- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
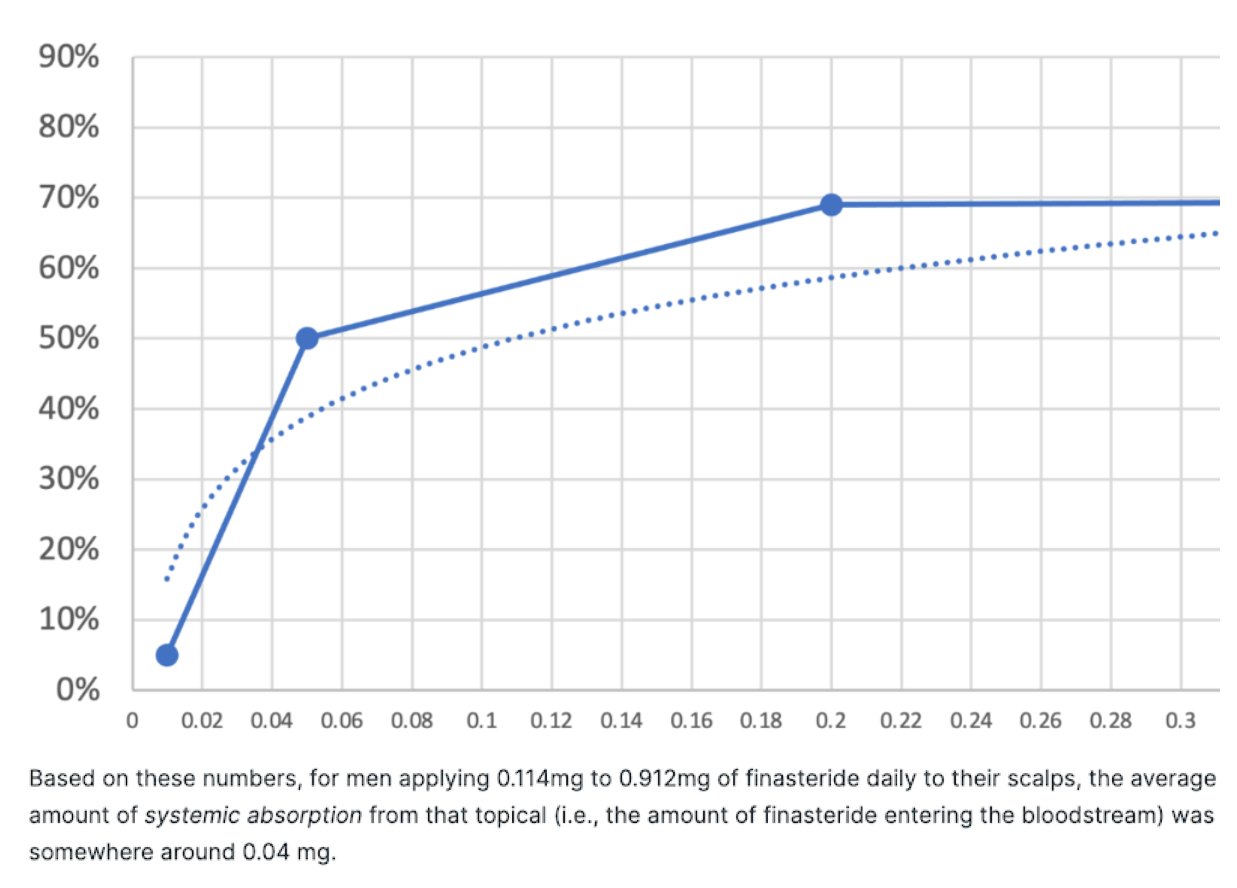
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Does Anavar Cause Hair Loss?
-
10 Best Shampoos for Hair Loss in 2026
-
XYON Review: Do Their Products Actually Regrow Hair?
-
Minoxidil Before and After Photos [2026] | Does It Work?
-
How to Get Finasteride: Is It Over the Counter?
-
Keeps Review: The Truth About Their Hair Loss Treatments?
-
7 Best Oils for Hair Growth
-
Hims Hair Growth Reviews: The Pros, Cons, and Real Results
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
Articles
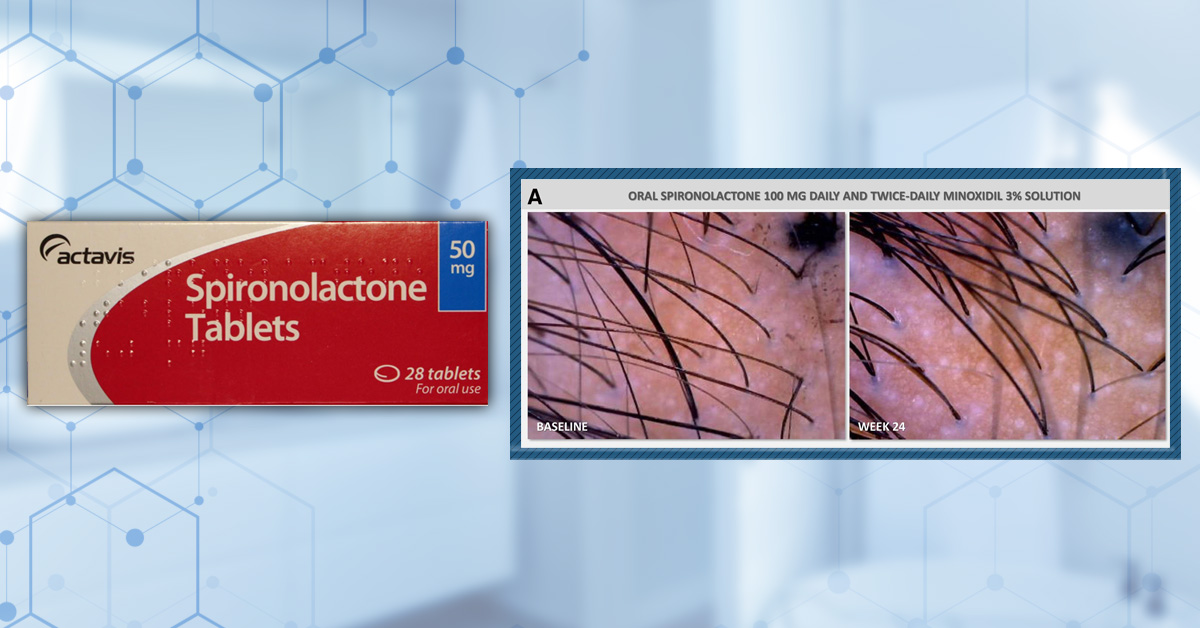
Minoxidil is a medication that began as a treatment for hypertension (high blood pressure). However, after observing excess hair growth during the testing stages of the drug, minoxidil was repurposed as a treatment for hair loss. Topical minoxidil soon received FDA approval for the treatment of both male and female pattern hair loss, while oral minoxidil is also used as an off-label treatment for hair loss.[1]Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug design, development and therapy. 2777-2786. Available at: … Continue reading
So, if minoxidil is a treatment for hair loss, why does it cause an increase in hair loss when you start using it? Yes, you did read that right – minoxidil can actually cause an increase in hair loss as part of what is often called the ‘dread shed.’ This phenomenon was demonstrated in a retrospective study of 435 patients with androgenic alopecia (AGA) who were prescribed low-dose oral minoxidil [≤5 mg per day] by the same clinic. Self-reported adverse events were recorded for each of the users and, of the 435 patients, 32% experienced increased hair shedding.[2]Sanabria, B., de Nardo Vanzela, T., Miot, H. A., & Ramos, P. M. (2021). Adverse effects of low-dose oral minoxidil for androgenetic alopecia in 435 patients. Journal of the American Academy of … Continue reading
This is evidently cause for concern – if you have just started minoxidil treatment to prevent hair loss, a sudden increase in hair loss is possibly the last thing you would hope and expect to experience. So what is it about minoxidil that causes this to happen, how long does it usually last, and is it actually a good thing? In this article, we will explore the hair cycle and minoxidil in detail to provide answers to each of these questions.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

The Hair Cycle
To understand why minoxidil can increase hair shedding, let’s first take a refresher on the hair cycle. Our hair is constantly going through a cycle of growing (anagen), regression and transition (catagen), resting (telogen), and shedding (exogen), which it repeats continuously.
Healthy hairs grow for anywhere between 2 and 8 years, and there is a correlation between the length and strength of a hair and the time spent in anagen. Catagen is the transition from anagen to telogen, a period of approximately 2 weeks during which the follicle regresses from the hair shaft and disconnects it from the blood supply, preventing any further growth. Telogen follows and lasts for 2-3 months, with a new hair shaft beginning to develop at the base of the follicle underneath the now resting hair shaft. Exogen then represents the transition from telogen to anagen, with the growing hair shaft pushing out the old hair shaft.[3]Natarelli, N., Gahoonia, N., & Sivamani, R. K. (2023). Integrative and mechanistic approach to the hair growth cycle and hair loss. Journal of clinical medicine. 12(3). 893. Available at: … Continue reading
In the healthy scalp, the percentage of hairs in each of the hair cycle stages is thought to remain fairly consistent. At any one time, evidence suggests that approximately 9% of the hairs on a healthy scalp are in the telogen phase (although there are some suggestions that this figure may actually be too high).[4]Natarelli, N., Gahoonia, N., & Sivamani, R. K. (2023). Integrative and mechanistic approach to the hair growth cycle and hair loss. Journal of clinical medicine. 12(3). 893. Available at: … Continue reading These hairs exist in a largely asynchronous fashion, with hair follicles progressing through the hair cycle according to their own unique pattern. Hair follicles undergo between 10 and 30 full cycles, and it is normal for up to 150 hairs to fall out per day without there being an underlying hair loss problem, as the hairs are constantly being replaced.[5]Bergfeld, W. (2009). Diffuse hair loss: its triggers and management. Cleve Clin J Med. 76(6). 361-370. Available at: https://doi.org/10.3949/ccjm.76a.08080 This keeps hair fall relatively consistent, preventing periods of significant hair shedding.
What Happens to the Hair Cycle during AGA?
AGA, the hair loss condition for which topical minoxidil is an approved treatment, is caused by damage to the hair follicle that contributes to its miniaturization. This is when individual strands of hair become smaller and smaller over time, eventually becoming vellus hairs that are shorter, thinner, and more white, which makes them difficult to see. We have a previous article that explores AGA-induced hair loss in great detail, but let’s summarize the key characteristics below:
- Perifollicular fibrosis – collagen deposition in the spaces around the hair follicles restricts the room within which hair shafts are able to grow, acting as a physical barrier to hair growth.
- Increased telogen:anagen ratio – healthy scalps typically have 1 telogen hair for every 12 anagen hairs (1:12 telogen:anagen), but people with AGA may exhibit ratios that are as high as 1:4 or even 2:5. The telogen phase also lengthens, with hairs spending longer in a state of non-growth.
- Shortened anagen phase – as previously mentioned, the length of an anagen phase directly corresponds to the length of the eventual hair shaft, and people with AGA typically exhibit anagen phases that become shorter with every hair cycle.
Although the exact mechanisms underlying AGA are yet to be fully understood, it is evident that AGA is a progressive and cumulative process that occurs due to harmful factors damaging the hair follicle and shortening the anagen phase.
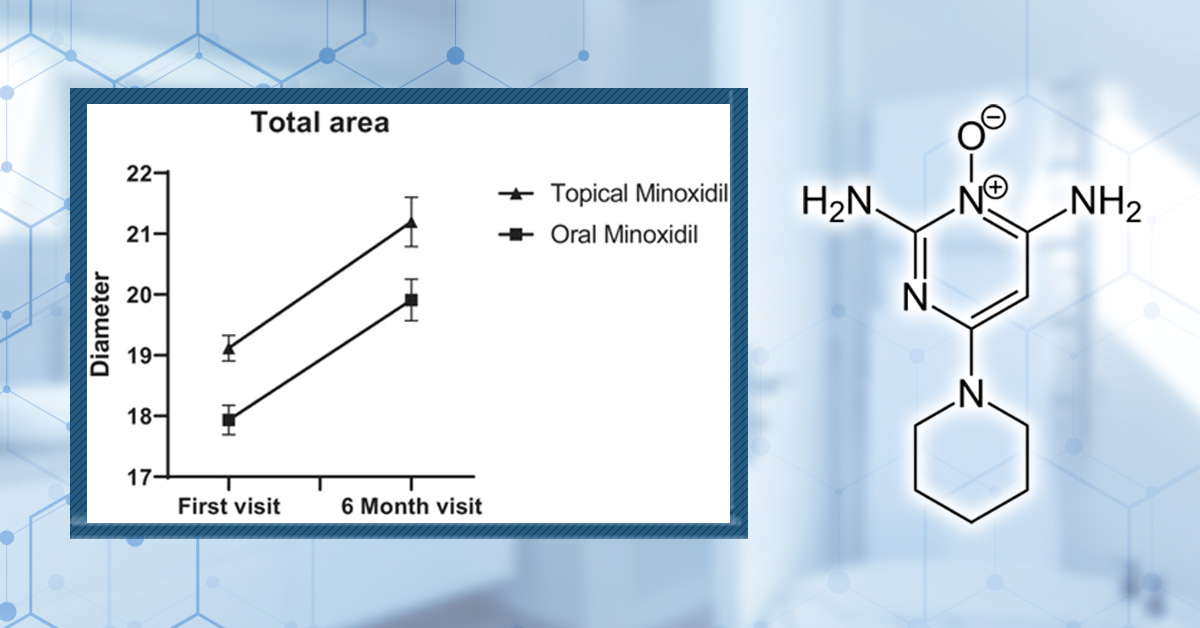
How Minoxidil Works
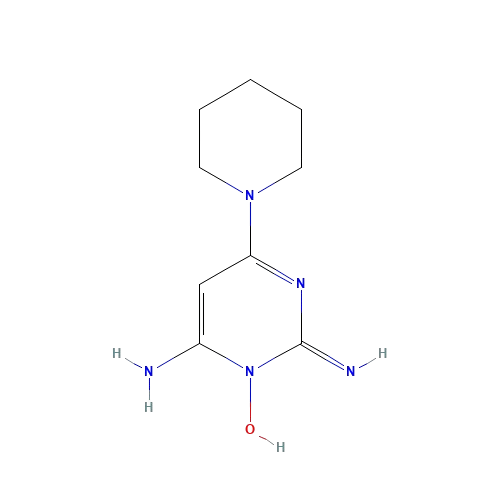
Chemical Structure of Minoxidil. Adapted from:[6]Pubchem (no date). Minoxidil (Compound). Available at: https://pubchem.ncbi.nlm.nih.gov/compound/Minoxidil#section=2D-Structure (Accessed: June 2025
Minoxidil is also yet to be fully understood, but several mechanisms have been suggested that could explain how it reduces hair loss:
- Vasodilation – Minoxidil started out as a treatment for hypertension, so its function as a vasodilator is well documented. Studies have shown that minoxidil increases blood flow by opening potassium channels, reducing high blood pressure. However, an unintended benefit of this function is increased blood flow to the hair follicles, providing them with nutrients and oxygen that help to prolong the anagen phase.[7]Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British journal of dermatology. 150(2). 186-194. Available at: … Continue reading
- Prostaglandin mediation – Minoxidil has been shown to increase the production of prostaglandin E2 (PGE2) in cultured human dermal papilla fibroblasts, the specialized cells that sit at the base of the hair follicle. It is thought that PGE2 may protect hair follicles from damage.[8]Michelet, J. F., Commo, S., Billoni, N., Mahé, Y. F., & Bernard, B. A. (1997). Activation of cytoprotective prostaglandin synthase-1 by minoxidil as a possible explanation for its hair … Continue reading
- Growth factor mediation – Studies have shown that minoxidil has the potential to upregulate the expression of several growth factors that support hair growth, including vascular endothelial growth factor in human dermal papilla cells.[9]Lachgar, Charveron, Gall, & Bonafe. (1998). Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. British Journal of Dermatology. 138(3). … Continue reading
It is likely that minoxidil reduces hair loss through a combination of several mechanisms, including those noted above and perhaps others that are yet to be discovered.
How Might Minoxidil Cause the Dread Shed?
So, we know the basics of the hair cycle, some of the mechanisms that contribute to pattern hair loss, and some of the mechanisms by which minoxidil may reduce hair loss. But what does all this have to do with the ‘dread shed ’? Fortunately, observing the effects of minoxidil is more straightforward than trying to understand how it works, and there is one key effect which is believed to contribute to the increase in shedding: shortening of the telogen phase.
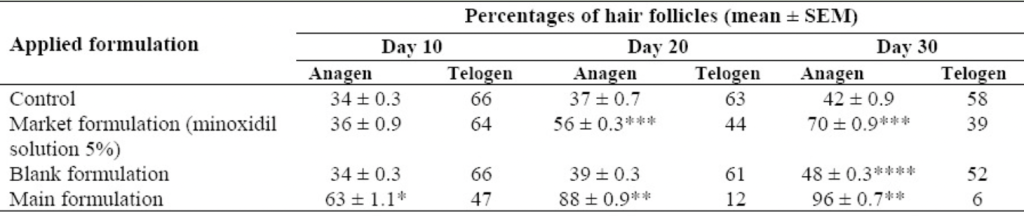
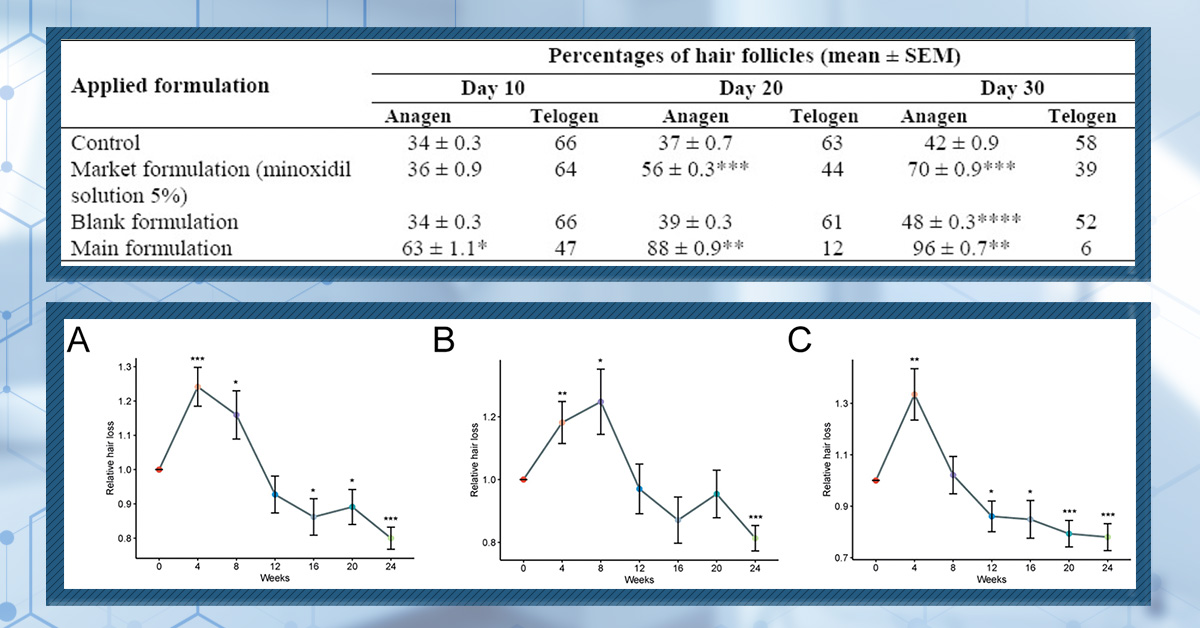
As we previously discussed, a key characteristic of AGA is lengthening of the telogen phase, which causes an abnormal amount of scalp hair to be in a state of arrested growth at the same time. It is widely believed that minoxidil directly addresses this issue by both shortening the telogen phase and accelerating the telogen to anagen transition. In one study, application of topical minoxidil to rats caused a dramatic shortening of the telogen phase, falling from 20 days to just 1-2 days.[10]Mori, O., & Uno, H. (1990). The effect of topical minoxidil on hair follicular cycles of rats. The Journal of dermatology. 17(5). 276-281. Available at: … Continue reading In a separate study that was also conducted in rats, topical minoxidil caused a significant switch from the telogen to anagen phase as quickly as 10 days after beginning treatment (Figure 2).[11]Shatalebi, M. A., & Rafiei, Y. (2014). Preparation and evaluation of minoxidil foamable emu oil emulsion. Research in pharmaceutical sciences. 9(2). 123-133. Available at: … Continue reading
Figure 1: The percentage of rat hair follicles in the anagen and telogen phases following treatment with minoxidil. Control rats were not given any treatment, market formulation refers to standard topical minoxidil [5%], main formulation is minoxidil in a foamable emulsion, and blank formulation is the foamable emulsion without the minoxidil.[12]Shatalebi, M. A., & Rafiei, Y. (2014). Preparation and evaluation of minoxidil foamable emu oil emulsion. Research in pharmaceutical sciences. 9(2). 123-133. Available at: … Continue reading
We could only find one clinical study that has investigated the telogen-anagen shift in humans at an early stage after beginning minoxidil use. They conducted a 24-week trial in which men with AGA either applied topical minoxidil [5%] or topical cetirizine for the first 16 weeks, then stopped use for 8 weeks. Although the results were not statistically significant, they showed that minoxidil caused an increase in the percentage of anagen hair and a decrease in the percentage of telogen hair, supporting the idea that minoxidil rapidly induces shortening of the telogen phase.[13]Mostafa, D. H., Samadi, A., Niknam, S., Nasrollahi, S. A., Guishard, A., & Firooz, A. (2021). Efficacy of cetirizine 1% versus minoxidil 5% topical solution in the treatment of male alopecia: a … Continue reading
In accelerating the telogen to anagen transition, minoxidil also causes “hair follicle synchronization” or synchronization of the hair cycle. As we highlighted earlier, healthy scalps are somewhat ‘protected’ from significant hair shedding events due to the asynchronous nature of the hairs and their individual hair cycles. However, due to the increased density of telogen follicles in the AGA-affected scalp, minoxidil causes a greater-than-normal percentage of hairs to enter anagen at the same time. This syncing of the hair cycles then results in more hairs being pushed out at the same time.
So, to summarize the process that is believed to be the key factor behind the dread shed:
- In AGA, a greater number of hair follicles are in telogen
- Minoxidil treatment accelerates the transition of these hair follicles from telogen to anagen and causes many follicular units to become synchronized
- The transition leads to initiation of the shedding phase (exogen)
- Increased hair shedding occurs
Is The ‘Dread Shed’ Real?
Until very recently, evidence of minoxidil-induced hair shedding was either anecdotal or provided by studies of minoxidil in which increased hair shedding was noted as an adverse event. However, a newly published study sought to investigate the shedding phase in detail.
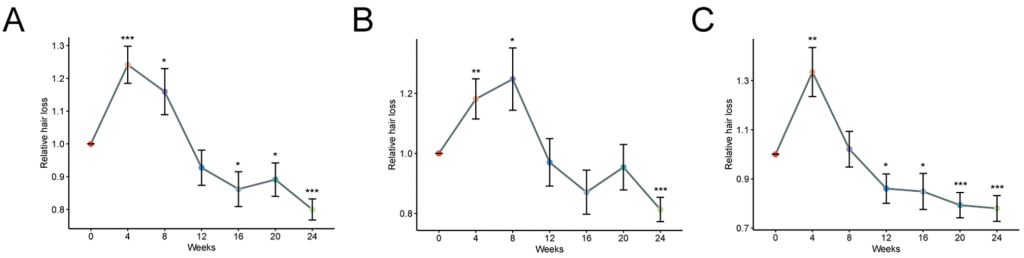
In this 2025 study, 49 patients with AGA used topical minoxidil [2% or 5%] for 24 weeks. Total hair shedding was quantified daily by the participants, who self-assessed their hair fall after combing, after washing, and on the pillow after sleeping. This was then averaged every 4 weeks and compared to the level of hair shedding prior to starting treatment. They found that the participants who used 5% minoxidil exhibited increased hair shedding (relative to pre-treatment) for 4-8 weeks, while the participants who used 2% minoxidil exhibited increased shedding for 8-12 weeks (Figure 2).[14]Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., Du, Y., Lu, C., Zhao, M., Sun, W. & Su, T. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it … Continue reading
Figure 2: Relative hair loss in the 24 weeks after starting treatment with minoxidil. *p < 0.05, **p < 0.01, ***p < 0.001. (A) Hair loss across all patients. (B) Hair loss in patients using 2% topical minoxidil. (C) Hair loss in patients using 5% topical minoxidil.[15]Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., Du, Y., Lu, C., Zhao, M., Sun, W. & Su, T. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it … Continue reading
This study provides definitive evidence that minoxidil does cause an initial shedding phase. However, importantly, hair shedding eventually fell below baseline levels in both groups, indicating that the initial shedding phase is temporary and that minoxidil did begin to reduce hair loss.
Is Initial Hair Loss Actually a Good Sign?
The same authors investigating the minoxidil shedding phase also sought to determine whether the amount of shedding had any association with treatment efficacy. They compared peak relative hair shedding (within the first 12 weeks) to changes in AGA severity using the Basic and Specific classification (BASP), which is a universal hair loss classification system that is used to assess the distribution and severity of hair loss in men and women of all races. They also compared peak relative hair shedding to several trichoscopy measurements, including hair density, hair diameter, and terminal hair proportion.[16]Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., Du, Y., Lu, C., Zhao, M., Sun, W. & Su, T. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it … Continue reading
Interestingly, in the 5% minoxidil group, a significant association was found between the amount of initial hair shedding and hair density, hair diameter, and the proportion of terminal hairs. In other words, people who lost more hair in the ‘dread shed’ actually experienced greater outcomes from minoxidil treatment. Furthermore, participants who initially shed the most hair in both the 2% and 5% minoxidil groups demonstrated the greatest improvements in AGA severity by week 24.[17]Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., Du, Y., Lu, C., Zhao, M., Sun, W. & Su, T. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it … Continue reading
These results are very interesting – not only does the initial shedding phase indicate that the minoxidil is working, but more shedding may even predict better treatment outcomes! So, if you’ve just started treatment, don’t fear the shed!
Final Thoughts
Minoxidil is an FDA-approved treatment for AGA but, in some cases, it can cause increased hair shedding in the early stages of use. Known as the ‘dread shed,’ this phase is believed to be caused by minoxidil shortening the telogen phase of the hair cycle, causing old hairs to fall out. This shedding is even more pronounced due to the increased density of telogen hairs present in the scalps of people with AGA. However, this shedding is only temporary, typically lasting between 4 and 8 weeks. Moreover, people who experience more shedding in this initial phase may actually experience greater overall outcomes from their minoxidil treatment. So, if you have just started minoxidil and are noticing increased shedding, don’t panic – it is most likely a sign that the minoxidil is working.
References[+]
References ↑1 Suchonwanit, P., Thammarucha, S., & Leerunyakul, K. (2019). Minoxidil and its use in hair disorders: a review. Drug design, development and therapy. 2777-2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑2 Sanabria, B., de Nardo Vanzela, T., Miot, H. A., & Ramos, P. M. (2021). Adverse effects of low-dose oral minoxidil for androgenetic alopecia in 435 patients. Journal of the American Academy of Dermatology. 84(4). 1175-1178. Available at: https://doi.org/10.1016/j.jaad.2020.11.035 ↑3 Natarelli, N., Gahoonia, N., & Sivamani, R. K. (2023). Integrative and mechanistic approach to the hair growth cycle and hair loss. Journal of clinical medicine. 12(3). 893. Available at: https://doi.org/10.3390/jcm12030893 ↑4 Natarelli, N., Gahoonia, N., & Sivamani, R. K. (2023). Integrative and mechanistic approach to the hair growth cycle and hair loss. Journal of clinical medicine. 12(3). 893. Available at: https://doi.org/10.3390/jcm12030893 ↑5 Bergfeld, W. (2009). Diffuse hair loss: its triggers and management. Cleve Clin J Med. 76(6). 361-370. Available at: https://doi.org/10.3949/ccjm.76a.08080 ↑6 Pubchem (no date). Minoxidil (Compound). Available at: https://pubchem.ncbi.nlm.nih.gov/compound/Minoxidil#section=2D-Structure (Accessed: June 2025 ↑7 Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British journal of dermatology. 150(2). 186-194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑8 Michelet, J. F., Commo, S., Billoni, N., Mahé, Y. F., & Bernard, B. A. (1997). Activation of cytoprotective prostaglandin synthase-1 by minoxidil as a possible explanation for its hair growth-stimulating effect. Journal of investigative dermatology. 108(2). 205-209. Available at: https://doi.org/10.1111/1523-1747.ep12334249 ↑9 Lachgar, Charveron, Gall, & Bonafe. (1998). Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. British Journal of Dermatology. 138(3). 407-411. Available at: https://doi.org/10.1046/j.1365-2133.1998.02115.x ↑10 Mori, O., & Uno, H. (1990). The effect of topical minoxidil on hair follicular cycles of rats. The Journal of dermatology. 17(5). 276-281. Available at: https://doi.org/10.1111/j.1346-8138.1990.tb01641.x ↑11, ↑12 Shatalebi, M. A., & Rafiei, Y. (2014). Preparation and evaluation of minoxidil foamable emu oil emulsion. Research in pharmaceutical sciences. 9(2). 123-133. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC4311290/ ↑13 Mostafa, D. H., Samadi, A., Niknam, S., Nasrollahi, S. A., Guishard, A., & Firooz, A. (2021). Efficacy of cetirizine 1% versus minoxidil 5% topical solution in the treatment of male alopecia: a randomized, single-blind controlled study. Journal of Pharmacy & Pharmaceutical Sciences. 24. 191-199. Available at: https://doi.org/10.18433/jpps31456 ↑14, ↑16, ↑17 Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., Du, Y., Lu, C., Zhao, M., Sun, W. & Su, T. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it predict treatment efficacy? A retrospective study in androgenetic alopecia patients. Journal of Dermatological Treatment. 36(1). 2480739. Available at: https://doi.org/10.1080/09546634.2025.2480739 ↑15 Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., Du, Y., Lu, C., Zhao, M., Sun, W. & Su, T. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it predict treatment efficacy? A retrospective study in androgenetic alopecia patients. Journal of Dermatological Treatment. 36(1). 2480739. Available at: https://doi.org/10.1080/09546634.2025.2480739 Caffeine & Hair Growth: A Scientific Deep-Dive
In recent years, there’s been an explosion of interest in caffeine shampoos / topicals and their potential to improve pattern hair loss (androgenic alopecia). The hope: that applying caffeine to our scalps might stimulate growth factors, improve blood flow, and maybe even reverse hair follicle miniaturization.
At first glance, caffeine use might look like a viable, natural intervention. But what does the research actually say?
In other words, does caffeine work? Is it a viable alternative to minoxidil? Is caffeine better if ingested, applied topically, or used as a shampoo? How much hair regrowth can we expect? And are there any longterm side effects?
This article dives in the science (and answers).
We’ll dispel a lot of common knowledge about caffeine’s efficacy for hair growth. We’ll also comb through the evidence, set realistic expectations, and reveal how to best use caffeine to maximize your chances of hair recovery.
Long-story short: caffeine isn’t a miracle cure. But it might not be completely useless, either.
Topical caffeine: highlights
- Effort. Low – daily topical application, shampoo use, or ingestion are all easy to implement
- Expectations. According to studies, hair improvements are typically observed by 6+ months
- Response rate: 75%
- Regrowth rate: 0-5%
- Cost. $10-25 per month
- Problems. Low-grade clinical evidence; studies measure outcomes like shedding rates and anagen:telogen ratios, not hair counts; lacking impressive before-after photosets; likely less effective than minoxidil; must be combined with other treatments or ingredients like azaleic acid for maximized benefit.
All-Natural Hair Supplement + Topical
The top natural ingredients for hair growth, all in one serum & supplement.
Take the next step in your hair growth journey with a world-class natural serum & supplement. Ingredients, doses, & concentrations built by science.

Key takeaways
Topical caffeine is clinically shown to reduce hair shedding rates and improve anagen:telogen ratios in men with androgenic alopecia. Unfortunately, it’s still unclear just how effective caffeine-based topicals and shampoos are for improving pattern hair loss.
Caffeine shampoos/topicals fall under an intervention umbrella of “low risk, low reward”. In other words, caffeine’s risk of significant side effects is minimal, as is the amount of hair growth it may initiate.
Having said that, not all caffeine is created equally. While topical caffeine products have been shown to improve shedding rates and anagen:telogen ratios, oral caffeine might actually increase hair loss in those who have insulin resistance or are hypothyroid.
Of all clinical studies on topical caffeine for pattern hair loss, the best results seem to occur when topical caffeine is combined with ingredients like azelaic acid or drugs like minoxidil.
In any case, a 0.2% caffeine dilution for topical solutions and a 1% caffeine dilution for shampoo formulations seem to be the best studied (and most promising), so look for brands that meet these criteria.
If you’re going to use caffeine as a potential hair loss intervention, please understand that this stimulant is only clinically tested on androgenic alopecia, and that it’s likely not effective as a standalone treatment.
More information on the science behind caffeine – its mechanisms, as well as the optimal delivery methods, dilution, and more for hair recovery – can be found below.
What is caffeine?
Caffeine is a stimulant derived from plants – namely, coffee and tea. It’s the most popular stimulant on the planet.
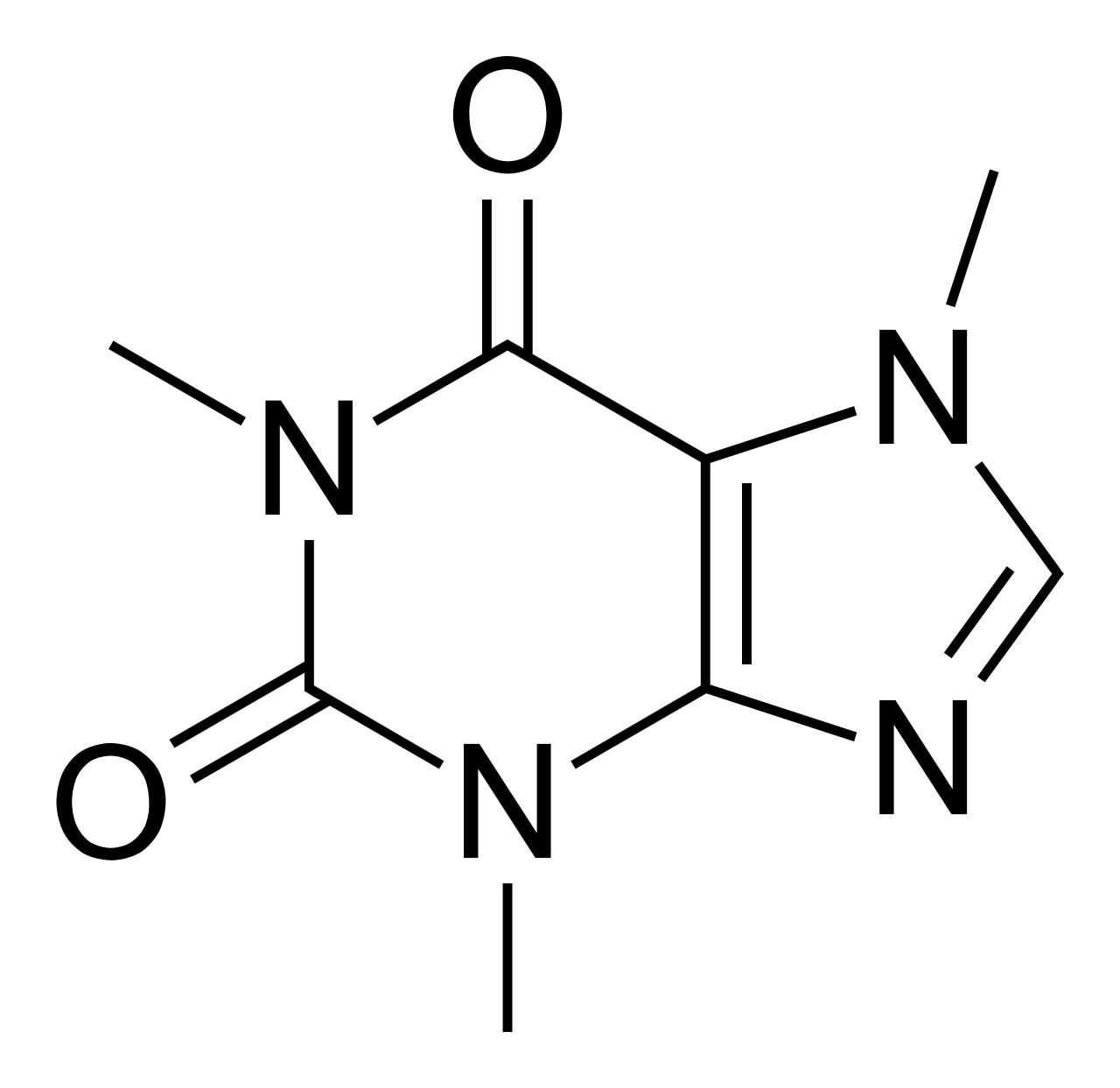
Caffeine structure
As a stimulant, caffeine has a variety of effects on the human body – from better focus to improvements in endurance. But interestingly, the magnitude of these effect often vary per person, and as a result of differences in our genetic constitution, food consumption, and even past caffeine exposure.
What popularized caffeine as a potential hair loss treatment?
In general, caffeine has been studied for its effects on:
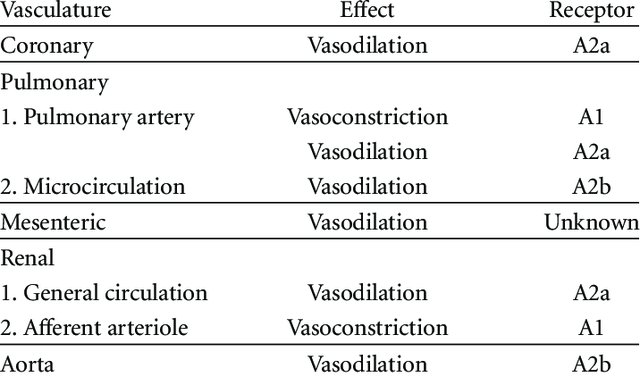
- The cardiovascular system – i.e., increased heart rate and possible vasodilation and/or vasoconstriction
- Longevity – i.e., muscular development, cellular metabolism
- Hormones – i.e., cortisol and thyroid hormones (these may play a role in caffeine’s “energizing” effects)
And interestingly, vasodilation, cellular metabolism, cortisol, and thyroid hormones have all been studied as potential treatments to different hair loss disorders.
For instance, hair loss drugs like minoxidil improve hair growth by increasing vasodilation; thyroid drugs like levothyroxine help to improve hypothyroid-related hair loss by restoring thyroid functionality.
This begs the question: what sort of impact might caffeine have on our hair follicles?
Can caffeine – ingested orally or applied topically – mimic the mechanisms of hair loss interventions? And if so, are the effects of caffeine strong enough to actually improve hair loss outcomes?
These connective points are what prompted scientists to start studying caffeine as a potential hair loss intervention. And taking a deeper look, there is some mechanistic overlap in how this stimulant might improve hair loss outcomes.
Caffeine is one of the most popular stimulants. It’s well-studied in terms of its effects on vasodilation, cellular metabolism, and hormonal health. And interestingly, these research avenues have left scientists wondering if caffeine can also be reoriented as a hair loss solution.
How might caffeine help with hair regrowth?
It’s hard to say. On the one hand, caffeine does have some hair-promoting effects. On the other hand, caffeine also has some issues that may actually contribute to hair loss. All in all, the way it will effect you will boil down to (1) the dose and (2) the ingestion type (oral or topical), and (3) your genetic constitution.
Here are a few effects that caffeine has – both positive and negative – in regard to hair health.
1. Topical caffeine may improve vasodilation (good)
Caffeine’s effects on blood flow vary depending on the mode of ingestion (i.e., topical versus oral) and the actual tissue being measured. Just see this chart demonstrating how oral caffeine impacts blood flow across body tissues.
Caffeine’s location-dependent effects on blood flow
(source)
Interestingly, both topical caffeine and oral caffeine seem to improve blood flow in microcapillary networks – the blood vessel networks that supply our peripheral tissues (i.e., skin) – and the same blood vessel networks that help support the growth of our hair follicles.
This is because in vascular smooth muscle cells, caffeine acts as a phosphodiesterase inhibitor. In other words, caffeine helps to block the enzyme phosphodiesterase.
This enzyme helps inactivate a molecule called cyclic adenosine monophosphate – a biological messenger molecule that regulates vasolidation (i.e., blood flow) in smooth muscle cells. In the absence of phosphodiesterase, more cyclic adenosine monophosphate accumulates, thus expanding vasodilation in smooth muscle tissues.
This is also why phosphodiesterase inhibitors are often prescribed for a variety of blood flow-related health conditions – i.e., erectile dysfunction, hypertension, and even vascular disease. They all help promote blood flow.
Caffeine happens to be one of these phosphodiesterase inhibitors. And while it’s a weak inhibitor, it still has an effect on these capillary networks.
But, there’s one caveat here. While it’s true that a defining characteristic of androgenic alopecia (AGA) is reduced blood flow, it’s still debated whether blood flow is a cause or consequence to hair follicle miniaturization. So, we don’t yet know if caffeine’s vasodilation effects will really have any impact to our hair.
2. Caffeine may increase cellular metabolism (good)
Caffeine doesn’t just inhibit phosphodiesterase. It also inhibits adenosine receptors – a type of neural receptor that helps to regulate cellular metabolism and our own sense of “wakefulness”.
In the absence of caffeine, a molecule called adenosine normally binds to an adenosine receptor in our brain. When adenosine binds to an adenosine receptor, our brain’s neural activity begins to quiet. The end-result: we feel a bit sleepier.
