- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Does Anavar Cause Hair Loss?
-
10 Best Shampoos for Hair Loss in 2026
-
XYON Review: Do Their Products Actually Regrow Hair?
-
Minoxidil Before and After Photos [2026] | Does It Work?
-
How to Get Finasteride: Is It Over the Counter?
-
Keeps Review: The Truth About Their Hair Loss Treatments?
-
7 Best Oils for Hair Growth
-
Hims Hair Growth Reviews: The Pros, Cons, and Real Results
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
Articles
Oral minoxidil has become a widely used off-label option for androgenic alopecia (AGA) in men and women, despite initially being approved only as an antihypertensive medication decades ago.
Oral minoxidil is increasingly offered to address specific unmet needs in AGA treatment:
- Topical non-responders: Some people don’t see satisfactory results with topical minoxidil, possibly due to inadequate absorption, variations in scalp skin properties, or genetic factors affecting drug metabolism.
- Scalp sensitivity/irritation: Topical minoxidil can cause local side effects such as itching, flaking, or dermatitis due to the vehicle (especially propylene glycol) or the active ingredient itself. Switching to oral minoxidil bypasses these local reactions.
- Simpler routines/adherence: Oral dosing eliminates the need for daily topical application, which can be messy, time-consuming, or interfere with hair styling. For some, this leads to better long-term adherence and quality of life.
Regrowth with oral minoxidil can be variable, however. This variability mainly stems from biological differences between patients. Because minoxidil is a prodrug that requires conversion by the enzyme sulfotransferase (SULT1A1) into its active form, individual variation in enzyme activity strongly influences response.[1]Ramos, P.M., Goren, A., Sinclair, R., Miot, H.A. (2020). Oral minoxidil bio-activation by hair follicle outer root sheath cell sulfotransferase enzymes predicts clinical efficacy in female pattern … Continue reading
Interested in Oral Minoxidil?
Low-dose oral minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Unfortunately, what most people see online are extremely successful oral minoxidil before and after photos (which you’ll see below), which can lead people to believe that they will experience more regrowth than they actually might.
Source: u/pomidorich via r/FemaleHairLoss.
In this article, we will explore what the likely (not just possible) results are and what the clinical trials show.
But first, let’s have a quick refresher on what oral minoxidil is.
What is Oral Minoxidil?
Oral minoxidil is a tablet medication originally developed as a vasodilator for severe hypertension, but now increasingly used off-label at low doses to treat AGA and other hair loss conditions. It enhances scalp flow and prolongs the growth phase (anagen) of hair follicles, primarily by increasing prostaglandin E2 and vascular endothelial growth factor (VEGF).
Oral minoxidil acts by opening ATP-sensitive potassium channels in vascular smooth muscle, which increases perifollicular blood flow. It also prolongs the anagen phase of the hair cycle. Unlike topical minoxidil, which requires local sulfotransferase enzyme activity in the scalp for conversion to its active form, oral minoxidil is metabolized systemically via hepatic sulfotransferase, potentially bypassing poor scalp enzyme activity in some individuals.[2]Kaiser, M., Abdin, R., Gaumond, S.I., Issa, N.T., Jiminez, J.J. (2023). Treatment of Androgenetic Alopecia: Current Guidance and Unmet Needs. Clinical, Cosmetic and Investigational Dermatology. 16. … Continue reading
For hair loss, oral minoxidil is prescribed at significantly reduced dosages compared to antihypertensive therapy – typically:
- 0.25 mg – 1.25 mg for women
- 2.5 mg – 5 mg for men
- Adverse effects are much less frequent at these low doses but may include hypertrichosis (excess unwanted body hair), fluid retention, and, rarely, cardiovascular effects.[3]Bloch, L.D., Carlos, R.M.D. (2024). Side Effects’ Frequency Assessment of Low Dose Oral Minoxidil in Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: … Continue reading
While oral minoxidil is not FDA-approved for hair loss, a number of studies show its efficacy.
The Evidence
You can see a complete table of oral minoxidil studies here, but we have picked a few to show.
Study One – AGA (Female)
Vahabi-Amlashi et al (2021) conducted a triple-blind, randomized clinical trial in 72 women with female pattern hair loss, comparing oral minoxidil 0.25 mg daily to 2% topical minoxidil twice daily over 36 weeks.[4]Vahabi-Amlashi, S., Layegh, P., Kiafar, B., Hoseininezhad, M., Abbaspour, M., Khaniki, S.H., Forouzanfar, M., Sabeti, V. (2021). A randomized clinical trial on the therapeutic effects of 0.25 mg oral … Continue reading
- Measured outcomes included hair shedding, Sinclair scores by three independent evaluators, trichoscopy assessments of hair density and diameter, and safety labs (liver, kidney, electrolytes).
- Both groups showed significant improvement in hair diameter and density:
- Group 1 (oral): 13% improvement in hair density.
- Group 2 (topical): 6% improvement in hair density.
- Sinclair and shedding scores decreased significantly in both groups by the end of the study.
- There was no significant difference between groups in any measured endpoint.
- Oral minoxidil showed slightly better improvement in hair density; topical minoxidil showed slightly better improvement in hair diameter.
- Adverse events:
- Group 1 (oral): hirsutism (7.7%), gastrointestinal intolerance (7.7%), weight gain (3.8%), hypotension (3.8%), leading to withdrawal.
- Group 2 (topical): hirsutism (8%).
Study Two – AGA (Male)
Panchaprateep and Lueangarun (2020) conducted an open-label, prospective single-arm study in 30 men with AGA, treated with 5 mg oral minoxidil once daily for 24 weeks.[5]Panchaprateep, R., Lueangarun, S. (2020). Efficacy and Safety of Oral Minoxidil 5 mg Once Daily in the Treatment of Male Patients with Androgenetic Alopecia: An Open-Label and Global Photographic … Continue reading
- Statistically significant increase in total hair count at 24 weeks (+35.1±18.9 hairs/cm2; p=0.003).
- Hair diameter improved by 15.2% from baseline (p<0.001).
- Global photographic assessment: 43% of patients achieved an “excellent improvement” score by study end.
- High rate of hypertrichosis (93.3% at 24 weeks); pedal edema was reported in 3 patients, resolving after 2-3 months in all cases.
- No serious cardiovascular side effects were observed.
Study Three – AGA and TE (Male and Female)
Feaster et al. (2023) performed a multicenter retrospective study in 210 patients (50 men, 160 women) with AGA and/or telogen effluvium (TE), comparing low-dose oral minoxidil (0.625 – 2.5 mg daily) to a control (no minoxidil or other non-minoxidil treatments) over 52 weeks.[6]Feaster, B., Onamusi, T., Cooley, J.E., McMichael, A.J. (2023). Oral minoxidil use in androgenetic alopecia and telogen effluvium. Archives of Dermatological Research. 315(2). 201-205. Available at: … Continue reading
- Group 1 (oral minoxidil): 52.4% of patients showed clinical improvement in hair growth; 41.9% had hair loss stabilization; 5.7% experienced worsening.
- These results were significantly better than the control group (p=0.001).
- The mean duration of oral minoxidil use was 25.1 months (range 12-54); the most common dose was 1.25 mg daily.
- Adverse effects were uncommon (9.5% of patients), generally mild, and included:
- Hypertrichosis (4.8%)
- Edema (1.9%)
- Hair shedding (1%)
- Lower extremity cramping (1%)
- Palpitations (1%)
- Scalp dysesthesia (1%)
- There were no serious side effects or study withdrawals due to adverse events in the analyzed population.
Overall, these studies demonstrate that oral minoxidil is an effective and generally well-tolerated treatment option for both AGA and TE in men and women. Across the studies, clinical improvements were observed, with similar efficacy to topical minoxidil.
However, the results that people show online are usually more dramatic than what is seen in clinical studies.
What’s Possible ≠ What’s Probable
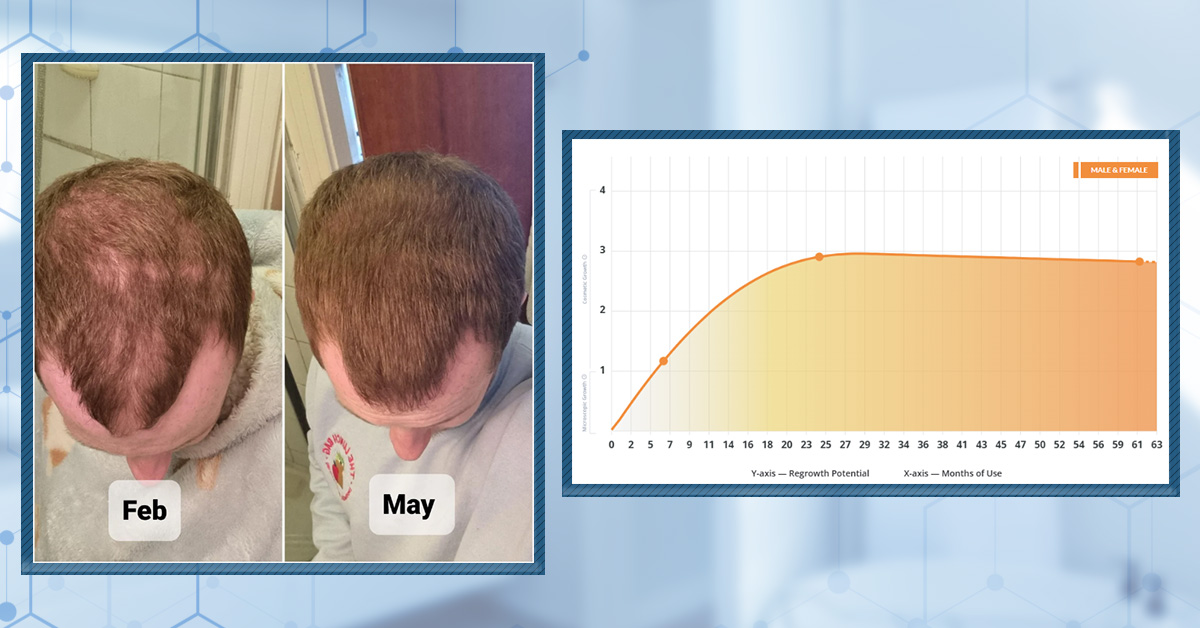
When reviewing outcomes, such as oral minoxidil before and after photos, it’s essential to distinguish what can happen from what is likely to happen. Take a look at the example we have created below. The scattered data points represent the full spectrum of possible individual results, while the central trendline shows the average, reflecting the most common or expected response.
Figure 1: A graph we created showing possible and probable results. The arrows point to possible results, and the trendline shows probable results.
These anecdotes (arrows on the graphs) often highlight “hyper-responders”. These are individuals who experience notably dramatic hair regrowth. They may have higher SULT1A1 activity, leading to higher levels of minoxidil activation.
Figure 2: Anecdotal results often look significantly more dramatic than clinical results.
These accounts, although genuine, illustrate results that surpass what most people can achieve. Problems can then arise when people fixate on these exceptional outcomes and assume that their experience will be similar. If their own hair growth is slower or less impressive, they might discontinue treatment early, or wrongly conclude that it isn’t working.
Figure 3: This leads to high expectations when the reality is that results will often be a lot less dramatic.
Impressive results are more frequently shared than modest improvements as a result of survivorship bias. This bias happens when only the most impressive successes receive attention, while the many cases with typical or less remarkable outcomes are overlooked or not reported.
Survivorship bias can distort our understanding of how hair loss treatments perform. Online narratives often draw attention to the most extreme cases, either astonishing successes or clear failures, while the more typical moderate outcomes that most people experience are rarely seen or discussed.
Now that we have covered the clinical evidence, strategies, and reasonable expectations for oral minoxidil, let’s now look into 10 firsthand accounts of oral minoxidil before and after photos shared by real users on Reddit.
Oral Minoxidil Success Stories
Keep in mind that the experiences below are purely anecdotal in nature and are not subject to any independent verification. They strictly reflect the individual journeys of each poster and should not be construed as reliable clinical evidence or medical advice.
Case 1: Male (29), Oral minoxidil monotherapy (11 months, 5 mg nightly)
Source: u/jeffersonsteelflex94 via r/tressless.
This Redditor began oral minoxidil monotherapy at a dose of 2.5 mg before increasing to 5 mg once nightly. He had switched from topical minoxidil after a year of subpar results. He experienced shedding in the early months of therapy, with visible regrowth by month 3, and a major improvement in hair density by 11 months. Overall, the patient was satisfied with thicker scalp coverage, a fuller beard and brows, and did not report any significant side effects.
Case 2: Male (26), finasteride and oral minoxidil (2 years total; 1 mg finasteride and 2.5 mg oral minoxidil daily)
Source: u/GriffsChoice via r/tressless.
This 26-year-old used a combination of 1 mg finasteride and 2.5 mg oral minoxidil once daily, having switched from topical to oral minoxidil one year into treatment. He reported notable improvement one year into therapy, with visible regrowth by the 4-month mark. He did not report any shedding or other side effects after switching from topical to oral formulations. After two years of treatment, the patient reported marked hair thickening and cosmetic improvement.
Case 3: Female, oral minoxidil monotherapy (2 years; 2.5 mg daily, titrated up from 1.25 mg)
This user reported one her progress with oral minoxidil monotherapy over 2 years. She started at a daily dose of 1.25 mg, before increasing to 2.5 mg. She had tried vitamins, spironolactone, oils, and Nioxin/Nizoral without benefit for one year prior. Shedding started shortly after beginning the protocol and normalized by the fourth month. She reported visible improvement within one year, and by the end of the second year, she had notable improvement in ponytail thickness and part density. She also reported that her hair looked and felt close to pre-loss baseline. The user did not report any significant side effects.Source: u/[deleted] via r/FemaleHairLoss.
Case 4: Female (24), Oral and topical minoxidil (9 months topical 5% foam; 6 months oral 0.625 – 1.25 mg nightly)
Source: u/pomidorich via r/FemaleHairLoss.
This 24-year-old began minoxidil monotherapy with 5% foam nightly, before adding oral minoxidil 0.625 mg three months later, and titrating to 1.25 mg nightly the following month. She continued both nightly for six months. She did not note any noticeable shedding on the foam, nor after incorporating 0.625 mg orally. She reported steady baby hairs and a reduction in hair loss at ~6-9 months alongside thickening compared to the baseline, and mentioned that her hair felt and looked much closer to its pre-loss condition overall. She also reported faster growth of facial/body hair (hypertrichosis), water retention, and bloating after salt meals, and occasional stronger heartbeat at night, after incorporating oral minoxidil, though this last symptom did resolve.
Case 5: Male, oral minoxidil (6 weeks shown; 5 mg daily) and weekly microneedling
Source: u/No_Pop2289 via r/tressless.
This male Redditor used oral minoxidil 5 mg once daily in addition to microneedling once weekly with a pen device. The poster also noted no side effects from finasteride, implying that they might have used it alongside the other treatments. He reported an initial shedding phase at around week 3 and rapid visible coverage by week 6 into treatment. In commenting on the progress to date, he noted fuller coverage compared to baseline and said his beard was slightly thicker without any apparent increase in body hair. He did not report any side effects.
Case 6: Female, oral minoxidil + spironolactone (1 year; 1.25 mg oral minoxidil daily, 50 mg spironolactone daily)
Source: u/monicatheshark5 via r/FemaleHairLoss.
The poster took oral minoxidil 1.25 mg once daily, in addition to spironolactone 50 mg once daily (started at 100 mg but reduced the dose due to drowsiness). She noted visible improvement after about 5 months of consistent use, with shedding slowing markedly around weeks 7-8. At the one-year mark, she reported noticeably fuller hair at the part and temples, with ongoing gradual thickening, and “good hair days”. She reported increased body and facial hair from oral minoxidil, which she managed by waxing, and did not report any noticeable weight gain or low-blood-pressure symptoms.
Case 7: Female, oral minoxidil monotherapy (6 months; 1.25 mg daily, titrated from 0.625 mg)
Source: u/Aggressive_Space_121 via r/FemaleHairLoss.
This female Redditor took oral minoxidil 1.25 mg once daily for six months, having started from the initial dose of 0.625 mg. She supplemented with vitamin B12 injections every two weeks for pernicious anemia, daily shampooing, higher protein intake, and therapy/stress management. She reported steady improvement in hair through month 6, despite a brief “dread shed” in the initial month. Unfortunately, the images don’t show the same hair pulled back, so we can’t see exactly how much improvement was gained. She did report increased facial hair, which she has been managing by shaving/dermaplaning.
Case 8: Female, oral minoxidil monotherapy (1 year; 1.25 mg daily)
Source: u/neptunesummer via r/FemaleHairLoss.
This user took oral minoxidil 1.25 mg once daily for one year. She also used Nizoral shampoo on the same timeline. Her hair loss was first diagnosed as telogen effluvium that did not resolve, and she now suspects AGA. She reported modest shedding in the first 2 months, then gradual thickening over the year. The photos showed some improvement, but the patient stated that she is not fully back to baseline. Regarding side effects, she noted increased body hair growth and mild dizziness in the first month that resolved.
Case 9: Male, self-made oral minoxidil (3 months; ingested diluted topical solution)
Source: u/Frosty_Wedding8706 via r/tressless.
This male Redditor switched from applying topical minoxidil to drinking a diluted solution mixed with water. He reported taking “a few drops” per dose, having discontinued application of the topical preparation to the scalp. At the three-month mark, the Redditor reported clearly greater overall density and said results were still improving. He did not report any side effects, except for some extra body hair.
We should mention that we do not recommend ingesting topical minoxidil – if oral therapy is pursued, it is advised to use a prescribed oral dose under medical supervision to control dosing and avoid health risks.
Case 10: Female, oral minoxidil monotherapy (12 months; dose not reported)
Source: u/blahblahyayah via r/FemaleHairLoss.
This user reported taking oral minoxidil at an unspecified dose for about one year. The photos show modest improvements around the parting. The poster did not report any side effects. Notwithstanding the sparse information provided by this Redditor, multiple commenters observed hair thickening and density gains in the before-and-after photos.
Strategies to Optimize Oral Minoxidil Response
To maximize the benefits of oral minoxidil, consistent use and patience are essential as optimal regrowth often takes 6-8 months to fully manifest. Combining oral minoxidil with other treatments like finasteride or dutasteride, platelet-rich plasma (PRP), or low-level laser therapy (LLLT) can improve results for those who do not respond sufficiently to minoxidil alone. Recent studies support the efficacy of combination regimens, particularly oral minoxidil plus finasteride, with one retrospective study finding significant and clinically meaningful improvements.[7]Johnson, H., Huang, D., Clift, A.K., Bersch-Ferreira, A., Guimaraes, G.A. (2025). Effectiveness of Combined Oral Minoxidil and Finasteride in Male Androgenetic Alopecia: A Retrospective Service … Continue reading
Figure 4: Percentage of patients who are stable or improved, or improved only, across increasing Norwood severities after combination finasteride and oral minoxidil treatment.[8]Johnson, H., Huang, D., Clift, A.K., Bersch-Ferreira, A., Guimaraes, G.A. (2025). Effectiveness of Combined Oral Minoxidil and Finasteride in Male Androgenetic Alopecia: A Retrospective Service … Continue reading Image used in accordance with the PMC Copyright notice.
People using oral minoxidil, especially at 2.5 mg or more and in combination with other treatments, should keep an eye on potential side effects. This can help catch rare but potentially serious side effects like edema or changes in blood pressure. Regular follow-ups can ensure that any adverse effects are detected early and that the regimen can be safely adjusted to maintain a balance between safety and efficacy. This patient-tailored management increases the likelihood of achieving the best possible outcomes while minimizing health risks.
Probable Regrowth Timeline
Based on the available evidence, the expected regrowth timeline for oral minoxidil is around 8 months.
Months 0-3: Possible increased shedding; no visible gains
- Temporary shedding can occur as follicles transition into a new growth cycle.
- This process often resolves within the first 4-8 weeks and is seen as an early sign that the treatment is starting to take effect.
Months 3-6: First positive density changes for many
- Early visible improvements, such as reduced shedding and appearance of fine new hairs, can be expected.
- Thicker, darker regrowth starts to become apparent for most users during this period.
Months 6-8: Maximal visible benefits
- Cosmetic changes peak, with stronger hair, increased density, and the greatest coverage observed.
- This is the period of maximum regrowth for most individuals.
Months 8-12+
- Further gains plateau.
- Continued daily use is needed to maintain improvements.
- Some might consider combination treatment at this point to increase growth outcomes.
Safety and Side Effects
While oral minoxidil can avoid local irritation occurring with the topical formulation, some safety concerns do remain.
Oral minoxidil use does carry a risk of cardiovascular issues like pericardial effusion, especially at the high doses used for blood pressure control. At hair loss doses (0.25 – 5 mg), such events are rare and tend to be individual rather than predictable.
The majority of side effects, such as excess body hair, mild ankle swelling, lightheadedness, or palpitations, are mild and usually resolve with dose adjustments or stopping the medication.
Large safety studies have shown that serious side effects are exceedingly rare in otherwise healthy individuals taking low-dose oral minoxidil, with only a small percentage discontinuing treatment due to adverse effects.
A gradual dose escalation is recommended to minimize side effects with oral minoxidil. Recent studies have shown that a 2.5 mg daily dose is as effective as 5 mg for treating male pattern hair loss, but with a better safety profile, and this supports 2.5 mg as a preferred starting dose for men.[9]Fonseca, L.P.C., Miot, H.A., Chaves, C.R.P., Ramos, P.M. (2025). Oral Minoxidil 2.5 mg vs 5 mg for Male Androgenetic Alopecia: A Double-Blind Randomized Clinical Trial. Journal of the American … Continue reading
If you want to use a higher dose, sublingual minoxidil may be beneficial for reducing the risk of cardiovascular side effects.[10]Gupta, A.K., Bamimore, M.A., Williams, G., Talukder, M. (2025). Comparative Efficacy of Minoxidil and 5-Alpha Reductase Inhibitors Monotherapy for Male Pattern Hair Loss: Network Meta-Analysis Study … Continue reading
Practical Takeaways
- Oral minoxidil is an effective and generally well-tolerated alternative for people who cannot use topical minoxidil or do not respond to it.
- Clinical trial results typically show stabilization or modest regrowth. Hyper-responder cases seen online are possible but not common.
- Careful dose selection (0.25 – 2.5 mg daily) minimizes side effects while maintaining efficacy.
- Consistent, long-term use is key; optimal results generally take 6-8 months to appear and require ongoing treatment to maintain gains.
- Combining oral minoxidil with other therapies such as finasteride, dutasteride, PRP, or LLLT may improve outcomes for partial responders.
Final Thoughts
Oral minoxidil use is growing, but should be recognized as an off-label option (albeit backed by a solid body of emerging evidence.. Most users can expect stabilization or noticeable yet modest regrowth rather than dramatic transformation. The best outcomes come with realistic expectations, consistent use over several months, and regular medical supervision to ensure safety. By focusing on average, evidence-based results rather than rare hyper-responder anecdotes, patients can make balanced decisions and maintain long-term adherence to their treatment plan.
References[+]
References ↑1 Ramos, P.M., Goren, A., Sinclair, R., Miot, H.A. (2020). Oral minoxidil bio-activation by hair follicle outer root sheath cell sulfotransferase enzymes predicts clinical efficacy in female pattern hair loss. Journal of the European Academy of Dermatology and Venereology. 34(1). E40-e41. Available at: https://doi.org/10.1111/jdv.15891 ↑2 Kaiser, M., Abdin, R., Gaumond, S.I., Issa, N.T., Jiminez, J.J. (2023). Treatment of Androgenetic Alopecia: Current Guidance and Unmet Needs. Clinical, Cosmetic and Investigational Dermatology. 16. 1387-1406. Available at: https://doi.org/10.2147/CCID.S385861 ↑3 Bloch, L.D., Carlos, R.M.D. (2024). Side Effects’ Frequency Assessment of Low Dose Oral Minoxidil in Male Androgenetic Alopecia Patients. Skin Appendage Disorders. 11(1). 14-18. Available at: https://doi.org/10.1159/00539969 ↑4 Vahabi-Amlashi, S., Layegh, P., Kiafar, B., Hoseininezhad, M., Abbaspour, M., Khaniki, S.H., Forouzanfar, M., Sabeti, V. (2021). A randomized clinical trial on the therapeutic effects of 0.25 mg oral minoxidil tablets on treatment of female pattern hair loss. Dermatological Therapy. 34(6). E15131. Available at: https://doi.org/10.1111/dth.15131 ↑5 Panchaprateep, R., Lueangarun, S. (2020). Efficacy and Safety of Oral Minoxidil 5 mg Once Daily in the Treatment of Male Patients with Androgenetic Alopecia: An Open-Label and Global Photographic Assessment. Dermatology and Therapy. 10. 1345-1357. Available at: https://doi.org/10.1007/s13555-020-00448-x ↑6 Feaster, B., Onamusi, T., Cooley, J.E., McMichael, A.J. (2023). Oral minoxidil use in androgenetic alopecia and telogen effluvium. Archives of Dermatological Research. 315(2). 201-205. Available at: https://doi.org/10.1007/s00403-022-02331-5 ↑7 Johnson, H., Huang, D., Clift, A.K., Bersch-Ferreira, A., Guimaraes, G.A. (2025). Effectiveness of Combined Oral Minoxidil and Finasteride in Male Androgenetic Alopecia: A Retrospective Service Evaluation. Cureus. 17(1). E77549. Available at: https://doi.org/10.7759/cureus.77549 ↑8 Johnson, H., Huang, D., Clift, A.K., Bersch-Ferreira, A., Guimaraes, G.A. (2025). Effectiveness of Combined Oral Minoxidil and Finasteride in Male Androgenetic Alopecia: A Retrospective Service Evaluation. Cureus. 17(1). E77549. Available at: https://doi.org/10.7759/cureus.77549 ↑9 Fonseca, L.P.C., Miot, H.A., Chaves, C.R.P., Ramos, P.M. (2025). Oral Minoxidil 2.5 mg vs 5 mg for Male Androgenetic Alopecia: A Double-Blind Randomized Clinical Trial. Journal of the American Academy Dermatology. 15. Available at: https://doi.org/10.1016/j.jaad.2025.09.031 ↑10 Gupta, A.K., Bamimore, M.A., Williams, G., Talukder, M. (2025). Comparative Efficacy of Minoxidil and 5-Alpha Reductase Inhibitors Monotherapy for Male Pattern Hair Loss: Network Meta-Analysis Study of Current Empirical Evidence. Journal of Cosmetic Dermatology. 62(2). 257-259. Available at: https://doi.org/10.1111/ijd.163737 Finding the right hair loss treatment can feel overwhelming, and expert guidance is essential to identify the best formula for your needs. The PhD-led team at Perfect Hair Health leverages emerging research and close collaborations with top innovators to provide readers with the most accurate and actionable reviews of hair restoration offerings. Topical finasteride is one of the most sought-after treatments on the market, boasting clinical support, targeted results, and easy application.
