- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Musely Reviews for Hair Loss: Are They Trustworthy?
-
Best Minoxidil for Women: Top 6 Brands of 2026
-
Best Topical Finasteride: 5 Top Products of 2026
-
Best Topical Dutasteride: 8 Choices for 2026
-
10 Best Shampoos for Thinning Hair Men
-
Does Semaglutide Cause Hair Loss?
-
Does Dandruff Cause Hair Loss?
-
Topical Finasteride: The Best Dosage for Maximizing Regrowth and Minimizing Side Effects
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
ArticlesA New Way to Regrow Hair? Inside the Science of PP-405
First Published Apr 17 2026Last Updated Apr 17 2026Pharmaceutical Researched & Written By:Dr. Michael Williams
Researched & Written By:Dr. Michael Williams Reviewed By:Rob English, Medical Editor
Reviewed By:Rob English, Medical Editor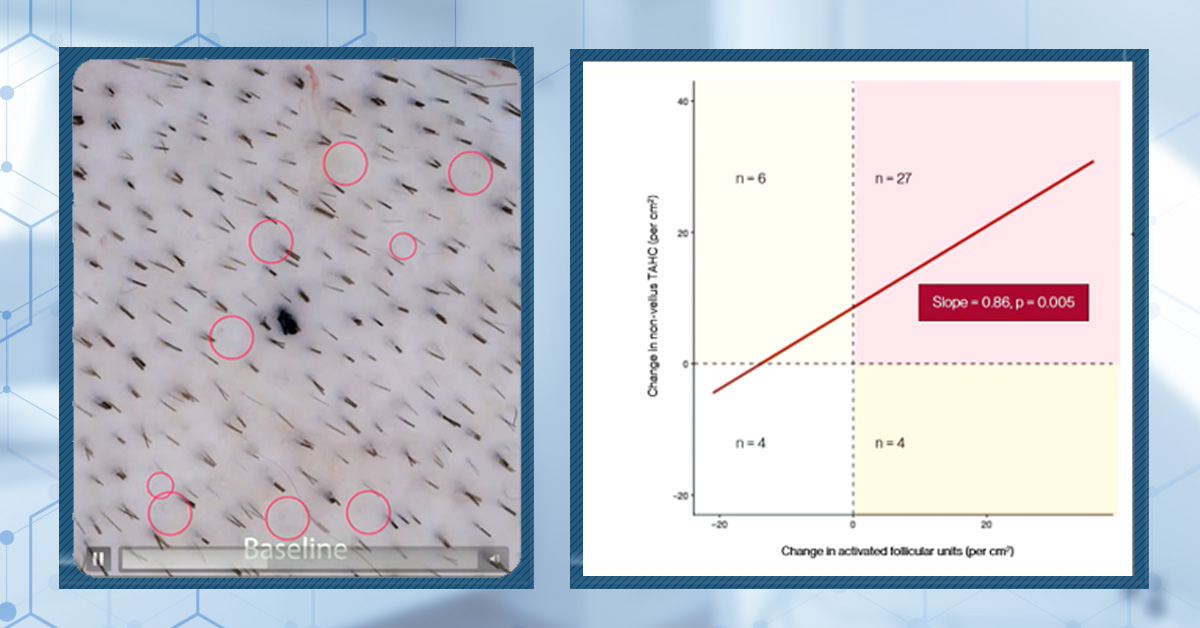
Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
PP-405 is a novel topical hair loss treatment developed by Pelage Pharmaceuticals. By targeting the hair follicle stem cell metabolism, the drug aims to “wake up” dormant hair follicle stem cells and drive new hair growth. Early clinical trial results have generated significant excitement, but with data yet to be peer-reviewed, questions remain about its true efficacy, mechanism, and safety. In this article, we break down the underlying science, examine the available clinical data, and explore what can realistically be expected from PP-405 as it progresses through development.
Full Article
Key Takeaways
- Branding: PP-405 is positioned as a next-generation regenerative hair loss therapy, focusing on reactivating dormant hair follicles rather than simply slowing hair loss.
- Unique Selling Point: Targets hair follicle metabolism, aiming to activate dormant hair follicle stem cells and potentially regenerate new hair growth.
- Clinical Support: Early Phase 1/2a data (n=78) suggest some efficacy, with ~31% of treated participants achieving >20% increases in hair density vs 0% in placebo. However, results are based on company-released data and conference presentations, not peer-reviewed publications.
