- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Musely Reviews for Hair Loss: Are They Trustworthy?
-
Best Minoxidil for Women: Top 6 Brands of 2026
-
Best Topical Finasteride: 5 Top Products of 2026
-
Best Topical Dutasteride: 8 Choices for 2026
-
10 Best Shampoos for Thinning Hair Men
-
Does Semaglutide Cause Hair Loss?
-
Does Dandruff Cause Hair Loss?
-
Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
ArticlesAMP-303 Hair Injections: How Strong is the Evidence?
First Published Apr 17 2026Last Updated Apr 17 2026Pharmaceutical Researched & Written By:Sarah King, PhD
Researched & Written By:Sarah King, PhD Reviewed By:Rob English, Medical Editor
Reviewed By:Rob English, Medical Editor
Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
AMP-303 is a novel injectable hair loss treatment developed by Amplifica Holdings Group, a company founded by the scientists behind the ‘hairy mole’ discovery that made headlines in 2023. With first-in-human trial results showing promising hair regrowth after a single treatment cycle, it has quickly become one of the most talked-about developments in the androgenic alopecia space. In this article, we break down the science behind the treatment, what the clinical data actually shows, and what you should realistically expect if and when AMP-303 reaches the market.
Full Article
Key Takeaways
- Branding: AMP-303 is a proprietary injectable treatment for androgenic alopecia (AGA), developed by Amplifica Holdings Group Inc.
- Unique Selling Point: Amplifica claims that AMP-303 works by delivering “unique biological signaling molecules” intradermally to reactivate dormant hair follicles and stimulate the conversion of vellus hairs into terminal hairs – a process known as hair terminalization.
- Clinical Support: One first-in-human, randomized, double-blind, placebo-controlled, multicenter Phase I trial has been completed, with results announced in September 2024 and presented at the Society for Investigative Dermatology Annual Meeting in May 2025. Male participants aged 18-45 with AGA were stratified by duration of hair loss (3-5 years vs. ≥10 years) and received AMP-303 on one side of the scalp and placebo on the other. A statistically significant percentage of participants showed >15% increase in non-vellus hair count at 60 days post-treatment (p<0.001) compared to placebo, with >10% increase still observed at 150 days post-treatment. Safety was described as favourable, with no severe adverse events.
- Concerns:
- Results for the long-standing hair loss group (Group 2) have not been separately disclosed, raising questions about whether AMP-303 is effective in this population.
- No raw data or clinical photographs have been published; all information currently derives from press releases and a single conference abstract. It is therefore impossible to assess the visible clinical impact of the treatment.
- There are no data beyond 150 days, so the durability of effect, need for repeat dosing, and long-term safety are all unknown.
- The precise active ingredient and mechanism of action have not been published in a peer-reviewed journal.
- Evidence Quality: AMP-303 scored 31/100 for evidence quality, by our metrics.
- Recommendations to Amplifica: We recommend that Amplifica publish the full Phase I trial dataset in a peer-reviewed journal, including subgroup data separated by hair loss duration, responder versus non-responder analyses, and objective mean hair count data for the full treated population. Phase II/III trials with adequate sample sizes, enrolment of both sexes, a wider age range, active comparator arms, and follow-up of at least 12 months are needed before the clinical place of AMP-303 can be properly assessed. Full disclosure of the mechanism of action would substantially increase confidence in the product.
What is AMP-303?
AMP-303 is an intradermal injectable treatment for androgenic alopecia (AGA), currently in first-in-human trials. It was developed by Amplifica Holdings Group Inc, a clinical-stage biopharmaceutical company co-founded by Dr Maksim Plikus (Chief Scientific Officer), Dr William Rassman (Chief Medical Officer), and Frank Fazio (President and CEO).[1]Amplifica, (no date). About Us. Available at: https://amplificabio.com/about-us/ (Accessed: April 2026) AMP-303 is described as a polysaccharide-based formulation designed to be delivered into the deep dermis via intradermal microinjection in the frontotemporal scalp.
Who is the Target Population?
While Amplifica’s first-in-human trial has only enrolled men aged 18-45, the company has stated that it expects AMP-303 to be effective in both men and women. Future research is expected to include women.