This comprehensive guide to the top-rated topical finasteride products is based on extensive research, expert recommendations, and the latest consumer feedback. Whether you’re just beginning your hair health journey or searching for better hair loss solutions, this article provides valuable information on safe and effective treatment options. Our recent partnership with Ulo enhances this guide with privileged insights on clinical findings and state-of-the-art protocols.
Read on for specialized guidance, insightful reviews, and proven recommendations of topical finasteride preparations from today’s leading brands.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Quick Look: 5 Best Topical Finasteride Products
Here’s a side-by-side comparison of our top recommended topical finasteride treatments. Read on for more details on each of these excellent products.
Product Price Finasteride % Customization Application Ulo $64+/month 0.005% – 0.2% High Solution Strut Health $59 0.1-0.25% Medium Solution/Gel Happy Head $59-99/month 0.3% Medium Liposomal Gel Roman $50/month 0.3% Low Spray Hims $35+/month 0.3% Low Spray Topical Finasteride: What It Is & Why It’s Better
First, we will briefly explore the science behind topical finasteride, including its mechanism of action in treating hair loss and the advantages of topical formulations.
What Is Topical Finasteride?
Topical finasteride is a clinically proven treatment for androgenic alopecia (AGA), a type of hair loss that affects more than 50% of men by the age of 50.[1]Jang, W.S., Son, I.P., Yeo, I.K., Park, K.Y., Li, K., Kim, B.J., Seo, S.J., Kim, M.N., Hong, C.K. (2013). The Annual Changes of Clinical Manifestation of Androgenetic Alopecia Clinic in Korean Males … Continue reading AGA is caused by dihydrotestosterone (DHT), a testosterone-derived hormone that damages hair follicles, causing them to shrink and produce thinner hair. Finasteride reduces DHT levels by inhibiting the enzyme 5-alpha-reductase, protecting the hair follicles to stop hair loss and encourage new growth.[2]Duran, R, C-D., Martinez-Ledesma, E., Garcia-Garcia, M., Gauzin, D.B., Sarro-Ramirez, A., Gonzalez-Carillo, C., Rodriguez-Sardin, D., Fuentes, A., Cardenas-Lopez. (2024). The Biology and Genomics of … Continue reading
While orally administered finasteride has been in use since 1997, topical formulations are a relatively new development. When applied to the scalp, finasteride provides targeted treatment with a reduced risk of systemic side effects associated with oral preparations. Topical finasteride is shown to reduce DHT levels on the scalp for greater hair count and density with comparable effectiveness to oral finasteride.[3]Lee, S.W, Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading
Numerous studies confirm that topical finasteride slows and reverses hair loss by boosting hair thickness, density, and growth cycles. These results are enhanced when combined with other topical treatments like minoxidil. Due to minimal systemic absorption and attendant side effects, topical formulas are considered a safer choice for prolonged use. Overall, topical finasteride is prized among clinicians and patients as a safe, effective, and convenient solution to pattern hair loss.[4]Lee, S.W, Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading,[5]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray … Continue reading
Why Choose Topical Over Oral?
Because topical finasteride is applied directly to the scalp, it offers localized therapy and avoids the side effects associated with oral finasteride’s systemic exposure. Here is a brief overview of this and other primary advantages of topical formulas:
- Better Safety Profile: Oral finasteride is absorbed into the bloodstream and can have global side effects like sexual dysfunction, hormone disruption, and mood swings. In contrast, topical formulas target the scalp directly and minimally affect plasma levels. Topicals are consistently shown to have fewer systemic side effects. If present, side effects tend to be mild and temporary, such as scalp irritation.
- Individualized Dosing: While the oral finasteride dose is a standardized 1 mg, topical formulas enable tailored dosages and greater precision through potency titration. This is especially important given the dose-dependent outcomes of finasteride treatment. The finasteride concentration in topicals can range from a maintenance dose of 0.005% to a more aggressive protocol of 2.5%.
- Greater Customization: Topical formulas often combine other proven treatments, such as tretinoin, minoxidil, and caffeine. Studies demonstrate that finasteride-minoxidil combinations deliver superior results compared to single-agent treatments. Plus, the range of available delivery methods (e.g., solutions, sprays, gels) can improve convenience, satisfaction, and adherence.
- Long-Term Use: With a better safety profile and lower systemic impact, topical finasteride is more suitable for prolonged use, which is often necessary to maintain the benefits of hair loss treatment.
In short, research data indicate that topical finasteride yields comparable results with a more favorable safety profile than oral finasteride, making it a better option for long-term treatment. Further benefits of topical formulations include greater dose precision, synergistic adjuvants like minoxidil, and a range of delivery methods to support compliance.[6]Lee, S.W, Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading,[7]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray … Continue reading
Evaluation Criteria: What Makes the Best?
When selecting the best topical finasteride products, we adhered to the following evaluation criteria, which are thoroughly supported by clinical findings and customer feedback.[8]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray … Continue reading,[9]Suchonwanit, P., Iamsumang, W., Leerunyakul, K. (2020). Topical finasteride for the treatment of male androgenetic alopecia and female pattern hair loss: a review of the current literature. Journal … Continue reading
✓ Effectiveness
We assessed scientific data, DHT decrease rates, and real-world testimonies of hair count increases. The top products deliver measurable results in 3-6 months with continuous improvements in hair density throughout the first year and beyond.
✓ Safety
Because finasteride is a prescription-only treatment, we verified that providers adhere to all safety regulations, including proper prescribing and monitoring protocols. Leading suppliers also provide full ingredient transparency to help assess potential side effects.
✓ Customization
Premium brands offer treatment customization options tailored to patients’ needs and preferences, maximizing outcomes beyond those achieved with traditional, standardized approaches. These include concentration adjustments, synergistic add-on ingredients such as minoxidil, and various delivery methods.
✓ Convenience
Successful treatment outcomes depend on long-term compliance and a positive user experience. Factors such as ease of application, clear instructions, manageable dose frequency, and responsive support help ensure uninterrupted treatment.
✓ Value
The best providers balance superior quality with transparent and affordable pricing models that cover doctor consultations, prescriptions, and follow-up care. These are especially important concerns in the telehealth landscape, where many subpar platforms employ opaque billing practices to stack on hidden fees and unexpected charges.
How to Choose the Right Product for Your Needs
When choosing the product that best meets your hair loss treatment needs, consider the extent of your hair loss and treatment aims, such as basic upkeep or intensive restoration. Also account for your preferred delivery method (i.e., gel, solution, or spray), desired add-ons, medical support requirements, and budget.
Reviewing the Best Topical Finasteride Products
With the above key factors in mind, let’s turn now to our top recommended options that each offer unique advantages sure to satisfy.
#1: Ulo Finasteride + Minoxidil Plus Rx
Pros: Cons: ✓ Clinically-proven combination therapy ✗ Prescription products are only available in the USA ✓ True personalization with custom concentrations and medical consultations ✓ PhD-scientist formulated ✓ Ongoing medical monitoring and a user-friendly platform ✓ Quality-tested ingredients free of irritants Ulo’s combination formula of high-strength finasteride with prescription-grade minoxidil takes the top spot, reflecting the platform’s commitment to providing safe, scientifically proven hair loss treatments tailored to meet individual needs. Every Ulo product is developed by a team of PhD scientists who deliver innovative interventions in hair restoration.
Their advanced finasteride and minoxidil solution blocks DHT and stimulates new growth in a targeted dual treatment that is proven superior to monotherapies in clinical trials. Ulo’s finasteride formulas are uniquely customizable and available in different concentrations (0.005% for maintenance and 0.2% for stronger protocols), with a series of synergistic add-ons like caffeine to boost circulation, tretinoin for better absorption, and melatonin for antioxidant protection.
The deluxe treatment begins at $64 per month, which includes an onboarding medical consultation to assess each client’s needs, a physician-crafted treatment plan, and continued dermatologist oversight. All Ulo products are strictly sourced from pharmacies that adhere to safety and quality regulations. The clinic’s licensed dermatologists provide customized treatment regimens with free home prescription deliveries and ongoing support.
Bottom Line: Ulo’s individualized hybrid treatment is the ultimate in advanced hair restoration technology. With precise formulation and continuous medical supervision, this tailored solution delivers exceptional results for a low monthly subscription fee starting at $64. Ulo’s guiding dedication to safety and efficacy makes this full-service platform the best option for those who are serious about combating hair loss.
#2 Strut Finasteride Hair Loss Formula
Pros: Cons: ✓ Custom-compounded topical finasteride formulas (with optional add-ons) ✗ Not available internationally, and currently cannot ship to Arkansas ✓ Online consult and prescriptions from U.S. board-certified physicians ✗ Auto-refills by default; some reviews report difficulty canceling and customer service gaps ✓ Multiple formulation options (gel or solution; adjustable finasteride strength) ✓ Convenient telehealth model with free, discreet shipping in most U.S. states. Strut Health is a U.S.-based telehealth provider offering customized topical finasteride for AGA. After an online questionnaire and photo-based evaluation, a board-certified clinician designs a compounded formula that may contain finasteride alone or in combination with other actives like minoxidil, tretinoin, fluocinolone, or biotin. The company’s finasteride hair-loss formulas typically use 0.1-0.25% topical finasteride, with optional minoxidil concentrations up to 7.5% and tretinoin in a gel or solution base, giving flexibility to tailor potency and tolerability.
Our in-house lab testing showed that Strut Health’s finasteride product contained the correct concentrations of ingredients (finasteride, minoxidil, and tretinoin), indicating that they can be trusted to provide a quality product.
However, there are trade-offs. Strut does not accept insurance, and pricing for topical finasteride (often around the mid-range of the telehealth market) may add up for long-term use. The company currently ships to nearly all U.S. states but cannot ship to Arkansas, and there is no international service. Public reviews highlight convenience for many users but also recurring concerns about auto-refills and difficulty canceling, as well as frustration with customer support responsiveness and refund policies.
Bottom Line: Strut Health’s topical finasteride offers a flexible, telehealth-delivered alternative for patients who want DHT-targeted treatment without taking a daily pill, with customizable formulas that can include or exclude minoxidil and other actives. Its convenience, physician oversight, and compounding flexibility are strong advantages, but subscription-based pricing and mixed customer service reviews may give more budget-conscious patients pause.
#3 Happy Head Topical Super Solution: Finasteride, Minoxidil & Liposomal
Pros: Cons: ✓ Medical oversight by board-certified dermatologists ✗ Price rises from $59 to $99 after the first six months ✓ Higher concentrations of 0.3% finasteride + 8% minoxidil ✗ Higher concentrations may increase side effect risk ✓ Made with a delayed-release liposomal base ✗ Gel formula cannot be customized ✓ User-friendly platform and follow-up care ✗ May irritate sensitive skin ✓ Affiliate programs available ✗ Possible shipping and customer service concerns Our next selection is from Happy Head, known for unique formulations and dermatologist oversight. The Topical Super Solution features a more concentrated combination of 0.3% finasteride with 8% minoxidil for targeted hair restoration.
Available in a mess-free pump dispenser, this gel is made with the brand’s proprietary liposomal delivery base that boosts scalp penetration with timed medication release and once-daily application. This helps minimize bloodstream absorption, reducing potential side effects, and is a notable safety feature for higher finasteride doses. That said, some ingredients in the carrier base should not be used on sensitive skin. Other products from the brand are more customizable and can be adapted to sensitive skin.
The starting price of Happy Head’s treatment is competitive at $59 for a monthly subscription that covers dermatologist oversight and prescription deliveries. However, the price increases to $99 after the first six months. Some reviews cite a lack of transparency regarding potential side effects, as well as intermittent shipping delays and inconsistent customer service.
Bottom Line: Happy Head’s potent dual therapy is a useful option for patients with intermediate to severe hair loss and normal skin. The delayed-release gel base increases medication absorption for more targeted treatment while decreasing systemic side effects and requires application only once a day. Advantages include dermatologist support and attractive pricing, but patients should be aware of reviews citing shipping and customer service issues.
#4: Roman (Ro) Hair Solution Rx
Pros: Cons: ✓ Transparent and competitive price of $50 per month ✗ Contains potential irritants not suitable for sensitive skin ✓ High finasteride concentration (0.3%) for aggressive treatment ✗ High fixed concentration with a greater risk of side effects ✓ Includes minoxidil and tretinoin for synergistic effects ✗ Limited customization options ✓ Convenient once-daily application ✗ Standard delivery system lacks advanced features ✓ Established telehealth provider with medical supervision Roman offers a simple topical finasteride formula designed for convenient, once-daily use and focused intervention. The spray contains three active ingredients: high-dose 0.3% finasteride with 6% minoxidil and tretinoin to enhance absorption. This product delivers a potent dose of finasteride on par with premium rivals through a no-frills spray system.
While tretinoin may improve minoxidil absorption, Roman’s spray has some drawbacks for users with sensitive skin or systemic side effect concerns. The 0.3% finasteride concentration cannot be tailored to individual response, and the standard delivery system does not have delayed-release technology to offset the risk of systemic absorption as seen in Happy Head’s liposomal gel. Inactive ingredients like ethyl alcohol and propylene glycol are also known irritants.
Roman is an accessible option for many users. The transparent and fixed monthly fee of $50 covers doctor consultations, prescription deliveries, and unlimited follow-ups. However, users with sensitive skin or more complex treatment needs may find Roman’s standardized and less gentle formulation to be unsuitable for long-term compliance.
Bottom Line: Roman offers a potent and affordable dual finasteride therapy with telehealth support features. Although it is not customizable like leading alternatives, this spray is a convenient choice for individuals with moderate to severe hair loss who can tolerate high dosages and potential irritants.
#5: Hims Topical Finasteride & Minoxidil Spray
Pros: Cons: ✓ Lowest starting rate of $35+/month ✗ Standardized approach that lacks customization ✓ Well-known brand with recognized platform ✗ Minimal ongoing medical support ✓ Simple ordering process and rapid shipping ✗ Contains irritants not suitable for sensitive skin and fixed high dose ✓ No lengthy consultations required ✗ Basic telehealth features without specialist care ✓ High-dose dual treatment ✗ Concerns about the price transparency of subscription tiers Hims is a well-known brand that offers topical finasteride through its streamlined telemedicine platform. Their fixed-dose spray of 0.3% finasteride and 6% minoxidil is a convenient and affordable option without the customization features of premium competitors, appealing to those who value simplicity over tailored care.
Like similar high-dose sprays, this standardized formula from Hims is not suitable for patients with sensitive skin, complex treatment needs, or those requiring a lower maintenance dose.
This provider’s appeal is its wide accessibility. With low prices starting at just $35 per month, quick doctor consultations, and shipping in all 50 states, Hims is a gateway brand for many new hair loss patients. However, this entry-level approach lacks the treatment personalization and close medical supervision of established specialty providers.
Bottom Line: Hims’ topical finasteride spray is an affordable and easily accessible option backed by a recognized brand name. Although Hims fails to offer the precision treatment and close medical oversight of premium alternatives, it is a mainstay for patients who prioritize low costs and convenience.
Final Verdict: Our Topical Finasteride Picks
After a comprehensive evaluation, Ulo emerges as our top choice for topical finasteride in 2026. Their genuine customization, science-backed formulations, and ongoing dermatologist supervision set the gold standard for personalized hair restoration.
While competitors offer standardized protocols or unproven technologies, Ulo delivers verified results through precision medicine tailored to individual needs. For patients seeking the highest quality care, Ulo’s scientific rigor and safety-first approach make it the clear winner.
What to Expect: Timeline and Results
Realistic treatment expectations facilitate long-term compliance and successful outcomes. Medical research has established the following evidence-based benchmarks of expected improvements with topical finasteride use as prescribed to treat androgenetic alopecia.
Clinical Timeline for Topical Finasteride Results
Timeframe What to Expect Clinical Evidence 3 Months Terminal hair count starts to improve; increases in total hair count may not yet be observable [10]B. Piraccini et al. (2021). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European … Continue reading,[11]Cheng Zhou et al. (2025). Efficacy and safety of topical finasteride spray solution in the treatment of Chinese men with androgenetic alopecia: A phase III, multicenter, randomized, double-blind, … Continue reading 6 Months Noteworthy increases in hair counts (total and terminal); visible increase in vertex hair growth [12]B. Piraccini et al. (2021). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European … Continue reading,[13]Cheng Zhou et al. (2025). Efficacy and safety of topical finasteride spray solution in the treatment of Chinese men with androgenetic alopecia: A phase III, multicenter, randomized, double-blind, … Continue reading,[14]A. Rossi et al. (2020). Efficacy of Topical Finasteride 0.5% vs 17α-Estradiol 0.05% in the Treatment of Postmenopausal Female Pattern Hair Loss: A Retrospective, Single-Blind Study of 119 … Continue reading 12-18 Months Maximum improvement is typically observed; continuous improvements in hair coverage and density [15]A. Rossi et al. (2020). Efficacy of Topical Finasteride 0.5% vs 17α-Estradiol 0.05% in the Treatment of Postmenopausal Female Pattern Hair Loss: A Retrospective, Single-Blind Study of 119 … Continue reading - Early Treatment Phase (Months 1-3): During early treatment, visible changes are minimal, which may be disappointing for new patients. The therapy begins to suppress scalp DHT levels, creating shifts that are not yet cosmetically evident. Studies show that improvements in terminal hair count begin around 3 months, though total hair count is not yet typically impacted.
In the first 6-8 weeks, some experience a temporary increase in shedding as finasteride prompts new growth cycles to start. This is expected and is an encouraging indication that the treatment is taking effect.
- Visible Improvement Phase (Months 3-6): Visible results begin to appear during this period. Studies show notable increases in hair counts and density by 6 months. Patients tend to note particular improvements in the crown or vertex region.
- Optimization Phase (Months 6-18): The most significant benefits occur during this phase. Research indicates that maximum improvements arise around 12 months of continued treatment, as hair density and new growth quality increase.
- Long-Term Maintenance: According to clinical data, benefits stabilize around 18-24 months, and treatment transitions from active regrowth to maintenance. Because treatment cessation can lead to the gradual return of hair loss within 6-12 months, continued daily compliance is essential to maintain long-term therapeutic outcomes.
Topical Delivery Formats
There are several different topical finasteride delivery formats that each offer unique advantages to suit individual needs and preferences, as follows:
- Solutions are liquid formulas typically applied using a dropper for targeted administration on desired scalp areas. These lightweight preparations are valued for their excellent absorption and residue-free finish. Because solutions may contain irritating excipients such as propylene glycol, sensitive patients should opt for gentle blends like Ulo’s prescription topicals.
- Sprays are a lighter, more fluid type of solution applied using an aerosol or pump. This form enables misting over larger or difficult-to-reach scalp areas. However, sprays are not as precise as droppers, as the liquid may run off and fail to effectively reach the hair follicles.
- Gels offer a more controlled application with a thicker consistency than solutions. They often include advanced delivery bases such as liposome carriers with delayed-release technology, designed to enhance penetration and reduce systemic absorption. However, customization options are typically limited due to compounding practices. Additionally, gel formulas can leave a sticky residue that some users find undesirable.
- Foams may be beneficial for patients with sensitive skin, as they dry quickly and generally lack harsh additives. However, their capacity for targeted application is limited compared to other formulations.
- Creams and lotions are appropriate for sensitive or dry scalps, featuring thick, emollient-laden bases with moisturizing benefits. Lotions are generally faster-absorbing, more spreadable, and lighter than creams.
The best formulation choice depends on hair and skin type, lifestyle, and personal preferences. Patients are encouraged to consult a doctor to determine the ideal format for their individual needs.
Safety and Side Effects
Topical finasteride is noted for its favorable safety profile, especially in contrast to oral formulations; however, understanding potential risks and proper monitoring practices is recommended.
- Common Mild Side Effects: The most common side effects are transient and mild scalp irritation or dryness. Though these reactions often resolve on their own, they also respond well to dose adjustment or calming adjunctive treatments. Patients with sensitive skin should avoid formulas containing irritants like propylene glycol or parabens.[16]Lee, S.W, Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading,[17]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray … Continue reading
- When to Contact Your Doctor: More serious side effects or reactions are rare but possible and require prompt medical intervention. These include severe scalp irritation, allergic reactions (blistering, swelling, and redness), and systemic effects like mood changes, sexual dysfunction, or gynecomastia.[18]Suchonwanit, P., Iamsumang, W., Leerunyakul, K. (2020). Topical finasteride for the treatment of male androgenetic alopecia and female pattern hair loss: a review of the current literature. Journal … Continue reading,[19]Cheng Zhou et al. (2025). Efficacy and safety of topical finasteride spray solution in the treatment of Chinese men with androgenetic alopecia: A phase III, multicenter, randomized, double-blind, … Continue reading
- Topical vs. Oral Finasteride Safety: Clinical data show that topical finasteride has a significantly lower risk of sexual side effects, typically less than 2% compared to 8-15% with oral formulations. This is attributed to the targeted delivery of topical preparations that limit systemic exposure while providing localized scalp treatment.[20]Mysore, V. (2012). Finasteride and sexual side effects. Indian Dermatology Online Journal, 3(1), 62. https://doi.org/10.4103/2229-5178.93496.
- Who Should Avoid Topical Finasteride: Topical finasteride is not suitable for women who are pregnant or nursing due to the risk of birth defects or infant harm. Additionally, individuals with known sensitivity, scalp infections, or certain autoimmune diseases may be ineligible for use. Consult with a doctor before beginning treatment.
Closing Thoughts
Topical finasteride is an important development in hair loss therapeutics, providing comparable results with a superior safety profile to oral medications. In today’s market, Ulo’s individualized, clinically rigorous approach sets a benchmark for comprehensive care, outperforming competitors primarily focused on budget or convenience factors.
Successful treatment with topical finasteride requires patience and persistence. Advances in hair restoration technology suggest that tailored medicine, rather than standardized approaches, represents the future. Ready to begin your own journey toward hair restoration?
Visit Ulo today to see how customized topical finasteride can enhance your results.
Frequently Asked Questions
Do I need a prescription for topical finasteride?
Yes, topical finasteride is a prescription-only medication in the USA. This ensures proper diagnosis, medical supervision, dosage, and quality control. Reputable telehealth providers like Ulo make the prescription process quick and easy via video consultations with licensed doctors specialized in hair restoration therapies. These elite platforms also offer ongoing medical supervision to monitor side effects and adjust treatment for optimization.
Avoid online platforms that market finasteride products without a prescription or those that fail to perform a proper medical consultation before prescribing treatment. Reputable platforms employ board-certified doctors licensed to practice in your state of residence with credentials clearly displayed or offered upon request. Sourcing topical finasteride products from unauthorized vendors without a prescription risks hazardous side effects and ineffectiveness.
Can topical finasteride be used with other hair loss treatments?
Yes, topical finasteride can be beneficially combined with treatments like minoxidil, a popular additive in many available products. These formulas often pair finasteride with additional synergistic ingredients such as antioxidants, vasodilators, antifungals, and absorption enhancers.
Topical finasteride can also be safely used in conjunction with most non-prescription hair products, hair transplants, and low-level laser treatments. However, it should not be combined with oral DHT blockers (i.e., oral finasteride or dutasteride) due to the risk of systemic side effects. Medical approval is necessary before starting new treatments.
When will I see results with topical finasteride?
Most patients report stabilization of hair loss after 3-4 months, with notable regrowth in 6-12 months. Shedding within the first 6-8 weeks of starting treatment is considered normal and indicative of the treatment’s efficacy. Because DHT suppression takes time to significantly impact hair growth cycles, consistency and patience are essential for optimal treatment outcomes.
Treatment outcomes can vary between individuals. Consult a doctor to discuss influencing factors and form realistic treatment expectations. This grounded approach facilitates better long-term adherence and more desirable results.
Is topical finasteride suitable for women?
Although the FDA has not yet authorized topical finasteride to treat AGA in women, it is occasionally prescribed for off-label use in postmenopausal patients with female pattern hair loss (FPHL). Because topical finasteride risks fewer hormonal adverse effects than oral finasteride, it is considered a safer option in these cases.
However, women of reproductive age are often recommended to completely avoid finasteride due to the risk of severe birth defects from exposure while pregnant. Non-pregnant women who may plan for pregnancy in the future should abstain from finasteride use unless under strict medical supervision with suitable contraception established. Newer low-dose formulations designed for women show promise, although more study is needed to verify long-term viability.
Ultimately, women considering finasteride therapy should speak with an experienced clinician to go over their medical history, weigh the benefits and risks, and review alternative treatment options, such as FDA-approved topical minoxidil.
What is the cost of topical finasteride without insurance?
The average monthly cost of topical finasteride without insurance can vary from $35 to $90, depending on the source, formulation, and services rendered. Direct-pay telehealth providers tend to offer lower rates than conventional in-person dermatology clinics with combined pharmacy fees. Bespoke formulas with advanced excipients can command higher prices. Patients should account for continued costs to budget for long-term treatment, which is often required for beneficial outcomes.
What will happen if I stop using topical finasteride?
If regular topical finasteride treatment is discontinued, hair loss is expected to return gradually within 2-3 months of cessation. Most patients report a rebound to their baseline hair loss level within 6-12 months. Because this regression is gradual, patients have some flexibility regarding therapy breaks or medication transitions if needed.
Does topical finasteride have any drug interactions?
Compared to oral finasteride, topical formulations have few contraindications due to lower systemic absorption rates. For your safety, inform your prescribing doctor of all current medications, especially blood thinners and other hormonal drugs. While topical finasteride is often safely combined with common medications, there may be exclusions.
How do I store topical finasteride?
Topical finasteride should be kept at room temperature out of direct light, moisture, and heat. With proper storage, most products maintain stability for 12-24 months. While refrigeration isn’t required, it may boost shelf life in temperate climates. Be sure to check expiration dates and safely dispose of expired products.
Can I travel while on topical finasteride?
Yes, prescribed topical finasteride may be stored in carry-on luggage during domestic travel. When traveling internationally, it is recommended to bring the original prescription and label. Be mindful of TSA restrictions on liquid volumes over 3.4 oz.
How is topical finasteride applied?
Topical finasteride is applied directly to a clean and dry scalp with the product applicator. While specific instructions vary with delivery method, in general, the hair should be parted to expose the scalp and allow targeted application to the desired areas. Patients should consult product instructions for precise indications and dosing protocols. Avoid wetting hair for several hours after application, and thoroughly wash hands after use.