- Concerns: There is currently a lack of peer-reviewed clinical data, and the proposed mechanism, reactivating dormant hair follicles, has not been convincingly demonstrated. Notably, the placebo group also showed signs of “new growth,” which is likely explained by natural hair cycling (kenogen) rather than a true treatment effect.
- Evidence Quality: scored 10/100 for evidence quality, by our metrics.
- Recommendations to Pelage: Pelage should publish full clinical trial data in peer-reviewed journals and provide clear mechanistic evidence, such as biopsies confirming stem cell activation. The company should also conduct larger, longer-term randomized controlled trials using validated endpoints, and release detailed safety data from all phases of testing.
What is PP-405?
PP-405 is a drug that inhibits the mitochondrial pyruvate carrier (MPC). MPC is an important regulator of metabolism, and it has been suggested that inhibiting MPC can ‘wake up’ hair follicle stem cells from a dormant state and force them into an active state. This may lead to hair growth in areas where it had previously stopped[1]Flores, A., Schell, J., Krall, A. S., Jelinek, D., Miranda, M., Grigorian, M., Braas, D., et al. (2017). Lactate dehydrogenase activity drives hair follicle stem cell activation. *Nature Cell … Continue reading The drug is being developed by a company called Pelage Pharmaceuticals, founded by researchers from the University of California, Los Angeles (UCLA) who originally discovered the importance of MPC in hair follicles.
PP-405 is currently undergoing clinical trials. Phase 1/2a clinical trials have been completed, and Pelage has released some promising results.[2]ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: … Continue reading,[3]Pelage Pharmaceuticals. (2026). Pelage Pharmaceuticals’ PP405 and its impact on follicular regeneration to be presented at the American Academy of Dermatology (AAD) Annual Meeting 2026. *Pelage … Continue reading However, these are yet to be published in a peer-reviewed scientific journal. Phase 3 clinical trials are believed to be currently ongoing.
Who is the Target Population?
Clinical trials to assess PP-405 have included men and women with androgenic alopecia. The company’s website highlights multiple target conditions: androgenic alopecia, telogen effluvium, and drug-induced alopecia.[4]Pelage Pharmaceuticals. (n.d.). Regenerative medicine treatments for hair loss. *Pelage Pharma.* Available at: https://pelagepharma.com/
What PP-405 Products are on Offer?
PP-405 is currently undergoing clinical trials, and is therefore not approved for sale. However, given the interest and excitement generated by early experiments, some companies are selling similar molecules, or their own compounded versions of PP-405. Given that this drug has not been approved, and there is very limited safety data available, it is not recommended to buy any of these products.
How is PP-405 Unique?
PP-405 is the only drug that targets metabolism in hair follicle stem cells to promote hair growth that is currently undergoing clinical trials. Pelage Pharmaceutical claims that the drug could go beyond slowing hair loss and “directly drive hair follicle regeneration”.[5]Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: … Continue reading
Product Science: Deep Dive
PP405 was discovered through research investigating important cells in the hair follicle known as stem cells.
Stem cells act like factories that can produce different types of cells in the body. In the hair follicle, the production of new hairs depends on the activity of stem cells, which are activated at the start of the growth phase of the hair cycle. Hair follicle stem cells (HFSCs) can give rise to all the different types of cells in the hair follicle and are essential for the growth of new hair.[6]Hsu, Y. C., Pasolli, H. A., Fuchs, E., et al. (2011). Dynamics between stem cells, niche, and progeny in the hair follicle. *Cell.* 144. 92–105. Available at: … Continue reading
Because the hair follicle produces hairs in cycles, HFSCs go through periods of activation and a dormant period known as quiescence. Researchers at UCLA were interested in how the metabolic activity of HFSCs changes when they switch from a quiescent state to an active one.
When HFSCs are quiescent, they show relatively low metabolic activity. The researchers found that HFSCs generate energy through a process called glycolysis more than most other cells do. Glycolysis is a form of anaerobic respiration, meaning it doesn’t use oxygen. You’re most likely to experience anaerobic respiration when exercising: if your body can’t get enough oxygen to your muscles fast enough, they revert to glycolysis to produce energy.
