Available at: https://amplificabio.com/pipeline/ (Accessed: April 2026) All of these treatments remain investigational at the time of writing.
How Does Amplifica Claim Its Product is Unique?
Amplifica has stated that AMP-303 works via a nature-inspired mechanism involving the delivery of isolated biological signaling molecules to reactivate dormant hair follicles. The company reported that AMP-303 can induce the conversion of vellus (thin, fine) hairs back into terminal (thicker, pigmented) hairs. This would represent a meaningful reversal of AGA pathology rather than just slowing its progression. This is a mechanistic distinction from FDA-approved treatments like finasteride and minoxidil, though, as mentioned before, the precise molecular target has not yet been described.
Product Science: Deep Dive
The Hairy Mole Observation and the Scientific Basis
The scientific foundation behind AMP-303 begins with an observation: skin moles (“melanocytic nevi”) in humans very commonly grow unusually thick, dense hair. The same phenomenon was replicated in mouse models. Rather than treating this as a curiosity, Dr Plikus and his team at UCI asked the question – what is the mole actually doing to the hair follicles around it?
A study published in Nature provided the answer. The cells that give moles their pigment – melanocytes – eventually stop dividing and enter a state called senescence, a kind of permanent cellular retirement. Senescent cells are not simply inactive, however; they release a cocktail of signaling proteins into the surrounding tissue. The Plikus laboratory found that it is precisely this secreted cocktail that supercharges hair growth in the skin above and around the mole.[3]Wang, X., Ramos, R., Phan, A, Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, … Continue reading
By systematically screening the proteins released by these senescent melanocytes, the researchers identified osteopontin (a protein encoded by the gene SPP1) as the key driver. They demonstrated this in several ways:
- Injecting osteopontin alone was sufficient to trigger robust hair growth in mice.
- Genetically engineering mice to overproduce it had the same effect
- Blocking either osteopontin itself or the receptor on hair stem cells that it binds to (a molecule called CD44) prevented the mole from inducing excess hair growth
Figure 1: Effect of hair growth hyperactivation in mice.[4]Wang, X., Ramos, R., Phan, A,Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, … Continue reading Figure used in line with Creative Commons.
Importantly, osteopontin also stimulated new hair follicle growth in isolated human hair tissue tested in the laboratory, suggesting a pathway relevant to humans.
In short, the hairy mole effect is not accidental. It is driven by a specific molecular signal that tells dormant hair follicles to start growing.
It should be noted, however, that the precise active ingredient in AMP-303 has not been publicly disclosed by Amplifica. Within the company’s pipeline, osteopontin is designated as a separate compound, AMP-203, which has not yet entered clinical trials. It is not yet confirmed whether osteopontin, a related molecule from the same biological pathway, or an entirely distinct compound is responsible for the effects observed in the first-in-human trial.
What is the Regulatory Status of AMP-303?
AMP-303 is being developed through the conventional pharmaceutical regulatory pathway, with Amplifica pursuing regulatory approval. The first-in-human trial that was conducted in 2023-2024 was a Phase I safety and tolerability study. No FDA approval or IND clearance status for subsequent phases has been publicly disclosed. AMP-303 is therefore not available to people outside of clinical trials at this time.
Clinical Data
As previously mentioned, a first-in-human trial was conducted over 2023-2024 for AMP-303.[5]Green, J.B., Joseph, J.H., DuBois, J.C., Rassman, W.R., Fazio, F., Plikus, M.V., Ahmad, W. (2025). LB1139 AMP-303 injectable treatment for androgenetic alopecia: A multicenter, randomized, … Continue reading This was a randomized, double-blind, placebo-controlled, multicenter study conducted in the United States. Participants were male, aged 18-45, and diagnosed with AGA.
The participants were split into two groups:
- Group 1 (n=32): recent onset hair loss (3-5 years duration)
- Group 2 (n=29): long-standing hair loss (≥10 years duration)
Each participant received AMP-303 injected into 20 frontotemporal locations on one side of the scalp and saline placebo injections on the other side. Assessments were conducted at 60 and 150 days post-treatment.
Results:
- At 60 days post-treatment, a statistically significant proportion of participants showed >15% increase in non-vellus hair count compared to the placebo (p<0.001).
- At 150 days post-treatment, >10% experienced an increase in non-vellus hair count vs. just maintenance in the placebo.
- For Group 1 (recent onset) responders, the mean increase in non-vellus hair count was reported as 14.5%, peaking at 60 days.
- AMP-303 demonstrated the ability to promote the conversion of vellus to non-vellus hair.
- Results were observed in the frontotemporal region, which has historically been a challenging area for demonstrating treatment response.
While the results seem promising, we do have some concerns.
Missing data for the long-standing hair loss group. Group 2 (≥10 years duration) results have not been reported in public communications. This is a meaningful omission because a large number of hair loss patients have been battling it for long periods of time, and the lack of disclosure could mean that AMP-303 did not confer a benefit to this group.
No data beyond 150 days. While this concern is less important as it is a first-in-human trial, we currently do not know if gains persist longer than 150 days, if patients require repeat dosing, or if hair gains start to regress. Furthermore, we don’t know what the long-term safety is.
No published raw data/images: We are only able to keep up with updates about AMP-303 through press releases (and one published abstract); we have no idea what the raw data looks like, and whether the results have led to visible clinical effects.
Safety Profile
AMP-303 was described as safe and well-tolerated in the above trial. The majority of adverse events were mild in severity, and no severe adverse events were reported. Local skin reactions to intradermal injections were similar between the AMP-303 and placebo groups. Long-term safety is unknown.
The most common treatment-related adverse effects were:
- Mild headache (7.8%)
- Mild skin swelling (placebo 3.1%; AMP-303 1.6%)
Should I Use AMP-303?
While AMP-303 is not yet available to the public, once it is available, you might want to follow the treatment if:
- You have experienced hair loss for 5 years or less.
- Open to an injectable treatment.
- Interested in non-hormonal approaches to hair loss.
- Interested in early adoption of hair loss treatments.
This product could take some time (we are talking years) to come to the market, so we wouldn’t recommend you drop your current treatments just yet!
Recommendations to Amplifica
We recommend that Amplifica publish the full Phase I trial dataset in a peer-reviewed journal, including subgroup data separated by hair loss duration, responder vs. non-responder analyses, and objective mean hair count data for the full treated population. Phase II/III trials with adequate sample sizes, enrolment of both sexes, a wider age range, active comparator arms, and follow-up periods of at least 12 months are needed before the clinical efficacy of AMP-303 can be properly assessed. Furthermore, full disclosure of the mechanism of action of the treatment would substantially increase our confidence in the product.
Final Thoughts
AMP-303 represents a genuinely novel biological approach to AGA with its roots in rigorous academic research at UCI on the molecular basis of follicular activation. The first-in-human trial results are encouraging, particularly the frontotemporal efficacy and evidence of terminalization, and the non-hormonal mechanism is potentially advantageous for those who cannot or do not wish to take finasteride or dutasteride. However, AMP-303 remains an investigational product with a single small-scale trial behind it and no peer-reviewed publication.
We have seen many treatments start with a lot of excitement that ultimately fail to translate into meaningful clinical benefit. We recommend cautious optimism and waiting for more data to be published before making a decision.
References[+]
References ↑1 Amplifica, (no date). About Us. Available at: https://amplificabio.com/about-us/ (Accessed: April 2026) ↑2 Amplifica. Pipeline. Developing novel injectable treatments to address androgenetic alopecia. Available at: https://amplificabio.com/pipeline/ (Accessed: April 2026) ↑3 Wang, X., Ramos, R., Phan, A, Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, K.N., Phung, J.L., Ingal, J.P., Levitt, K.M., Cao, X., Liu, Y., Deng, Z., Taguchi, N., Scarfone, V.M., Wang, G., Paolilli, K.N., Wang, X., Guerrero-Juarez, C.F., Davis, R.T., Greenberd, E.N., Ruiz-Vega, R., Vasudeva, P., Murad, R., Widyastuti, L.H.P., Lee, H-L., McElwee, K.J., Gadeau, A-P., Lawson, D.A., Andersen, B., Mortazavi, A., Yu, Z., Nie, Q., Kunisada, T., Karin, M., Tuckermann, J., Esko, J.D., Ganesan, A.K., Li, J., Plikus, M.V. (2023). Signalling by senescent melanocytes hyperactivates hair growth. Nature. 618. 808-817. Available at: https://doi.org/10.1038/s41586-023-06172-8 ↑4 Wang, X., Ramos, R., Phan, A,Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, K.N., Phung, J.L., Ingal, J.P., Levitt, K.M., Cao, X., Liu, Y., Deng, Z., Taguchi, N., Scarfone, V.M., Wang, G., Paolilli, K.N., Wang, X., Guerrero-Juarez, C.F., Davis, R.T., Greenberd, E.N., Ruiz-Vega, R., Vasudeva, P., Murad, R., Widyastuti, L.H.P., Lee, H-L., McElwee, K.J., Gadeau, A-P., Lawson, D.A., Andersen, B., Mortazavi, A., Yu, Z., Nie, Q., Kunisada, T., Karin, M., Tuckermann, J., Esko, J.D., Ganesan, A.K., Li, J., Plikus, M.V. (2023). Signalling by senescent melanocytes hyperactivates hair growth. Nature. 618. 808-817. Available at: https://doi.org/10.1038/s41586-023-06172-8 ↑5 Green, J.B., Joseph, J.H., DuBois, J.C., Rassman, W.R., Fazio, F., Plikus, M.V., Ahmad, W. (2025). LB1139 AMP-303 injectable treatment for androgenetic alopecia: A multicenter, randomized, placebo-controlled feasibility study of a novel polysaccharide (Abstract). Journal of Investigative Dermatology. 145(8), S198. Available at: www.https://doi.org/ 10.1016/j.jid.2025.06.1428 Anavar is the brand name for the synthetic anabolic-androgenic steroid (AAS), oxandrolone. It is used clinically to promote weight gain and preserve body mass in catabolic disorders (where body tissues are excessively broken down for energy, causing rapid weight loss), such as severe burns, chronic illnesses, or neuromuscular disorders.[1]Orr, R., Fiatarone Singh, M. (2004). The Anabolic Androgenic Steroid Oxandrolone in the Treatment of Wasting and Catabolic Disorders: Review of Efficacy and Safety. Drugs. 64(7). 725–750. Available … Continue reading
AAS, including anavar, are structurally modified versions of either testosterone or dihydrotestosterone (DHT). They induce responses in tissues by binding to an androgen receptor, resulting in increased muscle protein synthesis.
Androgenic alopecia (AGA) is a condition characterized by the activation of the androgen receptor, whereby the growth phase of the hair growth cycle shortens, resulting in the progressive miniaturization of susceptible hair follicles and hair loss.[2]Ho, C.H., Sood, T., Zito, P.M., (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026)
Anavar is often described as a mild AAS; however, any introduction of systemic, exogenous steroids (i.e., steroids introduced to the body from external sources which act on the whole body) can result in the steroids activating androgen receptors on susceptible scalp follicles and the acceleration of AGA.
In this article, we will discuss the effect that AAS have on hair loss, whether there is any evidence for Anavar-specific hair loss, and potential risk management strategies while using Anavar.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Exogenous Androgens and AGA: What the Science Actually Shows
Androgens are steroid sex hormones; those produced within the body are known as endogenous, whereas those produced outside the body are known as exogenous. AAS are androgens used (or misused) for performance-enhancing effects.
How Does Endogenous Testosterone Contribute to Hair Loss?
Endogenous testosterone is produced by the body as a crucial androgen promoting the development and maintenance of male characteristics.
The importance of testosterone in hair loss was originally suggested by James Hamilton. He demonstrated that castrated men did not develop AGA unless they received testosterone supplementation.[3]Hamilton, J.B. (1942). Male Hormone Stimulation Is a Prerequisite and Incitant in Common Baldness. Journal of Investigative Dermatology. 5(6). 473–474. Available at: … Continue reading
Testosterone contributes to AGA primarily through its conversion to DHT, a more potent androgen that plays a central role in hair loss. Testosterone itself is not inherently harmful to hair follicles, but its downstream metabolism into DHT and the sensitivity of hair follicles determine its effect on AGA.