References[+]
References ↑1 Jang, W.S., Son, I.P., Yeo, I.K., Park, K.Y., Li, K., Kim, B.J., Seo, S.J., Kim, M.N., Hong, C.K. (2013). The Annual Changes of Clinical Manifestation of Androgenetic Alopecia Clinic in Korean Males and Females: A Outpatient-Based Study. Annals of Dermatology. 25(2). 181-188. Available at: https://doi.org/10.5021/ad.2013.25.2.181 ↑2 Duran, R, C-D., Martinez-Ledesma, E., Garcia-Garcia, M., Gauzin, D.B., Sarro-Ramirez, A., Gonzalez-Carillo, C., Rodriguez-Sardin, D., Fuentes, A., Cardenas-Lopez. (2024). The Biology and Genomics of Human Hair Follicles: A Focus on Androgenetic Alopecia. International Journal of Molecular Sciences. 25(5). 2542. Available at: https://doi.org/10.3390/ijms25052542 ↑3, ↑4, ↑6, ↑16 Lee, S.W, Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in Dermatology. 17(4). 457-463. Available at: PMID: 29601622 ↑5, ↑7, ↑17 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology and Venereology. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑8 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falques, M., Otero, R., Tamarit, M.L., Galvan, J., Tebbs, V., Massana, E. (2021). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology and Venereology. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑9, ↑18 Suchonwanit, P., Iamsumang, W., Leerunyakul, K. (2020). Topical finasteride for the treatment of male androgenetic alopecia and female pattern hair loss: a review of the current literature. Journal of Dermatological Treatment, 33(2), 643–648. Available at: https://doi.org/10.1080/09546634.2020.1782324 ↑10, ↑12 B. Piraccini et al. (2021). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. Journal of the European Academy of Dermatology and Venereology, 36: 286 – 294. https://doi.org/10.1111/jdv.17738 ↑11, ↑13 Cheng Zhou et al. (2025). Efficacy and safety of topical finasteride spray solution in the treatment of Chinese men with androgenetic alopecia: A phase III, multicenter, randomized, double-blind, placebo-controlled study. Chinese medical journal. Available at: https://doi.org/10.1097/CM9.0000000000003495 ↑14, ↑15 A. Rossi et al. (2020). Efficacy of Topical Finasteride 0.5% vs 17α-Estradiol 0.05% in the Treatment of Postmenopausal Female Pattern Hair Loss: A Retrospective, Single-Blind Study of 119 Patients. Dermatology Practical & Conceptual, 10 (2020). https://doi.org/10.5826/dpc.1002a39 ↑19 Cheng Zhou et al. (2025). Efficacy and safety of topical finasteride spray solution in the treatment of Chinese men with androgenetic alopecia: A phase III, multicenter, randomized, double-blind, placebo-controlled study. Chinese medical journal. Available at: https://doi.org/10.1097/CM9.0000000000003495 ↑20 Mysore, V. (2012). Finasteride and sexual side effects. Indian Dermatology Online Journal, 3(1), 62. https://doi.org/10.4103/2229-5178.93496. Oral finasteride remains the most clinically validated medication for androgenic alopecia (AGA). Over 20 years of trials and follow-up data show consistent, predictable outcomes in slowing miniaturization and restoring density, particularly in the vertex and mid-scalp.
Yet, despite this wealth of data, online forums and “miracle” before-and-after photos often distort expectations. These transformations represent the upper end of the response curve, while most users experience more moderate, steady results that unfold over months to years.
This article explores what realistic regrowth looks like with oral finasteride, how to interpret finasteride before and after photos critically, and what to expect when used correctly and consistently.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What is Oral Finasteride?
Oral finasteride is a type II 5ɑ-reductase inhibitor that reduces the conversion of testosterone to dihydrotestosterone (DHT), the key androgen responsible for hair follicle miniaturization in AGA.
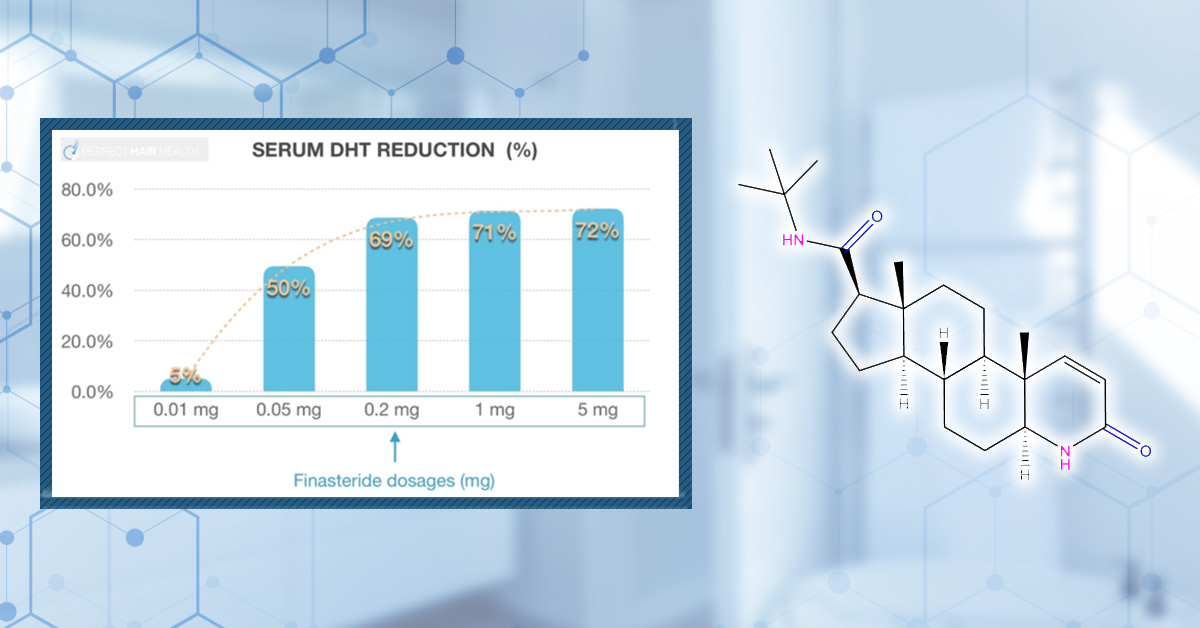
Originally approved by the FDA at a dose of 1 mg for male pattern hair loss, oral finasteride has since become widely known as the gold-standard treatment for hair loss. Its primary mechanism is systemic DHT suppression, lowering circulating levels by roughly 60-70% and scalp DHT by up to 65%.[1]Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men … Continue reading
Mechanism of Action
As mentioned above, finasteride is a competitive inhibitor of the 5ɑ-reductase enzyme, specifically targeting the type II (and to a lesser extent, type III) isoenzymes.[2]Zito, P.M., Bistas, K.G., Patel, P., Syed, K. (2024). Finasteride. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513329/
Treatment with finasteride leads to a significant decrease in both scalp and serum DHT concentrations. This effect is dose-dependent and has been observed as early as 28 days after starting treatment.[3]Dallob, A.L., Sadick, N.S., Unger, W., Lipert, S., Geissler, L.A., Gregoire, S.L., Nguyen, H.H., Moore, E.C., Tanaka, W.K. (1994). The effect of finasteride, a 5 alpha-reductase inhibitor, on scalp … Continue reading Lower DHT levels in the scalp prevent androgen-dependent miniaturization of hair follicles, promoting the maintenance and improvement of hair follicle size and number in the anagen (growth) phase.
Unlike topical formulations, oral finasteride exerts a systemic effect. More comprehensive, but also more likely to cause side effects in sensitive individuals.
Adverse Effects
The most commonly reported adverse effects of oral finasteride are sexual in nature, including decreased libido, erectile dysfunction, and reduced ejaculatory volume. These occur in a range of users depending on the study and determination of what a side effect is (<1% to 25%).[4]Traish, A.M., Hassani, J., Guay, A.T., Zitzmann, M., Hansen, M.L. (2011). Adverse side effects of 5ɑ-reductase inhibitors therapy: persistent diminished libido and erectile dysfunction and … Continue reading
Other potential side effects include psychological and neuropsychiatric effects and post-finasteride syndrome (PFS). PFS refers to persistent sexual, neurological, and physical symptoms that continue after stopping finasteride. However, the existence and prevalence of PFS are debated, with some studies suggesting a possible genetic predisposition.[5]Gupta, A.K., Talukder, M., Keene, S.A., Bamimore, M.A. (2025). Is the safety of finasteride correlated with its route of administration: topical versus oral? A pharmacovigilance study with data from … Continue reading
You can read in more depth about the potential side effects in our oral finasteride ultimate guide.
Why Expectation Setting Matters
Finasteride is powerful and predictable, but even the best data can get lost when you see incredible results online. Online before-and-after posts tend to showcase outliers, individuals who happen to respond at the extreme high end of the efficacy curve.
Meanwhile, clinical averages show that:
- Most men stop or reverse their hair loss progression.
- Around 65-80% experience visible thickening after 12-24 months.
- Only a small subset achieves “complete restoration”.
Unrealistic expectations can lead to disappointment, early discontinuation, or cycling through treatments unnecessarily. Grounding expectations in clinical probabilities rather than forum anecdotes helps users stay consistent long enough to see results.
What’s Possible ≠ What’s Probable
While oral finasteride can produce impressive regrowth, most users experience gradual thickening and stabilization rather than dramatic restoration.
Treatment outcomes typically follow a distribution pattern:
- A minority are “hyper responders”, achieving rapid and substantial visible improvement.
- The majority notice moderate, progressive gains, such as fuller density or slowed shedding.
- A smaller subset experience arrest of hair loss but remain unsatisfied with new hair growth.[6]Dhurat, R., Mathapati, S. (2015). Response to Microneedling Treatment in Men with Androgenetic Alopecia Who Failed to Respond to Conventional Therapy. Indian Journal of Dermatology. 60(3). 260-263. … Continue reading
Factors influencing response:
- Age and duration of hair loss: Finasteride efficacy is higher in younger patients and those with less advanced disease at treatment initiation.[7]Camacho, F.M., Garcia-Hernandez, M.J., Fernandez-Crehuet, J.L. (2008). Value of hormonal levels in patients with male androgenetic alopecia treated with finasteride: better response in patients under … Continue reading Starting treatment at age 40 or higher is an independent risk factor for insufficient efficacy.[8]Yoshitake, T., Takeda, A., Ohki, K., Inoue, Y., Yamawaki, T., Otsuka, S., Akimoto, M., Nemoto, M., Shimakura, Y., Sato, A. (2015). Five-year efficacy of finasteride in 801 Japanese men with … Continue reading
- Consistency: Missing doses or cycling off can interrupt the stabilization of hair loss.
- Adjunct therapies: Combining finasteride with other therapies like topical minoxidil can improve results.[9]Hu, R., Xu, F., Sheng, Y., Qi, S., Han, Y., Miao, Y., Rui, W., Yang, Q. (2015). Combined treatment with oral finasteride and topical minoxidil in male androgenetic alopecia: a randomized and … Continue reading
- Genetics: Variations in the androgen receptor (AR) gene sensitivity affect responsiveness.[10]Keene, S., Goren A. (2011). Therapeutic hotline. Genetic variations in the androgen receptor gene and finasteride response in women with androgenetic alopecia mediated by epigenetics. Dermatologic … Continue reading
Strong Evidence = Predictable Results
Unlike many newer (and even potentially stronger acting treatments), oral finasteride benefits from decades of standardized data, making outcomes highly predictable. A good example of this is comparing finasteride to dutasteride. Finasteride is extremely well-studied; therefore, we can usually predict the type of results we will see. Dutasteride, on the other hand, has studies showing it can outperform even the likes of finasteride in terms of hair regrowth. But the number of clinical studies is lacking, meaning that it is more difficult to predict a consistent outcome.
Figure 1: For dutasteride, a wide, shallow curve represents a much wider spread of outcomes. For finasteride, a narrow bell curve shows predictable outcomes.
This predictability helps clinicians set clear, realistic expectations and provides users with confidence that consistency will lead to steady results.
Real Case Studies: Oral Finasteride Before and After
The examples below help see what’s possible and do not represent what every user should expect. They are purely anecdotal and not independently verified by us or any qualified third party.
Case Summary #1: Male (23), 3 Months on Oral Finasteride 1 mg (then every other day) + Topical Minoxidil Foam (twice daily)
Source: u/Prize-Leg2544 via r/tressless.
This 23-year-old male took oral finasteride 1 mg daily for 2 months before titrating down to 1 mg every other day due to side effects. He also applied topical minoxidil foam twice daily, initially to the whole scalp but later exclusively to the hairline. He noted an initial shedding phase and early side effects on finasteride in the first month of treatment. In the second month, he described visible thickening while still on 1 mg daily. By month 3, he reduced finasteride to every other day and continued with minoxidil to maintain the progress. He reported marked improvement in hairline and density over the 3 months.
The user also described early transient effects, including testicular ache (first week), watery semen, heightened libido, and less intense orgasm, which resolved after dialing the finasteride down to every other day.
Case Summary #2: Male (25), 1-Year Course of Finasteride (1 mg) + Topical Minoxidil (1–2x Daily) + Monthly Dermarolling
Source: u/zacboring via r/malehairadvice.
This male Redditor started treatment at age 24 after severe crown thinning since he was about 18. His one-year course consisted of finasteride 1 mg daily (generic), topical liquid minoxidil 1–2/day, and dermarolling roughly monthly (post-shower, before nightly minoxidil). He supplemented with vitamin D and occasional biotin, and he regularly used argan oil after showers to limit flaking.
The patient noted a strong early response, but felt his results peaked roughly halfway into treatment. He stopped minoxidil for 1 month and missed some doses in the months leading up to the post. His photos indicated a significant improvement in density compared to baseline, particularly at the hairline and the top. He noted more coverage at the crown but felt it was still noticeably weaker than desired. The patient reported no sexual side effects or skin issues.Case Summary #3: Male (24), 6 Months on Oral Finasteride 1 mg Daily + Oral Minoxidil 2.5 mg Daily
Source: u/LongjumpingDurian429 via r/tressless.
This 24-year-old male’s intervention consisted of oral finasteride 1 mg once daily and oral minoxidil 2.5 mg once daily. The user noted the emergence of small baby hairs at the hairline and temples at month 2 of treatment. By months 4-5, he reported noticeable thickening, with longer eyelashes and denser eyebrows. By month 6, the user showed clear improvement, as evidenced by his wet-hair photos and day-to-day density. He reported visible density gains at the hairline with better coverage under bright light, with his crown improved from baseline, though not fully closed. He reported no side effects, apart from brief mild erectile difficulty early on that resolved.
Case Summary #4: Male (25), 3 Months on Oral Finasteride 1 mg Daily + Oral Minoxidil 2.5 mg Daily (added at month 2) + Ketoconazole Shampoo 3x/week + Microneedling 1.5 mm Weekly
Source: u/Buchaill-Bo via r/tressless.
This 24-year-old male’s intervention was oral finasteride 1 mg daily for 3 months, oral minoxidil 2.5 mg daily (added 2 weeks before the 3-month update), ketoconazole shampoo (Nizoral) about three times weekly, and dermaroller 1.5 mm once weekly over the whole scalp.
The user described gradual thickening over the 3-month finasteride course, with minoxidil introduced at month 2.5. He reported a noticeable improvement in density at the hairline and mid-scalp compared with baseline. He described a possible mild libido reduction on finasteride and thinning of the eyebrows thinned on finasteride, which appeared fuller after starting minoxidil. He reported no other significant effects.
Case Summary #5: Male (38), 7 Months on Oral Finasteride + Topical Finasteride + Topical Minoxidil + Weekly Microneedling
Source: u/Moonrocks321 via r/tressless.
This 38-year-old male started the current treatment with oral finasteride 1 mg daily and topical minoxidil (strength/frequency not specified). He added topical finasteride (concentration not specified) and weekly microneedling at 0.25 mm in month 5. The patient reported a slight but noticeable improvement vs. baseline at month 5 and, at month 7, likewise described his progress as modest. In the 7 months, he achieved stabilization with modest density improvement, most visible anteriorly, with the crown remaining limited. He reported no side effects from finasteride and minoxidil.
Case Summary #6: Male (28), 10 Years on Oral Finasteride + 4–5 Months on Oral Minoxidil
Source: u/DatBronzeGuy via r/tressless.
This 28-year-old male reported on his 10-year treatment progress on oral finasteride 1 mg daily, before adding oral minoxidil 2.5 mg twice daily 4-5 months before the progress report. He confirmed no dermarolling or topical agent application. The patient reported that, between the ages of 18 and 27, he was on finasteride monotherapy, achieving significant regrowth and strong maintenance. Towards the end of that period, he reported subtle renewed thinning, prompting him to add oral minoxidil. Within 4 months thereafter, his hair density and thickness nearly doubled.
He reported good tolerance to finasteride with no noticeable side effects. He added that oral minoxidil caused mild generalized hypertrichosis (slightly darker chest/back hair, fuller beard, higher cheek/temple hairs), but was otherwise tolerated.
Case Summary #7: Male (28), 2–3 Months on Oral Finasteride 1 mg Monotherapy
Source: u/danish0001 via r/tressless.
This 28-year-old male’s intervention included oral finasteride 1 mg daily, weekly microneedling with dermastamp 1.0–1.5 mm, and supplementation with vitamin D3 + K2, zinc, folate, magnesium glycinate, and ketoconazole 2% shampoo. Additionally, he made lifestyle adjustments, including going to the gym 2x/week and starting a higher-protein diet.
He noted a mild shedding phase in the first month of treatment. By month 2, he described the first visible tiny regrowth after a buzz cut, by which point the shedding had reduced. Overall, the patient described early regrowth at the hairline and thinning zones. He reported no side effects on finasteride 1 mg daily, stating that libido and energy have remained unchanged or improved.
Case Summary #8: Male (unspecified age), 3 Years on Oral Finasteride 1 mg Daily + Topical Minoxidil
Source: u/AthleteAfraid7844 via r/tressless.
This male Redditor’s intervention over 3 years was oral finasteride 1 mg daily and topical minoxidil (strength not specified).
In the first year, he noted steady cosmetic improvement, followed by continued thickening and maintenance of gains over years 2-3. Overall, the user reported substantial density gain, with perceived improvement in the hairline and temples over time. He mentioned no significant side effects and confirmed his sex drive had remained unchanged throughout treatment. He noted that the minoxidil sometimes caused dandruff and that on finasteride, he required greater mental focus to push weight-lifting sets to failure.
Case Summary #9: Male (early 30s), 3.5 Months on Oral Finasteride 1.25 mg Daily
Source: u/Ill-Foundation2637 via r/tressless.
This male, in his early 30s, opted for a treatment consisting of oral finasteride monotherapy at 1.25 mg daily (5 mg tablets quartered). He described a mild daily shedding phase within the first month of treatment. By month 2.5, he reported his first visible thickening. At the 3.5-month progress report, the user achieved a fuller crown with reduced scalp show-through. He reported no side effects.
Case Summary #10: Male (25), 5.5 Months on Oral Finasteride 1.25 mg Daily + Oral Minoxidil 5 mg DailySource: u/Dantheghost012 via r/tressless.
This 25-year-old South Asian male’s intervention was oral finasteride 1.25 mg daily (5 mg tablets quartered) and oral minoxidil, starting at 2.5 mg daily, titrated to 5 mg after 3 months. He described a substantial shedding phase, particularly in the first 3 weeks of month 3. The following month, he reported progressive thickening and density gains with improved coverage. As of month 5.5, he is still improving and continuing both agents unchanged. Overall, he reported a marked increase in density, with a thicker texture, and the crown and hairline both looked fuller than baseline. He reported no side effects on either dose level for finasteride or oral minoxidil.
Probable Regrowth Timeline
Based on the available research (which you can take a look at with our research tables here), we have created a probable hair regrowth timeline.
Figure 2: Typical regrowth for oral finasteride 1 mg.
Months 0-3: Adjustment Phase
In the first three months, most users experience minimal visible change. Finasteride actively lowers scalp and serum DHT levels, initiating follicle recovery. Some users notice a temporary increase in shedding as older hairs are shed to make way for new growth, while others observe subtle stabilization of hair loss.
Months 3-6: Reduction in Shedding
By this stage, a clear reduction in hair shedding often becomes noticeable. Although visible thickening remains limited for many, the treatment is beginning to normalize hair cycling. Early responders may report small areas of improved density, particularly in the crown or vertex.
Months 6-12: Early Cosmetic Improvements
Between 6 and 12 months, most responders start to see visible regrowth. Hair density gradually improves, strands appear thicker, and areas of thinning begin to fill in. The crown region typically shows improvement sooner than the frontal hairline, which tends to respond more slowly. Continued adherence is essential during this phase to sustain progress.
Months 12-24: Peak Response Period
During the second year, responders generally achieve their maximum results. Hair density, coverage, and stabilization reach their highest levels, with further visible gains possible in some individuals. Those starting with more advanced thinning usually experience consolidation rather than full growth.
Months 24+: Maintenance and Long-Term Stability
After two years, most users reach a plateau in visible improvement. Continued daily use of finasteride is needed to preserve these gains, as stopping the medication often results in gradual hair loss returning to baseline within 6-12 months. Some people combine treatments at this point to achieve further outcomes.
For most users, oral finasteride provides progressive, steady improvements over time, with long-term commitment being the key to maintaining results.
Setting Realistic Expectations
It’s important to set realistic expectations when starting any treatment. Finasteride primarily stabilizes hair loss during the early months, with noticeable growth typically developing later in the treatment course. True cosmetic improvement usually unfolds gradually over 12 to 24 months rather than weeks, reflecting the natural pace of the hair growth cycle.
Clinical trials suggest that most men experience visible thickening and halting of progression rather than full restoration, effectively “turning back the clock” by two to five years in terms of density and coverage.[11]Kaufman, K.D., Olson, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride … Continue reading,[12]Price, V.H., Roberts, J.L., Hordinsky, M., Olsen, E.A., Savin, R., Bergfeld, W., Fiedler, V., Lucky, A., Whiting, D.A., Pappas, F., Culbertson, J., Kotey, P., Meehan, A., Waldstreicher, J. (2000). … Continue reading,[13]Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparative effectiveness of finasteride vs Serenoa repens in male androgenetic alopecia: a … Continue reading
Consistent photographic tracking using identical lighting and angles can help visualize these incremental gains. Long-term adherence is crucial, as discontinuation typically leads to renewed miniaturization and hair loss within several months of stopping treatment.
Practical Takeaways
- Oral finasteride remains the most effective DHT blocker for male AGA
- Decades of clinical data make results highly predictable and durable
- The safety profile is well-established, with sexual side effects uncommon and often reversible
- Combining treatments can improve outcomes, especially for advanced loss.
Final Thoughts
Oral finasteride remains the most extensively studied and clinically validated treatment for androgenic alopecia, offering predictable outcomes for most users through gradual thickening, stabilization of shedding, and preservation of existing hair rather than dramatic restoration. While newer options like oral minoxidil are gaining popularity, they remain off-label despite growing supportive evidence and should be used under medical supervision.
The key to achieving and maintaining meaningful results lies in consistency, patience, and realistic expectations, recognizing that hair regrowth is a slow, cumulative process measured over months and years. By grounding expectations in clinical evidence rather than anecdotal extremes, patients can make informed, balanced decisions and stay committed to a long-term plan that maximizes both safety and effectiveness.
References[+]
References ↑1 Caserini, M., Radicioni, M., Leuratti, C., Terragni, E., Iorizzo, M., Palmieri, R. (2016). Effects of a novel finasteride 0.25% topical solution on scalp and serum dihydrotestosterone in healthy men with androgenetic alopecia. International Journal of Clinical Pharmacology and Therapeutics. 54(1). 19-27. Available at: https://doi.org/10.5414/CP202467 ↑2 Zito, P.M., Bistas, K.G., Patel, P., Syed, K. (2024). Finasteride. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513329/ ↑3 Dallob, A.L., Sadick, N.S., Unger, W., Lipert, S., Geissler, L.A., Gregoire, S.L., Nguyen, H.H., Moore, E.C., Tanaka, W.K. (1994). The effect of finasteride, a 5 alpha-reductase inhibitor, on scalp skin testosterone and dihydrotestosterone concentrations in patients with male pattern baldness. Journal of Clinical Endocrinology and Metabolism. 79(3). 703-706. Available at: https://doi.org/10.1210/jcem.79.3.8077349 ↑4 Traish, A.M., Hassani, J., Guay, A.T., Zitzmann, M., Hansen, M.L. (2011). Adverse side effects of 5ɑ-reductase inhibitors therapy: persistent diminished libido and erectile dysfunction and depression in a subset of patients. The Journal of Sexual Medicine. 872-884. Available at: https://doi.org/10.1111/j.1743-6109.2010.02157.x ↑5 Gupta, A.K., Talukder, M., Keene, S.A., Bamimore, M.A. (2025). Is the safety of finasteride correlated with its route of administration: topical versus oral? A pharmacovigilance study with data from the United States Food and Drug Administration adverse event reporting system. International Journal of Dermatology. Available at: https://doi.org/10.1111/ijd.17957 ↑6 Dhurat, R., Mathapati, S. (2015). Response to Microneedling Treatment in Men with Androgenetic Alopecia Who Failed to Respond to Conventional Therapy. Indian Journal of Dermatology. 60(3). 260-263. Available at: https://doi.org/10.4103/0019-5154.156361 ↑7 Camacho, F.M., Garcia-Hernandez, M.J., Fernandez-Crehuet, J.L. (2008). Value of hormonal levels in patients with male androgenetic alopecia treated with finasteride: better response in patients under 26 years old. British Journal of Dermatology. 158(5). 1121-1124. Available at: https://doi.org/10.1111/j.1365-2133.2008.08509.x. ↑8 Yoshitake, T., Takeda, A., Ohki, K., Inoue, Y., Yamawaki, T., Otsuka, S., Akimoto, M., Nemoto, M., Shimakura, Y., Sato, A. (2015). Five-year efficacy of finasteride in 801 Japanese men with androgenetic alopecia. The Journal of Dermatology. 42(7). 735-738. Available at: https://doi.org/10.1111/1346-8138.12890 ↑9 Hu, R., Xu, F., Sheng, Y., Qi, S., Han, Y., Miao, Y., Rui, W., Yang, Q. (2015). Combined treatment with oral finasteride and topical minoxidil in male androgenetic alopecia: a randomized and comparative study in Chinese patients. Dermatologic Therapy. 28(5). 303-308. Available at: https://doi.org/10.1111/dth.12246 ↑10 Keene, S., Goren A. (2011). Therapeutic hotline. Genetic variations in the androgen receptor gene and finasteride response in women with androgenetic alopecia mediated by epigenetics. Dermatologic Therapy. 24(2). 296-300. Available at: https://doi.org/10.1111/j.1529-8019.2011.01407.x. ↑11 Kaufman, K.D., Olson, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride in the treatment of men with androgenetic alopecia. Finasteride Male Pattern Hair Loss Study Group. Journal of the American Academy of Dermatology. 39(4 Pt 1), 578-589. Available at: https://doi.org/10.1016/s0190-9622(98)70007-6 ↑12 Price, V.H., Roberts, J.L., Hordinsky, M., Olsen, E.A., Savin, R., Bergfeld, W., Fiedler, V., Lucky, A., Whiting, D.A., Pappas, F., Culbertson, J., Kotey, P., Meehan, A., Waldstreicher, J. (2000). Lack of efficacy of finasteride in postmenopausal women with androgentic alopecia. Journal of the American Academy of Dermatology. 43(5 Pt 1). 768-776. Available at: https://doi.org/10.1067/mjd.2000.107953 ↑13 Rossi, A., Mari, E., Scarno, M., Garelli, V., Maxia, C., Scali, E., Iorio, A., Carlesimo, M. (2012). Comparative effectiveness of finasteride vs Serenoa repens in male androgenetic alopecia: a two-year study. International Journal of Immunopathology and Pharmacology. 25(4). 1167-1173. Available at: https://doi.org/10.1177/039463201202500435 Millions worldwide experience hair loss, with androgenic alopecia (AGA) as the leading cause. Also known as male- or female-pattern hair loss, AGA becomes more prevalent with age. It affects a majority of men over the age of 50 and a substantial number of postmenopausal women. Hair thinning and balding due to AGA is not just a cosmetic concern but can significantly detract from self-esteem and overall quality of life.
