- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Musely Reviews for Hair Loss: Are They Trustworthy?
-
Best Minoxidil for Women: Top 6 Brands of 2026
-
Best Topical Finasteride: 5 Top Products of 2026
-
Best Topical Dutasteride: 8 Choices for 2026
-
10 Best Shampoos for Thinning Hair Men
-
Does Semaglutide Cause Hair Loss?
-
Does Dandruff Cause Hair Loss?
-
Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
ArticlesFecal Microbiota Transplants (FMT) & Accidental Hair Regrowth: Studies, Anecdotes, & Photos
First Published Feb 25 2021Last Updated Sep 19 2025PharmaceuticalResearched & Written By:Perfect Hair Health TeamReviewed By:Rob English, Medical EditorWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
Studies suggest that fecal microbiota transplants (FMTs) can recolonize the gut microbiome, dampen autoimmunity (i.e., allergies), and even save people’s lives from C. difficile infections. Fascinatingly, some people who’ve undergone an FMT have later reported an unintended benefit: hair regrowth. This article explores the science connecting stool transplants and the microbiome to hair loss disorders like alopecia areata, telogen effluvium, and even androgenic alopecia (AGA). It’s possible that FMTs may become a new frontier for hair loss research. Read on to uncover the science, anecdotes, and before-after photos demonstrating hair regrowth.
Full Article
Stool transplants – also known as fecal microbiota transplants (FMT) – are a controversial therapy reserved for life-threatening bacterial infections and autoimmune disorders. Fascinatingly, people who’ve undergone stool transplants have later reported unintended benefits: weight loss, less acne, personality improvements, and even hair regrowth.
At face value, a connection between stool transplants and hair loss sounds like science fiction. How could altering the bacteria inside of our guts affect hair growth on top of our scalps?
At the same time, new research suggests that the hair loss-fecal microbiota connection is very real, and may even become a future therapeutic target for people looking to regrow hair.
In this article, we’ll dive into the science surrounding stool transplants: what this therapy is, what it does to our gut microbiome, and the evidence linking fecal microbiota transplants to hair regrowth in alopecia areata, telogen effluvium, and maybe even androgenic alopecia.
We’ll even showcase a few before-after photos featured in studies and forums from people who received stool transplants to treat unrelated health conditions, and ended up (accidentally) experiencing hair regrowth.
If you have any questions, feel free to reach us in the comments below.
Table of contents
All-Natural Hair Supplement + Topical
The top natural ingredients for hair growth, all in one serum & supplement.
Take the next step in your hair growth journey with a world-class natural serum & supplement. Ingredients, doses, & concentrations built by science.

What is a fecal microbiota transplant (i.e., stool transplant)?
Our guts are like a storage facility for large colonies of microbes. Because of the role the gut microbiome encompasses, many diseases and conditions that relate to a dysfunctional gut microbiome, have now been treated utilizing a fecal microbiota transplant.
An artist’s rendering of gut organisms and debris
A fecal microbiota transplant (FMT) is basically what it sounds like: the transfer of stool from one individual’s colon into another person. The aim? To improve gut bacteria density and diversity. In other words: increase the total number of “good” bacteria in the gut, as well as the number of “good” species.
Stool transplants – or FMTs – are nothing new. This practice has been around since the 4th century. More recently, it gained notoriety when the FDA approved FMTs for recurrent Clostridium difficile infections – a deadly bacterial infection that often grows resistant to antibiotics [1].
In this context, FMT’s are incredibly successful. For a benchmark, antibiotic treatments for C. difficile tend to boast a 20-30% remission rate. FMTs boast around a 90% remission rate [2]. In the veterinary world, FMT’s are used for similar purposes [3].
While the use of a FMT seems most pertinent to gastrointestinal diseases, there is a much broader spectrum for the therapy in relation to other conditions, such as multiple sclerosis, Alzheimer’s disease, epilepsy, obesity, and metabolic syndrome (to name only a few) [4, 5].
Can fecal microbiota transplants alter our gut microbiome?
FMTs are perhaps one of the most powerful ways to alter our gut microbiome: the collection of bacteria inside our intestines that help regulate anything from autoimmune reactions to hormonal levels to nutrient metabolism.
C. difficile & stool transplants: alterations to gut flora
In one study, researchers decided to investigate 55 stool samples from donors and recipients who used FMTs as a treatment for C. difficile infections. They found major differences in the pre- versus post-procedure samples of the recipients [6].
Before the procedure, healthy donors’ stools contained a significant portion of the phylum of bacteria – known as Firmicutes and Bacteroides – constituting roughly 85% of bacteria identified.
We can first think of a phylum as the link between different species of bacteria. For example, while an oak tree differs from a palm tree, ultimately, they’re still all trees. As such, no one group can be labeled as good or bad as often the species within those groups can exhibit both helpful and disruptive properties. This is evident with helminth infections – where the same helminth can cause inflammation in the first-world but protect from malaria in the developed world. To put it simply: the effects of a bacteria often rely on its environment, the health of an individual, the ratio of that bacteria to others, and about a billion other factors [7,8].
However, in the case of resistant Clostridium infections, Firmicutes and Bacteroides tend to be absent. This suggests that these phylum may play a role in preventing gut dysbiosis and thereby C. difficile overgrowths.
With respect to the study, here’s what researchers found:
- Prior to the stool transplants, recipients’ stools had lower amounts of Firmicutes and Bacteroides and markedly high Fusobacteria and Proteobacteria.
- Donor stool contained exceptionally higher Firmicutes and Bacteroides, as well as lower Fusobacteria and Proteobacteria.
- After treatments, recipients’ stool showed the higher Firmicutes and Bacteroides as well as lower Fusobacteria and Proteobacteria (thereby more closely resembling that of the donor’s gut).
- After transplantation, the more closely a recipient’s gut matched that of its donor, the more likely that recipient was to experience a remission from C. difficile.
In other words, FMTs significantly altered gut flora – and to the benefit of sick patients.
Interestingly, the health of our gut microbiome is linked to many hair loss disorders – particularly alopecia areata and telogen effluvium. Moreover, research now links gut bacteria to the regulation of DHT – the hormone implicated in pattern hair loss.
We’ll dive into this evidence below: what it is, what it means, and why it might rewrite a lot of what we think we know about hair loss.