What Amplifica Products are Offered?
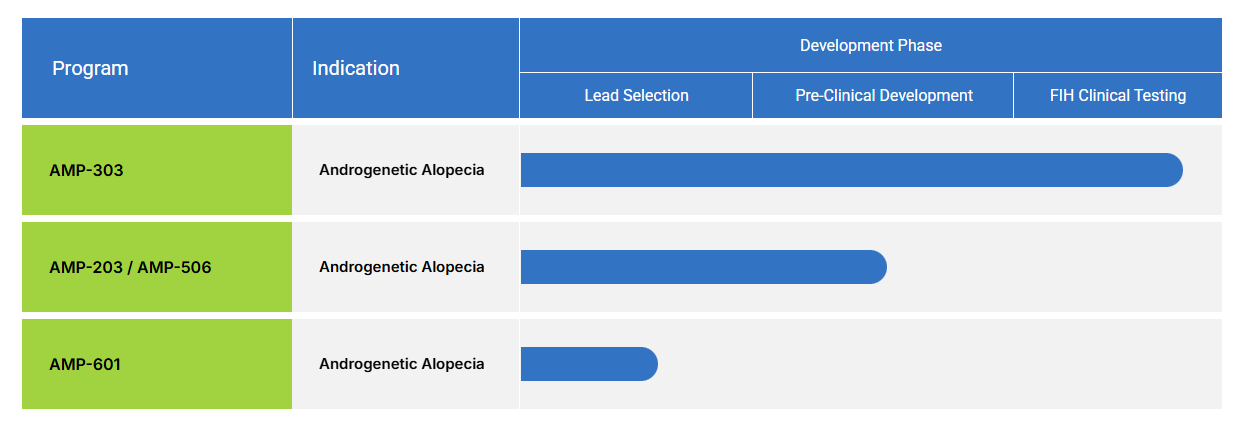
It should be noted that none of Amplifica’s products are currently available to use; however, Amplifica currently has three hair loss treatments in the pipeline.
Its lead candidate, currently in first-in-human trials, is AMP-303. Additional compounds include AMP-203/506, an osteopontin-based compound, and AMP-601, a SCUBE-3-based compound.[2]Amplifica. Pipeline. Developing novel injectable treatments to address androgenetic alopecia. Available at: https://amplificabio.com/pipeline/ (Accessed: April 2026) All of these treatments remain investigational at the time of writing.
How Does Amplifica Claim Its Product is Unique?
Amplifica has stated that AMP-303 works via a nature-inspired mechanism involving the delivery of isolated biological signaling molecules to reactivate dormant hair follicles. The company reported that AMP-303 can induce the conversion of vellus (thin, fine) hairs back into terminal (thicker, pigmented) hairs. This would represent a meaningful reversal of AGA pathology rather than just slowing its progression. This is a mechanistic distinction from FDA-approved treatments like finasteride and minoxidil, though, as mentioned before, the precise molecular target has not yet been described.
Product Science: Deep Dive
The Hairy Mole Observation and the Scientific Basis
The scientific foundation behind AMP-303 begins with an observation: skin moles (“melanocytic nevi”) in humans very commonly grow unusually thick, dense hair. The same phenomenon was replicated in mouse models. Rather than treating this as a curiosity, Dr Plikus and his team at UCI asked the question – what is the mole actually doing to the hair follicles around it?
A study published in Nature provided the answer. The cells that give moles their pigment – melanocytes – eventually stop dividing and enter a state called senescence, a kind of permanent cellular retirement. Senescent cells are not simply inactive, however; they release a cocktail of signaling proteins into the surrounding tissue. The Plikus laboratory found that it is precisely this secreted cocktail that supercharges hair growth in the skin above and around the mole.[3]Wang, X., Ramos, R., Phan, A, Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, … Continue reading
By systematically screening the proteins released by these senescent melanocytes, the researchers identified osteopontin (a protein encoded by the gene SPP1) as the key driver. They demonstrated this in several ways:
- Injecting osteopontin alone was sufficient to trigger robust hair growth in mice.