In the scalp, testosterone is converted to DHT by the enzyme 5-alpha reductase (5AR), which is highly expressed in hair follicles of the frontal and vertex scalp (areas typically affected in AGA).[4]Sawaya, M.E., Price, V.H. (1997). Different Levels of 5alpha-Reductase Type I and II, Aromatase, and Androgen Receptor in Hair Follicles of Women and Men with Androgenetic Alopecia. Journal of … Continue reading The hair follicles in these areas of the scalp also contain a higher number of androgen receptors, increasing their vulnerability to the action of androgens, such as DHT.[5]Hibberts, N.A., Howell, A.E., Randall, V.A. (1998). Balding Hair Follicle Dermal Papilla Cells Contain Higher Levels of Androgen Receptors than Those from Non-Balding Scalp. Journal of Endocrinology. … Continue reading
DHT binds to the androgen receptor with significantly greater strength than testosterone. This leads to DHT preferentially binding the androgen receptor, resulting in stronger and longer activation of the cell, and greater effects on cell function.
The binding of DHT to androgen receptors in susceptible hair follicles disrupts the hair growth cycle, resulting in follicle miniaturization. This is caused by the disruption of the different phases of the hair cycle. Specifically, the growth phase of the hair follicle (anagen) shortens, and the resting phase (telogen) stays the same or increases, altering the ratio of growth to rest from 12:1 to 5:1.[6]Ho, C.H., Sood, T., Zito, P.M., (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026)
Consequently, the hair follicle shrinks with each cycle, resulting in the replacement of regular hair with short terminal hairs (hairs that lack pigmentation and won’t grow any more than a few inches). Over time, the growth phase becomes so short that the hair fails to penetrate the surface of the skin, causing a bald patch.[7]Asfour, L., Cranwell, W., Sinclair, R. (2000). Male Androgenetic Alopecia. Endotext. Available at: http://www.ncbi.nlm.nih.gov/books/NBK278957/
Males lacking the 5AR enzyme to convert testosterone to DHT have never been reported as developing hair loss.[8]Imperato-McGinley, J., Zhu, Y.S. (2002). Androgens and Male Physiology: The Syndrome of 5alpha-Reductase-2 Deficiency. Molecular and Cellular Endocrinology. 198(1–2). 51–59. Available at: … Continue reading The binding of testosterone-derived DHT to androgen receptors is therefore a key step in hair loss.
What Effect Do Exogenous Androgens Have on Hair Loss?
Testosterone
The administration of exogenous testosterone causes serum levels of testosterone to rise systemically (throughout the body). This provides an increased concentration of testosterone for the 5AR enzymes to convert into DHT. In individuals susceptible to AGA, this can accelerate the onset or progression of hair loss.
Clinical Evidence
Hair loss is a relatively common side effect of taking exogenous testosterone. Studies investigating the use of testosterone in athletes or others taking it for muscle-building purposes have demonstrated the onset of AGA after use.[9]Tawanwongsri, W., Desai, D.D., Nohria, A., Shapiro, J., Lo Sicco, K.I. (2025). Hair Loss in Athletic Testosterone Use in Males: A Narrative Review. International Journal of Dermatology. 64(4). … Continue reading,[10]Griggs, J., Almohanna, H., Ahmed, A., Tosti, A. (2018). New-Onset Androgenic Alopecia following Human Chorionic Gonadotropic Diet and Testosterone Pellet Implantation. International Journal of … Continue reading
In addition, a study researching long-term use of testosterone therapy in trans men has found that 31% of their cohort developed moderate to severe AGA (although this was a very small study, so any conclusions should be used cautiously).[11]Wierckx, K., Van de Peer, F., Verhaeghe, E., et al. (2014). Short- and Long-Term Clinical Skin Effects of Testosterone Treatment in Trans Men. Journal of Sexual Medicine. 11(1). 222–229. Available … Continue reading
AAS
AAS are often derivatives of either testosterone or DHT (i.e., synthetic compounds which are chemically modified versions of testosterone/DHT), sharing the same mechanism of action as endogenous testosterone in initiating or progressing hair loss. However, AAS expose hair follicles to androgen stimulation well beyond normal levels.[12]Bond, P., Smit, D.L., de Ronde, W. (2022). Anabolic–Androgenic Steroids: How Do They Work and What Are the Risks? Frontiers in Endocrinology (Lausanne). 13. Available at: … Continue reading
The high levels of androgen stimulation can occur in different ways depending on the AAS used:
- AAS that are similar in structure to testosterone can be converted to DHT or similar molecules using the 5AR enzyme. These DHT products can bind to and activate the androgen receptors with high affinity (strength).
- AAS that are similar in structure to DHT can act directly on androgen receptors, bypassing the need for conversion using the 5AR enzyme.
The presence of these androgen-inducing AAS in the scalp at levels well beyond normal testosterone and DHT levels results in the increased activation of androgen receptors and the subsequent disruption of the hair cycle, follicle miniaturization, and hair loss in susceptible individuals.[13]Stojko, M., Nocoń, J., Piłat, P., et al. (2023). Innovative Reports on the Effects of Anabolic Androgenic Steroid Abuse—How to Lose Your Mind for the Love of Sport. Medicina (Kaunas). 59(8). … Continue reading
Evidence in Humans
- An investigation into AGA use by 541 gym-goers in Saudi Arabia found that 14.2% experienced hair loss as a side effect. However, this study was not carried out under clinical conditions, and all results were self-reported, so it cannot be said for certain whether the AAS were causing hair loss.[14]Albaker, W., Alkhars, A., Elamin, Y., Jatoi, N., Boumarah, D., Al-Hariri, M. (2021). Anabolic–Androgenic Steroid Abuse among Gym Users, Eastern Province, Saudi Arabia. Medicina (Kaunas). 57(7). … Continue reading
- An investigation into AGA use in 104 bodybuilders in Brazil found that 23.7% of AAS users experienced hair loss as a side effect. This study was also not carried out under clinical conditions and used self-reported results.[15]dos Santos, J.C., de Souza, E., Meneses-Santos, D., et al. (2024). The Use of Anabolic Steroids by Bodybuilders in the State of Sergipe, Brazil. European Journal of Investigation in Health, … Continue reading
- The HAARLEM study investigated AAS abuse in a cohort of 100 strength athletes. They found that self-reported alopecia increased from 2% at the start of the study to 12% after a year. The study was limited by using only self-reported results, and multiple participants used other drugs at the same time, making it impossible to prove that the hair loss was from AAS use alone.[16]Smit, D.L., Buijs, M.M., de Hon, O., den Heijer, M., de Ronde, W. (2021). Positive and Negative Side Effects of Androgen Abuse. The HAARLEM Study: A One-Year Prospective Cohort Study in 100 Men. … Continue reading
While the underlying science supporting a potential role of AAS in AGA is thought to be sound, there is minimal evidence from human studies. Although there are a multitude of anecdotal reports supporting the evidence for AGA on AAS use, there are no rigorous clinical trials demonstrating these effects.
Why Are Some People More Susceptible to Hair Loss?
You may have noticed that throughout this blog so far, we have often used the word ‘susceptible’ when referring to hair follicles or people experiencing AGA. This is because AGA and follicular sensitivity are highly determined by genetic factors, meaning that not everyone is susceptible to AGA.
Approximately 80% of men will ultimately experience AGA by the age of 70.[17]Ho, C.H., Sood, T., Zito, P.M. (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026) This is largely controlled by genetics, although environmental and lifestyle factors (such as smoking, obesity, and nutritional status) can also contribute to the development of AGA.
One study found that men whose fathers had AGA had a 2.5-fold increased risk of developing AGA compared to those whose fathers did not experience hair loss.[18]Chumlea, W.C., Rhodes, T., Girman, C.J., et al. (2004). Family History and Risk of Hair Loss. Dermatology. 209(1). 33–39. Available at: https://doi.org/10.1159/000078584 Women experiencing female pattern hair loss also often report other family members with the same condition.[19]Ramos, P.M., Miot, H.A. (2015). Female Pattern Hair Loss: A Clinical and Pathophysiological Review. Anais Brasileiros de Dermatologia. 90(4). 529–543. Available at: … Continue reading
Genetic variants, particularly of the androgen receptor, contribute substantially to susceptibility and can also influence how individuals respond to treatment (see our article on genetic tests for personalized hair loss treatments to find out more). There is no one gene involved in AGA; hair loss is likely to be a polygenic disorder, meaning that variants in multiple genes are involved in enhanced susceptibility.
It is thought that a person’s genetics is responsible for influencing how strongly scalp follicles respond to androgen signaling. In susceptible persons, the follicles in the scalp have enhanced sensitivity (even to normal levels of androgens) and so are more likely to miniaturize when exposed to DHT.[20]Kaufman, K.D. (2002). Androgens and Alopecia. Molecular and Cellular Endocrinology. 198(1). 89–95. Available at: https://doi.org/10.1016/S0303-7207(02)00372-6
Susceptibility to AAS-Induced Hair Loss
The use of AAS does not increase an individual’s susceptibility to AGA, but is thought to contribute to accelerating and unmasking hair loss in those already genetically predisposed to hair loss.
Under normal conditions, the AGA process of hair loss occurs slowly, potentially only reaching a noticeable level after years. However, the introduction into the body of AAS is thought to cause a dramatic rise in androgen levels.
Genetically vulnerable follicles are potentially subjected to intense and sustained androgen receptor activation from the AAS testosterone or DHT derivatives, which may bind to the receptors with similar or greater strength than natural DHT. The enhanced activation of the androgen receptors then causes follicle miniaturization and hair loss.[21]Stojko, M., Nocoń, J., Piłat, P., et al. (2023). Innovative Reports on the Effects of Anabolic Androgenic Steroid Abuse—How to Lose Your Mind for the Love of Sport. Medicina (Kaunas). 59(8). … Continue reading
Individuals who lack a genetic predisposition to hair loss may tolerate even high levels of androgen exposure without experiencing hair loss.
Limitations
While the theory of AAS-accelerated AGA makes sense and is consistent with what is known about the DHT-induced method of follicle miniaturization, the scientific evidence supporting the theory is surprisingly limited. Much of the basis of this theory comes from assumptions and extrapolation (extension) of the collective current knowledge of follicle miniaturization.
Any studies that do exist tend to be based on self-reported outcomes of AAS use and lack the controls necessary for true clinical conclusions to be drawn.
How Does Anavar Contribute to Hair Loss?
Anavar (or oxandrolone) is an AAS used clinically to restore weight loss after severe trauma, major surgery, or malnutrition, and has also been studied in HIV-related wasting, neuromuscular conditions, and in severe burns patients.[22]Orr, R., Fiatarone Singh, M. (2004). The Anabolic Androgenic Steroid Oxandrolone in the Treatment of Wasting and Catabolic Disorders: Review of Efficacy and Safety. Drugs. 64(7). 725–750. Available … Continue reading,[23]Fenichel, G.M., Griggs, R.C., Kissel, J., et al. (2001). A Randomized Efficacy and Safety Trial of Oxandrolone in the Treatment of Duchenne Dystrophy. Neurology. 56(8). 1075–1079. Available at: … Continue reading,[24]Jeschke, M.G., Finnerty, C.C., Suman, O.E., Kulp, G., Mlcak, R.P., Herndon, D.N. (2007). The Effect of Oxandrolone on the Endocrinologic, Inflammatory, and Hypermetabolic Responses During the Acute … Continue reading Anavar’s clinical use requires a prescription, and it is carefully controlled due to its established effect on the liver and potential effect on cardiovascular health and hormones.[25]Orr, R., Fiatarone Singh, M. (2004). The Anabolic Androgenic Steroid Oxandrolone in the Treatment of Wasting and Catabolic Disorders: Review of Efficacy and Safety. Drugs. 64(7). 725–750. Available … Continue reading,[26]van Amsterdam, J., Opperhuizen, A., Hartgens, F. (2010). Adverse Health Effects of Anabolic-Androgenic Steroids. Regulatory Toxicology and Pharmacology. 57(1). 117–123. Available at: … Continue reading
Anavar has been described as having a favourable ratio of muscle-building effect to androgenic effect, meaning that it is thought to be less likely to cause severe androgenic side effects compared to other AAS.[27]Wu, C., Kovac, J.R. (2016). Novel Uses for the Anabolic Androgenic Steroids Nandrolone and Oxandrolone in the Management of Male Health. Current Urology Reports. 17(10). 72. Available at: … Continue reading As such, it is often considered ‘mild’. However, it must be recognized that this is in reference to other AAS and does not mean that it is safe.
A misunderstanding of the mildness of Anavar has resulted in its relatively popular recreational misuse. Bodybuilders and athletes have been known to use it for promoting muscle growth and increasing strength without large amounts of water retention. Despite many considering it mild, Anavar is not safe when misused.
How Does Anavar Compare to Other AAS?
As discussed above, our understanding of exactly how the use of AAS contributes to hair loss is severely limited. However, we can speculate (based on our knowledge of the DHT-induced mechanism of hair loss) how different AAS may differentially affect hair loss.