Stool transplants for hair loss: the evidence
When it comes to FMTs as a treatment for hair loss, evidence is very limited. This is because reports of fecal transplants and hair regrowth have mainly happened by accident – specifically, after a patient with C. difficile + hair loss receives a stool transplant and later inadvertently sees improvements to both conditions.
Nonetheless, there is compelling evidence of a gut microbiome-hair loss connection. To best outline this evidence, we’ll dive into the research on FMT’s and hair loss as organized by :
- Alopecia Areata
- Telogen Effluvium
- Androgenic Alopecia
Let’s begin.
Alopecia areata and fecal microbiota transplants
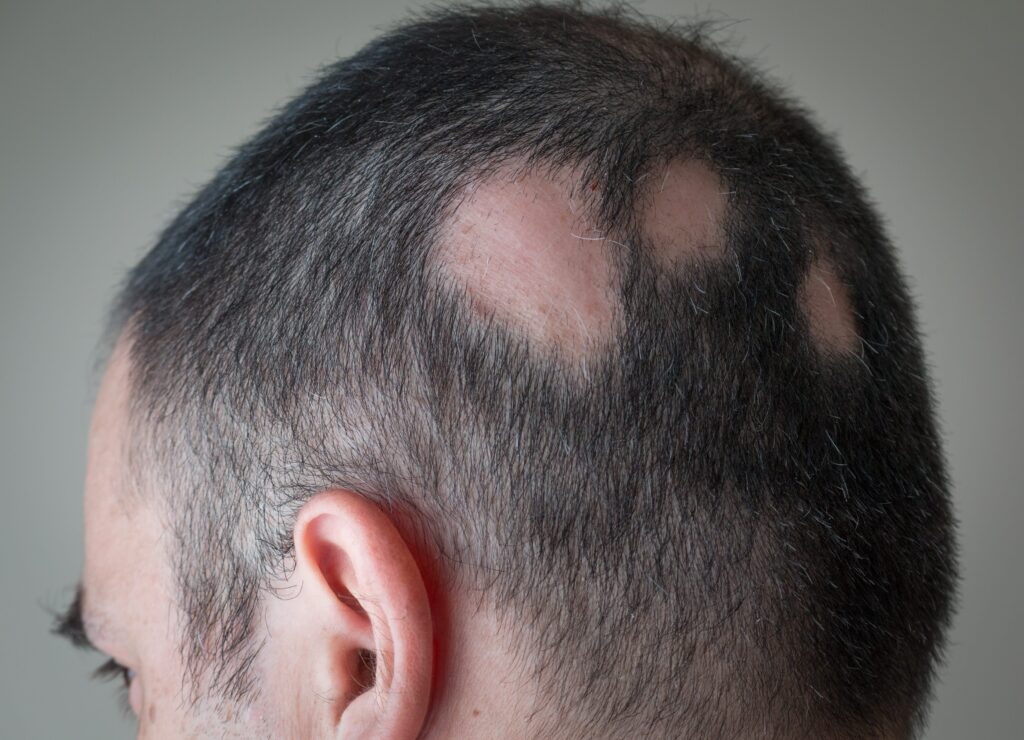
Alopecia areata (AA) is an autoimmune form of hair loss; it often presents as patchy-related hair loss in the scalp. Researchers currently believe AA is the result of a collapse in “immune privilege” of the hair follicles – whereby the immune system begins to read hair follicles as foreign invaders, and then starts to attack them.
Alopecia areata: patchy hair loss
When it comes to stool transplants, alopecia areata is the best-studied form of hair loss. This isn’t saying much – because there’s not a ton of data on stool transplants and hair regrowth in general. Again, the studies here have been unintended and inadvertent: people with alopecia areata seem to have higher rates of gut dysbiosis, and thereby they seem to suffer disproportionately from infections like C. difficile. Therefore, they’re probably more likely to eventually become eligible for extreme treatments like a stool transplant – which is why we have case reports of FMTs and AA-related regrowth in the first place.
So, what limited evidence do we have on stool transplants for hair regrowth from alopecia areata?
Case reports of FMTs and hair regrowth from alopecia areata
Study #1: two uncontrolled retrospective case reports
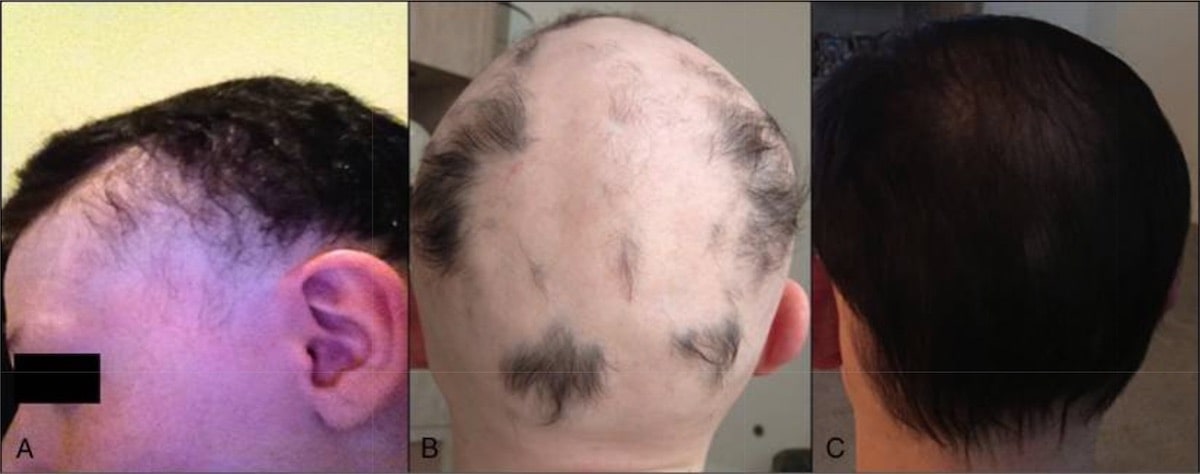
In a study where two patients received an FMT for treating Clostridium difficile, researchers reported that both people saw an unintended benefit of hair regrowth following the procedure [14]. In these cases, both patients were classified as having alopecia universalis – an advanced form of alopecia areata where the hair loss has progresses beyond the scalp and to the body.