Among popular hair restoration treatments, topical dutasteride is a breakthrough solution to the serious side effects and plateaued results many experience. This potent DHT blocker offers more targeted therapeutic benefits with minimal systemic exposure and reduced risk. However, not all topical dutasteride products meet quality standards, so specialized guidance is key to selecting the right formula.
The PhD-directed board of experts at Perfect Hair Health draws from the latest clinical data and verified customer feedback to deliver this comprehensive guide to the best topical dutasteride products available in 2026. Through our research collaboration with leading innovators in the field, including our partnership with Ulo, we provide unique insights into the most advanced hair restoration protocols.
Read on for our detailed analysis and evidence-based product recommendations.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Quick View: 8 Best Topical Dutasteride Products
Here’s a brief comparison of our most highly recommended topical dutasteride products. Continue reading for in-depth reviews.
Product Price Dutasteride % Delivery Method Key Feature Ulo $84+/month 0.02%-0.2% Solution PhD-developed extensive customization XYON Health $129+/month 2% Gel Patented controlled-release technology Strut Health $69/month Up to 0.1% Gel Alcohol-free and customizable Blends ~$45/month (90-day supply) 0.3% Gel Proprietary hydrating formula Happy Head $59-89+/month Up to 0.3% Solution Potency and customization Maximus $54.99 0.1% Gel Customizability, including the addition of antihistamines Miiskin $30-59 + Rx cost Variable, custom Solution No subscription required Musely $33/month 0.3% Solution Lowest cost option Science and Benefits of Topical Dutasteride
To begin, we will briefly explore the scientific background of topical dutasteride, including how it works to treat AGA and the distinct benefits of topical preparations.
Mechanism of Action
Topical dutasteride is valued as a breakthrough treatment for AGA due to its powerful and targeted effects on the molecular levels of hair loss. The root cause of AGA is the testosterone-derived hormone dihydrotestosterone (DHT), which adheres to hair follicles and leads to follicular degeneration and hair loss over time. Dutasteride is classified as a DHT blocker, reducing DHT levels on the scalp to combat hair loss.[1]R. Cuevas-Díaz Durán., Martinez-Ledesma, E., Garcia-Garcia, M., Gauzin, D.B., Sarro-Ramirez, A., Gonzalez-Carrillo, C., Rodriguez-Sardin, D., Fuentes, A., Cardenas-Lopez, A. (2024) The Biology and … Continue reading,[2]Dhurat, R., Sharma, A., Rudnicka, L., Kroumpouzos, G., Kassir, M., Galadari, H., Wollina, U., Lotti, T., Golubović, M., Binic, I., Grabbe, S., & Goldust, M. (2020). 5‐Alpha reductase … Continue reading
Among DHT-blocking treatments, dutasteride is considered the most potent due to its dual enzyme activity. Testosterone is converted into DHT through two forms of 5α-reductase enzymes: Type I is located in the skin and sebaceous glands, and Type II is in the hair follicles. Finasteride inhibits only Type II for about 70% DHT reduction, while dutasteride inhibits both types to elicit greater DHT suppression (up to 93% in the skin and 99% in the follicles).[3]Arif, T., Dorjay, K., Adil, M., & Sami, M. (2017). Dutasteride in Androgenetic Alopecia: An Update.. Current clinical pharmacology, 12 1, 31-35. Available at: … Continue reading
Dutasteride’s powerful DHT inhibition leads to greater therapeutic outcomes in hair restoration. Clinical findings indicate dutasteride’s superior effects on hair count and mass in comparison to finasteride in terms of both quantitative and temporal metrics. While finasteride’s benefits tend to plateau after about 12 months of treatment, dutasteride demonstrates continued improvements up to 12 months and beyond.[4]Harcha, W., Martínez, J., Tsai, T., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy … Continue reading
Topical vs. Oral Advantages
Studies show that topical dutasteride is a safer option than oral with comparable and often better treatment outcomes. When applied locally to the scalp, dutasteride delivers targeted treatment with limited systemic exposure and a reduced risk of serious side effects. If present, adverse reactions to topical dutasteride tend to be transient and mild, in contrast to oral dutasteride’s potential systemic side effects like sexual dysfunction and mood disorders.[5]Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 mg) in … Continue reading,[6]Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., & Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology, 240, 833 – 843. Available at: … Continue reading
This and other clinically proposed advantages of topical dutasteride are briefly summarized as follows:
- Safe and Targeted Treatment: Oral dutasteride has systemic exposure through the bloodstream and therefore runs the risk of more serious side effects. Meanwhile, topical treatment targets only the scalp, lowering the risk of severe adverse reactions. Minor irritation at the site of application may occur and tends to resolve quickly.
- Flexible Dosing: Topical formulations offer greater dose flexibility through modified concentrations, in contrast to the usually fixed doses of oral preparations. Common topical offerings range from a maintenance dose of 0.02% dutasteride to advanced therapies of 2%, allowing dose-dependent customization.
- Increased Personalization: Topicals can easily integrate synergistic treatments like vasodilators (minoxidil), absorption enhancers (tretinoin), and anti-inflammatories (melatonin) for combination therapies that outperform dutasteride alone. These add-ons are prescribed based on patients’ individual needs and treatment goals.
Plus, patients have a choice of different delivery methods (e.g., sprays, gels, solutions), which can offer greater convenience and adherence.
- Long-term Outlook: Topical use is considered safer for long-term treatment due to its reduced systemic effects. Because hair restoration treatments’ benefits build with time, their suitability for prolonged use is paramount.
Combined, these factors locate topical dutasteride as a more effective and flexible hair restoration intervention with a greater safety profile than its oral counterpart.[7]Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 mg) in … Continue reading,[8]Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., & Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology, 240, 833 – 843. Available at: … Continue reading,[9]Radhakrishnan, P., Mariyappan, G., Karthi, J., & Jaisumi, K. (2024). Formulating, Optimizing and Evaluation of Dutasteride Loaded Insitu-Emulgel for Androgenetic Alopecia. International Journal … Continue reading
Choosing the Best: What We Looked For
To help navigate the topical dutasteride market, we developed these quality benchmarks grounded in clinical expertise, scientific evidence, and patient feedback.
Clinical Efficacy
Quality dutasteride products demonstrate notable DHT reduction and improved hair growth within 3-6 months, with these effects continuing over 12 months or longer. The products recommended in this article were assessed based on documented DHT reductions, verified patient feedback, and peer-reviewed studies.
Safety Profile and Medical Oversight
Given the prescription status of topical dutasteride, we verified each telehealth provider’s legitimacy in terms of physician supervision, adherence to legal requirements, and adherence to sound safety protocols. These include proper diagnostic procedures before prescribing, as well as close follow-up care to monitor for side effects and adjust the treatment as needed for optimal outcomes. Trustworthy providers exclusively partner with licensed physicians specialized in hair restoration.
When it comes to quality assurance, suppliers must partner only with authorized domestic pharmacies to source their compounded medications. Complete ingredient transparency and labeling of allergens establishes acceptable safety profiles.
Personalization Options
Varied patient needs call for customizable treatments that exceed standard one-size-fits-all protocols. Outstanding brands provide a personalized approach through extensive customization options, including variable concentrations, adjuvant blends (e.g., minoxidil), and various delivery formats (e.g., solutions and controlled-release gels). These tailored therapies are physician-crafted to safely elicit the best results.
User Experience
Prolonged, uninterrupted treatment is necessary to achieve positive results, highlighting the importance of user comfort and satisfaction. Practical considerations, such as application convenience, precise instructions, manageable dose routines, and excellent customer support, all influence treatment adherence.
Cost Transparency
Because treatment sustainability also depends on affordability, we also examined billing models and price transparency. Premium suppliers have honest and reasonable pricing that covers primary treatment costs (prescriptions, consultations, deliveries, and monitoring), free of hidden fees that are common among telehealth providers.
Selecting Your Ideal Treatment
Begin by assessing your hair loss level and treatment objectives, determining whether moderate maintenance or intensive regrowth protocols are needed. Further considerations include application method preferences (gels, solutions, sprays, etc.), helpful adjuvants, and skin sensitivities. Budgetary limitations and your required level of physician involvement also come into play.
With these factors in mind, let’s dive into the following product reviews that highlight distinct approaches.
Our Top 8 Topical Dutasteride Products
#1: Ulo Dutasteride + Minoxidil Plus Rx
Pros: Cons: ✓ PhD-scientist developed with thorough customization ✗ USA-only service for prescription treatments ✓ Customizable concentrations with adjuvant ingredients ✓ Board-certified physician oversight, monitoring, and tailored treatment protocols ✓ Verified quality and safety ✓ Full price transparency and free shipping The top position goes to Ulo, a leader in clinical rigor and precision hair loss medicine. Known for their highly individualized treatments, cutting-edge protocols, and PhD oversight, Ulo’s offerings are among the most comprehensive and patient-centered available. Their dual topical dutasteride solution with minoxidil delivers potent DHT suppression and new growth stimulation, proven to be more effective than either therapy alone.
Ulo’s topical systems feature customization across the spectrum, from dutasteride concentration (0.02% for lighter therapy to 0.2% for more aggressive intervention) to adjuvants like absorption enhancers, antioxidants, and vasodilators. Premium services include licensed dermatologist consultations, tailored treatment plans, and continued medical oversight, with a starting monthly subscription price of $84 and no added fees.
All Ulo safety and medication sourcing practices adhere to the highest regulatory standards, including partnerships with certified domestic pharmacies and strict follow-up protocols. Full ingredient transparency and irritant-free formulations establish excellent safety profiles. Patients enjoy free prescription deliveries and responsive support through Ulo’s sleek telehealth platform.
Our Verdict: Ulo’s advanced dual formula harnesses the power of customized topical dutasteride to deliver the gold standard of hair loss therapy. With PhD-developed formulas, genuine customization, and complete medical oversight, this platform offers proven benefits through a transparent monthly subscription that starts at $84. Their dedication to upholding the highest standards of safe and effective care positions Ulo as the best choice for your hair restoration needs.
#2 XYON Topical Dutasteride
Pros: Cons: ✓ Proprietary SiloxysSystem™ Gel for timed release ✗ Premium pricing at $129/month ✓ Highest concentration (2% dutasteride) for aggressive protocols ✗ Limited customization options ✓ Physician oversight ✗ Not recommended for sensitive skin ✓ Service area includes the US and Canada XYON stands out for its innovative, technology-driven approach, exemplified by its proprietary SiloxysSystem Gel delivery base, which contains delayed-release topical dutasteride at a high concentration of 2%. This controlled-release method allows for a higher dutasteride concentration and more targeted treatment, protecting against systemic exposure and potentially serious side effects.
While this treatment is a strong option for advanced hair loss cases due to its potency, XYON emphasizes its advanced and proven delivery method over treatment customization features. Monotherapy involves a fixed dose of 2% dutasteride, which may be too strong for some patients. Excipients include ingredients like silicone that are not recommended for sensitive skin.
XYON’s premium price point reflects not only its forward-looking strategy but also its high medical standards, as the platform offers qualified physician support throughout the process.
Our Verdict: XYON’s topical dutasteride formulation delivers maximum treatment with minimal systemic risk through its unique delayed-release carrier gel. It is recommended for advanced hair loss patients who can tolerate higher doses and potential irritants, particularly those who don’t mind paying a premium for its advanced technology development.
#3: Strut Health Topical Dutasteride Gel
Pros: Cons: ✓ Good value at $69/month with full customization ✗ Lower maximum dutasteride concentration (0.1%) ✓ Alcohol-free, preservative-free options available ✗ Moderate doses may be insufficient for advanced hair loss cases ✓ Extensive customization of adjuvant ingredients ✗ Occasional gaps in customer service are cited ✓ Licensed medical oversight with hair loss specialization ✗ Service area excludes Arkansas Strut Health is a solid mid-tier option that balances accessible pricing with medically guided customization, emphasizing cost-effectiveness over potency. The preservative- and alcohol-free dutasteride gel contains a moderate 0.1% dutasteride, with tailored add-on options including minoxidil, biotin, and tretinoin at varying concentrations. Capped at such a middling dose, this gentle gel formula is best for mild to moderate hair loss patients with sensitive or dry skin.
Fluocinolone, a corticosteroid, is also available as an adjuvant. While leading providers like Ulo avoid such ingredients due to potential safety concerns, Strut provides appropriate safety disclaimers and maintains an overall emphasis on ingredient transparency, backed by medical oversight.
Strut’s subscription fee of $69 per month for topical dutasteride treatment is a competitive offering, covering consultations, follow-ups, and prescription shipping. Patients cite flexible cancellation policies and rapid home deliveries as notable benefits. We detected some mixed reviews regarding delayed customer service responses and logistical issues.
Our Verdict: Strut Health offers a compelling value proposition with its topical dutasteride gel, priced at only $69 per month, and features that rival those of more expensive providers. The gentle formula with a limited 0.1% dutasteride concentration suits patients with moderate treatment needs who are seeking a budget-friendly option with flexible add-ons and legitimate medical oversight.
#4: Blends Growth PRO Topical
Pros: Cons: ✓ Doctor-founded with hair restoration specialization ✗ 90-day supply requirement may not suit all patients ✓ Competitive pricing at ~$45/month (90-day supply) ✗ Fixed formula despite “personalized” marketing claims ✓ Preservative-free, alcohol-free formula with 5 active ingredients ✗ Includes latanoprost with limited long-term safety data for scalp use ✓ 0.3% dutasteride and 7.5% minoxidil offer powerful DHT inhibition ✗ Lacks true customization compared to leading competitors ✓ Proprietary hydrating gel base with advanced formulation Blends is a physician-founded company with a heavy focus on cutting-edge manufacturing methods and sophisticated formulations. Their small but specialized catalog includes this topical dutasteride treatment made with a proprietary gel base for timed medication release and scalp hydration. Free of alcohol and preservatives, the concentrated 0.3% dutasteride and 7.5% minoxidil formula offers powerful therapeutic effects.
Although Blends asserts the “personalized” and “customized” nature of its products, its Growth Pro Topical gel is actually a comprehensive fixed formula with three adjuvant ingredients in addition to dutasteride and minoxidil: tretinoin, latanoprost, and ketoconazole. Competitively priced at about $45/month (sold in 90-day batches), this offering stands out for its full ingredient transparency and high concentrations suited to aggressive treatment protocols.
However, the use of latanoprost poses concerns due to the lack of long-term studies confirming its safety and efficacy as a topical hair loss treatment. Despite marketing claims of customized protocols, Blends does not allow formulation adjustments to this fixed gel product. Although the monthly cost is lower than that of most leading brands of comparable quality, the minimum purchase requirement of a 90-day supply (~$135) may not suit all patients.
Our Verdict: This physician-crafted gel formula from Blends is competitively priced, featuring high concentrations of synergistic ingredients for advanced hair loss treatment. Its positioning as a mid-level selection rather than a top choice is due to its false personalization claims and unproven add-on ingredient.
#5: Happy Head Topical Rx Dutasteride & Minoxidil
Pros: Cons: ✓ Dermatologist oversight with ongoing support ✗ Limited dutasteride concentration options ✓ 0.3% dutasteride and 8% minoxidil formula with customizable add-ons, including ketoconazole ✗ Pricing increases after the initial months and can reach ~$109 ✓ Established brand with a 6-month money-back guarantee ✗ Some formulations contain potential irritants ✓ User-friendly platform and free consultation ✗ Shipping and customer service inconsistencies Happy Head represents a dependable option for targeted hair restoration, recognized for its dermatologist-backed and customizable treatments. This hybrid dutasteride-minoxidil solution offers a strong dose of 0.3% dutasteride with up to 8% minoxidil for effective DHT reduction. Elective adjuvants include retinoic acid and hydrocortisone. Anti-dandruff agent ketoconazole can be added for an additional fee.
While some clinicians warn against the use of topical corticosteroids like hydrocortisone, Happy Head seems to follow established medical safety protocols with appropriate disclaimers. Inactive ingredients like alcohol and propylene glycol can irritate and should be approached with caution. Highly concentrated DHT blockers with harsh excipients require close medical oversight, and patients with sensitive skin may want to seek other options.
Although transparent about costs, Happy Head’s variable pricing can grow quickly with treatment duration and formulation. Their topical dutasteride solution starts at $59 per month and shifts to $89 after the first six months. The anti-dandruff adjuvant can be added for $20, bringing the total to $109 per month, which exceeds the cost of some comparable providers. Mixed patient reviews cite issues with deliveries and logistical support.
Our Verdict: This combination treatment from Happy Head is a solid choice for patients in need of more aggressive treatment and concentrated formulas. Continued dermatologist oversight, personalization options, and potent formulations can help patients whose results may have stalled on other treatments. However, highly active ingredients and variable pricing may deter those seeking gentler therapies and fixed costs.
#6 Maximus Dutasteride+ Gel
Pros: Cons: ✓ High level of personalization with physician-driven telehealth oversight ✗ Limited long-term safety data for some ingredient combinations ✓ Competitive monthly pricing ✗ Availability restricted to the U.S. ✓ Physician-guided treatment plans and ongoing clinical support ✗ No subscription flexibility (all via 90-day supply) ✓ Proprietary gel base designed for scalp absorption and minimal mess ✓ Evidence-informed protocols supported by board-certified physicians Maximus Tribe’s Max-Absorb Gel is a highly customizable, prescription-only hair loss solution leveraging physician-driven telehealth and evidence-based formulation. At $54.99 for a 90-day supply, it offers a lot of value for those looking for efficacy and clinical support. The core formulation includes dutasteride (0.1%) with optional minoxidil, tretinoin, and fexofenadine, delivered in a proprietary gel base that is designed to minimize mess and maximize scalp coverage.
Maximus prioritizes convenience and targeted care: onboarding and ongoing support are managed by board-certified doctors, and the medication is shipped directly to the patient for easy home application. While ingredient customization and clinical guidance are strengths, choices remain limited in comparison to other telehealth options (like Ulo).
Our Verdict: Maximus offers a well-priced topical dutasteride gel that balances multi-ingredient efficacy with convenient home use and physician oversight.
#7: Miiskin Dutasteride + Minoxidil
Pros: Cons: ✓ Direct board-certified dermatologist consultations ✗ Total cost varies with third-party pharmacy fees ✓ Custom-compounded formulations with up to 3 active ingredients ✗ Basic compounding without advanced delivery systems ✓ No automatic subscriptions—pay per order ✗ Less specialized in hair loss compared to dedicated platforms ✓ Legitimate medical oversight with prescription flexibility ✓ Serves both the USA and Mexico Miiskin differs from the subscription-based providers on this list, as it facilitates direct medical consultations between patients and qualified dermatologists. The pay-as-you-go model allows legitimate medical access without subscription requirements. Miiskin charges $59 for initial consultations and $30 for returning visits. Medication fees vary and are charged separately at the pharmacy where the medication is filled.
While the dermatologist-direct platform increases access to qualified care and prescription hair loss treatments, its simple compounded formulas lack the customization and specialization of leading brands. Ingredient disclosure is limited beyond general mentions of three-active-ingredient formulations and the listing of a combination of dutasteride and minoxidil therapy.
The model does not permit total cost calculation until third-party pharmacy checkout, separate from Miiskin’s billing process, which only covers prescribing dermatologist consultation fees.
Our Verdict: Although Miiskin offers medical legitimacy, the dermatology consultation platform lacks the treatment specialization and transparency of top dutasteride therapy sources. It may be a valuable tool for patients with hair loss to connect with qualified providers and access basic compounded formulas.
#8 Musely The Hair Topical Solution – Classic
Pros: Cons: ✓ Lowest cost option at $33/month ✗ Minimal medical oversight compared to competitors ✓ 0.3% dutasteride concentration in a customizable formula ✗ Limited customization options ✓ Aesthetic medicine platform with dermatologist involvement ✗ Medical rigor lacking ✓ Multi-ingredient formulations ✗ Less specialized hair loss expertise ✗ Some ingredients are not recommended for sensitive skin
Musely’s topical dutasteride solution occupies the final slot on our list as the lowest-priced option at just $33 per month (plus a one-time $20 consultation fee at onboarding). The lower price point reflects less extensive offerings that are well-suited for patients who prefer a more self-directed approach. This tailored prescription formulation combines up to 0.3% dutasteride and 8% minoxidil with optional add-ons of ketoconazole to fight dandruff and hydrocortisone to reduce inflammation.It is less customizable than other options, characteristic of Musely’s simplified strategy. Excipients like propylene glycol and alcohol may deter ingredient-conscious consumers due to irritant potential. While such potent formulas and highly active ingredients offer more powerful treatment, they also carry a greater risk of adverse reactions and call for qualified medical supervision.
However, unlike premium providers, Musely is not known for close medical oversight. In fact, the aesthetic medicine platform has a markedly superficial onboarding and assessment process that falls short of the thorough medical evaluations offered by top providers. Although clinicians are involved in the platform, follow-up care and specialized hair restoration knowledge are lacking, according to customer reviews.
Our Verdict: Musely is included as a purely budget-friendly option for cost-conscious patients who understand the importance of self-monitoring and supplement with appropriate medical consultation.
Bottom Line on Buying Topical Dutasteride Online
According to our thorough analysis, Ulo is the best choice for topical dutasteride therapy in 2026. This platform stands out for its rigorous scientific foundation, highly tailored treatments, and commitment to close medical oversight, outperforming the fixed protocols and misleading marketing claims of its peers.
Rather than standardized approaches, Ulo’s PhD-designed therapies with built-in customization features meet the individual needs of each patient, representing the pinnacle of evidence-based precision care in the rapidly evolving field of hair restoration medicine.
Expectations and Timeline
Clinical findings support the following timeline of expected treatment outcomes in topical dutasteride therapy. Familiarity with these will ground your therapy in a realistic outlook, encouraging patience and prolonged adherence.[10]Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 mg) in … Continue reading,[11]Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., & Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology, 240, 833 – 843. Available at: … Continue reading,[12]Radhakrishnan, P., Mariyappan, G., Karthi, J., & Jaisumi, K. (2024). Formulating, Optimizing and Evaluation of Dutasteride Loaded Insitu-Emulgel for Androgenetic Alopecia. International Journal … Continue reading
- Early Stage (Months 1-3): Within days of beginning treatment, scalp DHT levels start to decline, although cosmetic improvements take longer to show as existing hair cycles complete. Some patients may experience temporary hair shedding in the first 2-8 weeks, which is considered a positive sign that the treatment is working by triggering new growth cycles.
- Emergence Stage (Months 3-6): After about 3 months of continued therapy, subtle visible improvements typically begin to emerge. Existing strands become thicker, and previously inert hair follicles are reactivated to produce new shafts for measurable increases in hair count metrics.
- Maximization Stage (Months 6-18): This phase sees the most considerable improvements, as hair count and diameter continue to increase for a thicker, healthier appearance. The majority of patients reach optimal results within a year.
- Maintenance Stage (Long-Term): After reaching peak outcomes, continuous daily application is necessary to sustain benefits. Studies show that indefinite treatment can maintain positive results, although occasional adjustments may be needed over time to account for typical signs of aging.
Risk Profile and Safety
Topical dutasteride is considered a safe hair loss intervention, particularly when weighed against oral formulas that can carry substantial risk due to systemic exposure. Nonetheless, informed risk assessment and familiarity with safety protocols are recommended before beginning any new treatment. Here is a brief overview of medical data on the safety and side effects of topical dutasteride.[13]Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., & Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology, 240, 833 – 843. Available at: … Continue reading
Common Side Effects
Adverse patient reactions to topical dutasteride are rare and typically limited to mild, localized scalp irritation that resolves in 2-4 weeks, occurring in only about 5-10% of new users. Depending on individual sensitivities and irritating formulation additives like alcohol, scalp dryness, flaking, and contact dermatitis are possible, though the latter is less common. Mild systemic side effects are extremely rare, documented in under 2% of patients.
Serious Considerations
Also rare are severe allergic reactions and pronounced systemic side effects, which can include sexual dysfunction, mood disorders, and gynecomastia. These all warrant prompt medical intervention. Topical dutasteride is not recommended in patients with active scalp infections and hormonal disorders.
Topical dutasteride is strongly contraindicated in pregnant or nursing women and in those planning future pregnancies due to the risk of birth defects. Patients with known hypersensitivity to DHT blockers should also avoid use.
Safety and optimal treatment outcomes depend on regular medical follow-up evaluations, typically at three-month intervals in the first year. This enables early identification of problems and treatment response evaluation.
Conclusion: Best Topical Dustaride Products
Cutting-edge topical dutasteride formulations bridge the gap between pharmacological precision and patient satisfaction in hair restoration therapy. Available products represent a spectrum of targeted, proven treatments that are considered safer and more customizable than previous-generation modalities.