The impact on hair loss of AAS use is predicted to vary depending on which AAS is taken. AAS differ in how strongly they activate androgen receptors in different tissues. The structure of the androgen is very important to androgen receptor activation; AAS may be DHT-derived or testosterone-derived, with the ability to convert into DHT or a DHT-like molecule using the 5AR enzyme.
Anavar is a DHT-derived AAS; as such, it cannot be converted into a more potent androgen receptor binder using the 5AR enzyme. It has been suggested to bind to the androgen receptor less strongly than testosterone, and so could be predicted to have a lesser effect on hair loss mechanisms than endogenous testosterone/DHT or other AAS.[28]Knuth, C.M., Auger, C., Jeschke, M.G. (2021). Burn-Induced Hypermetabolism and Skeletal Muscle Dysfunction. American Journal of Physiology – Cell Physiology. 321(1). C58–C71. Available at: … Continue reading However, it does still have an androgenic effect, meaning that it will likely still have a harmful effect on hair loss.[29]Church, J.A., (2004). Oxandrolone Treatment of Childhood Hereditary Angioedema. Annals of Allergy, Asthma & Immunology. 92(3). 377–378. Available at: … Continue reading
It should be noted that the conclusions drawn here on the effects of Anaver on hair loss are primarily speculation and have not been demonstrated in clinical trials or other scientific research.
Risk Management While Using Anavar
Anavar should only be used when prescribed by your medical practitioner and should never be used recreationally due to the associated risks. However, if you are using Anavar and are concerned about the effects on hair loss, the following may be points to consider to reduce hair loss risk.
1. Using DHT Reducers
Finasteride and dutasteride are prescription medications that treat hair loss through their DHT-reducing action. They work by targeting the 5AR enzyme, blocking the conversion of testosterone to DHT, thereby reducing DHT in the scalp and enhancing hair growth (for more information on finasteride and dutasteride, see our ultimate guide).[30]Asfour, L., Cranwell, W., Sinclair, R. (2000). Male Androgenetic Alopecia. Endotext. Available at: http://www.ncbi.nlm.nih.gov/books/NBK278957/
While finasteride and dutasteride are often effective for normal AGA, they will only be partially effective when using Anavar.
Anavar is a DHT-derivative and cannot be converted using the 5AR enzyme, so it activates the scalp androgen receptors directly. Finasteride/dutasteride target the 5AR enzyme and consequently play no role in preventing Anavar from binding to the androgen receptors and initiating follicle miniaturization, and so will not prevent Anavar-induced hair loss.
Taking finasteride or dutasteride will reduce the baseline level of hair loss caused by endogenous testosterone (i.e., any hair loss that would be occurring regardless of whether AAS have been taken), potentially mildly reducing your total hair loss.
2. Changing Anavar Formulation
Anavar has only been formulated and approved for use in oral form. The application of a topical preparation (such as crushed tablets) is untested and potentially unsafe.
Topical AAS do exist and may be prescribed for conditions such as hypogonadism to provide relatively steady-state delivery of the drug (i.e., the drug is administered in a way where the amount of drug in the body stays stable over time).
While you may think that applying the AAS topically will prevent hair loss – as the drug may only have its effect on the area where it is applied – this is untrue. Topically applied AAS will enter the bloodstream and end up acting systemically (throughout the body).
For example, case studies have shown instances of topical AAS acting systemically:
- One woman who applied a cellulite cream containing DHT to her legs started to develop a deeper voice due to virilization (development of male physical characteristics) caused by the AAS.[31]Wollina, U., Pabst, F., Schönlebe, J., et al. (2007). Side-Effects of Topical Androgenic and Anabolic Substances and Steroids. A Short Review. Acta Dermatovenerologica Alpina, Pannonica et … Continue reading
- A different woman applying an androgen cream containing testosterone to her legs developed dark male-like hair on her legs and mild excessive hair growth on her upper lip.[32]Wollina, U., Pabst, F., Schönlebe, J., et al. (2007). Side-Effects of Topical Androgenic and Anabolic Substances and Steroids. A Short Review. Acta Dermatovenerologica Alpina, Pannonica et … Continue reading
3. Brand Switching
AAS must only be used under the guidance of your medical practitioner. They will know the brand or type of AAS that you should be on based on your medical issue, meaning that it may not be possible to switch.
When comparing types of AAS for the ability to cause hair loss, Anavar is predicted to be low down the list due to its relatively low androgenic potential. As such, if you are on Anavar, then that has the potential to be the least likely to cause hair loss.
Other AAS predicted to have low hair loss potential are low doses of Turinabol or Nandrolone, which are also thought to have relatively low androgenic potential.[33]Kam, P.C.A., Yarrow, M. (2005). Anabolic Steroid Abuse: Physiological and Anaesthetic Considerations. Anaesthesia. 60(7). 685–692. Available at: https://doi.org/10.1111/j.1365-2044.2005.04218.x,[34]Bond, P., Smit, D.L., de Ronde, W. (2022). Anabolic–Androgenic Steroids: How Do They Work and What Are the Risks? Frontiers in Endocrinology (Lausanne). 13. Available at: … Continue reading
4. Avoiding Exogenous Androgens Entirely
The best way to avoid AAS-induced hair loss is to avoid taking any AAS. In certain situations, they may be medically necessary, in which case you should do what your medical practitioner perceives to be best. Taking AAS recreationally puts you at risk of several serious side effects.
If you are looking to boost your testosterone naturally, you could consider:
- Losing weight
- Resistance and HIIT exercise
- Sleep optimization
- Stress reduction
- Changes to your diet
- Reducing alcohol intake
Final Thoughts
Anavar is unlikely to be completely hair safe. As a DHT-derived compound, it can directly activate androgen receptors in the scalp and contribute to the miniaturization of susceptible hair follicles. While Anavar is often described as a “mild” anabolic steroid, this is relative to other AAS and should not be interpreted as an absence of any androgenic effects on the scalp.
Some users may use AAS without experiencing noticeable hair loss, but this is largely due to genetics rather than the specific steroid used. In those who are genetically susceptible to hair loss, the use of AAS is thought to accelerate or unmask an underlying hair loss process rather than initiating a new one.
It is important to recognize the limitations of available evidence linking AAS and hair loss. Although hair loss is frequently reported anecdotally, controlled clinical trials are lacking, and most available data come from observational studies relying on self-reported outcomes.
As a result, much of our current understanding of AAS-induced hair loss is based on extrapolation from the well-established mechanisms of DHT-mediated follicle miniaturization, which unfortunately limits the strength of any conclusions.
References[+]
References ↑1, ↑22, ↑25 Orr, R., Fiatarone Singh, M. (2004). The Anabolic Androgenic Steroid Oxandrolone in the Treatment of Wasting and Catabolic Disorders: Review of Efficacy and Safety. Drugs. 64(7). 725–750. Available at: https://doi.org/10.2165/00003495-200464070-00004 ↑2, ↑6 Ho, C.H., Sood, T., Zito, P.M., (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026) ↑3 Hamilton, J.B. (1942). Male Hormone Stimulation Is a Prerequisite and Incitant in Common Baldness. Journal of Investigative Dermatology. 5(6). 473–474. Available at: https://doi.org/10.1038/jid.1942.62 ↑4 Sawaya, M.E., Price, V.H. (1997). Different Levels of 5alpha-Reductase Type I and II, Aromatase, and Androgen Receptor in Hair Follicles of Women and Men with Androgenetic Alopecia. Journal of Investigative Dermatology. 109(3). 296–300. Available at: https://doi.org/10.1111/1523-1747.ep12335779 ↑5 Hibberts, N.A., Howell, A.E., Randall, V.A. (1998). Balding Hair Follicle Dermal Papilla Cells Contain Higher Levels of Androgen Receptors than Those from Non-Balding Scalp. Journal of Endocrinology. 156(1). 59–65. Available at: https://doi.org/10.1677/joe.0.1560059 ↑7 Asfour, L., Cranwell, W., Sinclair, R. (2000). Male Androgenetic Alopecia. Endotext. Available at: http://www.ncbi.nlm.nih.gov/books/NBK278957/ ↑8 Imperato-McGinley, J., Zhu, Y.S. (2002). Androgens and Male Physiology: The Syndrome of 5alpha-Reductase-2 Deficiency. Molecular and Cellular Endocrinology. 198(1–2). 51–59. Available at: https://doi.org/10.1016/S0303-7207(02)00368-4 ↑9 Tawanwongsri, W., Desai, D.D., Nohria, A., Shapiro, J., Lo Sicco, K.I. (2025). Hair Loss in Athletic Testosterone Use in Males: A Narrative Review. International Journal of Dermatology. 64(4). 654–658. Available at: https://doi.org/10.1111/ijd.17567 ↑10 Griggs, J., Almohanna, H., Ahmed, A., Tosti, A. (2018). New-Onset Androgenic Alopecia following Human Chorionic Gonadotropic Diet and Testosterone Pellet Implantation. International Journal of Trichology. 10(6). 284–285. Available at: https://doi.org/10.4103/ijt.ijt_75_18 ↑11 Wierckx, K., Van de Peer, F., Verhaeghe, E., et al. (2014). Short- and Long-Term Clinical Skin Effects of Testosterone Treatment in Trans Men. Journal of Sexual Medicine. 11(1). 222–229. Available at: https://doi.org/10.1111/jsm.12366 ↑12 Bond, P., Smit, D.L., de Ronde, W. (2022). Anabolic–Androgenic Steroids: How Do They Work and What Are the Risks? Frontiers in Endocrinology (Lausanne). 13. Available at: https://doi.org/10.3389/fendo.2022.1059473 ↑13, ↑21 Stojko, M., Nocoń, J., Piłat, P., et al. (2023). Innovative Reports on the Effects of Anabolic Androgenic Steroid Abuse—How to Lose Your Mind for the Love of Sport. Medicina (Kaunas). 59(8). 1439. Available at: https://doi.org/10.3390/medicina59081439 ↑14 Albaker, W., Alkhars, A., Elamin, Y., Jatoi, N., Boumarah, D., Al-Hariri, M. (2021). Anabolic–Androgenic Steroid Abuse among Gym Users, Eastern Province, Saudi Arabia. Medicina (Kaunas). 57(7). 703. Available at: https://doi.org/10.3390/medicina57070703 ↑15 dos Santos, J.C., de Souza, E., Meneses-Santos, D., et al. (2024). The Use of Anabolic Steroids by Bodybuilders in the State of Sergipe, Brazil. European Journal of Investigation in Health, Psychology and Education. 14(5). 1451–1469. Available at: https://doi.org/10.3390/ejihpe14050096 ↑16 Smit, D.L., Buijs, M.M., de Hon, O., den Heijer, M., de Ronde, W. (2021). Positive and Negative Side Effects of Androgen Abuse. The HAARLEM Study: A One-Year Prospective Cohort Study in 100 Men. Scandinavian Journal of Medicine & Science in Sports. 31(2). 427–438. Available at: https://doi.org/10.1111/sms.13843 ↑17 Ho, C.H., Sood, T., Zito, P.M. (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026) ↑18 Chumlea, W.C., Rhodes, T., Girman, C.J., et al. (2004). Family History and Risk of Hair Loss. Dermatology. 209(1). 33–39. Available at: https://doi.org/10.1159/000078584 ↑19 Ramos, P.M., Miot, H.A. (2015). Female Pattern Hair Loss: A Clinical and Pathophysiological Review. Anais Brasileiros de Dermatologia. 90(4). 529–543. Available at: https://pubmed.ncbi.nlm.nih.gov/26375223/ ↑20 Kaufman, K.D. (2002). Androgens and Alopecia. Molecular and Cellular Endocrinology. 198(1). 89–95. Available at: https://doi.org/10.1016/S0303-7207(02)00372-6 ↑23 Fenichel, G.M., Griggs, R.C., Kissel, J., et al. (2001). A Randomized Efficacy and Safety Trial of Oxandrolone in the Treatment of Duchenne Dystrophy. Neurology. 56(8). 1075–1079. Available at: https://doi.org/10.1212/WNL.56.8.1075 ↑24 Jeschke, M.G., Finnerty, C.C., Suman, O.E., Kulp, G., Mlcak, R.P., Herndon, D.N. (2007). The Effect of Oxandrolone on the Endocrinologic, Inflammatory, and Hypermetabolic Responses During the Acute Phase Postburn. Annals of Surgery. 246(3). 351–362. Available at: https://doi.org/10.1097/SLA.0b013e318146980e ↑26 van Amsterdam, J., Opperhuizen, A., Hartgens, F. (2010). Adverse Health Effects of Anabolic-Androgenic Steroids. Regulatory Toxicology and Pharmacology. 57(1). 117–123. Available at: https://doi.org/10.1016/j.yrtph.2010.02.001 ↑27 Wu, C., Kovac, J.R. (2016). Novel Uses for the Anabolic Androgenic Steroids Nandrolone and Oxandrolone in the Management of Male Health. Current Urology Reports. 17(10). 72. Available at: https://doi.org/10.1007/s11934-016-0629-8 ↑28 Knuth, C.M., Auger, C., Jeschke, M.G. (2021). Burn-Induced Hypermetabolism and Skeletal Muscle Dysfunction. American Journal of Physiology – Cell Physiology. 321(1). C58–C71. Available at: https://doi.org/10.1152/ajpcell.00106.2021 ↑29 Church, J.A., (2004). Oxandrolone Treatment of Childhood Hereditary Angioedema. Annals of Allergy, Asthma & Immunology. 92(3). 377–378. Available at: https://doi.org/10.1016/S1081-1206(10)61578-5 ↑30 Asfour, L., Cranwell, W., Sinclair, R. (2000). Male Androgenetic Alopecia. Endotext. Available at: http://www.ncbi.nlm.nih.gov/books/NBK278957/ ↑31, ↑32 Wollina, U., Pabst, F., Schönlebe, J., et al. (2007). Side-Effects of Topical Androgenic and Anabolic Substances and Steroids. A Short Review. Acta Dermatovenerologica Alpina, Pannonica et Adriatica. 16(3). 117–122. Available at: https://pubmed.ncbi.nlm.nih.gov/17994172/ ↑33 Kam, P.C.A., Yarrow, M. (2005). Anabolic Steroid Abuse: Physiological and Anaesthetic Considerations. Anaesthesia. 60(7). 685–692. Available at: https://doi.org/10.1111/j.1365-2044.2005.04218.x ↑34 Bond, P., Smit, D.L., de Ronde, W. (2022). Anabolic–Androgenic Steroids: How Do They Work and What Are the Risks? Frontiers in Endocrinology (Lausanne). 13. Available at: https://doi.org/10.3389/fendo.2022.1059473 In 2026, we’re seeing hundreds of shampoos marketed as “hair loss miracles.” In reality, most of these shampoos rely on misinterpreted science and inflated claims to boost sales. Very few of these shampoos actually show meaningful results for hair regrowth.