In the first case, the 38-year old man began to grow peach fuzz not only on his head, but also his face and arms. The regrowth became cosmetically noticeable 8 weeks after the stool transplant – after having suffered from progressive alopecia universalis for decades without any improvements.
In the second case, a 20-year old man – diagnosed with alopecia universalis 2 years prior to his FMT – saw major improvements to scalp hair regrowth over a 1.5-year period after receiving a stool transplant. Prior to the FMT, this subject had tried to treat his alopecia using corticosteroid injections, topical steroids, squaric acid, and laser treatments— all to no avail. While this man also received steroid injections within his scalp after the FMT, he began to grow hair throughout the rest of his body as well, even where had not received a steroid injection [21].
Study #2: a case report of FMT + hair regrowth in an elderly patient with diffuse alopecia areata
In a second study investigating FMT’s effects on an elderly man (86-year old) with diffuse alopecia areata, researchers found some unexpected benefits [15]. Originally, FMT was used to treat his intestinal disorders (with success). However, alongside the treatment of his other digestive ailments, his hair thickness began to return. What’s more, his previously gray hair had regrown as its original color.
These are just three case reports. That’s not really “overwhelming” evidence…
You’re right! We always have to evaluate any intervention in respect to its quality and quantity of evidence. In regard to FMT and alopecia areata, the quality and quantity of evidence is low: three uncontrolled, retrospective case reports.
Having said that, we also have to recognize that the total number of alopecia areata patients who (1) develop C. difficile, and (2) have it progress to the severity where they become eligible for an FMT – is also low. On that note, the evidence here has excited other research teams enough to launch a clinical trial on FMT and alopecia areata – the results of which will (hopefully) be available in the coming year.
So, needless to say, the preliminary evidence (and mechanistic data) supporting a connection between is FMT and AA-related regrowth is exciting enough to continue exploring.
How might FMT’s improve alopecia areata?
Alopecia areata is mainly categorized by an autoimmune attack on the hair follicles. This is predominantly mediated by a group of immune cell known as T-cells. Specifically, a type of T-cell known as TH17 [16].
Not surprisingly, TH17 is a major contributor to intestinal conditions that manifest as inflammation. Since the gut is our largest site of immune cell concentration, then by altering the gut microbiome to a more favorable anti-inflammatory profile, we may be able to alter T-cell activity throughout the rest of our body.
In fact, studies have shown that modulating the microbiome to favor this anti-inflammatory profile can nearly shutdown TH17 cell activity [17]. By this very same mechanism, it is possible that a more widespread shutdown of TH17 could alleviate a TH17-mediated attack on hair follicles – even at the top of the scalp.
Furthermore, there is an established link between inflammatory bowel disease and alopecia areata [18]. The implication: an unhealthy microbiome may drive both inflammatory bowel disease and alopecia areata. After all, patients with alopecia areata and inflammatory bowel conditions who undergo FMT seem to inadvertently regrow hair – and at a consistency worth noting by investigators.
Summary so far: Alopecia areata (AA) is an autoimmune form of hair loss; it’s likely driven by the over-activation of T-cells, and specifically, TH17 cells. Interestingly, patients with AA who’ve undergone stool transplants (FMTs) to treat C. difficile infections have inadvertently regrown hair. The gut microbiome is the body’s largest site of immune cell concentration – and thereby TH17 activation. So, it’s plausible that FMTs might dampen TH17 immune activation – all by restoring gut microflora balance to a more commensal state.
Telogen effluvium and stool transplants
Telogen effluvium (TE) is a temporary form of hair loss that is characterized by a dysregulation of the hair cycle.
This can occur in many ways: for instance, too many hairs can “shed” prematurely, or there can be a delay between when a hair sheds out and when a new hair grows in (to start a new hair cycle). This often presents as diffuse thinning, or sometimes even region-specific shedding (oftentimes the hairline for women).
A hair loss patient with suspected telogen effluvium (TE)
What causes telogen effluvium (TE)?
Telogen effluvium (TE) is sort of like a catch-all diagnosis for a wide array of hair loss triggers. But the main drivers of TE are often considered (1) stress (emotional or physical), (2) nutrient imbalances, and (3) chronic conditions (i.e., hypothyroidism, heavy metal toxicities, etc.).
Currently, there are no studies investigating a link between FMTs and TE. However, there is mechanistic evidence that FMTs may help address some (of the many) underlying factors leading to TE.
For example, nutrient deficiencies – namely, iron, zinc, and/or vitamin D – have been associated with telogen effluvium [20]. Given that evidence implicates the state of the microbiome in nutrient absorption, it’s possible that disruptions to the microbiome may result in poor nutrient absorption and, consequently, telogen effluvium-related hair shedding [21].
Research has also found elevated cadmium levels in some people with TE – a trace element that may trigger hair shedding if consumed in excness [22]. Most interesting, though, is that excessive cadmium has also been shown to alter the gut microbiome of mice in a way that directly mirrors individuals plagued by antibiotic-resistant Clostridium difficile infections… the exact condition FMT is designed to treat [23].
Altogether, direct (i.e., by bacterial infection) or indirect (i.e., by excess cadmium) changes to the microbiome, both of which seem to be associated with telogen effluivum, may benefit from FMT.
Summary so far: Telogen effluvium (TE) is a temporary form of hair loss caused by a disruption to the hair cycle – oftentimes resulting from stress, nutrient imbalances, or a chronic condition. While no evidence directly implicates stool transplants as a therapeutic procedure for TE, there is mechanistic data showing that our gut microbiome has the capacity to improve chronic conditions as well as the synthesis of vitamins commonly found as deficient in TE patients. Therefore, it’s possible that stool transplants might help TE by addressing its underlying causes. But again, we’re extrapolating here!