Our thorough review of the evolving market identified clear leaders, with Ulo setting itself apart as an exemplary provider of topical dutasteride. Its scientifically optimized protocols are tailored to meet the individual needs of each patient, with qualified medical supervision to ensure a safe and transformative treatment experience.
As hair restoration technology advances, leading topical dutasteride formulations, such as Ulo’s, are at the forefront, empowering users with convenient and clinically proven solutions that not only enhance appearance but also improve quality of life.
Explore Ulo’s advanced topical dutasteride treatments today.
Frequently Asked Questions
Do I need a prescription for topical dutasteride? Yes, topical dutasteride is a prescription-only treatment in the United States. This ensures proper candidate screening and legitimate medical supervision throughout treatment.
When can I expect results? Initial DHT suppression on the scalp occurs within the first few days, but visible changes in hair growth typically emerge within 3-4 months. Benefits generally reach their peak after 12-18 months of consistent application.
How do I apply topical dutasteride? Use the product applicator to deposit the treatment directly onto clean, dry scalp areas where you are experiencing hair loss. Allow it to absorb fully before using other hair products. Following application, avoid wetting the hair for several hours and wash your hands thoroughly.
Can I combine topical dutasteride with other treatments? Yes, the benefits of topical dutasteride can be enhanced through combination therapies, such as minoxidil. Consult your prescribing doctor to learn about safe and effective hybrid treatments.
What will happen if I stop treatment? When treatment is halted, scalp DHT levels gradually return to starting levels, which typically leads to resumed hair loss after several months.
How much does topical dutasteride cost? Pricing varies from about $30 to $135+ for a monthly supply, depending on concentration, formulation, brand, and telehealth services included. Leading brands provide comprehensive medical oversight and customization, offering transparent and flat-rate monthly subscription fees.
References[+]
References ↑1 R. Cuevas-Díaz Durán., Martinez-Ledesma, E., Garcia-Garcia, M., Gauzin, D.B., Sarro-Ramirez, A., Gonzalez-Carrillo, C., Rodriguez-Sardin, D., Fuentes, A., Cardenas-Lopez, A. (2024) The Biology and Genomics of Human Hair Follicles: A Focus on Androgenetic Alopecia. International Journal of Molecular Sciences, 25. Available at: https://doi.org/10.3390/ijms25052542. ↑2 Dhurat, R., Sharma, A., Rudnicka, L., Kroumpouzos, G., Kassir, M., Galadari, H., Wollina, U., Lotti, T., Golubović, M., Binic, I., Grabbe, S., & Goldust, M. (2020). 5‐Alpha reductase inhibitors in androgenetic alopecia: Shifting paradigms, current concepts, comparative efficacy, and safety. Dermatologic Therapy, 33. Available at: https://doi.org/10.1111/dth.13379. ↑3 Arif, T., Dorjay, K., Adil, M., & Sami, M. (2017). Dutasteride in Androgenetic Alopecia: An Update.. Current clinical pharmacology, 12 1, 31-35. Available at: https://doi.org/10.2174/1574884712666170310111125. ↑4 Harcha, W., Martínez, J., Tsai, T., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia.. Journal of the American Academy of Dermatology, 70 3, 489-498.e3. Available at: https://doi.org/10.1016/j.jaad.2013.10.049. ↑5, ↑7, ↑10 Obeid, M., Fattah, N., Elfangary, M., & Husseni, R. (2024). Comparison between Topical Minoxidil 5% Alone versus Combined with Dutasteride (Topical 0.02% through Microneedling or Oral 0.5 mg) in Treatment of Androgenetic Alopecia. QJM: An International Journal of Medicine. Available at: https://doi.org/10.1093/qjmed/hcae175.207. ↑6, ↑8, ↑11, ↑13 Ding, Y., Wang, C., Bi, L., Du, Y., Lu, C., Zhao, M., & Fan, W. (2024). Dutasteride for the Treatment of Androgenetic Alopecia: An Updated Review. Dermatology, 240, 833 – 843. Available at: https://doi.org/10.1159/000541395. ↑9, ↑12 Radhakrishnan, P., Mariyappan, G., Karthi, J., & Jaisumi, K. (2024). Formulating, Optimizing and Evaluation of Dutasteride Loaded Insitu-Emulgel for Androgenetic Alopecia. International Journal of Pharmaceutical Research and Applications. Available at: https://doi.org/10.35629/4494-0906631659. Finasteride is a prescription medication originally created to treat benign prostatic hyperplasia (BPH), and now commonly used to treat androgenic alopecia (AGA). It achieves this through targeting 5α‑reductase (5AR), an enzyme that converts testosterone to dihydrotestosterone (DHT), lowering levels of DHT in the scalp. DHT is the key androgen driving follicular miniaturization and is known to drive hair loss. Therefore, the finasteride-induced reduction of DHT results in increased hair growth.[1]Asfour L, Cranwell W, Sinclair R, (2023), Male Androgenetic Alopecia. In: Feingold KR, Adler RA, Ahmed SF, et al., editors, Endotext [Internet], South Dartmouth (MA): MDText.com, Inc.; Available at: … Continue reading
Despite the high amount of evidence supporting finasteride use, many users are concerned about starting due to its potential side effects, including a loss of libido and erectile dysfunction.[2]Diviccaro, S., Melcangi, R.C., Giatti, S. (2019). Post-finasteride syndrome: an emerging clinical problem. Neurobiology of Stress. 12. 100209. Available at: https://doi.org/10.1016/j.ynstr.2019.100209 Many people presume that these side effects, to some extent, are caused by finasteride’s effects on male hormones. In fact, many people presume that these side effects are caused by finasteride lowering testosterone.
The potential for the finasteride mechanism to lower testosterone levels is a common fear; however, the sexual side effects observed in a minority of finasteride users do not occur due to a finasteride-induced reduction in testosterone. Paradoxically, finasteride causes a rise in testosterone, yet occasional sexual side effects are still seen.
In this article, we will discuss whether finasteride lowers testosterone and what the mechanisms are behind sexual side effects in finasteride users.
Interested in Oral Finasteride?
Oral finasteride & minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Does Finasteride Work?
AGA is one of the most common hair loss disorders in the world, and is most often characterized as the progressive miniaturization of hair follicles – induced by androgen sensitivity. Activation of the androgen receptor shortens the growth phase of the hair cycle, increases rates of hair shedding, and upon reentry into anagen, affected hairs are often thinner than in the previous cycle. This eventually leads to visible hair loss that is chronic and progressive; without treatment, it worsens over months, years, and decades.[3]Ho, C.H., Sood, T., Zito, P.M., (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026)
DHT is the key androgen driving follicular miniaturization. When free testosterone comes into contact with an enzyme called 5AR, that free testosterone is converted into another hormone – DHT – which then binds to hair follicle sites and initiates a cascade that results in hair shedding and hair follicle miniaturization. Both 5AR and DHT have been shown to be elevated in balding areas of the scalp compared to the hair-containing scalp.[4]Dallob, A.L., Sadick, N.S., Unger, W., Lipert, S., Geissler, L.A., Gregoire, S.L., Nguyen, H.H., Moore, E.C. and Tanaka, W.K. (1994). The Effect of Finasteride, a 5 Alpha-Reductase Inhibitor, on … Continue reading
Effect of Finasteride on DHT Suppression
Finasteride suppression of DHT can be fit to a non-linear (logarithmic) dose-response curve, i.e., even very low levels of finasteride (≤0.2 mg/day) can produce near-maximal suppression of DHT in both the serum and the scalp.[5]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., … Continue reading The majority of its inhibitory action is achieved at low doses, with higher dosing only providing marginal additional effects.
Clinical trials have demonstrated that finasteride treatment decreases levels of DHT in the serum by 71-72% and in the scalp by 64-69%.[6]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., … Continue reading
Clinical Effectiveness
The clinical effectiveness of finasteride in improving hair growth has been demonstrated in multiple clinical trials. It has been shown to improve hair growth and slow further hair loss across 1-year, 2-year, and 5-year clinical trials, with 65% of finasteride-treated men demonstrating increased hair count at 5 years compared to 0% of the placebo-treated (sugar pill group) men.[7]Finasteride Male Pattern Hair Loss Study Group. (2002). Long-Term (5-Year) Multinational Experience With Finasteride 1 mg in the Treatment of Men With Androgenetic Alopecia. Eur J Dermatol. 12:38-49. … Continue reading,[8]Shapiro, J., Kaufman, K.D. (2003). Use of Finasteride in the Treatment of Men With Androgenetic Alopecia (Male Pattern Hair Loss). J Investig Dermatol Symp Proc. 8(1):20-23. Available at: … Continue reading
Does Finasteride Lower Testosterone?
The belief that finasteride lowers testosterone is common. However, there is no evidence supporting this. In fact, research has actually shown that finasteride treatment increases levels of testosterone.
A four-year clinical trial investigating the safety and efficacy of finasteride in patients with BPH demonstrated a ~15% rise in testosterone in the serum compared to the placebo group.[9]Roehrborn, C., Lee, M., Meehan, A., Waldstreicher, J. (2003). Effects Of Finasteride On Serum Testosterone And Body Mass Index In Men With Benign Prostatic Hyperplasia. Urology. 62(5). 894–899. … Continue reading Additional trials have also reported a moderate rise in testosterone (5-33%; not dose-dependent).[10]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., … Continue reading,[11]Uygur, M.C., Arık, A.İ., Altuğ, U., Erol, D., (1998). Effects Of The 5α-Reductase Inhibitor Finasteride On Serum Levels Of Gonadal, Adrenal, And Hypophyseal Hormones And Its Clinical … Continue reading,[12]Amory, J.K., Wang, C., Swerdloff, R.S., Anawalt, B.D., Matsumoto, A.M., Bremner, W.J., Walker, S.E., Haberer, L.J. and Clark, R.V. (2007). The Effect Of 5α-Reductase Inhibition With Dutasteride And … Continue reading
Although the rise in testosterone was significantly different between finasteride- and placebo-treated individuals, levels of testosterone consistently remained within the normal physiological range. The increase in testosterone was identified in women as well as men, and was found to be more pronounced in men with low baseline testosterone.[13]Albasher, G., Bin-Jumah, M., Alfarraj, S., Al-Otibi, F., Al-Sultan, N.K., Alarifi, S., Alkahtani, S. and Al-Qahtani, W.S. (2020). Prolonged Use Of Finasteride-Induced Gonadal Sex Steroids … Continue reading,[14]Roehrborn, C., Lee, M., Meehan, A., Waldstreicher, J. (2003). Effects Of Finasteride On Serum Testosterone And Body Mass Index In Men With Benign Prostatic Hyperplasia. Urology. 62(5). 894–899. … Continue reading However, not all patients tested experienced this rise in testosterone.
The evidence therefore supports a short-term rise in testosterone on finasteride treatment, while remaining within the physiological range. Many of these studies were carried out in relation to BPH, where the finasteride doses are higher (generally 5 mg vs 1 mg), so the impact of finasteride for AGA treatment is likely to be even lower.
Why Does Testosterone Increase When DHT Falls?
The inhibition of the 5AR enzyme results in the blocking of the DHT production pathway, resulting in the testosterone that would otherwise have been converted to DHT remaining as testosterone. No large increases in testosterone are seen, as not only do negative feedback adjustments occur, but free testosterone is also thought to be redistributed within the androgen pathway, where it is converted into estradiol.
Can the Rise in Testosterone Have Sexual Side Effects?
The moderate rise in testosterone identified in individuals taking finasteride does not result in a rise above the physiological range, making sexual side effects directly as a result of the rise in testosterone unlikely. In general, a moderate rise in testosterone levels improves sexual function. This appears to contradict the well-characterized (though uncommon) sexual side effects of finasteride treatment. Therefore, another mechanism must be at work to induce these sexual side effects.
Finasteride Sexual Side Effects
What are the Sexual Side Effects of Finasteride?
Sexual side effects have been reported in a small minority of users. These include:
- Erectile dysfunction
- Ejaculatory dysfunction
- Loss of libido
While these symptoms often improve or resolve on their own, persistent sexual dysfunction has been reported in a very small number of finasteride users, in a condition known as post-finasteride syndrome.[15]Hirshburg, J.M., Kelsey, P.A., Therrien, C.A., Gavino, A.C. and Reichenberg, J.S. (2016). Adverse Effects And Safety Of 5-Alpha Reductase Inhibitors (Finasteride, Dutasteride): A Systematic Review. … Continue reading However, reports of this condition are controversial, and conflicting results are seen in different studies. If you are interested in learning more, see our article exploring post-finasteride syndrome.
What is the Prevalence of Sexual Side Effects?
In controlled clinical trials, sexual side effects of taking finasteride are uncommon, but exact estimates vary depending on the dose, study design, and population receiving treatment.
- In a 4-year, randomized, placebo-controlled clinical trial assessing the prevalence of sexual adverse experiences (AEs) on the treatment of BPH with 5 mg of finasteride, 15% of finasteride-treated and 7% of placebo-treated men had sexual AEs within the first year, indicating an increase in AEs in the finasteride group. However, no differences between placebo- and finasteride-treated AEs were seen in years 2-4.[16]Wessells, H., Roy, J., Bannow, J., Grayhack, J., Matsumoto, A.M., Tenover, L., Herlihy, R., Fitch, W., Labasky, R., Auerbach, S., Parra, R., Rajfer, J., Culbertson, J., Lee, M., Bach, M.A. and … Continue reading
- A meta-analysis (a study where data from multiple independent studies are analyzed) of 15 clinical trials where AGA was treated with 1 mg of finasteride found that only 5.31% of users had adverse sexual effects compared to 3.05% of those taking a placebo.[17]Lee, S., Lee, Y.B., Choe, S.J. and Lee, W. (2019). Adverse Sexual Effects Of Treatment With Finasteride Or Dutasteride For Male Androgenetic Alopecia: A Systematic Review And Meta-Analysis. Acta … Continue reading
- A different meta-analysis of 17 clinical trials found that men taking finasteride had a 1.29 times higher risk of underactive sexual desire or a 1.47 times higher risk of erectile dysfunction.[18]Corona, G., Tirabassi, G., Santi, D., Maseroli, E., Gacci, M., Dicuio, M., Sforza, A., Mannucci, E. and Maggi, M. (2017). Sexual Dysfunction In Subjects Treated With Inhibitors Of 5α-Reductase For … Continue reading
Meta-analyses and studies with a high number of participants are the gold standard for assessing the risk of sexual side effects. Smaller studies have put the incidence of sexual side effects higher; however, these results should be interpreted with caution due to the limitations of the clinical trials.
Further caution should be taken due to the “nocebo” phenomenon, where a patient may experience negative symptoms or worse outcomes due to their being informed that the product may cause them harm.
A small study investigating this phenomenon in patients treated for BPH with 5 mg finasteride found that the group who received counseling on the sexual side effects (i.e., were informed of these side effects) demonstrated a significantly higher proportion of sexual side effects than the group who did not receive counseling (43.6% vs. 15.3%).[19]Mondaini, N., Gontero, P., Giubilei, G., Lombardi, G., Cai, T., Gavazzi, A. and Bartoletti, R. (2007). Finasteride 5 mg And Sexual Side Effects: How Many Of These Are Related To A Nocebo Phenomenon? … Continue reading
Care should also be taken when interpreting information on natural health websites and online forums, as the perceived prevalence of sexual side effects may seem incredibly high on these. This may occur due to multiple reasons, including:
- Fearmongering due to the desire to sell natural remedies claiming to be sexual side-effect-free (which often isn’t true).
- Users are more likely to post in anger about issues with the product, whereas those content with the product are less likely to post (the “Yelp Effect”).
Why do Sexual Side Effects Occur Despite Higher Testosterone?
As discussed above, increased testosterone (within the physiological range) enhances sexual function. However, despite the slightly raised testosterone seen in men taking finasteride, sexual side effects still occur. Below are three main pathways through which these side effects have been suggested to occur. Please consider that none of these mechanisms have been conclusively proven, and are only suggestions as to how sexual side effects may be occurring.
DHT Depletion
DHT is a potent androgen, binding the androgen receptor with greater strength than testosterone. DHT and testosterone can bind to androgen receptors in the corpora cavernosa (erectile tissue in the penis), causing an upregulation in nitric oxide production and resulting in increased blood flow into the penis and an erection.
It has been suggested that susceptible individuals experiencing decreased levels of DHT on finasteride treatment experience a reduction in nitric oxide production, limiting blood flow into the penis, and, consequently, in erectile dysfunction.[20]Clapauch, R. (2011). Finasteride and Erectile Dysfunction: Fact or Fiction? Brasília Medical (Medical Experience). 48(4), pp.422–427. Available at: … Continue reading,[21]Erdemir, F., Harbin, A. and Hellstrom, W.J.G. (2008). 5-Alpha Reductase Inhibitors And Erectile Dysfunction: The Connection. The Journal Of Sexual Medicine. 5(12), pp.2917–2924. Available at: … Continue reading
DHT is also an important contributor to secretory function and semen volume, as controlled by two glands – the prostate and seminal vesicles. These glands are androgen-dependent. DHT is the primary androgen involved, but testosterone can compensate for reduced DHT.[22]Anitha, B., Inamadar, A.C., Ragunatha, S. (2009). Finasteride-Its Impact on Sexual Function and Prostate Cancer. J Cutan Aesthet Surg. 2(1):12–16. Available at: … Continue reading,[23]Cai, L.Q., Fratianni, C.M., Gautier, T., Imperato-McGinley, J. (1994). Dihydrotestosterone Regulation of Semen in Male Pseudohermaphrodites with 5 Alpha-Reductase-2 Deficiency. Journal of Clinical … Continue reading
Finasteride treatment does not cause low semen or ejaculatory volume in all users, but this does appear in rare cases. This ejaculatory dysfunction could occur as a result of testosterone not sufficiently compensating for DHT’s role in secretory function.
Neurosteroids in the Central Nervous System
In addition to its ability to convert testosterone to DHT, 5AR is also important in the conversion of other molecules into neurosteroids. Neurosteroids are steroids synthesized within the central nervous system (CNS; formed of the brain and spinal cord) that play a key role in controlling activity within the CNS, including anxiety, stress, and depression.
Treatment with 5AR inhibitors, such as finasteride, can cause a decrease in active neurosteroids within the CNS and altered transmission of certain signals within the brain. This disrupted neurosteroid signaling has been suggested as a potential cause of the decreased libido side effect that is occasionally seen in users of finasteride.[24]Irwig, M.S. (2014), Persistent Sexual and Nonsexual Adverse Effects of Finasteride in Younger Men. Sex Med Rev. 2(1):24–35. Available at: https://doi.org/10.1002/smrj.19,[25]Traish, A.M., Hassani, J., Guay, A.T., Zitzmann, M., Hansen, M.L. (2011). Adverse Side Effects of 5 Alpha-Reductase Inhibitors Therapy: Persistent Diminished Libido and Erectile Dysfunction and … Continue reading
Androgen Redistribution
As discussed above, the inhibition of 5AR results in less testosterone being converted into DHT and a mild rise in testosterone. It has been suggested that the increase in free testosterone may be compensated for through androgen redistribution via conversion to estradiol (an estrogen).
Estradiol levels show a minor rise in finasteride users of approximately 15% (while remaining within physiological reference ranges).[26]U.S. Food and Drug Administration, (2022), Propecia (Finasteride) Tablets Label. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020788s030lbl.pdf (Accessed: 07 January 2026) For those with a genetic predisposition to high estrogen or whose diets, lifestyles, and environments have elevated their estrogen levels, a finasteride-induced further rise in estrogen may be sufficient for the development of gynecomastia (the development of male breast tissue).
How Can We Reduce the Risk of Sexual Side Effects?
The first step if side effects occur is to speak to your primary care physician to ensure that side effects are recorded and appropriate management steps are taken. If side effects are severe or rapidly worsening, you should immediately see your physician. The strategies discussed below are only applicable if mild to moderate side effects are experienced, and should only be carried out under clinical supervision. More information on finasteride-associated side effects can be found in our articles on topical finasteride and reducing side effects.
- Lowering your daily dose of finasteride
Finasteride produces the majority of its DHT-lowering effects at relatively low doses. The standard finasteride daily dose is 1 mg. Due to its non-linear dose-response curve, lower doses can still have significant effects on lowering serum and scalp DHT and enhancing hair growth.
Finasteride side effects are often dose-sensitive, meaning that lowering the daily dose could be a first step to reducing side effects, without a significant loss in benefits.
Figure 1. The non-linear dose response curve of finasteride shows that increasing the concentration of the treatment does not translate to an increased reduction in serum DHT.
- Lowering the frequency of dosing
Mild flexibility in finasteride dosing also allows for carefully controlled alterations in dosing frequency. Finasteride has a relatively short half-life (time taken for the amount of drug in the body to halve), but does accumulate with repeated doses.[27]Steiner, J.F. (1996). Clinical Pharmacokinetics and Pharmacodynamics of Finasteride. Clinical Pharmacokinetics. 30(1):16–27. Available at: https://doi.org/10.2165/00003088-199630010-00002
As a result, the frequency of finasteride dosing can be reduced to 1 mg three times per week or 0.5 mg every other day (once steady-state suppression has been achieved) while maintaining sufficient DHT inhibition for significant hair growth effects.
This has the potential to reduce finasteride-associated side effects, particularly sexual side effects associated with hormonal changes.
- Switching to a topical finasteride
Topical, as opposed to oral, formulations deliver the drug directly to the scalp without extensive systemic circulation and therefore reduce overall drug exposure.
Randomized clinical trials have been carried out to assess the effectiveness of oral vs topical finasteride. These have demonstrated comparable clinical benefits, including hair count, hair thickness, and size of bald area.[28]Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing the Therapeutic Effects of Finasteride Gel and Tablet in Treatment of the Androgenetic Alopecia. Indian Journal of Dermatology, … Continue reading,[29]Bhura, M. (2013). Comparative Analysis of Topical and Oral Finasteride in Androgenetic Alopecia: Impact on Hair Growth, Systemic Absorption, and Adverse Effects. Indian Journal of Basic and Applied … Continue reading
Topical application lowers the amount of systemic drug, resulting in reduced side effects. Studies have shown that sexual side effects, in particular, are improved when using finasteride topically instead of orally.[30]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and Safety … Continue reading
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

The beneficial effects of topical finasteride require consistent, measured application and strong adherence to the prescribed application regimen. As such, users who are not experiencing side effects may find it easier to use oral finasteride instead.
- Using PDE5 inhibitors to manage sexual side-effects
PDE5 inhibitors can be used to treat erectile dysfunction by relaxing the blood vessels so as to allow increased blood flow into the penis. The effectiveness of these drugs is well studied, and they have also been shown to improve erectile function specifically in men taking finasteride.[31]Casabé, A., Roehrborn, C.G., Da Pozzo, L.F., Zepeda, S., Henderson, R.J., Sorsaburu, S., Henneges, C., Wong, D.G. & Viktrup, L. (2014). Efficacy and Safety of the Coadministration of Tadalafil … Continue reading
While PDE5 inhibitors improve erectile performance and sexual confidence, they do not address other sexual side effects of finasteride, such as libido or ejaculatory volume. In addition, as prescription medications, they should only be used under clinical supervision to reduce the risk of drug interactions.
Final Thoughts
Finasteride does not lower testosterone. In fact, clinical evidence consistently demonstrates that the reduced conversion of testosterone into DHT causes a modest increase in serum testosterone, which remains within the normal physiological ranges. Concerns about finasteride causing testosterone deficiency are therefore not supported by the scientific literature.
Sexual side effects, while real, occur only in a small minority of users and are unlikely to be caused by changes in testosterone. Instead, they are more likely linked to DHT depletion in androgen-sensitive tissues, altered neurosteroid signaling, androgen distribution, or the “nocebo” effect.
For most users, finasteride is effective and well-tolerated. For those who do experience side effects, strategies like dose adjustment or switching to topical use under the guidance of your physician may improve tolerability while maintaining benefits.