Caffeine is an adenosine receptor antagonist. That means that when it’s ingested, caffeine blocks adenosine receptors so that adenosine cannot bind to them. This prevents the “quieting” of neural activity – and thus promotes longer periods of wakefulness.
Interestingly, there’s also evidence that caffeine’s inhibition of both phosphodiesterase and adenosine receptors may promote cellular metabolism. To put it more bluntly, caffeine ingestion might help to improve (1) energy utilization in the body, and (2) the mobilization of free fatty acids for energy usage.
This may have pro-hair effects, as many genes that are upregulating in balding scalp tissues tend to have an association with impaired cellular metabolism. But again, we just don’t know for sure.
3. High-dose oral caffeine may increase insulin resistance (bad)
Unfortunately, not all effects from caffeine are pro-hair. While improving cellular metabolism may help support the growth stage of our hair follicles, there are also consequences to the way in which caffeine improves cellular metabolism that may negatively impact our hair.
For instance, one study found that oral caffeine consumption decreased insulin sensitivity by 15% in healthy adults. That’s not good – especially for young men and women who are balding, as insulin resistance is almost always a commonly confounding factor in early-onset AGA.
Moreover, there’s also evidence that at high dosages, oral caffeine’s “liberation” of free fatty acids also promotes hyperglycemia and insulin resistance in peripheral tissues (i.e., our skin) – possibly as a result of increased stress hormones like cortisol. Which brings us to our second concern…
4. Oral caffeine increases cortisol levels, and may impair thyroid function and skin quality.
Evidence strongly implicates oral caffeine consumption and an increase in cortisol levels. Unfortunately, the hormone cortisol, when chronically elevated, can negatively impact hair-related bodily functions and in two major ways:
- It can lead to skin degradation around the follicle and disrupt the hair cycle
- It can reduce thyroid functionality, which can increase hair loss for those susceptible to hypothyroidism.
Another noteworthy mention is that coffee can also impair the absorption of thyroxine. So, if you are taking this medication for a thyroid disorder, it’s likely in your best interest to avoid consuming coffee around the same time. But, it’s unclear whether this effect is a result of the caffeine content or other compounds found in coffee.
Again, we don’t yet have any data correlating oral caffeine consumption to pattern hair loss. But these concerns are worth noting for anyone who’s balding and has had a history of hyperglycemia, insulin resistance, hypothyroidism, or adrenal dysregulation.
What about topical caffeine?
In contrast to high dose oral caffeine, it doesn’t seem like topical caffeine elicits the exact same anti-hair effects. Rather, topical caffeine (as a lotion or shampoo) might have some therapeutic benefit to our scalp hair.
For reference, in vitro studies in humans and in vivo studies in mice suggest that caffeine’s effects on (1) phosphodiesterase inhibition and (2) adenosine receptor binding probably will improve hair growth, and through a variety of means.
Specifically, topical caffeine might…
- Prolong the growth phase of the hair cycle (i.e., the anagen phase)
- Stimulate matrix keratinocyte proliferation – or encourage the growth of new hair fibers
- Increase IGF-1 – a signaling protein that helps regulate hair growth
- Inhibit apoptosis – or the “death” of cells responsible for regulating hair growth
- Promote blood flow – which is historically much lower in balding versus non-balding scalps
So, overall, it seems like mechanistic evidence supports at least the use of topical caffeine as a potential hair loss intervention. And this conclusion is why so many researchers have bothered studying caffeine lotions and shampoos for the improvement of AGA.
Caffeine is a (1) phosphodiesterase inhibitor and (2) an adenosine receptor antagonist. While its effects vary on a tissue-by-tissue basis, oral and topical caffeine seem to improve microcapillary networks in periphery tissues (where our hair follicles reside). Moreover, caffeine can help improve cellular metabolism by liberating free fatty acids for energy use. Improvements to both (1) vasodilation and (2) cellular metabolism should theoretically benefit our hair.
At the same time, oral caffeine seems to also increase insulin resistance in peripheral tissues. This is problematic – as reduced insulin sensitivity may interfere with the growth cycles of our hair follicles. Moreover, oral caffeine consumption can increase cortisol levels and decrease thyroid functionality – which may also negatively impact hair growth cycles.
Despite concerns of oral caffeine use for hair, evidence does support the use of topical caffeine for hair growth – at least from a mechanistic standpoint. In vitro research suggests that, in human hair follicles, topical caffeine helps to prolong anagen duration, increase IGF-1, inhibit cell death, and improve blood flow. While this doesn’t mean that these effects will translate in vivo, it does give credence to the idea that topical caffeine is worth testing as a hair loss intervention.
This all brings us to our next question: what does the clinical data say about topical caffeine and its use as a hair growth stimulant?
Is topical caffeine effective for hair loss?
This is harder to answer than it may seem.
At face-value, the answer is yes. This is because there are a lot of studies showing that caffeine in a topical or shampoo (or caffeine in conjunction with minoxidil and/or azelaic acid) can improve hair loss outcomes.
For instance, this recent literature review on topical caffeine dives into over a dozen clinical studies, many of which report:
- “…Statistically significantly better results” when used in conjunction with minoxidil versus minoxidil alone
- Higher patient satisfaction when used with minoxidil compared to minoxidil alone
- Decrease in hair loss after washing when using caffeine + minoxidil + azelaic acid, with similar results to a minoxidil solution after 36 weeks
- Decrease in hairs lost in a hair pull test after 6 months of using a caffeine shampoo (no placebo) and 4 months of using a caffeine-containing lotion.
Reading these conclusions, it’s no wonder why caffeine topical sales have spiked in the last few years.
However, taking a closer look, these studies might not be as encouraging as their conclusions imply. Here’s why.
Quality analysis: how do caffeine-hair loss studies stack up?
When it comes to research, not all peer-reviewed papers are created equally.
Some studies are published in predatory journals that circumvent the peer review process; others are published in low-ranking journals; others simply have major methodological concerns that draw the findings of those studies into question.
When it comes to the studies on caffeine and hair regrowth, it’s that last issue that’s most prevalent. That’s not to say that we should dismiss caffeine’s effects entirely. But, there are several concerns worth highlighting.
1. Methodological concerns
In the above literature review, most of the feature studies don’t measure hair count increases. Rather, they measure endpoints like patient self-satisfaction surveys, changes to anagen:telogen ratios, and a reduction in hair fall during “wash tests” or “tug tests”.
These measurements are difficult to standardize and are notoriously unreliable, meaning it can be hard to determine the true effectiveness of caffeine from any study designed this way.
In fact, it’s my belief that a lot of industry-funded research purposefully chooses these measurement endpoints because of their unreliability. For reference, these types of endpoints are why so many low-level laser therapy studies will report almost unbelievable hair improvement – i.e., “200% hair diameter increases” or “80% hair density improvements” in their clinical trials – while paradoxically, having no visual improvements to show subjects’ photographic assessments.
The bottom line: these measurement endpoints aren’t very strong, and some investigators who choose these endpoints may be doing so to deliberately skew caffeine’s perception of efficacy. But no matter what, the weaker the hair measurement endpoints, the less reliable the results.
2. Topical caffeine is rarely measured as a standalone treatment
Most of these studies measure topical caffeine alongside enhancer ingredients – like azelaic acid, minoxidil, or both – and not caffeine as a standalone treatment. This makes it hard to evaluate whether caffeine by itself is very effective.
In fact, there’s just one study that we could find that measured topical caffeine as a standalone treatment. Unfortunately, it measures topical caffeine versus minoxidil, not a placebo. That study’s takeaway? That topical caffeine is similarly effective to 5% minoxidil… at least when we compare weak measurement endpoints (see #1).
3. Conflicts of interest
Of all the clinical research done on caffeine and hair growth, most of it is industry-funded.
At face-value, this isn’t necessarily a problem. After all, a significant portion of hair loss studies comes from industry-funded research teams. Where there’s financial incentive for a treatment, there will be attempts at peer-reviewed research to prove efficacy.
Having said that, this does become a problem when the studies are typically designed with poor measurement outcomes – such that the odds of achieving “favorable” results increases dramatically. Nearly all of the topical caffeine studies on AGA have this very problem. Compile that with the issue of almost never measuring caffeine alone, and you have even more problems (see #1 and #2).
4. Manufacturers publish research on topical caffeine, but sell you shampoo-based caffeine
One of the most frustrating aspects of hair loss products are that manufacturers will publish a study showing their product demonstrating benefit, but then sell you a product that’s different from the one studied.
This happens all the time with LLLT devices, and it seemingly also happens with caffeine products for hair loss.
Case in point: Alpecin’s study on a topical caffeine solution. The findings showed that this topical did improve hair loss outcomes. But ironically, Alpecin doesn’t sell this topical; it sells a caffeine shampoo. Topicals are leave-in for hours, whereas shampoos may only come into contact with the scalp for 60 seconds. It was this issue in addition to #1-#3 that got Alpecin banned in certain countries from saying their “shampoos” could reduce hair loss.
It was a combination of these issues that led researchers in the above literature review to conclude that while caffeine might help improve aspects of our hair, there isn’t yet enough evidence to support most claims being made by manufacturers.
Topical caffeine does have clinical research supporting its use for hair loss. However, in literature reviews of the dozen or so studies on topical caffeine, concerns of endpoint measurements, lacking study as a standalone treatment, conflicts of interest, and discrepancies in what’s studied versus sold to consumers raises red flags as to caffeine being a truly viable long-term solution.
Despite all of this, there is evidence that topical caffeine might help our hair
Circling back to that literature review, there is accumulating evidence that caffeine can help reduce hair shedding from androgenic alopecia and even improve anagen:telogen ratios. It’s just that if we’re going to use it, we shouldn’t set our expectations at regrowth; we should set our expectations at a slowing of hair loss.
This leaves us with an interesting dilemma: if we want to leverage caffeine as a hair growth promoter, we need to do so in topical or shampoo form. And that means we need to know:
- What’s the best caffeine formulation: topical or shampoo?
- Is there a difference in regrowth reported between 0.2% and 1% caffeine dilutions?
- What’s the optimal frequency of application? Once a week, once a day, etc.?
- Does the effectiveness of caffeine wane over time – like minoxidil and other hair loss drugs?
Sifting through the literature review, there are studies that answer these questions. But again, they’re all subject to significant bias.
Even still, we can use these studies to guide some caffeine best practices – at least for those who want to make the investment and try it out.
Using caffeine for hair growth: best practices
The best caffeine formulation is likely within a shampoo or topical
Oral caffeine likely doesn’t accumulate in the scalp at a high enough degree to elicit adverse or beneficial effects. However, it may have peripheral action through increased cortisol levels, and this may indirectly impede hair growth for those with insulin resistance and/or hypothyroidism.
Knowing this, the safest and most effective way to use caffeine for hair growth is through topical means, either through some sort of lotion or shampoo.
Considering one study found that caffeine solutions penetrate the hair follicle after 2 minutes and peak at 2 hours, a leave-on caffeine solution may be optimal over a shampoo formulation.
The best caffeine dilution is 0.2% for topicals and 1% for shampoos (as far as we can tell)
But to be clear: the answer likely depends on whatever other ingredients are also in the topical (i.e., azelaic acid, minoxidil, etc.). Here’s why.
- This study, the only study measuring the efficacy of caffeine alone, found that a 0.2% caffeine topical (Alpecin) was just as effective as 5% minoxidil at improving anagen hair percentages.
- Problems: does not measure total hair count changes, thus improvements to anagen:telogen ratio can also be achieved by simply just shedding more telogen hairs for either treatment group; study only went for 6 months and results (i.e., improvements to anagen:telogen ratio for both caffeine topical and minoxidil) are within range for what we’d expect from seasonality
- Pazoki-Toroudi et al. (2013) assessed the combination of caffeine 1%, minoxidil 5%, and azelaic acid 1.5% versus minoxidil 5%. The combination therapy was more effective than minoxidil alone.
- Problems: does not measure caffeine alone, so we can’t really say.
- Bussoletti et al. (2010, 2011) found that a 1% caffeine shampoo alone and caffeine lotion alone both resulted in less hair lost to a pull test after 6 and 4 months, respectively.
- Problems: no concentration specified for the lotion; no control group used to assess true efficacy.
- Golpur et al. (2013) found that caffeine + 2.5% minoxidil was more effective than 2.5% minoxidil alone.
- Problems: no percentage of caffeine was indicated.
- Sisto et al. (2013) analyzed the effects of a caffeine-containing shampoo vs a caffeine-free shampoo and found that the active treatment was superior to the placebo.
- Problems: no concentration specified.
So, from what we can garner from the limited studies available, a 0.2% solution for topical application seems to be somewhat effective. However, keep in mind that this is only for a leave-on treatment, not a shampoo.
We don’t really have much information on optimal shampoo dilution. A 1% dilution was the only concentration reported for caffeine shampoo alone, but this treatment wasn’t compared against a placebo and, so, results aren’t super applicable. Even still, 1% seems to be comparably effective to minoxidil.
A 1% caffeine, minoxidil 5%, and 1.5% azelaic acid topical was considered more effective than minoxidil alone, but we can’t extrapolate this 1% dilution to a caffeine-only shampoo.
0.2% for topical solutions and 1% for shampoo formulations is all we can really extract from the current body of evidence, but it should be noted that these recommendations aren’t necessarily reliable given the minimal evidence.
What is the optimal frequency of use?
Like concentration, data on the frequency of use is also sparse. In the highest quality study, subjects used a 0.2% caffeine topical twice a day, every day. Another study instructed subjects to apply a caffeine lotion (unspecified concentration) once daily.
Other studies likely using shampoos most likely involved subjects using the treatment product however often they would normally shampoo their hair. So, it can be difficult to quantify just how often is optimal because use wasn’t standardized, as far as we can see.
With the knowledge we have, daily use of a topical seems to be optimal. Conversely, using a shampoo daily may be drying to the hair and the scalp, so it’s safe to say that using a caffeine-containing shampoo however often you would normally shampoo is probably best.
Does caffeine’s efficacy wane over time?
While no studies measure caffeine’s effectiveness for long enough to see if regrowth is sustained, we can assume – like nearly all topicals – that its hair-promoting effects will likely lessen over time.
The reasons why will be explained in a future article – one comparing the long-term outcomes of antiandrogenic versus non-antiandrogenic hair loss interventions. But the short answer is that we can liken any sort of topical formulation for hair loss – even minoxidil – as a bandaid that won’t necessarily fully address the underlying roots of the problem.
Summary
Topical caffeine may help to elongate the anagen phase of the hair cycle, improve anagen:telogen ratios, decrease hair shedding, and slow the progression of androgenic alopecia (AGA). But it’s by no means a miracle cure, and evidence so far suggests that this topical isn’t any better than 5% minoxidil.
The limited evidence we do have on topical caffeine is relatively biased, poorly designed, and has only been studied for androgenic alopecia. Until studies with better designs are published, it’s hard to say with certainty just how much of an effect topical caffeine will have on our hair.
Nevertheless, topical caffeine leave-on treatments have more quality evidence to support their use (as opposed to shampoos). 0.2% dilutions for topicals and 1% dilutions for shampoos seem to be the most promising. Moreover, the best way to utilize caffeine seems to be in conjunction with minoxidil and, possibly, azelaic acid.
Paradoxically, oral caffeine consumption may have an adverse effect on hair growth through increased cortisol release, increased hyperglycemia in periphery tissues, and potentially decreased thyroid functionality. For these reasons, anyone with a history of insulin resistance or hypothyroidism is probably better off avoiding oral caffeine altogether – particularly for hair health.
If you do decide to implement caffeine into your regrowth regimen, the first thing you’ll notice is how difficult it is to field the market. Almost all companies that have any merit in this space don’t quantify the caffeine dilution in their products. Considering most caffeine treatments are upwards of $35 USD, you may want to call around and see if their customer service representatives can give you more information.
Let us know if you have personal experience or testament in using (or excluding) caffeine. If you start using caffeine, report your progress here for others to see! Any questions or comments? Please reach out in the comments section.
The Ultimate Guide To Platelet-Rich Plasma Therapy (PRP)
In the last decade, thousands of dermatologists have started offering platelet-rich plasma (PRP) therapy as a treatment for hair loss. At first glance, PRP seems like an enticing therapy: a hands-free, drug-free approach to improve our hair thinning…
…but with a $1,000+ price tag, is the therapy worth it? Is PRP right for all hair loss sufferers? And if platelet-rich plasma therapy does work, how much hair can we expect to regrow?
This ultimate guide to platelet-rich plasma therapy uncovers the answers. Here we’ll reveal how platelet-rich plasma therapy works, how it compares to similar therapies, and what most dermatologists don’t tell you about their PRP “before-after” photos.
We’ll also reveal how hair regrowth from PRP depends largely on your form of hair loss, whether you combine PRP with other treatments, and the type of PRP your dermatologist provides (Acell, etc.).
If you’re considering PRP as a hair loss treatment, this guide will help you determine if the costs make sense for your situation and, if so, how to select the right provider.
PRP Therapy: Highlights
- Effort. Medium (requires a few dermatology appointments, but is otherwise a hands-free therapy)
- Expectations. According to studies, regrowth can occur in as little as 3 months.
- Response rate: 80%+
- Regrowth rate: this varies depending on if PRP is done alone or alongside other therapies.
- PRP (alone): 25-30%
- PRP + finasteride: +1%
- PRP + microneedling: +5%
- PRP + minoxidil: +5%
- PRP + microneedling + minoxidil: +5-10%
- PRP + ACell: no studies (yet), but anecdotes suggest better results than PRP alone
- PRP (alone): 25-30%
- Cost. $1,000-$4,000+
- Problems. Expensive; real-world results often don’t match those of studies; similar mechanisms targeted with massaging and microneedling; results vary wildly depending on the clinic; results contingent upon continued sessions
All-Natural Hair Supplement + Topical
The top natural ingredients for hair growth, all in one serum & supplement.
Take the next step in your hair growth journey with a world-class natural serum & supplement. Ingredients, doses, & concentrations built by science.

Key takeaways
PRP is effective for androgenic alopecia and alopecia areata, but it’s expensive and requires ongoing injections to maintain results. It works best as an adjunct treatment alongside other hair loss therapies, and while it has helped both men and women, evidence suggests that it’s less effective for females overall. If you’re going to try PRP, don’t just go to any clinic; rather, vet your cosmetic surgeon by asking them a list of questions we’ve suggested (below).
What is platelet-rich plasma therapy (PRP)?
Platelet-rich plasma therapy (PRP) is an injection-based therapy. It’s the injection of a modified version of our own blood into a tissue site – with the goal to accelerate healing, reduce scarring, and improve injury outcomes.
PRP has been used for dentistry, facial reconstruction surgery, orthopedics, sports injuries, acne scarring, and fat grafting. But in the last decade, it’s been given serious attention as a potential treatment for thinning hair.
How does PRP work?
PRP therapy is a multi-step process that involves drawing a sample of our blood, separating out its platelets, concentrating those platelets, and then re-injecting those platelets into a targeted location (like our scalp).
If you’re considering PRP, the procedure usually takes around an 30-90 minutes, and the process looks something like this:
- Obtain blood from the patient
- Centrifuge once – under a low speed – to separate out platelet-rich plasma from the blood
- Remove anything that isn’t plasma
- Centrifuge again
- Repeat step 3
- Mix the solution to make it uniform
Why focus on platelets?
Our blood volume contains roughly 55% plasma, 40-45% red blood cells, 6% platelets, and 1% white blood cells. Whenever our tissues incur a wound – for example, a paper cut – an inflammatory reaction begins, and our bodies will send blood to our injury site to initiate repair.
Interestingly, our platelets – which constitute just 6% of our blood volume – are responsible for a huge part of the entire repair process. Specifically, platelets do two things:
- Platelets help clot a wound to prevent excessive bleeding
- Platelets carry with them dozens of growth factors, all of which help to coordinate the entire healing process.
This begs the question: what if we could concentrate our platelets so that instead of sending only 6% of platelets to a wound tissue, we could send a much higher percentage? Would we see better injury outcomes? Would we see less scarring?
Well, this is exactly what platelet-rich plasma therapy does. In fact, recent advents in “centrifugation” – or the swirling, mixing, and separation of platelets from our blood – have enabled dermatologists to achieve blood platelet concentrations higher than 94%+. That’s a huge jump from the 6% typically carried within our normal blood volume.
And as of today, it seems like platelet concentrations do improve injury outcomes and scarring. Decades of studies show that, on average, if we concentrate high levels of plasma and send that plasma to an injury site, we can improve injuries, reduce scar tissue, and in doing so, maybe even regrow some hair.
How, exactly, does PRP improve hair loss?
There are several growth factors carried within plasma linked to hair growth, most notably:
- Platelet-derived growth factor
- Transforming growth factor
- Vascular endothelial growth factor
- Epidermal growth factor
- Fibroblast growth factor
- Connective tissue growth factor
- Insulin-like growth factor (IGF-1)
And when injected into balding scalp tissues, the arrival of these growth factors can do a few things:
- They reactivate dormant hair follicles and encourage them to reenter the anagen phase, thus increasing hair counts.
- They prevent hair follicles from entering the catagen phase or undergoing cell apoptosis (programmed cell death).
- They help to resolve longstanding inflammation and reduce scar tissue surrounding miniaturizing hair follicles, thereby increasing the hair thickness of miniaturized hairs.
- The promote blood vessel growth around the hair follicles to aid in hair regeneration.
Isn’t this similar to how massaging and microneedling work?
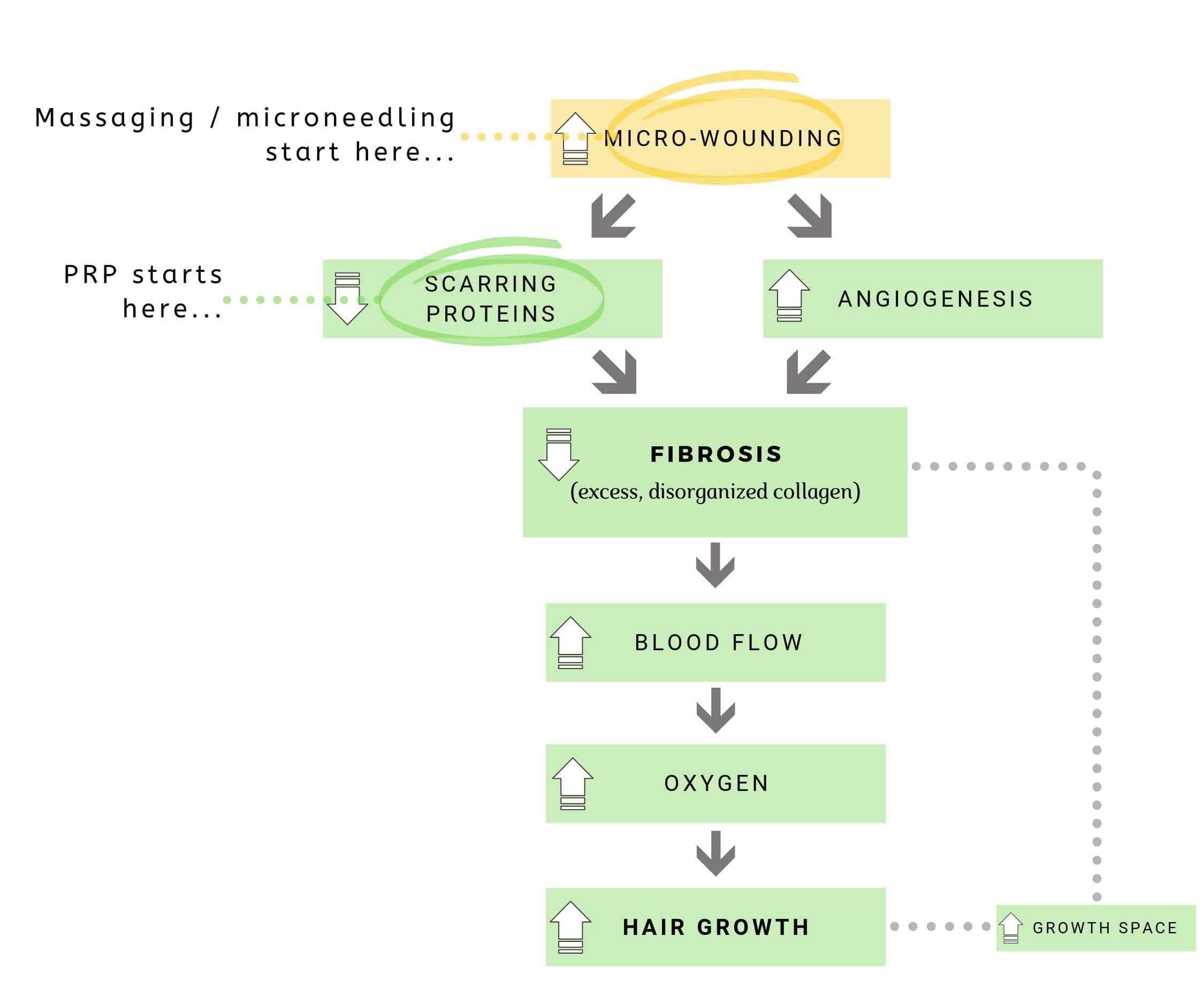
Yes. PRP’s mechanisms overlap with one of the ways by which massaging and microneedling improve hair loss: they both increase the number of growth factors in balding scalp regions. But, they do it in slightly different ways.
Massaging and microneedling first generate acute inflammation (i.e., micro-wounding), which then increases growth factors, which then helps to promote hair recovery. The order of operations is as follows:
Massaging / microneedling >> evokes micro-inflammation >> evokes platelets / growth factors >> decreases scarring proteins / increases angiogenesis >> reduces perifollicular fibrosis >> improves blood flow to miniaturizing hair follicles / increases follicle growth space >> increases hair growth
But there’s a key difference between PRP and these two therapies. With massaging and microneedling, you need to first evoke inflammation to increase growth factors to a wound site. With platelet-rich plasma, you essentially skip that first step, and instead, you simply inject platelets directly into the tissue of your choosing.
When it comes to balding scalp tissues, we can think of the mechanistic difference between these therapies as this:
Now, there is some wounding involved in PRP procedures. But that wounding / acute inflammation is a consequence to the injection of the platelets. In this way, PRP is sort of like a supercharged microneedling or massage session – only with many more platelets present.
Is platelet-rich plasma therapy effective?
This is a tricky question to answer.
If forced to give a one-word answer, then yes. Most studies on PRP show positive outcomes for hair loss. But if you’re going to invest thousands of dollars into the therapy, there are caveats of which you should be made aware.
The reality is that PRP’s effectiveness for regrowth depends on the study you reference and how you define the term, “effective”. Moreover, PRP efficacy varies greatly by:
- Hair loss type (i.e., androgenic alopecia, alopecia areata, etc.)
- Whether we combine PRP with other treatments (i.e., minoxidil, finasteride, microneedling)
- The type of PRP procedure (i.e., PRP alone, PRP + Acell, etc.).
We’ll cover all of this below. First, we’ll start with PRP’s issues. Then, we’ll dive into PRP’s benefits (and its effects on our hair).
Cons
Problem #1: most PRP studies have a high risk of bias
Most PRP studies are conducted by dermatologists who offer PRP procedures at their clinics. That creates an incentive to achieve positive results – because those positive results might encourage patients to do the procedure at their specific clinic.
However, this problem isn’t necessarily game-ending. In fact, nearly all hair loss research contains some level of bias. For instance, despite our efforts to control for bias in our own study on the massages, technically you could argue that because this site conducted it, our results are at risk of bias, too.
In any case, there are plenty of well-controlled studies on platelet-rich plasma and hair loss. We’ve filtered for these. But if you go digging through the literature and find a PRP study with crazy results, just know that if it wasn’t included in our analysis, there’s probably a good reason why.
Problem #2: most platelet-rich plasma studies aren’t standardized
Nearly every PRP study has a different patient profile (i.e., ages and hair loss severities), methodology (i.e., injection methods, rounds, treatment regions), trial duration (i.e., three months versus two years), and hair assessment method.
For instance, here are just a few ways PRP studies have measured hair loss “improvements” (ranked from worst to best).
- Hair tug tests (i.e., when an investigator yanks at a subject’s hair before and after treatment, then counts the number of hairs that fell out)
- Reduction in hair fall (i.e., hair shedding collected in the shower, by patients, pre- and post- treatment)
- Patient satisfaction scores (i.e., when a patient fills out a survey saying whether they’re happy with the treatment)
- Independent visual assessments (i.e., when a dermatologist eyeballs photos to give their input on hair improvements)
- Follical unit survival rates (for PRP + hair transplant studies) (i.e., how many transplanted hairs survived the transfer)
- Hair thickness (i.e., the change in the diameter of hair shafts in a specific scalp region)
- Terminal/vellus hair ratio (i.e., the number of thick and healthy versus thin and wispy hairs in a specific region)
- Hair counts (i.e., when a region is marked – usually with a temporary tattoo – to count hairs before and after treatment)
To be fair, this isn’t just a problem with PRP; it’s a problem with all of hair loss research. It’s why literature reviews have a hard time drawing conclusions about most treatments – because there are rarely apples-to-apples comparisons.
But again, we’ve sorted through all the PRP studies we could find to standardize the research (as best we can) and give you ballpark assessments of regrowth rates (i.e., increases to hair count in balding regions).
Problem #3: platelet-rich plasma therapy likely requires ongoing treatments
When you look into the research on minoxidil or finasteride, studies show that within 3-12 months of quitting either drug, your hair loss will return to what it was prior to the intervention. So, how long will results hold for platelet-rich plasma after quitting?
Well, it’s unclear how long results will last after you stop doing PRP treatments, but evidence suggests that a percentage of people will start seeing their hair return to baseline after a year.
Out of all PRP studies, the one with the longest follow-up period (two years) included 20 patients. Interestingly, four of them experienced a relapse in hair loss one-year post-PRP. In fact, their androgenic alopecia progressed beyond their pre-trial hair counts by the 16-month mark. This suggests that for about 20% of people, PRP’s effects start to wane 12-18 months after the treatment.
This wouldn’t be such an issue if the procedure were cheap, but it isn’t: several therapeutic rounds of PRP cost $1,000-$4,000+. So, if you’re going to give this procedure a try, make sure you’re financially comfortable with the investment.
Problem #4: PRP might not work for every person with hair loss
Out of all the research on PRP, only two studies found that PRP was an ineffective treatment option. One study was on females with androgenic alopecia. The other study was on men with advanced androgenic alopecia (Norwood gradients 4+).
If you were to ask me why the first study failed to produce results, I would say that it was probably because (like most women with hair loss) the females in that study likely had other undiagnosed hair loss types (like telogen effluvium / hair loss related to a chronic condition).
And as far as the study on men with advanced androgenic alopecia (AGA) – we need to keep in mind that the investigation team only did two rounds of PRP injections. For what it’s worth, in all of the PRP studies which saw improvement, a minimum of three PRP injection rounds were performed. So, it’s likely that either this study didn’t do enough injections to see results, or that men with advanced AGA needed several more injections before PRP begins to repeat significant benefit.
Problem #5: most PRP studies don’t measure PRP by itself
In fact, the overwhelming majority of studies measure PRP alongside other hair loss treatments – like minoxidil, finasteride, or even a hair transplant. So, it’s important to delineate between the studies you reference when evaluating whether PRP is right for you (we’ve done this below).
This also brings up another problem: dermatologists showcasing their PRP results online often don’t tell you something important: that they’re showing you PRP results alongside drugs like minoxidil and finasteride.
This is incredibly disingenuous, and I suggest that if you’re shopping around for a PRP clinic, you call ahead and ask the doctor if the results they showcase on their website are from PRP alone. If they are, great. If they’re not, but they’re labeled to make it seem as such, then that means these dermatologists are intentionally misleading prospective patients, and they should lose your business (and their license to practice).
Pros
While we might’ve just painted a problematic picture for PRP, this isn’t the whole story. In fact, PRP is an incredibly effective treatment for hair loss under the right circumstances. This is all covered below.
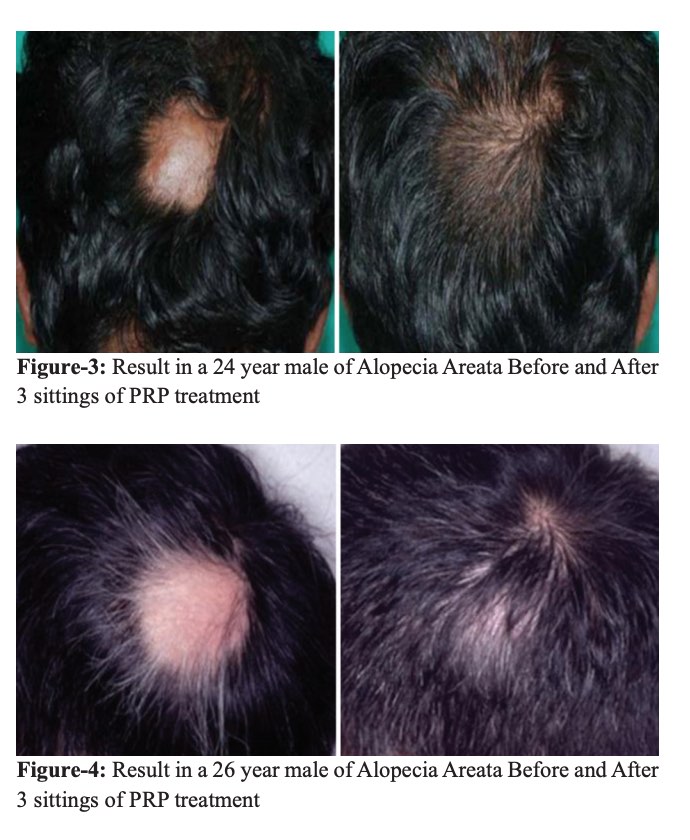
Alopecia areata
PRP has shown great promise for the hair loss disorder alopecia areata. This is an autoimmune condition that leads to hair loss in patchy spots throughout the head. In some cases, it can advance to complete baldness (alopecia universalis).
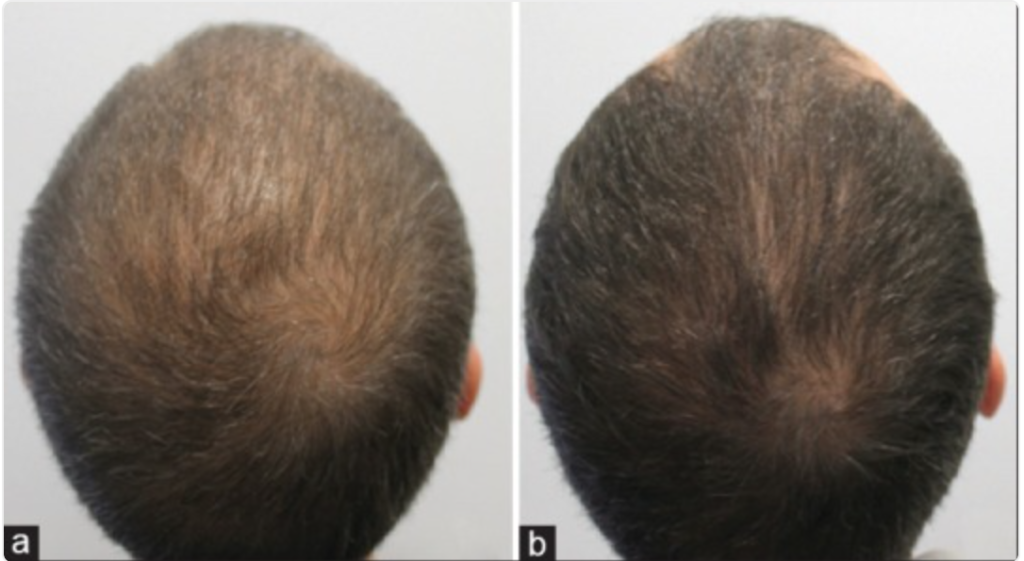
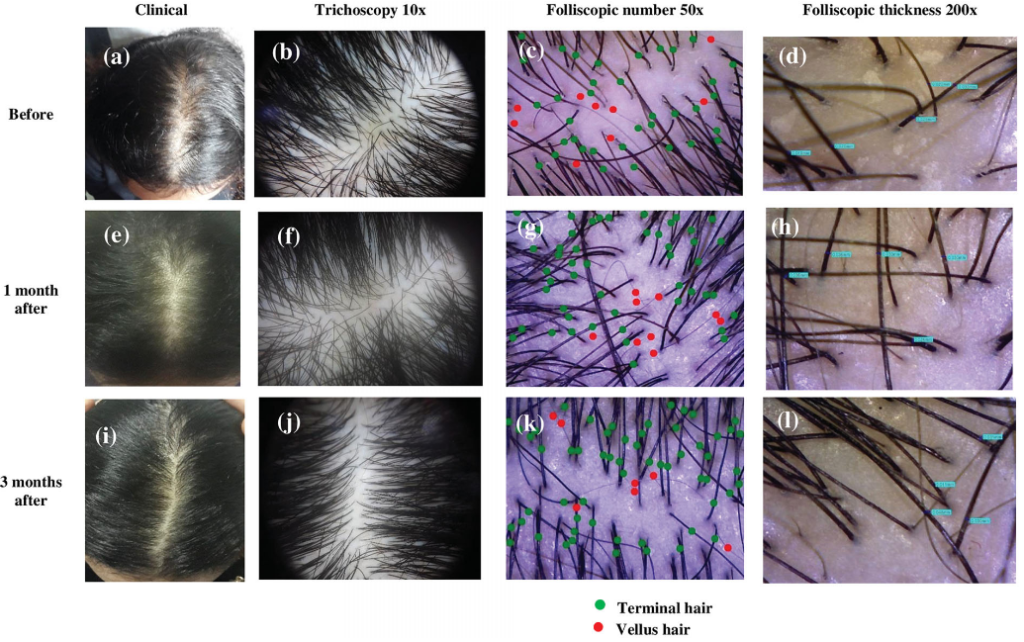
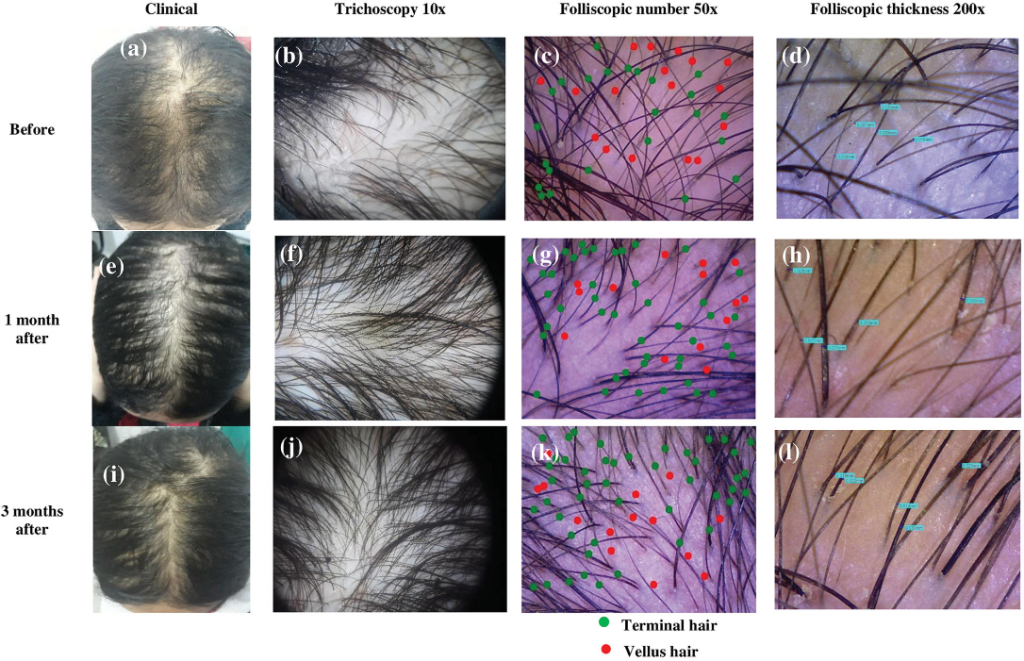
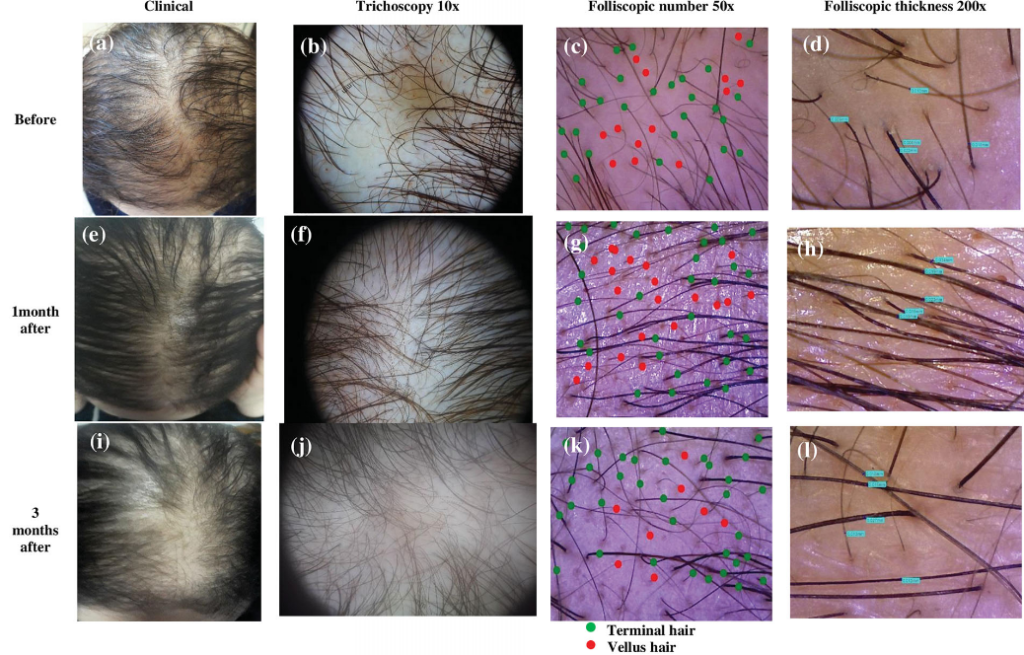
In fact, studies show that PRP is very effective in treating at least 70% of alopecia areata cases. Here are some of the really promising photos (source):
Androgenic alopecia
PRP alone (no other treatments)
When looking at PRP as a standalone treatment, most studies measuring hair counts suggest that the average patient will regrow 15 hairs per square centimeter (i.e., half the size of a penny). That’s about a 25-30% regrowth rate at 3-6 month follow-ups.