Androgenic alopecia and fecal transplants
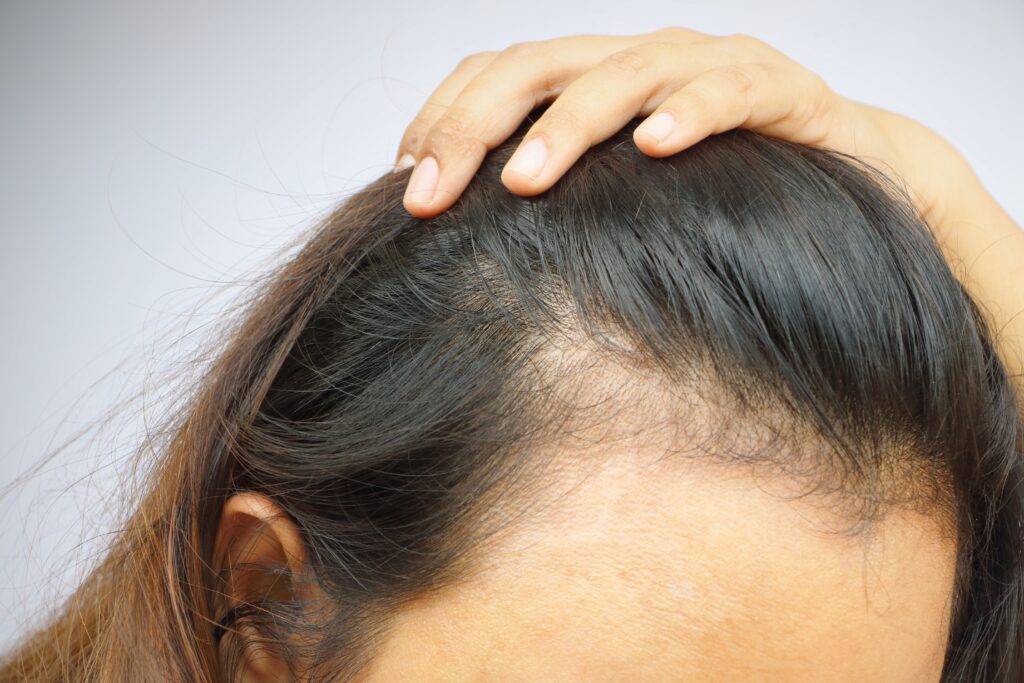
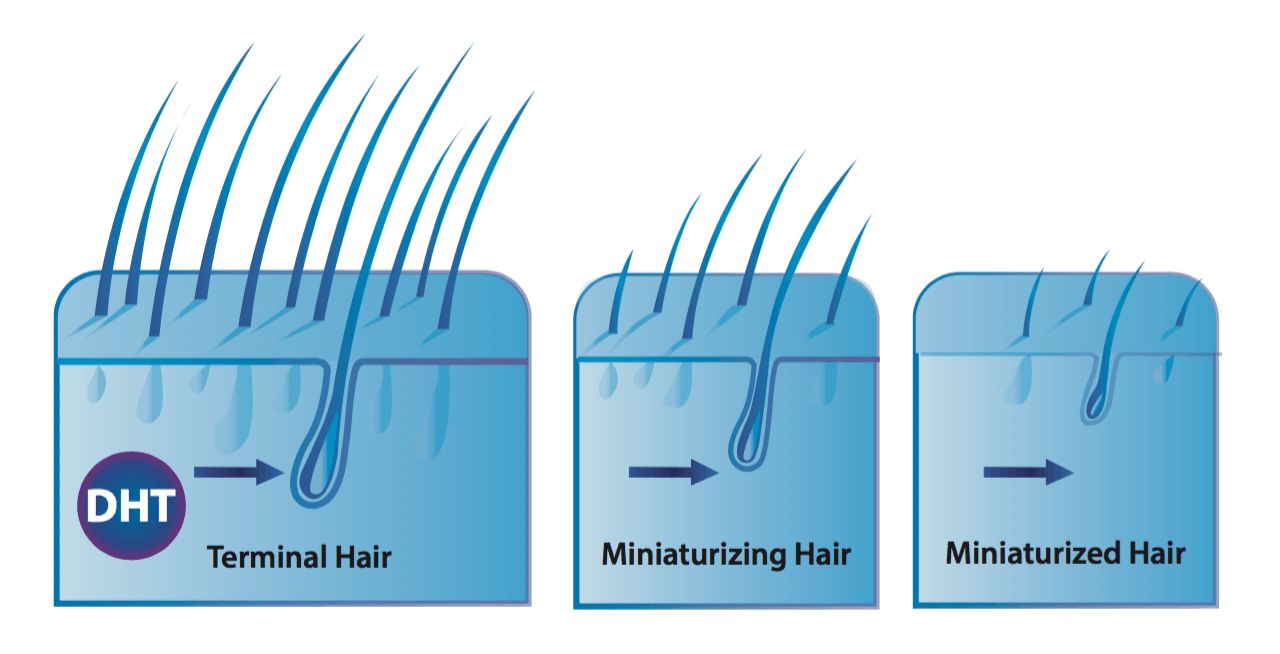
Androgenic alopecia (AGA) is one of the world’s most common hair loss disorders – affecting at least 50% of women and 80% of men throughout a lifetime. Its often characterized by progressive hair follicle miniaturization, whereby affected hair follicles get thinner and thinner over a series of hair cycles. It presents most commonly across the top of the scalp as temple recession + a bald spot in men, and diffuse thinning in most women.
Androgenic alopecia in a male: temple recession
What causes androgenic alopecia (AGA)?
While all of the causes aren’t fully elucidated, most researchers agree that AGA is caused by a combination of male hormones and genetics, and possibly the scalp’s environment (i.e., inflammatory microorganisms, the contraction of muscles surrounding the scalp perimeter, inflammation-mediated tension, etc.).
Specifically, the hormone dihydrotestosterone (DHT) seems to overexpress in balding scalp regions. In vitro studies demonstrate that DHT may trigger premature shedding, inflammatory signaling proteins, and cell death in dermal papillae cells (the “powerhouse” of the hair follicle) – all of which can lead to progressive hair follicle miniaturization. Moreover, studies have shown that men without DHT do not go bald, and that reducing DHT can help prevent (and partially reverse) the balding process.
Is there evidence that stool transplants can improve pattern hair loss (AGA)?
Clinical evidence? No. Mechanistic evidence? Yes. Anecdotes of AGA improvements after stool transplants? Yes – even with photos. We’ll uncover all of this below.
First, to our knowledge, no case reports or clinical trials investigating the benefits of FMT have occurred. Nonetheless, the absence of evidence doesn’t imply evidence against a therapy (remember: FMTs are a very limited intervention). On that note, there are anecdotes online of people with AGA regrowing their hair (accidentally) following a fecal transplant.
