- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Does Anavar Cause Hair Loss?
-
10 Best Shampoos for Hair Loss in 2026
-
XYON Review: Do Their Products Actually Regrow Hair?
-
Minoxidil Before and After Photos [2026] | Does It Work?
-
How to Get Finasteride: Is It Over the Counter?
-
Keeps Review: The Truth About Their Hair Loss Treatments?
-
7 Best Oils for Hair Growth
-
Hims Hair Growth Reviews: The Pros, Cons, and Real Results
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
Articles
Oral finasteride is a gold-standard treatment option for men wanting a low-effort approach to hair maintenance. The drug is FDA-approved for androgenic alopecia (AGA) – with two-year studies showing that 1 mg daily of finasteride might slow, stop, or partially reverse AGA progression in 80-90% of men using the drug.[1]https://pubmed.ncbi.nlm.nih.gov/9777765/
But what happens when someone decides to stop using finasteride? How long does the drug remain in the system? After withdrawal, how long does the drug continue to impact hormones like dihydrotestosterone (DHT)?
In this article, we’ll explore the scientific papers that have attempted to answer these questions, and provide step-by-step instructions for what to expect should you decide – for any reason – to withdraw from finasteride.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Why Might Someone Stop Using Finasteride?
There are three main reasons someone might withdraw from finasteride use:
- Experiencing side effects. In this case, withdrawing can help troubleshoot if those side effects are related to the drug or perhaps something else in their life.
- Trying to conceive. Under these circumstances, a user may not want to risk having the drug interfere with their ability to do so or the health of the baby. (Note: while men have the option to temporarily withdraw from finasteride during windows of conception, the drug is contraindicated in women who are pregnant, planning to become pregnant, or breastfeeding.)
- Not seeing enough benefit. Unfortunately, finasteride doesn’t work for everyone, which may lead to drug withdrawal for those not seeing desired results.
In cases of side effects and conception, withdrawal might be temporary.
For instance, men experiencing sexual dysfunction might withdraw from finasteride to gauge if it’s the drug causing the side effect, or perhaps something else in their life (like poor dietary, lifestyle, and/or environmental habits). Men who want to conceive might temporarily withdraw from finasteride to ensure that none of their semen is influenced by finasteride during the period of conception, after which they can then hop back on the drug.
However, for those who are having trouble tolerating finasteride – or for those who aren’t getting any results from the drug – finasteride withdrawal might be more permanent. In these cases, users might find better success by switching to dutasteride, adding in adjuvant therapies like microneedling and/or minoxidil, or exploring if their hair loss is actually androgenic or perhaps caused by other factors – like hypothyroidism.
Under these circumstance, how long does finasteride stay in the bloodstream after we stop taking it? How long until the effects of the drug reverse?
After Quitting, How Long Does Finasteride Stay in the Bloodstream?
The answer depends on (1) how long it takes for the body to metabolize finasteride, and (2) whether you’ve taken enough finasteride to reach steady-state saturation levels throughout the body.
The term most often used to describe drug metabolism is terminal half life. That’s the amount of time it takes for the body to metabolize 50% of a drug dose.
According to pharmacokinetic studies, finasteride has a terminal half-life in the bloodstream of 5-7 hours. This means that it takes 5-7 hours before half of the drug is metabolized from the circulatory system and sent for excretion – where finasteride is primarily excreted through bile and urine.[2]https://pubmed.ncbi.nlm.nih.gov/8846625/
So, if you took 1 mg of finasteride one time, your body would probably metabolize, inactivate, and excrete all of that finasteride within a 2-3 days.
However, taking 1 mg one time isn’t how most people use finasteride. So, what about real-world usage cases – where someone takes 1 mg of finasteride daily for weeks, months, or years?
If this person stops, how long would it take for the drug to clear the system and for the drug’s effects to go away?
With repeated doses, finasteride reaches a saturation point throughout the body. This is where the drug travels throughout the bloodstream, gets distributed throughout other organs and tissues, and after that distribution reaches its peak, blood levels of finasteride reach a steady-state.
In other words, there’s an equilibrium reached for the amount of new finasteride coming into the body, and the amount of finasteride getting metabolized and excreted by the body. And we can measure this equilibrium by measuring steady-state plasma levels (i.e., blood levels) of finasteride as someone takes repeat doses.
For most people, steady-state plasma levels of finasteride are reached within just a few days.[3]https://pubmed.ncbi.nlm.nih.gov/24012200/ And once that occurs, the “equation” for finasteride’s full elimination changes.
Why? Because now finasteride’s excretion isn’t just dependent on its terminal half-life – or how long it takes for 50% of that finasteride to get metabolized from the blood. Now, the equation must also take into account how long it will take for all finasteride that has been stored in other tissues and organs – like the skin and prostate – to dissociate from those sites, re-enter the blood stream, and get metabolized.
According to pharmacokinetic studies, finasteride has a tissue dissociation timing of 4-5 days.[4]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3481923/ Because of this, finasteride’s full excretion from the body (and bloodstream) can actually take a bit longer than a few days.
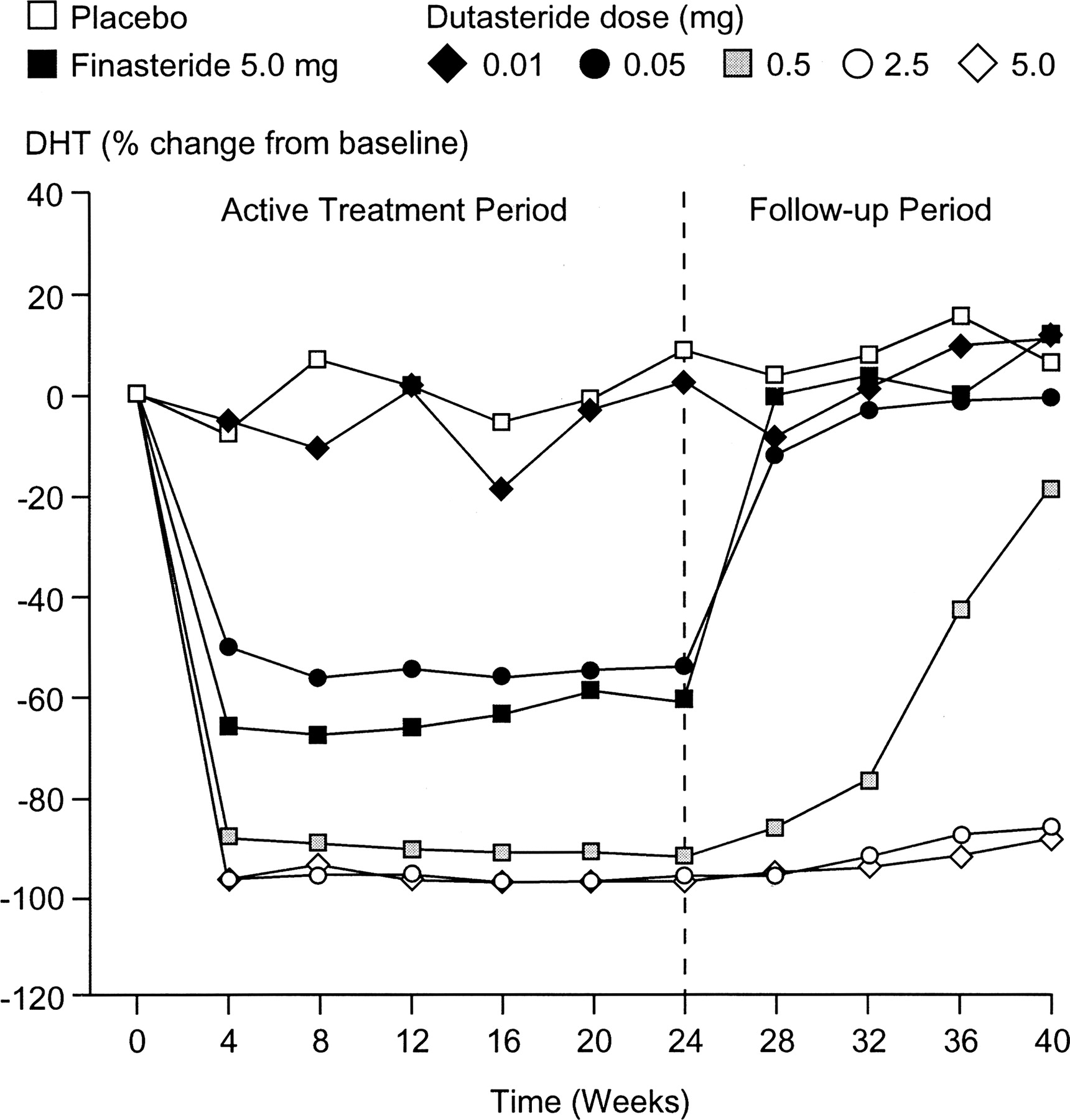
In fact, withdrawal studies on finasteride have demonstrated that despite its terminal half-life of 5-7 hours, it still takes ~30 days for blood levels of dihydrotestosterone (i.e., DHT) to return back to baseline after someone quits the drug.[5]https://academic.oup.com/jcem/article/89/5/2179/2844345 Just see this chart comparing drug withdrawal from finasteride and dutasteride versus placebo – and the number of weeks it takes for the finasteride group to return to baseline levels of serum DHT.
Richard V. Clark, David J. Hermann, Glenn R. Cunningham, Timothy H. Wilson, Betsy B. Morrill, Stuart Hobbs, Marked Suppression of Dihydrotestosterone in Men with Benign Prostatic Hyperplasia by Dutasteride, a Dual 5α-Reductase Inhibitor, The Journal of Clinical Endocrinology & Metabolism, Volume 89, Issue 5, 1 May 2004, Pages 2179–2184
Moreover, after quitting, former finasteride users will typically lose any hair gains from the drug within 3-12 months.
So, to summarize:
- Finasteride’s terminal half-life is 5-7 hours. This is the amount of time it takes for 50% of a drug’s dose to be metabolized and sent for excretion.
- Finasteride’s biological half-life is 14-30 days. This is the amount of time it takes for the biological effects of a drug to fully reverse. In the case of finasteride, this is a return to baseline levels of DHT. This takes longer than a drug’s terminal half-life, because the equation for biological half-life involves not only a drug’s terminal half-life, but also the drug’s saturation levels and its tissue dissociation timings.
How To Take Advantage Of Finasteride’s Terminal & Biological Half-Lives
Because of the mismatch between finasteride’s terminal half-life and biological half-life, users of finasteride who withdraw from the drug don’t immediately lose their hair. Instead, it usually takes ~3 months for hair loss following finasteride withdrawal to presume.
In fact, one clinical study found that after one year of finasteride use, men who switched to 30 days-on, 30 days-off finasteride use were no worse off than men who continued taking the drug every day.[6]https://www.jaad.org/article/S0190-9622(20)31928-9/fulltext
This is, again, because of the difference in finasteride’s terminal and biological half-lives. And with this knowledge, finasteride users who intend to temporarily withdraw from the drug can leverage these differences to their advantage.
For example, say a male would like to conceive with their partner, but they don’t want finasteride to potential impact their semen parameters during the window of conception. This user can:
- Withdraw from finasteride. After 30 days, the drug should be fully out of the system and all biological effects should return to baseline.
- Conceive with their partner. Technically speaking, they have a 1-2 month window after that 30-day washout period whereby hair gains from finasteride should remain preserved.
- Hop back on finasteride. After conception is complete, this male can restart finasteride prior to hair loss reinitiating – so long as they were able to conceive during that 1-2 month window following their finasteride washout period.
The same approach can be taken with troubleshooting side effects, or deciding to leave finasteride at-home while traveling short-term, or any other reason you might want to temporarily hop off the drug.
Those experiencing side effects can potentially minimize them by switching to a topical formula. Just be aware that topical finasteride can enter the bloodstream, too, depending on the percent active ingredient, the amount applied, and the length of time the solution is left on the scalp.
References[+]
References ↑1 https://pubmed.ncbi.nlm.nih.gov/9777765/ ↑2 https://pubmed.ncbi.nlm.nih.gov/8846625/ ↑3 https://pubmed.ncbi.nlm.nih.gov/24012200/ ↑4 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3481923/ ↑5 https://academic.oup.com/jcem/article/89/5/2179/2844345 ↑6 https://www.jaad.org/article/S0190-9622(20)31928-9/fulltext Post-Finasteride Syndrome is a term used to describe a constellation of symptoms reported by former finasteride users who quit using the drug, but who still report drug-related side effects more than 3 months after discontinuing.
Amongst dermatologists, endocrinologists, and hair loss researchers, the existence and prevalence of Post-Finasteride Syndrome (PFS) is a hotly debated topic. On the one hand, some urologists like Dr. Abdulmaged Traish are adamant that PFS is real, and that the original clinical trials that earned finasteride FDA approval were neither adequately designed nor robust enough to capture how many finasteride users might be affected.[1]https://pubmed.ncbi.nlm.nih.gov/32033719/
On the other hand, some world-renowned hair loss researchers like Dr. Ralph Trueb claim that PFS is a self-induced delusional disorder bordering on mass formation psychosis – and support those claims by pointing to (1) the absence of evidence of PFS from the original finasteride studies, (2) the influence of media reporting on PFS and subsequent spikes in people claiming to have the condition, and (3) a strong proclivity toward those claiming to have PFS also having diagnosed mental health disorders – particularly histrionic personality disorder.[2]https://www.karger.com/Article/Fulltext/497362
So, what’s the truth about PFS? Is it real or imagined? If PFS is real, what’s the estimated risk for people trying finasteride? And is there anything that can be done to lower the risk of PFS?
In this article, we’ll explore the evidence on both sides of the PFS argument, explain why the existence of PFS is difficult to study, and reveal why debate over PFS is likely here to stay. Finally, we’ll contextualize the estimated prevalence of PFS by comparing its risk ratio to other voluntary risks taken in each day of life – like the risk of death from driving a motor vehicle – all so that potential finasteride users can make more informed decisions about their hair loss treatments.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What Is Post-Finasteride Syndrome (PFS)?
Post-Finasteride Syndrome (PFS) is an alleged condition from the use of finasteride, whereby side effects incurred from the drug do not go away 3+ months after quitting. The implication of PFS is that, for a very small number of finasteride users, side effects might be long-lasting, and perhaps permanent.
Alleged PFS symptoms vary from person-to-person, but in general, may involve persistent and unresolved:
- Sexual dysfunction
- Genital numbness
- Reduced libido and/or weak erections
- Persistent brain fog
- Depressive-like symptoms
Some people claiming to experience PFS-related side effects have described their onset within hours of taking a single finasteride pill. Others say it took months before anything started going wrong. Others say they were symptom-free while using finasteride, and that they only started experiencing side effects after withdrawing from the drug.
Given the heterogeneity in both PFS symptoms and their timings of onset, researchers (and sufferers) have struggled to understand why this alleged condition seems to impact so many people in such wildly different ways.
Nonetheless, those who believe PFS is real also suspect that its development is causally linked to the enzyme that finasteride inhibits, as well as the potential for that enzyme inhibition to have unintended, downstream, long-lasting effects on the body.
Finasteride & Side Effects: A History Of Debate
Finasteride is a drug that inhibits the type II 5-alpha reductase enzyme. By inhibiting this enzyme, finasteride lowers levels of dihydrotestosterone (DHT) – a hormone causally associated with androgenic alopecia (AGA) and benign prostatic hyperplasia.
At daily doses of 0.2 mg to 5.0 mg daily, oral finasteride can therapeutically lower DHT levels by 70% – with two-year studies showing that 80-90% of men using the drug see a slowing, stopping, or partial reversal in AGA.[3]https://www.sciencedirect.com/science/article/pii/S0022202X15529357 In fact, dozens of studies totaling more than 10,000 participants suggest that oral finasteride routinely improves hair growth outcomes versus placebo – with 5- and 10-year studies suggesting hair maintenance above baseline for a majority of its users.[4]https://www.oatext.com/Long-term-(10-year)-efficacy-of-finasteride-in-523-Japanese-men-with-androgenetic-alopecia.php
For these reasons (and the quality of evidence supporting the medication), finasteride has received FDA-approval for AGA and is widely considered the gold-standard medical intervention for this condition.
But as is the case with all medications, there’s always a risk of side effects. For finasteride, these side effects tend to present in the form of lowered libido, sexual dysfunction, gynecomastia, and/or brain fog.
While the original clinical trials that granted finasteride FDA-approval estimated side effect risks to be less than 5% versus placebo, follow-up studies have put that risk as high as 40%.[5]https://www.sciencedirect.com/science/article/pii/S0022202X15529357[6]http://www.drproctor.com/propecia/propecia.pdf Depending on the online resource you read (i.e., a natural website versus a telehealth website), you may have noticed that websites will cite either end of the extreme, depending on the products they’re trying to sell. Natural websites tend to cite higher numbers to fearmonger over side effects and encourage people to buy their natural supplements and topicals; telehealth websites tend to avoid discussions about higher estimations because it might interfere with prescription drug sales.
The reality is that most follow-up studies citing higher rates of finasteride-related side effects tend to be of lower quality – with smaller sample sizes, biases in research methodologies, and a strong influence of the nocebo effect – whereby in some studies, physicians actually told patients to “watch out” for side effects while prescribing the drug, which introduces a psychogenic influence whereby patients become nearly 5 times as likely to report those side effects.[7]https://www.nature.com/articles/ncpuro1012
At the same time, other researchers have pointed out that finasteride’s original clinical trials also contain their own biases. For instance, those studies were funded by Merck – the very drug company that stood to gain from the approval of its patented hair loss-fighting medication – and that this might’ve influenced the ways in which side effects were both catalogued and tabulated by investigators – all to downplay their true prevalence and/or magnitude.
While these criticisms may read as conspiratorial and unfounded, the reality is that Merck doesn’t have the best reputation as a drug company. Former employees-turned-whistleblowers alleging clinical trial obfuscation in vaccine trials, and lawsuits settled after drugs taken to market by Merck later inadvertently killed lots of people.[8]https://www.fiercepharma.com/vaccines/whistleblowers-accuse-merck-of-withholding-info-on-mumps-vaccine[9]https://www.nature.com/articles/450324b Because of this history, criticisms of this nature will remain on-the-table for Merck, regardless of their validity.
Based on our experience working with hair loss sufferers (and our read of the data), we estimate that between 5% to 15% of finasteride users experience some degree of side effects – and that the majority of these side effects are mild and improvable – at least with the right titration and/or drug delivery strategies. If you’re interested in learning more, see our article on strategies to reduce side effects from finasteride, and if blood tests can help predict finateride-linked side effects.
Nonetheless, it is true that a small subset of finasteride users do experience side effects, and that many of those side effects are sexual or cognitive in nature. What is debated, however, is whether those finasteride-induced side effects reverse entirely for 100% of people after quitting the medication.
According to finasteride’s phase II and phase III clinical trials, all participants who stopped using finasteride due to side effects saw a complete resolution of those side effects, and within three months. Yet in the last 15 years, some former finasteride users have claimed otherwise.
So what might explain this discrepancy? And if this phenomenon is real, how might we explain its pathology?
Post-Finasteride Syndrome: What Might Cause It?
To date, there have been dozens of hypotheses surrounding how PFS might develop. All of them relate to the way finasteride works, and what might go wrong when the enzyme the drug targets is inhibited for appreciable periods of time.
Below are three theories that have received significant attention over the last few years, along with their counterarguments.