You might be wondering about shampoos that include ingredients such as saw palmetto, caffeine, niacin, or biotin, since some scientific evidence does exist for these ingredients. But, despite these ingredients being the same, the available evidence involves completely different delivery methods. Biotin supplements are not comparable to biotin shampoos for hair growth, and the same can be said for topicals containing saw palmetto, caffeine, and niacin.
In this article, we break down what hair shampoos can actually do, we rank the 10 best shampoos for hair loss based on their ingredients, and dive into what the clinical evidence actually says. We also explore their safety profiles and offer some practical tips on how to use them. We’ll help guide you on which shampoos are actually worth your time and money, and which might just be marketing hype.
Thickening Shampoo
Stronger strands. Fuller look. Science-backed ingredients.
A lightweight, daily-use formula designed to support thicker-looking hair. Powered by caffeine, saw palmetto, keratin, and panthenol without harsh chemicals or artificial fragrance.

Why We Have to Be Careful with Hair Loss Shampoo Claims
When searching for shampoos for hair loss, it’s not unusual to see bold marketing claims. Even in 2026, the market is saturated with statements such as:
- “…contributes towards up to 10X less breakage” – Garnier
- “…strengthens scalp’s hold onto hair roots for visibly denser, longer, and stronger hair” – Pantene
- “…recharges and energizes the strands for a fuller-looking head of hair” – Alpecin
It is easy to take these claims at face value. But while some may have scientific merit, most of the time, these claims are inflated or backed by misinterpreted supportive evidence. We commonly see the same top four ingredients when searching for hair loss shampoos: Saw palmetto, caffeine, niacin, and biotin.
These ingredients do have some supportive evidence. Oral and topical saw palmetto has been shown to improve hair parameters in men and women with androgenic alopecia (AGA).[1]Evron, E., Juhasz, M., Babadjouni, A., Mesinkovska, N.A., (2020). Natural Hair Supplement: Friend Or Foe? Saw Palmetto, A Systematic Review In Alopecia. Skin Appendage Disorders. 6(6). 329–337. … Continue reading Topical caffeine has been shown to perform similarly to 2% minoxidil.[2]Völker, J.M., Koch, N., Becker, M., Klenk, A., (2020). Caffeine And Its Pharmacological Benefits In The Management Of Androgenetic Alopecia: A Review. Skin Pharmacology And Physiology. 33(3). … Continue reading Topical niacin improved hair volume in one study on females with pattern hair loss.[3]Draelos, Z.D., Jacobson, E.L., Kim, H., Kim, M., Jacobson, M.K., (2005). A Pilot Study Evaluating The Efficacy Of Topically Applied Niacin Derivatives For Treatment Of Female Pattern Alopecia. … Continue reading Oral biotin supplements have regrown hair in people with severe biotinidase deficiencies (which occur in 1/110,000 people).[4]Patel, D.P., Swink, S.M., Castelo-Soccio, L., (2017). A Review Of The Use Of Biotin For Hair Loss. Skin Appendage Disorders. 3(3). 166–169. Available at: https://doi.org/10.1159/000462981
However, a common tactic we see in the hair-loss industry is cherry-picking data to make ingredients look effective, but in reality, the trials conducted evaluated ingredients in completely different formulations (e.g., oral, topical). So these ingredient claims become invalid when applied to shampoos. Here’s why:
#1: Phase I metabolism
Products that are taken orally usually enter phase I metabolism in the liver. Here, the substance is broken down (metabolized) into other compounds, then distributed throughout the body via the bloodstream. When the same product is applied topically, it does not go through the same metabolic process, unless some of the product is absorbed into the systemic circulation.
When products are used in different formulations (oral, topical, or shampoo), they have completely different effects on the body compared to oral administration. This means that studies evaluating the efficacy of oral products for hair regrowth cannot be translated into claims for topically applied products, and certainly not for products used as shampoos.
Figure 1. Metabolism of oral products. Adapted from Figure 1.[5]Dinh, L., Yan, B. (2023). Oral Drug Delivery Via Intestinal Lymphatic Transport Utilizing Lipid-Based Lyotropic Liquid Crystals. Liquids. 3(4). 456-468. Available at: … Continue reading
Used under Creative Commons License.#2: Scalp penetration and skin contact time
Now, if we look at studies that investigate products used topically, we may assume that any claims can automatically be applied to shampoo products. Unfortunately, this is not the case.
Many of the ingredients used in topicals take hours for effective penetration into the scalp. When products are applied as a shampoo, they are in contact with the scalp for much shorter periods, usually just minutes, before ingredients within the shampoo are washed out.
This means, in the majority of cases, ingredients lack enough time to effectively penetrate the scalp, and therefore, have little time to be efficacious for hair regrowth.
If you are a member, you can find out more about hair shampoo ingredients in our video and article here.
What Can “Hair Loss Shampoos” Actually Do?
Overall, shampoos may improve the appearance of the hair. Shampoos can potentially make the hair feel more robust and thicker, or even appear healthier and shinier. There are some shampoo ingredients that have limited preclinical or clinical support for hair regrowth (included in our top 10 list below).
However, it is important to note that these shampoos are mostly appropriate for people with AGA alongside inflammation associated with microorganism overgrowths in the scalp.
Figure 2. Photo documentation of hair growth at one scalp site at (a) Baseline, (b) after 3 months, and (c) after 6 months of shampoo application containing 1% caffeine and 1% sodium dimethylglycinate. Adapted from Figure 6.[6]Celleno, L., Bussoletti, C., Tolaini, M.V., Rossi, A., Ala, L., Becker, M., Völker, J.M., Wiesche, E.S.Z. (2025). A Novel Approach Against Male Pattern Hair Loss With Topical Dimethylglycine Sodium … Continue reading Used under Creative Commons License.
10 Shampoos Ingredients – Ranked by Evidence
Despite the marketing hype around hair loss shampoos, we have sifted through the noise and found some ingredients that have been tested in human trials.
At Perfect Hair Health, we evaluate products based on evidence quality. We prioritize randomized controlled human trials over small pilot studies, preclinical data, and anecdotes. We take into consideration regrowth potential, looking closely at clinical results that translate into cosmetically meaningful improvements (terminal hair counts and hair-thickness improvements).
Here is a breakdown of our top shampoos that have clinical evidence for hair growth:
Shampoo Ingredient Clinical studies Treatments Key findings #1 Ketoconazole Study #1:[7]Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: … Continue reading Controlled comparative study. 39 men with AGA aged 21-33, 22 non-AGA age-matched controls; 21 months 2% ketoconazole shampoo vs normal shampoo 2-4x/week Pilary index (% hairs in anagen hairs times average hair-shaft diameter) in AGA men increased progressively with ketoconazole; the control group showed a gradual decline. No change in non-AGA controls Study #2:[8]Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: … Continue reading Pilot study. 8 men with AGA aged 24-29; 6 months 2% ketoconazole shampoo vs 2% minoxidil lotion Ketoconazole: +18% hair density, +7% shaft diameter, -19% sebaceous gland size. Minoxidil: +11% hair density, +7% shaft diameter, +5% sebaceous gland size
Study #3:[9]Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine … Continue reading Randomized controlled trial. 150 men with telogen effluvium + AGA + dandruff; 6 months
1% ketoconazole vs 1 % piroctone olamine vs 1% zinc pyrithione shampoos 2-3x/week Ketoconazole: -17.3% shedding, +4.9% anagen hair ratio, -4.8% sebum excretion. No difference in hair density. Pruritus and dandruff cleared in all groups Study #4:[10]Khandpur, S., Suman, M., Reddy, B.S. (2002). Comparative Efficacy Of Various Treatment Regimens For Androgenetic Alopecia In Men. J Dermatol. 29(8). 489-98. Available at: … Continue reading Open-label, randomized, parallel-group trial. 100 men with AGA; 12 months
1 mg/day oral finasteride + 2% ketoconazole shampoo vs 1 mg/day oral finasteride vs 2% topical minoxidil vs 1 mg/day oral finasteride + 2% topical minoxidil Finasteride + minoxidil and finasteride + ketoconazole had the best improvement scores for both self- and physician-assessments. Finasteride + minoxidil slightly outperformed finasteride + ketoconazole Study #5:[11]Rafi, A.W., Katz, R.M. (2011). Pilot Study Of 15 Patients Receiving A New Treatment Regimen For Androgenic Alopecia: The Effects Of Atopy On AGA. ISRN Dermatol. 2011. 241953. Available at: … Continue reading Pilot study. 15 men with AGA + atopic or seborrheic dermatitis; 9 months
2% ketoconazole shampoo + NuH Hair + 1 mg/day finasteride + minoxidil foam vs NuH Hair vs 2% ketoconazole + NuH Hair + 1 mg/day finasteride vs 2% ketoconazole shampoo + NuH Hair Average time for hair regrowth: 30 days with NuH Hair + finasteride + minoxidil + ketoconazole and NuH Hair + finasteride + ketoconazole, 60 days with NuH Hair + ketoconazole, and 90 days with NuH Hair alone #2 Piroctone olamine Study #1:[12]Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine … Continue reading Randomized controlled trial. 150 men with telogen effluvium + AGA + dandruff; 6 months 1% ketoconazole vs 1 % piroctone olamine vs 1% zinc pyrithione shampoos 2-3x/week Piroctone olamine: -16.5% shedding, +7.9% anagen hair ratio, -2.9% sebum excretion. No difference in hair density. Pruritus and dandruff cleared in all groups #3 Zinc pyrithione Study #1:[13]Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine … Continue reading Randomized controlled trial. 150 men with telogen effluvium + AGA + dandruff; 6 months 1% ketoconazole vs 1 % piroctone olamine vs 1% zinc pyrithione shampoos 2-3x/week Zinc pyrithione: -10.1% shedding, +6.8% anagen hair ratio, -5.5% sebum excretion. No difference in hair density. Pruritus and dandruff cleared in all groups