One anecdote comes from a male forum user who suffered simultaneously from both irritable bowel syndrome (IBS) and AGA. In the past, he’d tried to treat his AGA with finasteride. Unfortunately, he did not see any improvements, so he stopped using the drug.
In search for a way to improve his IBS, he later stumbled upon the evidence supporting fecal microbiota transplants to treat C. difficile, Crohn’s disease, and gut dysbiosis. His symptoms were severe enough that he ended up tracking down a willing practitioner and giving the therapy a try – potentially outside of legal means.
A week after the therapy, he reported that his hair shedding dramatically decreased. A year later, he reported significant hair thickening (and potential regrowth)… regrowth that occurred outside of any drug interventions or hair growth therapies.
AGA-related hair regrowth one-year after a stool transplant
This anecdote is promising. But again, it’s just the experience of one person. Beyond this report, are there any other examples?
When it comes to fecal microbiota transplants – we couldn’t find any other anecdotes of hair regrowth from AGA. But interestingly, we were able to find reports of hair regrowth (with photos) from people adhering to incredibly restrictive diets that completely revamp gut microflora constitutions.
One such diet is the carnivore diet – whereby someone nearly completely restricts carbohydrate and fiber intake and begins to subsist entirely off of meat. It sounds like a crazy diet (and it just might be). However, it’s also the only diet where I’ve actually seen people regrow hair lost due to androgenic alopecia. For instance:
- A 1928 case report detailed two men under clinical observation for an entire year while eating only meat. One observation from the investigation team? One of the men saw a complete stop in the progression of his pattern hair loss.
- Some members of our community (who have failed to see success with drugs like finasteride) have later opted to adopt the carnivore diet as an experiment for health. A few of these members have reported hair regrowth.
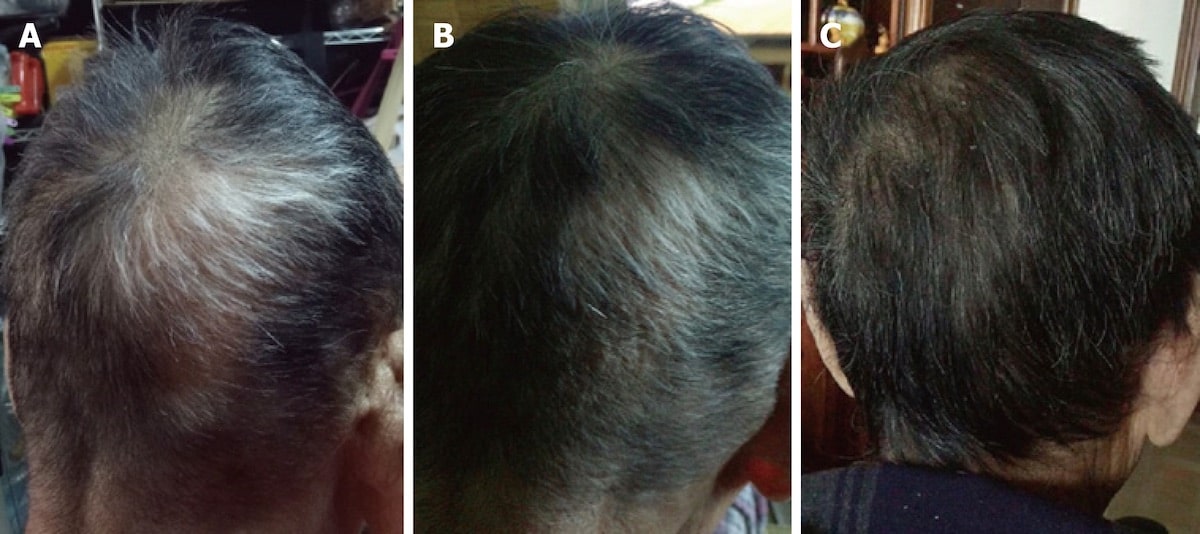
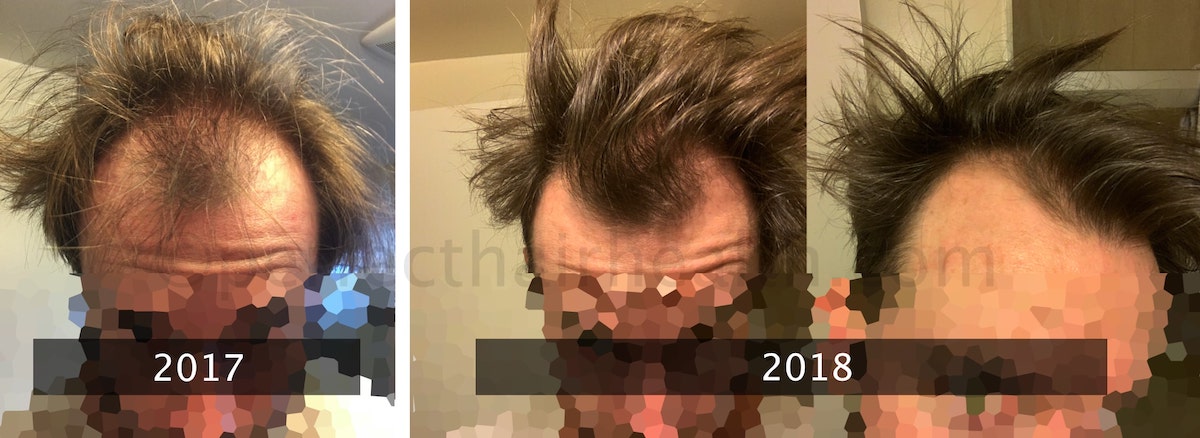
For an example – take Brian (a member of our community). He’d tried finasteride for two years, saw zero improvements, and then eventually quit the drug. Later, he and his wife decided to try the carnivore diet. Simultaneously, he also started to incorporate our scalp massages to see if that would improve his hair loss (since finasteride didn’t work).
One year later, he’d noticed significant hair regrowth at hairline, as well as overall hair thickening. Here were his results.
AGA-related hair regrowth one-year after the carnivore diet + massaging
Interestingly, preliminary evidence suggests that the carnivore diet may increase gut microorganism biodiversity – one of the objectives of FMT. It may also reduce the totality of bacteria inside the gut – since these microorganisms feed off carbohydrate and fiber (two things that are nearly eliminated on a diet consisting of all meat).