Quantitatively, that’s pretty impressive. For a benchmark, most studies on finasteride show just a 10% increase in hair count over two years.
Across studies, some of the less quantitative outcomes for PRP alone (at least at the 3-6 month mark) are:
- Patients report about a 75% increase in hair quality and thickness, and a decrease in the rate of hair loss.
- Patients usually perform better on the hair pull test, meaning that when they pull on a clump of the patient’s hair, fewer hairs came out compared to control groups.
- Patients report somewhat high satisfaction with the procedure at month three of follow up
Finally, a common trend mentioned among researchers is that PRP treatment seems to be more effective for patients with less severe forms of AGA. So, if you’re in the early stages of hair loss, PRP might be a great option for you.
PRP + hair transplantation
In one study examining hair transplantation, two areas with 50 grafts each(not a lot to measure, especially for hair transplantation) were compared with or without PRP injections. The area with PRP had, on average, 46.75 units that survived compared to the non-PRP which had, on average, 41 units that survived.
While this isn’t that drastic of an increase – we have to keep in mind that transplant procedures are incredibly costly… and that means that every hair follicle unit counts.
So, if you’re considering a hair transplant, you’ve spent the finances to secure a skilled surgeon, and you still have some extra spending money you’d like to throw into improving your results – do it alongside PRP. Chances are your hair transplant survival rates will improve, as will your overall hair count.
PRP + microneedling
In this study, 30 male participants received 6 PRP injections following microneedling sessions. At the six month follow-up, the average patient had a hair density increase of about 30%. This study also noted that the most significant improvement was seen in patients with less severe AGA.
PRP + minoxidil or PRP + finasteride
In this study comparing the efficacy of PRP + minoxidil and PRP + finasteride, while both outcomes were deemed effective, the PRP + minoxidil treatment actually achieved significantly better results than the PRP + finasteride group. In fact, the PRP + minoxidil group showed five-fold better hair increases versus the PRP + finasteride group.
Now, you might read these results and think that makes no sense. Finasteride is clinically more effective than minoxidil. So, why would PRP + minoxidil outperform PRP + finasteride?
Well, the devil is in the details. For one, the sample size of each PRP subgroup was less than 15 people. So, it’s possible these differences might’ve been due to statistical noise which would’ve canceled itself out with subgroups of 150+ people.
And secondly, while a five-fold improvement might sound drastic, we’re actually dealing with the law of small numbers here. Yes, PRP + finasteride saw an additional hair count lift of 1% versus 5% for the PRP + minoxidil group. And yes, that is technically a five-fold improvement. But in all reality, that’s just a few percentage points better.
PRP + minoxidil + microneedling
In this study comparing PRP + minoxidil + microneedling versus minoxidil alone, the earlier treatment proved to be much more effective than the latter, although exact numbers were not given in this study to show this. However, we can assume that PRP + minoxidil + microneedling is probably better than PRP + minoxidil, and that PRP + minoxidil is probably better than PRP alone.
PRP + Acell
PRP + Acell is a relatively new procedure that a lot of PRP practitioners are offering now. Acell is a protein matrix derived from pig bladder (you read that right) that creates a “scaffold” for new hairs. Acell essentially offers a platform by which all of our growth factors (and hair) can cling to. It also helps to stimulate stem cell activity.
[Note: since ACell is made from pig, people allergic to pig products should notify their physicians about their allergy prior to the treatment.]
There doesn’t seem to be any studies measuring the results of PRP + Acell compared to PRP alone, so it’s hard to objectively say whether it increases the effectiveness of PRP.
Are there any risks to PRP?
At least so far, there haven’t been any severe risks reported. However, some milder symptoms have been noted during and shortly after the procedure:
- Headaches
- Pain
- Itching
- Drowsiness
- Infection (rarer)
- Scarring or calcification of the injection site (for those with a propensity toward keloid scarring) (rarer)
- Blood vessel or nerve injury (rarer)
Who’s a good candidate for PRP? Who’s a bad candidate?
Androgenic alopecia treatments vary depending on your (1) finances, (2) willingness to invest time into a therapy, and (3) comfortability with FDA-approved drugs. Compared to some of the other AGA specific treatments, like standardized scalp massages and microneedling, PRP is a pricey option. At the same time, PRP is a lot less time consuming than microneedling or massaging because you may only go into a clinic for a handful of injection rounds before you start seeing results.
You are a great candidate for PRP if you…
- Are comfortable with the financial investment and ongoing treatments
- Want to stack PRP alongside other treatments in your hair loss regimen – like microneedling, massaging, minoxidil, or finasteride
- Are going to get a hair transplant and want to optimize your outcome of the procedure
- Have early-stage to mild AGA (and relatively small areas of hair loss)
- Have alopecia areata
- Are a poor candidate / do not want to try traditional hair loss medications
- Are physically healthy in general
You are not a great candidate for PRP if you…
- Do not want to spend thousands of dollars on the procedure
- Have advanced AGA and aren’t willing to commit to several rounds (and months-to-years) of injections
- Are a female with androgenic alopecia alongside conditions like hypothyroidism (treat that first!)
- Do not want to keep reinvesting money into a procedure that might not prove effective after a year.
- Have a bleeding disorder with clotting factor deficiencies or low platelet counts
- Use blood thinners
- Have a liver disorder
- Have a recent cancer history or sepsis (blood infection)
- Are a heavy smoker, alcoholic, or illicit drug user
- Have low blood pressure or chronic user of steroids
Do PRP study results hold true in the real world?
When evaluating any hair loss therapy, it’s important to note that, sometimes, study results don’t match up to real-world results. On hair loss blogs and forums, there’s sometimes an inkling that this might be the case with PRP.
For starters, while there are positive patient stories with PRP, there are also many anecdotes of patients who tried PRP without success. This video is a perfect example. And if you dig deeper, you’ll probably find more negative than positive anecdotes.
This can be confusing – as most of the literature tends to describe PRP as seemingly beneficial. And even more troublesome, it’s also worth noting that I’ve spoken with dozens of readers who’ve tried PRP… and most of them have also reported negligible improvements.
This begs the question: is PRP that effective? And regardless of the answer, why might there be a discrepancy between studies’ results and patient reports?
What could explain the discrepancies between PRP studies and real-world results?
For starters, it’s actually unclear if there are clinical versus real-world discrepancies for PRP. For instance, it’s possible that PRP treatments might just suffer from the “Yelp effect”. This is when someone with a negative experience is far more likely to leave a public review versus someone with a positive experience.
So, PRP might just be one of those treatments that have collected negative reviews over a period of years – much like finasteride and its reports of sexual side effects.
Secondly (and this is the more important point to make), clinical research does not always depict reality. For instance, while a 25-40% increase in hair count from PRP looks great on paper, it doesn’t always translate to cosmetic results.
This tends to be a problem with even the “best” FDA-approved treatments. Just take a look at these five men who did combination treatments of minoxidil, finasteride, laser combs, and even hair transplants – and their results after one year.
I’ll save you the suspense: their final “after” photos are darkened to obscure just how minimal their hair changes are. And, for the hundreds (to thousands) of dollars each of the men spent, their hair seems more-or-less cosmetically unchanged.
(Note: that video is just one of the reasons why, for most AGA sufferers, I recommend approaches like massaging / microneedling as a baseline for any regimen. Not only do these therapies enhance other hair loss treatments, but also without their inclusion, you’re statistically likely to see no cosmetic improvements from your other treatments).
Thirdly, we just learned that without follow-up sessions, PRP results will fade for 20% of patients starting 12-18 months after their last round of injections. So, if you’re reading a review of someone who got PRP once four years ago and never saw results (or is experiencing continued hair loss) – you’ll know that it’s probably because they didn’t do enough injection rounds and they didn’t keep up with the therapy.
In any case, it’s worth noting that of the readers with whom I’ve communicated, the ones who tried PRP + Acell all reported positive results. While there aren’t yet studies validating this combination therapy, it seems to be the most promising from an anecdotal standpoint (if that means anything to you).
How to choose the right PRP practitioner
If you’re going to invest $1,000+ into platelet-rich plasma, don’t give your money to the first PRP clinic you find.
Instead, find a few clinics near you that offer the procedure and make sure they have a proven track record. That means they should have a website with PRP before-after photos.
Make a list of these clinics. Then, call each clinic and ask if their online photos are of PRP alone or of PRP + finasteride / minoxidil.
If the photos are of PRP alone, great! If they’re of PRP + multi-therapies but advertised to represent only PRP… then hang up, cross them off your list, and consider reporting them the clinic to the Better Business Bureau.
Doing this should eliminate 60-70% of providers.
Next, call the remaining providers and ask about their PRP techniques. You’ll want to find a clinic that offers 1-4+ months between injection rounds. If someone offers you a package of 10 PRP sessions spaced out as one injection round per week, that goes against the literature’s recommendations (and our understanding of wound-healing timelines) – and you should probably find another provider.
Moreover, the actual PRP product that a clinic uses will vary by volume, number of injection rounds, color, platelet count, leukocyte count, and protein content. The best PRP providers will offer double-spin centrifugation preparation with an activator like thrombin or calcium chloride. While some evidence suggests that “platelet activators” are not necessary, they also don’t seem to hurt the procedure. In any case, it’ll be good to speak with your provider about all of this – so you can get a feel of whether they can actually answer these questions. If they can’t, then cross them off your list.
Lastly, ask your remaining clinics if they offer PRP + Acell. If they do (and the costs aren’t prohibitive), then this might be a better option versus PRP alone. If they don’t, it’s not the end of the world – and chances are you’ll still see benefit from the procedure.
Following this process should leave you a few great PRP clinic options. And, as long as you’re willing to commit to injections every 4-6 months, you should see a considerable lift in hair count… especially if you’re combining platelet-rich plasma with other therapies.
Summary
Platelet-rich plasma therapy is effective for both androgenic alopecia and alopecia areata, but the procedure is cost-prohibitive and requires repeated clinic visits to see sustained results. These factors are big turnoffs for most hair loss sufferers considering PRP as a treatment option.
PRP works best as an adjunct treatment alongside other hair loss therapies. So, don’t just try PRP as the only thing to help your hair. We recommend combining it with massaging and/or microneedling, and (if you’re comfortable), FDA-approved drugs (especially minoxidil) to maximize results. And, if you can, try to find a clinic that provides PRP + Acell.
While PRP can help both male and female hair loss sufferers, preliminary evidence suggests that it’s just not as effective for females. However, this might be because of the added complexities of female AGA – and the fact that women are so frequently misdiagnosed with AGA but instead actually have hair shedding disorders related to underlying chronic conditions.
Be careful about clinic selection for PRP. For instance, the photos you’ll see on most dermatology websites offering PRP are from patients doing PRP + finasteride / minoxidil (rather than PRP alone). The equivalent would be if I advertised a before-after photo of someone doing massaging + finasteride, but decided to position the photo as if the results were only from massaging. It’s just bad business. So, be aware of this. Call all clinics to confirm the regimens of their highlighted patients.
If you’re seeing improvements from PRP, chances are you’ve already navigated through this mess to find a good provider. So, steer the course and keep us posted with your progress!
Saw palmetto for hair growth: does it stack up to finasteride?
Saw palmetto is often touted as nature’s finasteride. This herbal extract reduces levels of the hormone dihydrotestosterone (DHT). For this reason, it’s clinically demonstrated to improve hair growth in men with androgenic alopecia (AGA).
But is saw palmetto as effective as finasteride?
Not quite. In fact, a closer look into the research on saw palmetto reveals that taking this supplement also comes with risks.
That’s not to say that saw palmetto is ineffective. But if you’re going to opt for saw palmetto over finasteride, you’re going to want to weigh its benefits and risks. Specifically, you’re going to want to know how saw palmetto compares to finasteride in terms of its (1) ability to reduce DHT, (2) ability to regrow hair, and (3) reported side effects.
This Quick Win dives into the details (and answers). The bottom line: saw palmetto isn’t as powerful as finasteride, but it also comes with some upsides that may make it a better option, at leaast for certain hair loss sufferers.
Note: Quick Wins are short articles focused on answering one question about hair loss. Given their specificity, these articles are written in a more scientific tone. If you’re new to hair loss research, start with our long-form articles.
All-Natural Hair Supplement + Topical
The top natural ingredients for hair growth, all in one serum & supplement.
Take the next step in your hair growth journey with a world-class natural serum & supplement. Ingredients, doses, & concentrations built by science.

What is saw palmetto?
Saw palmetto is a palm plant native to warm humid climates (Florida).
Saw palmetto plant
Thirty years ago, researchers discovered that certain polyphenol and lipid extracts of saw palmetto could reduce the activity of type II 5-alpha reductase – an enzyme our bodies use to make the hormone dihydrotestosterone (DHT).
DHT is the main hormone implicated in pattern hair loss, also called androgenic alopecia (AGA). And interestingly, the popular hair loss drug finasteride (Propecia®) reduces that same enzyme – type II 5-alpha reductase – to lower DHT levels and improve pattern hair loss outcomes in ~80% of men trying the drug.
Unfortunately, finasteride use is also associated with sexual side effects. This scares a lot of men away from trying the drug. It also leads many of them to wonder…
“If I use saw palmetto instead of finasteride, can I still regrow hair while also reducing my risk of side effects? After all, saw palmetto is natural, and natural often means safer.”
For starters, anything branded as “natural” isn’t always safer. Many natural supplements sold through Amazon and tested by third parties have been found to contain dangerous levels of heavy metals. Moreover, many popular natural extracts – i.e., green tea extract – have been associated with hepatic failure due to the metabolic demands these extracts can place on our livers. (For more information, see my master class).
In any case, if we’re to answer the question, “Is saw palmetto as effective as finasteride?”, we’ll need to evaluate saw palmetto versus finasteride in terms of its (1) expected hair regrowth, and (2) risk of side effects.
The rest of this Quick Win does just this.
Saw palmetto vs finasteride: hair regrowth
“Hair regrowth” is a non-specific term, and most clinical studies on pattern hair loss define this term differently:
- Reductions to hair shedding
- Improvements to anagen:telogen ratios
- Increases in non-vellus hair counts
- Global photography assessments
…the list goes on.
That’s why, whenever we’re diving in studies on hair loss, we standardize the term “hair regrowth” into two categories:
- Response Rate. This is the percentage of people receiving treatment who see a slowing, stopping, or reversal in their hair loss. After all, simply slowing down the progression of AGA is a win for most hair loss sufferers.
- Regrowth Rate. This is the percent change in terminal (i.e., non-vellus) hair counts before and after treatment. We use this metric because it’s best out there in terms of translation to visual improvements. Increases to vellus hair counts (i.e., small, wispy, white hairs) just don’t show up in photographs, so we don’t count those.
So, if we summarize the data on saw palmetto versus finasteride, how do these two interventions compare? (1) (2)
Saw Palmetto (320mg)
Finasteride (1mg)
Response Rate 60%; dependent on the dose and delivery (supplement or topical) 80-90% Regrowth Rate 0-10%; potentially higher if used alongside other therapies 10%, alongside thickening of miniaturizing hair The key takeaway: compared to finasteride, saw palmetto has a lower response rate and regrowth rate. And when we look at the individual studies that constitute these aggregated estimations, things look a little bleak.
To date, there’s only one clinical study directly comparing saw palmetto versus finasteride for the treatment of androgenic alopecia. The investigation team randomized 100 men with androgenic alopecia into two groups. They gave one group 320mg of saw palmetto and the other group 1mg of finasteride – every day, for two years. (3)
The results? After 24 months, 68% of finasteride users saw hair regrowth, while only 38% of men taking saw palmetto saw hair regrowth. Moreover, investigators noted that in the saw palmetto group, hair regrowth only occurred in the crown (i.e., vertex), and that the magnitude of regrowth was significantly less than finasteride.
In other words, saw palmetto achieves half the response rate of finasteride, and that if regrowth does occur, it’s not nearly as impressive as what finasteride achieves.
Having said that, it’s not all bad news. That same study also demonstrated that 45/50 men in the saw palmetto group saw a stop in hair loss over the two years that they took it. So, in this one study, saw palmetto showed a response rate of 90%.
This means it’s not a complete stretch to say that most men taking 320mg of saw palmetto daily should see an improvement in their pattern hair loss. It’s just that this improvement won’t be anywhere near on-par what is achievable with finasteride.
Why isn’t saw palmetto as effective as finasteride?
This likely has to do with the ways in which saw palmetto reduces DHT, and the amount of DHT that saw palmetto reduces.
Finasteride is a synthetic azosteroid. It reduces DHT by competing with (and binding to) a coenzyme that our bodies use to make type II 5-alpha reductase – the enzyme that converts free testosterone into DHT.
Conversely, saw palmetto competitively and non-competitively inhibiting type II 5-alpha reductase, reducing the binding of DHT to androgen receptors, and increasing the conversion of DHT to a weaker metabolite called androstanediol (4).
The long-story short is that these factors, along with differences in the half lives of both finasteride and saw palmetto, lead to differences in their abilities to lower DHT.
For reference, see this graph on the DHT-reducing capabilities of both saw palmetto and finasteride, as organized by different tissue sites.
The bottom line: saw palmetto is about half as effective as finasteride because it just doesn’t reduce as much DHT.
What about side effects?
At this point, we’ve really only evaluated half of the question: is saw palmetto as effective as finasteride?
In terms of response rates and regrowth rates, the answer is no. But can saw palmetto make up for its lower efficacy by being a safer long-term supplement for hair loss sufferers?
Maybe.
Saw palmetto: risk of adverse events
Long-term clinical studies show that saw palmetto’s overall rate of side effects is just 2%. Moreover, if side effects do occur, they’re relegated more so to gastrointestinal distress than to sexual dysfunction. Even better, many studies on saw palmetto show no change in libido; some studies show improvement to sexual health (at least for men with enlarged prostates). (5) (6) (7)
Finasteride: risk of adverse events
The “Yelp effect” is a phenomenon where patrons of a business are far more likely to leave a review if their experience was negative rather than positive. Finasteride is a drug that suffers from the Yelp effect, meaning that its sexual side effects are often overstated and amplified online.
Having said that, side effects do occur. Moreover, the self-assessment questionnaires filled out by participants in large-scale clinical trials for finasteride were worded in such a way where under-reporting certain side effects were more likely than not (but that’s for another article).
In any case, finasteride use does come with a heightened risk of side effects. Depending on which study you cite, between 1% and 25% of finasteride users will report issues ranging from brain fog to depression to sexual dysfunction. (8) (9) Moreover, when side effects are reported, they seem to be of higher magnitude versus saw palmetto.
The bottom line:
- Finasteride’s side effects are likely overstated online and understated in the literature
- Compared to saw palmetto, finasteride likely leads to side effects that are (1) of higher incidence, and (2) more severe
Saw palmetto or finasteride: which one is right for you?
It depends on your risk tolerance for side effects, and whether you plan on combining saw palmetto with other treatments, therapies, or procedures to make up for its lower response rates and regrowth rates.
There’s evidence that supplemental + topical saw palmetto, alongside other ingredients, might lead to better hair loss outcomes than just supplemental saw palmetto. Moreover, combining saw palmetto with massaging, microneedling, platelet-rich plasma therapy, or other interventions might help mitigate its lower efficacy.
Having said that, making these choices will depend entirely on someone’s needs, preferences, and unique hair loss situation.
Anything else to know?
Unlike finasteride, saw palmetto isn’t standardized. Serenoa repens growing conditions, extraction methods, and manufacturing practices can all impact the composition, bioavailability, and absorption of each saw palmetto supplement. In fact, these differences might explain the variances in response rates and regrowth rates seen across saw palmetto studies.
So, if you’re looking for a more comprehensive guide on how to use saw palmetto – including recommendations for dosages, extraction practices, and combinations therapies – see our saw palmetto ultimate guide here.
The bottom line
In order to evaluate whether saw palmetto is as effective as finasteride, we need to understand how the supplement compares in terms of (1) response rates, (2) regrowth rates, and (3) risk of side effects.
Saw palmetto isn’t as effective as finasteride in terms of its response rates or regrowth rates, but it also seems to cause fewer (and less severe) side effects. Because of this, making the choice to use saw palmetto over finasteride depends entirely on someone’s risk tolerance for side effects, as well as whether they plan on combining the supplement with other treatments, therapies, or procedures to make up for its less-impressive efficacy.
Saw Palmetto (320 mg)
Finasteride (1 mg)
Response Rate 60%; dependent on the dose and delivery (supplement or topical) 80-90% Regrowth Rate 0-10%; potentially higher if used alongside other therapies 10%, alongside thickening of miniaturizing hair Side Effects 2%; more relegated to gastrointestinal distress than sexual side effects 1-25%; partly psychosomatic, but more severe than saw palmetto Otherwise, if you have any questions, please feel free to leave them in the comments section.
References
- https://perfecthairhealth.com/saw-palmetto-for-hair-growth/
- https://my.perfecthairhealth.com/courses/finasteride/
- Rossi A, Mari E, Scarno M, et al. Comparitive effectiveness of finasteride vs Serenoa repensin male androgenetic alopecia: a two-year study. Int J Immunopathol Pharmacol. 2012;25:1167–73.
- Suzuki, M., Ito, Y., Fujino, T. et al.Pharmacological effects of saw palmetto extract in the lower urinary tract. Acta Pharmacol Sin 30, 271–281 (2009) doi:10.1038/aps.2009.1
- McPartland J, Pruitt P. Benign prostatic hyperplasia treated with saw palmetto: a literature search and an experimental case study. J Am Osteopath Assoc2000;100(2):89–96.
- Carraro, J.‐C., Raynaud, J.‐P., Koch, G., Chisholm, G.D., Di Silverio, F., Teillac, P., Da Silva, F.C., Cauquil, J., Chopin, D.K., Hamdy, F.C., Hanus, M., Hauri, D., Kalinteris, A., Marencak, J., Perier, A. and Perrin, P. (1996), Comparison of phytotherapy (Permixon®) with finasteride in the treatment of benign prostate hyperplasia: A randomized international study of 1,098 patients. Prostate, 29: 231-240. doi:1002/(SICI)1097-0045(199610)29:4<231::AID-PROS4>3.0.CO;2-E
- Sinescu I, Geavlete P, Multescu R, Gangu C, Miclea F, Coman I, Ioiart I, Ambert V, Constantin T, Petrut B, Feciche B: Long-Term Efficacy of Serenoa repens Treatment in Patients with Mild and Moderate Symptomatic Benign Prostatic Hyperplasia. Urol Int 2011;86:284-289. doi: 10.1159/000322645
- https://www.rxlist.com/propecia-drug.htm#side_effects
- http://www.drproctor.com/propecia/propecia.pdf
Latanoprost is a medication that was originally developed to treat glaucoma. Over the last two decades, research groups have also started testing it as a treatment for hair loss – including alopecia areata, androgenic alopecia, and telogen effluvium.
In this ultimate guide, we’ll explore the evidence on latanoprost, dive into the mechanisms and clinical data regarding hair growth, and identify who might be a good candidate for this experimental intervention. We’ll also specify which formulations, dilutions, and usage frequencies are reaping the best results in terms of both efficacy and safety.
Finally, we’ll uncover critical caveats with its use – including lesser-discussed (but significant) safety risks – and where to get latanoprost (should you choose to incorporate it into your regrowth regimen).
Latanoprost: Key Takeaways
- Drug. Latanoprost is a drug that changes the activity of substances called prostaglandins. It was originally used to treat glaucoma, but after patients began reporting thicker eyelash hairs following its use, it has since been repurposed as a topical and tested on hair loss sufferers.
- Clinical data. Two clinical studies testing topical latanoprost for androgenic alopecia suggest that 0.1 mL x 0.1% latanoprost and 1-2 mL x 0.005% latanoprost improve hair counts, with visual improvements occurring in 30-50% of people over 6-8 months. While both formulations equate to ~0.1 mg of latanoprost exposure daily, preliminary data suggest that lower concentrations (i.e., 0.005%) are much more tolerable for patients, at least in terms of scalp irritation. For alopecia areata of the eyelashes, 3 clinical studies suggest that 0.005% latanoprost has no effect. For alopecia areata of the scalp, one clinical study suggests that 0.005% latanoprost may improve hair growth in ~25% of patients, but that treatments such as 0.05% betamethasone diproprionate reap superior efficacy.
- Safety. Clinical data on topical latanoprost indicate a strong safety profile when used at 1-2 mL x 0.005% to 0.01% daily. At higher concentrations and lower volumes (i.e., 0.1 mL x 0.1% latanoprost), one study found that 50% of patients reported symptoms of scalp irritation – suggesting that these formulations may evoke skin sensitivities in a relatively large number of people. Finally, none of the clinical studies on topical latanoprost for hair growth ran longer than 6-8 month. This is a problem, because long-term safety data are not yet established despite well-defined long-term risks, like pigmentation changes to the skin, eyes, and hair. Long-term clinical studies on latanoprost for glaucoma show that up to 10% of patients experience a darkening of their irises, with the effect beginning after one year of treatment. For these reasons, we have significant reservations about the long-term viability of latanoprost when used on the scalp – particularly for people who have lighter colors of skin / hair and who would prefer to not experience any hypopigmentation.
- Best practices. While topical latanoprost is a popular treatment amongst dermatologists and certain online hair loss circles, we have significant reservations about its long-term viability – mainly because of the unknown risks (but high potential) for hypopigmentation in the skin, hair, and (perhaps) eyes – depending on systemic leakage. If you decide to try topical latanoprost, it seems to be most appropriate for those with androgenic alopecia, and at dilutions of 1-2 mL x 0.005% latanoprost daily.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What is latanoprost?
Latanoprost is a drug developed to treat disorders of the eye – specifically, glaucoma. Originally formulated for delivery as eye drops, some users began reporting the elongation and/or regrowth of eyelash hairs.[1]https://jamanetwork.com/journals/jamaophthalmology/fullarticle/413134 Resultantly, researchers started investigating whether latanoprost – when formulated as a topical – might also enhance hair regrowth in other areas of the body, such as the scalp.
How does latanoprost work?
Latanoprost modulates levels of prostaglandin F2 – a substance our bodies produce that is derived from essential fatty acids (i.e., omega 6 fatty acids). More specifically, latanoprost stimulates the activity of the prostaglandin F2 alpha receptor. This allows for prostaglandin F2 levels to increase in cell sites, and when this occurs in eye tissues, it can reduce (or alleviate) ocular pressure and thereby improve cases of glaucoma.[2]https://www.ncbi.nlm.nih.gov/books/NBK540978/
How might latanoprost regrow hair?
There’s evidence that the eyelash hair regrowth reported by many latanoprost users isn’t just happenstance. For instance, as prostaglandin F2 activity increases…
- Microvascular networks tend to widen, thereby improving blood, oxygen, and nutrient transport to hair follicles affected by hair loss.
- Mitosis (i.e., cellular division and proliferation) increases. When more hair follicle related cells divide, more cells can contribute to hair follicle growth overall. [3]https://pubmed.ncbi.nlm.nih.gov/15854125/
- Prostaglandin D2 activity may simultaneously decrease – because precursor molecules for prostaglandins start shifting toward prostaglandin F2 alpha production and away from prostaglandin D2 production.[4]https://www.frontiersin.org/articles/10.3389/fphys.2020.594313/full This is relevant because some evidence implicates prostaglandin D2 as a contributor to the balding process, so lowering its activity may help improve hair growth.[5]https://pubmed.ncbi.nlm.nih.gov/33854354/
Interestingly, many of the above mechanisms (i.e., improved vasodilation and prostaglandin modification) overlap with the suspected mechanisms of the FDA-approved hair loss drug, topical minoxidil. As such, it’s no surprise that in a case series on 300+ patients using eye drops of latanoprost to treat glaucoma, 77% saw hypertrichosis (i.e., additional hair growth) in the areas surrounding the eyes. [6]https://www.jaad.org/article/S0190-9622(01)70206-X/fulltext
With all of these cases, it’s also not out-of-the-question that latanoprost might also help regrow some scalp hair if reformulated as a topical.
In fact, this was corroborated by a 2002 study on primates (i.e., stump-tailed macaques) who happen to be one of the only other species to also suffer from pattern hair loss, or androgenic alopecia. Over an 8-month study, monkeys receiving a topical application of 0.5 ml x 0.005% to 0.05% latanoprost daily showed improvements to hair loss and cosmetic degrees of hair regrowth.
Fig. 1.Frontal bald scalp of a 10-year-old, female, macaque, showing sparce short vellus hairs in the bald scalp at pretreatment time (a). After 5 months of treatment with latanoprost 50 mg/ml, thickness and density of hair increased in upper central and lower lateral regions (b). After 3 months of latanoprost 500 ug/ml thickness and density of hair significantly increased in mid and lower lateral region.
Fig. 2.Sequential changes of average scores of categorical grades of hair growth in latanoprost and vehicle-treated monkeys.
The anecdotes on eyelash hair growth in glaucoma patients – and the preliminary evidence of hair regrowth on balding monkeys – certainly provides a strong enough signal to further investigate topical latanoprost as a hair loss treatment in humans.
So, over the last 20 years, what does the clinical evidence on latanoprost show?
Human evidence: topical latanoprost for the treatment of hair loss
While the data are limited (so far), clinical studies on latanoprost for hair loss have been conducted on both androgenic alopecia, telogen effluvium, and alopecia areata.
Clinical study on androgenic alopecia (2011)
In its first-ever randomized, double-blinded, placebo-controlled clinical trial, researchers tested topical latanoprost on 16 men with early-stage androgenic alopecia.
Each participant received two drops of 0.1% latanoprost daily – one drop per receding temple region – equating to 0.1 mL of solution applied daily. In other words, each man applied 0.1 mg of latanoprost per day.[7]https://pubmed.ncbi.nlm.nih.gov/21875758/
Fig 1. Male subject with androgenetic alopecia (Norwood Hamilton scale grade III). Marked areas indicate minizone position for topical application of latanoprost and placebo solutions.
2011 clinical results
Over the course of 24 weeks, daily application of 0.1 mg of latanoprost…
- Improved terminal hair counts and hair density versus placebo
- Improved the ratio of anagen:telogen hairs versus placebo (i.e., growing vs. shedding hairs)
- Did not cause any serious adverse events
The results were reflected statistically through phototrichogram data (i.e. the gold-standard for clinical research on hair loss disorders).
Fig 3. Number and percentage of anagen and telogen hairs/cm2 at baseline and 24 weeks. [8]https://pubmed.ncbi.nlm.nih.gov/21875758/
While the data seem encouraging, it’s important to point out three caveats. Across all latanoprost users:
- 8 of 16 men did report skin irritation-like effects – such as dermatitis. So, the risk of mild side effects were relatively high (i.e., 50%).
- 50% of men demonstrated what investigators called a “clinical improvement”. In other words, they saw improvements in at least two criteria points on the phototrichogram: increased hair density, increased hair counts, etc.
- Of the remaining men, 44% demonstrated no response, and 6% demonstrated a “poor” response – meaning things got worse for them versus placebo.
In other words, latanoprost seemed to work well in half of participants, with the other half of participants showing hair loss stabilization or no response at all.
For what it’s worth, this is a common trait among hair loss drugs that target factors like histamine responses and/or prostaglandins – for instance, minoxidil and cetirizine. [9]https://perfecthairhealth.com/histamine-and-hair-loss-is-there-a-use-for-antihistamines/ For some, these drugs work wonders. For others, they see no effect. Why?
In our estimation, the dichotomies in responses are likely related to the following:
- The degree of inflammation in the scalp – as studies have shown inflamed scalps respond more poorly to topicals such as minoxidil[10]https://pubmed.ncbi.nlm.nih.gov/8496421/
- The level of histamine activity surrounding hair follicles – as this problem may contribute to some cases of androgenic alopecia, but not all[11]https://perfecthairhealth.com/histamine-and-hair-loss-is-there-a-use-for-antihistamines/
- The activation of these compounds – in the case of minoxidil, its sulfation (which can be improved with retinoic acid, microneedling, and/or switching to oral minoxidil)[12]https://my.perfecthairhealth.com/courses/minoxidil/topical-minoxidil/
Therefore, this initial (albeit, small) clinical study on topical latanoprost suggests that it might have a place in treating some cases of hair loss, but not all.
Clinical study on androgenic alopecia and telogen effluvium (2018)
In 2018, another research group investigated the use of topical latanoprost by itself (and combined with topical minoxidil) in the treatment of men and women with androgenic alopecia or telogen effluvium.[13]http://www.surgicalcosmetic.org.br/details/618/en-US/latanoprost-and-minoxidil–comparative-double-blind–placebo-controlled-study-for-the-treatment-of-hair-loss
First, the researchers split 123 participants into six groups, with each group testing one of the following:
- G1: Placebo (i.e., nothing)
- G2: 5% minoxidil
- G3: 5% minoxidil + 0.005% latanoprost
- G4: 0.005% latanoprost
- G5: 5% minoxidil + 0.01% latanoprost
- G6: 0.01% latanoprost
Unfortunately, the investigators didn’t specify how much solution each participant applied once daily. Based on the photos of participants featured inside their study, many participants had moderate levels of hair loss and beyond, which would likely require at least 1-2 mL of solution to cover all balding regions. So, we can only make rough estimations as to how much daily exposure of these drugs each participant received.
Note: the absence of daily drug exposure volume is just one of the many flaws in this study. We’ll uncover more issues soon.
2018 clinical results
Over the course of 180-240 days, 25 of the 123 people withdrew from the study for reasons that the investigators state were “not related to the research” – though they don’t specify what those reasons were. Withdrawal rates were mostly equivalent across all six groups, which leaves us with 98 people and 16-18 participants per group for statistical comparisons.
Among those who remained, here are the key findings:
- No side effects were reported, with no significant signs of dermatitis or scalp irritation for any participant across all treatment groups.
- These groups saw statistically significant hair count improvements versus placebo:
- G2: 5% minoxidil
- G3: 5% minoxidil + 0.005% latanoprost
- G4: 0.005% latanoprost
- G5: 5% minoxidil + 0.01% latanoprost
- These groups did not see hair count improvements versus placebo:
- G6: 0.01% latanoprost
- The response rates (i.e., the percent of people seeing visually cosmetic regrowth) were:
- G2: 5% minoxidil – 35%
- G3: 5% minoxidil + 0.005% latanoprost – 36%
- G4: 0.005% latanoprost – 19%
- G5: 5% minoxidil + 0.01% latanoprost – 6%
- G6: 0.01% latanoprost – 0%
Here are some photos the investigators featured of participants using 5% minoxidil and 0.005% latanoprost – albeit from an extension period that ran up to 240 days:
Figure 1: Macro image of a female participant at the start and end periods (treatment with 5% minoxidil + 0.005% latanoprost)
Figure 2: Macro image of a male participant at the start and end periods (treatment with 5% minoxidil + 0.005% latanoprost)
And here is data regarding anagen hair counts (i.e., the number of “growing” hairs) across all groups and time periods (day 2, day 92, and day 182):
Graph 2: Number of anagen hairs by period and by treatment
What can we glean from this 2018 study?
First, let’s start off by interpreting a good signal from this study.
Topical formulations of latanoprost that are of higher volume and lower concentration might greatly minimize the risk of side effects.
If we recall from the 2011 study on topical latanoprost, two drops of 0.1% latanoprost totaling 0.1 mL of daily use led to skin irritation-related side effects in 50% of participants. But in this study on 0.005% to 0.01% latanoprost applied daily at 1-2 mL of volume, no adverse events were reported.
It might interest you to know that both of these studies likely exposed participants to the exact same total amount of latanoprost each day. It’s just that the 2011 study used a higher concentration (i.e., 0.1%) but with less volume of topical (i.e., 0.1 mL), whereas the 2018 study used a 10-20x lower concentration (i.e., 0.005% to 0.01%) but with 10-20x more volume (i.e., 1-2 mL).
Yet mathematically, both studies exposed their participants to ~0.1mg of latanoprost daily.
This is important, because it suggests that 0.1mg of latanoprost may become a lot more tolerable for hair loss patients when it is spread out over a larger area. So, if nothing else comes from this study, we’re at least encouraged by this signal.
So what else can we surmise?
1-2 mL daily of 0.005% latanoprost may improve hair counts for a portion of people with androgenic alopecia and/or telogen effluvium… particularly if combined with 5% minoxidil
According to the data presented by the research team, 0.005% latanoprost by itself was effective at improving hair parameters, as was 0.005% latanoprost + 5% minoxidil.
However, we need to keep in mind the response rates here: just 35% to 36% of participants in these groups saw visual regrowth. That’s not a lot, and it gives credence to the belief that while latanoprost might work for some people, it’s probably not appropriate for all hair loss sufferers.
What shouldn’t we glean from this 2018 study?
Looking at all of the data from this study can be confusing. After all, we have 6 different groups statistically measured across one another, with varying methodologies applied for some groups (like extension time periods for those using 0.005% latanoprost + 5% minoxidil), and with poor clarity in writing regarding both results and discussion organizations.
Nonetheless, we’ve read online about some people presuming that this study gives us enough insights to include the following. So, before adopting similar opinions, please read our counterarguments below:
Fallacy #1: this study suggests that 0.01% latanoprost might be less effective than 0.005% latanoprost
It’s not abnormal to make this assumption given the data presented. After all, the groups using 0.005% latanoprost saw a 35% and 36% response rate, whereas the groups using 0.01% latanoprost saw a 6% and 0% response rate.
Moreover, it’s not unusual to see dose-dependent effects in medicine – where some drugs have a strong therapeutic benefit at lower concentrations but can have opposing or toxic effects at higher concentrations. We discuss these relationships in our guide on flutamide.
Nonetheless, there are significant methodological flaws in this study that do not allow us to jump to this conclusion. Here are a few:
- Participant dropout rates were nearly 20%. This increases the likelihood of survivorship bias, whereby those who are left at the end of the study are only there because they actually are seeing an effect… while those who were seeing no benefit quit. This phenomenon artificially enhances “response rates”, and while it can be controlled for statistically using Bayesian modeling, the investigators did not do this.
- There was an unknown number of telogen effluvium participants. Telogen effluvium is a type of hair loss that differs from androgenic alopecia. It’s often caused by stress, micronutrient imbalances, and/or chronic conditions like hypothyroidism. It also self-resolves given enough time away from a stressor, or after the micronutrient imbalance or chronic condition is corrected. This study includes an unknown number of telogen effluvium patients, and no data on how many were in each group. With group sizes of just 16-18 people per treatment, keep in mind that all it would take would be 5-6 people “recovering” from telogen effluvium (which is likely not treatable through the mechanisms suspected from latanoprost) to hit a 35% “response rate”. That’s a huge problem… especially because the investigators don’t specify their method of randomization – which means there could be a disproportionate number of telogen effluvium sufferers in one of these groups – either artificially stuffing down or inflating their response rates.
- Participants were included if they had quit other hair loss treatments only 4+ weeks prior. Multiple studies definitively show that abruptly quitting hair loss topicals, such as minoxidil, results in a telogen effluvium-based shed that can take upwards of 6+ months from which to fully recover back to baseline. That’s the entire duration of this study. As such, any study that includes participants who just quit other treatments or topicals only 4+ weeks ago could be capturing a huge percentage of participants who happen to be entering into a telogen effluvium shed due to quitting those treatments… which would obfuscate any hair count results from introducing a new treatment during the same period.
As such, we need to be incredibly careful about drawing any efficacy or comparison conclusions from this study. It’s entirely possible that 0.005% and 0.01% latanoprost are equally effective… but that the groups receiving higher dilutions of latanoprost had poorer results because of participant sampling. Without better data, we just don’t know.
As an aside, when discussing this study across our own team, we were even apprehensive to mention the positive safety signal – as this 2018 study is so poorly designed that any conclusions whatsoever are potentially problematic and/or at-odds with the real-world experiences of latanoprost users.
Nonetheless, we have to work with the data currently available, and these two studies on topical latanoprost are all that we’ve got (so far) for androgenic alopecia.
So, what about other hair loss disorders, such as alopecia areata?
Clinical studies: topical latanoprost for the treatment of alopecia areata
To date, there have been five publications (to which we could find) assessing topical latanoprost for alopecia areata. Here are their quick summaries and results:
Alopecia areata of the eyelashes and eyebrows
A 2003 case report details the experience of an 11-year old female who experienced alopecia areata of the eyelashes after recovering from a viral illness. After multiple failed treatments, doctors eventually tested topical latanoprost – at an unknown dilution – which was applied daily for a period of six months. During that time, photographic assessments show a near full recovery of all lost eyelashes.[14]https://pubmed.ncbi.nlm.nih.gov/12724722/
A 2009 clinical study tested 0.005% latanoprost – at an unknown volume – on a total of 26 men and women with alopecia areata of the eyebrows and eyelashes. Participants applied latanoprost to just one side of the face, with the other side acting as an intra-patient “control”. After four months, researchers saw partial hair recovery in just one patient, and concluded that topical latanoprost was no more effective than applying nothing at all. Aside from a transient headache fro one patient, no adverse events were reported.[15]https://pubmed.ncbi.nlm.nih.gov/19620039/
Interestingly, these results were mirrored by prior clinical studies done on similar dilutions of topical latanoprost for eyelash-related alopecia areata from 2005 and 2008.[16]https://pubmed.ncbi.nlm.nih.gov/16310083/[17]http://www.iranjd.ir/article_98048_64fda5d6552e3e9d714cc2162d5686a4.pdf As such, it appears that the data (so far) suggest topical latanoprost does not improve this specific hair loss disorder for the overwhelming majority of people trying it.