HFSCs, however, seem to use glycolysis at a high rate even when there is plenty of oxygen available. The researchers also found that activating metabolic pathways involved in anaerobic respiration is essential for activating HFSCs. One enzyme involved in these pathways is called lactate dehydrogenase.[7]Flores, A., Schell, J., Krall, A. S., Jelinek, D., Miranda, M., Grigorian, M., Braas, D., et al. (2017). Lactate dehydrogenase activity drives hair follicle stem cell activation. *Nature Cell … Continue reading
Using mouse models, the researchers showed that stopping lactate dehydrogenase from working can prevent HFSC activation, meaning they stay in a quiescent state. Conversely, forcing HSFCs into anaerobic respiration can promote hair growth. A molecule called the mitochondrial pyruvate carrier (MPC) is responsible for “deciding” whether cells prefer aerobic (with oxygen) or anaerobic respiration (without oxygen). If you block MPC, then you can guide HFSCs towards anaerobic respiration, causing them to ‘wake up’ from their dormant state.
The researchers identified a number of molecules that are capable of blocking MPC.[8]Liu, X., Flores, A. A., Situ, L., Gu, W., Ding, H., Christofk, H. R., Lowry, W. E., & Jung, M. E. (2021). Development of novel mitochondrial pyruvate carrier inhibitors to treat hair loss. … Continue reading Some of these UCLA researchers, led by William Lowry, went on to start Pelage Pharmaceuticals, and one of the MPC inhibitor molecules they later produced was PP-405.
It seems they were confident that activating hair growth by blocking MPC in HFSCs would translate well into a treatment for hair loss in humans.
Clinical Data
Following this promising experimental data and the founding of Pelage Pharmaceuticals, clinical trials began enrolling in 2024.[9]ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: … Continue reading Clinical trials registration information states that phase 1/2a trials were completed in July 2025, and the next day, Pelage announced “positive results”.[10]Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: … Continue reading
Their press release provides some details of the study. 78 men and women with androgenic alopecia were enrolled and randomized into either the treatment group or placebo group. The treatment group applied PP-405 to their scalp once daily for 4 weeks, with follow-up at 12 weeks.
While this phase of clinical trials is intended to test the safety of a treatment and generally operates as a ‘proof-of-concept’ study, Pelage did release some efficacy information. They state that 4 weeks after treatment was completed, 31% of participants in the treatment group exhibited a greater than 20% increase in hair density, compared to 0% of patients in the placebo group.
Unfortunately, these results have yet to be published in a peer-reviewed journal, which would allow us to better scrutinize their data. However, the company did present some results from the Phase 2a clinical trial at the American Academy of Dermatology (AAD) Annual Meeting in March 2026.
As of April 2026, the presentation is available on Vimeo (starting at around 1:21).[11]Vimeo. (2026). Pelage PP405 presentation at American Academy of Dermatology (AAD) Annual Meeting 2026. *Vimeo.* Available at: https://vimeo.com/1177843989/a90148385b
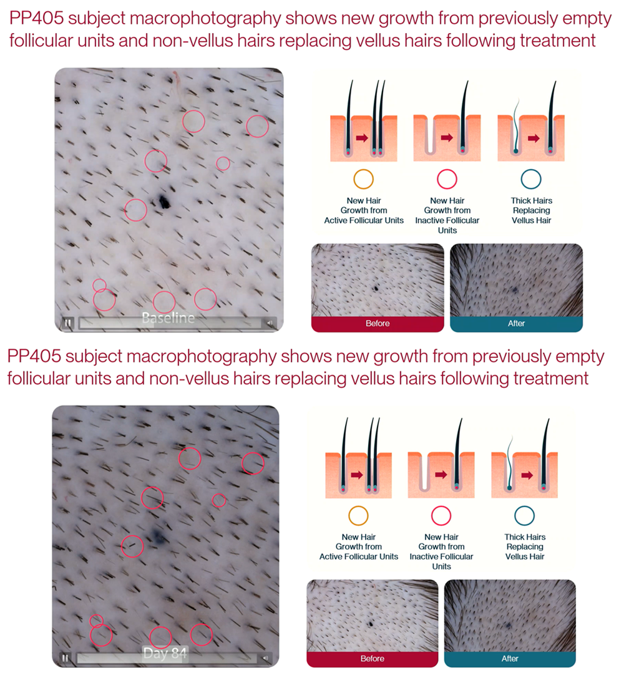
In their presentation, Pelage highlights the importance of follicular units: bundles of 1 to 4 hair follicles, along with associated glands, nerves, and muscles, that are the tissue structure used in hair transplantation.
You can see an example of the images presented in Figure 1. The researchers took pictures of patients’ scalps prior to treatment (using micro tattooing to confirm they were observing the same area), and again at week 12. The claim to ‘track’ the follicular units in the images over time, and suggest that there is new growth visible where previously there was none. They also claim that some follicular units that were producing vellus hairs are now producing terminal (visible) hairs, and some follicular units are producing multiple hairs where previously they were producing only one.