Moreover, the carnivore diet eliminates a lot of inflammatory food groups (i.e., FODMAPs) that might contribute to systemic inflammation for sensitive individuals. This may help reduce the number of pathogenic bacteria residing in the gut, as well [27].
This leads to some interesting discussion points about a possible connection between AGA, stool transplants, the carnivore diet, and subsequent changes to the microbiome that could improve hair loss outcomes. We’ll explore these below.
Mechanisms: how might stool transplants improve pattern hair loss?
There’s evidence that our microbiome may help regulate our endocrine system (i.e., the balance of our sex hormones). More specifically, our microbiome may play a direct role in the metabolism of androgens – the sex hormone implicated in AGA.
To start, here’s a quick overview on how hormone metabolism works:
- Once circulating hormones have “completed” a task, they reach the liver. The liver is where free hormones (i.e., the active, unbound hormones) in our blood are processed to later become excreted.
- The liver conjugates these free hormones. Simply put, the livers converts the structure of these hormones from lipophilic (i.e., fat-loving) to hydrophilic (i.e., water-loving). This does two things: it makes the hormone less potent, and it increases that hormone’s chances of entering the digestive tract (i.e., small and large intestine) where they can get excreted.
- Once these conjugated forms of hormones enter the digestive tract, the body excretes these conjugates or metabolites in the stool and urine.
Put simply, our liver conjugates active hormones, those hormones enter our digestive tract, then our digestive tract flushes those hormones out of the body.
But here is where things get interesting: some of our gut bacteria produce an enzyme called beta-glucuronidase. This enzyme has the ability to reverse the process of hormone excretion. Specifically, when conjugated forms of hormones come into contact with beta-glucuronidase, those hormones become unconjugated. In other words, they reconvert back into an active (i.e., unbound) form – where they can potentially reenter our circulatory system.
So, if gut bacteria have the ability to act as gatekeepers for hormone excretion and reabsorption, what does this have to do with AGA?
Fascinatingly, research in healthy men has demonstrated that free dihydrotestosterone (DHT) – the hormone implicated in AGA – exists at levels in the colon at levels 70-fold higher than free DHT found in the blood [24].
This suggests (at least) two things:
- Serum levels of DHT represent just a tiny fraction of total DHT within our body.
- If this free DHT can exit the digestive tract and reenter the circulatory system, this means that gut bacteria might act as a gatekeeper for androgen excretion / reabsorption… and that our microflora might be directly implicated in androgenic-linked conditions: heart disease, prostate enlargement, and maybe even pattern hair loss.
It’s already been shown that gut bacteria have a directly influence on steroid levels. In fact, in the case of periodontal disease and gingivitis, certain cultured bacterial species can actually increase levels of testosterone and DHT [25]. Moreover, researchers have linked this increase to the development of inflammation in periodontitis itself.
Free DHT (as opposed to conjugated DHT) is the type of DHT that likely elicits the majority of DHT-mediated effects on the body. This includes supporting the processes that lead to AGA. You can learn more about this here.
Can unconjugated DHT (in the gut) reenter the circulatory system?
No studies have demonstrated this. But to our knowledge, no studies have actually asked this question (to our knowledge). Not to sound like a broken record, but the absence of evidence cannot imply evidence against something.
It’s worth noting that gut bacteria can produce (and recirculate) estrogens. So, it’s not out of the realm of possibilities that DHT can also be recirculated. In fact, it’s more likely than not.
Moreover, studies on patients with post-finasteride syndrome (long-lasted sexual dysfunction and/or cognitive disorders after cessation of finasteride) show marked gut microbiota alterations [26]. Researchers believe that these microbiota alterations may possibly contribute to the symptoms of finasteride syndrome.
Of course, there are still no clear connections between post-finasteride syndrome and these microbiome alterations. However, if free DHT levels in the colon contribute to overall DHT levels and finasteride results in prolonged changes to the population of these bacteria, it’s possible that reduced colon DHT may contribute to blood DHT, and, thereby the symptoms of post-finasteride syndrome.
Similarly, the opposite could also prove true: if colonic DHT influences blood DHT levels (in an upwards or downwards direction), then anything that increases the total number of our gut bacteria might also increase DHT levels.
Whether this affects scalp DHT (and thereby AGA) – we just don’t know. But we do find these anecdotes of stool transplants and/or carnivore diets regrowing hair absolutely fascinating. After all, both interventions likely lead to improvements in gut biodiversity and changes to the total number of commensal / pathogenic microflora. Therefore, they likely alter DHT activity in the gut. But whether these effects extend to our scalp hair – we can’t yet say (aside from admiring those before-after photos and “hoping” that, after all these years, it’s just that easy to reverse AGA).
Summary so far: Androgenic alopecia (AGA) is driven by a combination of genes and androgens. Interestingly, some patients who’ve undergone stool transplants have later reported hair thickening from AGA without any other treatments. The main hormone implicated in AGA is dihydrotestosterone (DHT). Recent research has found that gut bacteria can actually produce DHT, and even convert it from an inactive to active form. Moreover, researchers recently discovered that free DHT is found in stool samples at levels 70-fold greater than in the blood supply. It has been demonstrated that gut microorganisms influence estrogen production, and the amount of estrogen circulating in our blood. Therefore, it’s not unreasonable to assume that gut flora may also act as gatekeepers for DHT activation between the gut and the circulatory system. If true, this may directly implicate gut bacteria in the pathogenesis of AGA.
Fecal microbiota transplants: undergoing the procedure
What’s it like to do a fecal microbiota transplant?
While FMT may sound like a cure-all for all autoimmune ailments, it’s worth mentioning that research here is still in its infancy, and there’s always the very real possibility that a transplant won’t work or might worsen your symptoms due to unforeseen interactions between donor and recipient microflora.
In the US and Europe, strict guidelines are set to screen a potential donor for any negative attributes that could trigger unwanted side effects for the recipient. There is a questionnaire that is given, stool sample analysis is performed approximately four weeks prior, and blood samples taken [1], followed by an additional interview at the time of the donation. Preferably, you would want a stool sample from someone close to you, with whom you may have shared a common environment. This reduces the chances of any unwanted immune reactions that may occur.