- Genetically engineering mice to overproduce it had the same effect
- Blocking either osteopontin itself or the receptor on hair stem cells that it binds to (a molecule called CD44) prevented the mole from inducing excess hair growth
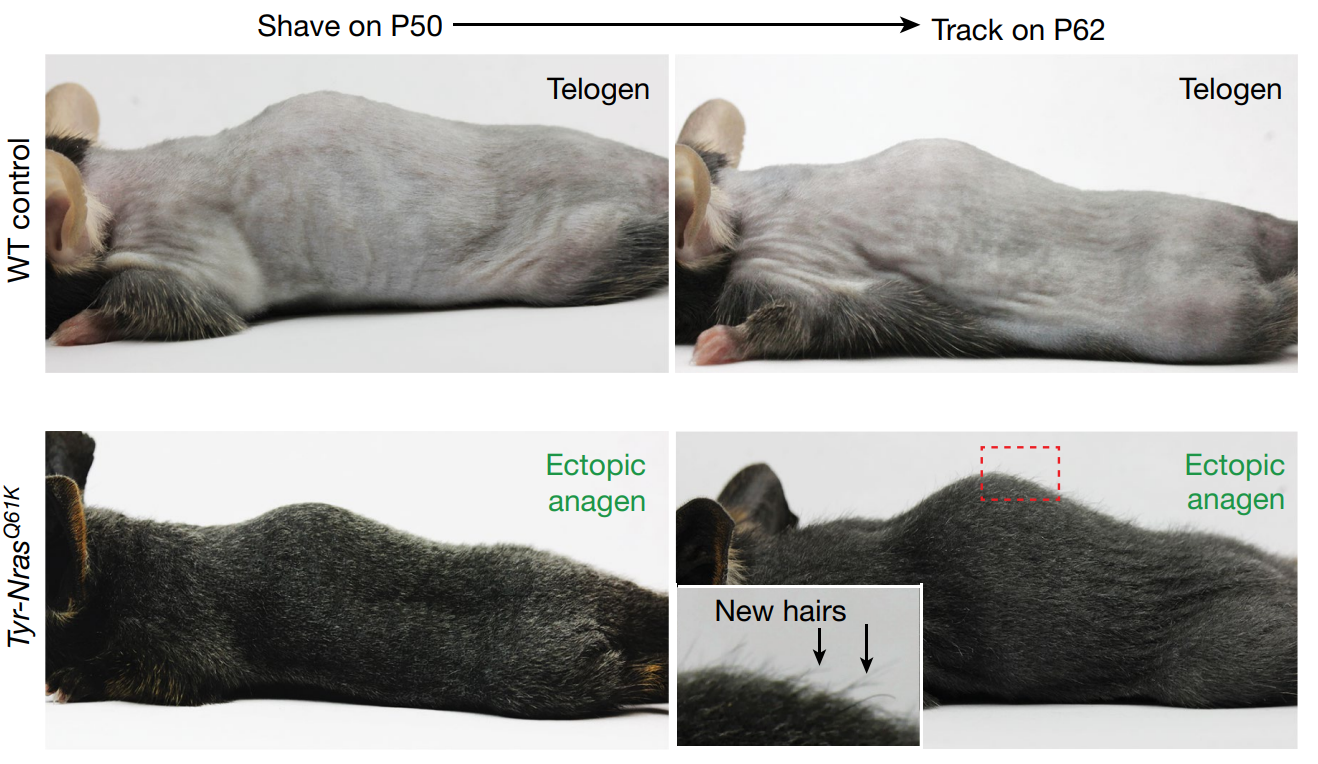
Figure 1: Effect of hair growth hyperactivation in mice.[4]Wang, X., Ramos, R., Phan, A,Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, … Continue reading Figure used in line with Creative Commons.
Importantly, osteopontin also stimulated new hair follicle growth in isolated human hair tissue tested in the laboratory, suggesting a pathway relevant to humans.
In short, the hairy mole effect is not accidental. It is driven by a specific molecular signal that tells dormant hair follicles to start growing.