Study #2:[14]Berger, R.S., Fu, J.L., Smiles, K.A., Turner, C.B., Schnell, B.M., Werchowski, K.M., Lammers, K.M. (2003). The Effects Of Minoxidil, 1% Pyrithione Zinc And A Combination Of Both On Hair Density: A … Continue reading Randomized, investigator-blinded, parallel-group trial. 200 men with AGA aged 18-49; 6 months 1% pyrithione zinc shampoo 1x/day vs 5% minoxidil topical solution 2x/day vs placebo shampoo vs 1% pyrithione zinc shampoo + 5% minoxidil topical solution At 9 weeks, total visible hair count increased in the pyrithione zinc, minoxidil, and zinc + minoxidil group compared to placebo. Relative hair count for pyrithione zinc was slightly less than half of the relative hair count for minoxidil. No advantage observed for the combination group. #4 Caffeine Study #1:[15]Celleno, L., Bussoletti, C., Tolaini, M.V., Rossi, A., Ala, L., Becker, M., Völker, J.M., Wiesche, E.S.Z. (2025). A Novel Approach Against Male Pattern Hair Loss With Topical Dimethylglycine Sodium … Continue reading Randomized, double-blind, placebo-controlled trial. 154 men early signs of hair thinning and/or AGA; 6 months Shampoo containing 1% caffeine + 1% sodium dimethylglycinate (plus further components) vs placebo Decreased mean number of hairs pulled in the hair pull test in the treatment group (-2.8) compared to placebo (+0.6). Phototrichogram results showed an increased number of hairs, hair density, and % anagen hairs after 6 months in the treatment group compared to placebo Study #2:[16]Bussoletti, C., Tolaini, M.V., Celleno, L. (2020). Efficacy Of A Cosmetic Phyto-Caffeine Shampoo In Female Androgenetic Alopecia. G Ital Dermatol Venereol. 155(4). 492-499. Available at: … Continue readingRandomized, double-blind parallel-group trial. 140 females with AGA aged 40-70; 6 months Plantur 39 caffeine shampoo vs placebo Reduction in pulled hairs after 3 and 6 months in the treatment group. Placebo group mean pulled hairs remained stable. Hair quality assessed by investigators revealed strength and thickness of hair, and hair fall improved in the treatment group compared to placebo Study #3:[17]Chen, D., Yu, F., Wang, C., Chen, H., Tan, J., Shi, Q., He, X., Liu, X., Wang, F., Zhao, H. (2024). Anti-Hair Loss Effect Of A Shampoo Containing Caffeine And Adenosine. Journal Of Cosmetic … Continue reading Randomized, single-blind, parallel-group, controlled trial. 77 individuals with self-perceieved thinning hair aged 20-60; 3 months 0.4% caffeine + 0.2% adenosine shampoo vs placebo Hair density increased from 118.25 to 130.03 hairs/cm2 in the treatment group at month 3. No change in hair density observed in the placebo group. Significant hair loss reduction in the treatment group compared to placebo. #5 Minoxidil Study #1:[18]Yaghmaee, R., Mirbagheri, M., Pakdaman, M.H., Rad, F., Gharibi, F. (2016). The Effect Of Minoxidil 1% Shampoo On Androgenetic Alopecia: A Clinical Trial Study. Research Journal Of Pharmaceutical, … Continue reading Randomized double-blind, placebo- controlled trial. 60 individuals with AGA aged 15-40; 4 months 1% minoxidil shampoo vs placebo Mean terminal hair count in the minoxidil group increased from 24.2 at baseline to 26.9 at 4 months. Placebo group terminal hair count was 22.9 at baseline and 22.9 at month 4. However, no significant differences between groups were observed #6 Salicylic acid, Panthenol & Niacinamide Study #1:[19]Kim, H.T., Park, H.S., Kim, Y.M. (2022). Double-Blind Randomized Placebo-Controlled Study Of The Efficacy And Safety Of Hair Loss Prevention Shampoo Containing Salicylic Acid, Panthenol, And … Continue reading Randomized, double-blind, placebo-controlled trial. 42 individuals with AGA aged 19-54; 24 weeks 0.2% salicylic acid + 0.2% panthenol + 0.1% niacinamide shampoo vs placebo 17.76% increase in the number of hairs on the crown in the treatment group compared to the placebo group after 24 weeks #7 Saw palmetto Study #1:[20]Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the … Continue reading Randomized, double-blind, placebo-controlled trial. 80 individuals with mild-to-moderate AGA aged 18-50; 16 weeks. Investigated a standardized saw palmetto oil (VISPO™, containing 2-3% β-sitosterol) Important caveat: Not a true shampoo study. Data from a topical lotion washed off after 30 minutes
Oral placebo vs oral VISPO™ (400 mg capsule containing 100 mg VISPO™ daily) vs topical placebo vs topical VISPO™ (5 mL of a 20% VISPO™ lotion daily, left on for 30 minutes before washing) At week 16, both VISPO™ groups showed significant reductions in hair shedding (measured by hair comb and hair pull tests) compared to baseline and placebo. Hair fall decreased by around 22% in the topical VISPO™ group compared to baseline. Hair density increased 7.61% with topical VISPO™ Currently, only 7 shampoo ingredients have at least some human clinical evidence suggesting they may be beneficial for hair regrowth. Yet, the evidence on shampoo ingredients for hair loss does have its pitfalls. Many trials involve small cohorts, short trial durations, and investigate a combination of ingredients. This makes it difficult to truly determine which ingredients are responsible for the observed benefits.
Figure 3. Patient with stage AGA who received 2% ketoconazole shampoo + NuH Hair + 1 mg/day finasteride + minoxidil foam. (a) Day 1, (b) Day 90. Adapted from Figure 1.[21]Rafi, A.W., Katz, R.M. (2011). Pilot Study Of 15 Patients Receiving A New Treatment Regimen For Androgenic Alopecia: The Effects Of Atopy On AGA. ISRN Dermatol. 2011. 241953. Available at: … Continue reading Used under Creative Commons license.
Here we provide information for 3 more shampoo ingredients: selenium sulfide, coal tar, and clobetasol. To date, these ingredients lack human clinical evidence for hair regrowth; however, they may indirectly improve scalp health and hair quality by reducing symptoms that damage hair follicles (e.g., inflammation, itching).
Shampoo ingredient Clinical Studies Treatments Key findings #8 Selenium sulfide Study #1:[22]Danby, F.W., Maddin, W.S., Margesson, L.J., Rosenthal, D. (1993). A Randomized, Double-Blind, Placebo-Controlled Trial Of Ketoconazole 2% Shampoo Versus Selenium Sulfide 2.5% Shampoo In The Treatment … Continue reading Randomized, double-blind, placebo-controlled trial. 246 adults with moderate-severe dandruff; 4 weeks 2.5% selenium sulfide shampoo vs 2% ketoconazole shampoo vs placebo Both selenium sulfide and ketoconazole reduced dandruff scores and itch compared to placebo. Ketoconazole was superior on day 8. Study #2:[23]Barbosa, V., Melo, D.F., Vañó-Galván, S., Lutchmanen-Kolanthan, V., Sant’Anna, B., Leclerc-Mercier, S., Reygagne, P. (2024). A Comparative Randomized Clinical Study Assessing The Efficacy Of … Continue reading Randomized, double-blind, comparative, clinical trial. 64 individuals with moderate-to-severe scalp seborrheic dermatitis; 28 days 1% selenium disulfide shampoo (3x/week) vs 2% ketoconazole shampoo 2x/week Selenium disulfide: 71% reduction in disease severity by day 28. Ketoconazole: 69% reduction in disease severity score by day 28
#9 Clobetasol Study #1:[24]Ortonne, J.-P., Nikkels, A.F., Reich, K., Ponce Olivera, R.M., Lee, J.H., Kerrouche, N., Sidou, F., Faergemann, J. (2011). Efficacious And Safe Management Of Moderate To Severe Scalp Seborrhoeic … Continue reading Randomized, multi-centre, comparative clinical trial. 326 individuals with moderate-to-severe scalp seborrhoeic dermatitis; 12 weeks total 0.05% clobetasol vs 2% ketoconazole vs 0.05% clobetasol twice weekly alternating with 2 % ketoconazole twice weekly vs 0.05% clobetasol four times weekly alternating with 2 % ketoconazole twice weekly Clobetasol + ketoconazole improved erythema, scaling, and pruritus more than ketoconazole alone. #10 Coal tar Study #1[25]Goyal, A. (2025). Comparative Study Of Ketoconazole-Based Shampoo Versus Coal Tar-Based Shampoo In Seborrheic Dermatitis. Int. J Med. Pharm. Res. 6(4). 208-211: Comparative clinical trial. 100 participants with mild-to-moderate scalp seborrheic dermatitis; 4 weeks 1% coal tar shampoo vs 2% ketoconazole shampoo 2x/week
By week 4, disease severity scores in both groups reduced from baseline. Ketoconazole was superior in reducing severity faster, providing better symptom relief, and causing fewer side effects Our #1 Best Shampoo Ingredient: Ketoconazole
Based on the available evidence, we ranked ketoconazole as our top-rated shampoo ingredient that may improve hair growth. Ketoconazole is commonly sold under the name Nizoral and is an antifungal medication used to help improve fungal-related conditions such as dandruff, fungal infections, certain hormone-linked diseases, and even hair loss from fungal and non-fungal causes.
Figure 4. Structure of ketoconazole. Adapted from Wikimedia.[26]Wikimedia Commons, (04 September 2025), Ketoconazole Chemical Structure (SVG). Available at: https://commons.wikimedia.org/wiki/Category:Ketoconazole#/media/File:(2S,4R)-Ketoconazol.svg (Accessed: 20 … Continue reading Image in the public domain.
Ketoconazole may improve telogen effluvium by killing scalp pathogens, thereby normalizing excessive hair shedding triggered by microorganism-mediated inflammation. It may also prolong the anagen phase of the hair cycle. Additionally, ketoconazole may improve AGA via three key mechanisms:
- Killing pathogens in the scalp, therefore normalizing excessive hair shedding triggered by microorganism-mediated inflammation.
- Reducing scalp DHT levels by inhibiting 5-alpha reductase (a key enzyme that converts testosterone to DHT) and blocking androgen receptors (“landing pads” for DHT to exert its effects).
- Prolonging the anagen phase of the hair cycle.
You can buy 1% ketoconazole shampoo over the counter, typically at around $16 for 200 mL. Whereas, 2% ketoconazole shampoo usually requires a prescription. Based on the available clinical evidence, there is stronger support for the use of 2% ketoconazole shampoo for hair growth than for 1%. However, there are currently no direct head-to-head studies comparing the effectiveness of 1% ketoconazole shampoo versus 2% for hair regrowth; therefore, any inferences are speculative.
If you are a member and would like to learn more about ketoconazole, read our ultimate guide here.
Are Hair Loss Shampoos Safe?
In general, most hair-loss shampoos containing ketoconazole, piroctone olamine, zinc pyrithione, or caffeine are well tolerated. Very few side effects have been reported with use. However, local irritation, itching, or dryness may occur, particularly when overused or on very sensitive scalps. Concurrent use of other scalp products may require more cautious use and medical guidance.
How To Use Hair Loss Shampoos
The majority of hair loss shampoos that contain ingredients such as ketoconazole, piroctone olamine, zinc pyrithione, or caffeine are used 2-3 times per week, not daily, unless a dermatologist has advised otherwise. Using a mild, non-medicated shampoo in between use may help avoid cumulative irritation or excessive dryness of the scalp.
You should always follow the directions for use provided on the product label. Generally, hair loss shampoos can be used by wetting the hair and scalp, then lathering a small amount in the hands and applying it mainly to the scalp. Many typically massage gently with the fingertips for around 1-2 minutes, and leave on for a total of 3-5 minutes before rinsing. Rinse thoroughly, as residues may increase the risk of scalp irritation or dryness.
Final Verdict
Most products marketed as hair loss shampoos do not actually regrow hair. They may, however, improve scalp health and hair appearance. Only a small number of shampoo ingredients are currently backed by human clinical trial evidence.
Our top-rated ingredients in this article with the strongest evidence for potential hair-growth benefits include ketoconazole, piroctone olamine, and zinc pyrithione. Other ingredients mentioned in this article, including caffeine, salicylic acid, panthenol, and niacinamide, have been investigated in combination formulation studies. So while the data looks promising, we cannot determine how much benefit comes from each individual ingredient.
Overall, shampoos generally have short contact time with the scalp, limiting their regrowth potential. Hair loss shampoos are best viewed as adjuncts to proven topical or oral products such as minoxidil and finasteride. The next time you see bold hair shampoo marketing claims, keep in mind to focus on the ingredients and the evidence.