Any failure in the proper preparation of the FMT can present with complications such as bacteremia (i.e., bacteria in the bloodstream) or hospitalizations. Other mishaps such as perforations can occur, but tend to be attributed to the tools and technicians rather than infectious agents [10].
Usually, preparations of the fecal material are done up to six hours before the procedure. The sample is frozen, then thawed out, and, finally mixed with a solution to deliver in a syringe.
It’s also been shown that the size of the sample matters dramatically. Usually, sample sizes are around 50g, which is no more than a few teaspoons. With difficult to treat clostridium infections, larger sample sizes have demonstrated better efficacy and reduced chances chances of failure with the treatment [11]. Many times, FMT requires multiple treatment sessions as well for optimal results.
An important (and interesting) aspect of the procedure is the fact that antibiotics are not to be given to the recipient within 48-hours [12]. While this may seem like an obvious aspect of FMT, this also points to the possibility that it actually is the bacteria present within the stool itself that produces results… not another confounding variable such as immune molecules, proteins, or other components.
Even more supportive of that notion: studies have shown when the procedure is given to the lower digestive tract, (which is also where a majority of the gut microbiome resides), results are drastically better than when administered in the upper digestive tract [1].
Is there a way around the “yuck factor” of FMT?
The idea of taking someone else’s fecal material and implanting it inside of your colon isn’t exactly appealing. And while a lot of the hesitancy is likely psychological, it’s reasonable to ask if there’s a way to do an FMT without doing an enema.
Yes. Researchers have shown that oral capsules produced results on par with the procedure itself – at least in the treatment of resistant Clostridium infections [13]. Obviously, oral supplementation may come across as even less appealing to some. Tto others, the possibility of supplementation certainly reduces the “barrier to entry”.
Summary: stool transplants for hair loss
By regulating the gut microbiome and reducing gut inflammation, a fecal microbiota transplant may improve hair loss outcomes for alopecia areata, telogen effluvium, and perhaps even androgenic alopecia.
Evidence on stool transplants and hair regrowth is incredibly limited. Our knowledge base comes from:
- Four case reports demonstrating significant hair regrowth across patients with autoimmune forms of hair loss
- Anecdotes from people who’ve undergone stool transplants for gut dysbiosis and later reported inadvertent hair regrowth
- Mechanistic data revealing possible connections between stool transplants, the gut microbiome, DHT production, and immunological changes – as well as their overlap with common hair loss disorders.
Nonetheless, I personally find this arena of research fascinating, and I hope it rapidly expands.
In the meantime, please note that we’re not recommending stool transplants as a first-line of defense for any hair loss disorder. If you’re interested in getting one, you’ll need to connect with a qualified physician – we’re not in a position to help you here.
You can reach us in the comments any time!
References
- Wang, J.-W., Kuo, C.-H., Kuo, F.-C., Wang, Y.-K., Hsu, W.-H., Yu, F.-J., … Wu, D.-C. (2018). Fecal microbiota transplantation: Review and update. Journal of the Formosan Medical Association. doi:10.1016/j.jfma.2018.08.011
- van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG,de Vos WM, et al. Duodenal infusion of donor feces for recur-rent Clostridium difficile.N Engl J Med2013;368:407e15.
- Pereira GQ, Gomes LA, Santos IS, Alfieri AF, Weese JS, Costa MC. Fecal microbiota transplantation in puppies with canine parvovirus infection. J Vet Intern Med. 2018 Mar;32(2):707-711. doi: 10.1111/jvim.15072. Epub 2018 Feb 20. PMID: 29460302; PMCID: PMC5867004.
- [Vendrik, K., Ooijevaar, R. E., de Jong, P., Laman, J. D., van Oosten, B. W., van Hilten, J. J., Ducarmon, Q. R., Keller, J. J., Kuijper, E. J., & Contarino, M. F. (2020). Fecal Microbiota Transplantation in Neurological Disorders. Frontiers in cellular and infection microbiology, 10, 98. https://doi.org/10.3389/fcimb.2020.00098
- Vrieze A, Van Nood E, Holleman F, Salojärvi J, Kootte RS, Bartelsman JF, Dallinga-Thie GM, Ackermans MT, Serlie MJ, Oozeer R, Derrien M, Druesne A, Van Hylckama Vlieg JE, Bloks VW, Groen AK, Heilig HG, Zoetendal EG, Stroes ES, de Vos WM, Hoekstra JB, Nieuwdorp M. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012 Oct;143(4):913-6.e7. doi: 10.1053/j.gastro.2012.06.031. Epub 2012 Jun 20. Erratum in: Gastroenterology. 2013 Jan;144(1):250. PMID: 22728514.
- Yoon H, Shim HI, Seol M, Shin CM, Park YS, Kim N, Lee DH. Factors Related to Outcomes of Fecal Microbiota Transplantation in Patients with Clostridioides difficile Infection. Gut Liver. 2021 Jan 15;15(1):61-69. doi: 10.5009/gnl20135. PMID: 32839363.
- Leimbach A, Hacker J, Dobrindt U. E. coli as an all-rounder: the thin line between commensalism and pathogenicity. Curr Top Microbiol Immunol. 2013;358:3-32. doi: 10.1007/82_2012_303. PMID: 23340801.
- Renga G, Bellet MM, Stincardini C, Pariano M, Oikonomou V, Villella VR, Brancorsini S, Clerici C, Romani L, Costantini C. To Be or Not to Be a Pathogen: Candida albicans and Celiac Disease. Front Immunol. 2019 Dec 5;10:2844. doi: 10.3389/fimmu.2019.02844. PMID: 31867008; PMCID: PMC6906151.