It should be noted, however, that the precise active ingredient in AMP-303 has not been publicly disclosed by Amplifica. Within the company’s pipeline, osteopontin is designated as a separate compound, AMP-203, which has not yet entered clinical trials. It is not yet confirmed whether osteopontin, a related molecule from the same biological pathway, or an entirely distinct compound is responsible for the effects observed in the first-in-human trial.
What is the Regulatory Status of AMP-303?
AMP-303 is being developed through the conventional pharmaceutical regulatory pathway, with Amplifica pursuing regulatory approval. The first-in-human trial that was conducted in 2023-2024 was a Phase I safety and tolerability study. No FDA approval or IND clearance status for subsequent phases has been publicly disclosed. AMP-303 is therefore not available to people outside of clinical trials at this time.
Clinical Data
As previously mentioned, a first-in-human trial was conducted over 2023-2024 for AMP-303.[5]Green, J.B., Joseph, J.H., DuBois, J.C., Rassman, W.R., Fazio, F., Plikus, M.V., Ahmad, W. (2025). LB1139 AMP-303 injectable treatment for androgenetic alopecia: A multicenter, randomized, … Continue reading This was a randomized, double-blind, placebo-controlled, multicenter study conducted in the United States. Participants were male, aged 18-45, and diagnosed with AGA.
The participants were split into two groups:
- Group 1 (n=32): recent onset hair loss (3-5 years duration)
- Group 2 (n=29): long-standing hair loss (≥10 years duration)
Each participant received AMP-303 injected into 20 frontotemporal locations on one side of the scalp and saline placebo injections on the other side. Assessments were conducted at 60 and 150 days post-treatment.
Results:
- At 60 days post-treatment, a statistically significant proportion of participants showed >15% increase in non-vellus hair count compared to the placebo (p<0.001).
- At 150 days post-treatment, >10% experienced an increase in non-vellus hair count vs. just maintenance in the placebo.
- For Group 1 (recent onset) responders, the mean increase in non-vellus hair count was reported as 14.5%, peaking at 60 days.
- AMP-303 demonstrated the ability to promote the conversion of vellus to non-vellus hair.
- Results were observed in the frontotemporal region, which has historically been a challenging area for demonstrating treatment response.
While the results seem promising, we do have some concerns.
Missing data for the long-standing hair loss group. Group 2 (≥10 years duration) results have not been reported in public communications. This is a meaningful omission because a large number of hair loss patients have been battling it for long periods of time, and the lack of disclosure could mean that AMP-303 did not confer a benefit to this group.
No data beyond 150 days. While this concern is less important as it is a first-in-human trial, we currently do not know if gains persist longer than 150 days, if patients require repeat dosing, or if hair gains start to regress. Furthermore, we don’t know what the long-term safety is.
No published raw data/images: We are only able to keep up with updates about AMP-303 through press releases (and one published abstract); we have no idea what the raw data looks like, and whether the results have led to visible clinical effects.
Safety Profile
AMP-303 was described as safe and well-tolerated in the above trial. The majority of adverse events were mild in severity, and no severe adverse events were reported. Local skin reactions to intradermal injections were similar between the AMP-303 and placebo groups. Long-term safety is unknown.
The most common treatment-related adverse effects were:
- Mild headache (7.8%)
- Mild skin swelling (placebo 3.1%; AMP-303 1.6%)
Should I Use AMP-303?
While AMP-303 is not yet available to the public, once it is available, you might want to follow the treatment if:
- You have experienced hair loss for 5 years or less.
- Open to an injectable treatment.
- Interested in non-hormonal approaches to hair loss.
- Interested in early adoption of hair loss treatments.
This product could take some time (we are talking years) to come to the market, so we wouldn’t recommend you drop your current treatments just yet!
Recommendations to Amplifica
We recommend that Amplifica publish the full Phase I trial dataset in a peer-reviewed journal, including subgroup data separated by hair loss duration, responder vs. non-responder analyses, and objective mean hair count data for the full treated population. Phase II/III trials with adequate sample sizes, enrolment of both sexes, a wider age range, active comparator arms, and follow-up periods of at least 12 months are needed before the clinical efficacy of AMP-303 can be properly assessed. Furthermore, full disclosure of the mechanism of action of the treatment would substantially increase our confidence in the product.