References[+]
References ↑1 Asfour L, Cranwell W, Sinclair R, (2023), Male Androgenetic Alopecia. In: Feingold KR, Adler RA, Ahmed SF, et al., editors, Endotext [Internet], South Dartmouth (MA): MDText.com, Inc.; Available at: https://www.ncbi.nlm.nih.gov/books/NBK278957/ (Accessed: 05 January 2026) ↑2 Diviccaro, S., Melcangi, R.C., Giatti, S. (2019). Post-finasteride syndrome: an emerging clinical problem. Neurobiology of Stress. 12. 100209. Available at: https://doi.org/10.1016/j.ynstr.2019.100209 ↑3 Ho, C.H., Sood, T., Zito, P.M., (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026) ↑4 Dallob, A.L., Sadick, N.S., Unger, W., Lipert, S., Geissler, L.A., Gregoire, S.L., Nguyen, H.H., Moore, E.C. and Tanaka, W.K. (1994). The Effect of Finasteride, a 5 Alpha-Reductase Inhibitor, on Scalp Skin Testosterone and Dihydrotestosterone Concentrations in Patients with Male Pattern Baldness. J Clin Endocrinol Metab. 79(3), pp.703-706. Available at: https://doi.org/10.1210/jcem.79.3.8077349 ↑5, ↑6, ↑10 Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., Carrington, P., Gencheff, C., Cole, G.W., Pariser, D.M., Epstein, E.S., Tanaka, W., Dallob, A.D., Vandormael, K., Geissler, L. and Waldstreicher, J. (1999). The Effects of Finasteride on Scalp Skin and Serum Androgen Levels in Men with Androgenetic Alopecia. J Am Acad Dermatol. 41(4), pp.550-554. Available at: https://pubmed.ncbi.nlm.nih.gov/10495374/ ↑7 Finasteride Male Pattern Hair Loss Study Group. (2002). Long-Term (5-Year) Multinational Experience With Finasteride 1 mg in the Treatment of Men With Androgenetic Alopecia. Eur J Dermatol. 12:38-49. Available at: https://pubmed.ncbi.nlm.nih.gov/11809594/ ↑8 Shapiro, J., Kaufman, K.D. (2003). Use of Finasteride in the Treatment of Men With Androgenetic Alopecia (Male Pattern Hair Loss). J Investig Dermatol Symp Proc. 8(1):20-23. Available at: https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑9, ↑14 Roehrborn, C., Lee, M., Meehan, A., Waldstreicher, J. (2003). Effects Of Finasteride On Serum Testosterone And Body Mass Index In Men With Benign Prostatic Hyperplasia. Urology. 62(5). 894–899. Available at: https://pubmed.ncbi.nlm.nih.gov/14624915/ ↑11 Uygur, M.C., Arık, A.İ., Altuğ, U., Erol, D., (1998). Effects Of The 5α-Reductase Inhibitor Finasteride On Serum Levels Of Gonadal, Adrenal, And Hypophyseal Hormones And Its Clinical Significance: A Prospective Clinical Study. Steroids. 63(4). 208–213. Available at: https://www.sciencedirect.com/science/article/abs/pii/S0039128X98000051 ↑12 Amory, J.K., Wang, C., Swerdloff, R.S., Anawalt, B.D., Matsumoto, A.M., Bremner, W.J., Walker, S.E., Haberer, L.J. and Clark, R.V. (2007). The Effect Of 5α-Reductase Inhibition With Dutasteride And Finasteride On Semen Parameters And Serum Hormones In Healthy Men. The Journal Of Clinical Endocrinology & Metabolism. 92(5), pp.1659–1665. Available at: https://doi.org/10.1210/jc.2006-2203 ↑13 Albasher, G., Bin-Jumah, M., Alfarraj, S., Al-Otibi, F., Al-Sultan, N.K., Alarifi, S., Alkahtani, S. and Al-Qahtani, W.S. (2020). Prolonged Use Of Finasteride-Induced Gonadal Sex Steroids Alterations, DNA Damage And Menstrual Bleeding In Women. Bioscience Reports. 40(2), BSR20191434. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC7007407/ ↑15 Hirshburg, J.M., Kelsey, P.A., Therrien, C.A., Gavino, A.C. and Reichenberg, J.S. (2016). Adverse Effects And Safety Of 5-Alpha Reductase Inhibitors (Finasteride, Dutasteride): A Systematic Review. Journal Of Clinical Aesthetic Dermatology. 9(7), pp.56–62. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC5023004/ ↑16 Wessells, H., Roy, J., Bannow, J., Grayhack, J., Matsumoto, A.M., Tenover, L., Herlihy, R., Fitch, W., Labasky, R., Auerbach, S., Parra, R., Rajfer, J., Culbertson, J., Lee, M., Bach, M.A. and Waldstreicher, J. (2003). Incidence And Severity Of Sexual Adverse Experiences In Finasteride And Placebo-Treated Men With Benign Prostatic Hyperplasia. Urology. 61(3), pp.579–584. Available at: https://pubmed.ncbi.nlm.nih.gov/12639651/ ↑17 Lee, S., Lee, Y.B., Choe, S.J. and Lee, W. (2019). Adverse Sexual Effects Of Treatment With Finasteride Or Dutasteride For Male Androgenetic Alopecia: A Systematic Review And Meta-Analysis. Acta Dermato-Venereologica. 99(1), pp.12–17. Available at: https://pubmed.ncbi.nlm.nih.gov/30206635/ ↑18 Corona, G., Tirabassi, G., Santi, D., Maseroli, E., Gacci, M., Dicuio, M., Sforza, A., Mannucci, E. and Maggi, M. (2017). Sexual Dysfunction In Subjects Treated With Inhibitors Of 5α-Reductase For Benign Prostatic Hyperplasia: A Comprehensive Review And Meta-Analysis. Andrology. 5(4), pp.671–678. Available at: https://doi.org/10.1111/andr.12353 ↑19 Mondaini, N., Gontero, P., Giubilei, G., Lombardi, G., Cai, T., Gavazzi, A. and Bartoletti, R. (2007). Finasteride 5 mg And Sexual Side Effects: How Many Of These Are Related To A Nocebo Phenomenon? Journal Of Sexual Medicine. 4(6), pp.1708–1712. Available at: https://pubmed.ncbi.nlm.nih.gov/17655657/ ↑20 Clapauch, R. (2011). Finasteride and Erectile Dysfunction: Fact or Fiction? Brasília Medical (Medical Experience). 48(4), pp.422–427. Available at: https://rbm.org.br/details/247/en-US/finasteride-and-erectile-dysfunction–fact-or-fiction ↑21 Erdemir, F., Harbin, A. and Hellstrom, W.J.G. (2008). 5-Alpha Reductase Inhibitors And Erectile Dysfunction: The Connection. The Journal Of Sexual Medicine. 5(12), pp.2917–2924. Available at: https://doi.org/10.1111/j.1743-6109.2008.01001.x ↑22 Anitha, B., Inamadar, A.C., Ragunatha, S. (2009). Finasteride-Its Impact on Sexual Function and Prostate Cancer. J Cutan Aesthet Surg. 2(1):12–16. Available at: https://doi.org/10.4103/0974-2077.53093 ↑23 Cai, L.Q., Fratianni, C.M., Gautier, T., Imperato-McGinley, J. (1994). Dihydrotestosterone Regulation of Semen in Male Pseudohermaphrodites with 5 Alpha-Reductase-2 Deficiency. Journal of Clinical Endocrinology & Metabolism. 79(2):409–414. Available at: https://doi.org/10.1210/jcem.79.2.8045956 ↑24 Irwig, M.S. (2014), Persistent Sexual and Nonsexual Adverse Effects of Finasteride in Younger Men. Sex Med Rev. 2(1):24–35. Available at: https://doi.org/10.1002/smrj.19 ↑25 Traish, A.M., Hassani, J., Guay, A.T., Zitzmann, M., Hansen, M.L. (2011). Adverse Side Effects of 5 Alpha-Reductase Inhibitors Therapy: Persistent Diminished Libido and Erectile Dysfunction and Depression in a Subset of Patients. Journal of Sexual Medicine. 8(3). Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC4064044/ ↑26 U.S. Food and Drug Administration, (2022), Propecia (Finasteride) Tablets Label. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020788s030lbl.pdf (Accessed: 07 January 2026) ↑27 Steiner, J.F. (1996). Clinical Pharmacokinetics and Pharmacodynamics of Finasteride. Clinical Pharmacokinetics. 30(1):16–27. Available at: https://doi.org/10.2165/00003088-199630010-00002 ↑28 Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing the Therapeutic Effects of Finasteride Gel and Tablet in Treatment of the Androgenetic Alopecia. Indian Journal of Dermatology, Venereology and Leprology. 75(1):47–51. Available at: https://pubmed.ncbi.nlm.nih.gov/19172031/ ↑29 Bhura, M. (2013). Comparative Analysis of Topical and Oral Finasteride in Androgenetic Alopecia: Impact on Hair Growth, Systemic Absorption, and Adverse Effects. Indian Journal of Basic and Applied Medical Research. 3(1):436–444. Available at: https://www.ijbamr.com/assets/images/issues/pdf/71nag2_6w0c1i_34kUVI_YI8x3j_177107.pdf (Accessed: 07 January 2026) ↑30 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., Jansat, J.M., Falqués, M., Otero, R., Tamarit, M.L., Galván, J., Tebbs, V., Massana, E., Topical Finasteride Study Group. (2022). Efficacy and Safety of Topical Finasteride Spray Solution for Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical Trial. Journal of the European Academy of Dermatology and Venereology. 36(2):286–294. Available at: https://doi.org/10.1111/jdv.17738 ↑31 Casabé, A., Roehrborn, C.G., Da Pozzo, L.F., Zepeda, S., Henderson, R.J., Sorsaburu, S., Henneges, C., Wong, D.G. & Viktrup, L. (2014). Efficacy and Safety of the Coadministration of Tadalafil Once Daily with Finasteride for 6 Months in Men with Lower Urinary Tract Symptoms and Prostatic Enlargement Secondary to Benign Prostatic Hyperplasia. Journal of Urology. 191(3), pp. 727–733. Available at: https://www.auajournals.org/doi/10.1016/j.juro.2013.09.059 Minoxidil has been a cornerstone of hair-loss treatment for decades, and the options have expanded well beyond over-the-counter 2% and 5% topicals. Low-dose oral minoxidil has become increasingly common off-label, especially for people who struggle with topical routines or don’t respond well to them.
As a result, many people find themselves re-evaluating their approach and thinking about switching to oral minoxidil. We’ll break down how the oral and topical formulations compare and outline when a switch is likely to help.
Interested in Oral Minoxidil?
Low-dose oral minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Does Minoxidil Work?
Topical and oral minoxidil both work through the same basic mechanisms. At a biological level, minoxidil alters the hair cycle by shortening the resting (telogen) phase and promoting earlier entry into the growth (anagen) phase. This increases the proportion of follicles actively producing hair at any given time. Minoxidil also enlarges miniaturized hair follicles, resulting in thicker, longer hair shafts.[1]Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: … Continue reading
Beyond hair-cycle effects, minoxidil influences several signaling and support pathways within the follicle. It increases local blood flow, activates the Wnt/β-catenin signaling pathway involved in follicular cell proliferation and differentiation, and appears to have cytoprotective and anti-inflammatory effects, in part by increasing prostaglandin E2 production.
What is the Difference Between Topical and Oral Minoxidil?
Where topical and oral minoxidil meaningfully diverge is not in what the drug does once active, but in how it becomes active and reaches the follicle.
Topical minoxidil must first penetrate the scalp barrier and then be converted into its active form within the hair follicle itself. This conversion depends on local sulfotransferase enzyme activity, which varies between individuals and even between scalp regions. As a result, exposure to active minoxidil can be highly variable, even when application is consistent.
Oral minoxidil, by contrast, is absorbed through the gastrointestinal tract and converted to its active form primarily in the liver, where sulfotransferase activity is abundant and consistent. The active drug is then delivered to hair follicles via the bloodstream, largely bypassing both the scalp barrier and variability in local enzymatic activation.
These differences in activation and delivery are what make switching between topical and oral formulations clinically meaningful.
Why Do People Switch Between Minoxidil Forms?
Switching is rarely about chasing a small theoretical advantage. Most switches happen for one of three reasons:
1. Convenience and Adherence
Topical minoxidil can work well in trials, but adherence is hard in real life. Many people struggle with twice-daily application for months, and discontinuation rates in real-world settings are high, with the most commonly cited reason being a lack of compliance (i.e., people couldn’t maintain their treatment regimen).[2]Shadi, Z. (2023). Compliance to topical minoxidil and reasons for discontinuation among patients with androgenetic alopecia. Dermatology and Therapy. 13(5). 1157–1169. Available at: … Continue reading
Topical minoxidil can also present cosmetic challenges that influence long-term adherence. Liquid solutions may leave the hair feeling greasy or stiff, create visible residue or flaking that can resemble dandruff, and interfere with hairstyling, particularly in people with longer hair or finer textures.
Oral minoxidil is simpler, usually a once-daily pill, and for many people, that alone changes outcomes.
2. Inconsistent Response to Topical Therapy
Some people use topical minoxidil correctly for long enough and still see little benefit. This can happen even when everything “looks right” on paper. The second most cited reason for discontinuing topical minoxidil was unsatisfactory results, and clinical data have shown that response rates are high in the first 3-6 months, but drop after a year or longer of use.[3]Olsen, E. A., Weiner, M. S., Amara, I. A., & DeLong, E. R. (1990). Five-year follow-up of men with androgenetic alopecia treated with topical minoxidil. *Journal of the American Academy of … Continue reading
3. Side Effects
The side effects most commonly associated with topical minoxidil are local rather than systemic, reflecting the fact that only a small fraction of the applied drug enters the bloodstream. The most frequently reported issues include scalp irritation, dryness, flaking, itching, and contact dermatitis. In many cases, these reactions are driven less by minoxidil itself and more by the vehicle used to deliver it, particularly formulations containing propylene glycol.[4]Olsen, E. A., DeLong, E. R., & Weiner, M. S. (1987). Long-term follow-up of men with male pattern baldness treated with topical minoxidil. Journal of the American Academy of Dermatology. 16(3). … Continue reading
Minoxidil is also toxic to pets, and even small exposures to topical or oral products can cause severe, sometimes fatal cardiovascular events. Because topical formulations can be harder to control, and need to be handled with care, some users may switch to oral formulations to decrease the risk of accidental exposure.[5]Tater, K. C., Gwaltney-Brant, S., & Wismer, T. (2021). Topical minoxidil exposures and toxicoses in dogs and cats: 211 cases (2001–2019). *Journal of the American Animal Hospital Association.* … Continue reading
Propylene glycol is included in many liquid formulations to enhance solubility and penetration, but it is a well-known irritant and contact sensitizer. In susceptible individuals, this can lead to erythema, scaling, burning, or eczematous dermatitis, particularly with twice-daily use.
Oral minoxidil produces a different side-effect profile because it is systemically absorbed and pharmacologically active throughout the body. At the low doses used for hair loss, it is generally well tolerated, but adverse effects occur more frequently than with topical formulations and tend to be dose-dependent.
The most common side effect is hypertrichosis, or unwanted hair growth outside the scalp, including on the face. This was reported in 15% of 1404 users in a follow-up study.[6]Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair … Continue reading
Hypertrichosis is typically more of an issue for women taking minoxidil, for whom hair on the face and body may be off-putting.
Other common side effects reported in the follow-up study included lightheadedness (1.7%), fluid retention (1.3%), tachycardia (elevated heart rate, 0.9%), and headache (0.4%). Serious cardiovascular complications are rare at the low doses used for hair loss and have been reported primarily in higher-dose antihypertensive use or in individuals with pre-existing cardiovascular disease.
Figure 1. Occurrence of adverse events in individuals taking low-dose oral minoxidil (LDOM), as reported in the FDA Adverse Event Reporting System. Adapted from Figure 1.[7]Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse … Continue reading Image used under Creative Commons License.
If you’re concerned about minoxidil side effects, check out our article on 10 ways to reduce them.
Why Oral Minoxidil Might Work Better for Some
Topical minoxidil relies heavily on follicular sulfotransferase activity (notably SULT1A1). Lower activity is associated with weaker response.[8]Pietrauszka, K., & Bergler-Czop, B. (2022). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: … Continue reading
As such, minoxidil’s “ceiling” isn’t the same for everyone: people with lower SULT1A1 activity will generally not respond as well.
Similarly, the penetration of topical minoxidil through the scalp is essential for it to function. This can depend on a range of factors, including the type of formulation, scalp health, contact time, and application technique.
Oral minoxidil bypasses these barriers to absorption. It is absorbed through the gastrointestinal tract and converted into its active form primarily in the liver, where sulfotransferase activity is abundant. Systemic activation makes exposure more uniform across the scalp and less dependent on follicular enzyme variability.
Oral Minoxidil vs Topical Minoxidil
So what does the clinical evidence tell us about the efficacy of the two approaches?
Both topical and oral minoxidil are supported by robust clinical evidence demonstrating their effectiveness in treating hair loss, including androgenic alopecia (AGA).[9]Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: … Continue reading,[10]Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global … Continue reading,[11]Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 … Continue reading,[12]Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). … Continue reading,[13]Olsen, E. A., Dunlap, F. E., Funicella, T., Koperski, J. A., Swinehart, J. M., Tschen, E. H., & Trancik, R. J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical … Continue reading,[14]Adil, A., & Godwin, M. (2017). The effectiveness of treatments for androgenetic alopecia: a systematic review and meta-analysis. *Journal of the American Academy of Dermatology.* 77(1). … Continue reading
Direct head-to-head comparisons, however, are less common.
Direct Comparisons in Men
A 2024 randomized clinical trial compared 5 mg oral minoxidil once daily vs 5% topical minoxidil twice daily for 24 weeks in men with AGA. They found no significant difference in hair outcomes overall, but oral trended higher for density and showed significantly greater improvement in photographic assessment at the crown.[15]Penha, M. A., Miot, H. A., Kasprzak, M., & Ramos, P. M. (2024). Oral minoxidil vs topical minoxidil for male androgenetic alopecia: a randomized clinical trial. JAMA Dermatology. 160(6). … Continue reading
This matches the broader trend seen across male oral minoxidil studies at 2.5-5 mg daily: more consistent, dose-dependent results with cosmetic relevance.[16]Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global … Continue reading
Direct Comparisons in Women
As we’ve already noted, doses for women tend to be lower than for men, typically 1 mg daily versus 5 mg daily, so as to avoid hypertrichosis.
The first direct comparison in women comes from a 24-week randomized, open comparative study conducted in Brazil, which evaluated oral minoxidil 1 mg once daily versus topical minoxidil 5% solution applied once daily in women aged 18–65 with pattern hair loss.[17]Ramos, P. M., Melo, D. F., Radwanski, H., Cortez de Almeida, R. F., & Miot, H. A. (2023). Female-pattern hair loss: therapeutic update. Anais Brasileiros de Dermatologia. 98. 506–519. Available … Continue reading
After 24 weeks, terminal hair density increased by 12% in the oral minoxidil group and 7.2% in the topical group. While this improvement favored oral therapy, the difference was not statistically significant.
Side-effect patterns differed in expected ways. Hypertrichosis was reported in 27% of women taking oral minoxidil, compared with 4% in the topical group.
A similar trial comparing 1mg oral minoxidil to 5% topical minoxidil also found no statistically significant differences between the two groups in hair diameter. They did, however, report improvement in photographic assessment in the topical group.[18]Asilian, A., Farmani, A., & Saber, M. (2024). Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in the improvement of androgenetic alopecia: a randomized controlled … Continue reading
Lower doses of oral minoxidil for women may reduce its efficacy compared to topical treatments, which are typically applied at similar concentrations to men (5%).
Figure 2. Improvements in average hair diameter were comparable between 5% topical solution or 1 mg/day oral minoxidil over a six-month study period. Adapted from Figure 2.[19]Asilian, A., Farmani, A., & Saber, M. (2024). Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in the improvement of androgenetic alopecia: a randomized controlled … Continue reading Image used under Creative Commons License.
What About Higher Concentrations of Topical Minoxidil?
As we’ve seen, most clinical research focuses on 5% topical minoxidil. However, in practice, many people use higher concentrations of minoxidil, with 7-8% being common, and sometimes concentrations as high as 10-15% are employed as a more aggressive treatment.
What’s more, minoxidil is often paired with tretinoin/retinoic acid, which enhances activation and penetration of the drug. While there is limited clinical data on such pairings, anecdotal evidence from our members suggests that this combination could help to optimize topical minoxidil’s efficacy.
We see that 7-8% minoxidil paired with 0.005-0.02% retinoic acid appears to outperform 5 mg oral minoxidil, even in cases of diffuse thinning.
For comprehensive information about using retinoic acid for hair loss, check out our ultimate guide.
Anecdotal evidence from our members also suggests that higher concentrations of topical minoxidil (10-15%) can generate similar results to 5 mg oral minoxidil.
Is there a clear winner?
In short, no. A 2025 meta-analysis comparing topical and oral minoxidil found no significant difference between approaches overall, which reflects how much outcomes depend on dose, population, and study design.[20]Fazal, F., Malik, B. H., Malik, H. M., Sabir, B., Mustafa, H., Ahmed, M., Abid, A., Adil, M. L., Shafi, U., & Saad, M. (2025). Can oral minoxidil be the game changer in androgenetic alopecia? A … Continue reading
It is important to note that adherence to a strict routine in trials is typically well enforced, and topical treatments are applied with consistent volumes at consistent times. This doesn’t necessarily reflect how people use topical minoxidil in the real world.
On the other hand, many users find that they can optimize a topical approach by increasing the strength or frequency of application, or by adding enhancer ingredients like tretinoin/retinoic acid. If you’re using such a combination therapy, you might, in fact, see worse results when switching to oral.
When Switching From Topical to Oral Makes the Most Sense
Switching to oral minoxidil is most rational when the limiting factor is activation or adherence, or when topical treatment isn’t delivering the results you expect.
Good Candidates for Switching to Oral Minoxidil
If systemic exposure is acceptable from a safety and tolerability standpoint, there are circumstances in which switching from an oral to a topical formulation makes sense.
Poor topical responders – If you’ve used topical minoxidil consistently for 6-12 months and can reasonably say adherence wasn’t the problem, oral is a logical next step because it bypasses follicular enzyme variability.[21]Pietrauszka, K., & Bergler-Czop, B. (2022). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: … Continue reading
Diffuse or Advanced Thinning – As larger areas of the scalp are affected, ensuring even coverage requires more time, higher product volumes, and careful technique, which many people struggle to sustain long term. Missed areas and uneven delivery are common and can limit results even when follicles remain responsive.
Oral minoxidil avoids these practical constraints by delivering the drug systemically, allowing uniform exposure across the entire scalp and making treatment easier to scale as hair loss progresses.
Convenience-driven adherence problems – When adherence is the primary issue, oral minoxidil often has a practical advantage. Topical therapy requires regular scalp application, drying time, and styling time, all of which can erode consistency over months or years of use. Missed applications are common and can reduce effectiveness. Oral minoxidil makes consistent long-term use more achievable for many people.
Scalp Inflammation – Inflammation of the scalp occurs in a high proportion of individuals with AGA, with seborrheic dermatitis being common. If you’re pairing minoxidil with inflammatory treatments like retinoic acid, this can exacerbate underlying chronic inflammation and prevent regrowth.
Scalp inflammation decreases the efficacy of topical minoxidil.[22]Whiting, D. A. (1993). Diagnostic and predictive value of horizontal sections of scalp biopsy specimens in male pattern androgenetic alopecia. *Journal of the American Academy of Dermatology.* 28(5). … Continue reading Switching to oral minoxidil, or even removing the inflammatory component from your treatment, can help improve underlying inflammation.
People Who Should Be Cautious Switching
Anyone with cardiovascular disease, kidney issues, fluid retention tendencies, low blood pressure, or unexplained palpitations should be conservative and involve a clinician before starting or escalating oral minoxidil.[23]Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse … Continue reading
When Switching From Oral to Topical Makes the Most Sense
There are also times when switching from topical oral minoxidil might be a sensible decision. These will most commonly occur when side effects have made use difficult or intolerable.
You might also consider an optimized topical approach, incorporating enhancers such as tretinoin/retinoic acid or increasing topical dosage. Anecdotal experiences of our members suggest these topical combinations can outperform low-dose oral minoxidil.
If you’’re interested in topical minoxidil for, check out our articles for men and women on the best products currently available.
Hair Shedding: Should You Be Concerned?
When people start a new hair loss treatment, shedding events are common. This includes treatments that are working! ‘Treatment-induced telogen effluvium shedding’ is the term used to describe this phenomenon, and can actually be a good predictor of whether minoxidil treatment will work down the line.[24]Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., & Fan, W. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it predict treatment efficacy? A … Continue reading
However, it’s hard to tell if shedding when you switch treatments is a positive sign, or if it’s a indicator of decreased efficacy. These cases are referred to as a ‘treatment withdrawal telogen effluvium shed’, and won’t lead to improvement in the future.
Information from our members indicates that shedding after switching from topical to oral minoxodil tends to fall into the latter camp: shedding is a result of the treatment working less well.
Be aware of shedding after switching treatments, as it could be an indicator that your new treatment isn’t working as well.
You can read more about minoxidil shedding in our comprehensive article.
How to Switch
There’s no universally validated switching protocol supported by large controlled trials. But there are sensible principles that reduce avoidable problems.
-
Avoid Abrupt Stop and Start Changes
Minoxidil works by keeping follicles in a growth-supportive environment. Abrupt discontinuation can allow hairs that were being prolonged in anagen to shed as cycles normalize.
-
Expect a Transition Period
Whether switching to oral or topical, you’re changing local concentrations and the consistency of follicular exposure. That can mean a temporary shed or plateau before stabilization, so don’t judge too early.
-
Be Patient and Track Longterm Changes
As with all hair loss treatments, consistency is key. Because minoxidil primarily influences hair-cycle timing rather than creating new follicles, benefits accrue gradually and are best assessed over 6–12 months of consistent use. Tracking progress with standardized photographs, consistent lighting, and fixed time intervals can help distinguish true treatment effects from normal cycle-related variation.
If you experience severe adverse effects, you should discontinue treatment and speak to a clinician.
Final Thoughts
Switching between topical and oral minoxidil should be a deliberate and strategic decision guided by potential reasons your current approach isn’t working. The evidence consistently shows that both topical and oral minoxidil can be effective, but their real-world performance is shaped by adherence, activation, dose, side effects, and how well the treatment fits into your life.
For people with limited topical response, inconsistent application, or large areas of thinning, oral minoxidil can provide more uniform exposure and better long-term consistency. However, real-world experience and member data suggest that higher-strength topical formulations (7–8%), especially when paired with penetration or activation enhancers like retinoic acid, often produce stronger results.
In some cases, these optimized topical approaches appear to match or even outperform low-dose oral minoxidil. For people who tolerate topical therapy well and can maintain consistency, increasing topical strength may be a more effective next step than switching to oral treatment.
Ultimately, switching treatments always carries trade-offs and some risk. The safest path forward is deliberate, not reactive: avoid abrupt changes, allow sufficient time to assess outcomes, and monitor both hair and systemic responses carefully.