Alopecia areata of the scalp
Finally, a 2021 randomized clinical study tested daily applications of 0.005% latanoprost versus 0.05% betamethasone diproprionate (an autoimmune-related medication) for the treatment of scalp-related alopecia areata in 50 patients.[18]https://ijdvl.com/a-randomized-comparative-study-of-the-efficacy-of-topical-latanoprost-versus-topical-betamethasone-diproprionate-lotion-in-the-treatment-of-localized-alopecia-areata/
Unfortunately, the researchers did not specify the amount (in mL) of topical applied daily. As such, we cannot estimate daily exposure volumes.
Over a 16-week period, 24% of patients using 0.005% latanoprost saw improvements, whereas 56% of patients using 0.05% betamethasone diproprionate saw improvements. In fact, patients in the 0.05% betamethasone diproprionate group also tended to see a faster recovery and a more dramatic reduction of alopecia areata lesions.
Therefore, the researchers concluded that 0.05% betamethasone diproprionate was superior to 0.005% latanoprost in the treatment of scalp alopecia areata.
Topical latanoprost for hair loss: summaries of the clinical studies
From what we can glean across all clinical studies on topical latanoprost for hair loss:
- Androgenic alopecia / pattern hair loss: When applied daily at a total exposure volume of 0.1 mg, topical latanoprost seems to improve 30-50% of androgenic alopecia cases over a six-month period. Concentrations of 1-2 mL x 0.005% latanoprost are preferred, particularly when combined with 5% minoxidil. Moreover, 1-2 mL x 0.005%-0.01% latanoprost seems to be a safer formulation than 0.1 mL x 0.1% latanoprost – even though both formulations equate to a 0.1 mg daily exposure volume.
- Alopecia areata of the eyelashes: Currently, the totality of evidence suggests that 0.005% latanoprost at undisclosed daily usage volumes does not work, minus one or two case reports.
- Alopecia areata of the scalp: One clinical study shows that 0.005% latanoprost improves alopecia areata of the scalp in ~25% of participants, but that its response rate is just half that of 0.05% betamethasone diproprionate (another autoimmune treatment), and that patients using 0.05% betamethasone diproprionate respond more quickly versus 0.005% latanoprost.
What about safety?
It’s easy to look at these studies and note that dilutions of 1-2 mL daily of 0.005% to 0.01% were well-tolerated for nearly all participants, with only a handful of minor, transient adverse events reported across all patient cohorts testing this formulation. As such, it’s easy to presume that topical latanoprost must be safe.
We have significant reservations with that conclusion.
After all, these topical latanoprost studies for hair growth ran – at most – 8 months. While no serious adverse events were reported during these short timeframes, there is a well-known phenomenon that occurs from long-term latanoprost use, and one that does not typically show up until after one full year of treatment: pigment changes.
In glaucoma patients, pigment changes of the iris begin to occur after one year of daily latanoprost use, and they worsen over time – affecting up to 10% of glaucoma patients.[19]https://www.ncbi.nlm.nih.gov/books/NBK540978/ Other studies suggest an incidence as high as 70%, depending on the eye color & duration of use.[20]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1771317/
This is because regular latanoprost use increases melanin activity (i.e., the activity of cells that produce pigmentation in our skin, eyes, etc.). The effects, over time, can be quite dramatic. Just see the results from this patient, who was treated for glaucoma in their right eye with latanoprost (picture A) but not in their left eye (picture B). It’s as if we are looking at two different eyes. [21]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1771317/
As such, a critical question remains: does latanoprost applied topically produce the same effects in the skin, hair, and (potentially) through the eyes via systemic absorption?
Unfortunately, we have no way of knowing… because the clinical studies have not yet been conducted. Moreover, none of the studies on topical latanoprost for hair loss ran even remotely long enough to measure if this effect would also apply to the scalp.
In cases of treatments producing serious, cosmetically-unwanted adverse events – like a darkening of scalp skin – we tend to err on the side of caution and say that without more data, most people should be seriously cautious about long-term use of topical latanoprost. Perhaps better data will demonstrate this is not a concern, but until we have that data, we don’t feel comfortable recommending topical latanoprost as a guinea pig experiment to members here… even despite its popularity online as an adjuvant hair loss treatment.
For more contraindications and side effects from latanoprost, please visit this resource.[22]https://www.ncbi.nlm.nih.gov/books/NBK540978/
Latanoprost: best and worst candidates
You are likely a candidate for latanoprost if:
- You have androgenic alopecia
- You are planning to use latanoprost alongside better-studied treatments (and not as a standalone treatment)
- You are not concerned about the unknown risks of pigmentation changes that tend to start presenting after 1+ years of latanoprost use
You are likely not a candidate for latanoprost if:
- You don’t have androgenic alopecia
- You are concerned about the unknown risks of pigmentation changes that tend to start presenting after 1+ years of latanoprost use
Summary
Latanoprost has some limited clinical data supporting its use for androgenic alopecia, but with response rates of 30-50%, it’s unlikely that this treatment is right for most hair loss sufferers. For these reasons, we recommend people use other prostaglandin analogues instead – such as minoxidil – and only resort to latanoprost if they’re using it as an add-on to their regimen and if they’re comfortable with the unknowns about its long-term safety profile.
For alopecia areata of the eyelashes, latanoprost is unlikely to help. For alopecia areata of the scalp, one small study found that topical latanoprost may help up to 25% of people. However, that same study also found that 0.05% betamethasone diproprionate was twice as effective at producing a response rate versus latanoprost, and that it also worked faster than latanoprost.
References[+]
References ↑1 https://jamanetwork.com/journals/jamaophthalmology/fullarticle/413134 ↑2, ↑19, ↑22 https://www.ncbi.nlm.nih.gov/books/NBK540978/ ↑3 https://pubmed.ncbi.nlm.nih.gov/15854125/ ↑4 https://www.frontiersin.org/articles/10.3389/fphys.2020.594313/full ↑5 https://pubmed.ncbi.nlm.nih.gov/33854354/ ↑6 https://www.jaad.org/article/S0190-9622(01)70206-X/fulltext ↑7, ↑8 https://pubmed.ncbi.nlm.nih.gov/21875758/ ↑9, ↑11 https://perfecthairhealth.com/histamine-and-hair-loss-is-there-a-use-for-antihistamines/ ↑10 https://pubmed.ncbi.nlm.nih.gov/8496421/ ↑12 https://my.perfecthairhealth.com/courses/minoxidil/topical-minoxidil/ ↑13 http://www.surgicalcosmetic.org.br/details/618/en-US/latanoprost-and-minoxidil–comparative-double-blind–placebo-controlled-study-for-the-treatment-of-hair-loss ↑14 https://pubmed.ncbi.nlm.nih.gov/12724722/ ↑15 https://pubmed.ncbi.nlm.nih.gov/19620039/ ↑16 https://pubmed.ncbi.nlm.nih.gov/16310083/ ↑17 http://www.iranjd.ir/article_98048_64fda5d6552e3e9d714cc2162d5686a4.pdf ↑18 https://ijdvl.com/a-randomized-comparative-study-of-the-efficacy-of-topical-latanoprost-versus-topical-betamethasone-diproprionate-lotion-in-the-treatment-of-localized-alopecia-areata/ ↑20, ↑21 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1771317/ Introduction
Finasteride is one of the most commonly-prescribed medications for treatment of male pattern hair loss—also known as androgenic alopecia (AGA). But it’s also used as an off-label treatment for female pattern hair loss. Evidence suggests this medication can help regrow hair in both sexes.
But what’s the best dose of finasteride for women with AGA? Unfortunately, it’s complicated. This article sets out to evaluate the data, uncover the answers, and provide recommendations based on the current landscape of clinical research.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What Is Finasteride?
Finasteride is a drug developed to inhibit type II 5-alpha reductase. This is an enzyme in the body that converts free testosterone in dihydrotesterone (DHT).
Essentially, finasteride lowers DHT levels by reducing the amount of type II 5-alpha reductase circulating throughout our bodies. And by taking finasteride at 0.2 to 5.0 mg daily dosages, we can often reduce total DHT levels by 70%. [1]https://www.ncbi.nlm.nih.gov/books/NBK513329/
Why Use a Drug To Reduce DHT?
DHT is not just a metabolite of testosterone; it’s also the primary male hormone causally associated with androgenic alopecia.
We know this because studies have shown that men who cannot produce DHT are nearly fully-protected from going bald throughout a lifetime. Furthermore, clinical studies on DHT-lowering drugs – such as finasteride – show that if DHT levels are suppressed enough, 80-90% of men can arrest the progression of their pattern hair loss and even regrow 10-20% of their lost hair. [2]https://pubmed.ncbi.nlm.nih.gov/29407002/ [3]https://www.ncbi.nlm.nih.gov/books/NBK430924/
Similarly, studies on females with androgenic alopecia have shown that finasteride can also improve their hair loss outcomes. The evidence is less robust than for men, but finasteride is something many women should consider trying in order to improve their pattern hair loss.
Finasteride for Women: What’s the Perfect Dose?
In men, the FDA has approved the use of 1mg of finasteride for pattern hair loss. However, male and female hair loss cases are not always the same. Reducing DHT levels is often of therapeutic interest to fighting AGA – and for both sexes – but some clinical evidence suggests that females might need a different dose of finasteride versus males.
How Much DHT Does Finasteride Reduce?
Finasteride has what is known as a dose-dependent, logarithmic response curve for DHT reduction. In other words: a little bit of finasteride reduces nearly as much DHT as a lot of finasteride. For an example, see this chart:
Clinical studies have demonstrated that 0.2 mg and 5.0 mg reduce nearly the same amount of finasteride: 69% versus 72%, respectively.
Because of this, a lot of people actually prefer to use lower dosages of finasteride than what is generally prescribed. This practice is also supported by clinical data. For instance, in men, 0.2mg of finasteride AGA at a statistically similar level as 1.0 mg of finasteride over the course of a year. [4]https://europepmc.org/article/med/15319158
But is the same true with females? Unfortunately, the data is less clear.
Finasteride for Female Pattern Hair Loss: the Clinical Evidence
The FDA approves the use of 1 mg of finasteride for male pattern hair loss.[5]https://www.fda.gov/drugs/information-drug-class/5-alpha-reductase-inhibitor-information But the underlying causes of male and female pattern hair loss cases (androgens such as DHT) are not always the same. Furthermore, while reducing DHT levels is of therapeutic relevance in treating AGA in men and women, some clinical evidence suggests that females might need a different dose of finasteride versus males.
What does the available research tell us? Here are a few of the key studies on finasteride for women, and their main findings (summarized in the table below).
Finasteride for Women: Key Studies
Type of study Number of patients Patient condition Finasteride dosage Length of treatment Assessment parameters Outcomes Reference Double-blind, randomized control trial 67 treated, 70 placebo AGA, post-menopausal, normal serum testosterone 1 mg, daily 12 months Hair counts, photographic assessment, self-assessment, scalp biopsies Serum DHT reduction but no effect on hair loss outcomes compared to placebo [6]https://pubmed.ncbi.nlm.nih.gov/10674382/[7]https://pubmed.ncbi.nlm.nih.gov/11050579/ Randomized, unmasked trial 12 finasteride, 12 flutamide, 12 cyproterone acetate with estradiol, 12 no treatment 48 women, hyperandrogenic, age- and weight matched controls 5 mg, daily 12 months Ludwig classification15 of female hair loss, self-assessment, and investigator assessment No effect with finasteride [8]https://pubmed.ncbi.nlm.nih.gov/11050579/ Single-blind, placebo-controlled trial 24 female patients included AGA, age 23-38 years (mean 33) 1 ml topical application (0.005%), twice daily to affected area 16 months Semi-quantitative investigator assessment, hair shedding quantification, self-assessment Hair count and hair density improvements versus placebo (data not sex-stratified) [9]https://www.tandfonline.com/doi/abs/10.3109/09546639709160517 Small trial 5 treated Post-menopausal, normal androgen levels 2.5 or 5 mg, daily 18 months, review every 6 months Self-assessment, investigator assessment, photographic assessment Overall improvement [10]https://pubmed.ncbi.nlm.nih.gov/15459533/ Small trial 10 treated Post-menopausal 1 mg, daily 52-82 weeks Self-assessment and photographic assessment Overall improvement (9/10 patients) [11]Ahn J, Cho SB, Kim MN, Ro BI. Finasteride treatment of female patterned hair loss in postmenopausal women. Korean J Dermatol. 2006;44:1094-1097. Small trial 6 treated AGA, age 30-76 years (mean 46.5), normal androgen levels 5 mg, daily Weeks (not specified) Retrospective questionnaire (self-assessment) Overall improvement (5/6 patients) [12]https://pubmed.ncbi.nlm.nih.gov/17454167/ Small trial 37 treated Female pattern hair loss, pre-menopausal, age 19-50 years (mean, 33.7) 2.5 mg, daily (+ oral contraceptive drospirenone and ethinyl estradiol) 12 months Self-assessment, photographic assessment, and hair-density scoring Overall improvement by self-assessment (29/37), photographic improvement ((23/37), significant hair density improvement (12/37) [13]https://pubmed.ncbi.nlm.nih.gov/16549704/ Small trial 41 treated AGA, persistent adrenarche syndrome 2.5 mg, daily (+ ethinyl estradiol) 2 years Not specified Overall improvement [14]https://pubmed.ncbi.nlm.nih.gov/19341939/ Small trial 4 treated 36, 40, 60, and 66 years old, elevated testosterone and hyperandrogenism 1.25 mg, daily 6 months to 2.5 years Photographic assessment and self-assessment Stabilization of hair loss within 6-12 months, hair growth improvements in 6 months – 2.5 years [15]https://pubmed.ncbi.nlm.nih.gov/12399766/ Case study 1 treated 47-year-old, ‘male’ pattern hair loss, hysterectomy and ovariectomy, long-term hormone replacement 2.5 mg, daily (+continued testosterone supplementation) 10 months Photographic assessment Hair loss stabilization at 6 months, hair growth improvement at 10 months [16]Hong JB, Chiu HC, Chan JY, Chen RJ, Lin SJ. A woman with iatrogenic androgenetic alopecia responding to finasteride. Br J Dermatol. 2007;156(4):754-755. doi:10.1111/j.1365-2133.2006.07719.x Case study 1 treated 67-year-old, 18 month history of hair thinning, normal androgen levels 5 mg, weekly 12 months Self-assessment and photographic assessment Improvement, hair regrowth [17]https://pubmed.ncbi.nlm.nih.gov/12366441/ Case study 1 treated 51-year-old, 8 month history of hair thinning, normal androgen levels 1 mg, daily 12-13 months Hair density measurements Hair density increased versus baseline [18]https://pubmed.ncbi.nlm.nih.gov/15844649/ Finasteride for Women: Key Studies Takeaway
There is conflicting data regarding the efficacy of finasteride for female pattern hair loss. Some studies report improvements while others do not.
There are some key variables to consider when weighing the available evidence:
- The dose of finasteride used
- The length and frequency of treatment
- Oral administration versus a topical treatment
- Whether finasteride was used in conjunction with other drugs or therapies
- The type of hair loss in the patient groups (e.g., female pattern hair loss versus age-related thinning – and was this accurately determined?)
- Patient age, history, and status (e.g., pre-, or post-menopausal, or abnormal androgen levels)
- How the treatment was assessed (e.g., quantitative hair counting versus patient self-assessment)
- Whether the study contained appropriate controls (i.e., placebo-receiving patients, ideally matched for age, weight and medical history)
- The number of patients in the study (which affects the statistical power; was the study a case report of one patient, or a larger study with a control group?).
Interpreting research data can be difficult and confusing. such as different studies using different dosages of finasteride and for varying lengths of time, measuring different hair loss outcomes, and using different numbers and ages of patients.
The specific type of hair loss is also a crucial variable.[19]https://pubmed.ncbi.nlm.nih.gov/30604525/ Often, studies reporting positive outcomes are based on patient self-reporting, which can be suspect and not meaningfully objective or quantitative in measuring true prevention or reversal of hair loss.
What is the best dose? Explaining the conflicting results.
Given the dose-response relationship between finasteride and DHT levels, shouldn’t 1 mg be as effective as 5 mg? Why aren’t women getting consistent regrowth across doses within these ranges?
Other discrepancies are the time for which finasteride was given. In men, 1 mg of finasteride can be effective in 6-12 months, but it is possible that women require more long-term treatment regimens.[20]https://pubmed.ncbi.nlm.nih.gov/30604525/ Generally, success with finasteride in women has been reported in both the short- and long-term.[21]https://pubmed.ncbi.nlm.nih.gov/12399766/
Alternatively, the difference in observed efficacies between studies may be due to patient background and the type of hair loss. Hair loss in patients suffering from PCOS or adrenarche (i.e., high levels of adrenal gland activity) likely has a clear causal link to abnormal androgen signaling (and therefore suitable for finasteride), where hair loss in post-menopausal women may be more akin to age-related hair ‘thinning’, and not linked to testosterone or DHT, which may explain why some studies find no effect with finasteride.[22]https://pubmed.ncbi.nlm.nih.gov/10674382/[23]https://pubmed.ncbi.nlm.nih.gov/11050579/[24]https://pubmed.ncbi.nlm.nih.gov/12399766/
That said, finasteride is also reportedly effective in patients that are androgen-normal.20 Therefore, there needs to be more careful classification of the type of hair loss and the likely underlying mechanisms, as well as clear standardization of treatment outcome measurements.
Conclusions
Does finasteride work for women suffering from hair loss? If so, what is the ideal dosage?
Finasteride is most beneficial for women when their hair loss occurs alongside elevated androgen levels—much like hair loss in men.
It is likely that DHT is a major factor in a proportion of female hair loss cases. Finasteride is likely to be a beneficial part of a successful hair loss regimen in these cases. However, it is not clear that male and female pattern hair loss universally share the same underlying cause.
Finasteride may be less suitable in contexts of age-related hair thinning. Hair follicles are sensitive to all manner of different hormones and chemical signals, not just androgens such as testosterone and DHT.[25]https://pubmed.ncbi.nlm.nih.gov/32731328/
The best dose is one that maximizes the desired benefits (prevention and reversion of hair loss) while minimizing any undesirable side effects.
Finasteride can be effective at reducing DHT even in small doses. Because of this, experimentation with low levels of finasteride may lead to results with far less exposure to chemicals. Topical treatments may also be considered to further reduce systemic side effects.
Therefore, the best dose will be patient-subjective. Prior consideration of your history, goals regarding hair loss, and experimentation with dosages is needed before you will find the ideal routine for your hair health.
References[+]
References ↑1 https://www.ncbi.nlm.nih.gov/books/NBK513329/ ↑2 https://pubmed.ncbi.nlm.nih.gov/29407002/ ↑3 https://www.ncbi.nlm.nih.gov/books/NBK430924/ ↑4 https://europepmc.org/article/med/15319158 ↑5 https://www.fda.gov/drugs/information-drug-class/5-alpha-reductase-inhibitor-information ↑6, ↑22 https://pubmed.ncbi.nlm.nih.gov/10674382/ ↑7, ↑8, ↑23 https://pubmed.ncbi.nlm.nih.gov/11050579/ ↑9 https://www.tandfonline.com/doi/abs/10.3109/09546639709160517 ↑10 https://pubmed.ncbi.nlm.nih.gov/15459533/ ↑11 Ahn J, Cho SB, Kim MN, Ro BI. Finasteride treatment of female patterned hair loss in postmenopausal women. Korean J Dermatol. 2006;44:1094-1097. ↑12 https://pubmed.ncbi.nlm.nih.gov/17454167/ ↑13 https://pubmed.ncbi.nlm.nih.gov/16549704/ ↑14 https://pubmed.ncbi.nlm.nih.gov/19341939/ ↑15, ↑21, ↑24 https://pubmed.ncbi.nlm.nih.gov/12399766/ ↑16 Hong JB, Chiu HC, Chan JY, Chen RJ, Lin SJ. A woman with iatrogenic androgenetic alopecia responding to finasteride. Br J Dermatol. 2007;156(4):754-755. doi:10.1111/j.1365-2133.2006.07719.x ↑17 https://pubmed.ncbi.nlm.nih.gov/12366441/ ↑18 https://pubmed.ncbi.nlm.nih.gov/15844649/ ↑19, ↑20 https://pubmed.ncbi.nlm.nih.gov/30604525/ ↑25 https://pubmed.ncbi.nlm.nih.gov/32731328/ Topical finasteride has emerged to meet demand for hair loss treatments that promise strong results with fewer systemic side effects than oral finasteride by concentrating action at the scalp while reducing overall drug exposure. Early studies suggest it can improve hair growth and lower scalp dihydrotestosterone (DHT) with less systemic absorption, but side effects are not eliminated, and long-term head-to-head data versus oral finasteride remain limited.
This article will cut through the “same results, fewer side effects” marketing narrative and set realistic expectations by focusing on what current clinical evidence and real-world user patterns actually support.
How Does Finasteride Work?
Finasteride is a 5-alpha reductase inhibitor, meaning that it blocks this enzyme from converting testosterone into DHT, the more potent androgen that drives miniaturization of sensitive scalp follicles in androgenic alopecia (AGA). The enzyme catalyzes this conversion in the scalp, skin, liver, and prostate. By inhibiting it, finasteride lowers both local (scalp/prostate) and circulating DHT.[1]Zito PM, Bistas KG, Patel P, et al. Finasteride. [Updated 2024 Feb 28]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: … Continue reading
Finasteride structure
Why Oral Finasteride is Systemic
A standard 1 mg oral dose reduces DHT by roughly 60-70%, with some studies reporting around 70% suppression at steady state, and intraprostatic DHT falls by about 90%.[2]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading,[3]Smith, A.B., Carson, C.C. (2009). Finasteride in the treatment of patients with benign prostatic hyperplasia: a review. Therapeutics and Clinical Risk Management. 5. 535-545. Available at: … Continue reading Because 5 alpha-reductase is expressed in multiple organs, this systemic inhibition affects DHT signaling throughout the body, not only in the scalp.
Why Side Effects Occur
Sexual and mood-related side effects are not purely “in the mind”; they reflect altered androgen physiology in a subset of men whose tissues are more sensitive to a DHT drop, even though testosterone usually stays within the normal range or rises modestly. Reduced DHT can influence libido, erectile function, ejaculate volume, and, in some individuals, mood or energy, consistent with the hormone’s broader role beyond hair follicles.[4]Traish, A.M. (2017). Negative Impact of Testosterone Deficiency and 5ɑ-Reductase Inhibitors Therapy on Metabolic and Sexual Function in Men. Advances in experimental medicine and biology. 1043. … Continue reading
Topical Targeting vs. Spillover
Topical finasteride aims to concentrate drug action in the scalp by delivering finasteride directly to hair follicles, potentially lowering local DHT while limiting how much drug reaches the bloodstream. However, percutaneous absorption still occurs, and pharmacokinetic studies do show measurable reductions in serum DHT with topical formulations, meaning systemic exposure is reduced compared to oral dosing, but not eliminated.[5]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading
What the Research Says About Topical Finasteride Side Effects
The evidence on topical finasteride’s side effect profile shows a nuanced picture – measurable systemic exposure, lower but not zero adverse event rates, and substantial variability between formulations and individuals.
Systemic Absorption
The 2021 Piraccini trial, one of the largest randomized double-blind topical finasteride studies to date, tested a 0.25% finasteride spray applied once daily over 24 weeks in 458 men.[6]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety … Continue reading Plasma finasteride concentrations were over 100-fold lower than with oral finasteride. Serum DHT fell by 34.5% with topical use compared to 55.6% with a 1 mg oral tablet.
Caserini found that lower volumes (100-200 μL of 0.25% solution) reduced serum DHT by 24-26%, while higher volumes (300-400 μL) dropped it by 44-48%.[7]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading Even at the lowest tested doses, scalp DHT declined by roughly 47-52%, demonstrating effective local action with more limited systemic spillover.
Other studies report a range of serum DHT suppression with topical formulations, but all confirm that absorption is lower than oral administration, not absent. One hydroxyl-propyl chitosan formulation was designed specifically to retain finasteride in the reticular dermis near hair bulbs; repeated-dose rat experiments showed no detectable plasma finasteride and no accumulation in skin, although human data remain sparse.[8]Monti. D., Tampucci, S., Burgalassi, S., Chetoni, P., Lenzi, C., Pirone, A., Mailland, F. (2014). Topical formulations containing finasteride. Part I: in vitro permeation/penetration study and in … Continue reading
So to summarize, topical finasteride’s systemic exposure is markedly reduced compared to oral dosing, yet reductions in circulating DHT do still occur across most formulations (in human studies), meaning that some degree of systemic exposure is virtually unavoidable.
Reported Side Effect Rates in Studies
Clinical trials have monitored sexual dysfunction, dermatological reactions, and less common systemic complaints, though sample sizes and durations limit the ability to detect rare or delayed adverse events.
Sexual Side Effects
In the 2021 Piraccini study, 2.8% of topical finasteride users reported sexual adverse events (decreased libido, erectile dysfunction, and ejaculatory dysfunction) versus 4.8% on oral finasteride and 3.3% on placebo.
The similarity between the topical and placebo groups led some investigators to suggest that many reports may not be causally linked to the drug. However, a 2025 FDA pharmacovigilance analysis of adverse event reports (2019-2024) identified 32 cases involving topical finasteride formulations, most of which were compounded products, with complaints of erectile dysfunction, anxiety, brain fog, depression, fatigue, insomnia, decreased libido, and testicular pain.[9]US Food and Drug Administration. (2025). FDA alerts health care providers, compounders and consumers of potential risks associated with compounded topical finasteride products. FDA. Available at: … Continue reading What’s more, many of these symptoms reportedly persisted after discontinuation.
A separate analysis found that topical finasteride generated fewer signals for post-finasteride syndrome (PFS) – like events than oral finasteride, but erectile dysfunction remained the most consistently reported adverse event for both routes.[10]Gupta, A.K., Talukder, M., Keene, S.A., Bamimore, M.A. (2025). Is the Safety of Finasteride Correlated With Its Route of Administration: Topical Versus Oral? A Pharmacovigilance Study With Data From … Continue reading
Figure 1: Association between finasteride use (topical vs. oral) and occurrence of adverse events (sexual and neuropsychiatric) between 2006and 2011 (inclusive) and across men.[11]Gupta, A.K., Talukder, M., Keene, S.A., Bamimore, M.A. (2025). Is the Safety of Finasteride Correlated With Its Route of Administration: Topical Versus Oral? A Pharmacovigilance Study With Data From … Continue reading
Scalp Irritation and Contact Dermatitis
Local reactions are the most common topical-specific side effects. A 2018 systematic review noted reports of erythema, contact dermatitis, and scalp irritation in several studies, though serious cutaneous adverse events were absent.[12]Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading
Rates of contact dermatitis ranged from 12-24% in some combination formulations (finasteride + minoxidil), though these were generally mild and did not lead to treatment discontinuation. The irritation effect likely derives from vehicle components (alcohol, propylene glycol) as well as the active drug.
Headaches, Fatigue, and Other Systemic Complaints
Less frequently, trials have documented headaches, feeling about to faint, testicular pain, increased liver enzymes, and even bed-wetting in isolated cases. The 2025 FDA alert highlighted fatigue and insomnia among the systemic complaints associated with topical formulations, reinforcing that absorption can produce effects beyond the scalp.
Trial Limitations: Duration and Sample Size
Most published trials run 12-24 weeks with cohorts of 30-135 participants per arm, which may under-detect rare events (incidence <1%) or adverse effects that emerge only after prolonged exposure. Only a handful of studies have extended beyond six months, and none approach the multi-year observation periods needed to assess phenomena such as PFS.[13]Pereira, A.F.J.R., Coehlo, T, O. de A. (2020). Post-finasteride syndrome. Anais Brasileiros de Dermatologia. 95(3). 271-277. Available at: https://doi.org/10.1016/j.abd.2020.02.001 Recruitment through specialist dermatology centres may also select for patients more tolerant of medication, introducing survivorship bias.
How do the Side Effects Compare with Oral Finasteride?
Head-to-head data have shown that topical finasteride has a numerically lower, but not zero, incidence of systemic adverse events. In Piraccini’s trial, sexual side effects occurred in 2.8% of topical users versus 4.8% of oral users, representing an approximate halving of risk.
There are two important points often overlooked in clinical summaries, however:
- Population averages do not predict individual susceptibility: Even if 97% of topical finasteride users report no sexual side effects in a 24-week trial, that statistic offers little reassurance to someone who is biochemically sensitive to modest DHT reductions. Genetic polymorphisms in 5ɑ-reductase isoforms, androgen receptor sensitivity, and neurosteroid metabolism all vary widely, meaning that a 25% serum DHT drop can be innocuous for one man and symptomatic for another.[14]Pereira, A.F.J.R., Coehlo, T, O. de A. (2020). Post-finasteride syndrome. Anais Brasileiros de Dermatologia. 95(3). 271-277. Available at: https://doi.org/10.1016/j.abd.2020.02.001
- Short trials may miss long-latency or persistent events: Most published studies conclude at 6 months, yet reports to the FDA and patient registries describe symptoms that onset gradually or persist long after stopping treatment. The cases flagged in the FDA alert involved adverse events that continued to persist after product discontinuation, suggesting that systemic absorption, however modest, can trigger durable changes in a subset of users. However, the exact cause of symptom persistence is not yet known.
Why Do Some People Still Get Side Effects From Topical Finasteride?
Despite the reduced systemic exposure, some people still experience sexual, mood, and systemic adverse effects. Let’s take a look at the mechanisms why.
Skin Permeability Varies Significantly Between Individuals
Human skin is not a uniform barrier. Stratum corneum thickness, lipid composition, hydration status, pH, and blood flow differ among individuals and even within scalp regions of a single person.
In vitro permeation studies using human cadaver scalp skin show that finasteride flux ranges of 1.1-20.1 μg/cm2/h, depending on vehicle choice, a roughly 18-fold variation. Live scalp skin likely exhibits even greater heterogeneity due to sebum content, follicle density, and individual differences in dermal blood flow, all of which influence how much drug reaches the bloodstream versus remaining confined to follicular tissue.[15]Farah, A.H., Brown, M.B., McAuley, W.J. (2020). Enhanced Follicular Delivery of Finasteride to Human Scalp Skin Using Heat and Chemical Penetration Enhancers. Pharmaceutical Research. 37(6). 112. … Continue reading
For topical finasteride to minimize systemic exposure, it needs to stay within the area of the hair follicle. Variability in penetration means some individuals absorb more systemically than others, despite using the same concentration and volume.
Vehicle Formulation Dramatically Alters Absorption
The formulation of a topical finasteride product isn’t just a minor detail; it’s one of the biggest determinants of how much of the medication actually enters the bloodstream. Not all vehicles behave the same way, and the differences can be dramatic. For example, in vitro research comparing common topical solvents on human scalp skin shows that propylene glycol (PG) and isopropyl alcohol (IPA) formulations allow far more drug to pass through the skin: roughly 62–81 µg/cm² over 24 hours. In contrast, dimethyl isosorbide (DMI), often marketed as a “low-penetration” carrier, delivered just 17.8 µg/cm² in the same timeframe.[16]Farah, A.H., Brown, M.B., McAuley, W.J. (2020). Enhanced Follicular Delivery of Finasteride to Human Scalp Skin Using Heat and Chemical Penetration Enhancers. Pharmaceutical Research. 37(6). 112. … Continue reading That’s a 4.5-fold reduction in systemic permeation, purely from changing the solvent.
More sophisticated delivery systems behave differently still. Liposomal and niosomal gels, for example, are designed to deposit the medication deeper into hair follicles while limiting diffusion into the bloodstream. And in controlled experiments, these vesicular carriers did exactly that: liquid-state liposomes delivered 2.1–2.3% of the applied dose into the skin, compared to only 0.76% from a standard hydroalcoholic solution.[17]Tabbakhian, M., Tavakoli, N., Jaafari, M.R., Daneshamouz, S. (2006). Enhancement of follicular delivery of finasteride by liposomes and niosomes: 1. In vitro permeation and in vivo deposition studies … Continue reading
In other words, depending on the vehicle, the same dose of finasteride can behave like a local scalp medication or something much closer to an oral drug.
At the opposite end of the spectrum are ethosomes, ethanol-rich vesicles that dramatically increase drug penetration. In one study, finasteride ethosomes produced 7.4-times higher transdermal flux than the same drug in an aqueous solution, reaching all the way into the dermis.[18]Rao, Y., Zheng, F., Zhang, X., Gao, J., Liang, W. (2008). In vitro Percutaneous Permeation and Skin Accumulation of Finasteride Using Vesicular Ethosomal Carriers. 9(3). 860-865. Available at: … Continue reading
This is the kind of penetration behavior that significantly raises the likelihood of systemic absorption.
The bottom line: two topical finasteride products with the same concentration can have completely different side-effect profiles, simply because the vehicles modulate how much drug gets through the skin. Hydroalcoholic sprays tend to penetrate more aggressively; liposomal or niosomal gels are usually more conservative; and individual biology, skin barrier integrity, inflammation, and sebaceous output add yet another layer of variability. This is why real-world responses to topical finasteride are so mixed, and why formulation needs to be taken as seriously as the dose itself.
Concentration and Application Volume Compounds Absorption
Higher concentrations and larger volumes increase both local scalp DHT reduction and systemic spillover. As mentioned above, Caserini’s dose-response study found that applying 100-200 μL of 0.25% finasteride reduced serum DHT by 24-26%, whereas 300-400 μL volumes reduced it by 44-48%, approaching the efficacy of oral dosing at the higher end.[19]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading
Users who apply larger volumes, cover a wider scalp area, or use higher-concentration formulations absorb more drug systemically, shifting the risk-benefit profile toward greater potential for side effects.
Finasteride’s Half-Life Enables Accumulation
Finasteride has a terminal half-life of 5-6 hours in young men and 8 hours in older men.[20]Mysore, V. (2012). Finasteride and sexual side effects. Indian Dermatology Online Journal. 3(1). 62-65. Available at: https://doi.org/10.4103/2229-5178.93496 Although short, daily topical application leads to steady-state accumulation in plasma and skin tissues.
With topical dosing, measurable finasteride concentrations appear in plasma within hours and remain detectable for a full day; repeated daily application amplifies cumulative levels, particularly if absorption is higher than intended due to vehicle choice or individual skin permeability.[21]Tai, Z., Cui, Z., Shi, X., Li, H., Chai, R., Huang, Y., Fang, Y., Jia, D., Zhu, Q., Chen, Z. (2025). The Pharmacokinetics of Topical Finasteride 0.25% Spray in Chinese Adult Male Volunteers with … Continue reading
DHT Sensitivity Varies Widely Between Individuals
Even when two people experience the same level of DHT reduction, their bodies may not respond the same way. This is where individual biology plays a much bigger role than most clinical studies acknowledge. Variations in genes tied to 5α-reductase activity, androgen receptor sensitivity (including CAG repeat length), and enzymes involved in neurosteroid production, such as 3α-HSD, can all influence how strongly someone reacts to changes in DHT.[22]Cecchin, E., De Mattia, E., Toffoli, G., Mazzon, G., Cauci, S., Trombetta, C. (2014). A Pharmacogenetic Survey of Androgen Receptor (CAG)N and (GGN)N Polymorphisms in Patients Experiencing … Continue reading,[23]Rahimi-Ardabili, B., Pourandarjani, R., Habibollahi, P., Mualeki, A. (2006). Finasteride-induced depression: a prospective study. BMC Clinical Pharmacology. 6(7). Available at: … Continue reading
For one man, a 30% drop in circulating DHT might feel completely benign. For another, that exact same reduction might coincide with noticeable changes in libido, erectile quality, or overall sexual function.
Layered on top of genetics are factors like baseline hormone levels, underlying hypogonadism, and even a person’s psychophysiological sensitivity to small hormonal shifts. Together, these variables help explain why side effects remain so individualized and difficult to predict, and why two people using an identical topical finasteride formulation can walk away with very different experiences. This isn’t meant to alarm anyone, only to highlight that DHT suppression is not a one-size-fits-all phenomenon, and that understanding your own sensitivity is often just as important as choosing the right dose or vehicle.
How Can I Minimize the Risk of Topical Finasteride Side Effects?
Target the Lowest Effective Exposure
- Understand exposure = concentration × volume. The systemic finasteride dose delivered through the scalp depends on both how strong the solution is and how much is applied. A low-concentration solution applied in large volume can expose the body to as much finasteride as a small volume of a stronger solution. Both variables must be considered together when aiming to reduce side effects.
- Start with the minimum viable dose. Evidence suggests that daily exposure to around 0.1 mg (for example, 0.005% solution applied at 2 mL per day) can still achieve measurable scalp DHT reduction with negligible systemic suppression.
- 0.25% remains the most studied concentration. Clinical trials show that 0.25% topical finasteride effectively treats androgenetic alopecia with hair density gains similar to oral finasteride yet significantly lower serum DHT reduction. For most users, starting at or below this level helps balance efficacy and safety.
- Higher isn’t always better. Increasing concentration or volume each raises systemic absorption, but hair-growth benefits tend to plateau. Pushing beyond the optimal concentration can shift the risk-to-benefit ratio unfavorably.
- Consider titrating upward. If highly sensitive to hormonal side effects, begin at lower exposure (e.g., 0.025–0.1% at 1 mL daily), then increase concentration or frequency only if well-tolerated and results plateau. Serum DHT measurements can help assess systemic exposure over time.
Adjust Volume and Frequency to Limit Systemic Load
- Keep volume conservative. Systemic absorption scales with total dose applied. Around 1 mL per application is a common clinical standard; using more typically provides diminishing returns for hair outcomes but notably raises systemic uptake.
- Use the lowest frequency that maintains results. Because twice-daily 0.25% use can suppress serum DHT by up to 70%—similar to oral dosing—while once-daily reduces it by only 20–35%, finding a balance between efficacy and exposure is key. For many users, once-daily application of a lower-strength solution achieves a good safety–efficacy trade-off.
Choose Vehicles Carefully
- Hydroalcoholic vs. liposomal: Standard hydroalcoholic (alcohol-based) solutions enhance drug penetration, which can lead to greater systemic absorption. In contrast, advanced vehicles like liposomal or other nanoparticle-based formulations are designed to target drug delivery to the hair follicle and reduce transdermal flux into the bloodstream.
Implement Smart Application Strategies
- Apply to a dry scalp: Applying topicals to wet skin can enhance absorption. Ensure the scalp is completely dry to create a more robust barrier.
- Target affected areas only: Use the applicator to apply the solution precisely to the areas of thinning hair, not the entire scalp, to limit the total surface area of absorption.
- Avoid compromised barriers: Do not apply the solution immediately after microneedling, on sunburnt skin, or on a scalp with cuts, inflammation, or dermatitis, as a compromised skin barrier significantly increases uptake.
- Wash hands thoroughly: Always wash hands with soap and water after application to prevent accidental transfer to other body parts or individuals.
Important: If you experience any side effects, we recommend first stopping and speaking to your primary care physician.
Who Should Avoid or Be Cautious with Topical Finasteride?
While topical finasteride carries a lower risk profile than its oral counterpart, some people may still need to exercise caution or avoid the drug entirely.
Groups That Should Exercise Caution
- Those with a prior history of persistent sexual dysfunction from oral finasteride.
- People with underlying endocrine disorders or baseline sexual dysfunction.
- Those sensitive to propylene glycol or alcohol-based carrier agents.
It should be mentioned that a history of side effects from oral finasteride does not mean that topical finasteride won’t work well for you; however, it does warrant a personalized, cautious approach. This approach might include starting with lower doses, extended monitoring, and communication with the prescribing physician about any potential symptoms.
Groups That Should Avoid Topical Finasteride
- Women who are pregnant or attempting to conceive.
- People with severe scalp dermatitis or barrier dysfunction.
- Individuals with known allergy/hypersensitivity to finasteride or formulation components.
If I Can’t Tolerate Topical Finasteride, What Should I Do?
For users unable to tolerate topical finasteride due to side effects or scalp irritation, several evidence-based alternatives exist.