References[+]
References ↑1 Evron, E., Juhasz, M., Babadjouni, A., Mesinkovska, N.A., (2020). Natural Hair Supplement: Friend Or Foe? Saw Palmetto, A Systematic Review In Alopecia. Skin Appendage Disorders. 6(6). 329–337. Available at: https://doi.org/10.1159/000509905 ↑2 Völker, J.M., Koch, N., Becker, M., Klenk, A., (2020). Caffeine And Its Pharmacological Benefits In The Management Of Androgenetic Alopecia: A Review. Skin Pharmacology And Physiology. 33(3). 153–169. Available at: https://doi.org/10.1159/000508228 ↑3 Draelos, Z.D., Jacobson, E.L., Kim, H., Kim, M., Jacobson, M.K., (2005). A Pilot Study Evaluating The Efficacy Of Topically Applied Niacin Derivatives For Treatment Of Female Pattern Alopecia. Journal Of Cosmetic Dermatology. 4(4). 258–261. Available at: https://doi.org/10.1111/j.1473-2165.2005.00201.x ↑4 Patel, D.P., Swink, S.M., Castelo-Soccio, L., (2017). A Review Of The Use Of Biotin For Hair Loss. Skin Appendage Disorders. 3(3). 166–169. Available at: https://doi.org/10.1159/000462981 ↑5 Dinh, L., Yan, B. (2023). Oral Drug Delivery Via Intestinal Lymphatic Transport Utilizing Lipid-Based Lyotropic Liquid Crystals. Liquids. 3(4). 456-468. Available at: https://doi.org/10.3390/liquids3040029 ↑6 Celleno, L., Bussoletti, C., Tolaini, M.V., Rossi, A., Ala, L., Becker, M., Völker, J.M., Wiesche, E.S.Z. (2025). A Novel Approach Against Male Pattern Hair Loss With Topical Dimethylglycine Sodium Salt (DMG-Na) and Caffeine: Efficacy of a 24-Week, Double-Blind, Randomized, Placebo-Controlled Trial. J Cosmet Dermatol. 24(8). e70390. Available at: https://doi.org/10.1111/jocd.70390 ↑7, ↑8 Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., Piérard, G.E. (1998). Ketoconazole Shampoo: Effect of Long-Term Use in Androgenic Alopecia. Dermatology. 196(4). 474-7. Available at: https://doi.org/10.1159/000017954 ↑9, ↑12, ↑13 Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., Piérard, G.E. (2002). Nudging Hair Shedding By Antidandruff Shampoos: A Comparison Of 1% Ketoconazole, 1% Piroctone Olamine And 1% Zinc Pyrithione Formulations. International Journal Of Cosmetic Science. 24(5). 249-256. Available at: https://doi.org/10.1046/j.1467-2494.2002.00145.x ↑10 Khandpur, S., Suman, M., Reddy, B.S. (2002). Comparative Efficacy Of Various Treatment Regimens For Androgenetic Alopecia In Men. J Dermatol. 29(8). 489-98. Available at: https://doi.org/10.1111/j.1346-8138.2002.tb00314.x ↑11 Rafi, A.W., Katz, R.M. (2011). Pilot Study Of 15 Patients Receiving A New Treatment Regimen For Androgenic Alopecia: The Effects Of Atopy On AGA. ISRN Dermatol. 2011. 241953. Available at: https://doi.org/10.5402/2011/241953 ↑14 Berger, R.S., Fu, J.L., Smiles, K.A., Turner, C.B., Schnell, B.M., Werchowski, K.M., Lammers, K.M. (2003). The Effects Of Minoxidil, 1% Pyrithione Zinc And A Combination Of Both On Hair Density: A Randomized Controlled Trial. British Journal Of Dermatology. 149(2). 354–362. Available at: https://doi.org/10.1046/j.1365-2133.2003.05435.x ↑15 Celleno, L., Bussoletti, C., Tolaini, M.V., Rossi, A., Ala, L., Becker, M., Völker, J.M., Wiesche, E.S.Z. (2025). A Novel Approach Against Male Pattern Hair Loss With Topical Dimethylglycine Sodium Salt (DMG-Na) And Caffeine: Efficacy Of A 24-Week, Double-Blind, Randomized, Placebo-Controlled Trial. J Cosmet Dermatol. 24(8). e70390. Available at: https://doi.org/10.1111/jocd.70390 ↑16 Bussoletti, C., Tolaini, M.V., Celleno, L. (2020). Efficacy Of A Cosmetic Phyto-Caffeine Shampoo In Female Androgenetic Alopecia. G Ital Dermatol Venereol. 155(4). 492-499. Available at: https://doi.org/10.23736/S0392-0488.18.05499-8 ↑17 Chen, D., Yu, F., Wang, C., Chen, H., Tan, J., Shi, Q., He, X., Liu, X., Wang, F., Zhao, H. (2024). Anti-Hair Loss Effect Of A Shampoo Containing Caffeine And Adenosine. Journal Of Cosmetic Dermatology. 23(9). 2927-2933. Available at: https://doi.org/10.1111/jocd.16347 ↑18 Yaghmaee, R., Mirbagheri, M., Pakdaman, M.H., Rad, F., Gharibi, F. (2016). The Effect Of Minoxidil 1% Shampoo On Androgenetic Alopecia: A Clinical Trial Study. Research Journal Of Pharmaceutical, Biological And Chemical Sciences. 7(5). 856. Available at: https://www.rjpbcs.com/pdf/2016_7(5)/%5B105%5D.pdf ↑19 Kim, H.T., Park, H.S., Kim, Y.M. (2022). Double-Blind Randomized Placebo-Controlled Study Of The Efficacy And Safety Of Hair Loss Prevention Shampoo Containing Salicylic Acid, Panthenol, And Niacinamide In Alopecia Patients. Toxicol Environ Health Sci. 14. 173–185. Available at: https://doi.org/10.1007/s13530-022-00126-9 ↑20 Sudeep, H.V., Rashmi, S., Jestin, T.V., Richards, A., Gouthamchandra, K., Shyamprasad, K. (2023). Oral and Topical Administration of a Standardized Saw Palmetto Oil Reduces Hair Fall and Improves the Hair Growth in Androgenetic Alopecia Subjects – A 16-Week Randomized, Placebo-Controlled Study. Clin Cosmet Investig Dermatol. 16, 3251-3266. Available at: https://doi.org/10.2147/CCID.S435795 ↑21 Rafi, A.W., Katz, R.M. (2011). Pilot Study Of 15 Patients Receiving A New Treatment Regimen For Androgenic Alopecia: The Effects Of Atopy On AGA. ISRN Dermatol. 2011. 241953. Available at: https://doi.org/10.5402/2011/241953 ↑22 Danby, F.W., Maddin, W.S., Margesson, L.J., Rosenthal, D. (1993). A Randomized, Double-Blind, Placebo-Controlled Trial Of Ketoconazole 2% Shampoo Versus Selenium Sulfide 2.5% Shampoo In The Treatment Of Moderate To Severe Dandruff. J Am Acad Dermatol. 29(6). 1008-12. Available at: https://doi.org/10.1016/0190-9622(93)70282-x ↑23 Barbosa, V., Melo, D.F., Vañó-Galván, S., Lutchmanen-Kolanthan, V., Sant’Anna, B., Leclerc-Mercier, S., Reygagne, P. (2024). A Comparative Randomized Clinical Study Assessing The Efficacy Of A 1% Selenium Disulfide-Based Shampoo Versus 2% Ketoconazole Shampoo In Subjects With Moderate To Severe Scalp Seborrheic Dermatitis. Skin Appendage Disord. 10(6). 497-504. Available at: https://doi.org/10.1159/000539209 ↑24 Ortonne, J.-P., Nikkels, A.F., Reich, K., Ponce Olivera, R.M., Lee, J.H., Kerrouche, N., Sidou, F., Faergemann, J. (2011). Efficacious And Safe Management Of Moderate To Severe Scalp Seborrhoeic Dermatitis Using Clobetasol Propionate Shampoo 0.05% Combined With Ketoconazole Shampoo 2%: A Randomized, Controlled Study. British Journal Of Dermatology. 165(1). 171–176. Available at: https://doi.org/10.1111/j.1365-2133.2011.10269.x ↑25 Goyal, A. (2025). Comparative Study Of Ketoconazole-Based Shampoo Versus Coal Tar-Based Shampoo In Seborrheic Dermatitis. Int. J Med. Pharm. Res. 6(4). 208-211 ↑26 Wikimedia Commons, (04 September 2025), Ketoconazole Chemical Structure (SVG). Available at: https://commons.wikimedia.org/wiki/Category:Ketoconazole#/media/File:(2S,4R)-Ketoconazol.svg (Accessed: 20 January 2026) Topical minoxidil has been a cornerstone of androgenic alopecia (AGA) treatment since its FDA approval in the 1980s. With millions of users worldwide, it stands as one of the most widely adopted therapies for hair loss, supported by decades of clinical evidence, and an enormous body of real-world anecdotes, reviews, and before-and-after photos.
However, results with minoxidil can be highly variable, and this variability can be traced in part to biological differences, particularly in the activity of the sulfotransferase enzyme (SULT1A1), which is responsible for converting minoxidil into its active form. Not everyone sulfates minoxidil effectively, meaning that while some achieve meaningful regrowth, others see little to no improvement.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Adding to this challenge is the influence of online topical minoxidil before and after photos (like you can see below), which can create inflated expectations compared to the more modest, probable outcomes seen in practice.
Source: u/United_Speaker3381 via r/tressless.
This article will break through that noise to answer a critical question: when it comes to minoxidil, what is actually the most probable result, not just possible?
First, let’s take a look at what topical minoxidil is.
Refresher: What is Topical Minoxidil?
Topical minoxidil was the first therapy ever approved by the FDA for the treatment of AGA, making it a landmark in the medical management of pattern hair loss.[1]Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786 Introduced in the 1980s, it has since become one of the most widely available and researched treatments across the globe. Its primary action is as a potassium channel opener, which helps to stimulate blood flow, prolong the hair’s growth phase (anagen), and increase follicle size, mechanisms that together can support thicker, denser hair regrowth over time.[2]Messenger, A.G., Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186-194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x
Minoxidil is known as a “pro-drug”, meaning that it needs to be activated within the body to exert its effect. The enzyme responsible for this is called sulfotransferase or, more specifically, SULT1A1. The level of sulfotransferase activity differs depending on the individual, which means that the effectiveness of treatment also differs depending on who you are.[3]Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: a … Continue reading
Formulations typically come in 2% and 5% strengths. The 2% solution was originally FDA-approved for women, while the 5% concentration was approved for men; however, both are now commonly used off-label across genders. Beyond these standard strengths, compounding pharmacies have made higher concentrations available for patients seeking more individualized treatment approaches, with some online telehealth providers (such as Ulo) offering customized formulations. Standard dose topical minoxidil is available over-the-counter in most countries, ensuring accessibility without the need for a prescription.
The Evidence
Topical minoxidil’s robust efficacy for hair regrowth is supported by dozens of high-quality randomized controlled trials (RCTs) and meta-analyses across men and women, consistently demonstrating average increases in hair count of around 10-20% over 6-12 months.
We have collated many of these studies into a table, which you can look at here. However, we have summarised some of the most recent topical minoxidil studies.
Liang et al. (2022)
- Prospective, evaluator-blinded RCT with 120 women with female pattern hair loss (FPHL) assigned to three groups: 5% topical minoxidil alone; 5% minoxidil + oral spironolactone (80-100 mg); and 5% minoxidil and microneedling every 2 weeks.[4]Liang, X., Chang, Y., Wu, H., Liu, Y., Zhao, J., Wang, L., Zhuo, F. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female … Continue reading
- Duration: 24 weeks; outcomes measured included dermascope, ultrasound biomicroscopy, physician, and patient assessments.
- Effective rate at week 2:
- 55.27% (topical minoxidil)
- 86.49% (topical minoxidil and oral spironolactone)
- 95% (topical minoxidil and microneedling)
- Combo therapies (Groups 2&3) outperformed topical minoxidil alone for hair density, shaft diameter, scalp/thickness, shedding, and quality of life; Group 3 showed the greatest benefit.
- Adverse effects were reported across all groups; more menstrual disorders were reported in the spironolactone group. Main limitations: small sample, exclusion of severe cases, possible bias from unblinded treatments.
Hassan et al. (2022)
- Comparative study in 60 women with FPHL: 5% minoxidil topical foam and oral vitamin D3, minoxidil foam only, or vitamin D3 for 12 weeks (plus 15 controls for vitamin D comparison).[5]Hassan, G.F.R., Sadoma, M.E.T., Elbatsh, M.M., Ibrahim, Z.A. (2022). Treatment with oral vitamin D alone, topical minoxidil, or combination of both in patients with female pattern hair loss: A … Continue reading
- Primary outcomes: reduction in thin hair count and Ludwig stage improvement via clinical and dermoscopic evaluation.
- Best results from combination therapy (minoxidil foam and vitamin D3), followed by minoxidil foam alone; vitamin D3 monotherapy had minimal effect.
- Vitamin D3 appeared to enhance minoxidil efficacy, though the sample size was small, and adverse events were not reported.