References[+]
References ↑1 Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑2 Shadi, Z. (2023). Compliance to topical minoxidil and reasons for discontinuation among patients with androgenetic alopecia. Dermatology and Therapy. 13(5). 1157–1169. Available at: https://doi.org/10.1007/s13555-023-00919-x ↑3 Olsen, E. A., Weiner, M. S., Amara, I. A., & DeLong, E. R. (1990). Five-year follow-up of men with androgenetic alopecia treated with topical minoxidil. *Journal of the American Academy of Dermatology.* 22(4). 643–646. Available at: https://doi.org/10.1016/0190-9622(90)70089-Z ↑4 Olsen, E. A., DeLong, E. R., & Weiner, M. S. (1987). Long-term follow-up of men with male pattern baldness treated with topical minoxidil. Journal of the American Academy of Dermatology. 16(3). 688–695. Available at: https://doi.org/10.1016/S0190-9622(87)70089-9 ↑5 Tater, K. C., Gwaltney-Brant, S., & Wismer, T. (2021). Topical minoxidil exposures and toxicoses in dogs and cats: 211 cases (2001–2019). *Journal of the American Animal Hospital Association.* 57(5). 225–231. Available at: https://doi.org/10.5326/jaaha-ms-7154 ↑6 Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair loss: a multicenter study of 1404 patients. Journal of the American Academy of Dermatology. 84(6). 1644–1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑7 Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse Event Reporting System (FAERS) with a focus on pericardial effusions. *Journal of Cosmetic Dermatology.* 24(1). e16574. Available at: https://doi.org/10.1111/jocd.16574 ↑8, ↑21 Pietrauszka, K., & Bergler-Czop, B. (2022). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: a review. Advances in Dermatology and Allergology / Postępy Dermatologii i Alergologii. 39(3). 472–478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑9 Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: https://www.researchgate.net/profile/Suparuj-Lueangarun/publication/319006716_Efficacy_and_safety_of_oral_minoxidil_5_mg_daily_during_24-week_treatment_in_male_androgenetic_alopecia/links/5b46f1690f7e9b4637cde38b/Efficacy-and-safety-of-oral-minoxidil-5-mg-daily-during-24-week-treatment-in-male-androgenetic-alopecia.pdf ↑10 Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global photographic assessment. *Dermatology and Therapy.* 10(6). 1345–1357. Available at: https://doi.org/10.1007/s13555-020-00448-x ↑11 Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 mg versus 1 mg. 396–399. Available at: https://doi.org/10.1016/j.jaad.2022.01.017 ↑12 Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). 104–109. Available at: https://doi.org/10.1111/ijd.13838 ↑13 Olsen, E. A., Dunlap, F. E., Funicella, T., Koperski, J. A., Swinehart, J. M., Tschen, E. H., & Trancik, R. J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical minoxidil and placebo in the treatment of androgenetic alopecia in men. *Journal of the American Academy of Dermatology.* 47(3). 377–385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑14 Adil, A., & Godwin, M. (2017). The effectiveness of treatments for androgenetic alopecia: a systematic review and meta-analysis. *Journal of the American Academy of Dermatology.* 77(1). 136–141. Available at: https://doi.org/10.1016/j.jaad.2017.02.054 ↑15 Penha, M. A., Miot, H. A., Kasprzak, M., & Ramos, P. M. (2024). Oral minoxidil vs topical minoxidil for male androgenetic alopecia: a randomized clinical trial. JAMA Dermatology. 160(6). 600–605. Available at: https://doi:10.1001/jamadermatol.2024.0284 ↑16 Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global photographic assessment. Dermatology and Therapy. 10(6). 1345–1357. Available at: https://doi.org/10.1007/s13555-020-00448-x ↑17 Ramos, P. M., Melo, D. F., Radwanski, H., Cortez de Almeida, R. F., & Miot, H. A. (2023). Female-pattern hair loss: therapeutic update. Anais Brasileiros de Dermatologia. 98. 506–519. Available at: https://doi.org/10.1016/j.abd.2022.09.006 ↑18 Asilian, A., Farmani, A., & Saber, M. (2024). Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in the improvement of androgenetic alopecia: a randomized controlled trial. Journal of Cosmetic Dermatology. 23(3). 949–957. Available at: https://doi.org/10.1111/jocd.16086 ↑19 Asilian, A., Farmani, A., & Saber, M. (2024). Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in the improvement of androgenetic alopecia: a randomized controlled trial. *Journal of Cosmetic Dermatology.* 23(3). 949–957. Available at: https://doi.org/10.1111/jocd.16086 ↑20 Fazal, F., Malik, B. H., Malik, H. M., Sabir, B., Mustafa, H., Ahmed, M., Abid, A., Adil, M. L., Shafi, U., & Saad, M. (2025). Can oral minoxidil be the game changer in androgenetic alopecia? A comprehensive review and meta-analysis comparing topical and oral minoxidil for treating androgenetic alopecia. Skin Health and Disease. vzaf009. Available at: https://doi.org/10.1093/skinhd/vzaf009 ↑22 Whiting, D. A. (1993). Diagnostic and predictive value of horizontal sections of scalp biopsy specimens in male pattern androgenetic alopecia. *Journal of the American Academy of Dermatology.* 28(5). 755–763. Available at: https://doi.org/10.1016/0190-9622(93)70106-4 ↑23 Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse Event Reporting System (FAERS) with a focus on pericardial effusions. Journal of Cosmetic Dermatology. 24(1). e16574. Available at: https://doi.org/10.1111/jocd.16574 ↑24 Bi, L., Kan, H., Wang, J., Ding, Y., Huang, Y., Wang, C., & Fan, W. (2025). Whether the transient hair shedding phase exist after minoxidil treatment and does it predict treatment efficacy? A retrospective study in androgenetic alopecia patients. *Journal of Dermatological Treatment.* 36(1). 2480739. Available at: https://doi.org/10.1080/09546634.2025.2480739 Spironolactone is a potassium-sparing diuretic with anti-androgen properties that has become increasingly popular for treating hair loss, particularly female pattern hair loss. By blocking androgen receptors, spironolactone can slow hair shedding and promote regrowth in hormonally driven alopecia.
Despite its benefits, many patients worry about potential side effects, especially whether spironolactone leads to weight gain. Anecdotally, several patients have reported weight gain when they begin taking spironolactone. In this article, we examine that concern by reviewing typical dosing for hair loss, summarizing clinical evidence on weight changes, explaining possible biological mechanisms, and outlining practical strategies to minimize unwanted weight or fluid-related effects during treatment.
What is Spironolactone?
Spironolactone is a prescription medication classified as a potassium-sparing diuretic. In other words, it is a pill that helps the body get rid of excess fluid like water and salt by increasing urination, but it prevents excessive loss of the vital mineral potassium.
It mediates this function by acting as an aldosterone antagonist. Spironolactone blocks the binding of the hormone aldosterone to its receptors, an action that regulates the body’s sodium and potassium levels.
This is why spironolactone is commonly prescribed for hypertension (high blood pressure), heart failure, and edema (swelling). By blocking aldosterone, spironolactone reduces water and salt retention and helps lower blood pressure, strain on the heart, and swelling.[1]Carone, L., Oxberry, S.G., Twycross, R., Charlesworth, S., Mihalyo, M., Wilcock, A. (2017). Spironolactone. Journal of Pain and Symptom Management. 53(2). 288–292. Available at: … Continue reading
How Does Spironolactone Work For Hair Loss?
Aside from blocking aldosterone receptors, spironolactone is also capable of binding to androgen receptors. These receptors typically bind to male androgen hormones like testosterone and dihydrotestosterone (DHT).[2]Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English … Continue reading
Levels of DHT above average are a common symptom in those with androgenic alopecia. In the body, the binding of DHT to androgen receptors stimulates a range of effects, including hair follicle miniaturization. That is, hairs become thinner, shorter, and weaker. This is because DHT reduces the length of the growth phase (the anagen phase) of hair follicles, while also increasing the length of the non-growing phase (the telogen phase).[3]Ustuner, E. T. (2013). Cause of Androgenic Alopecia: Crux of the Matter. Plast Reconstr Surg Glob Open. 1(7). e64. Available at: https://doi.org/10.1097/GOX.0000000000000005,[4]Sekhavat, H., Bar Yehuda, S., Asotra, S. (2025). Using the Mechanisms of Action Involved in the Pathogenesis of Androgenetic Alopecia to Treat Hair Loss. Int J Mol Sci. 26(21). 10712. Available at: … Continue reading
Thus, by binding androgen receptors, spironolactone can prevent the binding of DHT and, in this way, effectively “blocks” DHT to prevent hair loss.[5]Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English … Continue reading
Side effects
Because spironolactone blocks male hormones, it can lead to unwanted feminization in men, such as gynecomastia (enlarged breasts), reduced facial or body hair, and redistribution of body fat. It may also lead to a loss of libido and other sexual dysfunctions.[6]Carone, L., Oxberry, S.G., Twycross, R., Charlesworth, S., Mihalyo, M., Wilcock, A. (2017). Spironolactone. Journal of Pain and Symptom Management. 53(2). 288–292. Available at: … Continue reading,[7]Haynes, B.A., Mookadam, F. (2009). Male Gynecomastia. Mayo Clinic Proceedings. 84(8). 672. Available at: https://doi.org/10.4065/84.8.672
For this reason, it is usually prescribed to women but not to men for the purpose of treating non-life-threatening conditions, such as androgenic alopecia.
Women prescribed spironolactone may experience some other side effects. For example, 15-30% of women report irregular menstruation, while fewer than 5% report breast tenderness, reduced libido, nausea, headache, and fatigue.[8]Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English … Continue reading
The Science: Hair Growth Benefits
Oral spironolactone as a hair loss treatment has been studied across concentrations from 25 mg to 200 mg per day. Collectively, these trials demonstrate that spironolactone can provide meaningful improvements in hair growth in those with androgenic alopecia; however, not all trials have the scientific robustness to make clear conclusions on its efficacy.
Low doses (25-50 mg/day)
A pilot study of 100 women taking 25 mg spironolactone with 0.25 mg minoxidil for 12 months nearly halved hair shedding and substantially improved hair loss severity. However, the absence of a placebo control group limits interpretation, meaning the true effectiveness of low-dose spironolactone within this combination remains uncertain.[9]Sinclair, R.D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. International Journal of Dermatology. 57(1). … Continue reading
Medium doses (75-100 mg/day)
A 2025 randomized, placebo-controlled pilot study over 24 weeks evaluated 100 mg daily spironolactone plus 3% topical minoxidil versus placebo plus minoxidil in women with androgenic alopecia. Both groups experienced significant increases in hair density and diameter from baseline. The spironolactone group showed greater increases in terminal hairs and hair diameter than placebo, though this difference was not statistically significant.[10]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading
Figure 2: Images of hair density at baseline and following 24 weeks of daily treatment with either 100 mg spironolactone and 3% topical minoxidil or a placebo and topical 3% minoxidil. Adapted from Figure 3.[11]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading Image obtained in line with the Creative Commons License.
Earlier research from 1991 found that women treated with 75–100 mg spironolactone did not have a significant change in hair density after 12 months, while untreated women experienced decreases, suggesting spironolactone may prevent further loss rather than promote regrowth.[12]Rushton, D., Futterweit, W., Kingsley, D., Kingsley, P., Norris, M.J. (1990). Quantitative assessment of spironolactone treatment in women with diffuse androgen-dependent alopecia. Journal of the … Continue reading
Combination therapies appear promising. A larger study comparing 5% minoxidil alone, minoxidil with 100 mg spironolactone, and minoxidil with microneedling found all groups had increased hair density at 24 weeks. The minoxidil plus spironolactone group had greater gains than minoxidil alone, though less than the microneedling group, indicating that adding spironolactone may enhance treatment effects.[13]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading
Figure 4: Scalp images of patients treated with 5% minoxidil alone (A1-A3), 5% minoxidil and spironolactone (B1-B3), or 5% minoxidil and microneedling (C1-C3) at baseline, week 12, and week 24. Adapted from Figure 5.[14]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading Image obtained in line with the Creative Commons License.
High doses (150-200 mg/day)
Early studies suggested high doses of spironolactone might benefit people with androgenic alopecia, but the supporting evidence is limited and inconsistent.
A very small trial reported large improvements in hair growth and reductions in hair loss after six months of 200 mg daily treatment; however, this study involved only four patients and lacked a control group, severely limiting its reliability.[15]Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: … Continue reading
Larger research has failed to confirm such strong effects. In a study of 80 women comparing spironolactone with another anti-androgen, no significant difference was found between treatments. Overall, only 44% of participants experienced regrowth, while the same proportion saw no clear change and 12% continued to lose hair.[16]Sinclair, R., Wewerinke, M., Jolley, D. (2005). Treatment of female pattern hair loss with oral antiandrogens. British Journal of Dermatology. 152(3). 466–473. Available at: … Continue reading
Indeed, case evidence suggests that spironolactone alone may not provide long-term benefits. A 53-year old woman with androgenic alopecia was treated with 200 mg spironolactone daily. Hair regrowth was evident after 12 months, but regrowth was not sustained after 24 months. Adding topical minoxidil alongside oral spironolactone to a treatment routine appeared to restore and maintain regrowth, indicating that combination therapy may be more effective than spironolactone on its own.[17]Hoedemaker, C., Van Egmond, S., Sinclair, R. (2007). Treatment of female pattern hair loss with a combination of spironolactone and minoxidil. Australasian Journal of Dermatology. 48(1). 43–45. … Continue reading
Is Spironolactone Effective for Hair Loss?
Evidence does not support spironolactone alone as an effective hair-loss treatment, especially at higher doses. Medium doses around 100 mg/day may slow further loss, but regrowth is poorly supported. Across all doses, spironolactone works best in combination therapies, particularly with minoxidil, where sustained regrowth is more consistently observed.
Does Spironolactone Have Any Mechanism That Could Cause Weight Gain?
There is no plausible biological mechanism by which spironolactone would cause true weight gain (i.e., fat accumulation). Typically, drugs that cause weight gain will cause one or more specific effects that can lead to fat-gain.[18]Verhaegen, A.A., Van Gaal, L.F. (2000). Drugs That Affect Body Weight, Body Fat Distribution, and Metabolism. Endotext. MDText.com, Inc. Available at: http://www.ncbi.nlm.nih.gov/books/NBK537590/ These include:
- Increased appetite
- Changed insulin resistance
- Slowdown of metabolism
- Increased cortisol
However, spironolactone has no known interactions that would lead to these effects. In fact, because spironolactone blocks aldosterone, it reduces sodium and water retention in the blood and can cause mild weight loss due to its diuretic effect.
Spironolactone and Weight Gain: What Do We See from Clinical Evidence?
Many clinical studies assessing the effectiveness of spironolactone for hair loss or other medical conditions also report side effects. As doses increase, more side effects are likely to occur, but is weight gain one of them?
Low doses (25-50 mg/day)
In the pilot study by Sinclair in 2018, side effects were reported in eight women, but they were not related to weight gain.[19]Sinclair, R.D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. International Journal of Dermatology. 57(1). … Continue reading
Medium doses (75-100 mg/day)
The trial by Werachattawatchai and colleagues in 2025 reported menstrual irregularities in 37.5% of women but no mention of weight gain.[20]Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, … Continue reading Similarly, in the study by Rushton and colleagues in 1991, some women reported changes to their menstrual cycles, but there was again no mention of weight gain.[21]Rushton, D., Futterweit, W., Kingsley, D., Kingsley, P., Norris, M.J. (1990). Quantitative assessment of spironolactone treatment in women with diffuse androgen-dependent alopecia. Journal of the … Continue reading
The trial by Liang and colleagues in 2022 also had no reports of weight gain, but the spironolactone group had the most adverse effects reported. Menstrual disorder, hyperkalemia, and edema of the limbs occurred only within groups receiving spironolactone treatment. While it may appear paradoxical as spironolactone is designed to treat edema, spironolactone can also cause edema because it promotes shifts in fluids from the bloodstream to the tissues. [22]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading
Figure 5: Side effects recorded across patients receiving minoxidil (MN), minoxidil and spironolactone (SPT), or minoxidil and microneedling (MN) treatment. Adapted from Table 5.[23]Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A … Continue reading Image obtained in line with the Creative Commons License.
When used to treat hormonal disorders, a prospective clinical trial with women with polycystic ovary syndrome showed that intake of 100 mg spironolactone per day was not associated with weight gain.[24]Zulian, E., Sartorato, P., Benedini, S., et al. (2005). Spironolactone in the treatment of polycystic ovary syndrome: Effects on clinical features, insulin sensitivity and lipid profile. Journal of … Continue reading
High doses (150-200 mg/day)
Adamopoulos and colleagues reported that participants experienced no adverse side effects from taking 200 mg spironolactone per day.[25]Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: … Continue reading In the study by Sinclair in 2005 and case study by Hoedemaker and colleagues, adverse reactions, including weight gain, were not noted in the report, which suggests side effects were not measured.[26]Sinclair, R., Wewerinke, M., Jolley, D. (2005). Treatment of female pattern hair loss with oral antiandrogens. British Journal of Dermatology. 152(3). 466–473. Available at: … Continue reading,[27]Hoedemaker, C., Van Egmond, S., Sinclair, R. (2007). Treatment of female pattern hair loss with a combination of spironolactone and minoxidil. Australasian Journal of Dermatology. 48(1). 43–45. … Continue reading
In applications outside of hair loss, spironolactone has shown to actually cause weight loss. Administration of up to 200 mg spironolactone to patients with mineralocorticoid-induced hypertension resulted in a reversal of mineralocorticoid-induced abnormalities, including increased body weight.[28]Nicholls, M.G., Ramsay, L.E., Boddy, K., Fraser, R., Morton, J.J., Robertson, J.I.S. (1979). Mineralocorticoid-induced blood pressure, electrolyte, and hormone changes, and reversal with … Continue reading
Why Some People Think Spironolactone Causes Weight Gain
Even though spironolactone has no known mechanism that could cause weight gain, and has actually been shown to induce weight loss, some individuals taking spironolactone have reported weight gain anecdotally. Why could this be?
-
Water Retention
Spironolactone helps your body get rid of salt and water. It works by making the kidneys remove sodium and water from the bloodstream, which lowers the amount of fluid circulating in the vessels. That helps when edema is caused by too much circulating volume, like in heart failure or certain hormone disorders.
However, spironolactone can also relax veins and lower blood pressure. Edema can be caused by these exact symptoms. Thus, spironolactone can sometimes make fluid move into your tissues instead of staying in your bloodstream, causing swelling. This is why some studies report edema as a side effect.
Those in spironolactone may experience this water retention and swelling, causing them to feel that they have gained weight as a result of taking this medication.
-
Multiple Medications
It is likely that the individual is taking multiple medications. Spironolactone is best used in combination with other medications, and is safe to use with anti-depressants or birth control, both of which have clinically shown to cause weight gain.[29]Gafoor, R., Booth, H.P., Gulliford, M.C. (2018). Antidepressant utilisation and incidence of weight gain during 10 years’ follow-up: population based cohort study. BMJ. k1951. Available at: … Continue reading,[30]Lopez, L.M., Ramesh, S., Chen, M., et al. (2016). Progestin-only contraceptives: effects on weight. Cochrane Database of Systematic Reviews. 2016(8). CD008815. Available at: … Continue reading. Research summarising population characteristics of those prescribed spironolactone found that 22% had been using hormonal contraceptives, suggesting an alternative reason than spironolactone for reported weight gain.[31]Burns, L.J., Souza, B.D., Flynn, E., Hagigeorges, D., Senna, M.M. (2020). Spironolactone for treatment of female pattern hair loss. Journal of the American Academy of Dermatology. 83(1). 276–278. … Continue reading
-
Menopause
Weight gain is also one of the most common side effects of perimenopause, menopause, and postmenopause, affecting over 40% of women.[32]Knight, M.G., Anekwe, C., Washington, K., Akam, E.Y., Wang, E., Stanford, F.C. (2021). Weight Regulation in Menopause. Menopause. 28(8). 960–965. Available at: … Continue reading,[33]Davis, S.R., Castelo-Branco, C., Chedraui, P., et al. (2012). Understanding weight gain at menopause. Climacteric. 15(5). 419–429. Available at: https://doi.org/10.3109/13697137.2012.707385 Population characteristics of those prescribed spironolactone found that 51% were postmenopausal.[34]Burns, L.J., Souza, B.D., Flynn, E., Hagigeorges, D., Senna, M.M. (2020). Spironolactone for treatment of female pattern hair loss. Journal of the American Academy of Dermatology. 83(1). 276–278. … Continue reading Weight gain may therefore be related to life stage rather than spironolactone itself.
-
Other Side Effects
Many medications including spironolactone itself may cause symptoms such as fatigue. Those experiencing fatigue would be less inclined to stick to their active routines. Inactivity or changes to diet as a result could lead to weight gain.
-
Stress
We must also consider that external factors like stress can cause hormonal changes that influence weight. It is reasonable to assume that those prescribed spironolactone may be experiencing hypertension, heart failure, edema, or hair loss. Ongoing conditions like these could contribute to stress and hormonal changes that ultimately lead to weight gain.[35]Kyrou, I., Tsigos, C. (2009). Stress hormones: physiological stress and regulation of metabolism. Current Opinion in Pharmacology. 9(6). 787–793. Available at: … Continue reading
Managing Weight Fluctuations If They Occur
Because spironolactone has a diuretic effect, you may notice some temporary weight loss due to fluid reduction. This is not fat loss, but if you wish to maintain your weight, it’s important to monitor your body closely and make gradual lifestyle changes. The same can be advised if you are also experiencing weight gain for any reason.
To manage weight fluctuations if they occur, consider the following:
- Record keeping: Keep a consistent record of your weight to track change and distinguish between fluid-related changes and true weight changes
- Diet: Maintain a balanced diet rich in whole grains, lean proteins, healthy fats, fruits, and vegetables to stabilize weight.
- Hydration: Fluid shifts from diuretics can sometimes trigger dehydration, which may influence appetite and energy levels.
- Physical activity: Support muscle maintenance and overall metabolism by engaging in regular physical activity
- Take time: Weight changes from spironolactone are often temporary as your body gets used to the medication
If you feel your medication is causing your weight to change substantially, speak with your clinician, as it may indicate problems with your dosage, interactions with other medications, or additional health conditions.
Should I Take Spironolactone If I’m Concerned About Weight Gain?
There are some side effects of spironolactone, but weight gain is not one of them. Clinical evidence shows that daily usage of spironolactone at any dose is not known to cause weight gain.
If you’ve been prescribed spironolactone, take it with peace of mind that it will not cause permanent changes to your weight.
Final Verdict
Spironolactone isn’t a “fat-gain” drug, even though it can sometimes get that reputation online. Any early weight changes are usually from temporary fluid shifts, and many people actually notice weight loss over time due to the diuretic activity of this treatment.
Benefits to hair loss tend to appear at moderate doses like 50-100 mg per day and are most substantial when combined with common hair loss treatments like minoxidil. True weight gain has not been reported clinically, and anecdotal evidence from users is likely the result of other medications, age, activity level, or hormonal shifts. That being said, pairing treatment with sensible eating, hydration, and regular activity can help keep any weight changes in check, so the only thing you see growing is your hair.
References[+]
References ↑1, ↑6 Carone, L., Oxberry, S.G., Twycross, R., Charlesworth, S., Mihalyo, M., Wilcock, A. (2017). Spironolactone. Journal of Pain and Symptom Management. 53(2). 288–292. Available at: https://doi.org/10.1016/j.jpainsymman.2016.12.320 ↑2, ↑5, ↑8 Vargas-Mora, P., Morgado-Carrasco, D. (2020). Spironolactone in Dermatology: Uses in Acne, Hidradenitis Suppurativa, Female Pattern Hair Loss, and Hirsutism. Actas Dermo-Sifiliográficas (English Edition). 111(8). 639–649. Available at: https://doi.org/10.1016/j.adengl.2020.03.015 ↑3 Ustuner, E. T. (2013). Cause of Androgenic Alopecia: Crux of the Matter. Plast Reconstr Surg Glob Open. 1(7). e64. Available at: https://doi.org/10.1097/GOX.0000000000000005 ↑4 Sekhavat, H., Bar Yehuda, S., Asotra, S. (2025). Using the Mechanisms of Action Involved in the Pathogenesis of Androgenetic Alopecia to Treat Hair Loss. Int J Mol Sci. 26(21). 10712. Available at: https://doi.org/10.3390/ijms262110712 ↑7 Haynes, B.A., Mookadam, F. (2009). Male Gynecomastia. Mayo Clinic Proceedings. 84(8). 672. Available at: https://doi.org/10.4065/84.8.672 ↑9, ↑19 Sinclair, R.D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. International Journal of Dermatology. 57(1). 104–109. Available at: https://doi.org/10.1111/ijd.13838 ↑10, ↑20 Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, double-blind, placebo-controlled, parallel-group pilot study. International Journal of Women’s Dermatology. 11(3). e227. Available at: https://doi.org/10.1097/JW9.0000000000000227 ↑11 Werachattawatchai, P., Khunkhet, S., Harnchoowong, S., Lertphanichkul, C. (2025). Efficacy and safety of oral spironolactone for female pattern hair loss in premenopausal women: a randomized, double-blind, placebo-controlled, parallel-group pilot study. International Journal of Women’s Dermatology. 11(3). e227. Available at: https://doi.org/10.1097/JW9.0000000000000227 ↑12, ↑21 Rushton, D., Futterweit, W., Kingsley, D., Kingsley, P., Norris, M.J. (1990). Quantitative assessment of spironolactone treatment in women with diffuse androgen-dependent alopecia. Journal of the Society of Cosmetic Chemists. 42. 317–325. Available at: https://www.researchgate.net/publication/285856638_Quantitative_assessment_of_spironolactone_treatment_in_women_with_diffuse_androgen-dependent_alopecia ↑13, ↑22 Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A Prospective, Single-Center, Parallel-Group, Evaluator Blinded, Randomized Trial. Frontiers in Medicine. 9. Article 905140. Available at: https://doi.org/10.3389/fmed.2022.905140 ↑14, ↑23 Liang, X., Chang, Y., Wu, H., et al. (2022). Efficacy and Safety of 5% Minoxidil Alone, Minoxidil Plus Oral Spironolactone, and Minoxidil Plus Microneedling on Female Pattern Hair Loss: A Prospective, Single-Center, Parallel-Group, Evaluator Blinded, Randomized Trial. Frontiers in Medicine. 9. Article 905140. Available at: https://doi.org/10.3389/fmed.2022.905140 ↑15, ↑25 Adamopoulos, D.A., Karamertzanis, M., Nicopoulou, S., Gregoriou, A. (1997). Beneficial effect of spironolactone on androgenic alopecia. Clinical Endocrinology. 47(6). 759–760. Available at: https://doi.org/10.1046/j.1365-2265.1997.3761162.x ↑16, ↑26 Sinclair, R., Wewerinke, M., Jolley, D. (2005). Treatment of female pattern hair loss with oral antiandrogens. British Journal of Dermatology. 152(3). 466–473. Available at: https://doi.org/10.1111/j.1365-2133.2005.06218.x ↑17, ↑27 Hoedemaker, C., Van Egmond, S., Sinclair, R. (2007). Treatment of female pattern hair loss with a combination of spironolactone and minoxidil. Australasian Journal of Dermatology. 48(1). 43–45. Available at: https://doi.org/10.1111/j.1440-0960.2007.00332.x ↑18 Verhaegen, A.A., Van Gaal, L.F. (2000). Drugs That Affect Body Weight, Body Fat Distribution, and Metabolism. Endotext. MDText.com, Inc. Available at: http://www.ncbi.nlm.nih.gov/books/NBK537590/ ↑24 Zulian, E., Sartorato, P., Benedini, S., et al. (2005). Spironolactone in the treatment of polycystic ovary syndrome: Effects on clinical features, insulin sensitivity and lipid profile. Journal of Endocrinological Investigation. 28(3). 49–53. Available at: https://doi.org/10.1007/BF03345529 ↑28 Nicholls, M.G., Ramsay, L.E., Boddy, K., Fraser, R., Morton, J.J., Robertson, J.I.S. (1979). Mineralocorticoid-induced blood pressure, electrolyte, and hormone changes, and reversal with spironolactone, in healthy men. Metabolism. 28(5). 584–593. Available at: https://doi.org/10.1016/0026-0495(79)90201-4 ↑29 Gafoor, R., Booth, H.P., Gulliford, M.C. (2018). Antidepressant utilisation and incidence of weight gain during 10 years’ follow-up: population based cohort study. BMJ. k1951. Available at: https://doi.org/10.1136/bmj.k1951 ↑30 Lopez, L.M., Ramesh, S., Chen, M., et al. (2016). Progestin-only contraceptives: effects on weight. Cochrane Database of Systematic Reviews. 2016(8). CD008815. Available at: https://doi.org/10.1002/14651858.CD008815.pub4 ↑31 Burns, L.J., Souza, B.D., Flynn, E., Hagigeorges, D., Senna, M.M. (2020). Spironolactone for treatment of female pattern hair loss. Journal of the American Academy of Dermatology. 83(1). 276–278. Available at: https://doi.org/10.1016/j.jaad.2020.03.087 ↑32 Knight, M.G., Anekwe, C., Washington, K., Akam, E.Y., Wang, E., Stanford, F.C. (2021). Weight Regulation in Menopause. Menopause. 28(8). 960–965. Available at: https://doi.org/10.1097/GME.0000000000001792 ↑33 Davis, S.R., Castelo-Branco, C., Chedraui, P., et al. (2012). Understanding weight gain at menopause. Climacteric. 15(5). 419–429. Available at: https://doi.org/10.3109/13697137.2012.707385 ↑34 Burns, L.J., Souza, B.D., Flynn, E., Hagigeorges, D., Senna, M.M. (2020). Spironolactone for treatment of female pattern hair loss. Journal of the American Academy of Dermatology. 83(1). 276–278. Available at: https://doi.org/10.1016/j.jaad.2020.03.087 ↑35 Kyrou, I., Tsigos, C. (2009). Stress hormones: physiological stress and regulation of metabolism. Current Opinion in Pharmacology. 9(6). 787–793. Available at: https://doi.org/10.1016/j.coph.2009.08.007 Many people are interested in RU58841 after hearing anecdotes about it working well for hair regrowth. Even though RU58841 is classified as for “research purposes only,” you can find promotion of RU58841 on many YouTube channels. It is increasingly used as a preventive measure against hair loss, especially in the bodybuilding community and among individuals who cannot tolerate traditional treatments like finasteride due to side effects. Some people have demonstrated incredible results with no side effects. While others have reported no results and side effects ranging from brain fog to severe chest pain.