- Flannigan, K. L., Rajbar, T., Moffat, A., McKenzie, L. S., Dicke, F., Rioux, K., Workentine, M. L., Louie, T. J., Hirota, S. A., & Greenway, S. C. (2017). Changes in Composition of the Gut Bacterial Microbiome after Fecal Microbiota Transplantation for Recurrent Clostridium difficile Infection in a Pediatric Heart Transplant Patient. Frontiers in cardiovascular medicine, 4, 17. https://doi.org/10.3389/fcvm.2017.00017
- [Dailey, F. E., Turse, E. P., Daglilar, E., & Tahan, V. (2019). The dirty aspects of fecal microbiota transplantation: a review of its adverse effects and complications. Current Opinion in Pharmacology, 49, 29–33. doi:10.1016/j.coph.2019.04.008
- Cammarota, G., Ianiro, G., Tilg, H., Rajilić-Stojanović, M., Kump, P., Satokari, R., Sokol, H., Arkkila, P., Pintus, C., Hart, A., Segal, J., Aloi, M., Masucci, L., Molinaro, A., Scaldaferri, F., Gasbarrini, G., Lopez-Sanroman, A., Link, A., de Groot, P., de Vos, W. M., … European FMT Working Group (2017). European consensus conference on faecal microbiota transplantation in clinical practice. Gut, 66(4), 569–580. https://doi.org/10.1136/gutjnl-2016-313017
- William M. Tauxe, MST, Tanvi Dhere, MD, Angela Ward, MSN, FNP, Lori D. Racsa, DO, Jay B. Varkey, MD, Colleen S. Kraft, MD, MSc, Fecal Microbiota Transplant Protocol for Clostridium Difficile Infection, Laboratory Medicine, Volume 46, Issue 1, Winter 2015, Pages e19–e23, https://doi.org/10.1309/LMCI95M0TWPDZKOD
- Kao D, Roach B, Silva M, Beck P, Rioux K, Kaplan GG, et al.Effect of oral capsule- vs colonoscopy-delivered fecal micro-biota transplantation on recurrent Clostridium difficile infec-tion: a randomized clinical trial.JAMA2017;318:1985e93.
- Rebello, D., Wang, E., Yen, E., Lio, P. A., & Kelly, C. R. (2017). Hair Growth in Two Alopecia Patients after Fecal Microbiota Transplant. ACG case reports journal, 4, e107. https://doi.org/10.14309/crj.2017.107
- [Xie, W. R., Yang, X. Y., Xia, H. H., Wu, L. H., & He, X. X. (2019). Hair regrowth following fecal microbiota transplantation in an elderly patient with alopecia areata: A case report and review of the literature. World journal of clinical cases, 7(19), 3074–3081. https://doi.org/10.12998/wjcc.v7.i19.3074
- Loh SH, Moon HN, Lew BL, Sim WY. Role of T helper 17 cells and T regulatory cells in alopecia areata: comparison of lesion and serum cytokine between controls and patients. J Eur Acad Dermatol Venereol. 2018 Jun;32(6):1028-1033. doi: 10.1111/jdv.14775. Epub 2018 Jan 17. PMID: 29283462.
- [Pandiyan, Pushpa & Bhaskaran, Natarajan & Zou, Mangge & Schneider, Elizabeth & Jayaraman, Sangeetha & Huehn, Jochen. (2019). Microbiome Dependent Regulation of Tregs and Th17 Cells in Mucosa. Frontiers in Immunology. 10. 426. 10.3389/fimmu.2019.00426.
- Maghfour J, Olson J, Conic R, R, Z, Mesinkovska N, A: The Association between Alopecia and Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Dermatology 2021. doi: 10.1159/000512747
- Ojuawo A, Keith L. The serum concentrations of zinc, copper and selenium in children with inflammatory bowel disease. Cent Afr J Med. 2002 Sep-Oct;48(9-10):116-9. PMID: 14562534.
- Asghar, F., Shamim, N., Farooque, U., Sheikh, H., & Aqeel, R. (2020). Telogen Effluvium: A Review of the Literature. Cureus, 12(5), e8320. https://doi.org/10.7759/cureus.8320
- Krajmalnik-Brown, R., Ilhan, Z. E., Kang, D. W., & DiBaise, J. K. (2012). Effects of gut microbes on nutrient absorption and energy regulation. Nutrition in clinical practice : official publication of the American Society for Parenteral and Enteral Nutrition, 27(2), 201–214. https://doi.org/10.1177/0884533611436116
- [Abdel Aziz AM, Sh Hamed S, Gaballah MA. Possible Relationship between Chronic Telogen Effluvium and Changes in Lead, Cadmium, Zinc, and Iron Total Blood Levels in Females: A Case-Control Study. Int J Trichology. 2015 Jul-Sep;7(3):100-6. doi: 10.4103/0974-7753.167465. PMID: 26622152; PMCID: PMC4639951.
- He X, Qi Z, Hou H, Qian L, Gao J, Zhang XX. Structural and functional alterations of gut microbiome in mice induced by chronic cadmium exposure. Chemosphere. 2020 May;246:125747. doi: 10.1016/j.chemosphere.2019.125747. Epub 2019 Dec 24. PMID: 31891852.
- Colldén, H., Landin, A., Wallenius, V., Elebring, E., Fändriks, L., Nilsson, M. E., Ryberg, H., Poutanen, M., Sjögren, K., Vandenput, L., & Ohlsson, C. (2019). The gut microbiota is a major regulator of androgen metabolism in intestinal contents. American journal of physiology. Endocrinology and metabolism, 317(6), E1182–E1192. https://doi.org/10.1152/ajpendo.00338.2019
- Soory M. Bacterial steroidogenesis by periodontal pathogens and the effect of bacterial enzymes on steroid conversions by human gingival fibroblasts in culture. J Periodontal Res. 1995 Mar;30(2):124-31. doi: 10.1111/j.1600-0765.1995.tb01261.x. PMID: 7776153.
- Borgo, F., Macandog, A. D., Diviccaro, S., Falvo, E., Giatti, S., Cavaletti, G., & Melcangi, R. C. (2020). Alterations of gut microbiota composition in post-finasteride patients: a pilot study. Journal of endocrinological investigation, 10.1007/s40618-020-01424-0. Advance online publication. https://doi.org/10.1007/s40618-020-01424-0
- Pedersen N, Ankersen DV, Felding M, Wachmann H, Végh Z, Molzen L, Burisch J, Andersen JR, Munkholm P. Low-FODMAP diet reduces irritable bowel symptoms in patients with inflammatory bowel disease. World J Gastroenterol. 2017 May 14;23(18):3356-3366. doi: 10.3748/wjg.v23.i18.3356. PMID: 28566897; PMCID: PMC5434443.
Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MorePerfect Hair Health Team
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down