Pharmacological Alternatives
Low Dose Oral Minoxidil
Oral minoxidil is a hair growth stimulant that bypasses the hormonal pathway entirely. Studies show high efficacy (e.g., 43% of men achieving excellent results with 5 mg), but it carries systemic risks such as hypertrichosis (excess body hair), fluid retention, and cardiovascular effects like tachycardia.[24]Vano-Galvan, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Saceda-Corralo, D., Rodrigues-Barata, R., Jiminez-Cauhe, J., Koh, W.L., Poa, J.E., Jerjen, R., de Carvalho, L.T., John, J.M., … Continue reading
Alternative Topical Anti-Androgens
Fluridil is a topical anti-androgen designed to degrade in water (i.e., blood), minimizing systemic exposure. Clinical data are limited but suggest efficacy without affecting serum testosterone or sexual function.[25]Sovak, M., Seligson, A.L., Kucerova, R., Bienova, M., Hajduch, M., Bucek, M. (2002). Fluridil, a rationally designed topical agent for androgenetic alopecia: first clinical experience. Dermatologic … Continue reading
Clascoterone (17ɑ-proprionate) is an androgen receptor antagonist that competes with DHT at the follicle rather than reducing DHT production. It offers a different mechanism but is still under investigation for hair loss.[26]Devjani, S., Ezemma, O., Kelley, K.J., Stratton, S., Senna, M. (2023). Androgenetic Alopecia: Therapy Update. Drugs. 83(8). 701-715. Available at: PMID: 37166619
Pyrilutamide is a non-steroidal anti-androgen that binds to the androgen receptor with high affinity. Like fluridil, it is designed to metabolize into an inactive form upon entering systemic circulation, theoretically offering a safety advantage over finasteride.[27]Biospace. (2023). Kintor Pharma’s KX-826 and GT20029 for Treatment of Androgenetic Alopecia (AGA) and Acne Presented at AAD 2023. Biospace. Available at: … Continue reading
Non-Pharmaceutical Options
Microneedling creates micro-injuries in the scalp and stimulates growth factors (PDGF, VEGF) and activates Wnt/ꞵ-catening signaling, promoting hair regeneration even without concurrent drug use.[28]Dhurat, R., Sukesh, M.S., Ayhad, G., Dandale, A., Pal, A., Pund, P. (2013). A Randomized Evaluator Blinded Study of Effect of Microneedling in Androgenetic Alopecia: A Pilot Study. International … Continue reading
Low-level laser therapy (LLLT) devices use red light (650-655 nm) to stimulate mitochondrial activity in hair follicles, prolonging anagen and improving density with an excellent safety profile.[29]Egger, A., Resnik, S.R., Aickara, D., Maranda, E., Kaiser, M., Wikramanayake, T.C. (2020). Examining the Safety and Efficacy of Low-Level Laser Therapy for Male and Female Pattern Hair Loss: A … Continue reading
Some botanical supplements have some 5ɑ-reductase inhibitory properties. Saw palmetto and pumpkin seed oil have been shown to have mild 5ɑ-reductase inhibition.[30]Cho, Y.H., Lee, S.Y., Jeong, D.W., Choi, E.J., Kim, Y.J., Lee, J.G., Yi, H.Y., Cha, H.S. (2014). Effect of Pumpkin Seed Oil on Hair Growth in Men with Androgenetic Alopecia: A Randomized, … Continue reading,[31]Pilar, P., 2010. Potency of a novel saw palmetto ethanol extract, SPET-085, for inhibition of 5alpha-reductase II. Advances in Therapy. 27(8). 555-563. Available at: … Continue reading While less potent than finasteride (reducing DHT by ~30-40% vs. >60%), they may provide a viable “middle ground” for those seeking modest stabilization.
Final Thoughts
Topical finasteride offers a compelling middle path for those seeking the hair-growth benefits of 5-α-reductase inhibition with a lower likelihood of systemic side effects. But “lower” does not mean “none,” and responses vary widely depending on formulation, dose, skin permeability, and individual DHT sensitivity. The current research, while promising, remains short-term, and real-world patterns remind us that systemic exposure is still possible.
For many people, topical finasteride can be a safe, effective component of a broader hair-loss program. The key is approaching it strategically: start low, personalize dosing, choose the right vehicle, and monitor closely for changes. For others, especially those prone to hormonal side effects, alternatives ranging from anti-androgen topicals to non-pharmaceutical options may offer a better balance of efficacy and tolerability.
As with any AGA therapy, the best results come from matching the treatment to the individual, not the other way around.
References[+]
References ↑1 Zito PM, Bistas KG, Patel P, et al. Finasteride. [Updated 2024 Feb 28]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513329/ (Accessed: December 2025) ↑2 Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. International Journal of Clinical Pharmacology and Therapeutics. 54(1). 19-27. Available at: https://doi.org/10.5414/CP202467 ↑3 Smith, A.B., Carson, C.C. (2009). Finasteride in the treatment of patients with benign prostatic hyperplasia: a review. Therapeutics and Clinical Risk Management. 5. 535-545. Available at: https://doi.org/10.2147/tcrm.s6195 ↑4 Traish, A.M. (2017). Negative Impact of Testosterone Deficiency and 5ɑ-Reductase Inhibitors Therapy on Metabolic and Sexual Function in Men. Advances in experimental medicine and biology. 1043. 473-526. Available at: https://doi.org/10.1007/978-3-319-70178-3_22 ↑5 Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. International Journal of Clinical Pharmacology and Therapeutics. 54(1). 19-27. Available at: https://doi.org/10.5414/CP202467 ↑6 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology and Venereology. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑7, ↑19 Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. International Journal of Clinical Pharmacology and Therapeutics. 54(1). 19-27. Available at: https://doi.org/10.5414/CP202467 ↑8 Monti. D., Tampucci, S., Burgalassi, S., Chetoni, P., Lenzi, C., Pirone, A., Mailland, F. (2014). Topical formulations containing finasteride. Part I: in vitro permeation/penetration study and in vivo pharmacokinetics in hairless rat. Journal of Pharmaceutical Sciences. 103(8). 2307-2314. Available at: https://doi.org/10.1002/jps.24028 ↑9 US Food and Drug Administration. (2025). FDA alerts health care providers, compounders and consumers of potential risks associated with compounded topical finasteride products. FDA. Available at: https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-consumers-potential-risks-associated-compounded (Accessed: December 2025) ↑10 Gupta, A.K., Talukder, M., Keene, S.A., Bamimore, M.A. (2025). Is the Safety of Finasteride Correlated With Its Route of Administration: Topical Versus Oral? A Pharmacovigilance Study With Data From the United States Food and Drug Administration Adverse Event Reporting System. International Journal of Dermatology. Available at: https://doi.org/10.1111/ijd.17957 ↑11 Gupta, A.K., Talukder, M., Keene, S.A., Bamimore, M.A. (2025). Is the Safety of Finasteride Correlated With Its Route of Administration: Topical Versus Oral? A Pharmacovigilance Study With Data From the United States Food and Drug Administration Adverse Event Reporting System. International Journal of Dermatology. Available at: https://doi.org/10.1111/ijd.17957 ↑12 Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in Dermatology. 17(4). 457-463. Available at: PMID: 29601622 ↑13, ↑14 Pereira, A.F.J.R., Coehlo, T, O. de A. (2020). Post-finasteride syndrome. Anais Brasileiros de Dermatologia. 95(3). 271-277. Available at: https://doi.org/10.1016/j.abd.2020.02.001 ↑15, ↑16 Farah, A.H., Brown, M.B., McAuley, W.J. (2020). Enhanced Follicular Delivery of Finasteride to Human Scalp Skin Using Heat and Chemical Penetration Enhancers. Pharmaceutical Research. 37(6). 112. Available at: https://doi.org/10.1007/s11095-020-02822-y ↑17 Tabbakhian, M., Tavakoli, N., Jaafari, M.R., Daneshamouz, S. (2006). Enhancement of follicular delivery of finasteride by liposomes and niosomes: 1. In vitro permeation and in vivo deposition studies using hamster flank and ear models. International Journal of Pharmaceutics. 1-2(323). 1-10. Available at: https://doi.org/10.1016/j.ijpharm.2006.05.041 ↑18 Rao, Y., Zheng, F., Zhang, X., Gao, J., Liang, W. (2008). In vitro Percutaneous Permeation and Skin Accumulation of Finasteride Using Vesicular Ethosomal Carriers. 9(3). 860-865. Available at: https://doi.org/10.1208/s12249-008-9124-y ↑20 Mysore, V. (2012). Finasteride and sexual side effects. Indian Dermatology Online Journal. 3(1). 62-65. Available at: https://doi.org/10.4103/2229-5178.93496 ↑21 Tai, Z., Cui, Z., Shi, X., Li, H., Chai, R., Huang, Y., Fang, Y., Jia, D., Zhu, Q., Chen, Z. (2025). The Pharmacokinetics of Topical Finasteride 0.25% Spray in Chinese Adult Male Volunteers with Androgenic Alopecia: A Phase I Study. Advances in Therapy. 42(3). 1494-1505. Available at: https://doi.org/10.1007/s12325-025-03106-w. ↑22 Cecchin, E., De Mattia, E., Toffoli, G., Mazzon, G., Cauci, S., Trombetta, C. (2014). A Pharmacogenetic Survey of Androgen Receptor (CAG)N and (GGN)N Polymorphisms in Patients Experiencing Long-Term Side Effects after Finasteride Discontinuation. The International Journal of Biological Markers. 29(4). 310-316. Available at: https://doi.org/10.5301/jbm.500095 ↑23 Rahimi-Ardabili, B., Pourandarjani, R., Habibollahi, P., Mualeki, A. (2006). Finasteride-induced depression: a prospective study. BMC Clinical Pharmacology. 6(7). Available at: https://doi.org/10.1186/1472-6904-6-7 ↑24 Vano-Galvan, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, O.M., Saceda-Corralo, D., Rodrigues-Barata, R., Jiminez-Cauhe, J., Koh, W.L., Poa, J.E., Jerjen, R., de Carvalho, L.T., John, J.M., Salas-Callo, C.I., Vincenzi, C., Yin, L., Lo-Sicco, K., Waskiel-Burnat, A., Starace, M., Zamorano, J.L., Jaen-Olasolo, P., Piraccini, B.M., Rudnicka, L., Shapiro, J., Tosti, A., Sinclair, R., Bhoyrul, B. (2021). Safety of low-dose oral minoxidil for hair loss: A multicenter study of 1404 patients. Journal of the American Academy of Dermatology. 84(6). 1644-1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑25 Sovak, M., Seligson, A.L., Kucerova, R., Bienova, M., Hajduch, M., Bucek, M. (2002). Fluridil, a rationally designed topical agent for androgenetic alopecia: first clinical experience. Dermatologic Surgery. 28(8). 678-685. Available at: https://doi.org/10.1046/j.1524-4725.2002.02017.x ↑26 Devjani, S., Ezemma, O., Kelley, K.J., Stratton, S., Senna, M. (2023). Androgenetic Alopecia: Therapy Update. Drugs. 83(8). 701-715. Available at: PMID: 37166619 ↑27 Biospace. (2023). Kintor Pharma’s KX-826 and GT20029 for Treatment of Androgenetic Alopecia (AGA) and Acne Presented at AAD 2023. Biospace. Available at: https://www.biospace.com/kintor-pharma-s-kx-826-and-gt20029-for-treatment-of-androgenetic-alopecia-aga-and-acne-presented-at-aad-2023 (Accessed: December 2025) ↑28 Dhurat, R., Sukesh, M.S., Ayhad, G., Dandale, A., Pal, A., Pund, P. (2013). A Randomized Evaluator Blinded Study of Effect of Microneedling in Androgenetic Alopecia: A Pilot Study. International Journal of Trichology. 5(1). 6-11. Available at: https://doi.org/10.4103/0974-7753.114700 ↑29 Egger, A., Resnik, S.R., Aickara, D., Maranda, E., Kaiser, M., Wikramanayake, T.C. (2020). Examining the Safety and Efficacy of Low-Level Laser Therapy for Male and Female Pattern Hair Loss: A Review of the Literature. Skin Appendage Disorders. 6(5). 259-267. Available at: https://doi.org/10.1159/000509001 ↑30 Cho, Y.H., Lee, S.Y., Jeong, D.W., Choi, E.J., Kim, Y.J., Lee, J.G., Yi, H.Y., Cha, H.S. (2014). Effect of Pumpkin Seed Oil on Hair Growth in Men with Androgenetic Alopecia: A Randomized, Double-Blind, Placebo-Controlled Trial. Evidence-Based Complementary and Alternative Medicine. 23. 549721. Available at: https://doi.org/10.1155/2014/549721 ↑31 Pilar, P., 2010. Potency of a novel saw palmetto ethanol extract, SPET-085, for inhibition of 5alpha-reductase II. Advances in Therapy. 27(8). 555-563. Available at: https://doi.org/10.1007/s12325-010-0041-6 After years of anticipation, we finally have the first-ever study testing topical dutasteride as a standalone treatment for androgenic alopecia (AGA). No microneedling, no formulations mixed with other ingredients, just topical dutasteride itself.
And the results?
Frankly, they are shocking.
According to the study, low-dose topical dutasteride outperformed oral finasteride for hair regrowth.
On the face of it, this is so surprising that it borders on unbelievable, especially given the 5+ years of real-world data we’ve collected from our members.
So, in this article, we’ll break down:
- What we previously knew about topical dutasteride.
- How real-world user experiences compare to clinical findings.
- What this new study claims.
- And why two major methodological problems force us to seriously question the results.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What We Knew About Low-Dose Topical Dutasteride Before This Study
Until now, only two small clinical studies have evaluated low-dose topical dutasteride. Both used it alongside microneedling and applied it just once every 1-4 weeks.
-
Nada et al. (2018): Microneedling + Topical Dutasteride vs. Microneedling Alone
Nada et al. reported that adding low-dose topical dutasteride to a structured microneedling regimen improved hair density and shaft thickness more than microneedling alone, while only slightly reducing serum DHT, likely without meaningful systemic hormonal impact.[1]Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmaged, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). Available at: … Continue reading
30 men with AGA were randomized to either microneedling plus topical dutasteride 0.02% or microneedling alone for six months. Both groups received 13 microneedling sessions with a 1.5 mm Dermapen (12-needle cartridge) on a staggered schedule: weekly during the first 8 weeks, then gradually reduced to once every 2-4 weeks through month six. In the combination arm, up to 2 mL of 0.02% dutasteride
-
Sanchez-Meza et al. (2022): Microneedling + Topical Dutasteride vs. Microneedling Alone
Sanchez-Meza et al. found that adding very low-dose topical dutasteride to microneedling produced greater clinical and trichoscopic improvement than microneedling with placebo, without reported sexual side effects and with much lower total dutasteride exposure than earlier work.[2]Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical … Continue reading
34 men with AGA were randomized to microneedling plus 1 mL of 0.01% topical dutasteride (~0.1 mg) or microneedling plus 1 mL of saline as a placebo. Both groups received three treatment sessions spaced 4 weeks apart using a Dr. Pen Ultima A6 device set to a 2.5 mm needle depth, and outcomes were assessed over a 16-20 week period.
While both these studies showed mild hair regrowth, the effect is not from topical dutasteride alone, nor from daily use.
Furthermore, low-dose topical dutasteride showed almost no systemic dihydrotestosterone (DHT) suppression.
Across these studies, bloodwork suggested:
- Little to no measurable DHT suppression
- Meaning minimal systemic absorption
- And therefore lower risk of systemic side effects
This makes sense. Dutasteride is a large, lipophilic molecule. At low concentrations, it tends to stay localized to the scalp unless dosing or penetration enhancers are significant.
What Did Real-World Users Experience?
Because early studies were promising, some of our members tried 0.01-0.02% topical dutasteride in the years that followed.
A dozen users also tracked blood DHT levels before and after treatment.
The results? Strong localization with minimal regrowth.
- No meaningful changes in serum DHT (just normal daily variation of 10-15%).
- Even among users applying up to 2 mL daily.
- Meaning: absorption remained low – as expected.
- But…users also reported very little growth.
Instead, most people saw hair maintenance, not cosmetic improvement.
This aligns with the published studies: low-dose topical dutasteride appears to stabilize hair loss, with minimal systemic impact, but not drive substantial growth.
You can get more information about this and comparisons to topical finasteride here:
- Topical Dutasteride Vs. Topical Dutasteride: The Dose Decides Everything (2025)
- Topical Finasteride: Where Do We Stand Today? (2025 Update)
- Topical Finasteride: Same Results, No Side Effects? (2022)
What Happened in the August 2025 Study?
This new randomized, double-blind, placebo-controlled study appeared to be the gold standard of topical dutasteride research.[3]Panuganti, V.K., Mandala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., Rao, S., Dundigalla. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety … Continue reading
It was the first study to test topical dutasteride:
- By itself (no microneedling).
- With daily use.
- Across multiple doses (0.01%, 0.02%, and 0.05%).
- Against both a placebo and oral finasteride.
On paper, this looked like the definitive study that we have long needed.
Study Design
135 men aged 20-60 with Norwood III vertex, IV, or V AGA were randomized across five treatment arms in a 2:2:2:2:1 ratio:
- 0.01% topical dutasteride (n=30)
- 0.02% topical dutasteride (n=30)
- 0.05% topical dutasteride (n=30)
- Oral finasteride 1 mg + placebo topical (n=30)
- Placebo topical + placebo oral (n=15).
All participants were Asian men, average age of ~38 years. Baseline hair loss severity was balanced across groups.
Each 1 mL dose contained:
- 0.01% = 0.1 mg dutasteride
- 0.02% = 0.2 mg dutasteride
- 0.05% = 0.5 mg dutasteride
Importantly, the solution was nearly 30% dehydrated alcohol, a known penetration enhancer. This choice may have implications for systemic absorption and hair-count interpretation, though the authors claim systemic exposure remained minimal.
The researchers measured hair counts (total area hair counts) and hair widths (total area hair widths). Using a Dino-Lite microscope, researchers identified a 1.9 cm2 circular region at the vertex, clipped hairs to 0.5 – 1 mm, marked the center of the circle, and captured macrophotographs at baseline, week 12, and week 24.
The authors do not mention tattooing, ink permanence, or the use of a positioning device, which becomes critically important later.
The Study’s Reported Results
Here’s where the shock factor begins.
At 24 weeks, the study reported:
- 0.01% dutasteride: +32.32 hairs/cm2
- 0.02% dutasteride: +27.48 hairs/cm2
- 0.05% dutasteride: +75.52 hairs/cm2
- Oral finasteride: +41.21 hairs/cm2
- Placebo: 0.07 hairs/cm2
The headline claim: “0.05% topical dutasteride significantly outperformed oral finasteride (p=0.0083)”
This is a remarkable result, and one that contradicts every known real-world case we’ve observed at similar dosing.
Hair-Width Improvements
All active groups increased hair thickness, with results very close to oral finasteride:
- 0.01% +6.68 μm.
- 0.02% +9.15 μm.
- 0.05% +11.59 μm.
- Finasteride: +10.68 μm.
- Placebo: +4.00 μm.
Only the 0.05% topical and finasteride groups significantly beat placebo. The 0.05% topical did not significantly outperform finasteride in hair-width metrics.
Global Photography Assessment (Investigator-Rated)
At 24 weeks, the percentage of participants rated as having “moderate improvement” or better (GPA ≥ +2) was:
- 0.05% topical: 68.97%.
- Finasteride: 21.43%.
- Placebo: 13.33%.
These numbers suggest a level of regrowth from 0.5 mg/week of topical dutasteride that we’ve simply never witnessed, not in our community, not in the medical literature, and not among clinicians who routinely prescribe topical dutasteride.
Patient-Reported Outcomes (MHGQ)
By week 24, 96.55% of 0.05% topical users were satisfied with the hair on top of their heads. This is far above the finasteride group (71.43%) and far above the placebo (33.33%).
Again, the magnitude of the difference warrants scrutiny.
Serum Hormone Changes
This is where things do align with real-world experiences of low-dose topical users.
DHT Reductions
- Finasteride: 11% to -27%.
- 0.05% topical dutasteride: around -9% to -11%.
Testosterone Increases
- Finasteride: +20%.
- Topical dutasteride: modest, non-significant changes.
The authors emphasize that topical dutasteride caused minimal systemic effects.
This is consistent with our members’ lab data – but inconsistent with the hair-growth magnitude reported.
Pharmacokinetics (How Much Enters the Bloodstream)
According to the PK data:
- Plasma dutasteride levels were near or below quantification limits.
- A few values spiked as high as 2555 pg/mL, suggesting occasional high absorption events.
- Dutasteride remained detectable at day 168, implying accumulation over time.
The authors interpret this as “low systemic exposure”. But variability this large raises questions.
Safety Findings
Across the entire study, no serious adverse events were reported, no withdrawals due to safety, minimal skin irritation, and all groups showed mild effects like “glazing” at similar rates.
This matches expectations for low-dose topicals.
So, Why Don’t the Results Match Real-World Experience?
For more than five years, we’ve tracked user outcomes from low-dose topical dutasteride at comparable or even higher weekly doses than used in this study.
Not once have we seen:
- Regrowth exceeding oral finasteride.
- Cosmetic transformation from 0.05% topical alone.
- Hair-count improvements anywhere near +75 hairs/cm2.
Even in dermatology clinics across the world, this simply isn’t observed. So, why would this study find such dramatically different results?
It comes down to two major methodological problems – both related to hair counts.
Problem #1: Baseline Hair Counts That Defy Biology
The average baseline hair density reported in the study was 305-330 hairs/cm2.
For context:
- A healthy adult without hair loss typically has 100-250 hairs/cm2.
- Men with Norwood III-V AGA typically have 25-100 hairs/cm2 in the vertex.
- Yet this study reports 300+ hairs/cm2 in balding men.
This would mean that severely balding scalps had triple the hair density of a normal, non-balding scalp, and that hair counts exceeded what is physiologically plausible.
Why might this happen?
Possible Explanations
Vellus hairs were counted.
- The study never defines a diameter cutoff.
- Best practice is to exclude hairs ≤40 µm.
- Counting vellus hairs inflates numbers dramatically.
The measurement area was bigger than stated.
- The paper claims 1 cm2.
- Even a small mismeasurement could triple hair counts.
Manual counting errors.
- The study doesn’t specify software use.
- Manual hair counting is outdated and error-prone.
None of these possibilities inspires confidence in the baseline data.
Problem #2: The Measurement Circle Moves Between Photos
This is the more serious problem, and the one most likely to invalidate the findings entirely.
When examining the study’s published before-and-after images, the measurement circles:
- Change location.
- Change size.
- Change shape.
- Appear hand-drawn.
- Clearly do not track the same exact scalp area over time.
Figure 1: Representative hair growth images.[4]Panuganti, V.K., Mandala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., Rao, S., Dundigalla. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety … Continue reading
Take a close look at these images; the center point marks do not appear to be consistent (at least from these photos). This means that you really can’t compare the improvements over time, as the follow-up hair counts would have been conducted in slightly different areas of the scalp!
This is an enormous methodological flaw.
A shift of just 1-2 millimetres in circle placement can change hair counts by 50% or more, something we demonstrated in our 2021 publication.[5]Jnr, R.E., Ruiz, S. (2021). Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia: Are Biopsy Location, Hair Diameter Diversity, and Relative Hair Follicle … Continue reading
Fig 2: Circle A has 50% more hair than Circle B.[6]Jnr, R.E., Ruiz, S. (2021). Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia: Are Biopsy Location, Hair Diameter Diversity, and Relative Hair Follicle … Continue reading
Yet the improvements reported in this study were around 10-30%.
Meaning: These “improvements” could be fully explained by inconsistent circle placement, not actual regrowth.
This alone is enough to call the validity of the hair-count data into question.
Final Thoughts
This is one situation where we at PHH trust the real-world experiences of our members over a randomized, controlled clinical trial, because while the study appears rigorous on the surface, its hair-counting methods reveal inconsistencies significant enough to undermine its conclusions.
All available evidence still supports the following: low-dose topical dutasteride (0.01-0.05%) localizes well to the scalp, minimally suppresses serum DHT, effectively slows or stops hair loss, and rarely produces meaningful regrowth, whereas high-strength topical dutasteride (≥0.1%) is more likely to leak systemically, carry a greater risk of side effects, and generate visible regrowth approaching the results of oral finasteride.
References[+]
References ↑1 Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmaged, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). Available at: https://smj.journals.ekb.eg/article_42083_24b61cbba4be9982db23c318414034c0.pdf (Accessed: December 2025) ↑2 Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-Y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical dutasteride solution for androgenetic alopecia: a randomized placebo-controlled study. Journal of the European Academy of Dermatology and Venereology. 36(10). E806-e808. Available at: https://doi.org/10.1111/jdv.18285 ↑3 Panuganti, V.K., Mandala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., Rao, S., Dundigalla. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects with Androgenetic Alopecia. Cureus. 17(8). E89309. Available at: https://doi:10.7759/cureus.89309 ↑4 Panuganti, V.K., Mandala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., Rao, S., Dundigalla. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects with Androgenetic Alopecia. Cureus. 17(8). E89309. Available at: https://doi:10.7759/cureus.89309 ↑5 Jnr, R.E., Ruiz, S. (2021). Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia: Are Biopsy Location, Hair Diameter Diversity, and Relative Hair Follicle Miniaturization Partly to Blame? Clinical, Cosmetic and Investigative Dermatology. 14. 357-365. Available at: https://doi.org/10.2147/CCID.S306157 ↑6 Jnr, R.E., Ruiz, S. (2021). Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia: Are Biopsy Location, Hair Diameter Diversity, and Relative Hair Follicle Miniaturization Partly to Blame? Clinical, Cosmetic and Investigative Dermatology. 14. 357-365. Available at: https://doi.org/10.2147/CCID.S306157 Finasteride is among the most effective drugs for androgenic alopecia. And while side effects are often overstated, it can lead to reduced libido or lowered sperm counts in some men. For this reason, many choose topical versus oral finasteride, hoping to limit the drug’s DHT-reducing effects to the scalp. But at certain doses, even topical finasteride can become systemic. So, to minimize side effects, which finasteride dosage, formula and application is best?
In this article, we’ll review
- About oral versus topical finasteride
- Why topical finasteride can still go systemic
- Why choosing the lowest percentage solution isn’t the only answer
- How to maximize finasteride gains, while minimizing side effects
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

About Finasteride
Finasteride, also known under the brand name Propecia or Proscar, is a prescription medication approved by the FDA for the treatment of androgenic alopecia (AGA). The anti-androgen works by reducing the production of Dihydrotestosterone (DHT), a hormone linked to pattern hair loss. In use since 1992, Finasteride is among the most powerful and well-studied drugs for hair loss.
Oral Finasteride
Oral finasteride stops AGA progression in 80-90% of men and, on average, leads to a 10% increase in hair count over two years.[1]https://www.sciencedirect.com/science/article/pii/S0022202X15529357 For men wanting a “hands-off” approach to hair maintenance, oral finasteride can be an excellent option. With a once-daily pill, it’s expected that hair loss will stop at approximately 6 months, and thereafter, improve.
However, oral finasteride isn’t for everyone. While the risk of side effects are often overstated online, the drug appears to reduce libido in a certain percentage of men. Oral finasteride can also temporarily lower sperm counts, which might make conception more difficult during its first six months of use. In some men, the use of finasteride appears to increase anxiety and/or depression. While the true incidence and magnitude of these reports are hard to discern, it’s understandable that many are weary of taking this once daily pill.
Fortunately, recent improvements in finasteride’s delivery may mean we no longer need to throw the proverbial baby out with the bathwater.
Topical Finasteride
The biggest reason people seek out topical finasteride (instead of oral finasteride) is because they want to minimize systemic exposure to the drug, and in doing so, localize finasteride’s effects to the scalp.
While research is still in the early stages, evidence suggests that topical finasteride can produce similar levels of hair regrowth compared to oral finasteride, with a significantly reduced incidence of side effects.
Topical finasteride works identically to its oral counterpart: by inhibiting the type II 5-alpha reductase enzyme to reduce DHT. It is designed to target scalp DHT instead of systemic DHT levels.
However, limiting finasteride’s reach to scalp, not serum, DHT is harder than perhaps anticipated. To understand why, let’s take a closer look at scalp versus serum DHT.
Serum vs Scalp DHT
There’s a lot of evidence that the hormone DHT is directly implicated in androgenic alopecia. In fact, research directly links DHT to all three of AGA’s defining characteristics:
- Increased telogen:anagen ratio. DHT’s influence on DKK-1 expression increases hair shedding
- Increased anagen cycling. DHT’s effects on signaling pathways limit the growth phase of hairs
- Hair follicle miniaturization. DHT’s effects on dermal papilla sizing thins hair with each cycle
There are a wide variety of DHT reducers available, including prescription drugs, over-the-counter products, intradermal injections, and even herbal supplements. Finasteride reduces DHT in two ways.
- Oral finasteride reduces DHT everywhere in the body. Beyond just the scalp, it reduces DHT in the blood, brain, and prostate.
- Topical finasteride attempts to reduce DHT only where it is applied – in the scalp – with minimal effects on DHT levels elsewhere.
However, it’s far easier than most realize for topical finasteride to go systemic. And when it does, it potentially reduces DHT everywhere – leading to the very side effects it’s meant to prevent.
Topical Finasteride Can Still Go Systemic
Why? When it comes to lowering DHT (the goal of the drug), finasteride has a highly-sensitive, dose-dependent response curve. This means that while 0.01 mg of finasteride barely reduces any DHT at all, 0.1 mg reduces almost as much DHT as 5 mg, a much larger dose.
This implies that when applying topical finasteride, only a tiny fraction of it needs to go systemic in order to produce the same DHT-lowering effects as oral finasteride. If this happens, the purpose of using the topical formulation is completely defeated.
Research suggests a 1% topical finasteride formulation, applied twice daily, is ‘non-inferior,’ meaning equivalent, to 1 mg oral finasteride tablets.[2]https://www.ncbi.nlm.nih.gov/pubmed/19172031 And while that’s a positive in terms of hair growth, using this amount topically twice daily pretty much guarantees systemic absorption. In other words, if 1% topical is the equivalent of 1 mg oral, we can expect it to reduce serum DHT levels by 71%.
So what if we choose a formula with even lower percentages of the active drug? A study on 0.25% topical finasteride showed just a 35% reduction in serum DHT levels versus 55% for the oral medication, and yet similar outcomes for hair regrowth in both oral and topical groups.[3]https://onlinelibrary.wiley.com/doi/10.1111/jdv.17738
So what if we go even lower? A study on 0.005% alcohol-based topical finasteride instructed participants to use the formulation twice daily, with 1 mL applied per session.[4]https://dx.doi.org/10.3109%2F09546639709160517 This led to no appreciable changes in serum DHT levels… meaning that for this ultra-low topical formulation, there was little-to-no systemic absorption (and presumably, few-to-no side effects). The good news? This group still experienced great hair growth outcomes.
So Just Reach for the Lowest Percentage, Yes?
It’s tempting from the above to infer we’ve reached the end of our story. As long as we reach for the lowest percentage of topical finasteride, we can be free from worries about the drug’s systemic effects. But, reality is a bit more complicated than that.
Systemic absorption isn’t just about dilution percentages. Topical finasteride’s systemic absorption actually depends on (at least) for variables:
- The drug’s formulation. Common carrier agents include alcohol and propylene glycol.
- The amount applied per use. The amount of topical finasteride applied to the scalp greatly varies.
- The application frequency. Applying the drug twice per day can more than double its effects on serum DHT.
- The days of application. Topical finasteride can take many days to “leak” into the blood before its final effects on serum DHT are realized.
For an example, see this figure from a 2014 study measuring how one versus two applications of 1 mL of 0.25% topical finasteride affected serum DHT levels.[5]https://www.ncbi.nlm.nih.gov/pubmed/25074865
Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen levels in healthy male volunteers. International journal of clinical pharmacology and therapeutics, 52(10), 842–849.
Over a 24-hour period, what were the findings?
- One application (i.e., 1 mL of 0.25% topical finasteride) lowered blood DHT levels by ~20%
- Two applications (i.e., 2 mL total) lowered blood DHT levels by ~70%
As such, we need to factor in these variables if we plan on sticking with topical finasteride for the long-term. Otherwise, we risk lowering our DHT levels to the same degree as 1 mg of oral finasteride, which would defeat the purpose of using the topical in the first place.
Topical Finasteride and Serum DHT: What Do The Clinical Studies Say?
Let’s take a closer look at some of the studies mentioned above. In addition to the percentage of active drug in the formula, there are a few more variables to understand. How much of the topical was applied, and which carrier agents were used?
The table below shows how even a .25% formula, for example, can reduce DHT in the serum by 24-70%
Based on this table, our total mg of daily finasteride exposure is probably the biggest factor in determining systemic leakage. And our total daily exposure (in mg) is a function of topical finasteride dilution (%) and the amount (mL) applied daily.
Knowing this, we can turn this table into a chart and sort it by mg of daily exposure. In doing so, we see a clear trend:
The good news? At both extremes of this chart – 0.091 mg and 2.275 mg daily – topical finasteride was shown to produce clinical results in improving hair parameters. Based on this, if we’re going for topical finasteride, we probably want to be prescribed topical finasteride solutions that net us daily exposure volumes of 0.228 mg and lower. After all, at 0.091 mg of topical finasteride daily, no systemic effects on DHT were observed.[6]https://www.tandfonline.com/doi/abs/10.3109/09546639709160517
In other words, low dilutions (i.e., 1-2 mL of 0.005% to 0.02% of topical finasteride) confer significant benefits but at reduced risks of side effects due to lower systemic absorption – provided that guidelines for daily amounts (in mL) are also followed.
So What’s the Perfect Amount and Formula?
Our analyses from member-submitted lab tests and the clinical data suggest that 10-15% of topical finasteride will enter the bloodstream, at least when it’s formulated with alcohol and/or propylene glycol as carrier ingredients (as most compounding pharmacies do). So, if we apply 1-2 mL of 0.005% to 0.02% topical finasteride, this might equate to just 0.01-0.03 mg of systemic drug exposure.
For the overwhelming majority of people, that’s not enough systemic leakage to significantly lower DHT levels in the body. But it is enough to produce great hair loss outcomes.Our partial analysis estimating systemic leakage of topical finasteride from the study: Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial (2021)
Join our Membership Program to get the full analysis.
Long-story short: stick to 1-2 mL of 0.005% to 0.02% topical finasteride solutions. This equates to roughly 0.1-0.2mg of daily finasteride exposure to the scalp. Any more than that, and there’s risk of significant systemic leakage, which defeats the purpose of using topical finasteride altogether.
How to Maximize Gains and Minimize Side Effects
Maximizing gains while minimizing the side effects of finasteride can be done, but it’s not a perfect science. We’ve outlined several tips below that we think might help. Due to individual variance, the most important step is to always start with testing.
1. Get Lab Tests for DHT
The goal with topical finasteride is to reduce the risk of side effects. To do this, we must minimize the amount of finasteride that leaks into the bloodstream. The best way to do this isn’t to rely on estimated metrics from clinical studies, but to collect personal data.
Get a serum DHT test before using the topical to establish a baseline. Then, DHT levels should be retested at one month to gauge just how much is going systemic. Testing is easier than most expect, and in our experience, the expenses are worth the peace of mind.
If future lab tests deviate from baseline, it’s an indication of just how much topical finasteride is going systemic. According to the clinical data, 30 days of application is more than enough time for finasteride to saturate at its maximum levels in the scalp and serum.
As such, measuring DHT levels after one month of finasteride use offers a great reference point to see if topical finasteride is impacting serum DHT levels. If needed, changes can be made to the application or use frequency depending on any changes to your blood levels of DHT.
2. Understand That DHT Levels Fluctuate Diurnally
Keep in mind, some fluctuation in DHT levels is normal. DHT levels fluctuate throughout the day and across seasons. As such, 15-20% differences across tests are normal and expected. Anything beyond 20% suggests that topical finasteride might be having slight systemic effects.
Because of this fluctuation, however, it’s important to get blood draws done at the same time of day – preferably in the morning and while in a fasted state. Also, try not to make drastic changes to diet, lifestyle, or environment prior to testing. Heavy drinking, deviations from a typical daily diet, the introduction of creatine powders, and/or sleep deprivation can all influence DHT levels and muddy test results. In the 3-5 days prior to the second test, try to keep things as they were when you first went in for testing.
3. Avoid supplementing with Quercetin and/or Creatine
Maintaining systemic DHT levels, while maximizing the effects of finasteride, isn’t just about the drug. There are other activities that could potentially affect DHT levels. Supplementing with quercetin and/or creatine is one common mistake that could impact results.
Studies on mice suggest that quercetin can inhibit the DHT-reducing effects of finasteride.[7]https://joe.bioscientifica.com/view/journals/joe/181/3/493.xmlWhile the translatability to humans has not yet been studied, the dosages used in these mouse models were comparable to what humans typically consume from quercetin supplements. As such, it may be best for those using finasteride to avoid this supplement.
When it comes to creatine, one study found that in training athletes, creatine supplementation increased serum DHT levels by over 70%.[8]https://pubmed.ncbi.nlm.nih.gov/19741313/ While the study was small, we have to reconcile these findings with the reality that for the overwhelming majority of training athletes, creatine is unnecessary. Bodybuilders can still look great without using it, and so can the every-day gym goer. Competitive bodybuilders are, however, a different story.
4. Set Realistic Expectations and Track Progress
Finasteride is the best-studied intervention for androgenic alopecia. In order to appreciate its full effects, consider the following:
- Know the timeline for results. Clinical studies show that finasteride improves pattern hair loss outcomes in 80-90% of men. Having said that, cosmetic degrees of hair regrowth don’t often occur until after 10-12 months of treatment. Moreover, the drug’s full effects typically take two years to manifest, with the biggest degrees of cosmetic regrowth occurring between year one and year two. As such, don’t expect any miracles in the first six months, it may take the full two years to see an effect.
- Track progress. It’s hard to know if something is improving without objective measurements. While not all of us have access to trichoscopic hair-counting equipment, most of us do have a smartphone, which can easily be used to take photos to track progress (particularly when the photosets are standardized). This can be done relatively effortlessly.
Following the tips above allows for the best possible shot at measurable hair growth results, while minimizing the potential effects of systemic DHT reduction.
Summary
Oral finasteride works great for male pattern hair loss, but by significantly reducing serum DHT, it can cause unwanted side effects.
Topical finasteride was developed as a solution, with a goal to reduce DHT in the scalp only. But when applied in high percentages or large amounts, it too, can go systemic and reduce serum DHT levels.
To minimize the side effects of finasteride without missing out on the benefits, stick to 1-2 mL of 0.005% to 0.02% topical finasteride solutions. This equates to roughly 0.1-0.2mg of daily finasteride exposure to the scalp.
The best way to ensure a reduced risk for side effects is to track serum DHT levels. Establish a baseline by taking one test before using finasteride, then test again one month later.
References[+]
References ↑1 https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑2 https://www.ncbi.nlm.nih.gov/pubmed/19172031 ↑3 https://onlinelibrary.wiley.com/doi/10.1111/jdv.17738 ↑4 https://dx.doi.org/10.3109%2F09546639709160517 ↑5 https://www.ncbi.nlm.nih.gov/pubmed/25074865 ↑6 https://www.tandfonline.com/doi/abs/10.3109/09546639709160517 ↑7 https://joe.bioscientifica.com/view/journals/joe/181/3/493.xml ↑8 https://pubmed.ncbi.nlm.nih.gov/19741313/ Dihydrotestosterone (DHT) is a potent androgen hormone that can significantly impact hair loss. Its activity, however, isn’t just restricted to the scalp. What’s more, hair loss is a multifactorial condition, with a wide range of causes. As such, the solution to hair loss is much more complicated than simply eliminating DHT.
Many natural or pharmaceutical therapies seek to reduce, or rather, rebalance DHT levels. While lowering DHT can slow or even reverse hair loss, the hormone itself plays vital roles in sexual function, mood regulation, and overall well-being. Therefore, the goal should not be total suppression, but rather modulation that considers the balance between hair health and systemic androgen levels.
In this article, we will rank both natural and pharmaceutical DHT blockers from weakest to strongest, based on research, effectiveness, and risk of side effects.
All-Natural Hair Topical
The top natural ingredients for hair growth, all in one serum.
Take the next step in your hair growth journey with a world-class natural serum. Ingredients, doses, & concentrations built by science.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Understanding DHT: Why Blocking It Helps (and Hurts)
DHT is derived from testosterone through the enzyme 5-alpha-reductase. Once formed, DHT binds to androgen receptors and plays a major role in androgenic alopecia (AGA). In hair follicles genetically sensitive to DHT, such as those on the crown and frontal scalp, the hormone acts as a signal for miniaturization. Over time, the follicle shrinks, producing thinner, shorter hairs until it eventually becomes dormant.[1]York, K., Meah, N., Bhoyrul, B., & Sinclair, R. (2020). A review of the treatment of male pattern hair loss. Expert Opinion on Pharmacotherapy. 21(5). 603-612. Available at: … Continue reading
Figure 1. 5α-reductase converts testosterone into dihydrotestosterone. Finasteride is a 5α-reductase inhibitor, meaning it prevents or reduces this activity. Image adapted from Figure 1.[2]Azizi, A., Mumin, N. H., & Shafqat, N. (2021). Phytochemicals with anti-5-alpha-reductase activity: A prospective for prostate cancer treatment. F1000Research. 10. 221. Available at: … Continue reading Image used under Creative Commons License.
By reducing DHT levels or blocking its receptor binding, therapies can interrupt this miniaturization cascade. The goal is to prevent DHT from triggering the cellular changes that cause follicles to shrink and enter a premature resting (telogen) phase. AGA is influenced not just by DHT levels but also by individual receptor sensitivity, local inflammation, and oxidative stress.
However, because of DHT’s wide-reaching activity, side effects of its reduction can include sexual dysfunction, decreased motivation, and mood changes, especially in individuals who are more hormonally sensitive. As such, careful consideration of different therapies and their potential impacts is essential if you choose to use a DHT-blocking treatment.[3]Traish, A. M., Melcangi, R. C., Bortolato, M., Garcia-Segura, L. M., & Zitzmann, M. (2015). Adverse effects of 5α-reductase inhibitors: what do we know, don’t know, and need to know?. Reviews … Continue reading
Ranking the DHT Blockers: From Mild to Potent
We’ll rank DHT blockers based on their potency, as well as on the quality of evidence supporting their use for hair loss. You can learn more about our treatment metrics, including evidence quality, in our article.
Tier 1 – Mild / Natural DHT Blockers
Natural treatments, typically based on oils and herbs, are generally more gentle than pharmaceutical options. They tend to be less potent, but have far fewer side effects and are not typically subject to the same stringent regulation as pharmaceuticals.
EGCG (Evidence Quality – 6%)
Epigallocatechin gallate (EGCG), a catechin compound found in green tea, is another naturally derived compound that has attracted interest for its potential to influence hair follicle biology. As a potent antioxidant and anti-inflammatory molecule, EGCG helps protect follicular cells from oxidative stress and inflammation, both of which are exacerbated by androgen activity.