Figure 1. Images taken from Pelage Pharmaceuticals’ presentation at the AAD Annual Meeting in March 2026. They claim red circles highlight new growth after treatment (bottom), where previously hair follicles were dormant (top).
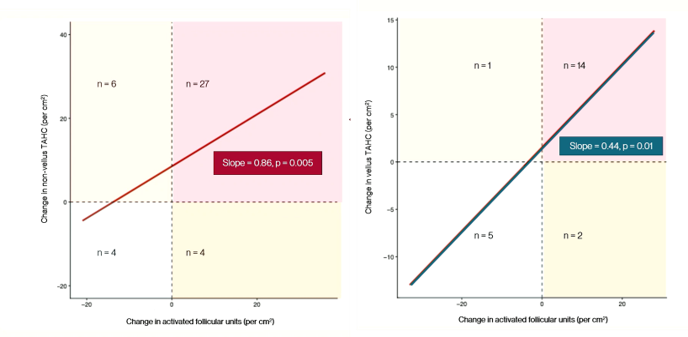
Pelage also presented some summary data from the trial, which you can see in Figure 2. They suggest that new growth in the treated group represents terminal (non-vellus) hairs, which will contribute to the appearance of hair, while new growth in the placebo group represents vellus hairs, which are not visible to the naked eye.
Figure 2. Summary data taken from Pelage Pharmaceuticals’ presentation at the AAD Annual Meeting in March 2026. Change in “activated follicular units” was related to an increase in non-vellus hairs in PP-405-treated patients (left), while it was related to an increase in vellus hairs in placebo patients (right).
They support this claim by showing a relationship between “activated follicular units” and the number of non-vellus hairs in the treatment group, while there is a relationship between “activated follicular units” and vellus hairs in the placebo group. I.e., while there is new growth in the placebo group, this is less likely to contribute to the appearance of hair.
Problems with the Data
While these data represent only a snapshot of the trial results, they could be very promising. Unfortunately, the data presented thus far don’t back up the claims made by Pelage.
In their presentation, the researchers consistently refer to “activated follicular units”. Based on the mechanism of action that they identified in laboratory models, they claim that these represent dormant follicles that have been reawakened via activation of their stem cells.
However, hair follicles go through natural cycles of shedding and regrowth. Hair follicles can spend long periods of time “empty” following a shedding event, where they are stuck in kenogen, the “resting” stage of the follicular cycle, where no fiber is produced. In fact, in a healthy scalp, about 10% of hair follicles may be in this state at any given time, which typically lasts about 2 months.[12]Guarrera, M., & Rebora, A. (2019). The higher number and longer duration of kenogen hairs are the main cause of the hair rarefaction in androgenetic alopecia. *Skin Appendage Disorders.* 5(3). … Continue reading
As such, there is no evidence to demonstrate that apparent new growth in PP-405 treated patients is not simply hairs exiting kenogen.
This is demonstrated by Pelage’s own data, where new growth from apparently empty areas is visible in participants who received placebo. If we look more closely at the x axes of the summary data, we can see that while ~76% (31 out of 41) patients in the treated group displayed a positive change in activated follicular units, ~73% (16 out of 21) in the placebo group also showed a positive change.
This makes the claim that PP-405 is activating dormant follicles less robust.
The data does still seem to suggest an increase in terminal hairs, which may translate to more visible hair. When Pelage publishes its data in full, and we see further evidence from larger trials, it will be possible to begin assessing the drug’s efficacy.
Safety Data
Pelage has not released the safety data from their early phase clinical trials, though they do claim that there are “no concerns” regarding safety based on these studies.[13]Vimeo. (2026). Pelage PP405 presentation at American Academy of Dermatology (AAD) Annual Meeting 2026. *Vimeo.* Available at: https://vimeo.com/1177843989/a90148385b It is important to wait until safety data from larger studies is released before making any firm decisions about the safety of the drug.
What is the Regulatory Status of PP-405?
PP-405 is still undergoing clinical trials and has not yet been approved in any jurisdiction. Phase 3 clinical trials are expected to finish in 2026, and the drug won’t be available until 2027/2028 at the earliest.
Should I Use PP-405?
It is not currently possible to assess the safety or efficacy of PP-405, or any similar MPC inhibitors. While some non-FDA-approved treatments might be available online, these represent untested and potentially unsafe treatments.
Recommendations to Pelage
The small snapshots of data we have seen from PP-405 could be promising. However, information is currently limited, and there are some steps we’d like to see Pelage take:
- Publish all safety and efficacy data from clinical trials in a peer-reviewed journal.