Bao et al. (2022)
- RCT with 71 men, split into: 5% topical minoxidil twice daily, electrodynamic microneedling every 3 weeks, or topical minoxidil and microneedling combined.[6]Bao, L., Zong, H., Fang, S., Zheng, L., Li, Y. (2019). Randomized trial of electrodynamic microneedling combined with 5% minoxidil topical solution for treating androgenetic alopecia in Chinese males … Continue reading
- Follow-up: 24 weeks, with an additional 6-month observation period.
- All treatments increased non-vellus hair density; the combined group had the greatest improvements in hair density and diameter.
- The combination group received additional scalp massages (potential confounder); 12 participants experienced adverse reactions (dermatitis, dandruff, lymph node enlargement), none severe.
Singh et al. (2020)
- Double-blind, placebo-controlled RCT in 80 men: Groups received either 5% topical minoxidil, 5% topical minoxidil and PRP injections, placebo, or PRP alone.[7]Singh, S.K., Kumar, V., Rai, T. (2020). Comparison of efficacy of platelet-rich plasma therapy with or without topical 5% minoxidil in male-type baldness: A randomized, double-blind placebo control … Continue reading
- 12 weeks’ treatment with 2-month follow-up; outcome: changes in total, terminal, vellus, and non-vellus hair density, pigmentation, and global assessments.
- Highest hair density and satisfaction with topical minoxidil and PRP; both monotherapies are better than placebo, but the combo was most effective.
- Limitations: short-term follow-up, some subjective outcomes; low adverse event rate (mostly mild, e.g., scalp pruritus, facial hair growth).
Overall, when you look across all of the data, consistent benefits are seen with topical minoxidil usage. However, these results are variable. The key reason for this is differences in sulfotransferase activity. As we mentioned before, minoxidil is a pro-drug, and it needs to be enzymatically activated to exert its effect. We will go into more detail about this below.
What’s Possible ≠ What’s Probable
When evaluating treatment outcomes, especially online minoxidil before and after photos, it’s important to separate possibility from probability. The scattered data points below show the full range of what might occur for individuals, while the central trendline reflects the average outcome (i.e., what typically happens).
Figure 1: A graph showing possible and probable results. The data points show possible results, and the trendline shows probable results.[8]Badenhorst CE, Dawson B, Goodman C, Sim M, Cox GR, Gore CJ, Tjalsma H, Swinkels DW, Peeling P. Influence of post-exercise hypoxic exposure on hepcidin response in athletes. Eur J Appl Physiol. 2014 … Continue reading
Online anecdotes often showcase what we would call “hyper-responders” – people who experience dramatic results. These people may also have higher levels of sulfotransferase activity.
Figure 2: Anecdotal results are often way more dramatic than the typical.
These stories, while real, represent outcomes that are well above average. The issue arises when patients focus only on these peak results and expect the same for themselves. If their own progress seems slower or less dramatic, they may stop prematurely or mistakenly believe the treatment is ineffective.
Figure 3: Expected results are often far more dramatic than probable results.
Dramatic results are often showcased more than modest outcomes because of survivorship bias. This bias occurs when only the most striking successes are noticed or shared, while the many average or less impressive results go unreported.
Figure 4: Survivorship bias skews perception of hair loss treatments. Online accounts often highlight extreme outcomes, either exceptional success or very poor results, while the typical, average outcomes experienced by most remain underrepresented.
Why Results Are Variable: The SULT1A1 Enzyme
Minoxidil must be converted into minoxidil sulfate by the sulfotransferase enzyme SULT1A1, which is present in the outer root sheath of hair follicles, to become pharmacologically active and stimulate hair growth.
Several peer-reviewed studies confirm that the level of SULT1A1 activity in scalp follicles is a key determinant of how well a patient responds to minoxidil therapy.[9]Goren, A., Castano, J.A., McCoy, J., Bermudez, F., Lotti, T. (2014). Novem enzymatic assay predicts minoxidil response in the treatment of androgenetic alopecia. Dermatologic Therapy. 27(3). 171-173. … Continue reading Enhancing SULT1A1 activity, such as by using enzyme-boosting adjuvants, has been shown in recent trials to convert a majority of prior non-responders into responders, further confirming the mechanistic role of this enzyme.[10]Chandrashekar, B.S., Chandu, M., Shenoy, C., Chander, A., Roopa, M.S. (2024). SULT1A1 enzyme booster to amplify topical minoxidil response in androgenic alopecia: a single-center prospective study. … Continue reading
This enzyme-based variability explains why two people can apply minoxidil in the same way but see very different outcomes, and why more than half of users may see only limited or no benefit despite consistent application.
Fixing the Enzymatic Problem
A number of evidence-based strategies exist to improve minoxidil response. These include microneeding, retinoic acid co-application, formulation changes, consistency, and combination therapy.
Microneedling
Multiple systematic reviews and clinical trials confirm microneedling significantly improves minoxidil efficacy. A 2023 meta-analysis showed that combining the two led to a significantly greater increase in hair count compared to minoxidil alone, regardless of needle depth or device used. Hair count improvements are notable after 12 to 24 weeks of combined treatment.[11]Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating the efficacy and safety of combined … Continue reading
You can read more about how microneedling improves minoxidil efficacy here.
Retinoic Acid (Tretinoin)
Topical tretinoin has been shown in peer-reviewed studies to upregulate follicular SULT1A1, thereby enhancing the conversion of minoxidil to its active form. In a 2019 study, tretinoin not only improved clinical response in difficult cases but also increased scalp SULT1A1 activity, supporting its role as a mechanistic booster.[12]Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvan, S., Chen, G., Tan, Y., Kovacevic, M. (2019). Tretinoin enhances minoxidil response in androgenetic alopecia … Continue reading
You can see the benefits of adding retinoic acid in our Compare Treatments graph below.
Figure 5: Combining topical minoxidil with retinoic acid significantly boosts regrowth potential.
Learn more about retinoic acid and its effects on sulfotransferase here.
Formulation Adjustments (Foam vs. Solution 2% vs. 5%)
Clinical research shows that 5% minoxidil is more effective than 2% for both men and women. Foam is as effective as a solution but better tolerated, with reduced risk of contact dermatitis due to the absence of propylene glycol. Some evidence suggests liquid formulations might yield slightly higher absorption, but both forms are clinically comparable.[13]Purnak, T., Senel, E., Sahin, C. (2011). Liquid formulation of minoxidil versus its foam formulation. Indian Journal of Dermatology. 56(4). 462. Available at: https://doi.org/10.4103/0019-5154.84714
Consistency and Adherence
Missed doses can reduce topical minoxidil efficacy. Some clinical guidance suggests that missing minoxidil applications even once per week can diminish results, and consistent, daily use is necessary to achieve and maintain hair regrowth.[14]Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy. 13(5). 1157-1169. Available at: … Continue reading
Combination Therapy
Combining minoxidil with finasteride/dutasteride, PRP, low-level laser therapy (LLLT), or, in resistant cases, oral minoxidil, provides superior results versus monotherapy. Meta-analyses and randomized trials consistently find greater improvement in hair density, count, and patient satisfaction when using combination regimens.[15]Xia, Y., Chen, H., Chen, Y., Chen, Z. (2025). Relative efficacy of minoxidil in combination with other treatments for androgenic alopecia: a network meta-analysis based on randomized controlled … Continue reading
So, what do online topical minoxidil before and after photos look like?
Online Topical Minoxidil Successes
Now that we have discussed how topical minoxidil works, potential regimens, and likely timelines, here are 10 reports published by Reddit users with before and after photos.
Disclaimer: The stories below are unverified anecdotes. Consequently, they illustrate individual outcomes and should not be construed as clinical proof or medical advice.
Case 1: Male (age unknown), 6 months on topical minoxidil monotherapy (5% once daily)
Source: u/Dual270x via r/tressless.
This individual applied 5% topical minoxidil once daily for six months. They chose topical minoxidil out of a general preference for avoiding oral medications. From the before-and-after photos, diffuse thinning can be observed from the center of the crown, and the protocol yielded a noticeable increase in crown density by month six. No side effects or early shedding have been reported.
Case 2: Female (age unknown), about 5 months on topical minoxidil monotherapy (5% once nightly)
Source: u/maddogyr via r/FemaleHairLoss.
This user applied 5% topical minoxidil once nightly for about 5 months, using a liquid solution that she purchased over the counter after a telemedicine consult. She stated that she had AGA and did not note any other treatment. Based on both her comments and photos, she experienced marked improvement in hair density and coverage, stating that minoxidil “saved her hair” and that she wishes she had started earlier. She did not describe any notable early shedding, but noted dry scalp and some dandruff as potential side effects.
Case 3: Male (age unknown), 60 days on topical minoxidil monotherapy (5% foam nightly)
This Redditor applied 5% minoxidil foam once nightly before bed for two months without any concurrent treatment. He reports the typical amount applied daily as “one cap-full”, rinsed out the next morning. The user has Crohn’s disease with prior nutrient absorption issues, and chose to avoid ifnasteride due to side-effect concerns. He had about one year of noticeable thinning before starting. Upon starting minoxidil, shedding increased for the first two weeks, then eased. The first measurable improvement was visible by day 30, with clear density gains at day 60. The crown region improved the fastest, while hairline changes were slower.Source: u/[deleted] via r/tressless.
Case 4: Female (age unknown), 1 year 5 months on topical minoxidil foam (5% once daily)
Source: u/Hyper-Fang via r/FemaleHairLoss.
This user had AGA with prior telogen effluvium, reporting diffuse thinning along the part and temples and a reportedly high daily shedding. She began therapy with Rogaine Men’s Foam 5% once nightly, supported by Ketoconazole 2% shampoo once weekly. At 2 months of treatment, “peach fuzz”/baby hairs were visible at the hairline and temples. At 6 months, there was a noticeable density across the top with new hairs evident but still short. After 1 year and 5 months of therapy, she notes ongoing thickening and widespread new hair growth. She also reported that her shower shed decreased from ~70 hairs to ~10-15, with visual density improving at the part and temples. She reported a mild scalp itch at the start and subtle peach-fuzz on the upper lip and chin as side effects.
Case 5: Male (age unknown), 2.5 months on topical minoxidil (5% liquid 1 mL twice daily)
Source: u/pcybits via r/tressless.
This male user, likely with AGA, reported recession at the hairline and thinning at the crown after 2.5 months of using a Rogaine-style 5% liquid at 2 mL/day, without any concurrent DHT blocker. He experienced an early shedding phase ~1 month into treatment, which stabilized with continued use. He exhibited visible thickening at the hairline and crown by 2.5 months of therapy. He did not report any systemic or local side effects and plans to continue minoxidil while considering adding weekly microneedling and/or topical finasteride if progression resumes.
Other Redditors have used combination treatments to further improve their hair regrowth. As you can see below, some of the results look dramatic.
Case 6: Male (age unknown), 3 months on topical combo (minoxidil 5% and finasteride 0.1% once nightly)
Source: u/United_Speaker3381 via r/tressless.
This user had thinning at the hairline and vertex areas of the scalp and began treatment to improve hair density and maintenance. He applied a single compounded solution (Novegrow) containing minoxidil 5% and finasteride 0.1%, once nightly, for three months before reporting on progress. Additionally, he dermarolled his scalp with a 0.5 mm needle twice weekly and supplemented with daily biotin and a multivitamin. He reported mild shedding in the first two weeks of therapy, and by month 3, he noted visible thickening and improved coverage. He did not report any side effects and added that the once-daily schedule was practical and helped with adherence.
Case 7: Male (25), 11 months on combination therapy (finasteride 1 mg daily and topical minoxidil foam 5% once daily)
Source: u/Jordan_cadagan via r/tressless.
This Redditor exhibited typical AGA with frontal recession and diffuse vertex thinning. His core regimen is oral finasteride 1 mg daily and minoxidil 5% foam (Kirkland) once daily. He noted he took regular ice/cold baths, ensured an increased water intake, engaged in exercise, avoided junk food, and did not smoke. He saw noticeable thickening from around 6 months after several shedding cycles. By month 11, he experienced substantial cosmetic improvement, with some commenters even comparing the results to transplant-level density. He did not report any adverse effects from finasteride or minoxidil, and subjectively felt less “brain fog” after starting finasteride.
Case 8: Male (37), 4 months on topical minoxidil 5% and topical finasteride 0.1% (1 mL nightly)
Source: u/Far-Instance268 via r/tressless.
This male had crown-predominant thinning with a stable hairline at baseline. His core regimen is a single combined solution containing minoxidil 5% and finasteride 0.1%, applied to the crown only. He did not report any microneedling or supplementation. He reported that shedding began around week 3 and was “heavy” for approximately 4 weeks. The shedding tapered and largely stopped at the 2.5-month mark. By month 4, he achieved visible crown regrowth and thickening compared to the baseline, despite the application being limited to the crown. He perceived an increase in overall density and did not report any side effects.