Figure 2: Molecular structure of EGCG. Adapted from:[4]Wikipedia. (no date). Epigallocatechin gallate. Available at: https://en.wikipedia.org/wiki/Epigallocatechin_gallate (Accessed: November 2025) Image used under Creative Commons License
EGCG appears to enhance cell-survival pathways such as Akt and Erk, supporting follicular cell proliferation while suppressing apoptotic signals that can trigger hair follicle regression.[5]Kwon, O. S., Han, J. H., Yoo, H. G., Chung, J. H., Cho, K. H., Eun, H. C., & Kim, K. H. (2007). Human hair growth enhancement in vitro by green tea epigallocatechin-3-gallate (EGCG). … Continue reading
Laboratory studies also indicate that EGCG can partially inhibit 5AR, reducing the local generation of DHT.[6]Hiipakka, R. A., Zhang, H. Z., Dai, W., Dai, Q., & Liao, S. (2002). Structure–activity relationships for inhibition of human 5α-reductases by polyphenols. Biochemical Pharmacology. 63(6). … Continue reading However, while this study showed that EGCG can inhibit 5AR in cell-free experiments, it did not have the same impact in cells. This suggests that it may not be able to enter cells and would have reduced impacts in humans.
This seems to be the case, and clinical evidence for EGCG in the treatment of hair loss remains limited. A small pilot study of topical EGCG found no visible changes in hair, although this research was conducted in people with no hair loss disorders.[7]Kwon, O. S., Han, J. H., Yoo, H. G., Chung, J. H., Cho, K. H., Eun, H. C., & Kim, K. H. (2007). Human hair growth enhancement in vitro by green tea epigallocatechin-3-gallate (EGCG). … Continue reading Despite the lack of large-scale human trials, molecular findings do suggest EGCG contributes meaningfully to the cellular environment that supports hair growth and protection.
Catechins from green tea infusions are generally well tolerated. However, supplements should be used cautiously at high doses, particularly above 800 mg per day, as they have been linked to liver enzyme elevations in sensitive individuals.[8]EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS), Younes, M., Aggett, P., Aguilar, F., Crebelli, R., Dusemund, B., Filipič, M., Frutos, M. J., Galtier, P., Gott, D., … Continue reading
You can read more about the evidence for using EGCG for hair loss in our article here.
Pumpkin Seed Oil (Evidence Quality – 49%)
Pumpkin seed oil (PSO) is one of the most extensively studied non-pharmaceutical treatments for hair loss. In general, it is mild and well-tolerated. Its effects appear to arise from its profile of phytosterols and essential fatty acids, such as linoleic and oleic acid. These compounds have been shown to exhibit mild 5AR inhibitory activity in laboratory studies, potentially reducing local DHT formation in scalp tissues.[9]Azizi, A., Mumin, N. H., & Shafqat, N. (2021). Phytochemicals with anti 5-alpha-reductase activity: a prospective for prostate cancer treatment. F1000Research. 10. 221. Available at: … Continue reading,[10]Liang, T., & Liao, S. (1992). Inhibition of steroid 5α-reductase by specific aliphatic unsaturated fatty acids. Biochemical Journal. 285(2). 557-562. Available at: … Continue reading
Beyond hormonal modulation, pumpkin seed oil provides antioxidant and anti-inflammatory support, which can help protect hair follicles from oxidative damage and microinflammation – two key contributors to progressive miniaturization.[11]Fahim, A. T., Abd-El Fattah, A. A., Agha, A. M., & Gad, M. Z. (1995). Effect of pumpkin-seed oil on the level of free radical scavengers induced during adjuvant-arthritis in rats. Pharmacological … Continue reading
A clinical trial carried out in Korea made waves in 2014 when it reported a 40% increase in hair count after 24 weeks. However, the formulation, Octa-Sabal Plus, included multiple additional herbal compounds, alongside pumpkin seed powder, not oil. This means we can’t attribute the growth reported to pumpkin seed oil.[12]Cho, Y. H., Lee, S. Y., Jeong, D. W., Choi, E. J., Kim, Y. J., Lee, J. G., & Cha, H. S. (2014). Effect of pumpkin seed oil on hair growth in men with androgenetic alopecia: a randomized, … Continue reading
You can read more about the controversial PSO study here.
Cold-pressed pumpkin seed oil is typically taken orally at doses ranging from 400 to 1000 mg daily, with results accumulating gradually over several months. The compound is generally well tolerated, with gastrointestinal discomfort being rare and mild.[13]Hong, H., Kim, C. S., & Maeng, S. (2009). Effects of pumpkin seed oil and saw palmetto oil in Korean men with symptomatic benign prostatic hyperplasia. Nutrition Research and Practice. 3(4). … Continue reading Although PSO alone is unlikely to reverse established AGA, it may serve as a gentle adjunct for individuals seeking natural or preventive strategies to support long-term scalp and hair health.
Saw Palmetto (Evidence Quality – 54%)
Saw palmetto (Serenoa repens) is one of the most widely studied natural 5AR inhibitors and is sometimes called “nature’s finasteride.” It appears to reduce DHT by inhibiting Type II 5AR, reducing the binding of DHT to androgen receptor sites and increasing the conversion of DHT to a weaker metabolite called androstanediol.[14]Suzuki, M., Ito, Y., Fujino, T., Abe, M., Umegaki, K., Onoue, S., Noguchi, H., & Yamada, S. (2009). Pharmacological effects of saw palmetto extract in the lower urinary tract. *Acta … Continue reading
Importantly, saw palmetto appears to reduce DHT selectively, with research showing decreases in DHT in the prostate but not in the blood. As such, it may cause fewer side effects than systemic DHT reducers.[15]Fagelman, E., & Lowe, F. C. (2001). Saw palmetto berry as a treatment for BPH. Reviews in Urology. 3(3). 134-138. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1476047/
Clinical studies have shown modest impacts of saw palmetto on hair loss. In one study, participants using oral extracts over several months showed reductions in hair shedding, increases in hair density, and subjective improvements in scalp coverage and hair quality.{ Importantly, however, this was an open-label study, where participants and researchers both knew what product they were receiving, so is not as strong a form of evidence as a randomzied controlled trial (RCT).[16]Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., & Carlesimo, M. (2012). Comparative effectiveness of finasteride vs Serenoa repens in male androgenetic alopecia: a … Continue reading
In a recent RCT, a standardized, proprietary saw palmetto extract was given as either a 400mg oral capsule or 5 ml of topical formulation and compared to relevant controls. The study found a significant reduction in hair shedding in both oral and topical groups compared to their controls, alongside an increase in hair density of 5% and 7.5% respectively. However, this study also had significant drawbacks. For example, measurements of hair density were not well defined and may therefore include vellus hairs, which do not contribute to visible hair. Despite this, the study does provide some support for the use of saw palmetto in decreasing hair shedding and increasing hair thickness.[17]Sudeep, H. V., Rashmi, S., Jestin, T. V., Richards, A., Gouthamchandra, K., & Shyamprasad, K. (2023). Oral and topical administration of a standardized saw palmetto oil reduces hair fall and … Continue reading
Evidence from benign prostatic hyperplasia suggests that an oral dose of 320 mg per day is optimal, with increases in dose above that resulting in no further improvements. For topical use, concentrations around 0.5 – 1% applied once or twice daily are common. Both routes are well tolerated, with infrequent reports of mild gastrointestinal discomfort or skin irritation.
You can read about the differences between saw palmetto and finasteride in our article here.
Tier 2 – Over-the-Counter Pharmaceutical Options
Over-the-Counter Pharmaceutical Options bridge the gap between gentle natural therapies and prescription-strength DHT blockers, potentially offering clinically supported benefits with relatively low systemic risk.
Ketoconazole Shampoo (Evidence Quality – 54%)
Originally developed as an antifungal agent, ketoconazole helps normalize the scalp environment by reducing inflammation, fungal overgrowth, and excess sebum, all of which can worsen AGA. Beyond its antifungal activity, laboratory and clinical research suggest that ketoconazole exerts mild local antiandrogenic effects, likely through partial inhibition of 5AR and modulation of DHT activity within the scalp.
Clinical evidence for men is stronger than for women. One 21-month study using 2% ketoconazole shampoo showed progressive improvement in hair shaft diameter and percentage of hairs in the growth (anagen) phase, reversing the slow decline seen in untreated AGA.[18]Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., & Piérard, G. E. (1998). Ketoconazole shampoo: effect of long-term use in androgenic alopecia. Dermatology. 196(4). 474-477. Available … Continue reading
Another study, which compared Ketoconazole with other shampoos, found that 1% ketoconazole reduced hair shedding by 17%, improved anagen hair by 4.9%, and lowered scalp sebum levels.[19]Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., & Piérard, G. E. (2002). Nudging hair shedding by antidandruff shampoos: A comparison of 1% ketoconazole, 1% piroctone … Continue reading Studies also show that ketoconazole performs well as an adjunct therapy, with synergistic effects when combined with finasteride.[20]Perez, B. S. H. (2004). Ketoconazole as an adjunct to finasteride in the treatment of androgenetic alopecia in men. Medical Hypotheses. 62(1). 112-115. Available at: … Continue reading
Evidence in women is limited, with only one published study investigating ketoconazole solution, rather than shampoo. They compared 2% ketoconazole to 2% minoxidil and reported meaningful regrowth in both groups, with ketoconazole producing improvements by the six-month mark and causing far fewer side effects.[21]El-Garf, A., Mohie, M., & Salah, E. (2019). Trichogenic effect of topical ketoconazole versus minoxidil 2% in female pattern hair loss: a clinical and trichoscopic evaluation. Biomedical … Continue reading
Ketoconazole is generally well tolerated. Mild irritation, dryness, or temporary scalp tightness may occur, especially with 2% formulations or frequent use. Most regimens recommend applying it 2–3 times per week, leaving the shampoo on the scalp for 3–5 minutes before rinsing.
You can read our breakdown of the research into ketoconazole shampoos here.
You can also read about why 2% ketoconazole might be a better choice than 1% here.
Tier 3 – Prescription Pharmaceutical DHT Blockers
Prescription Pharmaceutical DHT Blockers represent the most potent and clinically validated options, offering stronger and more predictable suppression of DHT than natural therapies but with a greater need for careful monitoring of side effects.
Topical Finasteride (Evidence Quality – 69%)
Topical finasteride represents a new, more targeted approach to DHT inhibition, delivering the drug directly to the scalp while aiming to minimize systemic exposure. Like its oral counterpart, it blocks the Type II 5-alpha reductase enzyme. However, by focusing this activity locally within the hair follicle, topical formulations can achieve substantial reductions in scalp DHT while maintaining serum levels that are a fraction of those seen with oral dosing. This distinction is clinically important for individuals concerned about systemic side effects such as decreased libido or hormonal shifts.
Clinical research has consistently shown that topical finasteride can achieve similar improvements in hair density and regrowth as oral therapy. One 24-week study showed a 34.5% decrease in DHT, compared to 55.6% for oral finasteride. However, plasma concentrations were 100 times lower, indicating that systemic exposure is greatly reduced.[22]Piraccini, B. M., Blume-Peytavi, U., Scarci, F., Jansat, J. M., Falqués, M., Otero, R., Tamarit, M. L., Galván, J., Tebbs, V., Massana, È., on behalf of the Topical Finasteride Study Group. … Continue reading
Topical finasteride is particularly appealing to men who respond well to oral therapy but wish to lower the risk of systemic adverse effects. When applied once daily at concentrations between 0.1% and 0.25%, it can stabilize shedding within four to six months, with visible improvements generally continuing over the course of a year.[23]Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a … Continue reading For optimal results, it can be combined with topical minoxidil to stimulate regrowth through complementary pathways.[24]Wang, H., Chen, B., & Hong, X. (2020). The efficacy and safety of finasteride combined with topical minoxidil for androgenetic alopecia: A systematic review and meta-analysis. *Journal of … Continue reading
Our members can find the ultimate guide to finasteride here.
Dutasteride (Evidence Quality – 77%)
Dutasteride is the most potent 5AR inhibitor currently available, targeting both Type I and Type II isoenzymes. This dual inhibition results in serum DHT reductions of up to 90 – 95%, creating a profoundly anti-androgenic environment within scalp follicles.[25]Roehrborn, C. G., Marks, L. S., Fenter, T., Freedman, S., Tuttle, J., Gittleman, M., Morrill, B., & Wolford, E. T. (2004). Efficacy and safety of dutasteride in the four-year treatment of men … Continue reading Clinically, this translates into faster and often more pronounced regrowth compared to finasteride, particularly in individuals with advanced hair thinning.
Because dutasteride suppresses DHT more comprehensively, it may pose a slightly higher risk of hormonal side effects such as gynecomastia or reduced libido. However, head-to-head studies have suggested superior efficacy for dutasteride in both short-term and long-term endpoints.
A meta-analysis, which combines results from multiple studies, indicated that dutasteride showed better efficacy in treating AGA.[26]Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., & Cui, Y. (2019). The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and … Continue reading These results suggest that dutasteride is ~3 times more potent than finasteride in blocking Type II 5α-reductase and up to 100 times more potent against Type I. This dual inhibition reduces scalp DHT levels by over 50%, compared to approximately 41% with finasteride, leading to greater prevention of follicular miniaturization and enhanced regrowth response with similar levels of adverse effects.
Dutasteride has a relatively low evidence quality score compared to finasteride, though this is largely due to there being fewer studies. The studies that do investigate dutasteride have shown higher potency and effectiveness.
Topical dutasteride formulations are now under active investigation. Concentrations ranging from 0.01% to 0.05% have produced significant gains in target-area hair counts, with studies suggesting that improvements associated with oral formulations can be achieved without drops in serum DHT.[27]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., KSSVV, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo and active controlled Phase II study to … Continue reading
Figure 3. Topical Dutasteride supports hair regrowth. Adapted from Figure 2.[28]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., KSSVV, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo and active controlled Phase II study to … Continue reading Image used under Creative Commons License
A 2018 randomized clinical study further demonstrated that topical dutasteride (0.02%) combined with microneedling significantly improved hair shaft thickness and patient satisfaction compared with microneedling alone, without notable systemic absorption or hormonal side effects. These findings suggest that localized delivery of dutasteride may enhance follicular response while minimizing systemic exposure and related adverse effects.[29]Nada, E.A., El Sharkawy, R.E.E.D., Abd El-Maged, W. M., & Abo Elmagd, M. A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. … Continue reading
Similarly, research published in 2024 looked into the impact of microneedling. However, in this study, topical dutasteride was combined with minoxidil. They found similar improvements in hair density and hair width in participants who had topical dutasteride and minoxidil treatment to those in patients who had oral dutasteride and topical minoxidil.[30]Abu Obeid, M. N., Abdel Fattah, N. S., Elfangary, M. M., & Al Husseni, R. M. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through … Continue reading These results suggest that, in combination with other therapies (minoxidil or microneedling), topical dutasteride may be able to produce similar results to oral formulations but with a significantly lower chance of side effects.
A newly published Phase II trial has also drawn significant attention because it is the first study to test topical dutasteride alone, without microneedling. The results appear impressive: all topical doses improved hair parameters versus placebo, showed minimal impact on serum DHT (suggesting good scalp localization), and 1 ml daily of 0.05% topical dutasteride reportedly outperformed oral finasteride in key regrowth metrics.[31]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading
However, the study reports unusually high baseline hair counts, as well as inconsistencies in the placement and size of the 1 cm² target areas used for serial hair counts. These issues can dramatically skew results, creating the illusion of improvement even when none exists. Real-world data from users of low-dose topical dutasteride (0.01–0.02% applied daily or near-daily) also paint a more modest picture: good localization and serum DHT stability, but primarily maintenance rather than regrowth exceeding oral finasteride
Oral Finasteride (Evidence Quality – 99%)
As you can see from our evidence quality score, oral finasteride remains the most clinically validated pharmacological treatment for AGA. It acts as a selective inhibitor of the Type II 5AR enzyme, reducing systemic and scalp DHT levels by approximately 60–70%.[32]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W. P., Cotterill, P. C., Thiboutot, D. M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S. J., Griffin, E. I., Weiss, D., … Continue reading This substantial suppression slows or halts follicular miniaturization, prolongs the growth (anagen) phase, and enables partial regrowth in many users. The standard 1 mg daily dose for AGA has been extensively studied and represents the optimal balance between efficacy and safety for most men.
Clinical trials have consistently confirmed the effectiveness of finasteride in both the short and long term. A 4-year, randomized study found that 1 mg of finasteride led to increased hair weight (46% vs placebo) and hair count (20% vs. placebo).[33]Price, V. H., Menefee, E., Sanchez, M., & Kaufman, K. D. (2006). Changes in hair weight in men with androgenetic alopecia after treatment with finasteride (1 mg daily): three- and 4-year results. … Continue reading
You can find an in-depth overview of the clinical studies on finasteride here.
While highly effective, oral finasteride is not without potential drawbacks. A small percentage of users report side effects related to sexual function, including reduced libido, erectile difficulty, and diminished ejaculate volume. Most of these effects are mild and resolve upon discontinuation, although persistent symptoms have been reported in rare cases. Some users also report changes in mood or motivation, likely reflecting individual hormonal sensitivity. For those wishing to minimize systemic exposure, transitioning to topical finasteride or titrating to a lower dose (e.g., 0.25-0.5 mg daily) may be a suitable option.[34]Gupta, A. K., Venkataraman, M., Talukder, M., & Bamimore, M. A. (2022). Finasteride for hair loss: a review. Journal of Dermatological Treatment. 33(4). 1938-1946. Available at: … Continue reading
There have also been reports of a condition that’s been named ‘post-finasteride syndrome’. While not widely accepted by the medical community, similar symptoms have been reported in a number of individuals who are taking finasteride. These symptoms can include sexual dysfunction, such as loss of libido and erectile dysfunction, neuropsychiatric symptoms like depression and anxiety, and physical changes like muscle atrophy and fatigue. Importantly, there have been cases where these symptoms have persisted after users have stopped taking the drug.
While the authors who published the original article on post-finasteride syndrome do highlight the lack of consistent evidence for persistent side effects found in clinical trials, they suggest that finasteride may act to worsen symptoms associated with sexual dysfunction and psychiatric conditions for some users who already have these issues.[35]Pereira, A. F. J. R., & Coelho, T. O. A. (2020). Post-finasteride syndrome. Anais Brasileiros de Dermatologia. 95(3). 271-277. Available at: https://doi.org/10.1016/j.abd.2020.02.001
Overall, oral finasteride remains the gold standard for DHT suppression, providing robust and predictable results when used consistently. It’s also important to consider the potential side effects when choosing to take finasteride, and consult with your doctor if symptoms arise.
Honourable Mention – Pyrilutamide
Pyrilutamide (KX-826)
Pyrilutamide takes a different approach to androgen modulation. Rather than reducing DHT production, it acts as a topical androgen receptor antagonist, blocking DHT and testosterone from binding to their receptors in the follicle. By interfering with receptor activation, pyrilutamide aims to suppress the downstream gene expression that leads to follicular miniaturization.[36]Koralewicz, M. M., & Szatkowska, O. A. (2024, July). Topical solutions for androgenetic alopecia: evaluating efficacy and safety. Forum Dermatologicum. 10(3). 61-70. Available at: … Continue reading
Clinical data show encouraging signals. Trials conducted in China and the United States suggest that topical formulations applied once or twice daily can produce measurable increases in hair counts over several months. Reported side effects are primarily mild local dermatitis, and systemic exposure appears very low. However, many of these results remain unpublished in peer-reviewed journals and have failed to demonstrate statistical superiority over placebo for some measurements.[37]Kintor Pharmaceuticals. (2022). Developing Novel Drugs and Commercialization Platform. Available at: … Continue reading,[38]Kintor Pharmaceutical Limited. (2023, March 28). Kintor Pharma Announces Completion of Subject Enrollment in Phase III Clinical Trial for KX-826 for Treatment of Male Androgenetic Alopecia in China. … Continue reading Because these reports have not been published in peer-reviewed journals, they can only currently be considered pre-releases, rather than academic papers. The absence of published, peer-reviewed, blinded, controlled data means that we can’t draw firm conclusions on pyrilutamide, even in the wake of the many press releases that Kintor has published.
For now, pyrilutamide should be viewed as an experimental therapy. Its mechanism is compelling and localized, offering a theoretical advantage over systemic antiandrogens. Still, robust, peer-reviewed clinical trials confirming efficacy and long-term safety are needed before it can be considered a mainstream treatment. Those interested in exploring it should do so only under medical supervision or within the context of formal clinical research.
Final Thoughts
DHT blockers span a broad spectrum, ranging from gentle, naturally derived modulators like pumpkin seed oil and EGCG to powerful prescription therapies such as finasteride and dutasteride. Natural options may improve scalp health and have a modest influence on hormonal activity, but their effects are typically gradual and limited. In contrast, pharmacological inhibitors act directly on the enzymatic pathways responsible for DHT synthesis, offering stronger and more predictable outcomes. Topical finasteride has emerged as a compelling middle ground, delivering scalp-level efficacy with minimal systemic impact, while dutasteride represents the upper limit of potency for those requiring more aggressive intervention.
Ultimately, the “best” DHT blocker depends on individual tolerance, goals, and the stage of hair loss. A well-balanced regimen typically combines a DHT inhibitor with a growth stimulant, such as minoxidil, alongside scalp care and lifestyle modifications. With consistent use and proper medical guidance, these approaches can collectively slow, halt, or even reverse the progression of AGA, helping individuals maintain fuller, healthier hair over time.
References[+]
References ↑1 York, K., Meah, N., Bhoyrul, B., & Sinclair, R. (2020). A review of the treatment of male pattern hair loss. Expert Opinion on Pharmacotherapy. 21(5). 603-612. Available at: https://doi.org/10.1080/14656566.2020.1721463 ↑2 Azizi, A., Mumin, N. H., & Shafqat, N. (2021). Phytochemicals with anti-5-alpha-reductase activity: A prospective for prostate cancer treatment. F1000Research. 10. 221. Available at: https://doi.org/10.12688/f1000research.51066.3 ↑3 Traish, A. M., Melcangi, R. C., Bortolato, M., Garcia-Segura, L. M., & Zitzmann, M. (2015). Adverse effects of 5α-reductase inhibitors: what do we know, don’t know, and need to know?. Reviews in Endocrine and Metabolic Disorders. 16(3). 177-198. Available at: https://doi.org/10.1007/s11154-015-9319-y ↑4 Wikipedia. (no date). Epigallocatechin gallate. Available at: https://en.wikipedia.org/wiki/Epigallocatechin_gallate (Accessed: November 2025) ↑5, ↑7 Kwon, O. S., Han, J. H., Yoo, H. G., Chung, J. H., Cho, K. H., Eun, H. C., & Kim, K. H. (2007). Human hair growth enhancement in vitro by green tea epigallocatechin-3-gallate (EGCG). Phytomedicine. 14(7-8). 551-555. Available at: https://doi.org/10.1016/j.phymed.2006.11.003 ↑6 Hiipakka, R. A., Zhang, H. Z., Dai, W., Dai, Q., & Liao, S. (2002). Structure–activity relationships for inhibition of human 5α-reductases by polyphenols. Biochemical Pharmacology. 63(6). 1165-1176. Available at: https://doi.org/10.1016/S0006-2952(02)00848-1 ↑8 EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS), Younes, M., Aggett, P., Aguilar, F., Crebelli, R., Dusemund, B., Filipič, M., Frutos, M. J., Galtier, P., Gott, D., Gundert-Remy, U., Kuhnle, G. G., Lambré, C., Leblanc, J.-C., Lillegaard, I. T., Moldeus, P., Mortensen, A., Oskarsson, A., Stankovic, I., Waalkens-Berendsen, I., Woutersen, R. A., Andrade, R. J., Fortes, C., Mosesso, P., Restani, P., Arcella, D., Pizzo, F., Smeraldi, C., & Wright, M. (2018). Scientific opinion on the safety of green tea catechins. *EFSA Journal.* 16(4). e05239. Available at: https://doi.org/10.2903/j.efsa.2018.5239 ↑9 Azizi, A., Mumin, N. H., & Shafqat, N. (2021). Phytochemicals with anti 5-alpha-reductase activity: a prospective for prostate cancer treatment. F1000Research. 10. 221. Available at: https://doi.org/10.12688/f1000research.51066.3 ↑10 Liang, T., & Liao, S. (1992). Inhibition of steroid 5α-reductase by specific aliphatic unsaturated fatty acids. Biochemical Journal. 285(2). 557-562. Available at: https://doi.org/10.1042/bj2850557 ↑11 Fahim, A. T., Abd-El Fattah, A. A., Agha, A. M., & Gad, M. Z. (1995). Effect of pumpkin-seed oil on the level of free radical scavengers induced during adjuvant-arthritis in rats. Pharmacological Research. 31(1). 73-79. Available at: https://doi.org/10.1016/1043-6618(95)80051-4 ↑12 Cho, Y. H., Lee, S. Y., Jeong, D. W., Choi, E. J., Kim, Y. J., Lee, J. G., & Cha, H. S. (2014). Effect of pumpkin seed oil on hair growth in men with androgenetic alopecia: a randomized, double-blind, placebo-controlled trial. *Evidence-Based Complementary and Alternative Medicine.* 2014(1). 549721. Available at: https://doi.org/10.1155/2014/549721 ↑13 Hong, H., Kim, C. S., & Maeng, S. (2009). Effects of pumpkin seed oil and saw palmetto oil in Korean men with symptomatic benign prostatic hyperplasia. Nutrition Research and Practice. 3(4). 323-327. Available at: https://doi.org/10.4162/nrp.2009.3.4.323 ↑14 Suzuki, M., Ito, Y., Fujino, T., Abe, M., Umegaki, K., Onoue, S., Noguchi, H., & Yamada, S. (2009). Pharmacological effects of saw palmetto extract in the lower urinary tract. *Acta Pharmacologica Sinica.* 30(3). 271-281. Available at: https://doi.org/10.1038/aps.2009.1 ↑15 Fagelman, E., & Lowe, F. C. (2001). Saw palmetto berry as a treatment for BPH. Reviews in Urology. 3(3). 134-138. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1476047/ ↑16 Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., & Carlesimo, M. (2012). Comparative effectiveness of finasteride vs Serenoa repens in male androgenetic alopecia: a two-year study. International Journal of Immunopathology and Pharmacology. 25(4). 1167-1173. Available at: https://doi.org/10.1177/039463201202500435 ↑17 Sudeep, H. V., Rashmi, S., Jestin, T. V., Richards, A., Gouthamchandra, K., & Shyamprasad, K. (2023). Oral and topical administration of a standardized saw palmetto oil reduces hair fall and improves hair growth in androgenetic alopecia subjects – a 16-week randomized, placebo-controlled study. Clinical, Cosmetic and Investigational Dermatology. 16. 3251-3266. Available at: https://doi.org/10.2147/CCID.S380214 ↑18 Piérard-Franchimont, C., De Doncker, P., Cauwenbergh, G., & Piérard, G. E. (1998). Ketoconazole shampoo: effect of long-term use in androgenic alopecia. Dermatology. 196(4). 474-477. Available at: https://doi.org/10.1159/000017954 ↑19 Piérard-Franchimont, C., Goffin, V., Henry, F., Uhoda, I., Braham, C., & Piérard, G. E. (2002). Nudging hair shedding by antidandruff shampoos: A comparison of 1% ketoconazole, 1% piroctone olamine and 1% zinc pyrithione formulations. International Journal of Cosmetic Science. 24(5). 249-256. Available at: https://doi.org/10.1046/j.1467-2494.2002.00145.x ↑20 Perez, B. S. H. (2004). Ketoconazole as an adjunct to finasteride in the treatment of androgenetic alopecia in men. Medical Hypotheses. 62(1). 112-115. Available at: https://doi.org/10.1016/S0306-9877(03)00264-0 ↑21 El-Garf, A., Mohie, M., & Salah, E. (2019). Trichogenic effect of topical ketoconazole versus minoxidil 2% in female pattern hair loss: a clinical and trichoscopic evaluation. Biomedical Dermatology. 3(1). 1-8. Available at: https://doi.org/10.1186/s41702-019-0046-y ↑22 Piraccini, B. M., Blume-Peytavi, U., Scarci, F., Jansat, J. M., Falqués, M., Otero, R., Tamarit, M. L., Galván, J., Tebbs, V., Massana, È., on behalf of the Topical Finasteride Study Group. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology and Venereology. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑23 Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia. Preliminary evaluations after a 16-month therapy course. Journal of Dermatological Treatment. 8(3). 189-192. Available at: https://doi.org/10.3109/09546639709160517 ↑24 Wang, H., Chen, B., & Hong, X. (2020). The efficacy and safety of finasteride combined with topical minoxidil for androgenetic alopecia: A systematic review and meta-analysis. *Journal of Dermatological Treatment.* Available at: https://doi.org/10.1007/s00403-020-02100-6 ↑25 Roehrborn, C. G., Marks, L. S., Fenter, T., Freedman, S., Tuttle, J., Gittleman, M., Morrill, B., & Wolford, E. T. (2004). Efficacy and safety of dutasteride in the four-year treatment of men with benign prostatic hyperplasia. Urology. 63(4). 709-715. Available at: https://doi.org/10.1016/j.urology.2004.01.001 ↑26 Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., & Cui, Y. (2019). The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and meta-analysis. Clinical Interventions in Aging. 14. 399-406. Available at: https://doi.org/10.2147/CIA.S192435 ↑27 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., KSSVV, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo and active controlled Phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑28 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., KSSVV, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo and active controlled Phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑29 Nada, E.A., El Sharkawy, R.E.E.D., Abd El-Maged, W. M., & Abo Elmagd, M. A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). 387-400. Available at: https://doi.org/10.21608/smj.2018.42083 ↑30 Abu Obeid, M. N., Abdel Fattah, N. S., Elfangary, M. M., & Al Husseni, R. M. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 mg) in Treatment of Androgenetic Alopecia. QJM: An International Journal of Medicine. 117(Supplement_2). hcae175-207. Available at: https://doi.org/10.1093/qjmed/hcae175.207 ↑31 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. *Cureus.* 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑32 Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W. P., Cotterill, P. C., Thiboutot, D. M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S. J., Griffin, E. I., Weiss, D., Carrington, P., Gencheff, C., Cole, G. W., Pariser, D. M., Epstein, E. S., Tanaka, W. K., Dallob, A. L., Vandormael, K., Geissler, L. A., & Waldstreicher, J. (1999). The effects of finasteride on scalp skin and serum androgen levels in men with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 41(4). 550-554. Available at: https://doi.org/10.1016/S0190-9622(99)80051-6 ↑33 Price, V. H., Menefee, E., Sanchez, M., & Kaufman, K. D. (2006). Changes in hair weight in men with androgenetic alopecia after treatment with finasteride (1 mg daily): three- and 4-year results. Journal of the American Academy of Dermatology. 55(1). 71-74. Available at: https://doi.org/10.1016/j.jaad.2005.07.001 ↑34 Gupta, A. K., Venkataraman, M., Talukder, M., & Bamimore, M. A. (2022). Finasteride for hair loss: a review. Journal of Dermatological Treatment. 33(4). 1938-1946. Available at: https://doi.org/10.1080/09546634.2021.1959506 ↑35 Pereira, A. F. J. R., & Coelho, T. O. A. (2020). Post-finasteride syndrome. Anais Brasileiros de Dermatologia. 95(3). 271-277. Available at: https://doi.org/10.1016/j.abd.2020.02.001 ↑36 Koralewicz, M. M., & Szatkowska, O. A. (2024, July). Topical solutions for androgenetic alopecia: evaluating efficacy and safety. Forum Dermatologicum. 10(3). 61-70. Available at: https://doi.org/10.5603/fd.101208 ↑37 Kintor Pharmaceuticals. (2022). Developing Novel Drugs and Commercialization Platform. Available at: https://docs.publicnow.com/viewDoc?hash_primary=BFD6444A4669BD4BE0683A8BCC8B5E80F3D7E430 (Accessed: October 2025)
↑38 Kintor Pharmaceutical Limited. (2023, March 28). Kintor Pharma Announces Completion of Subject Enrollment in Phase III Clinical Trial for KX-826 for Treatment of Male Androgenetic Alopecia in China. Available at: https://www.prnewswire.com/news-releases/kintor-pharma-announced-the-primary-endpoint-of-phase-ii-clinical-study-for-kx-826s-treatment-of-female-androgenetic-alopecia-in-china-was-met-301691321.html (Accessed: November 2025)
Oral finasteride is one of the most widely prescribed treatments for androgenic alopecia (AGA). The drug is a type II 5a-reductase inhibitor, which means it helps reduce dihydrotestosterone (DHT) – the major hormone implicated in pattern hair loss.
At the same time, finasteride isn’t the most powerful DHT-reducing drug available. In fact, there’s a drug that can reduce up to 50% more DHT than finasteride. It’s called dutasteride – and for those who are interested in unlocking another level of hair regrowth, it might be the right choice for them.
So, how much hair regrowth can we expect from dutasteride? Does dutasteride outperform finasteride? How does this drug stack up in terms of side effects? And is there anything else we should know before making the “leap” to a more powerful DHT reducer?
This ultimate guide uncovers the answers.
Interestingly, despite dutasteride’s stronger DHT-reducing effects, this drug doesn’t seem to come with an increased risk of cognitive or sexual side effects – at least when compared to finasteride. In fact, some research indicates that dutasteride might be a better option altogether.
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Highlights
- Effort: Low (drug, taken orally, once daily)
- Expectations: Regrowth observed as early as 3 months.
- Regrowth rate: 5-11% in 6 months, depending on the dosage. Some studies suggest that dutasteride leads to twice the hair count increase as finasteride over a six-month timeframe, though it’s unclear whether this is two
- Response rate: 80-95%, depending on the dosage.
- Cost: $14-$160 per month.
- Problems: Results contingent upon lifelong use; if taken at higher dosages and for long periods, DHT may remain suppressed 6+ months after stopping treatment; risk of side effects similar to finasteride
Key Takeaways
Dutasteride is a drug used most commonly in the treatment of benign prostate hyperplasia. But it’s also used off-label as a treatment for male pattern hair loss.
Dutasteride is a dual 5α-reductase inhibitor, meaning that the drug inhibits both the type I and type II isoforms of the 5α-reductase enzyme. This makes it unique from finasteride, a drug that only inhibits type II 5-alpha reductase. And since dutasteride inhibits more 5-alpha reductase, it can reduce DHT levels much more effectively than finasteride. Specifically, dutasteride appears to reduce systemic DHT levels by 92-96% and scalp DHT levels by 51-79%; whereas finasteride reduces systemic DHT by 60-70% and scalp DHT by 50-60%.
When it comes to hair regrowth, short-term studies suggest that dutasteride is anywhere from 2-5 times more effective than finasteride (depending on the dose). However, the studies comparing dutasteride and finasteride are generally over six-month periods. This may not make for a fair comparison, as finasteride tends to take two years to achieve its full effect.
Despite being more effective, dutasteride doesn’t seem to come with an increased risk of side effects relative to finasteride. In fact, in some instances, dutasteride’s risk of side effects is actually lower than that of finasteride… despite a more substantial decrease in systemic DHT. While it’s debated why this is the case, one explanation might involve dutasteride’s molecular weight, which is much heavier than finasteride, so much so that the drug cannot likely cross the blood-brain barrier.
If you opt for dutasteride, research suggests that for treating pattern hair loss, a dosage of 0.5 mg daily is best. At this dose, dutasteride seems to outperform finasteride and not result in long-term DHT suppression after stopping the medication. At higher dosages (i.e., 2.5 mg daily), DHT can remain suppressed for 6+ even after stopping treatment.
What is dutasteride?
Dutasteride is a drug used to treat a variety of androgen-related problems in men. It’s branded as Avodart®, and it’s most commonly prescribed for the treatment of benign prostate hyperplasia (BPH). But since it’s such a powerful DHT-reducing drug, many doctors will prescribe it “off-label” to treat male pattern hair loss, or androgenic alopecia (AGA).
How does dutasteride reduce DHT?
There are dozens of ways to reduce DHT levels – from lowering overall testosterone to inhibiting enzymes to blocking androgen receptors to increasing DHT metabolism (to name just a few). Some of these methods are safe; some of them are dangerous; some of them aren’t studied well enough in humans to consider trying.
The good news? Dutasteride reduces DHT through one of the better studied (and safer) ways… and similarly to finasteride. It inhibits the enzyme that converts testosterone into DHT: the 5-alpha reductase enzyme.
What makes dutasteride unique? It reduces type I and type II 5-alpha reductase
Dutasteride differs from finasteride in one key way: it reduces more DHT than finasteride. This is because dutasteride inhibits both type I and type II 5-alpha reductase, whereas finasteride inhibits only the type II 5-alpha reductase enzyme.
Both isoforms of 5-alpha reductase – type I and type II – help convert free testosterone into DHT. However, both function at different pH levels, and both are found in different tissue locations throughout the body:[1]Gerst, C., Dalko, M., Pichaud, P., Galey, J.B., Buan, B., Bernard, B.A. (2002). Type-1 steroid 5 alpha-reductase is functionally active in the hair follicle as evidenced by new selective inhibitors … Continue reading
- Type I 5-alpha reductase is found in the skin, liver, and in three specific places in the hair follicle: the outer root sheath, the dermal papilla, and the matrix cells (a.k.a the cells that produce the hair shaft).
- Type II 5-alpha reductase is predominantly located in the prostate, genital skin, seminal vesicles, epididymis, and liver. But it’s also found in hair follicles.
When it comes to skin tissues near and within hair follicles, the type I isoform dominates… at least in terms of the quantity of enzymes present. But interestingly, it’s the type II isoform that tends to get all of the attention when it comes to male pattern hair loss (AGA).
This is because type II 5-alpha reductase activity in the scalp seems to be a prerequisite for male AGA. This was confirmed back in the 1970s, when studies showed that men born without the genes that make type II 5-alpha reductase also never lose their hair to AGA later in life.[2]Imperato-McGinley, J., Miller, M., Wilson, J.D., Peterson, R.E., Shackleton, C., Gajdusek, D.C. (1991). A cluster of male pseudohermaphrodites with 5-alpha-reductase deficiency in Papua New Guinea. … Continue reading
In fact, these studies were what built the basis for finasteride – a drug that is now the most popular (and most studied) FDA-approved treatment for AGA. After all, finasteride is a type II 5a-reductase inhibitor, and it’s this specific enzyme that seems directly implicated in DHT-related hair follicle miniaturization.
Dutasteride does the exact same thing as finasteride – except it also inhibits both the type I and type II isoforms of 5-alpha reductase. And, since the body’s total DHT levels are closely linked to the sum of type I and type II isoform activity, dutasteride can technically reduce more DHT than finasteride – around 50% more (depending on the tissue being measured).
If dutasteride also inhibits the type I isoform and reduces more DHT, is it also better than finasteride at regrowing hair?
Maybe.
Several studies have demonstrated that dutasteride reduces DHT levels more than finasteride– at least in tests done on serum (i.e., blood) levels of DHT.[3]Bramson, H.M., Hermann, D., Batchelor, K.W., Lee, F.W., James, M.K., Frye, S.V. (1997). Unique preclinical characteristics of GG745, a potent dual inhibitor of 5AR. The Journal of pharmacology and … Continue reading In fact, a study found that while finasteride reduces serum DHT levels by ~70%, dutasteride reduces them by ~90% (depending on the dose):[4]Clark, R.V., Hermann, D.J., Cunningham, G.R., Wilson, T.H., Morrill, B.B., Hobbs, S. (2004). Marked Suppression of Dihydrotestosterone in Men with Benign Prostatic Hyperplasia by Dutasteride, a Dual … Continue reading
Figure 1: Change in DHT from baseline. Dutasteride vs. finasteride.[5]Clark, R.V., Hermann, D.J., Cunningham, G.R., Wilson, T.H., Morrill, B.B., Hobbs, S. (2004). Marked Suppression of Dihydrotestosterone in Men with Benign Prostatic Hyperplasia by Dutasteride, a Dual … Continue reading
But do these results also hold for scalp DHT – the region where DHT reduction matters most?
At least so far, the data suggests yes. Scalp biopsy studies on men taking dutasteride versus finasteride show that at certain dosages, dutasteride reduces much more DHT.[6]Olsen, E.A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M.L., Wilson, T., Rittmaster, R.S., Dutasteride Alopecia Research Team. (2006). The importance of dual 5alpha-reductase … Continue reading
Again, this makes logical sense. After all, type I and type II 5-alpha reductase are found inside and surrounding our hair follicles. And since dutasteride reduces both of these enzymes, this drug should technically reduce more scalp DHT than finasteride.
Thus, it’s totally rational to presume that since:
- DHT is causally linked to pattern hair loss, and…
- Finasteride helps treat pattern hair loss by reducing DHT, and…
- Dutasteride technically reduces more overall DHT + scalp DHT than finasteride…
Then all else equal, dutasteride should be a more effective hair loss drug.
But is this what the clinical research suggests? In order to answer this question, we’ll need to evaluate dutasteride’s hair-regrowing abilities against its risk profile: its reports of side effects, and how this does (or doesn’t) differ from finasteride.
Dutasteride versus finasteride: clinical trials (hair regrowth)
While there have been a handful of studies that have researched dutasteride, there are only four studies that provide data relevant to this question. We’ve compiled these studies as well as their methodologies, results, and conclusions into the chart below.
Studies Comparing the Effectiveness of Dutasteride and Finasteride
High quality study?
Daily dosages used Length of study Average hair count changes with dutasteride (%) Average hair count changes with finasteride (%) Any other notable results? Superior treatment?