Please consider this a very brief overview of these arguments. The goal of this article is not to indoctrinate ourselves within the fine details of these hypotheses, but instead, to overview both sides of the argument, explain why this debate won’t be settled anytime soon, express our current perspectives given the data, and reveal strategies to navigate any known-unknown risks linked to finasteride.
Persistent biological changes (i.e., androgen receptor upregulation and/or downregulation)
- Argument: In vitro and animal studies suggest that when anti-androgen drugs – like spironolactone, finasteride, or flutamide – are administered, male hormones (like testosterone or dihydrotestosterone) decrease, and in response, androgen receptors increase.[10]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3116931/[11]https://pubmed.ncbi.nlm.nih.gov/21557276/ In some animal models, withdrawal from anti-androgen drugs does not lead to a rebalancing of androgen receptor expression. In other words, these receptors remain upregulated. This suggests long-lasting biological changes from finasteride use might be possible in a small subset of users, and that androgen receptor upregulation (or reflexivity) might be part of the equation for those suffering from PFS.[12]https://pubmed.ncbi.nlm.nih.gov/34247957/
- Counterargument: Human data on finasteride use and androgen receptor upregulation often conflicts with findings from in vitro and animal models.[13]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4137476/ Moreover, animal models whereby androgen receptors remained upregulated following finasteride withdrawal (1) used supraphysiological doses of finasteride, and (2) paired that drug with other anti-androgens. This doesn’t reflect normal human usage cases, and co-administration of the drugs obfuscates the ability to discern which drug is actually responsible for the phenomenon. Finally, studies on finasteride show that serum and tissue DHT levels return to normal following ~30 days of treatment withdrawal. This suggests that if finasteride is leading to any androgen receptor reflexivity or permanent upregulation, then for whatever reason, hormonal levels post-finasteride withdrawal still return to baseline.[14]https://www.ncbi.nlm.nih.gov/books/NBK513329/ Finally, studies exploring the potential for upregulation of androgen receptors in penile tissues of those claiming PFS do not use adequate controls – such as men affected by erectile dysfunction who never used the drug. Therefore, it is impossible to parcel out how many of these cases are drug-related versus just related to impotence – which affects a high number of American adults.
Persistent changes to gut microflora
- Argument: Observational studies on PFS sufferers show alterations to gut microflora versus healthy controls.[15]https://pubmed.ncbi.nlm.nih.gov/32951160/ These gut alterations in microflora might influence hormonal and immunological markers elsewhere throughout the body – particularly the skin tissues and brain. After all, studies show bidirectional feedback loops between gut microflora and serum / organ hormonal profiles, which are causally linked to disorders related to libido and mood.[16]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6962501/
- Counterargument: While this is hypothetically plausible, the one study measuring gut microflora composition on post-finasteride syndrome participants suffers from major methodological flaws – one of the biggest being a negative-control group. For instance, depression also influences gut microflora, but depressed patients were not included as a negative control in this study. How can we discern the differences between these depressed patients and any long-lasting microflora changes due to withdrawn finasteride use? Due to the limitations of this study, it’s impossible to further this hypothesis with the available data.
Persistent histological changes (i.e., tissue fibrosis and scarring)
- Argument: Subsets of PFS sufferers have reported tissue-level changes appearing since their first use of finasteride. These histological changes include, but are not limited to, penile microvascular changes and fibrosis.[17]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7354335/ There is mechanistic evidence suggesting that low DHT levels in prostatic tissues might elevate inflammatory markers that could resolve, over time, into fibrosis if left unchecked.[18]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3997870/ Therefore, perhaps PFS is caused by medically lowering DHT levels, which in a subset of patients, might increase inflammation that leads to scarring in key organ sites. When the medication is withdrawn, DHT levels return to normal, but the fibrosis / scarring that formed during the hormone’s depression remains.
- Counterargument: While this hypothesis warrants further investigation, the studies supporting this viewpoint are of very low quality due to small sample sizes and no control groups. It is important to note that upwards of 40% of American adults suffer from erectile dysfunction – with increasing prevalence per decade of life and a subset of those individuals also displaying with penile fibrosis.[19]https://www.amjmed.com/article/S0002-9343(06)00689-9/fulltext[20]https://pubmed.ncbi.nlm.nih.gov/8254833/ Without a control group, these studies make it impossible to parcel out who might believe they’re experiencing long-standing side effects from finasteride, but who are just coincidentally reaching the age at which impotence risk increases, and by coincidence of usage and the very fact that finasteride can cause sexual side effects, these participants are misplacing blame on the drug for a phenomenon that would’ve otherwise happened to them had they never used the drug anyway. Furthermore, there is no high-quality evidence of long-lasting histological changes in finasteride users after withdrawing from the drug. All indications are that hormonal profiles return to normal, and that even while on the drug, muscle morphology and subcutaneous fat stores do not change relative to controls because testosterone and other hormones can still govern these processes in replacement to lowered DHT.
Summarizing The Case Against PFS
There are rebuttals to these counterarguments, and counter-rebuttals to those rebuttals. But given these arguments and counterarguments, we’ve likely seen enough to recognize their overarching patterns:
- Yes, there’s some mechanistic and clinical evidence to support the anecdotes of symptoms befitting PFS. But…
- That evidence is of very low quality, so will remain forever scrutinized (and with good reason)
For instance, some mechanistic studies conflict with human studies, and due to small sample sizes, imperfect methodologies, and inadequate control groups, the PFS clinical studies currently available will remain open to criticism.
Moreover, because the original 1-, 2-, and 5-year studies on finasteride did not report (in their published material) any incidences of side effects not resolving after a participant withdrew from treatment, it’s incredibly difficult to parcel out finasteride’s influence on persistent erectile dysfunction versus the background risk of erectile dysfunction in adult men – which some studies suggest is 40% or higher.
Finally, PFS reports increase and decrease in congruence with media coverage on the topic, along with lawsuits against Merck for men who claim to be experiencing PFS-like effects.[21]https://www.karger.com/Article/Fulltext/497362 For what it’s worth, studies show that finasteride users have a strong tendency toward the nocebo effect – whereby simply telling a patient about the drug’s side effects leads to those patients reporting those side effects at a much higher rate than had they never been told anything. This was demonstrated in a randomized, controlled clinical study whereby a group of men who were prescribed finasteride and told it might lower their libido were nearly 500% more likely to report sexual side effects in follow-up appointments versus those who hadn’t been told that information.[22]https://www.nature.com/articles/ncpuro1012
All of this makes it scientifically challenging to parcel out a true prevalence of PFS, or even catalogue the condition as real versus imaged.
5 Barriers In The Way Of Proving PFS’ Existence
Take into consideration the following realities:
- The strong nocebo influence of finasteride
- The influence of media on PFS reporting
- The high background rates of erectile dysfunction and/or impotence in adults
- The fact that many PFS sufferers have mental health disorders – like histrionic personality disorder
- The very low quality studies exploring PFS
Given these circumstances, it might feel reasonable to dismiss PFS as a hoax condition propagated by opportunistic, delusional men looking to cash in by filing frivolous lawsuits against a pharmaceutical giant.
In fact, this is essentially the sentiment on most public hair loss forums – BaldTruthTalk, HairLossTalk and Tressless – whose users routinely mock and insult people alleging PFS, and in doing so, dissuade discussion about the topic.
If you hold this sentiment, it’s not unreasonable – particularly given the studies, arguments, and counterarguments we just outlined.
Yes, there’s reason to doubt the existence of PFS. Yes, there’s reason to question the sanity of many alleged PFS sufferers.
But that’s not the whole story.
One of the biggest counterarguments for PFS – one routinely repeated online – is actually inaccurate. Most people wouldn’t know it, mainly because of the sheer number of people blindly regurgitating it on hair loss forums.
It’s the assertion that there’s no evidence that finasteride can cause histological changes that persist after quitting the drug.
This is 100% false.
There is evidence of persistence histological changes post-finasteride withdrawal. In fact, these changes are well-studied and widely acknowledged by physicians across both aisles of the PFS debate.
The phenomenon is called finasteride-induced gynecomastia. And in a subset of cases, it doesn’t resolve after quitting the drug.
Persistent Gynecomastia: The Strongest Evidence Supporting Post-Finasteride Syndrome
Gynecomastia is the growth of male breast tissue. In men, studies causally link the development of gynecomastia to elevations in estrogen and/or prolactin.
Men who use finasteride tend to see their levels of estradiol (an estrogen) increase by 10-20%. This is a normal consequence of 5-alpha reductase inhibition, because with less available DHT formation, more free testosterone will convert into total testosterone and estradiol.
For those who have a genetic predisposition to gynecomastia, or for those whose diets, lifestyles, and environments have already elevated their estrogen and/or prolactin beyond baseline, any additional increases in estrogen from finasteride might be enough to push these men toward the development of unwanted growth of breast tissue.
This is a well-studied side effect of finasteride (and 5-alpha reductase inhibitors). It tends to affect between 0.25% to 5% of finasteride users – with a varying magnitudes of effect and prevalence depending a study’s participants (i.e., healthy young men or overweight men with benign prostatic hyperplasia).[23]https://pubmed.ncbi.nlm.nih.gov/23067029/
To be clear: there is no debate over finasteride-induced gynecomastia, or the mechanisms governing its development.
So, let’s circle back to one of the biggest counterarguments levied by people who do not believe in the plausibility of PFS.
If gynecomastia occurs in a small subset of finasteride users, then once someone stops taking finasteride, that gynecomastia should go away. Right?
Wrong.
Clinical studies show that in 80% of cases, finasteride-induced gynecomastia resolves if given enough time away from the drug.[24]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2929552/[25]https://jamanetwork.com/journals/jamadermatology/article-abstract/478759 Having said that, 20% of cases may see incomplete or no resolution – even up to six years after quitting the medication.[26]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2929552/
Under those circumstances, the only option for removal is surgery.
Persistent gynecomastia is perhaps the best example of a long-lasting histological change caused by finasteride that does not always resolve after the drug is discontinued.
The histological change is the unwanted growth of adipose and/or glandular tissue in the male breast.
This phenomenon is well-defined, well-observed, and widely-recognized amongst researchers on both sides of the PFS debate. And while the original finasteride studies did not capture this risk, post-marketing studies did.
This is because those original finasteride studies – despite having more than 1,200 participants in the finasteride group – still weren’t large enough to account for a side effect rare enough to impact only one-fifth of 0.25% of healthy finasteride users (i.e., 1 in every 2,000 people).[27]https://www.sciencedirect.com/science/article/abs/pii/S0190962298700076
With that in mind, let’s revisit the possibility and plausibility of PFS. If there are cases of finasteride-induced gynecomastia that persist even after finasteride is discontinued, does this signal that finasteride might also have a potential to histologically influence other organ sites in similar matters?
Yes.
And while these types of adverse events (if they do exist) are probably incredibly rare – much like persistent gynecomastia induced by finasteride – their existence is now plausible, because we’ve seen a similar phenomenon documented in male breast tissue.
Why The PFS Debate Is Here To Stay
In the exercise above, we revealed how an incredibly rare adverse event from finasteride – treatment-induced gynecomastia that persists even after quitting the drug – is now a recognized side effect that was not captured in the original clinical studies on finasteride. Instead, it was a side effect observed in post-marketing studies – mainly because of how few people it affects: perhaps just 1 in every 2,000 finasteride users.
Given the heated scientific debate over PFS versus the well-established acknowledgment of finasteride-induced gynecomastia, it’s probably safe to assume that if PFS is real, the number of finasteride users affected is substantially less than 1 in 2,000.
Otherwise, PFS would’ve probably been noted in finasteride’s original phase II and phase III clinical trials.
So, let’s assume (for the sake of argument) that the estimated prevalence of PFS is 1 in 5,000 finasteride users. Let’s also assume that around 40% of American adult men already have some degree of impotence – which has previously been established by several studies.
What sort of study do we need to design to prove or disprove the existence of PFS?
We would need a study that was randomized, double-blinded, placebo-controlled, and prospectively designed – as these kinds of studies are generally the only types that can infer causality.
One group of people would receive finasteride; the other group would receive a sugar pill. The study would need to run multiple years – to control for the heterogeneity in timings of onset for PFS. And it would also need a multiple-year withdrawal period to track outcomes for side effect resolution.
So, how many participants would we need to gain enough statistical power to get a definitive answer?
This is where things get incredibly difficult to parcel out.
If we estimate a “true” PFS incidence of 1 in 5,000 finasteride users (i.e., 0.02%), we must recruit enough men in this study to statistically differentiate that effect from the background risk of erectile dysfunction in 40% of adult men.
With randomization, erectile dysfunction should be equally present in both finasteride users and non-users, and at a background rate of 40%. So, we’ll need a large enough sample size to discern statistical significance between impotence reports with differences of 40% (in the placebo group) versus 40.02% (erectile dysfunction + PFS post drug withdrawal).
Then we’ll need to also find ways to account for risks of the nocebo effect and mental health disorders influencing side effect reporting – as some participants will ultimately discover that they’re using finasteride and begin researching the drug on their own. There are statistical tools that can help us account for all of the above to determine a needed sample size for significance.[28]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4017493/
So, accounting for all of the above, how many participants do we need to recruit to determine – once and for all – if PFS is real?
1,000,000+ participants.
That’s right. We need more than a million study participants. That’s how much work it will take to parcel out a 0.02% increased risk of finasteride-induced erectile dysfunction persisting post-drug withdrawal from the 40% background risk of already having erectile dysfunction or having it develop for unrelated reasons during the time someone is using the medication.
That sort of study would require billions of dollars to fund, and even if it were funded, we wouldn’t have an answer until years after the study began – probably 5-10 years after its initiation.
Keep in mind that finasteride’s patent expired years ago. With these financial barriers and with no incentive for funding from large pharmaceutical companies, the likelihood of this study happening is as close to 0% as it gets.
For these reasons, debate over PFS is destined to rage on. The studies to prove or disprove its existence are too difficult to conduct.
Reconsidering The PFS Debate
With all of the evidence in mind, here are our current perspectives on PFS:
- PFS is likely real, but also probably affects fewer than 1 out of every 5,000 to 10,000 finasteride users.
- If PFS is real, its prevalence is likely overstated online due to negativity bias in public anonymous hair loss forums.
- While many people claiming PFS might instead be suffering from a psychological disorder, we’ve spoken to people alleging PFS who (1) are otherwise healthy, (2) have no psychiatric disorders, (3) have successful careers, (4) have good relationships with friends and romantic partners, (5) have never filed a lawsuit against Merck, (6) have received a PFS diagnosis by a doctor – sometimes with corroborating histological data, and (7) just want their life to go back to normal. For these reasons, PFS reports from credible individuals need to be separated from the hysteria, rather than dismissed by way of lumping in with the hysteria.
- Anyone considering finasteride should be made aware of the debate surrounding post-finasteride syndrome.
- At the same time, with a small estimated prevalence of PFS, those with AGA should consider the risk-benefit analysis of finasteride – especially in the context of what other behavioral risks people engage in on a daily basis.
PFS: Contextualizing Its Risk Vs. Other “Voluntary” Behavioral Risks
If you’re considering finasteride, but have decided against trying it because of concerns of PFS, we 100% understand.
With that said, let’s contextualize what a 1 in 5,000 (or lower) estimated risk of PFS actually looks like in the context of other risks you might take in your everyday life.
Throughout a lifetime, 1 out of every 102 Americans will die in a motor vehicle accident.[29]https://injuryfacts.nsc.org/ However, this does not stop the overwhelming majority of Americans from operating motor vehicles. While accidents are tragic and sometimes unavoidable, there are also safe-driving behaviors that might lower that risk, and in doing so, allow you all the benefits of driving.
We believe the same is true for finasteride and hair loss.
Consider the heavy psychological and emotional impact that hair loss can have (especially on younger people). Also consider the overwhelming clinical evidence on finasteride – a drug that can stop the progression of AGA in the majority of men, and for 10+ years. Now consider the debate over PFS and its loosely estimated prevalence of 1 out of every 5,000 to 10,000 finasteride users.
The decision to try or not try finasteride is entirely your own to make, along with the medical guidance of a physician. Depending on your needs, preferences, and goals – all of us will weight the above evidence and the opportunity costs of finasteride (i.e., more lost hair) differently. But if PFS is a top concern for you, we believe – much like safe driving – that there are ways to still benefit from the drug while also further lowering that ~1 in 5,000-10,000 risk.
If PFS Is Real, Can We Reduce Our Risks Of Developing It?
Currently, there are no risk factors (aside from psychiatric disorders) that might help to predict someone’s risk of PFS. But in our opinion, any strategy that might reduce side effects from finasteride may also reduce the risk of developing PFS.
By opting for lower-dose oral finasteride formulations, or by using certain topical finasteride formulations, finasteride users can not only reduce their total drug exposure over a lifetime, but also better localize the effects of the medication to the scalp.
Under both circumstances, we suspect there to be a significant risk reduction in both finasteride-linked side effects as well as PFS.
We hope this article helps, and if you’d like personal support on your hair loss journey, you can always partner with our team inside our membership community.
References[+]
References ↑1 https://pubmed.ncbi.nlm.nih.gov/32033719/ ↑2, ↑21 https://www.karger.com/Article/Fulltext/497362 ↑3, ↑5 https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑4 https://www.oatext.com/Long-term-(10-year)-efficacy-of-finasteride-in-523-Japanese-men-with-androgenetic-alopecia.php ↑6 http://www.drproctor.com/propecia/propecia.pdf ↑7, ↑22 https://www.nature.com/articles/ncpuro1012 ↑8 https://www.fiercepharma.com/vaccines/whistleblowers-accuse-merck-of-withholding-info-on-mumps-vaccine ↑9 https://www.nature.com/articles/450324b ↑10 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3116931/ ↑11 https://pubmed.ncbi.nlm.nih.gov/21557276/ ↑12 https://pubmed.ncbi.nlm.nih.gov/34247957/ ↑13 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4137476/ ↑14 https://www.ncbi.nlm.nih.gov/books/NBK513329/ ↑15 https://pubmed.ncbi.nlm.nih.gov/32951160/ ↑16 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6962501/ ↑17 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7354335/ ↑18 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3997870/ ↑19 https://www.amjmed.com/article/S0002-9343(06)00689-9/fulltext ↑20 https://pubmed.ncbi.nlm.nih.gov/8254833/ ↑23 https://pubmed.ncbi.nlm.nih.gov/23067029/ ↑24, ↑26 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2929552/ ↑25 https://jamanetwork.com/journals/jamadermatology/article-abstract/478759 ↑27 https://www.sciencedirect.com/science/article/abs/pii/S0190962298700076 ↑28 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4017493/ ↑29 https://injuryfacts.nsc.org/ In terms of hair regrowth, the “best” dose for medication is typically the one that maximizes improvements and minimizes the risk of adverse events.
In the case of finasteride, 1mg daily has the most clinical support befitting a “best” dose given the risk-benefit ratio of hair improvements. However, there’s also evidence that finasteride doses as low as 0.2 mg daily may produce similar degrees of hair regrowth and reduce the risk of side effects.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Finasteride Doses: Is 1 mg The Only Dose That Works?
Is 1 mg the best dose of finasteride for hair loss? After all, oral finasteride is prescribed at 1 mg daily for androgenic alopecia and 5 mg daily for benign prostate hyperplasia. For most people, these dosages are tolerable and produce a relatively excellent safety profile. But for those interested in cutting down on a lifetime of finasteride consumption, there may be ways to reduce drug exposure by 80% without sacrificing much (or any) hair gains.