Case 9: Male (34), about 12 months on topical finasteride 0.3% and minoxidil 6% (twice daily)
Source: u/iTsundere via r/tressless.
This male reported on his progress after using a combination of topical finasteride 0.3% and minoxidil 6% treatment twice daily for nearly one year. He first experienced hair thinning and hairline recession beginning in his late 20s. Alongside the topical treatments, he also applied a dermastamp at approximately 1.5 mm every 10 days. He reported significant shedding during the first two months of treatment along with mild chest tenderness in the first month, which resolved after temporarily halving the topical dose. He reported month-on-month thickening, with clear improvement by months 7-8 and robust density by the one-year mark.
Case 10: Male (22), 90 days on finasteride 1 mg and minoxidil 5% twice daily plus dermarolling
Source: u/M3ga_Shniz via r/tressless.
This user with early temple recession began a combination therapy of finasteride 1 mg oral daily, minoxidil 5% foam twice daily, and dermaoll 1.5 mm weekly. He experienced noticeable shedding at weeks 3-6 of therapy, and at the 90-day progress mark, he reported visible filling in of the temple and general thickening of the hair. He did not report any side effects and noted that pushing through the initial shedding phase was crucial to his progress thus far.
Probable Regrowth Timeline
Based on the available evidence, we have mapped out a likely regrowth timeline (although some may experience results faster or later).
Months 0-3: Initial shedding
- Temporary shedding may occur as follicles shift into a new growth phase.
- This usually resolves within the first 4-8 weeks.
Months 3-6: Early cosmetic improvements
- Early visible improvements, such as reduced shedding and appearance of fine new hairs, can be expected.
- Thicker, darker regrowth starts to become apparent for most users during this period.
Months 6-12: Peak cosmetic changes
- Cosmetic changes peak, with stronger hair, increased density, and the greatest coverage observed.
- This is the period of maximum regrowth for most individuals.
Months 12+: Plateau, maintenance, & slight regression
- Further gains plateau, with 5-year studies showing hair counts remaining above baseline –– but with some loss in “peak” results from year one. Continued daily use is needed to maintain improvements, as stopping treatment leads to a gradual reversal within a few months.
Safety and Side Effects
Local side effects are relatively common with topical minoxidil, especially with the liquid formulations. The principal culprits are propylene glycol and ethanol, which frequently cause redness, itching, dryness, and a dandruff-like scaling of the scalp.[16]Makhlouf, A., Elnawawy, T. (2023). Hair regrowth boosting via minoxidil cubosomes: Formulation development, in vivo hair regrowth evaluation, histopathological examination and confocal laser … Continue reading Patients with sensitive skin are particularly prone to these adverse reactions and may experience contact dermatitis, which can make long-term use uncomfortable.
The foam formulation is generally better tolerated because it lacks propylene glycol. Most clinical trials and user surveys report fewer cases of scalp irritation, itching, or flaking with foam compared to the liquid version.[17]Purnak, T., Senel, E., Sahin, C. (2011). Liquid formulation of minoxidil versus its foam formulation. Indian Journal of Dermatology. 56(4). 462. Available at: https://doi.org/10.4103/0019-5154.84714
While systemic absorption of topical minoxidil is rare, some users may experience unwanted side effects such as dizziness and the growth of facial or body hair away from the application site. These effects are more likely if the medication is used excessively or applied to broken or abraded skin, which can increase absorption. We do not recommend using topical products if you have an inflammatory scalp condition – deal with that problem first, and then take a look at topical hair regrowth products.
Overall, when used as directed, topical minoxidil has a strong safety profile, with most side effects limited to mild local irritation.
Practical Takeaways
- Topical minoxidil is effective and well-established, but clinical results vary between individuals.
- Most patients achieve stabilization of hair loss with modest regrowth, though complete restoration is uncommon.
- Response depends partly on scalp sulfotransferase enzyme activity, which explains why some non-responders see little or no improvement.
- Best results come with consistent, long-term use and may be enhanced by adjuncts such as microneedling or topical retinoids to improve absorption and efficacy.
Final Thoughts
When evaluating minoxidil, it is important to recognize it as the most widely accessible and evidence-backed treatment for androgenetic alopecia. While outcomes can vary, most users see stabilization and modest regrowth rather than dramatic reversal. Success depends on realistic expectations, consistent use, and optimized regimens that may include adjunctive therapies. Applying a framework that emphasizes the likely trendline over anecdotal extremes can help you stay committed, avoid discouragement, and make informed decisions grounded in evidence and personal treatment goals.
References[+]
References ↑1 Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug Design, Development and Therapy. 13. 2777-2786 ↑2 Messenger, A.G., Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186-194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑3 Pietrauszka, K., Bergler-Czop, B. (2020). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: a review. Advances in Dermatology and Allergology. 39(3). 472-478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑4 Liang, X., Chang, Y., Wu, H., Liu, Y., Zhao, J., Wang, L., Zhuo, F. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A Prospective, Single-Center, Parallel-Group, Evaluator Blinded, Randomized Trial. Frontiers in Medicine. 11(9). 905140. Available at: https://doi.org/10.3389/fmed.2022.905140 ↑5 Hassan, G.F.R., Sadoma, M.E.T., Elbatsh, M.M., Ibrahim, Z.A. (2022). Treatment with oral vitamin D alone, topical minoxidil, or combination of both in patients with female pattern hair loss: A comparative clinical and dermoscopic study. Journal of Cosmetic Dermatology. 21(9). 3917-3924. Available at: https://doi.org/10.1111/jocd.14743 ↑6 Bao, L., Zong, H., Fang, S., Zheng, L., Li, Y. (2019). Randomized trial of electrodynamic microneedling combined with 5% minoxidil topical solution for treating androgenetic alopecia in Chinese males and molecular mechanistic study of the involvement of the Wnt/ꞵ-catening signaling pathway. Journal of Dermatological Treatment. 33(1). 483-493. Available at: https://doi.org/10.1080/09546634.2020.1770162 ↑7 Singh, S.K., Kumar, V., Rai, T. (2020). Comparison of efficacy of platelet-rich plasma therapy with or without topical 5% minoxidil in male-type baldness: A randomized, double-blind placebo control trial. Indian Journal of Dermatology, Venereology, and Leprology. 86(2). 150-157. Available at: https://doi.org/10.4103/ijdvl.IJDVL_589_18 ↑8 Badenhorst CE, Dawson B, Goodman C, Sim M, Cox GR, Gore CJ, Tjalsma H, Swinkels DW, Peeling P. Influence of post-exercise hypoxic exposure on hepcidin response in athletes. Eur J Appl Physiol. 2014 May;114(5):951-9. doi: 10.1007/s00421-014-2829-6. Epub 2014 Feb 1. PMID: 24487960. ↑9 Goren, A., Castano, J.A., McCoy, J., Bermudez, F., Lotti, T. (2014). Novem enzymatic assay predicts minoxidil response in the treatment of androgenetic alopecia. Dermatologic Therapy. 27(3). 171-173. Available at: https://doi.org/10.1111/dth.12111 ↑10 Chandrashekar, B.S., Chandu, M., Shenoy, C., Chander, A., Roopa, M.S. (2024). SULT1A1 enzyme booster to amplify topical minoxidil response in androgenic alopecia: a single-center prospective study. International Journal of Research in Medical Sciences. 12(11). Available at: https://doi.org/10.18203/2320-6012.ijrms20243362 ↑11 Ahmed, K.M.A., Kozaa, Y.A., Abuawwad, M.T., Al-Najdawi, A.I., Mahmoud, Y.W., Ahmed, A.M., Taha, M.J.J., Fadhli, T., Giannopoulou, A. (2025). Evaluating the efficacy and safety of combined microneedling therapy versus topical Minoxidil in androgenetic alopecia: a systematic review and meta-analysis. Archives of Dermatological Research. 317(1). 528. Available at: https://doi.org/10.1007/s00403-025-04032-1 ↑12 Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvan, S., Chen, G., Tan, Y., Kovacevic, M. (2019). Tretinoin enhances minoxidil response in androgenetic alopecia patients by upregulating follicular sulfotransferase enzymes. Dermatologic Therapy. 32(3). E12915. Available at: https://doi.org/10.1111/dth.12915 ↑13, ↑17 Purnak, T., Senel, E., Sahin, C. (2011). Liquid formulation of minoxidil versus its foam formulation. Indian Journal of Dermatology. 56(4). 462. Available at: https://doi.org/10.4103/0019-5154.84714 ↑14 Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy. 13(5). 1157-1169. Available at: https://doi.org/10.1007/s13555-023-00919-x ↑15 Xia, Y., Chen, H., Chen, Y., Chen, Z. (2025). Relative efficacy of minoxidil in combination with other treatments for androgenic alopecia: a network meta-analysis based on randomized controlled trials. Frontiers in Medicine. 12. Available at: https://doi.org/10.3389/fmed.2025.1638496 ↑16 Makhlouf, A., Elnawawy, T. (2023). Hair regrowth boosting via minoxidil cubosomes: Formulation development, in vivo hair regrowth evaluation, histopathological examination and confocal laser microscopy imaging. International Journal of Pharmacology. 634. 122665. Available at: https://doi.org/10.1016/j.ijpharm.2023.122665 IngredientsPharmaceuticalsNatural RemediesCompany ReviewsBy Cassie Hopton, PhDMay 15, 2026Does Dandruff Cause Hair Loss?
Dandruff is more than a cosmetic nuisance: it often reflects scalp inflammation that can disrupt the hair cycle. While dandruff itself doesn’t directly cause hair loss, it may trigger shedding and worsen conditions like pattern hair loss. Understanding the root cause of dandruff and treating it effe...By Perfect Hair Health TeamMay 15, 2026Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
Since earning FDA approval in 1997, Finasteride has been one of the most widely used hair loss drugs. To avoid potential sexual side effects, many opt for topical finasteride in hopes of isolating the drug’s DHT-reducing effects on the scalp. However, at certain doses, topical finasteride can enter ...By Dr. Michael WilliamsMay 9, 2026Ulo Review: #1 Customized Hair Loss Treatment in 2026
Ulo, co-founded by Perfect Hair Health, was created to address long-standing issues in the hair loss industry, from unsafe formulations to misleading personalization claims. In this review, we examine whether Ulo’s evidence-first, highly customizable approach truly sets a new benchmark for hair loss...By Sophie Grice, PhDMay 8, 202610 Best Hair Loss Treatments for Men
If you are at the start of your hair-loss journey, figuring out where to begin can feel overwhelming. There is no shortage of products and treatments that promise results, but not all of them are worth your time and money. In this article, we break down 10 of the best hair loss treatments for men, i...By Sarah King, PhDMay 7, 2026Best Minoxidil for Men: Top Picks for 2026
With countless hair loss products on the market, choosing the right minoxidil can be overwhelming. This evidence-based guide ranks the best minoxidil treatments for men in 2026, including prescription and over-the-counter options, based on effectiveness, safety, customization, and value. Discover wh...By Catherine Kennedy, PhDMay 1, 2026Does Ozempic Cause Hair Loss?
GLP-1 medications like Ozempic are transforming weight-loss treatment. But viral posts and patient reports have raised concerns about unexpected hair shedding, and emerging research hints there may be something to it. But what does the science really show? We unpack the latest evidence to examine wh...By Dr. Michael WilliamsApr 30, 2026Does Masturbation Cause Hair Loss?
Theories about the causes of hair loss abound online. A potential link with masturbation is among the more curious of these ideas. Interestingly, there is a range of molecular mechanisms that may form the biological basis for the connection between masturbation and hair loss. But do they stand up to...By Cassie Hopton, PhDApr 24, 20266 Best Hair Loss Treatments for Women
Female pattern hair loss treatments vary widely in effectiveness, safety, and suitability across life stages. In this evidence-based guide, we review six of the best options for women, from minoxidil to low-level laser therapy, explaining how they work, the clinical evidence behind them, key safety ...By Sophie Grice, PhDApr 21, 202610 Best Shampoos for Thinning Hair Men
If you’re looking into products for thinning hair, shampoos are often one of the first options to consider. They’re quick, convenient, and easy to add into a routine. But a lot of these products rely on heavy marketing, and only a handful of ingredients actually have clinical evidence behind them in...By Dr. Michael WilliamsApr 17, 2026A New Way to Regrow Hair? Inside the Science of PP-405
PP-405 is a novel topical hair loss treatment developed by Pelage Pharmaceuticals. By targeting the hair follicle stem cell metabolism, the drug aims to “wake up” dormant hair follicle stem cells and drive new hair growth. Early clinical trial results have generated significant excitement, but with ... - Mission Statement
 Scroll Down
Scroll Down