Final Thoughts
AMP-303 represents a genuinely novel biological approach to AGA with its roots in rigorous academic research at UCI on the molecular basis of follicular activation. The first-in-human trial results are encouraging, particularly the frontotemporal efficacy and evidence of terminalization, and the non-hormonal mechanism is potentially advantageous for those who cannot or do not wish to take finasteride or dutasteride. However, AMP-303 remains an investigational product with a single small-scale trial behind it and no peer-reviewed publication.
We have seen many treatments start with a lot of excitement that ultimately fail to translate into meaningful clinical benefit. We recommend cautious optimism and waiting for more data to be published before making a decision.
References[+]
References ↑1 Amplifica, (no date). About Us. Available at: https://amplificabio.com/about-us/ (Accessed: April 2026) ↑2 Amplifica. Pipeline. Developing novel injectable treatments to address androgenetic alopecia. Available at: https://amplificabio.com/pipeline/ (Accessed: April 2026) ↑3 Wang, X., Ramos, R., Phan, A, Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, K.N., Phung, J.L., Ingal, J.P., Levitt, K.M., Cao, X., Liu, Y., Deng, Z., Taguchi, N., Scarfone, V.M., Wang, G., Paolilli, K.N., Wang, X., Guerrero-Juarez, C.F., Davis, R.T., Greenberd, E.N., Ruiz-Vega, R., Vasudeva, P., Murad, R., Widyastuti, L.H.P., Lee, H-L., McElwee, K.J., Gadeau, A-P., Lawson, D.A., Andersen, B., Mortazavi, A., Yu, Z., Nie, Q., Kunisada, T., Karin, M., Tuckermann, J., Esko, J.D., Ganesan, A.K., Li, J., Plikus, M.V. (2023). Signalling by senescent melanocytes hyperactivates hair growth. Nature. 618. 808-817. Available at: https://doi.org/10.1038/s41586-023-06172-8 ↑4 Wang, X., Ramos, R., Phan, A,Q., Yamaga, L., Flesher, J.L., Jiang, S., Oh, J.W., Jin, S., Jahid, S., Kuan, C-H., Nguyen, T.K., Liang, H.Y., Shettigar, N, U., Hou, R., Tran, K.H., Nguyen, A., Vu, K.N., Phung, J.L., Ingal, J.P., Levitt, K.M., Cao, X., Liu, Y., Deng, Z., Taguchi, N., Scarfone, V.M., Wang, G., Paolilli, K.N., Wang, X., Guerrero-Juarez, C.F., Davis, R.T., Greenberd, E.N., Ruiz-Vega, R., Vasudeva, P., Murad, R., Widyastuti, L.H.P., Lee, H-L., McElwee, K.J., Gadeau, A-P., Lawson, D.A., Andersen, B., Mortazavi, A., Yu, Z., Nie, Q., Kunisada, T., Karin, M., Tuckermann, J., Esko, J.D., Ganesan, A.K., Li, J., Plikus, M.V. (2023). Signalling by senescent melanocytes hyperactivates hair growth. Nature. 618. 808-817. Available at: https://doi.org/10.1038/s41586-023-06172-8 ↑5 Green, J.B., Joseph, J.H., DuBois, J.C., Rassman, W.R., Fazio, F., Plikus, M.V., Ahmad, W. (2025). LB1139 AMP-303 injectable treatment for androgenetic alopecia: A multicenter, randomized, placebo-controlled feasibility study of a novel polysaccharide (Abstract). Journal of Investigative Dermatology. 145(8), S198. Available at: www.https://doi.org/ 10.1016/j.jid.2025.06.1428 Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn More
Sarah King, PhD
Dr. Sarah King is a researcher & writer who holds a BSc in Medical Biology, an MSc in Forensic Biology, and a Ph.D. in Molecular and Cellular Biology. While at university, Dr. King’s research focused on cellular aging and senescence through NAD-dependent signaling – along with research into prostaglandins and their role in hair loss. She is a co-author on several upcoming manuscripts with the Perfect Hair Health team.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down