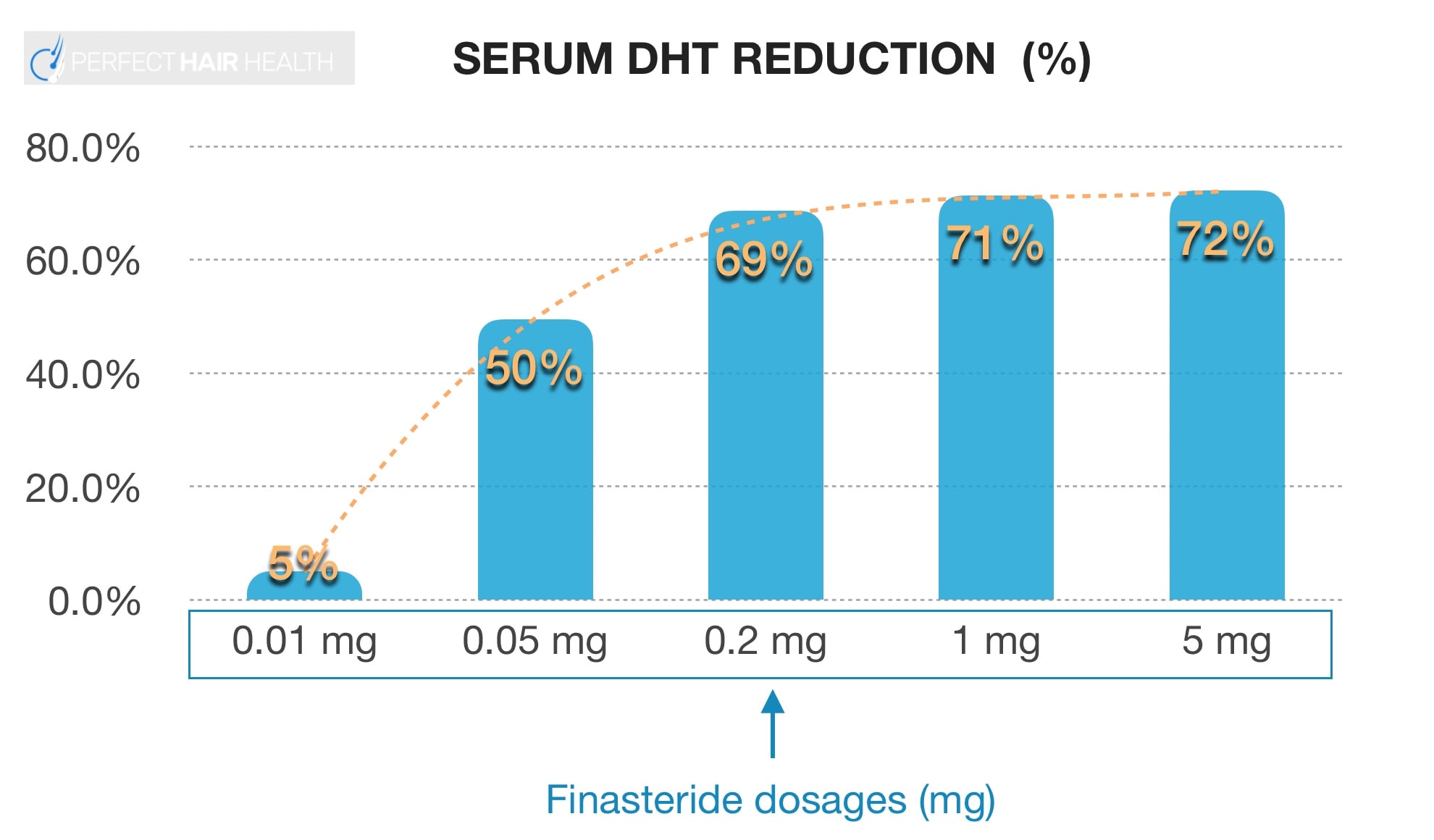
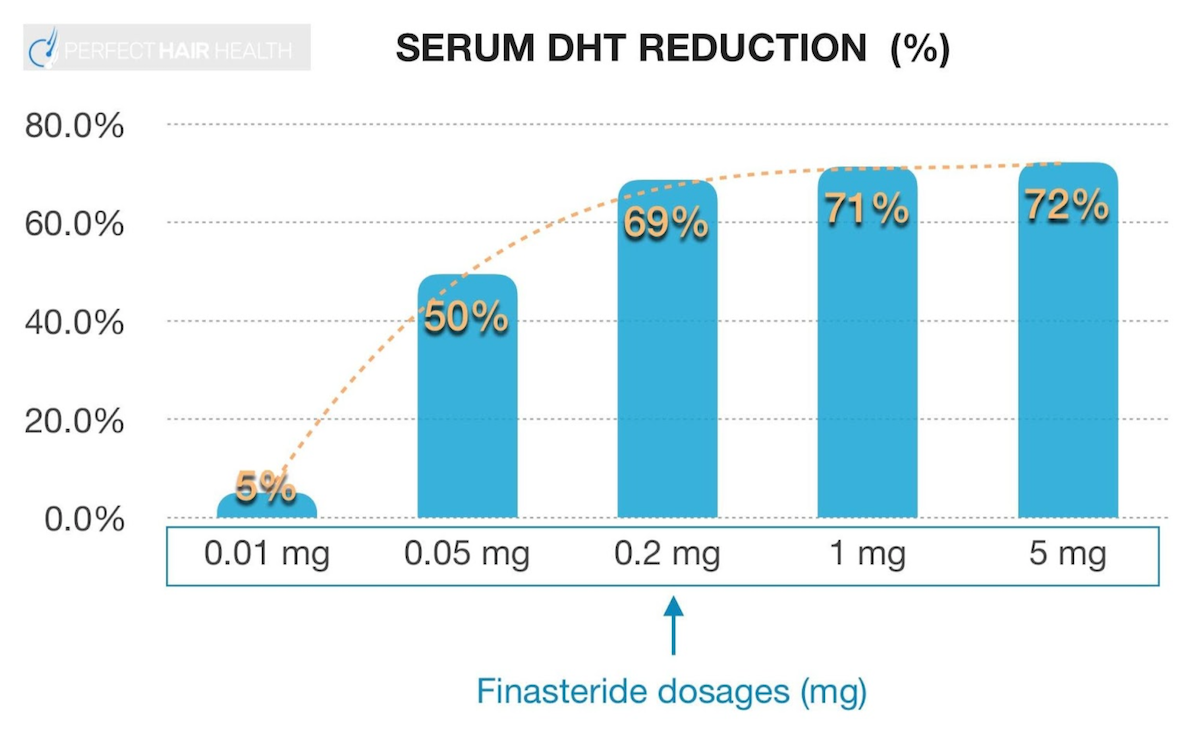
Clinical studies have demonstrated a steep, dose-dependent, logarithmic response curve for finasteride’s effects on DHT levels. That just means that a little bit of finasteride reduces almost as much DHT as a lot of finasteride. See this chart:
Serum DHT Reduction Percentage vs. Finasteride Dosages
As such, 0.2 mg daily reduces DHT levels by nearly the same as 5 mg. This is why, in some countries, it’s common to prescribe 0.2 mg of finasteride for androgenic alopecia rather than the full 1 mg dose.
Does Clinical Data Support a 0.2mg Daily Dose of Finasteride?
Yes. According to a review on finasteride:[1]https://escholarship.org/content/qt24k8q743/qt24k8q743.pdf
“In a 48-week randomized, double-blind study of finasteride treatment of androgenetic alopecia, the efficacy of 1mg and 0.2mg finasteride were compared in 414 men. The incidence of decreased libido was 2.9%, 1.5%, and 2.2% for finasteride 1mg, 0.2mg, and placebo, respectively [31]. Most of these cases resolved during the course of therapy, and there were no discontinuations of the study related to the adverse drug effects [31].”
That specific study also mentioned that:[2]europepmc.org/article/med/15319158
“All efficacy endpoints showed significant improvement with finasteride therapy by 12 weeks (p < 0.05 versus placebo). At 48 weeks, 58%, 54%, and 6% of men in the finasteride 1 mg, finasteride 0.2 mg, and placebo groups, respectively, had improved based on assessments of global photographs. All efficacy endpoints were numerically superior for the 1 mg dose over the 0.2 mg dose at 48 weeks.“
The authors did not find that the hair regrowth from 0.2 mg daily of finasteride was statistically significantly different (i.e., better or worse) than 1.0 mg daily of finasteride. So, it’s unclear whether these results regarding slightly lower efficacy (and risks of side effects) for the 0.2 mg versus 1.0 mg doses of finasteride are statistically or clinically significant.
Another 12-month study compared finasteride doses of 1 mg, 0.2 mg, and 0.01 mg daily versus placebo.[3]https://pubmed.ncbi.nlm.nih.gov/10495375/
Roberts JL, Fiedler V, Imperato-McGinley J, Whiting D, Olsen E, Shupack J, Stough D, DeVillez R, Rietschel R, Savin R, Bergfeld W, Swinehart J, Funicella T, Hordinsky M, Lowe N, Katz I, Lucky A, Drake L, Price VH, Weiss D, Whitmore E, Millikan L, Muller S, Gencheff C, et al. Clinical dose ranging studies with finasteride, a type 2 5alpha-reductase inhibitor, in men with male pattern hair loss. J Am Acad Dermatol. 1999 Oct;41(4):555-63. PMID: 10495375.
Interestingly this study found directionally stronger hair count improvements for finasteride at 1-5 mg versus 0.2 mg. The researchers also noted that, unlike the other aformentioned study, adverse events were directionally higher in the 0.2 mg group – albeit not statistically different across groups.
Roberts JL, Fiedler V, Imperato-McGinley J, Whiting D, Olsen E, Shupack J, Stough D, DeVillez R, Rietschel R, Savin R, Bergfeld W, Swinehart J, Funicella T, Hordinsky M, Lowe N, Katz I, Lucky A, Drake L, Price VH, Weiss D, Whitmore E, Millikan L, Muller S, Gencheff C, et al. Clinical dose ranging studies with finasteride, a type 2 5alpha-reductase inhibitor, in men with male pattern hair loss. J Am Acad Dermatol. 1999 Oct;41(4):555-63. PMID: 10495375.
For these reasons, the investigators concluded that 1 mg daily of finasteride is the optimal dose for androgenic alopecia (AGA).
Which Dose Of Finasteride Should I Try?
When it comes to efficacy vs. safety considerations, the evidence on dose titration of finasteride is mixed. On the one hand, some studies show directionally lower risks of side effects on 0.2 mg versus 1.0 mg daily of finasteride – albeit at the expense of slightly lower hair counts. On the other hand, other studies show no statistical risk in adverse events at 0.2 mg versus 1.0 daily of finasteride – and thus recommend the 1.0 mg dose.
What doesn’t get measured in these studies is the magnitude of each side effect. In other words, “How bad is this side effect? And does it get better on a 10-point scale when I titrate down my finasteride dose?”
In our experience working with hair loss sufferers, we routinely see a lowered magnitude of side effects when individuals go from 1.0 mg to 0.2 mg daily of finasteride. While this may come at the expense of a little more hair growth, this might be appropriate for people who are experiencing finasteride-induced side effects or who just want to take as much risk off the table, while still getting some benefit from the drug.
For others with more moderate side effects, it’s likely that topical finasteride is more appropriate.
How Can We Achieve A Dose Of 0.2 mg Daily?
To achieve a finasteride dose of 0.2 mg daily, consider the following:
- Get a prescription for 1mg of finasteride daily, then use a pill cutter to quarter the pills (so that each daily dose is now 0.25mg versus 1.0mg). This will effectively net patients the same drug exposure as 0.2mg daily.
- Get a prescription for 1mg of finasteride daily, cut the pills in half (so you have 0.5mg per daily dose), and then take one half-pill each Monday, Wednesday, and Friday. This will net patients 1.5 mg of weekly exposure, whereas 0.2mg daily would net 1.4mg weekly – so they’re more or less the same.
We hope this information helps.
References[+]
Oral finasteride, a type II 5-alpha reductase inhibitor, is one of the most effective treatments available for androgenic alopecia (AGA). However, concerns over the drug’s potential side effects prevent many men from ever considering the medication.
In the last several years, nearly a dozen studies have shown that certain formulation topical finasteride may confer the same hair growth-promoting benefits as oral finasteride, but with less systemic drug exposure. For those worried about side effects, topical finasteride is often a stepping stone toward an effective treatment protocol for hair regrowth.
In this article, we’ll uncover how to make topical finasteride using crushed pills – which is perhaps the least expensive alternative to DIY topical finasteride. We’ll also explain why doing so might be short-sighted, and that there is likely a better way to reduce costs for topical finasteride, but also control for the quality of the product: diluting topical finasteride that is already purchased.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Why Make Topical Finasteride at Home?
Most people consider making DIY topical finasteride for two reasons:
- Cost savings. Compounding pharmacies often charge $50-$100 per month for topical finasteride, thus pricing out a lot of men and women who would otherwise be interested in trying it. However, oral finasteride prescriptions are often 2-5 times cheaper on a monthly basis. This has led many to wonder, “Why don’t I save money by crushing my finasteride pills into a solution and apply that topical directly on my head?” While there are cost-savings behind this logic, the actual step-by-step process is a little more involved (more on this later).
- Control over dilutions. Most big-brand formulations of topical finasteride use dilution percentages that actually expose users to more finasteride each day than if they were to use the drug orally. For instance, Hims sells a 0.3% topical finasteride solution, of which many customers will apply 1 mL daily or more to cover all balding regions. That actually equates to 3 mg of finasteride exposure daily, which is 3 times the amount of drug prescribed daily for oral consumption (i.e., 1 mg). Under these circumstances, if just 0.2 mg of that topical drug escapes the scalp skin and enters the bloodstream, most users will experience the same systemic hormonal modifications as if they were to instead just use the oral medication.
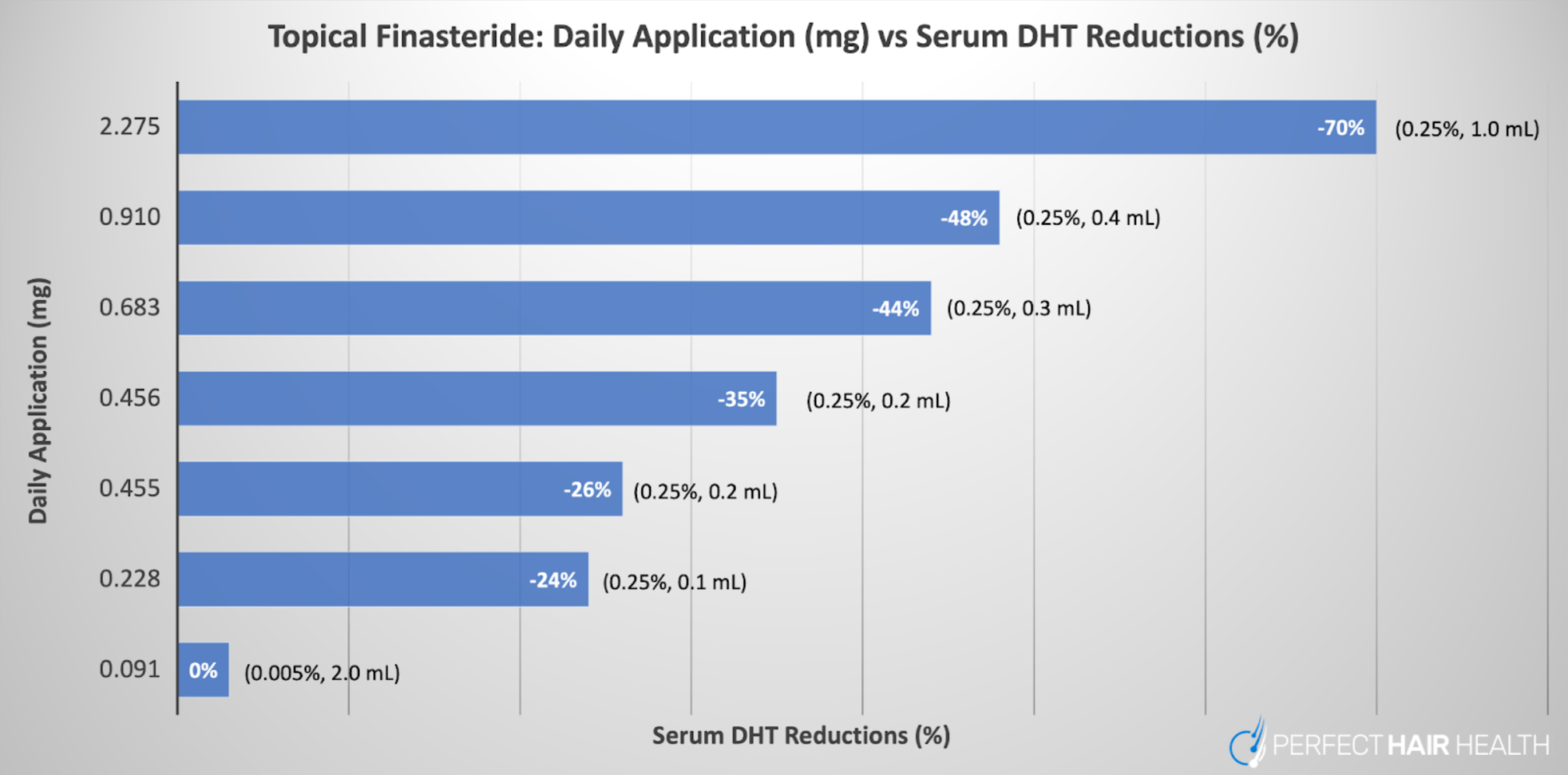
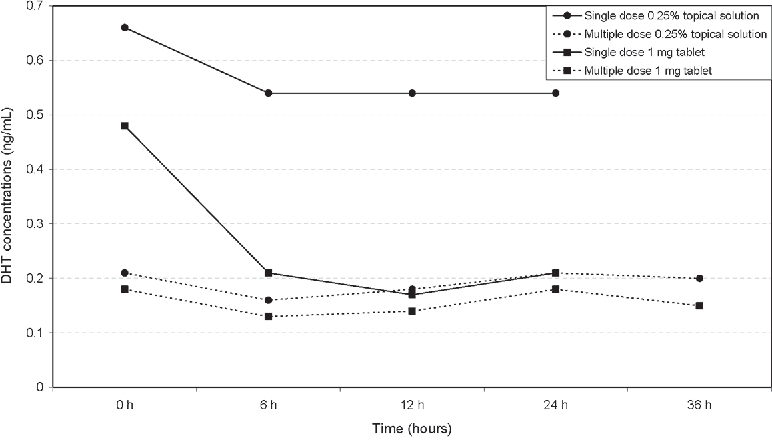
To elaborate on that second point, just see this chart below, which shows just how little finasteride needs to be consumed to affect serum levels of dihydrotestosterone (DHT).
With that in mind, see this other cart, which shows topical finasteride’s effects on serum DHT – a proxy to estimate systemic drug exposure – while controlling for the daily exposure (in mg) of finasteride applied to the scalp. The higher the daily dose, the higher the systemic reductions to DHT.
A graph representing the daily dose exposure of topical finasteride (y-axis) versus the amount of serum DHT reductions in participants (x-axis). Across all studies referenced in the graph, topical finasteride led to hair parameter improvements.
For these reasons, many topical finasteride users prefer to have the ability to titrate their daily dosages of the topical. They can do this by:
- Using less of the topical finasteride solution. The shortcoming: with less solution, you may not be able to apply the topical to all balding regions.