- Perform experiments to test the hypothesis that PP-405 is activating dormant hair follicles. This could include biopsies or other techniques to explore what is happening at the follicular level. Currently available data showing new hair growth do not demonstrate their proposed mechanism of action.
- Perform larger, randomized, placebo-controlled trials with validated endpoints to explore whether PP-405 increases hair growth over a long period of time.
Final Thoughts
The laboratory experiments that led to the development of PP-405 are robust and genuinely groundbreaking. The researchers highlighted a previously unexplored aspect of hair follicle physiology and identified a potential route to regrow hair.
There are also some promising results in terms of increases in non-vellus hairs. However, it is not currently possible to independently assess these data, as they have not been published in a peer-reviewed journal. Furthermore, the data we have seen so far do not sufficiently support Pelage’s claims regarding the mechanism of action of PP-405.
Still, PP-405 remains “one to watch”, and we look forward to more complete data being released in the near future.
References[+]
References ↑1, ↑7 Flores, A., Schell, J., Krall, A. S., Jelinek, D., Miranda, M., Grigorian, M., Braas, D., et al. (2017). Lactate dehydrogenase activity drives hair follicle stem cell activation. *Nature Cell Biology.* 19(9). 1017–1026. Available at: https://doi.org/10.1038/ncb3575 ↑2 ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: https://clinicaltrials.gov/study/NCT06393452 ↑3 Pelage Pharmaceuticals. (2026). Pelage Pharmaceuticals’ PP405 and its impact on follicular regeneration to be presented at the American Academy of Dermatology (AAD) Annual Meeting 2026. *Pelage Pharma.* Available at: https://pelagepharma.com/press-releases/pelage-pp405-aad-2026/ ↑4 Pelage Pharmaceuticals. (n.d.). Regenerative medicine treatments for hair loss. *Pelage Pharma.* Available at: https://pelagepharma.com/ ↑5 Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: https://www.businesswire.com/news/home/20250617338859/en/Pelage-Pharmaceuticals-Announces-Positive-Phase-2a-Clinical-Trial-Results-for-PP405-in-Regenerative-Hair-Loss-Therapy ↑6 Hsu, Y. C., Pasolli, H. A., Fuchs, E., et al. (2011). Dynamics between stem cells, niche, and progeny in the hair follicle. *Cell.* 144. 92–105. Available at: https://doi.org/10.1016/j.cell.2010.11.049 ↑8 Liu, X., Flores, A. A., Situ, L., Gu, W., Ding, H., Christofk, H. R., Lowry, W. E., & Jung, M. E. (2021). Development of novel mitochondrial pyruvate carrier inhibitors to treat hair loss. *Journal of Medicinal Chemistry.* 64(4). 2046–2063. Available at: https://doi.org/10.1021/acs.jmedchem.0c01570 ↑9 ClinicalTrials.gov. (n.d.). Safety, pharmacokinetics and efficacy of PP405 in adults with androgenetic alopecia (NCT06393452). *ClinicalTrials.gov.* Available at: https://clinicaltrials.gov/study/NCT06393452 ↑10 Business Wire. (2025). Pelage Pharmaceuticals announces positive Phase 2a clinical trial results for PP405 in regenerative hair loss therapy. *Business Wire.* Available at: https://www.businesswire.com/news/home/20250617338859/en/Pelage-Pharmaceuticals-Announces-Positive-Phase-2a-Clinical-Trial-Results-for-PP405-in-Regenerative-Hair-Loss-Therapy ↑11, ↑13 Vimeo. (2026). Pelage PP405 presentation at American Academy of Dermatology (AAD) Annual Meeting 2026. *Vimeo.* Available at: https://vimeo.com/1177843989/a90148385b ↑12 Guarrera, M., & Rebora, A. (2019). The higher number and longer duration of kenogen hairs are the main cause of the hair rarefaction in androgenetic alopecia. *Skin Appendage Disorders.* 5(3). 152–154. Available at: https://doi.org/10.1159/000493587 Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn More
Dr. Michael Williams
Michael is a researcher and writer who holds a BSc in Bioscience, an MSc in Regenerative Medicine, and a PhD in Translational Biomedicine. He undertook his PhD research at Houston Methodist Research Institute, Texas, focusing on cell signaling in the ovarian cancer tumor microenvironment. He conducted postdoctoral research at Barts Cancer Institute in London, exploring cellular metabolism in acute myeloid leukemia. He has published work in a range of fields, including oncology, nanomedicine, and cell-based therapeutics.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down