Study #1.[7]Olsen, E.A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M.L., Wilson, T., Rittmaster, R.S., Dutasteride Alopecia Research Team. (2006). The importance of dual 5alpha-reductase … Continue reading Yes, randomized, placebo-controlled
Dutasteride: 0.1 mg, 0.5 mg,
2.5 mg
Finasteride:
5 mg
24 weeks 0.1 mg: 8.6% 0.5 mg: 10.1%
2.5 mg: 11.3%
8.3% The amount of patients who achieved at least 10% increase in hair count increases with dutasteride dose. In other words, as dose increased, so did the amount of patients who achieved at least 10% increase in density Dutasteride 0.5 mg and 2.5 mg
Study #2.[8]Shanshanwal, S.J.S., Dhurat, R.S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: A randomized controlled … Continue reading Yes, randomized controlled trial
Dutasteride: 0.5 mg Finasteride: 1 mg
24 weeks 11% 2% 24% increase in the number of thick, terminal hairs with dutasteride vs. 4% with finasteride Dutasteride
Study #3.[9]Harcha, W.G., Martinez, B.J., Tsai, T-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy … Continue reading Yes, randomized, placebo-controlled trial
Dutasteride: 0.02 mg 0.1 mg
0.5 mg
Finasteride: 1 mg
24 weeks 0.02 mg: 2.3% 0.1 mg: 8%
0.5 mg: 11.7%
7.5% 0.5 mg dosage of dutasteride was superior to 1mg finasteride in increasing hair thickness. Dutasteride at 0.5 mg
Study #4.[10]Choi, G-S., Sim, W-Y., Kang, H., Huh, C.H., Lee, Y.W., Shantakumar, S., Ho, Y-F., Oh, E-J., Duh, M.S., Cheng, W.Y., Bobbili, P., Thompson-Leduc, P., Ong, G. (2022). Long-Term Effectiveness and Safety … Continue reading Yes, multicenter long-term retrospective study
Dutasteride: 0.5 mg/day Finasteride: 1 mg/day
≥3 years (mean 3.4 years) Hair count data not reported (too sparse; BASP classification used instead) N/A Dutasteride is significantly superior across BASP basic, basic M, and V types. Dutasteride
Here’s what these results tell us about dutasteride:
- Dutasteride, at a dosage of 0.5 mg, has a clear advantage over both 1 mg and 5 mg of finasteride over the course of 24 weeks.
- Much like finasteride, a higher dose doesn’t necessarily equal better results. In fact, among two different studies, a 2.5 mg dose showed no better efficacy over a 0.5 mg dose. However, a higher dose appears to increase the number of users who will achieve at least a 10% increase in density.
- Dutasteride doesn’t just increase the number of hairs present; it also seems to increase the thickness of existing hairs, and more effectively than finasteride. This suggests that dutasteride may be a better drug for reversing hair follicle miniaturization.
The most interesting finding, though? Despite the fact that 2.5 mg of dutasteride reduces scalp DHT by almost 30% more than 0.5 mg, this doesn’t necessarily translate to more hair regrowth. In fact, the difference in efficacy between the two doses doesn’t even reach statistical significance.
This suggests that even when we reduce DHT to a greater degree and where it’s most important (the scalp), we don’t necessarily see evidence that this increases efficacy. In other words, maybe reductions to scalp DHT don’t always correlate to increased hair regrowth. Maybe there is a degree of pattern hair loss that stems directly from serum DHT, not just scalp DHT. Dr. George Cotsarelis (of PGD2 fame) has mentioned this before in interviews. Hopefully, with more research, we’ll have a clearer answer.
Nonetheless, these trials still tell us that dutasteride, at various doses, is more effective than finasteride – and anywhere between 2- and 5-fold more effective (depending on the doses).
The problem with these trials? Many are relatively short (24 weeks)
The one caveat to all but one of these comparative studies is that they were conducted over a 24-week period. This makes it difficult to discern just how effective dutasteride versus finasteride is… mainly because finasteride is notoriously slow at regrowing hair.
For instance, studies show that men taking finasteride generally just start to see improvements around the 3-6 month mark.[11]Price, V.H., Menefee, E., Sanchez, M., Ruane, P., Kaufman, K.D. (2002). Changes in hair weight and hair count in men with androgenetic alopecia after treatment with finasteride, 1 mg, daily. Journal … Continue reading These studies also show that finasteride takes 2+ years to take full effect on our hair, with scalp hair counts increasing an average of 10% over this two-year period before plateauing.
That’s 96 weeks for finasteride to demonstrate full efficacy, not 24 weeks – the length of time for all studies comparing dutasteride to finasteride. If, for unforeseen reasons, dutasteride efficacy plateaus after six months, then maybe finasteride catches up in the long run. If dutasteride continues its trajectory at the same pace as finasteride, then maybe it’s actually 2- to 5-times more effective than finasteride… and we just don’t know it yet.
In any case, we can surmise that:
- Dutasteride works faster than finasteride. Meaning, both will achieve similar density gains, but dutasteride can achieve this in about a quarter of the time.
- It’s very possible (and probable) that dutasteride increases hair counts by even more over a longer period of time. In other words, if we studied dutasteride for the same amount of time as finasteride, there’s a chance it might be more effective. This is further demonstrated by members here who’ve switched from finasteride (after 5+ years of use) to dutasteride, and within months, saw even bigger hair gains (see CradleCap’s case study).
Regardless, there’s still one piece to the puzzle we haven’t examined yet: the incidence of side effects.
After all, if dutasteride is 200%-500% more effective than finasteride, but it also comes with a 200-500% increase in side effects… that may defeat the point of using it. After all, for those dealing with hair loss, we’re always trying to find treatments that strike the right balance between effectiveness versus the risk of side effects.
This begs the question: given that dutasteride reduces more DHT and seems much more effective than finasteride, does it also come with an increased risk of side effects?
The answers might surprise you.
Is there an increased risk of side effects with dutasteride?
We already know that dutasteride reduces serum DHT by larger amounts than finasteride. So, does this come with a higher risk of sexual side effects?
Several studies have attempted to answer this question. Here’s what they have to say:
Incidence of Side Effects
Average incidence of total side effects among dutasteride group vs. finasteride group Decreased libido (%) Ejaculation disorders (%) Erectile dysfunction (%) Breast enlargement (%) Breast tenderness (%) Total serum DHT reduction Study #1
4.6% vs. 2.6% Dutasteride 0.1 mg: 3%
0.5 mg: 1%
2.5 mg: 13%
Finasteride
5 mg: 4%
Dutasteride 0.1 mg: 4%
0.5 mg: 1%
2.5 mg: 1%
Finasteride
5 mg: 3%
Dutasteride 0.1 mg: 0%
0.5 mg: 0%
2.5 mg: 0%
Finasteride
5 mg: 1%
N/A N/A Dutasteride 0.1 mg: 69.8%
0.5 mg: 92%
2.5 mg: 96.4%
Finasteride
5 mg: 73%
Study #2 10% vs 10%
N/A
N/A N/A N/A N/A N/A
Study #3 11.5% vs. 13.4%
Dutasteride
0.02 mg: 8.1%
0.1 mg: 6.9%
0.5 mg: 4.9%
Finasteride
1 mg: 6.7%
Dutasteride 0.02 mg: 2.2%
0.1 mg: 4.8%
0.5 mg: 3.3%
Finasteride
1 mg: 3.9%
Dutasteride 0.02 mg: 4.3%
0.1 mg: 3.7%
0.5 mg: 5.4%
Finasteride
1 mg: 5.6%
Dutasteride 0.02 mg: 0%
0.1 mg: 0.5%
0.5 mg: 0.5%
Finasteride
1 mg: 0.6%
Dutasteride 0.02 mg: 0.5%
0.1 mg: 0.5%
0.5 mg: 0%
Finasteride
1 mg: 0%
N/A
Study #4 7.6% vs. 10.5% Dutasteride: 1.2% Finasteride: 0.7%
N/A Dutasteride: 0.4% Finasteride 0%
N/A N/A N/A
When we examine this data, here’s what we find:
- In one study, 0.5 mg and 2.5 mg of dutasteride reduced serum DHT by 19% and 23% more than finasteride, respectively. Despite this, finasteride still had a higher incidence of side effects than the various doses of dutasteride in some categories (except for the incidence of decreased libido, which appeared to affect about 13% of men in the 2.5mg group). In all other categories, the incidence of side effects was virtually the same.
- Side effects associated with dutasteride don’t appear to follow a dose-response relationship (at least in these three studies). In fact, in studies #1 and #2, a higher incidence of side effects is observed in the lowest In other words, it appears that higher doses don’t necessarily mean a higher risk of side effects.
- Overall, if we average out the total incidence of side effects over the three studies, finasteride and dutasteride have the exact same incidence of side effects: 8.7% for dutasteride and 8.6% for finasteride.
- The conclusion? Dutasteride is more effective at increasing hair counts over a 6-month period, all without a higher risk of side effects.
Moreover, here are some other findings from these studies that are not reported within that table:
- The most common side effect was observed in study #1’s 2.5 mg dutasteride group – about 13% reported decreased libido. What the table doesn’t tell you is that more than half of the men reporting decreased libido also reported that it spontaneously resolved over the course of the study. In other words, it resolved even while taking dutasteride. Moreover, an additional 22% reported libido recovery after cessation of use.
- For men reporting side effects with dutasteride, almost all of these men reported resolution while still taking dutasteride.
- In contrast, men taking finasteride did not see a resolution of side effects during treatment. Most did, however, report resolution after the study was over.
Again, these side effects are only derived from clinical trials conducted over the course of 6 months. So, these studies alone don’t really tell us what might happen over longer periods of use. And considering that, much like finasteride, dutasteride likely has to be taken for a lifetime to sustain its effectiveness – this really matters.
So, is there any insight as to what the incidence of side effects might be after 6 months?
Well, when we examine the research, we find that the incidence of side effects actually decrease over time with dutasteride use. For example, one four-year trial employing a daily 0.5 mg dose demonstrated that the incidence of side effects decreased from 6% in year one to a marginal 0.4% by year four.
We could argue that this shift is simply a consequence of our olfactory wiring + change blindness – whereby we have a tendency to only notice changes (i.e., new smells) when they first arrive, and whereby if changes occur too slowly, we generally won’t notice them at all.
At the same time, we could also argue that with long-term use of dutasteride, our bodies simply rewire and find ways for testosterone (and other hormones) to replace the roles previously fulfilled by DHT. This is generally how the body works – there are dozens of “fallback” mechanisms in case a vitamin, mineral, or hormone is missing. In other words, we’re more durable than we think.
Why does dutasteride come with an equivalent (or lower) risk of side effects?
It’s puzzling that dutasteride seems to come with an equivalent (or lower) risk of side effects… and that over time, these side effects seem to decrease more than with finasteride. This is in spite of dutasteride reducing much more DHT than finasteride. What could explain these paradoxical findings?
Well, one could argue that maybe DHT reduction isn’t necessarily as great of a predictor of side effects as we think. But, there is one additional explanation for these findings, and one that’s much more interesting to explore…
It’s the actual structure of the dutasteride molecule itself… and the fact that the size of this molecule might greatly reduce the drug’s accessibility to certain organs within our body. Specifically, the brain.
Why might the molecular characteristics of dutasteride influence the risk of side effects?
The blood is the ultimate transporter for nutrition. It’s also the transporter for oral drugs.
During circulation, the blood will deposit various compounds into cells, where they then elicit their specific effects. However, in the brain, it isn’t quite as simple. This is because of a membrane called the blood-brain barrier (BBB).
This membrane is comprised of cells that maintain tight cellular junctions. In other words, these cells are packed in close together with minimal space between each cell. As such, only very small compounds or compounds that can bind to specific entry receptors on the BBB can pass through the membrane into the brain.
In terms of molecular size, a given compound has to be smaller than 400 daltons (a measurement of mass) to traverse past this membrane. If any larger? The compounds cannot theoretically enter the brain through blood transportation. Therefore, these molecules may not be able to elicit their specific effects on the brain.
So, what does this have to do with dutasteride and finasteride? And why might this explain the difference in the incidence of side effects?
Interestingly, while both finasteride and dutasteride act similarly with respect to DHT reduction, they are actually quite different on a molecular level – particularly regarding their molecular weight.
While finasteride is only 372 daltons, dutasteride is 528 daltons.
The biggest difference here? Finasteride is small enough to pass through the tight junctions of the blood-brain barrier (BBB). Dutasteride isn’t.
As such, finasteride may elicit effects on the brain itself, which may precipitate major effects on sexual and mental side effects. In contrast, dutasteride may have less of a direct effect on the brain… or a reduced capacity to effect androgen metabolism in the brain relative to finasteride.
Again, this is all hypothetical, and the science is still very much debated. For instance, this study on mice found that dutasteride does influence androgen metabolism in the brain (although that study also found that dutasteride had neuroprotective effects on mice, helping to stave off Parkinsons).[12]Litim, N., Morissette, M., Caruso, D., Melcangi, R.C., Di Paolo, T. (2017). Effect of the 5α-reductase enzyme inhibitor dutasteride in the brain of intact and Parkinsonian mice. The Journal of … Continue reading
At the same time, with dutasteride use, some reduction in brain androgen activity is technically to be expected… even if dutasteride doesn’t technically enter the brain. Why? Because 5-alpha reductase can be transported via the blood (where dutasteride does elicit an effect). And if blood entering the brain has less 5-alpha reductase activity, then this would also change androgen activity in the tissues where that blood eventually gets transported (i.e., the brain).
In any case, our suggestion here is that dutasteride itself has a harder time entering into brain tissues, and that this is why it may confer to a lower risk of side effects (despite more DHT reduction) than finasteride.
So, despite the fact that dutasteride has a more powerful effect on both systemic DHT than finasteride, the lower or on-par incidence of side effects may simply be due to dutasteride’s molecular weight… and how this dampens the drug’s ability to enter brain tissue.
Thus, it appears we can derive faster, more substantial hair regrowth in AGA… all with the same or even lower risk of side effects. Moreover, we can even leverage dosages to both maximize regrowth and minimize the risk of side effects.
Dutasteride: best candidates + best practices (i.e., dosages)
It goes without saying that dutasteride is a drug that is appropriate only for one specific kind of hair loss: androgenic alopecia (pattern hair loss). It also goes without saying that this drug is studied almost exclusively on men. Therefore, men with pattern hair loss are the best candidates.
So, for men with pattern hair loss, what’s the best dose?
Well, revisiting data from the three studies comparing dutasteride to finasteride, we see that larger doses of dutasteride are generally no more effective than a dosage of 0.5 mg. And based on the safety data, this appears to be one of the safest doses, too.
Specifically, the studies show that 0.5 mg can increase hair counts by anywhere between 10 and 11.5% in 6 months, with just a 1.3% overall risk of sexual side effects. In contrast, finasteride’s risk of side effects ranges from 2-13% with regrowth rates only around 7-8%.
So, a daily dose of 0.5 mg of dutasteride orally seems to both maximize hair regrowth and minimize the risk of side effects. Moreover, it does so in a safer and more effective manner than both 1 mg and 5 mg doses of finasteride.
Finally, a 0.5 mg daily dose is also a dose that allows for our DHT levels to bounce back within six months after stopping the drug. Because of dutasteride’s longer half-life, it tends to exert a longer effect in the body. At higher dosages, it seems as though DHT levels remain suppressed for the majority of users, at least for six months of quitting. A 0.5 mg daily dose prevents this from happening, allowing DHT to return to 80% of baseline levels after six months of cessation, and likely 100% baseline levels a few months thereafter.
Want an example of a long-term dutasteride user? See Ashton Kutcher.
If you’d like an example of a long-term dutasteride user, look no further than Ashton Kutcher.[13]Team Coco. (2018). Ashton Kutcher’s Hair Is Starting To Go | CONAN on TBS. Available at: https://www.youtube.com/watch?v=Wv9gu1zJdRo (Accessed: November 2025)
During his 2018 interview with Konan, he revealed that he had used the drug for years, only hopping off to father his children (born in 2014 and 2016). It seems to have worked wonderfully for him. Just look at his hairline in 1998 versus today. While there’s no information about whether he has returned to using dutasteride, it also doesn’t seem like his hairline has changed much over the past several years anyway.
Summary
Finasteride is one of the most powerful and frequently used therapies for AGA. However, emerging research indicates that dutasteride may have a clear advantage over finasteride, at least in terms of (1) scalp DHT reduction, (2) hair regrowth, and (3) side effects.
Specifically, a 0.5 mg daily dose appears to increase hair density by anywhere between 10 and 11.5% over the course of 6 months, which happens to be the hair count increase expected from 2+ years of finasteride use. Moreover, dutasteride seems to achieve this without greatly increasing someone’s risk of cognitive or sexual side effects. In fact, the incidence of side effects with dutasteride seems to decrease over time, with one study demonstrating just a 0.4% incidence of side effects with four years of use.
While higher doses (up to 2.5 mg) do increase the response rate to the treatment, they don’t appear to increase the average amount of hair someone regrows. Moreover, they also increase the likelihood of long-term DHT suppression post-treatment. As such, a 0.5 mg daily dose seems like the best approach when utilizing this drug for hair regrowth.
Considering these overall advantages, cost-effectiveness, and accessibility, dutasteride shows considerable promise for men with AGA.
If you’re interested in taking dutasteride or swapping out your oral finasteride, talk with your doctor about the research. If you’re a good candidate for the drug, ask them to prescribe you a 0.5 mg daily dose, and please keep us posted with your results!
References[+]
References ↑1 Gerst, C., Dalko, M., Pichaud, P., Galey, J.B., Buan, B., Bernard, B.A. (2002). Type-1 steroid 5 alpha-reductase is functionally active in the hair follicle as evidenced by new selective inhibitors of either type-1 or type-2 human steroid 5 alpha-reductase. Experimental Dermatology. 11(1). 52-58. Available at: https://doi.org/10.1034/j.1600-0625.2002.110106.x. ↑2 Imperato-McGinley, J., Miller, M., Wilson, J.D., Peterson, R.E., Shackleton, C., Gajdusek, D.C. (1991). A cluster of male pseudohermaphrodites with 5-alpha-reductase deficiency in Papua New Guinea. Clinical Endocrinology. 34(4). 293-298. Available at: https://doi.org/10.1111/j.1365-2265.1991.tb03769.x. ↑3 Bramson, H.M., Hermann, D., Batchelor, K.W., Lee, F.W., James, M.K., Frye, S.V. (1997). Unique preclinical characteristics of GG745, a potent dual inhibitor of 5AR. The Journal of pharmacology and experimental therapeutics. 282(3). 1496-1502. Available at: PMID 9316864 ↑4, ↑5 Clark, R.V., Hermann, D.J., Cunningham, G.R., Wilson, T.H., Morrill, B.B., Hobbs, S. (2004). Marked Suppression of Dihydrotestosterone in Men with Benign Prostatic Hyperplasia by Dutasteride, a Dual 5α-Reductase Inhibitor. The Journal of Clinical Endocrinology & Metabolism. 89(5). 2179-2184. Available at: https://doi.org/10.1210/jc.2003-030330 ↑6, ↑7 Olsen, E.A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M.L., Wilson, T., Rittmaster, R.S., Dutasteride Alopecia Research Team. (2006). The importance of dual 5alpha-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. Journal of the American Academy of Dermatology. 55(6). 1014-1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007 ↑8 Shanshanwal, S.J.S., Dhurat, R.S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: A randomized controlled open-label, evaluator-blinded study. Indian Journal of Dermatology, Venereology and Leprology. 83(1). 47-54. Available at: https://doi.org/10.4103/0378-6323.188652 ↑9 Harcha, W.G., Martinez, B.J., Tsai, T-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. Journal of the American Academy of Dermatology. 70(3). 489-498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑10 Choi, G-S., Sim, W-Y., Kang, H., Huh, C.H., Lee, Y.W., Shantakumar, S., Ho, Y-F., Oh, E-J., Duh, M.S., Cheng, W.Y., Bobbili, P., Thompson-Leduc, P., Ong, G. (2022). Long-Term Effectiveness and Safety of Dutasteride versus Finasteride in Patients with Male Androgenic Alopecia in South Korea: A Multicentre Chart Review Study. 34(5). 349-359. Available at: https://doi.org/10.5021/ad.22.027 ↑11 Price, V.H., Menefee, E., Sanchez, M., Ruane, P., Kaufman, K.D. (2002). Changes in hair weight and hair count in men with androgenetic alopecia after treatment with finasteride, 1 mg, daily. Journal of the American Academy of Dermatology. 46(4). 517-523. Available at: https://doi.org/10.1067/mjd.2002.120537 ↑12 Litim, N., Morissette, M., Caruso, D., Melcangi, R.C., Di Paolo, T. (2017). Effect of the 5α-reductase enzyme inhibitor dutasteride in the brain of intact and Parkinsonian mice. The Journal of Steroid Biochemistry and Molecular Biology. 174. 242-256. Available at: https://doi.org/10.1016/j.jsbmb.2017.09.021 ↑13 Team Coco. (2018). Ashton Kutcher’s Hair Is Starting To Go | CONAN on TBS. Available at: https://www.youtube.com/watch?v=Wv9gu1zJdRo (Accessed: November 2025) Dutasteride is one of the strongest off-label treatments for androgenic alopecia. In this guide, we’ll explore the evidence supporting two delivery methods for dutasteride: topical dutasteride and mesotherapy dutasteride. We’ll also provide clear step-by-step instructions for anyone looking to try these therapies at home or find clinicians willing to do the procedures.
Overview: Topical / Mesotherapy Dutasteride
- Effort: Low (once weekly-to-monthly applications, depending on topical or mesotherapy delivery).
- Expectations: Hair improvements within 4-6 months.
- Response rate (i.e., the percent of people who experience a slowing, stopping, or partial reversal of AGA):
- Topical dutasteride: 70-90%
- Topical dutasteride + microneedling: 60-80%
- Mesotherapy dutasteride: 60-80%
- Regrowth rates (i.e., the percent change to hair counts, hair diameters, and hair density (i.e., ∆hair counts x ∆hair diameters)):
- Topical dutasteride: +23-25% increase in hair counts (mean +75 hairs/cm² vs baseline in the 0.05% group over 24 weeks).
- Topical dutasteride + microneedling: 10-15% increase in hair counts, 15-20% increase in hair density (sometimes more)
- Mesotherapy dutasteride: 10-15% increase in hair counts, 15-20% increase in hair density (sometimes more)
- Advantages: Easy to administer; works for the majority of people; minimizes DHT reductions elsewhere in the body; likely confers with a lower side effect profile.
- Disadvantages: Clinical studies are of lower quality; no standardization across mesotherapy needle depths or dutasteride exposure volumes per session; high variability between patients regarding systemic absorptions puts the onus on each patient to test serum DHT levels before/during treatment; expensive.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Key Takeaways
- The Good News:
- Topical / mesotherapy dutasteride works. While the evidence is limited and of lower quality, multiple studies from different research groups all suggest that topical dutasteride and mesotherapy dutasteride improve hair loss outcomes for men and women with androgenic alopecia.
- Topical / mesotherapy dutasteride may minimize unwanted declines in serum DHT. Multiple studies (while not perfectly designed) suggest, when dosing is done properly, that topical dutasteride and/or mesotherapy dutasteride can improve hair parameters while only modestly reducing serum DHT levels (a marker for systemic absorption). This has been confirmed by members here who’ve done serum DHT tests before/after treatment and seen only 10-15% declines in DHT. Moreover, absorption studies testing 1 mL of 0.025% topical dutasteride (i.e., 0.25 mg) applied to the scalp show, after 12 hours, that ~7% of the drug enters the bloodstream while 25% of the drug remains within the dermis of the scalp. This means that within proper exposure volumes, we can maximize DHT reduction in the scalp while minimizing the effects of DHT reduction elsewhere in the body.
- No sexual side effects (yet) reported in the clinical data. While the currently available studies have small sample sizes, all investigations on topical / mesotherapy dutasteride have yet to have a participant report sexual side effects. This experience is echoed by members here who’ve adhered to the usage ad frequency parameters of both topical and mesotherapy dutasteride.
- Critical Unknowns:
- How does topical dutasteride and mesotherapy dutasteride compare to each other (and other therapies)? Due to lacking head-to-head clinical studies, we don’t yet have an answer. However, directional hair count data across studies suggest that topical / mesotherapy dutasteride is probably less powerful than its oral counterpart. (This comparison is unscientific, but it’s all we can currently glean.)
- What’s the best dosage, frequency of application, and carrier agents? While low-exposure volumes of topical / mesotherapy dutasteride show minimal effects on serum DHT, we don’t yet know the perfect dosing schedule to maximize hair regrowth while minimizing serum DHT reductions. It’s likely that leakage from the scalp and systemic absorption vary depending on the individual and the treatment methodologies: age, genetics, mode of delivery, dosage per application, application frequency, and application volume are just a few of the dozens of factors impacting the answers to these questions.
- How does topical dutasteride perform as a standalone therapy? Early data suggest that low-dose topical dutasteride (including 0.05%) can improve hair parameters and may, in some trials, appear comparable or even superior to oral finasteride, but methodological limitations and conflicting real-world experiences mean these findings should be interpreted cautiously until independently replicated.
- Best Practices & Recommendations
- See below.
Key Recommendations
Topical dutasteride and mesotherapy dutasteride are probably most appropriate for people who:
- Have androgenic alopecia
- Don’t want to use oral finasteride or oral dutasteride, but want to leverage a pharmaceutical 5-alpha reductase inhibitor
- Want to better localize the effects of dutasteride to scalp
- Are comfortable experimenting with delivery methods that are lesser explored and have lower levels of clinical support
- Are comfortable investing $50-$400 per month into treatment, depending on the mode of delivery (topical or mesotherapy)
- Don’t have metabolic syndrome (as one study suggested that mesotherapy dutasteride was ineffective for these patients)
Step-By-Step Guide
Topical dutasteride
If trying topical dutasteride:
- Limit topical exposure of dutasteride to 0.1-0.5mg weekly.
- This can be achieved in many ways. Consider 1 mL formulas more appropriate for localized hair loss, and 2 mL formulas more appropriate for diffuse hair loss (for more spread of liquid). Examples:
- 1 mL of 0.01% to 0.05% dutasteride, once weekly. Exposure = 0.1-0.5 mg
- 1 mL of 0.025% dutasteride, twice weekly. Exposure = 0.5 mg
- 2 mL of 0.005% to 0.025% dutasteride, once weekly. Exposure = 0.1-0.5 mg
- 2 mL of 0.005% to 0.0125% dutasteride, twice weekly. Exposure = 0.2-0.5 mg
- This can be achieved in many ways. Consider 1 mL formulas more appropriate for localized hair loss, and 2 mL formulas more appropriate for diffuse hair loss (for more spread of liquid). Examples:
- Leave in scalp for at least 4 hours, and preferably 8-12 hours
- This will allow 25% of the dutasteride applied to penetrate into the percutaneous tissues, so it can have an effect.
- This will minimize the amount of dutasteride that enters the bloodstream, thus reducing systemic absorption.
- The easiest way to do this is to apply topical dutasteride before bed and then wash it out in the morning.
- Consider combining with microneedling
- If combining with microneedling, do not apply topical dutasteride the day of microneedling
- Consider testing serum DHT levels before/during treatment
- This will help track if topical dutasteride is remaining localized to the scalp.
- In tracking DHT levels, you can increase or decrease dutasteride dosing depending on your serum DHT readouts.
- Product recommendations
- MinoxidilMax: 0.1% topical dutasteride, 60 mL.
- You can use this solvent to dilute the formula down to 0.005% to 0.02%. If you’d like help with this, ask us. We’re currently building out a “how to” video for diluting topicals that will make this process simple. But if you want to get a jumpstart on this, please don’t hesitate to ask for our help.
- MinoxidilMax: 0.1% topical dutasteride, 60 mL.
Mesotherapy dutasteride
If trying mesotherapy dutasteride:
- Start with mesotherapy injections once monthly.
- After 6+ months, move to one session every 1-3 months.
- If once monthly injections are infeasible, opt for injections once every three months.
- Limit dutasteride exposure to 0.1-0.2 mg per session.
- This is equivalent to 1-2mL of 0.01% dutasteride
- Consider testing serum DHT levels before/during treatment
- This will help track if topical dutasteride is remaining localized to the scalp.
- In tracking DHT levels, you can increase or decrease dutasteride dosing depending on your serum DHT readouts.
How do I track down a clinician who can provide mesotherapy dutasteride?
It’s not easy, but it can be done.
Step #1: Build a list of practitioners offering mesotherapy in your locale
Run a google search for dermatologists and/or medical spas in your area. When checking out their websites, make sure they offer mesotherapy. It’s okay if the mesotherapy they offer is not related to hair loss. All that matters is that they offer mesotherapy, because that means they will have a mesotherapy gun in their office.
Track down their names and contact information via contact forms and emails listed on their sites. Try to build a list of 5-10 clinicians within travel distance.
Step #2: Send each practitioner this personalized email
Here’s an email script that has given us some success. Please personalize as necessary:
Dear [CLINICIAN],
My name is [YOUR NAME]. I’m reaching out because I’m interested in trying mesotherapy dutasteride for the treatment of androgenic alopecia. The procedures are typically done once monthly for 6+ months. I wanted to see if your offices would be willing to provide the therapy to me.
I’ve been encouraged by the results of this mini-review, suggesting that repeated mesotherapy dutasteride may improve hair loss outcomes for men with androgenic alopecia, and without significantly altering serum DHT levels.[1]Herz-Ruelas, M.E., Alvarez-Villalobos, N.A., Millan-Alanis, J.M., de Leon-Gutierrez, H., Ocampo-Garza, S.S., Grimalt, R. (2020). Efficacy of Intralesional and Oral Dutasteride in the Treatment of … Continue reading I’m planning on tracking serum DHT before/during treatment, and I’d be happy to share the blood work with you (especially as interest in this therapy is growing, it might be nice to have the data).
I’m happy to source the mesotherapy dutasteride serum. All I need is a clinician trained in mesotherapy to administer the procedure. Are you interested? If so, I can come to offices as soon as next week.
All my best,
[YOUR NAME]
[PHONE NUMBER]Step #3: Secure a practitioner
Typically, for every 5-10 clinicians who receive the above email, one will respond to say that they’re interested. Meet with these clinicians, print out and share with them the studies, and show them the methodologies (below) to standardize the procedure, thus making it as easy as possible for the doctor to administer.[2]Herz-Ruelas, M.E., Alvarez-Villalobos, N.A., Millan-Alanis, J.M., de Leon-Gutierrez, H., Ocampo-Garza, S.S., Grimalt, R. (2020). Efficacy of Intralesional and Oral Dutasteride in the Treatment of … Continue reading
Methods For Mesotherapy Dutasteride (With A Clinician):
- Injection depth: intradermal (i.e., between the epidermal and dermal skin layers, roughly 0.5-0.75 mm deep)
- Mesotherapy formulation: 0.01% dutasteride in a 10 mL vial (provider here)
- Injection volume per session: 1-2 mL (i.e., 0.1-0.2 mg of dutasteride)
- Injection frequency: once monthly for six months, then once every 1-3 months thereafter
Can I try mesotherapy at home?
To be clear, we DO NOT recommend taking a DIY approach to mesotherapy. If you cannot find a provider, we recommend trying topical dutasteride + microneedling. Topical dutasteride is far more accessible, and it’s also easily administered at home. It’s also likely that the combination produces similar results to mesotherapy dutasteride.
Under the circumstances that you decide not to heed our warnings, here are DIY instructions for mesotherapy dutasteride.
Methods for Mesotherapy Dutasteride (At-Home):
- Buy this mesotherapy device. Use the 9-needle cartridges and adjust the needle depth to 0.5mm when operating.
- Buy mesotherapy dutasteride from this provider (we lab tested their products; they meet quality and purity standards).
- Follow the instructions from the manufacturer to deliver mesotherapy to your scalp once per month.
- Consider tracking serum DHT levels before/during treatment.
Below is a scientific deep-dive on topical / mesotherapy dutasteride.
What Is Topical Dutasteride and How Does It Work?
Topical dutasteride is topical version of the oral drug, dutasteride. Whereas oral dutasteride tends to have hormonal effects all throughout the body, topical dutasteride is designed to selectively target just the scalp skin – allowing people to reap all the hair-growing benefits of dutasteride, and without the risk of systemic side effects.
At least, that’s the hope.
Specifically, dutasteride lowers the hormone dihydrotestosterone (DHT), which is causally linked to androgenic alopecia. It does so by inhibiting the enzyme that converts testosterone into DHT: 5-alpha reductase.
Dutasteride is one of the most powerful 5-alpha reductase inhibitors available. Whereas hair loss drugs like finasteride inhibit 5-alpha reductase and lower DHT levels by ~70%, dutasteride can lower DHT levels by 95%.
This is because there are three known isoforms of 5-alpha reductase: type I, II, and III. Compared to finasteride, dutasteride is more effective at inhibiting them. In fact, dutasteride is able to target the type I isoform 100x better than finasteride, and the type II isoform 3x better than finasteride.[3]Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., Cui, Y. (2019). The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and … Continue reading
How Does Oral Dutasteride Compare To Oral Finasteride for Androgenic Alopecia?
Dutasteride’s larger effects on DHT reduction also confer better hair regrowth outcomes.
A 2019 meta-analysis by Zhou et al. found that oral dutasteride regrew more hair than oral finasteride – even at dosage ranges lower than 0.5 mg daily.[4]Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., Cui, Y. (2019). The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and … Continue reading And while the studies in this review were relatively short (i.e., 6 months) and had sample sizes of less than 500 participants, it’s hard to ignore the preliminary evidence suggesting that oral dutasteride packs a big punch for hair regrowth – even at dosages as low as 0.1mg daily.
Figure 1: The studies cited maintain a low risk of bias, but because of the low sample size, it’s difficult to say with confidence that dutasteride is superior to finasteride.[5]Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., Cui, Y. (2019). The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and … Continue reading
What About Safety?
Despite reducing more DHT and regrowing more hair, both finasteride and dutasteride appear to carry the same risk of side effects.
Figure 2: Forest plots showing changes in (A) altered libido, (B) erectile dysfunction, and (C) ejaculation disorders.
Abbreviation: M–H, Mantel–Haenszel.[6]Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., Cui, Y. (2019). The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and … Continue readingWhy is this? Wouldn’t we expect dutasteride to carry a higher risk of side effects, since it reduces DHT levels more?
Generally, yes. But that’s not currently reflected in the data. Regarding why, there are many hypotheses:
- Biases in sample sizes and study durations. Perhaps dutasteride does carry a larger risk of side effects, as it has been shown to do so in some patient populations (for instance, dutasteride carries a higher risk versus finasteride for gynecomastia in patients with enlarged prostates). But perhaps the sample sizes in these hair loss studies were too small in these studies to pick up the differences. Or, perhaps the studies did not run long enough to capture all risks.[7]Hagberg, K.W., Divan, H.A., Fang, S.C., Nickel, J.C., Jick, S.S. (2017). Risk of gynecomastia and breast cancer associated with the use of 5-alpha reductase inhibitors for benign prostatic … Continue reading
- Differences in isoform side effect attribution. Perhaps the type II 5-AR enzyme (i.e., the one predominantly targeted by finasteride) is the one isoform predominantly responsible for most of the side effects associated with finasteride and dutasteride. (We’ve heard this argument before, but we don’t necessarily believe it.[8]Rahimi-Ardabili, B., Pourandarjani, R., Habibollahi, P., Mualeki, A. (2006). Finasteride induced depression: a prospective study. BMC Clinical Pharmacology. 6(7). Available at: … Continue reading
- Tissue-dependent concentration differences of isoforms. For example, in the prostate and testes, the type II isoform is expressed far more than type I. The opposite is true in brain tissue, where type I 5-AR is predominantly found. Perhaps the side effects of 5-AR inhibitors depend on how much 5-AR is inhibited in specific tissues, and at the dosages administered for dutasteride and finasteride, there just isn’t enough difference in DHT reduction to make one drug more likely to produce sexual side effects than the other.[9]Wang, K., Fan, D-D., Jin, S., Xing, N-Z., Niu, Y-N. (2014). Differential expression of 5-alpha reductase isozymes in the prostate and its clinical implications. Asian Journal of Andrology. 16(2). … Continue reading
Even still, there remains no consensus among clinicians for why this is the case.
Pharmacokinetics
How Does Dutasteride Behave In Our Bodies?
Pharmacokinetics is a term used to describe the behavior of drugs once they enter the body: how quickly they are transported to tissues, how quickly they are metabolized (i.e., their half-life), and more.
Dutasteride and finasteride, despite being structurally similar, differ dramatically in their half-lives. For instance, finasteride has a half-life of 5-8 hours. That means that after ingesting finasteride, it takes 5-8 hours before 50% of that finasteride leaves our bloodstream.
Dutasteride, on the other hand, has a half-life that can last 5+ weeks. In fact, dutasteride’s half-life depends on a lot of factors: age, genetics, and, most importantly, the amount of dutasteride given to an individual at one time.
This was investigated by Gislekog et al. (1998) by giving a dose of dutasteride ranging from 0.01mg to 40mg to 32 healthy males. The findings showed a number of distinctions:
- The half-life of the drug increases with increasing dose.
- Serum concentrations of dutasteride can last for quite a while before it is undetectable.
This is presented in the following table:
Dose Serum Concentration Duration (up to 1ng mL-1) 0.1mg Approximately one day. 1.0mg Averaging around seven days, but can be up to 14 days. 2.5mg Averaging approximately 14-21 days, but can be as long as 28 days. 5.0mg Approximately 21-28 days. 10.0mg Greater than 28 days. 20.0mg Greater than 28 days. 40.0mg Greater than 28 days. And here are the graphs from the study:Concentration of dutasteride in serum is dependent on how large a dose is used. This means that beyond half-life, the general concentration of the drug can last for quite a while when using greater than 2.5mg doses.[10]Gisleskog, P.O., Hermann, D., Hammarlund-Udenaes, M., Karlsson, M.O. (1999). The pharmacokinetic modelling of GI198745 (dutasteride), a compound with parallel linear and nonlinear elimination. … Continue reading
Can Topical Dutasteride Lower Scalp DHT While Minimally Affecting Hormones Elsewhere In The Body?
This is not only possible, but plausible… provided that we play with the dosages of dutasteride and the frequencies of application.
Since dutasteride rapidly distributes across tissues and has a dose-dependent half-life, the key is to find an exposure volume of topical dutasteride that…
- Is large enough to effectively saturate the epidermal and dermal layers of the scalp (so that we can locally lower DHT)
- Is small enough such that the amount of dutasteride that leaks daily into the bloodstream is rapidly metabolized (so that DHT levels elsewhere are minimally impacted)
Encouragingly, we have some data to suggest there are pharmacokinetic “sweet spots” to achieving this.
A recent analysis measured how much of a 1 mL solution of 0.025% dutasteride (i.e., 0.25mg) applied to the scalp (1) stayed on the skin, (2) penetrated into the dermis, and (3) travelled systemically into the bloodstream.
Across 12 hours of study, here’s what the investigators discovered:
Figure 4: Effect of topical dutasteride on skin absorption and circulation entry.
If the above chart feels overwhelming, don’t worry. For now, just know that 12 hours after applying topical dutasteride:
- 60.8% of the dutasteride remained on the surface of the scalp and was unabsorbed (i.e., 0.152 mg)
- 26.2% of the dutasteride penetrated into the scalp (i.e, 0.06 mg).
- 6.4% of the dutasteride entered into the bloodstream (i.e., 0.016 mg)
This is actually good news. Why? Because 12 hours after applying 1 mL of 0.025% topical dutasteride, 0.06 mg of that dutasteride permeated into balding tissues, while only 0.016 mg entered into the bloodstream.
In balding skin tissue, 0.06 mg of dutasteride is probably:
- Enough drug to appreciably lower scalp DHT levels, and…
- Enough drug to increase the elimination half-life to > 1 day, so that the scalp DHT suppression can last for multiple days.
Simultaneously, with the amount of dutasteride reaching the bloodstream, 0.016 mg of dutasteride is probably:
- Not enough drug to appreciably lower serum DHT levels, and…
- Not enough drug to increase the elimination half-life to > 1 day. As such, any serum DHT suppression will likely be short-lived.
In other words, one application of 1 mL of 0.025% topical dutasteride might lead to impressive reductions to scalp DHT, while having minimal impact on serum DHT (and thereby DHT levels elsewhere in the body).
So, Should We All Apply 1 mL of 0.025% Topical Dutasteride, Once Or Twice Daily?
We wish it were that simple.
The problem is that the above analysis only measures the effects from one application of topical dutasteride. If we apply this dosage daily, dutasteride levels in the scalp skin will accumulate, as will the amount of dutasteride entering the bloodstream.
Additionally, after that 12-hour mark, a portion of the ~25% of dutasteride already absorbed into the skin will continue to leak into the bloodstream. The above study does not capture these effects. So, while the analysis is insightful, it unfortunately does not run long enough to reveal the full extent of systemic absorption from a single 1 mL dose of 0.025% topical dutasteride. That means total bloodstream exposure from 1 mL of 0.025% topical dutasteride is probably a bit higher than the 0.016 mg suggested by the study.
Finally, as blood levels of dutasteride increase, so too will the drug’s half-life. Remember: the more dutasteride present, the longer the half-life. So, after a few weeks of daily application, it’s not unreasonable to expect serum drug exposure to accumulate and thereby serum DHT levels to substantially decrease. This is exactly what most people trying topical dutasteride want to avoid!
Fortunately, we don’t have to rely exclusively on mathematical modeling to figure this out. There are clinical studies testing topical dutasteride for the treatment of androgenic alopecia – some of which measured not only hair growth, but also changes to serum DHT levels before and after treatment.