- Using topical finasteride fewer days per week. The shortcoming: this throws off the rhythm of a daily routine, and at a certain threshold, reduces efficacy.
- Making their own topical finasteride. The shortcoming: it takes additional work, and it’s not always clear if you did it right.
Moreover, a lot of people presume that making their own topical finasteride is as simple as crushing their own prescription of finasteride pills into water, then applying that directly to the scalp. It isn’t.
Nonetheless, most people opt for option #3: making their own topical finasteride. And most people prefer to do this by crushing pills. While we strongly recommend getting properly formulated topical finasteride prescribed to you and made by a compounding pharmacy, for those who don’t heed this advice and instead go with the pill-crushing approach, here are some educational resources (and a free calculator) that might help you.
Making Topical Finasteride By Crushing Pills: Step-By-Step Instructions
Before proceeding, it’s important to recall what your goals are for opting to try topical finasteride. In 99% of cases, it’s going to be to minimize the risk of adverse events while maximizing the chances of growing new hair. If that is your goal, consider the following.
1. Settle on a daily dose of finasteride exposure
Serum DHT reductions act as proxies for estimate systemic absorption from topical finasteride, and thereby the risk of side effects. On that note, the only clinically studied formulation of topical finasteride that (1) demonstrated effectiveness in men and women, and (2) led to no changes in serum DHT, is one that exposed participants to ~0.1 mg daily of topical finasteride. That’s 1/10th the amount of the standard oral dose prescribed for androgenic alopecia.
This dose of topical finasteride is typically applied daily in the following formulas:
- 0.01% at 1 mL daily – good for those with localized hair loss
- 0.005% at 2 mL daily – good for those with diffuse hair loss (more topical to spread across the scalp)
So, for those who want to be as cautious as possible, this is probably the dose that is most appropriate to start for topical finasteride. Higher doses also work, and also still likely confer a reduction in side effect risk, but at the expense of guaranteed DHT reductions elsewhere in the body. With the 0.1 mg low-dose formula, this risk (according to the clinical literature) is a lot lower.
2. Consider your carrier ingredients
Carrier ingredients help carry a topical’s active ingredient into deeper layers of the skin, where it can have its desired effects. In the case of topical finasteride, carrier ingredients help bring finasteride through the stratum corneum and into the mesodermal sheath, outer root sheath, and dermal papillae cell clusters of hair follicles – where the drug can get to work inhibiting type II 5-alpha reductase and lowering scalp levels of DHT.
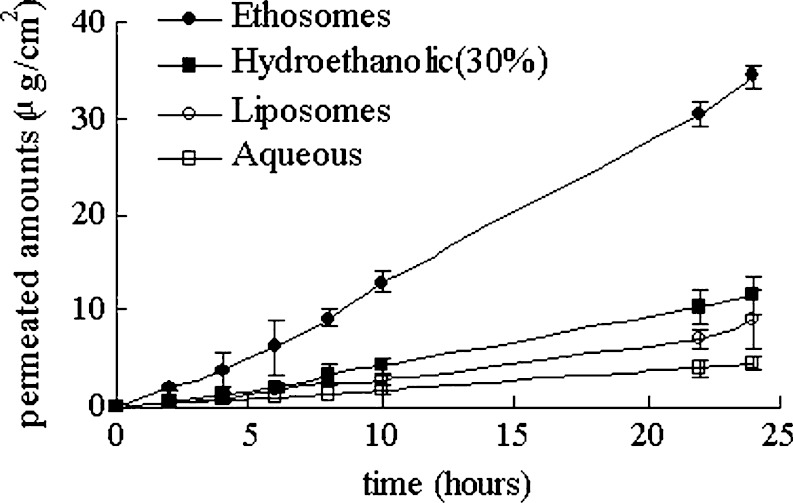
When it comes to topical finasteride, some carrier ingredients work better than others at enhancing skin penetration. For an example, see this chart:[1]https://ncbi.nlm.nih.gov/pmc/articles/PMC2977015/
Penetration profiles of finasteride permeating through human skin from different preparations (mean ± SD, n = 4)
There are a few key takeaways from this chart:
- Topical finasteride’s penetration into the skin is linear across 4 common carrier agents. That means the longer you leave in the topical without washing your hair, the more of it will absorb into the skin.
- This enhanced skin penetration will also come at the expense of higher systemic absorption of the drug. For these reasons, we recommend serum DHT testing regularly with topical finasteride. Our membership has resources on how to do this, and we can team members and researchers who will personally support you through this entire process if you need it.
- Don’t just mix your finasteride pills with water. As we can see from the chart, water is a terrible carrier ingredient for topical finasteride.
In our topical finasteride calculator (it’s free), we recommend a series of low-cost carrier ingredients for you, as well as links to the actual products. Please note: we do not receive commissions on any of those product recommendations, as doing so would undermine the objectives of this site: to be as unbiased as possible in our scientific analyses (and advice) to hair loss sufferers.
3. Before crushing your finasteride pills, remove any pill coatings
Depending on where your finasteride pills were manufactured, there’s a strong likelihood that the pills come with a protective coating that prevents incidental drug exposure from anyone who might’ve handled the drug without the intention of ingesting it. This is generally to prevent pregnant women from coming into incidental contact with the drug.
As such, if you’re going to use your prescribed finasteride pills to make topical finasteride, it’s critical to remove this protective coating from each pill in order to allow those pills a better chance at dissolving into the topical solution. You can do this by scraping the pills with a small knife or nail clipper, and then peeling off all of the coating with your finger tips.
4. Use our topical finasteride calculator to dial in your DIY, homemade topical finasteride formulation
The math involved in making topical finasteride isn’t likely solving an equation for quantum physics. At the same time, it’s also not as straightforward as many people would like. After all, you have to account for the following:
- Hair loss patterning (localized vs. diffuse) – as this determines if you’ll need 1 mL or 2 mL of liquid daily to cover all balding regions
- Dilution amounts – which, together with the mL of daily application, will determine your daily total finasteride exposure
- Adjustments for pill sizes (in mg). After all, oral finasteride pills are available in 1 mg and 5 mg daily doses, so you’ll need to account for this prior to crushing anything
To take the guesswork out of this process, we decided to do all of the math for you. Then we hired developers to integrate these math equations into an interactive survey that factors in all of the above, then spits out step-by-step instructions for how to make topical finasteride by crushing pills.
You can access the topical finasteride calculator right here. It’s 100% free.
To reiterate, this calculator is not medical advice and is for educational purposes only. We still strongly recommend that you just get your topical finasteride properly formulated by a compounding pharmacy. Nonetheless, the calculator is there for those who decide to ignore this advice and instead take the cost-savings route toward topical hair loss treatments.
Final Thoughts
We produce content like this to benefit hair loss sufferers worldwide – regardless of their financial circumstances. With more access to free information, more men and women can seek effective hair loss treatments faster, and thereby improve their outcomes long-term.
With that said, these free resources represent just a tiny fraction of what’s available inside our membership community. If you’re looking for personal support on your hair growth journey, interactions with hair loss researchers, one-on-one video calls, and a customized hair growth protocol built around your needs, preferences, goals, and the evidence – our membership is the place where we offer these services.
It’s also the way in which we monetize this site, which is why we’ve never accepted any hair loss product advertisements or commissions – which otherwise comes with the risk of biasing our own research lens and interpretation of data. For evidence of how big this problem is, just take a look all of the other sites, forums, and YouTube personalities catering their content toward the sale of physical products to which they’re financially tied – which only serves those creators and, simultaneously, overwhelms the ecosystem of information accessible to consumers with biased advice.
Not a member of the Perfect Hair Health Membership Community? Join today to create a customized regrowth roadmap and gain access to hundreds of resources, including treatment guides, product reviews, expert interviews, forums, and more.
Topical finasteride is an effective treatment for androgenic alopecia – particularly for those interested in better localizing the drug’s effects to the scalp. Unfortunately, prescriptions for topical finasteride can cost as much as $50-$100 per month.
This prices out a lot of people who would’ve otherwise committed to the topical had it not been for its costs. It’s also led others to ask, “Can I make topical finasteride at home? Can’t I just crush up my finasteride pills or dilute another topical finasteride?”
The answer to each question is yes, with caveats. While topical finasteride can be made at home – and at a low cost – anyone who intends to try this must formulate the product properly. This might entail:
- Removing the coating of finasteride pills prior to crushing them
- Matching carrier agents when diluting already-existing topicals
- Knowing how much finasteride to add to a formulation to maximize scalp DHT reductions while minimizing the risk of systemic absorption.
- Adjusting finasteride dilutions to control for total daily drug exposure, particularly for those with diffuse vs. localized hair loss.
In this article, we’ll reveal a step-by-step process for how to make topical finasteride – either by crushing finasteride pills or diluting an already-purchased topical. We’ll also provide a topical finasteride calculator that automatically calculates step-by-step instructions for you, all depending on your desired dilutions and starting ingredients. It’s also 100% free. See below.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Oral Finasteride: Efficacy & Side Effects
Oral finasteride is an effective FDA-approved drug used to treat androgenic alopecia (AGA). It reduces a hormone known as type II 5-α dihydrotestosterone (DHT) – which is causally linked to the balding process. Between 0.2 mg to 1.0 mg of finasteride daily can lower DHT levels by 70%, which is enough suppression to therapeutically improve AGA outcomes in 80-90% of male users in two years.[1]https://www.sciencedirect.com/science/article/pii/S0022202X15529357
While most men and women tolerate finasteride without issue, clinical studies consistently show that finasteride adversely impacts a small portion of users. Between 5% to 15% of men trying the drug report mild-to-moderate side effects ranging from diminished libido to brain fog. This is because finasteride does not just reduce DHT in scalp levels, but also across all other tissues in the body such as the brain and testes. For a portion of men and women, DHT reductions of this magnitude across all body tissues can cause undesired effects.
Topical Finasteride: Is It Still Effective? Does It Have A Better Safety Profile?
To reduce the risk of side effects from oral finasteride, many people opt to try finasteride delivered topically. After all, clinical studies demonstrate that compared to 1 mg daily of oral finasteride, daily use of 1 mL x 1% topical finasteride is “non-inferior” to the oral formulation.[2]https://pubmed.ncbi.nlm.nih.gov/19172031/
With topical delivery, users often believe that since they are isolating finasteride to the scalp skin, they can expect less finasteride to reach other parts of the body – thereby preserving DHT levels in other organ sites beyonds the scalp and lowering their risk of side effects.
But is this true?
On the one hand, clinical studies corroborate that some formulations of topical finasteride appear to reduce the risk of side effects versus oral finasteride. On the other hand, the dose per mL of topical finasteride influences this risk – as does the amount of topical finasteride applied daily along with the carrier ingredients used inside the topical formulation.
1. Daily Drug Exposure & Influence On Systemic Absorption
We can use reductions in blood levels of DHT as a proxy to estimate how much topical finasteride leaks from the scalp tissues into the blood stream, thereby traveling to other parts of the body and potentiating DHT reductions other organs.
For reference, see the following chart which summarizes clinical studies on topical finasteride in relation to its impact on DHT levels in the blood. Keep in mind that all daily exposure volumes of topical finasteride in this chart were clinically effective at improving hair. On the y-axis, we have the total daily exposure of finasteride (in mg) as a topical. On the x-axis, we show reductions to serum DHT, which acts to estimate the amount of systemic exposure of the drug (even when applied topically).
A graph representing the daily dose exposure of topical finasteride (y-axis) versus the amount of serum DHT reductions in participants (x-axis). Across all studies referenced in the graph, topical finasteride led to hair parameter improvements.
As we can see, according to the clinical literature, the only daily exposure volume of topical finasteride that improves hair loss but does not impact serum DHT levels is 0.005% x 2 mL of topical finasteride daily, which equates to just under 0.1 mg daily of finasteride applied to the scalp.
Therefore, if you intend to make topical finasteride at home with the goal of (1) improving hair loss while (2) minimizing systemic drug absorption, consider finasteride formulations of ~0.1mg daily. At higher daily doses, you can still achieve hair regrowth, but this might come at the expense with more systemic drug absorption and thereby a higher risk of side effects.
2. Carrier Agent Influence On Systemic Drug Exposure
Beyond the daily drug exposure of topical finasteride, the carrier agents used in a topical finasteride formulation also impact the amount of drug absorbed into the skin, and consequently the blood stream.
The outermost layer of the scalp’s epidermis is known as the stratum corneum. This skin layer acts as a barrier for what can enter the skin, and what can escape. It’s also a critical barrier of protection for human survival. Without a stratum corneum, human skin would absorb much more of the outside world – i.e., any nutrients, viruses, microorganisms, and/or pollutants we touch – and we’d be at a much higher risk of exogenous threats.
Unfortunately, the stratum corneum also creates a challenge for topical drug delivery. So, to bypass the stratum corneum, product formulators often add what are called “carrier ingredients” to a topical in order to allow for that topical’s active ingredients to penetrate beyond this layer and deeper into the skin — where that active ingredient can reach target areas and have a therapeutic effect.
Some common carrier ingredients are:
- Alcohol
- Propylene glycol
- Liposomes
But not all carrier ingredients are equally effective at their job. Depending on the active ingredient of a topical, some carrier ingredients do a better job than others at carrying a drug into deeper layers of the skin.
This is particularly true for topical finasteride. Just see this chart from an in vitro study measuring topical finasteride absorption across a 24-hour period, controlling for different carrier agents: ethosomes, hydroethanolic acid, liposomes, and water:[3]https://ncbi.nlm.nih.gov/pmc/articles/PMC2977015/
Penetration profiles of finasteride permeating through human skin from different preparations (mean ± SD, n = 4)
Keep in mind: the more finasteride that permeates into skin tissues, the more finasteride will absorb into the blood stream. Therefore, it is critical to be aware of this relationship when formulating your own topical finasteride at home.
Moreover, if you are diluting topical finasteride from a pre-existing topical, you must ensure that the carrier ingredients used in your new formulation match those used in the already-purchased topical. For instance, if your pre-purchased prescription of topical finasteride uses a liposomal base – which is a gel – but you’re trying to dilute that gel into a liquid formulation (with propylene glycol), the formulations won’t mix well, and you’ll just be wasting product.
How To Make Homemade Topical Finasteride
Here’s a recap of key factors to consider before opting to make topical finasteride at home:
- Control for daily drug exposure. Topical finasteride can still leak into the blood stream, and at higher daily doses, the effects of serum DHT can be as dramatic as if we were using oral finasteride. Formulations greater than 0.1mg daily appear to appreciably lower scalp DHT levels, but also serum DHT levels. With exposure volumes of 1.0 mg daily and higher, serum DHT can become reduced to the same level as oral finasteride.
- Control for daily application volume. Compared to people with localized hair loss (i.e., only temple recession and/or crown thinning), those with diffuse hair loss have a much larger area affected by pattern hair loss, and will thereby need to apply more topical (in mL) versus those with smaller areas of hair loss. Under these circumstances, finasteride dilutions may also need to be titrated in order to maintain a consistent daily exposure volume of the drug. For instance, if we want to keep total topical finasteride exposure consistent at 0.1mg daily, people with localized hair loss may only need 0.01% x 1 mL formulations, whereas those with diffuse hair loss may need to titrate their dilution to 0.005% x 2 mL in order to keep daily drug exposure constant while enabling an extra mL of liquid to cover all areas regions affected by hair loss.
- Select the right carrier ingredients. Different carrier ingredients have different capacities to bring finasteride through the stratum corneum. Over 24-hour penetration periods, one study found that topical finasteride formulated with ethosomes or hydroethanol outperformed formulations of liposomes or water. Having said that, better skin penetration also comes with higher rates of systemic absorption of finasteride into the blood stream.
- If making topical finasteride by diluting a pre-purchased topical, match the carrier ingredients. If you plan on diluting a pre-purchased topical finasteride, it is critical to match carrier ingredients between the old and new formulations.
- If making topical finasteride by crushing up oral pills, peel off any pill coatings prior to crushing. Failure to do this may resort in unwanted debris in your topical formulation and an incomplete dissolution of the finasteride pills in your topical.
If these factors feel overwhelming to control for, we completely understand. For these reasons, we decided to work with developers to build an interactive calculator that generates step-by-step instructions for you to make topical finasteride calculator.
DIY Topical Finasteride: Use Our Free Topical Finasteride Calculator
The following topical finasteride calculator not only controls for daily drug exposure, topical formulations, and carrier ingredients, but it also generates step-by-step instructions for your formulation based on whether you intend to make topical finasteride by crushing finasteride pills or by diluting an already-purchased topical.
Access the free topical finasteride calculator right here.
We hope it helps!
When Homemade Finasteride Is Not Recommended
Technically speaking, making DIY topical finasteride is never advised. After all, finasteride is a prescription drug, and prescription drugs require careful manufacturing and dosing control from compounding pharmacies.
For these reasons, if you’re not so price sensitive, it’s best to leave topical finasteride formulating to the professionals and take yourself out of the equation. Otherwise, you open yourself up to the risks of improper dosing and/or inadvertently exposing others in your household to a drug they never intended to use in the first place.
So, consider getting a prescription of topical finasteride from a dermatologist or telehealth company as your first (and only) option.
Nonetheless, we also recognize that people are going to do whatever they want to do, and that despite us strongly recommending against DIY topical finasteride, people will do it anyway. While this page (and the calculator) are for educational purposes only, and while we obviously recommend seeking medical advice before doing anything, we hope that the resources here help guide those who are making topical finasteride toward doing so as safely and effectively as possible.
Final Thoughts
While we don’t recommend making topical finasteride at home, we also recognize that DIY topical finasteride can save consumers money, enable better control over daily drug exposure, reduce systemic DHT reductions, and lower the risk of adverse events associated with the drug.
Users can make topical finasteride by crushing pills or diluting pre-purchased topical finasteride. Our 100% free calculator can generate step-by-step instructions for you.
Want To Take Your Hair Regrowth To The Next Level?
If you’re looking for personalized support in your battle against hair loss, our membership community is the place for you.
This is where we offer hair loss sufferers one-on-one support – all in an effort to save them years of time, money, and hair. In the first month of membership, community members are often able to simplify their regimen, improve their odds of success, and set themselves up to save tens of thousands of dollars over a lifetime. It starts with an education-first approach that prioritizes the interests of the individual over product sales.
Not a member? Join now to find your path to hair recovery.
We don’t affiliate with any products. Instead, we monetize this site through our personal services. We’ve found that this approach is far more effective for consumers looking to escape information overwhelm online, along with all of the product bombardments on their social media feeds about the latest and greatest “hair loss breakthrough” that, in most cases, is scientifically baseless.
We hope you consider joining. Otherwise, please continue enjoying our free resources.
References[+]
In the medical literature, it’s well established that women should withdraw from finasteride prior to conceiving, or abstain from finasteride throughout pregnancy and/or while breastfeeding. But what about men? Can they continue using finasteride during conception? What about during their partners’ pregnancy — when indirect finasteride exposure (via semen) might expose the female and fetus to the drug?
The answers and evidence aren’t so straightforward. Fortunately, for those concerned of adverse effects from finasteride on either sperm parameters or developing fetuses, there are strategies to mitigate these risks and continue protecting your hair. This article discusses the data, and elucidates a finasteride dosing strategy for men looking to keep their hair while also growing their family.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Is it Safe for Women to Use Finasteride During Conception?
Probably not. Here’s why.
Drugs like finasteride and dutasteride can bypass the placenta, where they can begin to inhibit 5-alpha reductase activity in a developing fetus and interfere with their hormonal profile.[1]https://rep.bioscientifica.com/configurable/content/journals$002frep$002f155$002f3$002fREP-17-0380.xml The hormone that these drugs reduce — dihydrotestosterone — is less relevant in adulthood, but is critical for early development — particularly for males. Subsequently, animal studies have shown that prolonged finasteride use in pregnant females may mutate and/or inhibit the development of male fetus genitalia.[2]https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=f96c6acd-4d02-4ece-bd54-2d5a35aab7f5#section-12.2
Since it takes ~30 days for finasteride to clear the body, doctors recommend that women trying to conceive should discontinue the drug at least one month before conception. Some doctors even advise their pregnant female patients to avoid any finasteride exposure whatsoever — including any handling of the medication. This is out of an abundance of caution for the safety of the developing fetus.
What About Men? Can Men Use Finasteride During Conception?
When it comes to men using finasteride during windows of conceptions opinions on whether or not to discontinue the drug are split.
Most providers recommend that men stop using finasteride for at least one month prior to conceiving. Other doctors claim that quitting finasteride is unnecessary, and come at the expense of lost hair. They even go so far as to say there’s “no evidence” that finasteride use for men during windows of conception leads to different health outcomes for their future offspring.
So, which position holds more merit? We’ll detail both sides of the scientific argument below and provide our own take on the data. Then, we’ll reveal a strategy men can employ to temporarily quit finasteride — within a window that shouldn’t compromise any hair gains — and use that window to conceive without (hopefully) losing additional hair.
It all boils down to the length of time it takes for finasteride’s hair growth-promoting effects to wear off versus the length of time the medication takes to clear from the system. Fortunately, there’s a difference here that is in favor of a reproductive window.
Should Men Stop Using Finasteride Before Or During Conception?
In one research article 2001, a group of family physicians examine the perceived versus realized risk of men using oral finasteride during windows of conception.[3]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2018472/pdf/11785276.pdf
First, the authors review evidence surrounding just how much of a daily 1mg finasteride pill ends up detectable in semen:
“In one study, semen levels were measured in 35 men taking 1 mg of finasteride daily for 6 weeks. Highest level measured was 1.52 ng/mL; mean level was 0.26 ng/mL.”
Then, the authors sought to use these numbers to estimate the total exposure of finasteride to a developing fetus — throughout a pregnancy — due to daily unprotected sex.
“Assuming a 100% vaginal absorption through a 5-mL ejaculate per day, women would be exposed to 7.6 ng/d, a negligible amount.”
Finally, the authors contextualized that risk by comparing that exposure level of finasteride to the amount that was required to cause birth defects in monkey studies.
“This level is 750 times lower than the “no effect” level for developmental abnormalities in rhesus monkeys.”
For these reasons, the authors conclude that finasteride use — at least in men — probably doesn’t need to be stopped during pregnancy.
The authors go on to state that, at least for men, the major risk of using finasteride during conception might instead be the drug’s temporary reductions to semen counts, which may impact the ability to conceive, but not the actual health of the baby (more on this later). Thus, while on finasteride, it may be more difficult to conceive within the first few months of using finasteride (when sperm parameters decline) if a male has borderline-low semen levels prior to starting the medication.
Beyond this, the authors seem far less concerned about about finasteride’s effects on the actual conception, such as potential congenital disabilities or risks to male fetal development. In fact, they assert that there’s no data that, during conception, the use of finasteride (for males) interferes with reproductive or fetal outcomes. They recommend that men taking oral finasteride keep taking the drug throughout conception.
Unfortunately, this review was conducted in 2001 — when epigenetics was still an emerging field, and when research groups weren’t necessarily aware of other markers worth measuring that might affect the health of fetal development. Today, in order to completely quell concerns of conception windows and finasteride use in men, what we really need to know is:
- Does finasteride use affect sperm parameters in men?
- Is finasteride use associated with epigenetic and/or DNA damage in sperm, and at doses prescribed for androgenic alopecia?
- Are there actual prospective, long-term studies measuring the outcomes of children whose fathers were using finasteride when they were conceived?
In exploring the answers to these questions, we’ll realize the whole debate over finasteride’s use in men during conception is not as clearcut as it may seem.
Finasteride Use In Men While Conceiving: Affect On Children (Unstudied!)
First, it is true that there are no prospective clinical studies tracking birth outcomes across men using vs. not using finasteride compared to (1) their success with conception, and (2) the health of their offspring. So, does that mean we should assume using finasteride as a male — during conception — is safe?
No. It simply means the question hasn’t been studied. Given that the life and health of a newborn is at stake, our position is that it’s probably better to exercise caution than it is to presume the absence of evidence is a signal suggesting that a behavior that has not been adequately studied must be safe.
Secondly, there’s newer data (from later than 2001) suggesting that finasteride may do more than temporarily lower the ability for men to reproduce via reduced sperm counts, and that these changes may come with a heightened risk of reproductive health.
Finasteride Use In Men: Affect On Sperm Parameters (Studied)
The effects of finasteride use on sperm parameters has been studied in humans at doses of 1-5mg. Here’s what the evidence says.
Overstreet et al (1999)
A 1999 study tested the effects of 1mg finasteride daily on sperm parameters in men. After one year, those using finasteride had an 11% decline in ejaculate volume, compared to 8% in the placebo group. The researchers also noted no significant changes to “sperm concentration, total sperm per ejaculate, sperm motility or morphology.” Resultantly, the team concluded that 1mg daily finasteride use “does not affect spermatogenesis of semen parameters” in men.[4]https://pubmed.ncbi.nlm.nih.gov/10492183/
Given that the study was randomized, double-blinded, placebo-controlled, used the standard dose of finasteride for androgenic alopecia, and ran a full year — many people use this study as justification that finasteride use (in men) probably does not have deleterious effects on reproduction.
Unfortunately, there are two problems with this reasoning.
The first problem is that the study doesn’t actually measure reproductive outcomes, nor does it measure epigenetic changes to sperm. Rather, the study only measure changes to sperms’ histological features: its concentration, sperm per ejaculate, sperm motility, sperm morphology, and semen volume. From a safety perspective, the clinical results are a positive signal. But without long-term studies on fetal outcomes for men using finasteride during conception, they don’t actually answer the question of whether finasteride use in men affect the way their offspring develop.
The second problem is that there are other studies on finasteride that add nuance to finasteride’s safety on sperm parameters — particularly at 5mg doses.
Amory et al (2007)
A 2007 study tested the effects of daily use of 5mg finasteride or 0.5mg dutasteride on semen parameters in healthy men versus placebo. Over six months, the authors found that daily doses of 5mg of finasteride or 0.5mg of dutasteride significantly reduced sperm counts, and by 25-35%. However, at the 12-month mark of continued use, sperm counts for finasteride were still below baseline (i.e., -14.5%), but no longer statistically significantly lower than when the study began.[5]https://academic.oup.com/jcem/article/92/5/1659/2598215
For these reasons, some researchers claim that the effects of finasteride on sperm parameters — if any — are temporary and resolve with continued use at 5mg daily doses, if those changes even exist at all at lower doses (i.e., 1mg).
This should be the end of the story, right? After all, most men are prescribed finasteride at 1mg daily for androgenic alopecia. So, if at 1mg daily there appears to be no effect on sperm parameters, and at 5mg daily the effect seems to diminish with continued use, then men must be in-the-clear to use finasteride while conceiving. Correct?
No.
Again, there are no randomized controlled clinical trials measuring long-term outcomes of children whose fathers were using finasteride before/during their conception. Moreover, sperm counts, motility, and morphogenesis are all histological features of sperm. What isn’t measured in these study is the effect of DNA expression on sperm (i.e., epigenetics). Keep in mind that you can have plenty of sperm that looks healthy, swims effectively, appears normal… but still carries with it genetic expressions that insinuate damage or a heightened potential of birth defects for the fetus.
So, is there any data giving us insights into the potential for finasteride to cause epigenetic changes to sperm (or damage to sperm) that might otherwise affect reproductive capacity and/or fetal outcomes? And no, we’re not talking mouse models that administer high-dose finasteride (which aren’t always applicable to human research, despite what hair loss forums may tell you).[6]https://www.mdpi.com/1467-3045/43/2/62 We’re talking about human evidence.
Yes, there is evidence. And while that evidence isn’t high-quality, it’s still worth discussing.
Finasteride And Infertility In Men: Tu HY, Zini A. (2011), Şalvarci A, Istanbulluoğlu O. (2012)
In 2011 and 2012, two separate case reports were published — each on a male using finasteride long-term who was having trouble conceiving with his respective partner.
Researchers examined sperm morphology — as the studies above did — which appeared normal. But then they examined the sperm through another endpoint: a sperm DNA fragmentation index. This is a measurement to approximate DNA damage (i.e., fragmentation). In both cases, sperm DNA fragmentation was elevated. After discontinuing finasteride, one case report saw sperm DNA fragmentation reduced from 30% to 16.5% within six months.[7]https://pubmed.ncbi.nlm.nih.gov/21292254/ The other case report noted similar improvements to DNA fragmentation, and the successful conception of a baby after discontinuing the drug.[8]https://pubmed.ncbi.nlm.nih.gov/23070721/ The implication: that finasteride use in some men may damage the DNA of sperm, and that discontinuing the drug can improve these outcomes.
Please note: these are case reports, and with such uncontrolled (and unrobust data), there is always the possibility that something aside from the discontinuance of finasteride might be explaining these results. For instance, upon receiving this news, both men featured in the case reports might not have only stopped finasteride, but also addressed other aspects of health to reduce their risk of sperm DNA fragmentation. Examples include supplementing with vitamin D, incorporating more daily activity into their lives, quitting alcohol, getting more consistent sleep, etc.
As such, there’s always the possibility that finasteride was not the majority causative agent in either outcomes.
Our Perspectives: Should Men Continue Using Finasteride While Trying To Have a Baby?
Given the balance of evidence, we feel that men using finasteride should perhaps exercise caution about continuing the drug while also trying to grow their families.
Unfortunately, this position puts many men in an uncomfortable situation. They might feel as though they need to withdraw from a hair-saving drug in order to minimize risks that aren’t necessarily clear, based on the clinical data. Again, those long-term studies on children whose fathers conceived them while using finasteride haven’t yet occurred. They may never occur.
At the same time, this “abundance of caution” comes at a tall expense: lost hair. After all, clinical studies show that after quitting finasteride, any hair that was preserved by the drug is lost and, soon after, hair loss continues at its normal rate.
These risks-benefits are not ours to make for anyone. We’re just here to communicate the data, and the debate. Risk tolerances vary depending on the person, and for many couples, the continued use of finasteride might be the decision made by the male during the windows of conception. This is a decision that shouldn’t be made by us; it should be made by you along with the counseling of your doctor(s).
Nonetheless, if you are looking for an approach to minimize the risks to a developing fetus while simultaneously preserving hair, there is a happy middle ground… and perhaps a way to get the best of both worlds.
Consider Quitting Finasteride Temporarily During Conception
Finasteride is a drug that has a terminal half-life of 5-7 hours, and a biological half-life of around two weeks. In other words, while it takes 5-7 hours for half of the finasteride in your bloodstream to metabolize, it takes 2+ weeks for half of the effects from finasteride use — i.e., the lowering of dihydrotestosterone — to go away. For these reasons, it is estimated that after quitting finasteride, it still takes ~30 days for finasteride (and its effects) to completely leave the body.
This is why most physicians recommend beginning trying to conceive after 30+ days away from finasteride (if female). But after quitting finasteride, how long does it take before hair loss starts to pick back up again?
According to the clinical literature, longer than four weeks. In fact, one study showed that, after one full year of use, men who quit finasteride were still above their baseline hair counts a year after leaving the medication.[9]https://www.sciencedirect.com/science/article/pii/S0022202X15529357 Another study showed that after one year of finasteride use, men who switched to every-other-month of daily medication had the same hair growth outcomes as men who still used the drug every day of the year.[10]https://pubmed.ncbi.nlm.nih.gov/15319158/
Taken together, these studies imply that finasteride’s terminal and biological half-lives might enable a key window whereby men can transition off the drug, conceive without detectable levels of finasteride in their sperm (and/or any adverse effects on sperm from finasteride use), then hop back on the drug — all with little (if any) risk to their hair.
Based on the current data, this window appears to be 1-3 months after quitting finasteride. Within that time, hair loss from withdrawal of the drug should be relatively minimal. And with a two-month reproductive window, this should hopefully be enough time to give couples a good chance to conceive.
What About Topical Finasteride?
For women, the use of topical finasteride within a month before conceiving, during pregnancy, or while breastfeeding is not recommended. This is because a portion of the drug will still go systemic, thereby potentiating adverse events to the developing baby.
For men, there is not yet any data to answer this question — but out of caution, many physicians will just say, “Don’t do it” However, it’s likely that the risks with topical finasteride — especially at lower exposure volumes (0.1 mg daily) — are much smaller than any risks with the oral formulations of the drug (due to less systemic exposure and thereby lower concentrations in the semen).
That said, for those using topical finasteride while starting a family, the Perfect Hair Health team recommends doing everything possible to minimize topical finasteride exposure to a partner while they are pregnant. We’ll have articles on how to do this in the near-future.
References[+]
References ↑1 https://rep.bioscientifica.com/configurable/content/journals$002frep$002f155$002f3$002fREP-17-0380.xml ↑2 https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=f96c6acd-4d02-4ece-bd54-2d5a35aab7f5#section-12.2 ↑3 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2018472/pdf/11785276.pdf ↑4 https://pubmed.ncbi.nlm.nih.gov/10492183/ ↑5 https://academic.oup.com/jcem/article/92/5/1659/2598215 ↑6 https://www.mdpi.com/1467-3045/43/2/62 ↑7 https://pubmed.ncbi.nlm.nih.gov/21292254/ ↑8 https://pubmed.ncbi.nlm.nih.gov/23070721/ ↑9 https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑10 https://pubmed.ncbi.nlm.nih.gov/15319158/ What’s the Prevalence Of Sexual Side Effects From Finasteride?
Does finasteride lower libido? The true incidence of the sexual side effects of finasteride remains up for debate. Finasteride is the world’s best-studied hair loss drug. Over the past 30 years, it has been clinically tested in over 30,000 men – with dozens of randomized, double-blinded, placebo-controlled studies converging on relatively consistent efficacy and safety profiles.
While finasteride is generally well-tolerated, some users have reported sexual side effects after starting the drug. These reports are well documented in the clinical literature. However, their prevalence varies greatly depending on the study referenced.
In well-controlled clinical studies lasting 1-5 years, the incidence of these reports is generally under 7%, with 3-4% of people in the placebo group (sugar pill group) also reporting reductions to libido.[1]https://pubmed.ncbi.nlm.nih.gov/30206635/ In fact, one randomized, double-blinded, placebo-controlled clinical trials in ~2,000 men showed – over one year – that 1 mg daily of finasteride led to reduced libido in 1.8% of users versus 1.3% in the placebo group.[2]https://www.sciencedirect.com/science/article/pii/S0022202X15529357
This seems to indicate the risk of sexual side effects from finasteride is very low. However, other (smaller) clinical studies have put the incidence of sexual side effects as high as 25-30%. It is important to note that these smaller studies are generally of lower quality, so their results should be interpreted with caution.[3]https://pubmed.ncbi.nlm.nih.gov/17655657/ Nonetheless, these studies and the side effects they mention do exist.
Having combed through nearly all the available literature on the subject, we estimate the true incidence of noticeable sexual side effects for finasteride users likely hovers around 3-15%.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Why Does It Seem Like Everyone Online Says Finasteride Lowers Libido?
While side effects from hair loss drugs do occur, their perceived prevalence is often overstated on natural health websites and online forums. There are (at least) two reasons why:
- Financial incentives. Many natural websites fearmonger over hair loss drugs because they want to sell people natural alternatives – supplements and serums – that they claim are free of sexual side effects (which often isn’t true).
- The “Yelp Effect”. The Yelp Effect explains why a restaurant is far more likely to receive reviews from patrons who are dissatisfied (rather than happy) with their experience. Anger motivates us to take action more than contentment does. The same applies to FDA-approved drugs on hair loss forums (especially in online spaces like HairLossTalk and Reddit, where anonymity is preserved).
Can We Reduce the Risk of Finasteride’s Sexual Side Effects?
Yes. There are strategies to potentially reduce the risk of finasteride’s sexual side effects, while also still regrowing hair. These strategies include the following:
- Reduce the dose. As a hair loss drug, finasteride is typically prescribed orally at 1mg daily. However, there is evidence that 0.2mg orally daily is nearly just as effective at improving hair counts.[4]https://pubmed.ncbi.nlm.nih.gov/10495375/ This dose also simultaneously reduces total drug exposure by 80%. For many people, this coincides with a reduction in perceived side effects.
- Try a topical formulation. Studies show that – when formulated properly – topical finasteride may reduce the risk of side effects by 30-90%. One 16-month study on 0.005% topical finasteride demonstrated significant hair improvements, no drug-associated side effects, and no impact on blood hormonal levels.[5]https://www.tandfonline.com/doi/abs/10.3109/09546639709160517 This suggests that, at this dose, there is not enough leakage of finasteride from the scalp into the bloodstream to appreciably alter hormonal profiles – at least in the men and women in this study (though some people inside our membership have reported a different experience).
- Try intradermal delivery methods. Also known as mesotherapy, intradermal delivery methods inject finasteride into the scalp. But rather than use finasteride, consider using another 5-alpha reductase inhibitor: dutasteride. Evidence suggests that scalp injections of 0.01% x 1-2 mL of dutasteride – once every 1-3 months – do not appreciably alter serum hormones, nor do they result in any reported sexual side effects. They do, however, still lead to statistically significant hair improvements. Mesotherapy is more commonly done with dutasteride than finasteride, as dutasteride has a longer half-life (days-to-weeks versus 5-7 hours). This means fewer mesotherapy sessions are required to lower scalp tissue DHT (the hormone lowered by finasteride) for sustained periods.
For topical formulations and intradermal injections, testing personal blood levels of DHT before and during treatment can offer peace of mind. By quantifying the exact changes to serum DHT levels, it’s possible to see just how much topical finasteride (if any) is going systemic. In general, DHT fluctuations smaller than 20% are considered biologically insignificant. For more information on how to test serum DHT, look inside our ultimate guide to finasteride treatment.
So, consider the data first and foremost before giving up entirely on finasteride. It’s by no means guaranteed that finasteride lowers libido. Regardless, there are ways to leverage the power of this effective drug and simultaneously mitigate its risks.
References[+]
What is Finasteride, and Which Finasteride is Best?
Finasteride is a drug approved by the FDA to treat benign prostate hyperplasia and androgenic alopecia. It is prescribed as a 1 mg daily tablet for men with androgenic alopecia. It is also prescribed in higher dosages for women suffering from female pattern hair loss. This ranges from 1.0-5.0 mg daily.
Finasteride is available in different formulations. The best option is determined on a patient-by-patient basis.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What Formulations of Finasteride are Available?
There are two main finasteride formulations: oral and topical. Doctors typically prescribe oral finasteride, as it’s a time-tested formulation with a high success rate. Many telehealth providers have sprouted up in recent years, offering topical and oral versions of the drug. Topical finasteride has become increasingly popular as more studies confirm its efficacy and relative safety versus oral finasteride.