Topical Dutasteride: Clinical Studies (Hair Counts, Serum DHT, & More)
Nada et al. (2018): Microneedling + Topical Dutasteride vs. Microneedling Alone
In a 2018 clinical study, researchers sought to determine how topical dutasteride + microneedling fared versus microneedling alone.[11]Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmagd, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). 387-401. … Continue reading
So, they recruited 30 males with androgenic alopecia, randomized them into two groups, and tracked hair parameter changes of a six-month period.
Using a Dermapen with a 12-gauge needle cartridge and a 1.5mm needle length, both groups received 13 microneedling sessions over six months, and using the following schedule:
- Once per week (weeks 0-7)
- Once every two weeks (weeks 9 and 11)
- Once every four weeks (weeks 15, 19, and 23)
However, during each microneedling session, the topical dutasteride group also received up to 2 mL of 0.02% topical dutasteride (i.e., 0.4mg) applied to the scalp.
After six months, the topical dutasteride + microneedling group saw greater improvements in hair density versus the microneedling-only group. This equated to a 12% increase in hair density.
Figure 5: Changes of hair density in the study groups before and after treatment.[12]Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmagd, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). 387-401. … Continue reading
Additionally, the topical dutasteride + microneedling group also saw hair thickness improve by nearly 100%.
Figure 6: Changes in hair caliber width in the study groups before and after treatment.[13]Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmagd, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). 387-401. … Continue reading
Finally, hormonal testing revealed that the topical dutasteride + microneedling group did experience a 5.3% reduction in serum levels of DHT. However, this level of DHT reduction is considered biologically insignificant. Keep in mind that serum DHT levels can fluctuate as much as 20-40% daily, depending on if the measurements were taken during the morning or night. So a ~5% change in DHT isn’t very concerning.
Figure 7: Hormone levels before and after treatment.[14]Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmagd, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). 387-401. … Continue reading
Our Analysis
While these results are encouraging, they’re also problematic. Specifically, the results are obfuscated by a methodological concern: staggered microneedling + topical dutasteride sessions, and no measurements for serum DHT during shorter durations between dutasteride applications.
Throughout the study, time gaps between topical dutasteride applications grew. For the first 8 weeks, topical dutasteride was applied once weekly. But for the last 12 weeks, it was applied once monthly. The investigators only reported changes to serum DHT before/after the study concluded. Specifically, they recorded serum DHT before the study began, and one week after their final dutasteride application. So, are these results really representative of serum DHT levels throughout the study… or were serum DHT levels much lower in the first few months when application frequency was 4x higher?
Unfortunately, the investigators do not give us enough information to know the answers to these questions. We’re reaching out and hope to have answers for you soon.
In the interim, we’ve had several members emulate the methodologies of this study, test serum DHT levels before/during treatment, and adhere to a once-weekly topical dutasteride application rate (at up to 2 mL of 0.025% dutasteride, that’s up to 0.4mg of weekly dutasteride exposure).
The good news is that all of these members reported only 10-15% decreases in serum DHT. While those decreases are likely partly due to serum dutasteride, they’re still within the realm of biologically insignificant. And encouragingly, our members’ results not only align with the ballpark results of this study, but also the previous dosing analysis – at least in terms of expectations for systemic leakage.
In our eyes, these are all positive signs. They’re preliminary signals (validated by some members) that at weekly dosages of up to 2 mL x 0.02% topical dutasteride (i.e., 0.4 mg per week), we can mostly preserve serum DHT levels – even when adding in microneedling.
Encouragingly, a 2022 study also validated this approach – at least in terms of efficacy.
Sanchez et al. (2022): Microneedling + Topical Dutasteride vs. Microneedling Alone
In 2022, investigators conducted another study on topical dutasteride + microneedling, albeit without serum DHT and with slightly different methodologies.[15]Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical … Continue reading
These investigators randomized 34 men into two groups. One received microneedling + 1 mL of 0.01% topical dutasteride (i.e., 0.1 mg). The other received microneedling + 1 mL of saline solution (i.e., water).
Both groups were treated once every four weeks for a total of three sessions. In both groups, the investigators used a Dr. Pen A6 Ultimate microneedling device and set the needle depth to 2.5mm.
Hair parameters were assessed 4-8 weeks later, thus bringing the study duration to 20 weeks (despite each group only having received three microneedling + topical treatments).
Encouragingly, over 50% of people in the microneedling + topical dutasteride group observed “marked improvement” in their hair loss. Hair thickness, hair density, and the ratio of vellus:terminal hairs also improved versus the microneedling-only group.
Figure 8: Results from a 2022 study on microneedling + dutasteride (left column) versus microneedling alone (right column).[16]Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical … Continue reading
While serum DHT levels were not measured, there were no reports of sexual side effects from participants.
Our Analysis
This is another positive (albeit preliminary) signal that topical dutasteride can produce results and reduce our risk of adverse events (and drug exposure) – especially when paired with microneedling. Just see these before-and-after photos from one study participant:
Figure 9: (a). Vertex area trichoscopy and clinical photograph of a patient with androgenetic alopecia (Hamilton-Norwood Scale III) at baseline visit. (b). Frontal area trichoscopy and clinical photograph showing marked improvement (HamiltonNorwood Scale II) at week 16 after 3 sessions with microneedling plus topical 0.01% solution of dutasteride in monotherapy.[17]Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical … Continue reading
Another encouraging component of this study is that the total dutasteride dosage was lower than the 2018 study, yet still produced results. Whereas the 2018 study used dosages as high as 0.4mg weekly (at least for the first 8 weeks), this 2022 study used dosages equating to 0.1mg once every four weeks. If we are to take the minimal serum DHT changes from the 2018 study at face-value, then it’s even more unlikely that the dutasteride volumes used in this 2022 study led to appreciable changes to serum DHT.
Another study published this year (2025) also gave some insight into low topical dosages of dutasteride in people with AGA.
Panuganti et al, 2025: Topical Dutasteride at 0.01%, 0.02%, and 0.05% w/v
The investigators randomized 135 men with AGA into five groups: 0.01%, 0.02%, or 0.05% topical dutasteride (1 mL once daily), 1 mg oral finasteride daily, or a topical placebo. Treatments were continued for 24 weeks, with the topical solutions applied to a marked vertex target area.[18]Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., KSSVV, S.R., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate … Continue reading
Baseline characteristics were balanced across groups. Efficacy was assessed via macrophotography and target area hair counts (TAHC), global photographic assessment, and patient satisfaction questionnaires.
At 24 weeks:
- The 0.05% topical dutasteride group showed the largest mean increase in TAHC, reported as +75.5 hairs/cm² vs baseline (p=0.0001), compared with +41.2 hairs/cm² in the oral finasteride group (p=0.0001) and essentially no change in the placebo group (+0.07 hairs/cm², p=0.9957).
- The 0.01% and 0.02% topical groups improved versus placebo, but did not outperform oral finasteride.
- Hair shaft width improved in a dose-dependent manner; 0.05% topical dutasteride increased hair width by 11.6 µm vs placebo at week 24 (p=0.0185).
- Patient satisfaction and investigator global assessments were highest in the 0.05% group.
- The study also evaluated serum testosterone and DHT. Reported changes with topical dutasteride were modest and dose-dependent. In the 0.05% group, serum DHT decreased by 8.9% at week 12 and 11% at week 24, while testosterone changes were described as minor. In contrast, oral finasteride was associated with a 27% DHT reduction at week 12 and 11% at week 24, alongside a gradual increase in serum testosterone (up to ~20% by week 24).
- Across all topical dutasteride arms, the authors state there were no statistically significant changes in serum DHT or testosterone, low systemic exposure, and no sexual side effects or clinically relevant lab abnormalities. The 0.05% group delivered the greatest reported efficacy while appearing to avoid the hormonal shifts seen with oral finasteride.
Figure 10: Representative images of hair growth in male-pattern androgenetic alopecia after treatment with 0.01% dutasteride topical solution at 12 week (b) and 24 week (c) vs baseline (a); 0.02% dutasteride topical solution at 12 week (e) and 24 week (f) vs baseline (d); 0.05% dutasteride topical solution at 12 week (h) and 24 week (i) vs baseline (g); oral finasteride 1 mg tablets at week 12 (k) and week 24 (l) vs baseline (j); placebo at week 12 (n) and week 24 (o) vs baseline (m).[19]Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., KSSVV, S.R., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate … Continue reading
Our Analysis
On the surface, the Panuganti trial is extremely attractive: it suggests that low-dose topical dutasteride (0.05%) can outperform 1 mg oral finasteride in hair regrowth while producing only minimal average changes in serum DHT. However, several issues with the design and reporting warrant caution, and in our view, the topline conclusion should not be taken at face value.
Sponsorship and authorship
The trial was sponsored by Shilpa Medicare Limited, and all authors were company employees. Industry sponsorship does not invalidate results, but it does increase the importance of methodological transparency and independent replication—especially when findings are strikingly positive and out of step with real-world experience.
Hair counts that don’t match typical scalp biology
The reported baseline hair counts (≈300–330 hairs/cm²) in men with Norwood III–V vertex thinning are difficult to reconcile with established data. In non-balding adults, typical terminal hair densities are ~100–250 hairs/cm², and they fall substantially with moderate-to-severe AGA. Yet the study presents baseline photos of visibly thinned vertex regions alongside hair counts that exceed even full-density scalps.
The most likely explanations are that:
- Vellus hairs were included in the counts without a clear diameter cut-off (e.g., ≤40 µm), and/or
- The counted area was effectively larger than 1 cm², or hair counts were performed manually rather than via standardized software.
Any of these will inflate absolute hair counts and complicate the interpretation of changes over time.
Questionable consistency of the hair count target area
Accurate hair counting depends on revisiting the exact same 1 cm² area at each time point, typically anchored to a tattoo or permanent scalp mark. In the figures provided, the circular “counting zones” appear to shift in position, shape, and size between baseline, week 12, and week 24 for the same participant.
Even a 1–2 mm shift in the center of a 1 cm² circle can change measured hair counts by 50% or more in patchy thinning regions. Since the study reports changes on the order of ~10–15%, small misalignments in the counting area could fully account for the apparent “regrowth,” particularly in groups with smaller gains.
Limited insight into systemic exposure
Serum DHT and testosterone were reported only as group mean percentage changes, with no measures of variability (e.g., standard deviations) or individual pharmacokinetic correlations. This means:
- A reported 11% mean DHT decrease could still conceal substantial outliers with much larger reductions.
- We can’t assess whether men with the best regrowth had proportionally greater systemic exposure.
For a drug like dutasteride, where individual absorption and metabolism vary widely, this lack of granularity limits the conclusions we can draw about “safety” at the individual level.
Conflict with real-world data and dose–response expectations
Over several years, our community has tracked people using low-dose topical dutasteride (e.g., 0.01–0.02%, 1–2 mL daily, sometimes higher weekly exposure than in the 0.05% arm of this study). Broadly:
- Serum DHT typically changes by ~10–15%, which aligns with the idea of good scalp localization.
- Clinical outcomes are generally hair loss stabilization, with modest regrowth in some cases, not the large average gains suggested to outperform oral finasteride.
- More pronounced regrowth tends to appear only at much higher weekly exposure (e.g., around 8–10+ mg/week or with added microneedling), and at that point, systemic side effects become more common.
Taken together, this creates a tension: the study’s claim that 1 mL of 0.05% topical dutasteride daily can outperform 1 mg oral finasteride on average, without meaningful systemic effects, does not match what we see in practice or what we would expect based on dutasteride pharmacology.
Figure 11: Changes from baseline to week 24 in (a) testosterone levels and (b) DHT levels.[20]Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., KSSVV, S.R., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate … Continue reading
Overall, the Panuganti et al. trial supports that low-dose topical dutasteride (0.01–0.05%) has real biologic activity, can improve hair parameters versus placebo, and may keep average serum DHT changes relatively modest. However, issues with baseline hair counts, apparent inconsistencies in how target areas were measured, limited systemic exposure data, and a headline result that conflicts with prior studies and real-world experience mean we don’t think this study justifies expecting low-dose topical dutasteride to reliably outperform oral finasteride. For now, it’s more reasonable to view low-dose topical dutasteride (≤0.05% with limited weekly exposure) as a localized, hair-loss–stabilizing option with modest regrowth and relatively mild systemic impact, while higher-dose topical regimens are more likely to produce cosmetically meaningful gains but also to behave more like oral dutasteride in terms of systemic DHT suppression and potential side effects.
Yet another study came out in 2025, combining skin patting and iontophoresis (a medical procedure using a mild electrical current) with dutasteride gel in male and menopausal female AGA.
Cedirian et al., 2025: 6% Dutasteride Gel, Skin Patting and Iontophoresis
In this single-center pilot study, researchers enrolled 20 adults (10 men, 10 postmenopausal women) with AGA who had failed at least 12 months of standard therapy (topical minoxidil and/or oral finasteride).[21]Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M. (2025). Efficacy of Skin Patting and Iontophoresis with Dutasteride Gel in Male and … Continue reading Participants underwent monthly sessions of a compounded 6% dutasteride gel delivered using a device that combines skin patting (SPi), microneedling, iontophoresis, electrostimulation, and red LED light over a standardized frontal-vertex treatment field for four months.
Hair density and shaft diameter were assessed by trichoscopy at baseline and again 8 weeks after the final session (i.e., roughly 6 months from baseline), alongside a hair pull test and patient-reported satisfaction scores. By the end of follow-up, frontal and vertex hair density increased significantly (p<0.001), hair shaft diameter improved in both regions (vertex p<0.001; frontal p=0.046), and pull test counts nearly halved (p<0.001), pointing to both regrowth and reduced shedding.
Clinically, investigators rated mean global improvement at +1.9 on a 7-point scale, corresponding to moderate regrowth and less shedding. Subjective outcomes were similarly positive: about 70% of participants perceived the treatment as moderately to very effective, mean cosmetic satisfaction was 3.4/4, and 85% described the procedure as pleasant. No adverse events or tolerability concerns were reported during treatment or follow-up.
Our Analysis
On paper, this is an encouraging signal: the participants experienced measurable gains in density, diameter, and shedding. The high satisfaction scores and absence of side effects also support the idea that this approach is, at minimum, well-tolerated.
However, the design makes it difficult to isolate what’s actually driving the benefit. Because the protocol combines several modalities, we can’t separate the relative contribution of dutasteride from the other treatments. Outcome measures were also partly subjective and non-validated, and all assessments were unblinded, which opens the door to expectation and observer bias.
A second major limitation is the complete absence of systemic safety data. Unlike the other studies, a high dosage of topical dutasteride is used (6%), but we don’t know the effect of this on systemic/serum DHT levels.
While lab work is really the only way to get a grip on things, this study suggests that topical dosages as low as 1 mL x 0.01%, and as infrequently applied as once every four weeks, can still produce results for more than 50% of men with androgenic alopecia.
This begs the question: just how low can we go for dosing? While we don’t yet have the answer, studies on a different delivery vehicle for dutasteride – mesotherapy – suggest we can go even lower.
Mesotherapy Dutasteride: Clinical Studies (Hair Counts, Serum DHT, & More)
Mesotherapy dutasteride is also known as intradermal dutasteride. This is different from topical dutasteride.
Whereas topical dutasteride is applied directly to the scalp, mesotherapy dutasteride is when dutasteride is injected directly into the scalp skin – between the epidermis and dermis (i.e., 0.5-0.7mm deep into the scalp).
Whereas only 25% of topical dutasteride is absorbed into the scalp dermis over 12 hours, 100% of mesotherapy dutasteride is absorbed into the scalp dermis, and immediately upon injection. After all, the drug is injected directly into the scalp, leaving no material to evaporate or get wiped away by sweat or during a shower.
That means, across equal dosages, that mesotherapy dutasteride will better saturate the scalp skin, but also lead to more systemic absorption.
The question then becomes: how do the pharmacokinetics of mesotherapy dutasteride change the equation for how much (and how frequently) we should be exposing our scalps to this medication in order to maximize hair gains while minimizing systemic DHT reductions?
While we don’t yet have perfect answers, there are a few studies that suggest the dose is even lower than topical dutasteride.
Corralo et. al (2017): Mesotherapy Dutasteride
A 2017 study by Corralo et al. investigated the effects of mesotherapy dutasteride on people with androgenic alopecia.[22]Saceda-Corralo, D., Rodrigues-Barata, A.R., Vano-Galvan, S., Jaen-Olasolo, P. (2017). Mesotherapy with Dutasteride in the Treatment of Androgenetic Alopecia. International Journal of Trichology. … Continue reading The researchers selected six participants to receive 1 mL of 0.01% dutasteride (i.e., 0.1 mg of dutasteride) once every three months, totaling three injection sessions at months 0, 3, and 6.
The investigation group also took serum DHT measurements before starting treatment, and one month after the final mesotherapy injection (according to personal email correspondences with the authors).
Three months after the last mesotherapy injection, the investigators measured hair parameter improvements. Encouragingly, 1 mL of 0.01% mesotherapy dutasteride once every three months led to statistically significant hair improvements for all participants. Moreover, no changes to serum hormonal profiles were observed.
Here are the before-and-after photos of a featured patient:
Figure 13: Androgenetic alopecia in a 33-year-old man before (a) and after (b) treatment with dutasteride injections through 6 months with a one-session treatment every 3 months.[23]Saceda-Corralo, D., Rodrigues-Barata, A.R., Vano-Galvan, S., Jaen-Olasolo, P. (2017). Mesotherapy with Dutasteride in the Treatment of Androgenetic Alopecia. International Journal of Trichology. … Continue reading
Our Analysis
The sample size of this study is incredibly small. With that said, the results are impressive, and they add to a growing number of signals suggesting that mesotherapy dutasteride (and topical dutasteride) improve hair growth for men and women with androgenic alopecia.
Moreover, this study suggests that a dosing schedule as infrequent as 1 mL of 0.01% dutasteride (i.e., 0.1 mg) once every three months can appreciably improve hair parameters, and without altering systemic levels of hormones.
However, just as with the last study measuring serum DHT levels, this investigation group waited a full month after a mesotherapy dutasteride session to read serum levels of DHT. While the results showed no effect on serum hormones, the reality is that serum DHT levels could have dropped during the days and weeks following an injection, only to rebound to baseline by the time these investigators decided to test.
On the other hand, it’s also possible that mesotherapy dutasteride changes the pharmacokinetics of dutasteride itself – perhaps by increasing tissue saturation time and decreasing systemic leakage rates. While this may sound absurd, other studies have demonstrated mesotherapy to exhibit this behavioral shift with other drugs and compounds.[24]Mammucari, M., Maggiori, E., Russo, D., Giorgio, C., Ronconi, G., Ferrara, P.E., Cazona, F., Antonaci, L., Violo, B., Velluci, R., Mediati, D.R., Migliore, A., Massafra, U., Bifarini, B., Gori, F., … Continue reading Therefore, it’s not unreasonable to assume these effects may also carry over to mesotherapy dutasteride, and that for unknown reasons, the drug has longer staying power in the scalp when injected (and less systemic absorption).
Based on the results of these serum DHT tests, preliminary evidence points toward mesotherapy dutasteride perhaps lasting longer in the scalp and conferring slower systemic absorption. But be forewarned: there is likely an upper limit for how much mesotherapy dutasteride we can inject before serum DHT levels start to decline.
Member Experience: Too Much Mesotherapy Dutasteride?
We once had a member test 2 mL of 0.05% dutasteride (i.e., 1.0 mg of dutasteride, or 10x the dosage used in the 2017 study) during his first mesotherapy session. He did a great job tracking serum DHT changes before treatment and 4 days, 4 weeks, and 8 weeks after the injections.
His results? Serum DHT decreases shortly after treatment, which continued to decline up through month one. At that point, the member started experiencing cognitive-related side effects. By the second month, serum DHT levels began to rebound and all side effects went away.
Serum DHT Reported side effects Pre-treatment 51 ng/dL (reference: 30-90 ng/dl) No Post-treatment (4 days) 35 ng/dL No Post-treatment (4 weeks) 25 ng/dL Yes Post-treatment (8 weeks) 31 ng/dL No With that said, there are a few things worth noting about this member’s experience:
- First, these effects occurred at 10x the dosage of the 2017 study by Saceda. So, it’s unlikely that if you follow the study parameters, the same thing will happen to you.
- Secondly, the delayed decline in serum DHT puzzled us, because it didn’t necessarily fit with the pharmacokinetics expected from mesotherapy or the better-studied pharmacokinetic expectations of oral dutasteride. Keep in mind that 1mg of oral dutasteride rapidly distributes across tissues and has a half-life of 7-14 days. With mesotherapy, much of the drug remains in the scalp skin and a lesser amount gets absorbed into the serum. Therefore, even if mesotherapy had delayed the systemic absorption of dutasteride, we shouldn’t expect the drop at 4 weeks to be more dramatic than the drop at 4 days – much less significantly lower at 4 weeks versus 4 days.
- Thirdly, after having a conversation with this member, we learned they also started using an over-the-counter allergy medication at some point after the mesotherapy sessions. Studies suggest that this allergy medication can lower testosterone, which can inadvertently lower DHT. As such, we’re not certain enough to attribute 100% of these serum DHT reductions exclusively to mesotherapy dutasteride – even if we believe the therapy to be responsible for the majority of the drops observed.
- Lastly, regardless of the drop in DHT and side effects, this member did report better hair growth after just one session.
So, by following lower dosage guidelines, we’re confident you can avoid a similar experience. In fact, a 2013 study on mesotherapy dutasteride echoes the experience from our member and shows why mesotherapy dosages, volumes, and frequencies matter a lot.
Sobhy et al. (2013): Low-Dose Vs. High-Dose Mesotherapy Dutasteride
In 2013, a research team in Egypt enrolled 90 men with androgenic alopecia to test mesotherapy dutasteride at varying dosages, with the higher dosage also containing other potential hair growth-promoting ingredients. They randomized the men into three groups, and then set them up with the following treatment:
- Group A: 1.5-2.0 mL of 0.005% mesotherapy dutasteride (i.e., 0.075-0.1 mg of dutasteride per session).
- Group B: 1.5-2.0 mL of 0.05% mesotherapy dutasteride (i.e., 0.75-1.0 mg of dutasteride per session; 10x more than Group A). The 10 mL solution also contained 500 mg of dexapanthenol, 20 mg of biotin 20 mg, and 200 mg of pyridoxine.
- Group C: 1.5-2.0 mL of saline solution (i.e., water; this is a placebo group).
Then, the investigators performed 9 mesotherapy sessions over a span of 19 weeks. The sessions were as follows:
- Once per week (weeks 0-3)
- Once every two weeks (weeks 5 and 7)
- Once every four weeks (weeks 11, 15, 19
Given this dosing schedule, here’s how much dutasteride each group was exposed to over 19 weeks:
- Group A: 0.675-0.9 mg
- Group B: 6.75-9.0 mg (i.e., 10x more than Group A)
- Group C: 0 mg (they’re the placebo group)
Results
After 19 weeks, here were the changes to hair parameters and serum DHT levels:
- Hair Parameters:
- Unsurprisingly, both Group A and Group B saw hair parameter improvements. However, Group B saw more robust improvements relative to Group A. This is probably because they received 10x the dosage of dutasteride.
- Serum DHT:
- Group A saw a significant increase in serum DHT levels before and after the study ended. The authors attributed this change to the preservation of diurnal DHT fluctuations. This suggests that at dosages of as low as 0.9 mg over 19 weeks, serum DHT levels are basically preserved.
- Group B saw a significant decrease in serum DHT levels. While this would be expected given what we’ve just learned about the dosing and the pharmacokinetics of both mesotherapy and dutasteride, the investigation group was also puzzled by the fact that the placebo group also saw a decline in serum DHT levels. Again, the researchers chalked it up to normal diurnal fluctuations to DHT. However, based on members’ experiences and the additional studies published since this 2013 investigation, our best bet is that Group B saw serum DHT levels decline mainly because of higher exposure rates to dutasteride.
The bottom line: stick to mesotherapy dutasteride dosages of 0.1-0.2 mg per month. That’s probably close to the “sweet spot” for maximizing hair gains while minimizing unwanted systemic hormonal effects.
Ultimately, a number of factors determine how much of a substance will be absorbed systemically and how much of that substance will remain saturated in a tissue to exert its maximal effect. These factors include:
- Drug pKa. This is a measurement of the acidity of a drug.
- Drug polarity. this affects the hydrophilic-hydrophobic ratio of a compound in reference to its location within the dermis.
- Location of the mesodermal application. Keep in mind the scalp dermis is distinctly different from the facial dermal layer.
- The diffusion coefficient of a compound.[25]Souto, E.B., Fangueiro, J.F., Fernandes, A.R., Cano, A., Sanchez-Lopez, E., Garcia, M.L., Severino, P., Paganelli, M.O., Chaud, M.V., Silva, A.M. (2022). Physicochemical and biopharmaceutical aspects … Continue reading This is a measurement for how quickly and widely a compound can diffuse in the surrounding tissue.
The above factors are influenced by drug preservatives, carrier agents, temperatures, injection needle depths, aqueous secretions from sweat glands, and even apocrine secretions from sebaceous glands (just to name a few). We can tell you right now: none of the studies on mesotherapy dutasteride made an effort to standardize these factors, or even mention them in their methodologies. In fact, we couldn’t even find standards for just how deep the mesotherapy needles were injected for any study. These are all big limitations for data quality.
(Side note: we’re planning on investigating these factors in early 2023, so that we can develop a better protocol for mesotherapy dutasteride – one that is optimized based on human data across multiple injection frequencies, exposure volumes, carrier agents, and needle depths. Expect more details about this toward the end of 2022.)
Are There Any Other Studies On Mesotherapy Dutasteride?
Yes. However, they mostly repeat the results we’ve discussed above. Moreover, to our knowledge, none of the other studies measured serum DHT levels, so they don’t help us get closer to answering our core question: “What’s the best protocol for mesotherapy dutasteride to maximize hair gains while minimizing systemic hormonal effects?”
With that said, there is one final study that provides some nuanced insights – particularly related to mesotherapy dutasteride and its variability in hair regrowth, depending on whether someone has metabolic syndrome.
Mofta et al. (2021): Mesotherapy Dutasteride In Females With Metabolic Syndrome
In this 2021 study, researchers sought to determine the effects of mesotherapy dutasteride on females with pattern hair loss (androgenic alopecia), and how hair growth outcomes changed depending on whether participants had metabolic syndrome.[26]Moftah, N., Mubarak, R., Abdelghani, R. (2021). Clinical, trichoscopic, and folliscopic identification of the impact of metabolic syndrome on the response to intradermal dutasteride 0.02% injection … Continue reading
The researchers identified 51 females with pattern hair loss. Then, they determined which women also had metabolic syndrome. For reference, the investigators defined metabolic syndrome as meeting at least three of the following criteria:
- Raised waist circumference (population- and country-specific definitions), i.e.,> 92.3 cm in Egyptian women
- Fasting blood glucose ≥ 100 mg/dL or on diabetes treatment
- Blood pressure ≥ 130/85 mm Hg, or on antihypertensive treatment
- Triglycerides ≥150 mg/dL or on treatment
- High-density lipoprotein-cholesterol < 50 mg/dL, or on treatment
This left 26 and 25 females with and without metabolic syndrome, respectively.
Next, the investigators administered mesotherapy dutasteride to all participants. This included 1 mL of 0.02% mesotherapy dutasteride (i.e., 0.2 mg of dutasteride) per session, with eight sessions spanning a period of ~6 months. A few months later, hair parameters were assessed to gauge results.
Results
Surprisingly, females with metabolic syndrome experienced an initial improvement in hair growth at month one, followed by an acceleration of hair loss by month three. Contrastingly, females without metabolic syndrome saw their hair growth increasingly improve throughout each month of treatment.
These results were not only reflected in phototrichograms (i.e., hair counts, hair diameters, and anagen:telogen ratios) but also in global photographs.
Just see these before-and-after photos of two females without metabolic syndrome:
Figure 14: Before and after of woman one without metabolic syndrome.[27]Moftah, N., Mubarak, R., Abdelghani, R. (2019). Clinical, trichoscopic, and folliscopic identification of the impact of metabolic syndrome on the response to intradermal dutasteride 0.02% injection … Continue reading
Figure 15: Before and after of woman two without metabolic syndrome.[28]Moftah, N., Mubarak, R., Abdelghani, R. (2019). Clinical, trichoscopic, and folliscopic identification of the impact of metabolic syndrome on the response to intradermal dutasteride 0.02% injection … Continue reading
Now see this before-and-after photoset of a female with metabolic syndrome:
Figure 16: Before-and-after images of a woman with metabolic syndrome.[29]Moftah, N., Mubarak, R., Abdelghani, R. (2019). Clinical, trichoscopic, and folliscopic identification of the impact of metabolic syndrome on the response to intradermal dutasteride 0.02% injection … Continue reading
On a positive note, no females in either group reported any side effects – sexual or otherwise. But the discrepancy in regrowth between those affected and unaffected by metabolic syndrome was, to put it simply, puzzling.
Why Did Female With Metabolic Syndrome See A Worsening Of Hair Loss With Mesotherapy Dutasteride?
The researchers did not have a definitive answer. However, some studies suggest an association between metabolic syndrome and androgenic alopecia, with mechanistic data suggesting that insulin insensitivity can enhance androgen production in certain tissues – like the ovaries in women and, perhaps, other organs like the skin.
Oral dutasteride is known to have a mildly negative effect on insulin sensitivity. For most healthy people, this isn’t problematic. But the data are less clear for people who are already afflicted with metabolic syndrome and then start taking dutasteride.
Here’s the rationale the authors used to explain their results:
“These findings could be explained by the study conducted by Upreti et al. [59] who confirmed that dual inhibition of 5α reductase I and II by dutasteride lead to modulation of insulin sensitivity in the peripheral tissues. Accordingly, dutasteride induced reduction of insulin sensitivity that is already impaired in both MetS [55, 60] and FPHL [61-63] could be a possible mechanism of deterioration in participants with MetS. This possibility was recently supported by Sadgrove [64] who suggested that insulin resistance could enhance microinflammation in androgenetic alopecia both directly and indirectly by increased microbial colonization. Moreover, an in vitro study carried out by Philpott et al. [39] revealed that removal of insulin from the culture medium of human hair follicles could lead to follicular transformation into catagen state.“
The bottom line: if you’re already insulin resistant, perhaps it is worth getting those levels within normal ranges before trying dutasteride altogether– including via mesotherapy delivery vehicles.
Where Can We Buy Mesotherapy Dutasteride?
Here are a few resources.
- SuperHumanStore (worldwide). We recently lab-tested this product. It passed all tests for purity and sterility. Unfortunately, the store no longer carries that product. So, we’re currently lab-testing their new product. We’ll update this guide when we have the results.
- FUE Clinic (U.S.). This company sells 0.01% mesotherapy dutasteride alongside biotin, pyridoxine, and a blend of “cellular matrix growth factors”. Don’t be fooled by those fancy words; we can assure you that cellular matrix growth factors are currently more so buzzwords than they are clinically-proven hair growth stimulants.
Where Can We Buy Topical Dutasteride?
Here are a few resources.
- MinoxidilMax (worldwide): 0.1% topical dutasteride, 60 mL. On the one hand, MinoxidilMax definitely sells prescription-grade hair loss drugs without requiring a prescription, which we’re 99% sure is illegal. On the other hand, when we lab-tested their products, they contained what they claimed – which is a good sign. We like MinoxidilMax because (1) they ship worldwide, and (2) they sell solvents to pair with their products – so you can dilute their topicals down to lower dosages: 0.005%, 0.01%, 0.02%, etc.
- Happy Head (U.S.). Their products contain topical finasteride, minoxidil and retinoic acid. But they allow customization by including topical dutasteride with or in place of finasteride.
- Regrowth Labs, D57α (U.S.). This is a topical solution containing dutasteride in a 0.5% concentration. Their suggested dose is 1mL which would give a 0.5mg dose topically. However, the company suggests applying this volume twice a day, daily. The accompanying ingredients include azelaic acid, vitamins, phytonutrients and polar carriers to help with absorption.
- CFS Pharmacy (U.S.). This also sells topical dutasteride as a prescription medication. The topical solution comes in a gel. While CFS Pharmacy promotes themselves as capable of making any dosage of topical dutasteride, we believe they require a prescription. So, before you exercise this option, be sure to find a doctor who will write you a prescription for the dutasteride concentration you’d like.
- Bauman Medical, 82D (U.S.). This is a topical dutasteride + minoxidil solution. The amount of dutasteride contained in the product is a 0.75% concentration. There is no mention of what the additional ingredients or carrier solutions are.
References[+]
References ↑1, ↑2 Herz-Ruelas, M.E., Alvarez-Villalobos, N.A., Millan-Alanis, J.M., de Leon-Gutierrez, H., Ocampo-Garza, S.S., Grimalt, R. (2020). Efficacy of Intralesional and Oral Dutasteride in the Treatment of Androgenetic Alopecia: A Systematic Review. Skin Appendage Disorders. 6(6). 338-345. Available at: https://doi.org/10.1159/000510697 ↑3, ↑4, ↑5, ↑6 Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., Cui, Y. (2019). The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and meta-analysis. Clinical Interventions in Aging. 14. 399-406. Available at: PMID 30863034 ↑7 Hagberg, K.W., Divan, H.A., Fang, S.C., Nickel, J.C., Jick, S.S. (2017). Risk of gynecomastia and breast cancer associated with the use of 5-alpha reductase inhibitors for benign prostatic hyperplasia. Clinical Epidemiology. 9. 83-91. Available at: https://doi.org/10.2147/CLEP.S124674 ↑8 Rahimi-Ardabili, B., Pourandarjani, R., Habibollahi, P., Mualeki, A. (2006). Finasteride induced depression: a prospective study. BMC Clinical Pharmacology. 6(7). Available at: https://doi.org/10.1186/1472-6904-6-7 ↑9 Wang, K., Fan, D-D., Jin, S., Xing, N-Z., Niu, Y-N. (2014). Differential expression of 5-alpha reductase isozymes in the prostate and its clinical implications. Asian Journal of Andrology. 16(2). 274-279. Available at: https://doi.org/10.4103/1008-682X.123664 ↑10 Gisleskog, P.O., Hermann, D., Hammarlund-Udenaes, M., Karlsson, M.O. (1999). The pharmacokinetic modelling of GI198745 (dutasteride), a compound with parallel linear and nonlinear elimination. British Journal of Clinical Pharmacology. 47. 53-58. Available at: https://doi.org/10.1046/j.1365-2125.1999.00843.x ↑11 Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmagd, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). 387-401. Available at: https://smj.journals.ekb.eg/article_42083_24b61cbba4be9982db23c318414034c0.pdf (Accessed: Novemeber 2025) ↑12, ↑13, ↑14 Nada, E.A., El-Dawla, R.E., El-Maged, W.M.A., Elmagd, M.A.A. (2018). Topical dutasteride with microneedling in treatment of male androgenetic alopecia. Sohag Medical Journal. 22(1). 387-401. Available at: https://smj.journals.ekb.eg/article_42083_24b61cbba4be9982db23c318414034c0.pdf (Accessed: November 2025) ↑15, ↑16, ↑17 Sanchez-Meza, E., Ocampo-Candiani, J., Gomez-Flores, M., Herz-Ruelas, M.E., Ocampo-Garza, J., Orizaga-y-Quiroga, T.L., Martinez-Moreno, A., Ocampo-Garza, S.S. (2022). Microneedling plus topical dutasteride solution for androgenetic alopecia: a randomized placebo-controlled study. JEADV. 36. c806-c808. Available at: https://doi.org/10.1111/jdv.18285 ↑18, ↑19, ↑20 Panuganti, V.K., Madala, P.K., Grandhi, V.R., Alluri, C.V., Mohammad, J., KSSVV, S.R., Dundigalla, M.R. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑21 Cedirian, S., Pampaloni, F., Quadrelli, F., Rapparini, L., Bruni, F., Martelli, G., Piraccini, B.M., Starace, M. (2025). Efficacy of Skin Patting and Iontophoresis with Dutasteride Gel in Male and Menopausal Female Androgenetic Alopecia: A Pilot Study. Dermatologic Therapy. 15(11). 3419-3424. Available at: https://doi.org/10.1007/s13555-025-01532-w. ↑22, ↑23 Saceda-Corralo, D., Rodrigues-Barata, A.R., Vano-Galvan, S., Jaen-Olasolo, P. (2017). Mesotherapy with Dutasteride in the Treatment of Androgenetic Alopecia. International Journal of Trichology. 9(3). 143-145. Available at: https://doi.org/10.4103/ijt.ijt_73_16 ↑24 Mammucari, M., Maggiori, E., Russo, D., Giorgio, C., Ronconi, G., Ferrara, P.E., Cazona, F., Antonaci, L., Violo, B., Velluci, R., Mediati, D.R., Migliore, A., Massafra, U., Bifarini, B., Gori, F., di Carlo, M., Brauneis, S., Paolucci, T., Rocchi, P., Cugutti, A., Di Marzo, R., Bomprezzi, A., Santini, S., Giardini, M., Catizzone, A.R., Troili, F., Dorato, D., Gallo, A., Guglielmo, C., Natoli, S. (2020). Mesotherapy: From Historical Notes to Scientific Evidence and Future Prospects. ScientificWorldJournal. 3542848. Available at: https://doi.org/10.1155/2020/3542848 ↑25 Souto, E.B., Fangueiro, J.F., Fernandes, A.R., Cano, A., Sanchez-Lopez, E., Garcia, M.L., Severino, P., Paganelli, M.O., Chaud, M.V., Silva, A.M. (2022). Physicochemical and biopharmaceutical aspects influencing skin permeation and role of SLN and NLC for skin drug delivery. Heliyon. 8(2). e08938. Available at: https://doi.org/10.1016/j.heliyon.2022.e08938 ↑26 Moftah, N., Mubarak, R., Abdelghani, R. (2021). Clinical, trichoscopic, and folliscopic identification of the impact of metabolic syndrome on the response to intradermal dutasteride 0.02% injection in patients with female pattern hair loss: a prospective cohort study. 32(7). 827-836. Available at: https://doi.org/10.1080/09546634.2019.1708849 ↑27, ↑28, ↑29 Moftah, N., Mubarak, R., Abdelghani, R. (2019). Clinical, trichoscopic, and folliscopic identification of the impact of metabolic syndrome on the response to intradermal dutasteride 0.02% injection in patients with female pattern hair loss: a prospective cohort study. Journal of Dermatological Treatment. 32(7). Available at: https://doi.org/10.1080/09546634.2019.1708849 IngredientsPharmaceuticalsNatural RemediesCompany ReviewsBy Michael Williams, PhDJan 12, 2026Switching From Topical Minoxidil To Oral Minoxidil
Thinking about switching from topical to oral minoxidil, or back again? Changing your approach to hair loss can significantly improve your results, but changing treatment can also be a gamble that sets back progress. We break down what the evidence actually shows, when you might want to consider swi...By Cassie Hopton, PhDJan 12, 2026Does Spironolactone Cause Weight Gain?
Spironolactone is a popular off-label treatment for female pattern hair loss, but many users worry about gaining weight while taking it. This article breaks down how the medication works, what the research actually shows about weight changes, typical dosing for hair growth, and simple strategies to ...By Sophie Grice, PhDJan 9, 2026RU58841 for Hair Loss: Is It Legit?
RU58841 is one of the most talked-about research chemicals in hair loss forums, and the anecdotes are often surprisingly positive. Mechanistically, it makes sense: block androgen receptors in the scalp so dihydrotestosterone (DHT) can’t trigger follicle miniaturization. But the problem is that the h...By Michael Williams, PhDJan 9, 2026Oral vs. Topical Minoxidil: What’s Better for Hair Loss?
Oral vs topical minoxidil for hair loss: which works better, and at what cost? We break down the evidence on effectiveness, safety, dosing, and real-world tradeoffs to help you choose the right option.
By Michael Williams, PhDDec 31, 2025Finasteride vs. Dutasteride: The Ultimate Guide
Finasteride and dutasteride are the two most powerful medications available for androgenetic alopecia, but choosing between them isn’t straightforward. We break down how these drugs truly compare across efficacy, side effects, oral versus topical use, and real-world decision-making, so you can deter...By Michael Williams, PhDDec 31, 202510 Ways to Reduce the Side Effects of Finasteride
Finasteride is one of the most effective treatments for male pattern hair loss, but side effects can happen. This evidence-based guide outlines 10 practical strategies to reduce or manage finasteride side effects, from dose and formulation adjustments to lifestyle considerations, while emphasizing w...By Michael Williams, PhDDec 17, 2025Oral vs. Topical Dutasteride: What Studies Show
Oral dutasteride is one of the most effective treatments for androgenic alopecia, but its systemic hormone suppression raises safety concerns. Topical dutasteride is often promoted as a way to deliver similar scalp-level benefits with fewer side effects, but does the evidence support that claim? In ...By Ben Fletcher, PhDDec 17, 2025Minoxidil Shedding – What to Expect & When it Stops
Starting minoxidil and seeing more hair fall out? You’re not alone. This article breaks down the phenomenon known as the “dread shed”—a temporary spike in hair loss that can happen when beginning treatment. We explore why this shedding occurs, what it means for your hair cycle, and whether it’s actu...By Perfect Hair Health TeamDec 17, 2025Peppermint Oil for Hair Growth: Better Than Minoxidil?
In 2014, a study on mice found that peppermint oil improved hair growth outcomes better than minoxidil. In 2015, a study on men with androgenic alopecia showed that topical rosemary oil (similar to peppermint) improved hair counts, and as effectively as minoxidil. While these studies appear promisin...By Perfect Hair Health TeamDec 17, 2025Caffeine For Hair Loss (AGA): Evidence & Recommendations
Caffeine’s role in hair loss (and hair growth) is hotly debated. On the one hand, oral caffeine may indirectly exacerbate hair loss in those who are hyperglycemic or hypothyroid. On the other hand, topical caffeine does have efficacy as a potential hair loss intervention – at least when combin... - Mission Statement
 Scroll Down
Scroll Down