As concluded in one study:
Topical finasteride significantly improves hair count compared to placebo and is well tolerated. Its effect is similar to that of oral finasteride, but with markedly lower systemic exposure and less impact on serum DHT concentrations. [1]https://pubmed.ncbi.nlm.nih.gov/34634163/
Clinical studies have shown that oral and topical formulations improve hair parameters equivalently in target area hair counts.[2]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9297965/ As such, many people looking to minimize their risk of side effects from finasteride often prefer the topical formulation, and they rationalize that decision by arguing that topical finasteride (1) is just as effective as oral finasteride, and (2) remains localized to the scalp, so it must not have any systemic effects elsewhere in the body.
In reality, both of these arguments are wrong.
- While studies do show that topical finasteride is equivalent to oral finasteride in “target area hair counts”, hair count changes outside of these target zones have not yet been measured. Therefore, it is possible that topical finasteride may not protect against hair loss wherever it isn’t applied, whereas the oral formulation tends to provide global protection across the entire scalp. In fact, if topical finasteride does offer hair loss protection in non-applied scalp regions, the most likely reason is that the drug went systemic (i.e., entered into the bloodstream), traveled throughout the body, and redistributed to those non-applied areas. In that regard…
- Topical finasteride can go systemic, depending on the dose. Several studies show that topical finasteride also lowers blood levels of DHT, particularly for daily doses totaling greater than 0.1 mg of finasteride exposure. While the amount of drug in circulation is still far less than that of oral finasteride, people trying topical finasteride should know this, and titrate their topical formulations accordingly. Just see this chart:
A graph representing the daily dose exposure of topical finasteride (y-axis) versus the amount of serum DHT reductions in participants (x-axis). Across all studies referenced in the graph, topical finasteride led to hair parameter improvements.
Finasteride 1mg Oral Tablets
As mentioned, finasteride is typically prescribed as a once-daily 1mg tablet. At 1mg daily, finasteride is sometimes branded as Propecia®. Using more than 1mg per day isn’t likely to improve results.[3]https://pubmed.ncbi.nlm.nih.gov/10495375/
However, it may increase the risk of side effects. Nearly all clinical studies use 1mg, as it’s the gold standard for treating male pattern baldness. 5 mg finasteride is typically used to treat men diagnosed with benign prostatic hyperplasia (under the label Proscar®).
Oral Propecia® (i.e., 1mg daily of finasteride) is prescribed under its brand name and as a generic formulation through many telehealth companies. Generic versions of the drug typically deliver similar results, and often at a fraction of the cost.
Finasteride Topical Formulations
Finasteride topicals include gels, liquid solutions, and liquid sprays. Foams are available as well.
A previous post centered on the best topical finasteride dosage determined that finasteride has a highly-sensitive and dose-dependent response curve.
In other words, 0.01 mg of finasteride barely reduces any DHT, while 0.2 mg reduces almost as much DHT as 5 mg, a much larger dose.[4]https://onlinelibrary.wiley.com/doi/10.1111/jdv.17738 1% topical formulations essentially guarantee systemic absorption.
Those aiming to avoid the side effects may want to consider a formula with lower percentages of the active drug.
Which Finasteride Is Best for Hair Loss?
It depends on two factors: (1) the presence of side effects, and (2) whether a patient has diffuse thinning or localized hair loss.
Side effects
When weighing the pros and cons of finasteride formulas, doctors often have patients start with oral finasteride. This is because oral finasteride has the strongest clinical evidence for treating male pattern hair loss, and it provides some degree of protection across all balding-prone areas.
If side effects occur on oral finasteride at 1 mg daily, doctors may consider lowering the dose to 0.2 mg daily to see if this reduces side effects. If issues persist, other options can be explored – such as topical formulations.
Under these circumstances, users may introduce topical finasteride at a 1-2 mL daily of 0.025% to 0.3% finasteride. If side effects persist, it may be necessary to lower that dose all the way to 0.005% x 2 mL daily, and start tracking serum DHT levels to measure – as a proxy – how much finasteride is actually going systemic (as these levels vary greatly depending on the person and any adjuvant treatments that might be influencing topical absorption – i.e., retinoic acid, microneedling, etc.).
Hair loss patterning
If someone wants to use topical finasteride, they should recognize that topical formulations of the drug are most appropriate for people who have localized hair loss (i.e., hair loss only at the temples and/or crown), rather than people with diffuse thinning (i.e., hair loss throughout the entire scalp).
This is because diffuse thinners have a larger area of the scalp to cover with a topical. That requires a higher amount of mL per application daily of topical finasteride to cover all zones. When holding constance the percentage dilution of topical finasteride, the more mL applied daily, the higher likelihood some of that additional finasteride will leak into the bloodstream and cause systemic effects – thereby defeating the whole effort of the topical in the first place.
For these reasons, diffuser thinners need to take extra care to titrate down their topical finasteride doses, or perhaps consider oral formulations of finasteride to maximize their scalp coverage and thereby improve their odds of long-term success.
For those who don’t experience any sexual side effects, long-term use of oral finasteride may be advisable, given its success rate. And for those who experience adverse systemic effects of oral finasteride, or those wary about potential issues with the oral formulation, topical finasteride may be the better option.
References[+]
Finasteride and dutasteride are drugs that lower the hormone dihydrotestosterone (DHT), which is a hormone that is causally linked to both benign prostatic hyperplasia and male pattern hair loss. While both drugs tend to be effective hair loss treatments for men, they do come with a risk of side effects –most commonly sexual side effects and the growth of male breast tissue (gynecomastia).
These side effects are believed to occur because of finasteride and dutasteride’s inhibitory effects on 5-alpha reductase – an enzyme that helps convert free testosterone into dihydrotestosterone. By inhibiting this enzyme, finasteride and dutasteride are able to therapeutically lower DHT levels to improve the symptoms of an enlarged prostate and/or regrow hair. However, the inhibition of 5-alpha reductase can come with undesired side effects in 5-15% of men using these drugs – mainly due to the hormonal shifts that occur throughout the body when 5-alpha reductase activity is suppressed.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Sexual Side Effects and Hair Loss Drugs
Based on clinical data (so far), there aren’t yet reliable blood tests to determine someone’s risk of sexual side effects from drugs like finasteride or dutasteride. Having said that, clinical studies show that men who have low levels of free testosterone and/or high levels of sex hormone binding globulin tend to be at the highest risk of “low libido”.[1]https://pubmed.ncbi.nlm.nih.gov/25800960/
As such, some clinicians have argued (anecdotally) that patients reporting side effects from finasteride and dutasteride tend to already have hormonal imbalances associated with reduced libido prior to starting the drug. As such, these same clinicians sometimes suggest that by taking measures to (1) improve free testosterone, and/or (2) reduce sex hormone binding globulin – these men tend to see improvements to libido and, as a consequence, sport a higher tolerability for drugs like finasteride and dutasteride.
So, if you’re worried about sexual side effects from finasteride and dutasteride, there is at least some anecdotal and observational evidence suggesting that testing free testosterone and sex hormone binding globulin might help to predict your actual risk tolerance. With that said, it’s important to note that the data here remains limited.
Gynecomastia and Hair Loss Drugs
Gynecomastia is the growth of male breast tissue. It results from prolonged, elevated levels of the hormones prolactin and/or estrogen.
When it comes to the use of finasteride and dutasteride, blood tests can likely be used to determine someone’s risk of gynecomastia from both drugs.
Gynecomastia is estimated to affect between 0.25% to 1% of healthy people using 5-alpha reductase inhibitors, with 5-year retrospective studies in men with benign prostate hyperplasia suggesting an incidence of up to 3% to 5%. [2]https://pubmed.ncbi.nlm.nih.gov/23067029/
Interestingly, those who start finasteride and/or dutasteride while already having elevated levels of prolactin and estrogen might be at a higher risk of developing gynecomastia. This is because drugs like finasteride and dutasteride can raise blood levels of both testosterone and/or estrogen by 10-20%, depending on the dose.[3]https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020788s020s021s023lbl.pdf[4]https://journals.sagepub.com/doi/abs/10.1177/2051415820926301 Consequently, as blood levels of estrogen and/or prolactin rise, these hormones can stimulate the growth of ductal tissue and alveolar differentiation – both of which relate to the growth of male breast tissue.[5]https://www.ncbi.nlm.nih.gov/books/NBK279105/
Hormones affecting growth and differentiation of breast tissue. Adapted from “Gynecomastia: Etiology, Diagnosis, and Treatment” (2019)[6]https://www.ncbi.nlm.nih.gov/books/NBK279105/
So, for those starting the drug with borderline-high estrogen, the additional lift in estrogen levels may put someone in the “danger zone” for gynecomastia.
Blood Tests for Finasteride and Dutasteride
For peace of mind, people can always order blood tests for prolactin and estrogen prior to starting finasteride or dutasteride. Additional tests can be performed further down the line.
If levels are within range, the risk of gynecomastia is likely much lower. This can be done with a primary care physician. Those based in the U.S. (and other countries that offer direct-to-consumer lab testing), can order tests through the links below.
- Direct-To-Consumer Lab Test: Prolactin (U.S. only)[7]truehealthlabs.com/product/prolactin
- Direct-To-Consumer Lab Test: Estrogen (U.S. only)[8]truehealthlabs.com/product/estradiol-e2
- Finasteride: Ultimate Guide (Member’s Only)
Not a member? Sign up today to become part of the Perfect Hair Membership Community. You’ll gain instant access to a customized hair regrowth plan, 50+ guides, 30+ product reviews, 60+ case studies, community support forums, expert interviews, and more.
What a Blood Test for Finasteride Can Do
While it’s up for debate if blood tests can actually predict someone’s risk of sexual side effects from finasteride or dutasteride, there is evidence that estrogen and prolactin levels pre-hair loss drugs might give some insights into the risk of developing gynecomastia. The totality of evidence suggests that finasteride and dutasteride may raise estrogen levels by 10-20%. Therefore, if your pre-finasteride levels of estrogen and/or prolactin are within 10-20% of the upper limit, it’s probably best to find ways to lower these levels before committing to the drug.
Diet, lifestyle, and environmental changes are often enough to normalize these hormones in many men.
What about Blood Tests to Predict Hair Regrowth from Finasteride?
The best predictor of hair regrowth from finasteride comes not from a blood test, but from an accurate hair loss diagnosis. After all, two-year clinical studies show that in otherwise healthy men with androgenic alopecia that presents in its standard horseshoe pattern, response rates for finasteride tend to hover around 80-90%.[9]https://www.sciencedirect.com/science/article/pii/S0022202X15529357
Recently, marketers have begun pushing genetic testing to determine someone’s response rate to finasteride and dutasteride. Preliminary data from poorly designed clinical studies suggests that perhaps there are some genes associated with higher-magnitude responses from both drugs, and also a better success rate. For instance, one study suggested that genetic “CAG repeat score” might help determine the response rate to finasteride, and that this data could be collected through blood draws.[10]https://pubmed.ncbi.nlm.nih.gov/31949455/
But again, the evidence here is limited and preliminary. Given the overwhelmingly high odds of a response to finasteride overall, we tend to place more weight on an accurate diagnosis than on genetic testings for either drug.
References[+]
References ↑1 https://pubmed.ncbi.nlm.nih.gov/25800960/ ↑2 https://pubmed.ncbi.nlm.nih.gov/23067029/ ↑3 https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020788s020s021s023lbl.pdf ↑4 https://journals.sagepub.com/doi/abs/10.1177/2051415820926301 ↑5, ↑6 https://www.ncbi.nlm.nih.gov/books/NBK279105/ ↑7 truehealthlabs.com/product/prolactin ↑8 truehealthlabs.com/product/estradiol-e2 ↑9 https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑10 https://pubmed.ncbi.nlm.nih.gov/31949455/ I Am Experiencing Side Effects From Minoxidil. What Should I Do?
If experiencing side effects from minoxidil, it’s best to speak with the prescribing physician as soon as possible to discuss next steps. Discontinuing use, however, is not the only option. There are strategies to reduce minoxidil’s side effects (while still benefiting from the drug) in both topical and oral formulations. This article will focus on reducing adverse events from topical minoxidil, depending on the side effect you’re experiencing:
- Skin irritation, dandruff, and/or dermatitis
- Water retention, skin aging, and/or bags under eyes
- Headaches and/or heart palpitations
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Topical Minoxidil: Reducing the Side Effects
For the most part, side effects from minoxidil are minor. Here are a few adverse events reported in the clinical literature (and online), the percent of people they tend to affect, and strategies on how to go about reducing or resolving them.
Skin Irritation, Dandruff, and/or Dermatitis (2-7% of users)
Skin irritation, dandruff, and/or dermatitis are the most frequently reported side effect from minoxidil. These tend to be reported by 2-7% of topical minoxidil users.
One study demonstrated that 80% of these reports were not actually due to minoxidil, but the carrier ingredient used to help minoxidil penetrate into the dermis: propylene glycol.[1]thaiscience.info/…ticle/JMAT/10986429.pdf
In these cases, simply switching to a minoxidil product without propylene glycol solved most reports of skin irritation. As such, if you’re experiencing these problems, you may want to experiment with switching minoxidil brands or formulations — specifically to a product that does not contain propylene glycol. Examples include:
- Essengen-5 NO PG FAST DRY (from MinoxidilMax)
If skin irritation persists on these new formulations, consider titrating the dose of topical minoxidil. You can achieve this by moving from twice-daily 5% minoxidil to once-daily 5% minoxidil. If that doesn’t work, try moving from once-daily 5% minoxidil to once-daily 2% minoxidil. If that doesn’t work, consider trying oral minoxidil at doses from 0.25mg to 5.0mg (more on this below).
Join the Community
Not a member? Join the Perfect Hair Health Membership Community to build your customized regrowth roadmap and receive personalized minoxidil recommendations.
Water Retention, Skin Aging, and/or Under-Eye Bags (prevalence unknown)
Some topical minoxidil users have reported under-eye bags and/or signs of accelerated skin aging. However, these reports are not reflected in the clinical literature; they’re anecdotal. Even still, while there’s currently no evidence (to which we’re aware) that minoxidil accelerates aging, there is a mechanistic argument to be made that topical minoxidil might increase the perception of skin aging and/or under-eye bags. This is likely due to two factors:
- Skin irritation/dryness from applying the drug, which can make the skin appear to be “more aged”. Keep in mind that studies suggest that 80% of these side effects are caused by propylene glycol and not the minoxidil itself. Therefore, switching brand formulations should resolve this dryness, and thereby the perception of accelerated skin aging.
- The drug leading to more water retention near the areas where it is applied. If this occurs in facial tissues, that water retention comes with the possibility of exaggerating the appearance of under-eye bags, and thereby skin aging.
If these side effects reflect your own experiences with topical minoxidil, consider the following:
- Wait and watch. For many people, the water-retaining effects of minoxidil are transient and go away with continued use of the drug.
- Switch formulations. Finding a minoxidil formulation without propylene glycol should go a long way toward resolving side effects related to dryness or skin irritation.
- Reduce salt intake. Salt is also water retentive, and it’s possible that people consuming lots of salt and using topical minoxidil might be at a higher risk of experiencing more exaggerated water-retentive effects from the drug. By lowering salt intake, this may be enough to resolve excessive water retention and diminish the presence of under-eye bags while still using minoxidil.
In most cases, these changes make enough of an impact to reduce, mitigate, or even eliminate these side effects altogether.
Headaches and/or Heart Palpitations(~1% of users)
In rarer cases, topical minoxidil results in heart palpitations or headaches. In some cases, these side effects are related to the formulation of minoxidil; in others, they’re due to the drug itself. If you don’t intend on quitting the drug outright after having experienced these effects, it’s critical to tease out which category you fall into — and to approach troubleshooting very carefully.
Headaches
If you’re experiencing a headache after topical minoxidil applications, the first question to ask is: is it the minoxidil itself, or an ingredient applied alongside the minoxidil that’s causing this experience?
In many cases, the scent of topical minoxidil is what’s causing someone’s headache after applying the drug topically. This can be due to a scent added to the formulation, or even the off-gassing of the alcohol (if you’re using a topical that contains ethanol or an alcohol as a carrier ingredient).
Under these circumstances, simply switching topical minoxidil brands to something that is (1) unscented, and (2) does not contain alcohol should be enough to resolve symptoms. If this doesn’t work, it’s likely that the headaches are a direct result of the minoxidil itself.
If this is the case, consider titrating the dose of minoxidil from twice-daily 5% minoxidil to once-daily 5% minoxidil. If that doesn’t work, consider trying nanoxidil — a minoxidil analogue that has a lower molecular weight and may confer a slightly better safety profile (at least according to very biased research from the company selling nanoxidil, DS Laboratories). Anecdotally, members of our community who have made this switch have mostly reported resolution of headaches secondary to topical minoxidil after switching to topical nanoxidil. So it’s not a bad idea.
If that doesn’t work, minoxidil may not be the right medication for you — at least when it comes to fighting hair loss. The good news is there are many other options.
Heart Palpitations
If you’ve noticed that, after applying topical minoxidil, your heart feels as though it “skips a beat” or begins beating irregularly, these signs are indication of a drug sensitivity to minoxidil itself. These effects are rare, but they likely impact up to 1% of people who have tried topical minoxidil.
If this is your experience, please speak to a medical professional and strongly consider discontinuing the medication. We do know of people who’ve managed these side effects by titrating the dose of topical minoxidil from twice-daily 5% to once-daily 5%, and even lower. Having said that, it’s critical to remember that our heart is more important than our hair. There are many other treatment options aside from topical minoxidil for your hair loss, and if you’re getting heart palpitations after applying the medication, it’s probably best to start exploring those rather than manage a medication that — despite being FDA-approved — still has lower qualities of evidence supporting its long-term use.
References[+]
References ↑1 thaiscience.info/…ticle/JMAT/10986429.pdf Topical Finasteride
Finasteride is a hair loss drug for treatment of androgenic alopecia. It inhibits the 5-alpha reductase type II enzyme that converts testosterone into dihydrotestosterone, or DHT.
Topical finasteride formulations (think: solution, spray, or gel) are growing in popularity because of their localized approach. Oral finasteride reduces DHT everywhere in the body. Topical finasteride reduces DHT in the area that it is applied while minimizing systemic absorption. That is, if you apply it correctly. The question of how to apply topical finasteride for the greatest benefit and the least systemic absorption has no single, easy answer.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Applying Topical Finasteride to the Scalp
Strategies for applying topical finasteride depend on its formulation (spray, solution, serum, or gel). We’ll review those first, then explain how long topical finasteride needs to stay on the scalp in order to absorb and have an effect (hint: it depends on the dosage and carrier agent).
How to Apply Topical Finasteride Spray
Many hair loss sufferers opt for topical finasteride spray. These sprays are often used to disperse the drug over wide surface areas of scalp skin, and are most appropriate for people with (1) thinning over widespread areas who also (2) are keeping their hair very short so that the scalp skin is easily accessible.
If you have diffuse thinning and longer hair, sprays might not be right for you. This is because too much of the topical finasteride will end up on the hair (where it does nothing) rather than the scalp (where hair grows from).
Users are advised to hold the spray bottle 2-3 inches from the scalp and apply the prescribed amount on the crown, top, and front of their heads. The hands can then be used to rub in any liquid blocked by existing hairs. This will ensure that a considerable amount of the formula has reached the scalp, particularly in problem areas.
Some users prefer to spray the formula into their palms and apply it manually with their fingertips. As with other formulas, parting sections of hair may make it easier to apply.
How to Apply Topical Finasteride Solution
Topical finasteride is also prescribed in the form of a liquid solution. These variations may include minoxidil as a combination therapy. Such formulas typically include a dropper for easy application. To apply the solution, part the hair in sections, apply a few drops along the part, then rub the solution around the scalp skin with the fingertips. Additional drops can then be applied to key problem areas (crown, top, and both sides of the front).
Alternatively, the prescribed amount can be dispensed into the hand and applied manually.
How to Apply Topical Finasteride Gel
Gel formulations are often made of bases of silica and/or liposomes. They’re very popular, as they are easy to apply. For best results, users can start with a few pumps of the gel, applying with their hands to provide even coverage across each problem area. The gel should gently be massaged into the scalp for 30 seconds or more.
No matter the treatment form, it’s only necessary to cover the areas affected by hair loss. Users should always follow their doctor’s recommendations and consult the directions included in their formula.
Any excess formula should immediately be washed off of hands and other areas of the body.
How Long Should Topical Finasteride be left on Scalp?
How long to let topical finasteride sit on your scalp depends on several factors, including:
- The dilution (the % finasteride in your topical formulation)
- The daily mL applied (alongside % dilution, this will determine your daily total exposure volume of finasteride in mg)
- The carrier ingredients in your topical (this will determine how much – and how quickly – topical finasteride will move from outside of the scalp into the dermis, where it can actually elicit an effect on hair follicles)
To understand more of this process, we also need to understand the stages of absorption when applying any drug to our scalp skin:
- Stage #1: the drug sits on the epidermis (i.e., the outermost layer of skin). Over several hours, some of the drug will absorb into the dermis, and some will evaporate.
- Stage #2: the drug absorbs percutaneously. This is when some of the drug moves from the epidermis into the dermis, where it can begin to affect hair follicles.
- Stage #3: the drug absorbs systemically. This is when some of the drug moves from the dermis into the bloodstream, where it can have systemic effects.
An in vitro study measured percutaneous absorption of topical finasteride across a variety of different formulations: ethosomes, ethanol, liposomes, and aqueous (water). [1]ncbi.nlm.nih.gov/pmc/articles/PMC2977015
This chart shows the percent of topical finasteride that percutaneously absorbs (enters the dermis) over a 24-hour period:
Penetration profiles of finasteride permeating through human skin from different preparations (mean ± SD, n = 4)
There are three big takeaways from this chart:
- The longer topical finasteride is left on the scalp, the more drug will be absorbed percutaneously.
- Across carrier ingredients, absorption rates appear to be linear. At 10 hours of contact, 2-13 micrograms of finasteride are absorbed into the skin, depending on the carrier. At 20 hours, those numbers double to 4-26 micrograms.
- When it comes to finasteride skin penetration, carriers rank from best to worst as ethosomes > hydroethanol > lipsomes > water.
Finasteride Absorption Rates
Leaving finasteride on the scalp for 10 hours allows for 5 micrograms per square centimeter of percutaneous absorption across most carrier agents. Is this enough finasteride to therapeutically lower scalp DHT levels? More importantly, is it enough to regrow hair?
There is not a clinical study that attempts to answer this. However, there is some surrogate data to help us approximate the answer.
A certain percentage of topical finasteride will absorb into the dermis. The bloodstream will absorb some of this later. Time-dependent DHT reductions in the bloodstream can be used to “ballpark” how much DHT is likely also being reduced in the scalp. This is because there is more topical finasteride that percutaneously absorbs than systemically absorbs. For lower dosages, the effects we see in the system can be used as signals for what’s happening – at a minimum – in the scalp skin.
This figure from a 2014 study measures the effects of one versus two applications of 1 mL of 0.25% topical finasteride on serum DHT levels. [2]https://pubmed.ncbi.nlm.nih.gov/25074865/
Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen levels in healthy male volunteers. International journal of clinical pharmacology and therapeutics, 52(10), 842–849.
Optimal Amount of Time
Six hours after applying 1 mL or 2 mL applications of 0.25% topical finasteride (i.e., 2.275-4.550 mg of finasteride), serum DHT reductions flatline. By the 6-hour mark, the 2ml application group experienced roughly the same serum DHT reductions as would be expected from a 1mg oral dose of finasteride.
Finasteride has an upper limit for its effects on serum DHT reduction. After ~70% reduction in serum DHT, adding more finasteride doesn’t reduce more serum DHT. The same is true with scalp DHT reductions.
Topical finasteride applications of 2.275 mg or higher (with a hydroxypropyl chitosan delivery vehicle) achieve systemic reductions in DHT. Furthermore, the larger dose of topical finasteride reduces systemic DHT levels on par with oral finasteride.
Serum DHT reductions are only achieved after scalp DHT reductions occur. Because of this, the conclusion can be drawn that 6-12 hours after applying topical finasteride, there’s likely enough percutaneous absorption to therapeutically lower scalp DHT levels for hair regrowth. However, this time window depends on a number of factors, including:
- Finasteride dilution (%). After all, the percent of finasteride, in part, determines your total daily exposure of finasteride. Remember that finasteride’s absorption is linear, so the higher the dilution, the more finasteride will absorb.
- The daily mL applied. Along with the percentage dilution of finasteride, the mL of topical applied will determine our total daily exposure of finasteride placed on the scalp.
- The carrier ingredients used. As we learned in the previous study, water-based carriers perform the worst at carrying finasteride into the skin, while ethosomes and water-alcohol mixes perform the best.
The next logical question becomes, just how low of a dilution of finasteride can you apply to evoke hair growth outcomes, while still potentially preserving serum DHT levels to the best of your ability? Interestingly, there seems to be a “sweet spot” for this at ~0.1 mg daily of topical finasteride mixed with alcohol and propylene glycol as carriers.
A 1997 study corroborates this. The study suggests that 2 mL daily of 0.005% topical finasteride (i.e., 0.0912 mg of finasteride exposure) improved hair parameters for men with AGA. This was achieved without affecting serum DHT levels – even after 16 months of treatment. [3]https://www.tandfonline.com/doi/abs/10.3109/09546639709160517
For Most Topical Finasteride Formulations, 6-12 Hours is Long Enough
Most big-brand topical finasteride companies are:
- Selling dilutions mixed with alcohol, propylene glycol, or glycerin
- Selling dilutions of 0.1% to 0.3% finasteride
- Advising patients to apply at least 1-2 mL daily
For most users, this will equate to 1 mg to 6 mg of topical finasteride exposure daily – which is more than 100x the minimum viable dose of topical finasteride. At these dosage ranges, the evidence suggests that topical finasteride will only need 6-12 hours on the scalp to therapeutically lower scalp DHT levels and start encouraging hair growth.
Topical finasteride users should use this time window to their advantage! But they should also keep in mind that, at most big-brand dose ranges, they’re exposing themselves to just as much (if not more) finasteride than the oral formulations. So they might be overpaying for topical finasteride in hopes of “localizing” its effects, only to also have just as much – if not more – of that drug going systemic.
To get the true benefits of localization, users will likely need to drop their dose as low as 0.005% x 2 mL daily, and periodically track serum DHT levels to ensure systemic effects are minimal.
The instructions on the packaging aren’t always as precise as the research. Best to trust the science when it comes to how to apply topical finasteride for maximum effectiveness, and minimal side effects.
References[+]
According to 2021 estimates, over 20% of people in the U.S. use antidepressants or antianxiety medications. Within this group of medications, hair loss is sometimes listed as a known side effect.
Mostly, these drugs cause temporary hair loss that goes away after a period of acclimation or after the drug is discontinued. But for the 1 in 5 people who use these drugs to support mental wellbeing, there’s a very real fear that mood stabilization might only be available at the expense of their hair.
The good news is that hair loss from anxiety medications is relatively uncommon. And while it can occur, there are ways to mitigate the risk of hair fall.
This article takes a scientific look at the relationship between the most commonly prescribed psychotropic drugs and hair loss, revealing science-based recommendations for those who are affected by hair loss and worried that their antidepressants are to blame.
- What type of hair loss is associated with psych meds?
- What evidence shows a link between psychopharmaceuticals and hair loss?
- Is there indeed a cause and effect relationship?
- What does this mean for those prescribed to these medications?
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Types of Drug-Induced Hair Loss
Drug-induced alopecia typically falls into two main categories: telogen effluvium and anagen effluvium. These names refer to the stage in the hair cycle where growth is interrupted.[1]https://link.springer.com/article/10.2165/00002018-199410040-00005
Telogen Effluvium
Telogen effluvium is the most common type of drug-induced hair loss. With this condition, hair follicles are prematurely triggered to enter their resting (telogen) phase, which induces early shedding. The condition typically becomes noticeable within 2-4 months of beginning treatment.
Anagen Effluvium
Anagen effluvium occurs during hair’s growth (anagen) phase. Hair cells that should be rapidly dividing are triggered to abruptly stop. This type of drug-related hair loss occurs within days or weeks of treatment and is not limited to the scalp. It is most commonly associated with chemotherapy drugs.
Medication-induced alopecia, in particular telogen effluvium, is a known side effect of some psychopharmaceuticals. Hair loss is typically temporary and may resolve on its own, or with a reduction in dosage. Discontinuation of these drugs nearly always leads to hair regrowth.[2]https://pubmed.ncbi.nlm.nih.gov/10798824/
What About Androgenic Alopecia (AGA)?
Psychopharmaceuticals do not cause androgenic alopecia (male pattern baldness), but may temporarily accelerate the condition. If several hair follicles in AGA-prone regions suddenly enter the telogen phase, hair follicle miniaturization can increase, speeding the progression of AGA.
Psychotropic Medications and Hair Loss: The Research
It’s important to note that just because hair loss is listed as a potential side effect of certain medications does not mean it will happen to everyone. But, it can still happen. What’s most important is the percent of people reporting hair loss in a clinical trial for any of these drugs.
Among the class of drugs referred to as psychopharmaceuticals, hair loss is most frequently associated with long-term use of lithium and valproic acid.
As many as 19% of lithium users and 12% of valproic acid users report the unwanted side effect.[3]https://pubmed.ncbi.nlm.nih.gov/10798824/ For most other drugs, risk is much lower, and possibly as low as 0.01%.
To learn more about the actual connection between antidepressants, anti-anxiety medications, mood stabilizers and hair loss, the Perfect Hair Health team combed through hundreds of case studies.
The Process
First, the team looked at the most commonly prescribed medications and then ran those drug names through a research database to pull up any and all studies that mentioned hair loss. Below is an overview of what was found.
If a drug is not the list, it’s either because no reports of hair loss were not found or commonly prescribed. Others were simply not worth mentioning for various reasons. To jump to a particular drug, use the links below.
Antidepressants
- SSRIs
- Escitalopram (Lexapro)
- Fluoxetine (Prozac)
- Paroxetine (Paxil)
- Sertraline (Zoloft)
- Atypical Antidepressants
- Bupropion (Wellbutrin)
- Atypical Antipsychotics
- Olanzapine (Zyprexa)
- Quetiapine (Seroquel)
Anti-Anxiety Medications
- Benzodiazepines
- Clonazepam (Klonopin)
- Buspirone
- Antimantic Agents
- Lithium
- Valproic Acid
Evaluation Of Evidence Quality
Of the several studies cited below, most consist of case reports that reference just a single individual. What’s more, these case reports often make it to publication precisely because of their uniqueness. In these reports, researchers often make reference to ‘the first known case’ or ‘the only known case.’
Case studies rank relatively low on the hierarchy of evidence, as they are very anecdotal, and not supported by double-blind research, for example. This makes it hard to know the true incidence or prevalence of hair loss from many of these drugs.
Antidepressants and Hair Loss
Antidepressants are of several different classes, namely selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), MAOIs, and atypical medications.
SSRIs and Hair Loss
Selective serotonin reuptake inhibitors (SSRIs) are among the most commonly prescribed antidepressants and are sometimes used to treat anxiety disorders. Of these, hair loss is associated with the following medications:
- escitalopram (Lexapro)
- fluoxetine (Prozac)
- paroxetine (Paxil)
- sertraline (Zoloft)
The team did not find any studies linking two other common SSRIs, protriptyline (Vivactil) and amitriptyline (Elavil) with hair loss. Take a closer look at what was found.
Escitalopram (Lexapro)
2021 Case Study:
A male patient diagnosed with major depressive disorder experienced hair loss with escitalopram. The patient discontinued escitalopram when he could no longer tolerate the hair loss. The hair loss stopped within one month.[4]https://www.psychiatria-danubina.com/UserDocsImages/pdf/dnb_vol33_no2/dnb_vol33_no2_187.pdf
2020 Case Report:
A female patient on 5mg escitalopram reported minor hair loss. After her dose was increased to 10mg, hair loss became ‘significant.’ After quitting the drug due to intolerable hair loss her symptoms ‘dramatically’ resolved within one week.[5]https://www.psychiatrist.com/pcc/depression/escitalopram-induced-hair-loss/
2016 Case Study:
This case study is unusual because the female patient presented with eyelash loss 12 weeks after beginning treatment with escitalopram. Her eyelashes returned to near normal 5 weeks after she stopped taking the medication.
While SSRIs are known to cause alopecia, this is the first reported case of eyelash loss. Researchers noted the timing of the patient’s presentation was consistent with the growth cycle of eyelashes, suggesting the mechanism of loss was interruption of the hair growth cycle.[6]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5035805/
Norton DJ, Cates C. Eyelash Loss Secondary to Escitalopram But Not to Sertraline: A Case Report. Prim Care Companion CNS Disord. 2016;18(3):10.4088/PCC.15l01887. Published 2016 May 19.
2011 Case Study:
Woman suffering from major depressive disorder noticed hair loss 3 weeks after beginning escitalopram. She discontinued the drug due to intolerable hair loss. Two weeks later, the hair loss stopped.
Several months later she tried the drug again as her depressive symptoms had returned. After 2 weeks? Her hair loss returned.[7]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3219523/
Fluoxetine (Prozac)
2021 Case Study:
Six weeks after beginning fluoxetine, a male patient reported hair loss in the frontal region of the skull. His complaints ended after he stopped taking the drug. This case appears to be the first evidence of fluoxetine-related hair loss in men.[8]https://pubmed.ncbi.nlm.nih.gov/34355374/
2019 Case Study:
Six weeks after beginning fluoxetine, a female patient reported hair loss in the frontal region of the skull.[9]https://pubmed.ncbi.nlm.nih.gov/31599441/
2018 Case Report:
Female patient notices significant hair loss 2 weeks after beginning fluoxetine. By 18 months, she had lost all her scalp and body hair. Researchers are aware of just one other similar case, and suggest there may be a wider spectrum of fluoxetine-related hair loss than previously known.[10]https://www.cambridge.org/core/journals/the-british-journal-of-psychiatry/article/hair-loss-associated-with-fluoxetine/D2A55E09DDCF393399C14DB9289BAADE
2004 Case Study:
Female patient reports slight hair loss 3 months after beginning fluoxetine 20mg treatment. Reducing the drug to 10mg had no effect on her hair loss. After 1 year she stopped treatment due to intolerable hair loss on the scalp and body. 4 weeks later, her hair returned to normal thickness.[11]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC514846/
1992 Drug Trial:
A group of 15 young adults, ages 16-24, trialed fluoxetine for the treatment of their depression. Researchers noted that the side effect of alopecia appeared more commonly than in adult studies.[12]https://pubmed.ncbi.nlm.nih.gov/19630647/
Paroxetine (Paxil)
2021 Case Report:
Male with social anxiety disorder reports hair loss after beginning treatment with paroxetine. The hair loss stopped after discontinuing the drug, then recurred when treatment with paroxetine began again.[13]https://pubmed.ncbi.nlm.nih.gov/34181746/
2006 Case Study:
Over 60% of patients with alopecia areata (AA) have a psychiatric comorbidity. Researchers hypothesize that treating this depression may have a positive effect on hair growth. In this case study, a female patient had complete hair regrowth 7 weeks after beginning paroxetine treatment. One month after discontinuing the drug, her symptoms of AA had returned.[14]https://sci-hub.se/https://pubmed.ncbi.nlm.nih.gov/16922952/
2001 Double-Blind Randomized, Placebo-Controlled Trial:
A group of 13 patients presenting with AA and a psychiatric comorbidity were studied to see if treatment with SSRIs could lead to regrowth of hair. Researchers observed the complete regrowth of hair in two patients treated with paroxetine, while four showed partial regrowth. Meanwhile, only one patient from the placebo group had similar regrowth.[15]https://pubmed.ncbi.nlm.nih.gov/11737460/
2000 Case Report:
‘Massive’ hair loss was reported in a woman being treated with paroxetine, which improved soon after she stopped taking the drug.[16]https://pubmed.ncbi.nlm.nih.gov/10883182/
1999 Case Report:
A female complained of ‘moderate’ hair loss after beginning paroxetine treatment. The hair loss stopped after discontinuing the drug, and began again when the drug was reintroduced.[17]https://pubmed.ncbi.nlm.nih.gov/10442258/
Sertraline (Zoloft)
2015 Case Study:
A male patient reports hair loss 2 weeks after beginning sertraline treatment. His hair loss improved after he stopped taking the drug.[18]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4589582/
2008 Case Study:
A woman complained of hair loss during sertraline treatment. This case is unique because she had previously been treated with fluoxetine, but reported no hair loss during that treatment.[19]https://pubmed.ncbi.nlm.nih.gov/18664165/
2006 Literature Review:
Researchers reviewed all reports of SSRI-induced hair loss in the national Swedish database and the database for the World Health Organization. Reports of sertraline-induced hair loss were nearly double those for citalopram, although still considered a ‘rare’ side-effect.[20]https://pubmed.ncbi.nlm.nih.gov/16783834/
2005 Case Study:
A 14 year old boy reports hair loss after 5 years of sertraline treatment. The drug was gradually discontinued and the hair loss stopped.[21]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3000200/
Atypical Antidepressants
Bupropion (Wellbutrin)
Bupropion is an atypical antidepressant. It is not an SSRI or an SNRI, but an NDRI. It’s used not only to treat depression, but sometimes prescribed to help with smoking cessation.
2018 Research:
In a study that followed over 1 million patients, researchers tracked exclusive users of antidepressants to better understand hair loss risk. They found that compared with bupropion, all other antidepressants had a lower risk of hair loss. Fluoxetine and paroxetine ranked as being lowest risk, with the highest level of confidence.
The researchers concluded that of all the SSRIs and SNRIs, bupropion has the highest risk of hair loss, while paroxetine has the lowest.[22]https://pubmed.ncbi.nlm.nih.gov/28763345/
Atypical Antipsychotics
Olanzapine and quetiapine are classified as atypical antipsychotics. Both are used in the treatment of bipolar disorder, while quetiapine is also sometimes used to treat major depressive disorder.
Olanzapine (Zyprexa)
2002 Case Report:
After increasing her daily dose of olanzapine from 5mg to 15mg daily, a woman reports increasing hair loss. Hair loss discontinued after switching from olanzapine to risperidone. Researchers report that this is the first known case of olanzapine-induced hair loss, and note the manufacturer, Eli Lily Canada, estimates hair loss occurs among just 0.01% of users.[23]https://sci-hub.se/https://pubmed.ncbi.nlm.nih.gov/12500769/
Quetiapine (Seroquel)
2007 Literature Review:
Researchers reviewed all case reports of alopecia following quetiapine treatment reported to the New Zealand Intensive Medicines Monitoring Programme and the World Health Organization. They found 17 cases total, some of which support a causal relationship between quetiapine and hair loss. Researchers note that while hair loss has previously been associated with both olanzapine and risperidone, it has not yet been described with quetiapine.[24]https://pubmed.ncbi.nlm.nih.gov/17293712/
Anti-Anxiety Medications and Hair Loss
Benzodiazepines and Hair Loss
Benzodiazepines are classified as depressants and can help patients who struggle with anxiety. Of this class of drugs, only clonazepam seems to be associated with hair loss. There were no studies connecting alprazolam (Xanax), diazepam (Valium) or lorazepam (Ativan) with hair loss.
Clonazepam (Klonopin)
2009 Case Study:
A woman complains of hair loss one week after beginning treatment with clonazepam. The hair loss stopped when she stopped taking the drug. Researchers note they reviewed the literature and found only one other case of hair loss associated with clonazepam. Also noted, she had been taking escitalopram (which has a more well-documented association with hair loss) prior to her hair shedding and continued with this drug even as her hair grew back.[25]https://pubmed.ncbi.nlm.nih.gov/19471188/
Buspirone
Busiprone is categorized as an anxiolytic, and is used to treat both short-term and chronic anxiety.
2013 Case Report:
A woman being treated with buspirone and sertraline reports significant hair loss. Her treatment team discontinued buspirone, but not sertraline (which is also associated with hair loss). The patient reported her hair loss stopped 3-5 days later, although researchers noted they couldn’t confirm.[26]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3579479/
Antimanic Agents
Antimanic agents are mood stabilizers. They are used in the treatment of bipolar disorders and can calm those in states of mania or extreme anxiety. Of this class of drugs, lithium and valproic acid are most frequently associated with hair loss.
Lithium
2013 Literature Review:
A meta analysis on the side effects of lithium finds no statistically significant increase in risk of hair loss with lithium treatment. Researchers mention that lithium suppresses the thyroid, which could be related to reports of hair loss.[27]https://pubmed.ncbi.nlm.nih.gov/23525591/
2012 Literature Review:
A review of 385 abstracts were screened for reports of lithium toxicity, including lithium-induced alopecia. Researchers found no significant risk of alopecia associated with lithium.[28]https://pubmed.ncbi.nlm.nih.gov/22265699/
1994 Case Study:
A woman reports hair loss after beginning lithium treatment. 2 months after discontinuing treatment, her alopecia resolved. Researchers note this side effect is rare.[29]https://pubmed.ncbi.nlm.nih.gov/7995585/
1988 Case Study:
A patient reports total hair loss after 2 months of lithium treatment. Researchers discuss a possible connection between the drug and alopecia totalis.[30]https://pubmed.ncbi.nlm.nih.gov/3126095/
1984 Literature Review:
Researchers review all cases since 1970, when lithium-related hair loss was first reported. They suggest patients experiencing hair loss after lithium treatment be checked for hypothyroidism, because lithium ions may concentrate in the thyroid, disrupting normal processes in this area.[31]https://pubmed.ncbi.nlm.nih.gov/6519870/
1983 Case Report:
Of 100 patients on lithium therapy, 12 report hair loss. 1 patient was diagnosed with hypothyroidism, others experienced hair regrowth after discontinuing treatment.[32]https://pubmed.ncbi.nlm.nih.gov/6838778/
1983 Case Study:
Researchers present 2 cases of hair loss attributed to lithium therapy. They recommend thyroid tests to exclude hypothyroidism as the cause of hair loss.[33]https://pubmed.ncbi.nlm.nih.gov/6404546/
1982 Case Report:
Researchers study 7 cases of hair loss associated with lithium. They mention other findings that for those with scalp psoriasis, lithium can aggravate this condition. They report findings consistent with telogen effluvium and conclude lithium can cause increased hair shedding. [34]https://pubmed.ncbi.nlm.nih.gov/6809028/
Valproate/Valproic Acid
2018 Case Study:
A review of over 400,000 patients being treated with psychotropic drugs in German-speaking countries found that Valproic acid was related to the highest risk of hair loss. That said, researchers found just 43 cases, a number distinctly lower than expected.[35]https://pubmed.ncbi.nlm.nih.gov/30193142/
2018 Literature Review:
A review of literature finds Valproate-induced hair loss is diffused, nonscarring, and dose-related. The drug may also cause graying and changes to hair texture. Researchers note that topical valproic acid may help hair regeneration and suggest further study into the difference between oral and topical administration as they relate to changes in hair growth.[36]https://pubmed.ncbi.nlm.nih.gov/30386073/
2017 Research:
Hair loss is a well-known side effect of valproic acid. It leads to telogen effluvium and appears to be dose-dependent, meaning hair loss increases with increased dosage. Paradoxically, valproic acid can be applied topically to help regrow hair. Researchers explore this paradox.[37]https://pubmed.ncbi.nlm.nih.gov/29061425/
2017 Research:
Valproic acid is often administered to patients undergoing radiation therapy for brain tumors to help manage seizures. Doctors note that delay or prevention of hair loss in this population seems to be a positive side effect.[38]https://pubmed.ncbi.nlm.nih.gov/27889835/
1996 Literature Review:
A 1996 literature review finds that alopecia is a common side effect of treatment with antimanic agents, and is expected in up to 12% of patients undergoing treatment with valproate, and 10% of patients being treated with lithium.[39]https://pubmed.ncbi.nlm.nih.gov/8899137/
1992 Case Report:
Two reports with seemingly conflicting results. In one, lithium-induced hair loss improved when lithium was replaced by valproate. In the other, hair loss improved only after stopping valproate.[40]https://pubmed.ncbi.nlm.nih.gov/1486112/
Establishing Cause and Effect
The above case studies offer evidence that the relationship between psychopharmaceuticals and hair loss may be more nuanced than expected.
Case reports are justifiably considered a weaker form of evidence. In other words, they rank low on the hierarchy of evidence. While case studies (i.e., n=1 published anecdotes) offer ‘signals’ for scientists to explore in future randomized controlled clinical trials, they also come with a high risk of bias. Case studies don’t establish prevalence rates, and it’s always possible that patients in these reports were using other medications and/or experienced additional life events that might also explain their hair loss. While they indeed contribute to future research, using case studies to interpret cause and effect is difficult.
The bottom line? It’s not always easy to say, ‘this drug causes hair loss.’
Hair loss can have many causes, making a causal relationship between a single drug and hair loss very hard to prove. In some of the above studies on SSRIs, researchers tried to hone in on this relationship by stopping treatment, seeing if the hair grew back, and then rechallenging the patient with the same medication to see if their hair loss then returned.
While this process occurred in a few of the case studies found, more research must still be done, as the sample size in the studies mentioned here consists of just a few people.
Another way to explore the existence of cause-and-effect is to dive more deeply into possible causes outside of the drug. Basically, one must rule out the following:
Are other medications present?
Patients receiving treatment for depression and anxiety may have other comorbidities or may be taking more than one medication.
Some of the case studies above report this, others make it clear the patient was an ‘exclusive user’ of the drug in question, while others make no such reference to either.
It’s possible that even in cases where one medication was discontinued and hair loss subsequently stopped, that it was the interaction between one or more drugs, and not a single drug, that triggered the onset of hair shedding.
Are other skin conditions present?
Several of the studies on lithium, which is widely described as a drug which ‘causes’ hair loss, mentioned patients who had pre-existing skin conditions, particularly scalp psoriasis. While it may be true that they experienced alopecia only after beginning lithium treatment, the root cause of hair shedding could have been their psoriasis.
Several patients in the above case studies had also experienced alopecia in the past. Alopecia is an embarrassing condition which can lead to depression and anxiety. Also, those with depression and anxiety may be predisposed to alopecia. Researchers estimate that up to 30% of patients with skin conditions have psychiatric comorbidity.[41]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7756276/
Is the patient trustworthy?
Of the case reports presented here, researchers often (but not always) confirmed the patient’s own reports of hair loss and hair regrowth. In just one instance was there mention of blood drawn to confirm the patient was indeed taking only the medication prescribed, as directed.
Particularly regarding the anti-anxiety medications, some patients presented with symptoms of schizophrenia. Hair pulling, or trichotillomania, must be ruled out in those cases and others.
Understanding the Mechanisms of Action
Understanding the mechanisms of action can also help establish cause and effect. While it’s still unknown exactly how these medications induce hair loss, the following are a sample of the most plausible theories.
Melatonin and Hair Cycle Interruption
Psychotropic drugs typically cause diffuse, reversible alopecia by influencing hair’s telogen (shedding) phase.[42]https://pubmed.ncbi.nlm.nih.gov/10798824/ The exact mechanism via which this works has yet to be revealed, although it may be connected to the relationship between serotonin and melatonin.
Serotonin is a precursor to melatonin, but the two also work in tandem to regulate a healthy circadian cycle. SSRIs in particular may decrease melatonin, which plays a role in hair cycle control.[43]https://pubmed.ncbi.nlm.nih.gov/16217127/
Slominski A, Fischer TW, Zmijewski MA, Wortsman J, Semak I, Zbytek B, Slominski RM, Tobin DJ. On the role of melatonin in skin physiology and pathology. Endocrine. 2005 Jul;27(2):137-48.
Hypothyroidism
Lithium and other antimanic agents may exacerbate hypothyroidism. While lithium cannot seem to shake its reputation as a cause of hair loss, researchers do not necessarily believe that lithium is the direct cause. Rather, it seems that hypothyroidism (caused by lithium) is to blame. Insufficient thyroid hormone can trigger telogen effluvium.
Dose Dependency
With lithium and some other psychopharmaceuticals, dosage plays a role in determining risk for hair loss. Some patients who experience hair loss may find that reducing their dosage allows their hair loss to abate. Lithium in particular, rarely has to be discontinued entirely.[44]https://pubmed.ncbi.nlm.nih.gov/3157663/
Understanding more about the relationship between dose and hair loss may help researchers learn more about the mechanisms behind cause and effect.
Women vs Men
Hair loss related to psychopharmaceuticals is overwhelmingly experienced by women, not men. In one literature review, nearly 89% of the reports came from women.[45]https://pubmed.ncbi.nlm.nih.gov/16783834/ Understanding why women seem to be more affected by this type of hair loss may offer clues into the mechanism of action.
Multidirectional Relationships
Some drugs have a multi-directional relationship to hair loss, for reasons that aren’t yet entirely understood. Valproate and valproic acid, for example, can lead to hair loss when orally administered but may lead to hair growth when applied topically. In two of the case studies mentioned above, alopecia areata improved with paroxetine treatment, a drug that has induced hair loss in others.
These multidirectional relationships may shed light on cause and effect, but also, offer insight into how complicated it is to point to any one drug as a cause of hair shedding.
Hair loss, particularly telogen effluvium, may be related to trauma, stress, anxiety, or depression. Some people may find their hair loss improves as their anxiety and depression get better. On the other hand, researchers acknowledge it’s possible that cases of hair loss are underreported, either due to self-neglect or because the patient has experienced it in the past as a result of emotional stress and does not relate the occurrence to the drug.
What Can Be Done?
In general, the incidence rate for these reports of hair loss associated with antidepressants, anti-anxiety medications and mood stabilizers, appears to be quite low. It’s not a given that hair loss is an outcome with any of these drugs.
That said, hair loss can be devastating for the one person that experiences it. And in some cases, may lead to non-compliance with what otherwise are very effective and life-improving drugs.
Before deciding to forego or discontinue treatment, it’s worth it to consider that in all the cases mentioned above, patients reported their hair shedding stopped after an adjustment to dosage, or once treatment ended.
For those who have recently begun taking a new medication and have noticed hair thinning or hair shedding, it would be advisable to speak with a doctor about switching to another medication. Remember, there were several antidepressants and anti-anxiety medications that did not make the list because there was no evidence linking them to hair loss.
That said, if a medication on this list lands in the medicine cabinet, the risk of hair loss remains very low.
Summary
It’s true there are case studies linking some psychopharmaceuticals to hair loss. But generally, the incidence rates are low, and hair loss tends to stop when dosages are lowered or the use of the drug is discontinued.
That said, for the individual experiencing hair loss, it’s of little comfort to know that risk is low. Maintaining healthy hair can be an important part of maintaining emotional and psychological health. If treating anxiety or depression is having a negative effect on hair, it may be time to talk to a doctor or dermatologist about an alternative treatment plan.
While happiness and health should always supersede hair growth, in this case, people shouldn’t have to sacrifice either.
References[+]
References ↑1 https://link.springer.com/article/10.2165/00002018-199410040-00005 ↑2, ↑3, ↑42 https://pubmed.ncbi.nlm.nih.gov/10798824/ ↑4 https://www.psychiatria-danubina.com/UserDocsImages/pdf/dnb_vol33_no2/dnb_vol33_no2_187.pdf ↑5 https://www.psychiatrist.com/pcc/depression/escitalopram-induced-hair-loss/ ↑6 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5035805/ ↑7 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3219523/ ↑8 https://pubmed.ncbi.nlm.nih.gov/34355374/ ↑9 https://pubmed.ncbi.nlm.nih.gov/31599441/ ↑10 https://www.cambridge.org/core/journals/the-british-journal-of-psychiatry/article/hair-loss-associated-with-fluoxetine/D2A55E09DDCF393399C14DB9289BAADE ↑11 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC514846/ ↑12 https://pubmed.ncbi.nlm.nih.gov/19630647/ ↑13 https://pubmed.ncbi.nlm.nih.gov/34181746/ ↑14 https://sci-hub.se/https://pubmed.ncbi.nlm.nih.gov/16922952/ ↑15 https://pubmed.ncbi.nlm.nih.gov/11737460/ ↑16 https://pubmed.ncbi.nlm.nih.gov/10883182/ ↑17 https://pubmed.ncbi.nlm.nih.gov/10442258/ ↑18 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4589582/ ↑19 https://pubmed.ncbi.nlm.nih.gov/18664165/ ↑20, ↑45 https://pubmed.ncbi.nlm.nih.gov/16783834/ ↑21 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3000200/ ↑22 https://pubmed.ncbi.nlm.nih.gov/28763345/ ↑23 https://sci-hub.se/https://pubmed.ncbi.nlm.nih.gov/12500769/ ↑24 https://pubmed.ncbi.nlm.nih.gov/17293712/ ↑25 https://pubmed.ncbi.nlm.nih.gov/19471188/ ↑26 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3579479/ ↑27 https://pubmed.ncbi.nlm.nih.gov/23525591/ ↑28 https://pubmed.ncbi.nlm.nih.gov/22265699/ ↑29 https://pubmed.ncbi.nlm.nih.gov/7995585/ ↑30 https://pubmed.ncbi.nlm.nih.gov/3126095/ ↑31 https://pubmed.ncbi.nlm.nih.gov/6519870/ ↑32 https://pubmed.ncbi.nlm.nih.gov/6838778/ ↑33 https://pubmed.ncbi.nlm.nih.gov/6404546/ ↑34 https://pubmed.ncbi.nlm.nih.gov/6809028/ ↑35 https://pubmed.ncbi.nlm.nih.gov/30193142/ ↑36 https://pubmed.ncbi.nlm.nih.gov/30386073/ ↑37 https://pubmed.ncbi.nlm.nih.gov/29061425/ ↑38 https://pubmed.ncbi.nlm.nih.gov/27889835/ ↑39 https://pubmed.ncbi.nlm.nih.gov/8899137/ ↑40 https://pubmed.ncbi.nlm.nih.gov/1486112/ ↑41 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7756276/ ↑43 https://pubmed.ncbi.nlm.nih.gov/16217127/ ↑44 https://pubmed.ncbi.nlm.nih.gov/3157663/ IngredientsPharmaceuticalsNatural RemediesCompany ReviewsBy Sarah King, PhDNov 8, 2024Do Chinese Herbs Help Regrow Hair? A Look At Traditional Remedies
Hair loss treatments have expanded beyond pharmaceuticals to include Traditional Chinese Medicine (TCM) herbs, which some enthusiasts claim help support hair growth. We reviewed the scientific evidence behind four well-known herbs: Polygonum multiflorum (Fo-Ti), Panax ginseng, Lycium chinense (Goji ...By Sarah King, PhDOct 11, 2024Cannabidiol (CBD) Increases Hair Counts By 246%? Not So Fast.
Cannabidiol (CBD), a non-psychoactive compound from Cannabis sativa, is being explored for hair regrowth due to its anti-inflammatory, antioxidant, and circulation-boosting properties. Small studies on CBD topicals for androgenic alopecia (AGA) show hair count increases between 55% and 246% over six...By Sarah King, PhDSep 27, 2024Creatine: Does It Worsen Hair Loss? It Depends On The Hair Loss Type. – 2025 Update
Creatine, a substance that supports energy production in the muscles and brain, is commonly used as a supplement to boost athletic performance. However, its potential link to hair loss has raised concerns, particularly due to its effect on dihydrotestosterone (DHT), a hormone associated with androge...By Sarah King, PhDSep 18, 2024Can Progesterone Improve Hair Regrowth?
Progesterone is a hormone that plays a vital role in female reproductive health. Deficiencies in progesterone have been linked to hair thinning, particularly in conditions like female pattern hair loss (FPHL) and postpartum telogen effluvium (PPTE). While natural progesterone seems to have 5-alpha-r...By Ben Fletcher, PhDSep 10, 2024CRABP2: Can This Gene Predict Regrowth From Retinoids?
The CRABP2 gene, which plays a role in retinoic acid metabolism, has been linked to hair health. Retinoic acid, a derivative of vitamin A, is known to influence hair growth, and some studies suggest CRABP2 may also be involved. However, the evidence connecting CRABP2 variants to hair loss and treatm...By Ben Fletcher, PhDSep 8, 2024BTD: Can This Gene Predict Regrowth From Biotin?
The BTD gene, responsible for encoding the enzyme biotinidase, is crucial for biotin metabolism. Biotin, or vitamin B7, plays a significant role in hair health, and biotinidase deficiency—caused by BTD polymorphisms—can lead to hair loss conditions like alopecia. While some studies suggest that biot...By Ben Fletcher, PhDSep 4, 2024COL1A1: Can This Gene Predict Regrowth From Collagen Support?
The COL1A1 gene, responsible for producing a key component of type 1 collagen, has been associated with hair growth and structure. Certain genetic variations in COL1A1 might affect the balance of collagen chains, potentially influencing hair loss and the response to treatments. However, the current ...By Sarah King, PhDSep 4, 20242dDR For Hair Loss: What Do We Know So Far About This Sugar?
2dDR Hair introduces hair care products featuring 2-deoxy-D-ribose (2dDR), a naturally occurring sugar, claiming it can support hair regrowth. While a recent mouse study shows favorable results, there is currently no clinical evidence in humans to support its effectiveness or safety. In fact, after ...By Ben Fletcher, PhDSep 4, 2024CYP19A1: Can This Gene Predict Regrowth From Hormone Therapy?
Can your genes predict the effectiveness of hormone therapy for hair loss? The CYP19A1 gene, which encodes the enzyme aromatase, has been linked to hair growth and hair loss, particularly in androgenetic alopecia (AGA). Some studies suggest that certain CYP19A1 variants might influence the effective...By Ben Fletcher, PhDAug 27, 2024ACE: Can This Gene Predict Regrowth From Treatments Targeting Blood Flow?
This article explores the potential link between angiotensin-converting enzyme (ACE) activity and hair loss, particularly in the context of androgenic alopecia (AGA) and alopecia areata (AA). While ACE plays a crucial role in blood pressure regulation and vasoconstriction, there is limited evidence ... - Mission Statement
 Scroll Down
Scroll Down