Despite its reputation, RU58841 is still a clinical mystery. No one knows why the human clinical trials were never published, and why the research chemical was ultimately abandoned. This article explores preclinical and clinical data on RU58841, presents our perspectives, and provides information we believe is rarely discussed about this research chemical and its potential issues.
What Is RU58841 and How Does It Work?
RU58841 is a topical androgen receptor antagonist developed in the mid-1990’s to combat androgenic alopecia by preventing DHT from having its effects.[1]Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, … Continue reading
As an androgen receptor antagonist, RU58841 blocks the “landing pads” (i.e., androgen receptors) on cell surfaces for the hormone DHT, preventing DHT from exerting its effects. If DHT cannot bind to androgen receptors in balding-prone hair follicles, it cannot induce the cascade of events that leads to hair follicle miniaturization, which is the defining characteristic of androgenic alopecia.[2]Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. In: StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/
It’s important to note that androgen receptor antagonists like RU58841 work differently from drugs like finasteride or dutasteride that inhibit the enzyme that turns testosterone into DHT (reducing DHT levels overall). Androgen receptor antagonists block the “landing pads” on the cell surfaces, preventing DHT from binding. The body can still freely produce DHT, but in the presence of androgen receptor antagonists, DHT cannot bind to hair follicle sites and have its negative effects.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Follow this link to our article here to learn more about the side effects of finasteride.
The goal when developing RU58841 was to design a drug that blocked androgen receptors locally (i.e., in the scalp). With hopes of preventing its binding to androgen receptors everywhere else in the body (like with oral spironolactone), therefore preventing interference with androgen production in places like the testes, ovaries, or adrenal glands. In fact, one of the premises behind RU58841 was to simply avoid side effects that sometimes come with drugs like finasteride and dutasteride.
Figure 1. Structure of RU58841.[3]Wikipedia, (26 December 2018), RU-58841. Available at: https://en.wikipedia.org/wiki/RU-58841#/media/File:RU-58841_structure.svg (Accessed: 02 January 2026) Image in the Public Domain.
What the Evidence Actually Shows
Preclinical studies show promising results for RU58841. Yet despite tens of millions of dollars spent on RU58841’s human clinical trials, little information from those studies has been made public.[4]ISRCTN Registry, (no date), ISRCTN49873657. Available at: https://www.isrctn.com/ISRCTN49873657 (Accessed: 02 January 2026),[5]ISRCTN Registry, (no date), ISRCTN71083772. Available at: https://www.isrctn.com/ISRCTN71083772 (Accessed: 02 January 2026)
Soon after these clinical studies were completed, the drug’s research and development were discontinued, with financial concerns cited as the reason for halting RU58841’s progress. It was stated that with more money, development could continue. However, further development never occurred, and decades later (after the drug’s patents expired), companies began selling RU58841 for “research purposes only,” citing its initial, and small, clinical trials showing favorable results for male pattern hair loss.
Let’s break down what we actually know about RU58841 from preclinical and clinical research.
Evidence From Preclinical Data
Preclinical studies suggest RU58841 works as intended and may be effective for blocking androgen activity. But we must remember that they cannot confirm long-term human safety, and they do not guarantee purely local action in real-world conditions (where dose, formulation, frequency of application, and individual absorption vary).
How Strong Is RU58841 Compared to Natural Androgens?
RU58841 was designed as a nonsteroidal androgen-receptor antagonist and contains structural features (notably perfluoroalkyl and nitrile groups) that are associated with strong androgen-receptor binding.
Early receptor-binding experiments measuring equilibrium association constants (Ka) demonstrated that RU58841 binds the androgen receptor with affinity equal to or greater than testosterone in several species and skin-relevant tissues, including the hamster flank organ and the human receptor.[6]Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, … Continue reading This is positive as it supports the premise that RU58841 can effectively compete with natural androgens at target sites.
When compared with other nonsteroidal antiandrogens such as flutamide, RU58841 was reported to have approximately 30x higher receptor affinity, a key reason it was pursued as a best-in-class topical antiandrogen candidate.[7]Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, … Continue reading
But potency works both ways. Strong androgen receptor binding makes RU58841 mechanistically credible for scalp blockade. However, it also raises the stakes if any meaningful amount escapes the scalp, accumulates, or produces systemically active metabolites.
Does RU58841 Stay Local in the Skin?
In an early study using an intact hamster flank-organ model, topical RU58841 showed strong local antiandrogen activity with minimal evidence of systemic spillover at low doses.[8]Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, … Continue reading When applied topically at doses up to 100 µg/animal, RU58841 reduced flank-organ area in a dose-dependent manner while showing no effect on the opposite flank organ, no meaningful change in serum testosterone, and no detectable antiandrogenic effects on sex organs (e.g., prostate/seminal vesicles).[9]Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, … Continue reading These findings supported the premise that RU58841 can act locally within a certain exposure window.
Are the Effects of RU58841 Reversible?
In a separate hamster ear model focused on sebaceous gland size, topical RU58841 (10 µg) reduced sebaceous gland volume by roughly 60% in the treated ear, with no measurable effect on the untreated ear.[10]Matias, J.R., Gaillard, M. (1995). Local Inhibition Of Sebaceous Gland Growth By Topically Applied RU 58841. Annals of the New York Academy of Sciences. 761. 56-65. Available at: … Continue reading Importantly, gland size returned to its baseline size within approximately four weeks after stopping treatment, showing that RU58841’s local antiandrogenic effects were reversible in this model. Dose-response experiments in this model revealed that more is not necessarily better. Around 3 µg/day was found to be as effective as 100 µg/day in reducing sebaceous gland size, indicating a pharmacologic limit.
Figure 2. RU58841 and its effects on sebaceous gland sizing: a dose-dependent study. Adapted from Figure 2.[11]Matias, J.R., Gaillard, M. (1995). Local Inhibition Of Sebaceous Gland Growth By Topically Applied RU 58841. Annals of the New York Academy of Sciences. 761. 56-65. Available at: … Continue reading
At What Point Does “Local” Become Systemic?
In the hamster flank-organ study, when RU58841 was given subcutaneously (to mimic complete systemic exposure), systemic antiandrogen signals emerged at higher doses.[12]Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, … Continue reading At 300-1000 µg/animal, researchers observed reductions in prostate weight, indicating that systemic antiandrogenic effects can appear once exposure is high enough.
In intact rats, strong androgen-dependent effects were generally absent up to 1 mg/rat regardless of route of administration, but became apparent at 10 mg/rat. Testosterone increases were noted only after subcutaneous dosing at the highest dose. This suggests endocrine feedback can occur when systemic exposure is sufficiently high.
Figure 3. Antiandrogenic activity of RU5881 in the intact rat after subcutaneous (s.c.), percutaneous (p.c.) or oral (p.o.) treatment on sex organs and testosterone level. Adapted from Figure 3:[13]Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, … Continue reading
Does RU58841 Regrow Hair in Primate Models?
The most hair-relevant preclinical data for RU58841 comes from studies conducted in stump-tailed macaques.[14]Obana, N., Chang, C., Uno, H. (1997). Inhibition Of Hair Growth By Testosterone In The Presence Of Dermal Papilla Cells From The Frontal Bald Scalp Of The Postpubertal Stumptailed Macaque. … Continue reading These primate species can develop an androgenic alopecia-like pattern with age, making it a useful, though still not perfect, translational model.
In these experiments, topical RU58841 was applied to alopecic scalp and produced concentration-dependent improvements in visible hair parameters. A lower concentration (around 0.5%) did not yield impressive results, while a higher concentration (around 5%) produced the strongest regrowth signal over months of treatment.
Reported benefits included increased hair density and hair length. Beyond surface-level changes, the primate work also reported findings that align with meaningful follicle biology improvements, including support for dermal papilla cell growth and evidence consistent with vellus-to-terminal hair conversion.
One particularly notable mechanistic detail from the macaque work is that RU58841 appeared to prevent testosterone-related androgen receptor effects in the dermal papilla. This suggests testosterone may also contribute to miniaturization through androgen receptor signaling, with the androgen receptor acting as a “lynchpin” for both testosterone and DHT in susceptible follicles.
Even so, the macaque evidence has clear constraints. Sample sizes were small, dosing equivalence to human scalp use is uncertain (vehicle, skin barrier, follicular penetration), and the overall pattern suggests benefits are linked to continued treatment rather than a permanent reversal of the underlying process.
Figure 4. Photographs showing the effects of topical RU58841 on hair growth in the bald frontal scalps of the stumptailed macaques. Increased density and length of hairs were observed in a scalp at 4 months after treatment with 5% RU58841 (b) compared with the scalp at pretreatment time (a). Adapted from Figure 3:[15]Obana, N., Chang, C., Uno, H. (1997). Inhibition Of Hair Growth By Testosterone In The Presence Of Dermal Papilla Cells From The Frontal Bald Scalp Of The Postpubertal Stumptailed Macaque. … Continue reading
Does Normal Blood Testosterone Mean It’s Truly Safe?
The primate experiments reported no meaningful change in circulating testosterone during topical RU58841 treatment. These results are often used as a surrogate for limited systemic exposure, particularly when compared with therapies designed to change systemic androgen levels.
However, unchanged serum hormones do not prove the drug is strictly confined to the scalp. Low-level systemic absorption can still occur without shifting hormone levels, and systemic androgen receptor modulation can theoretically happen even when circulating testosterone/DHT remains normal, especially if active metabolites are involved or if certain tissues are more sensitive. So while these findings are encouraging for safety, they are not definitive proof that effects are strictly confined to the application site.
Human Evidence: The Missing Link
Two human clinical trials of RU58841 (under the name PSK‑3841) were registered and marked as completed in the early 2000’s. But the interesting thing is that the results from both of these studies have never been published, leaving a critical gap between promising preclinical work and real-world clinical decision-making.
The unpublished PSK-3841 trials:
- Completion in 2002: A phase I study (ISRCTN49873657) tested a 5% PSK‑3841 solution twice daily for 4 weeks in about 30 men with androgenetic alopecia, with primary endpoints focused on safety, tolerability, endocrine profiles, plus pharmacokinetics.[16]ISRCTN Registry, (no date), ISRCTN49873657. Available at: https://www.isrctn.com/ISRCTN49873657 (Accessed: 02 January 2026)
- Completion in 2003: A larger, multi‑centre, double‑blind trial (ISRCTN71083772) then treated 120 men for 6 months with 2.5% or 5% PSK‑3841 once daily versus vehicle, measuring total and anagen hair counts, safety, tolerability, and pharmacokinetics.[17]ISRCTN Registry, (no date), ISRCTN71083772. Available at: https://www.isrctn.com/ISRCTN71083772 (Accessed: 02 January 2026)
The fact that both studies were completed, yet no peer-reviewed results or detailed reports have appeared, is a major red flag.
For a potentially first-in-class topical antiandrogen with completed phase I and II trials, companies are usually highly motivated to publish positive data. The fact that developers walked away after finishing a 6-month, 120-subject trial, without even a basic efficacy and safety paper, strongly suggests something in the data made the program unattractive to advance.
Why Unpublished Trials Change the Risk Calculation
Without access to the actual results from these clinical trials, it’s impossible to verify key parameters such as:
- How effective RU58841 was for human hair regrowth
- Types and frequency of any adverse events
- How many participants discontinued using RU58841, and the underlying reasons
- Changes in systemic hormones and sex organs
- Durability of any hair regrowth after stopping treatment
- Dose-response relationships
Missing the data from clinical studies forces a dangerous guessing game. Internet anecdotes cannot answer questions that only controlled trials can resolve, like whether rare but serious adverse events occurred, whether endocrine markers shifted subtly over months, or whether regrowth reversed after discontinuation.
Controlled trials also reveal patterns that never appear in forums, such as subgroup vulnerabilities, time-dependent toxicity, or dose thresholds where benefits collapse into harm. Without these insights, users are effectively self-experimenting with a research chemical that already failed to make it through the regulatory system.
Why Was RU58841 Abandoned?
The short answer is that nobody outside the original development team truly knows why RU58841 was abandoned.
But what we do know is that in pharmaceutical development, it is normal for early-stage compounds to fail.[18]Sun, D., Gao, W., Hu, H., Zhou, S. (2022). Why 90% Of Clinical Drug Development Fails And How To Improve It? Acta Pharmaceutica Sinica B. 12(7). 3049-3062. Available at: … Continue reading What is not normal is for companies to complete phase I and II trials and then allow all efficacy and safety data to vanish without any peer-reviewed publication, abstract, or regulatory disclosure.
Once a drug has reached a completed multi-centre, double-blind phase II study, the cost of publishing the results is trivial compared to the tens of millions already spent developing, patenting, manufacturing, and running the trials. Even negative results are typically published or disclosed.
Let’s examine the three most plausible explanations.
Hypothesis 1: Financial Reasons
The official explanation given by the companies involved was financial difficulty.
On the surface, this does sound plausible. Drug development is expensive. But when viewed in context, this explanation collapses. RU58841 had already cleared the most expensive hurdles, such as toxicology work, formulation development, and phase I and II human trials. Writing up and submitting those results would have cost a few thousand dollars at most.
It does not make sense for a company to invest tens of millions into patenting, testing, and completing two human trials, only to stop short of the final, cheapest step. For this reason, financial limitations are the weakest explanation.
Hypothesis 2: Lack of Efficacy
Another possibility is that RU58841 was not effective for hair regrowth in humans.
It is actually quite common for companies to bury disappointing studies while only publishing favorable ones. However, when it comes to RU58841, this reason is highly unlikely, mainly due to the consistent anecdotal reports across forums describing visible regrowth and stabilization with RU58841.
While anecdotes cannot replace clinical trials, the volume and consistency of regrowth reports make it unlikely that RU58841 was completely ineffective in humans. If efficacy were truly absent, the enthusiasm around this research chemical would almost certainly not have persisted throughout the past two decades.
Hypothesis 3: Safety Concerns
This leaves the most plausible explanation, something in the human data made RU58841 commercially or medically unattractive to advance.
While this hypothesis is speculative, it is supported by several warning signals. Follow-up research on RU58841 reported that at least one of its metabolites possessed strong antiandrogenic activity, undermining the original premise that the drug would only act locally in the scalp.[19]Cousty-Berlin, D., Bergaud, B., Bruyant, M.C., Battmann, T., Branche, C., Philibert, D. (1994). Preliminary Pharmacokinetics And Metabolism Of Novel Non-Steroidal Antiandrogens In The Rat: Relation … Continue reading If systemically active metabolites were forming in humans, this would fundamentally change the drug’s risk profile.
Preclinical work also raised more subtle molecular concerns that are rarely discussed online. Some androgen-receptor antagonists can exhibit something called antagonistic-agonism, where compounds designed to block the receptor can, under certain conditions, partially activate it as well.[20]Miyamoto, H., Yeh, S., Wilding, G., Chang, C. (1998). Promotion Of Agonist Activity Of Antiandrogens By The Androgen Receptor Coactivator, ARA70, In Human Prostate Cancer DU145 Cells. Proceedings of … Continue reading
This appears to involve androgen-receptor co-activators such as ARA70, which amplify androgen signaling once the receptor complex is transported into the nucleus. In experimental systems, RU58841 showed a mild but measurable ability to enhance androgen-receptor co-activation in wild-type receptors in a dose-dependent manner. [21]Miyamoto, H., Yeh, S., Wilding, G., Chang, C. (1998). Promotion Of Agonist Activity Of Antiandrogens By The Androgen Receptor Coactivator, ARA70, In Human Prostate Cancer DU145 Cells. Proceedings of … Continue reading While it remains unknown whether this effect occurs in humans or is clinically meaningful, it represents exactly the type of risk that would make long-term use of a topical antiandrogen far less predictable than originally hoped.
Additionally, it’s worth noting that some people trying RU58841 online have reported chest pain, brain fog, and other issues, such as sexual dysfunction. We have not found credible evidence that these effects are permanent, but their nature is consistent with unintended endocrine or cardiovascular involvement.
Together, the evidence is quite difficult to ignore. RU58841 may well have been effective for hair regrowth, but a combination of systemically active metabolites, loss of localization in the scalp, and early molecular signals suggesting complex androgen-receptor behavior may have made the drug too risky to advance.
If you’re a member and would like to find out more about these molecular safety mechanisms, click the link here for more details in our RU58841 Ultimate Guide
Final Thoughts
So is RU58841 legit for efficacy? Plausibly yes. Given the preclinical data and sheer volume of regrowth anecdotes, RU58841 does likely block androgen signaling in the scalp.
But is RU58841 legit as a recommended treatment? No. Its safety profile is still a clinical mystery. Its human trials were never published, and the drug was abandoned at a stage where most drugs either move forward or fail. The gap in clinical information changes the entire risk calculation.
In our opinion, evidence quality and long-term safety hold more weight than exciting anecdotes.
For individuals who cannot tolerate finasteride, there are topical antiandrogen options with more transparent development histories and human data, including topical fluridil, topical CB-03-03, topical spironolactone, and newer molecules like pyrilutamide that are currently progressing through regulated clinical trials. While none are perfect, they at least exist within a framework of published safety monitoring.
If you’d like to learn more about pyrilutamide, read our article here, which covers all of the publicly available data on pyrilutamide and evaluates its mechanisms of action, efficacy, and safety.
We recommend proceeding with extreme caution when using RU58841, and only use it if you understand and are willing to accept the absence of human safety and efficacy data.
The best practice is to prioritize therapies with published human trials and regulatory oversight. Ultimately, given the lack of safety information and regulatory oversight for RU58841, we recommend erring on the side of caution.
References[+]
References ↑1 Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, Androgenetic Alopecia and Hirsutism. J Steroid Biochem Mol Biol. 48(1). 55-60. Available at: https://doi.org/10.1016/0960-0760(94)90250-x ↑2 Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. In: StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ ↑3 Wikipedia, (26 December 2018), RU-58841. Available at: https://en.wikipedia.org/wiki/RU-58841#/media/File:RU-58841_structure.svg (Accessed: 02 January 2026) ↑4, ↑16 ISRCTN Registry, (no date), ISRCTN49873657. Available at: https://www.isrctn.com/ISRCTN49873657 (Accessed: 02 January 2026) ↑5, ↑17 ISRCTN Registry, (no date), ISRCTN71083772. Available at: https://www.isrctn.com/ISRCTN71083772 (Accessed: 02 January 2026) ↑6, ↑7, ↑8, ↑9, ↑12, ↑13 Battmann, T., Bonfils, A., Branche, C., Humbert, J., Goubet, F., Teutsch, G., Philibert, D. (1994). RU 58841, A New Specific Topical Antiandrogen: A Candidate of Choice for the Treatment of Acne, Androgenetic Alopecia and Hirsutism. J Steroid Biochem Mol Biol. 48(1). 55–60. Available at: https://doi.org/10.1016/0960-0760(94)90250-x ↑10 Matias, J.R., Gaillard, M. (1995). Local Inhibition Of Sebaceous Gland Growth By Topically Applied RU 58841. Annals of the New York Academy of Sciences. 761. 56-65. Available at: https://doi.org/10.1111/j.1749-6632.1995.tb31369.x ↑11 Matias, J.R., Gaillard, M. (1995). Local Inhibition Of Sebaceous Gland Growth By Topically Applied RU 58841. Annals of the New York Academy of Sciences. 761. 56-65. Available at: https://doi.org/10.1111/j.1749-6632.1995.tb31369.x ↑14 Obana, N., Chang, C., Uno, H. (1997). Inhibition Of Hair Growth By Testosterone In The Presence Of Dermal Papilla Cells From The Frontal Bald Scalp Of The Postpubertal Stumptailed Macaque. Endocrinology. 138(1). 356-361. Available at: https://doi.org/10.1210/endo.138.1.4890 ↑15 Obana, N., Chang, C., Uno, H. (1997). Inhibition Of Hair Growth By Testosterone In The Presence Of Dermal Papilla Cells From The Frontal Bald Scalp Of The Postpubertal Stumptailed Macaque. Endocrinology. 138(1). 356-361. Available at: https://doi.org/10.1210/endo.138.1.4890 ↑18 Sun, D., Gao, W., Hu, H., Zhou, S. (2022). Why 90% Of Clinical Drug Development Fails And How To Improve It? Acta Pharmaceutica Sinica B. 12(7). 3049-3062. Available at: https://doi.org/10.1016/j.apsb.2022.02.002 ↑19 Cousty-Berlin, D., Bergaud, B., Bruyant, M.C., Battmann, T., Branche, C., Philibert, D. (1994). Preliminary Pharmacokinetics And Metabolism Of Novel Non-Steroidal Antiandrogens In The Rat: Relation Of Their Systemic Activity To The Formation Of A Common Metabolite. Journal of Steroid Biochemistry and Molecular Biology. 51(1-2). 47-55. Available at: https://doi.org/10.1016/0960-0760(94)90114-7 ↑20, ↑21 Miyamoto, H., Yeh, S., Wilding, G., Chang, C. (1998). Promotion Of Agonist Activity Of Antiandrogens By The Androgen Receptor Coactivator, ARA70, In Human Prostate Cancer DU145 Cells. Proceedings of the National Academy of Sciences of the United States of America. 95(13). 7379-7384. Available at: https://doi.org/10.1073/pnas.95.13.7379 Minoxidil has been a cornerstone of hair-loss treatment for decades, and there are now more options than ever for users. Alongside traditional topical solutions and foams, low-dose oral minoxidil has emerged as a popular off-label alternative.
Which raises the question: which formulation is the best choice for hair loss? And which option comes with the fewest side effects? In this article, we break down how oral and topical minoxidil compare across mechanisms, evidence, side effects, and real-world use, so you can make an informed decision about which approach fits your hair loss pattern, risk tolerance, and lifestyle.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Does Minoxidil Work?
Despite being used to treat hair loss for over 40 years, the mechanisms through which minoxidil promotes hair growth are only partially understood. We know that it alters the hair cycle, shortening the resting (telogen) phase so follicles enter the growth (anagen) phase earlier. This increases the number of growing hairs at any one time. It also increases follicle size, which translates into thicker, longer hair shafts.[1]Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. *British Journal of Dermatology.* 150(2). 186–194. Available at: … Continue reading
Minoxidil can increase blood flow to hair follicles, which increases the flow of oxygen and nutrients to support growth. The drug is also known to activate the Wnt/ꞵ-catenin signaling pathway, which supports the growth of cells in the follicle, and provides cytoprotective effects and regulates local inflammation.
Figure 1. Structure of minoxidil.[2]https://commons.wikimedia.org/wiki/File:Minoxidil_Structural_Formula_V1.svg) Image in the Public Domain.
In contrast to antiandrogenic drugs like finasteride and dutasteride, minoxidil doesn’t impact hormones.
Before we compare topical and oral treatments, we’ll look at the formulations separately to understand the options available for each.
Topical Minoxidil
Topical minoxidil is designed to work primarily at the scalp, not systemically. After you apply it, only a small fraction of the dose actually penetrates intact skin, and an even smaller fraction reaches the follicle structures where it matters most.
Where it does work is at (or near) the follicle, particularly around the outer root sheath and dermal papilla. That’s why topical minoxidil has a strong safety record: most of the drug never leaves the scalp in meaningful quantities.
Why Some People Respond Better to Topical Minoxidil
Minoxidil relies on activating enzymes to work, most notably sulfotransferase SULT1A1. SULT1A1 activity in the hair follicles is closely linked to how well topical minoxidil works, and people with lower levels of activity tend to see less benefit from treatment.[3]Pietrauszka, K., & Bergler-Czop, B. (2022). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: … Continue reading
Because minoxidil needs SULT1A1 to work, people with low levels of the enzyme in the hair follicle might not see much improvement from minoxidil (“enzyme low-responders”).
The effectiveness of topical minoxidil also depends on a range of other factors, including the type of formulation, scalp health, contact time, application technique, and the use of penetration enhancers. This is one reason why topical minoxidil performs reliably in clinical trials, but users find results more variable.
Typical Dosing: How Much and How Often?
Most over-the-counter minoxidil is sold in 2% or 5% formulations. 5% generally produces stronger average results and shows higher satisfaction in many studies. It also comes with a slightly higher irritation risk, especially in solution form. As such, 2% formulations are often used when scalp irritation from higher doses is too disruptive, or for milder cases.[4]Olsen, E. A., Dunlap, F. E., Funicella, T., Koperski, J. A., Swinehart, J. M., Tschen, E. H., & Trancik, R. J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical … Continue reading
Higher concentrations (7–15%) are also sometimes considered, but they offer diminishing returns for many users and a higher risk of irritation. Because of this, they’re usually reserved for people who don’t respond to standard options and can tolerate the vehicle.
Dosing frequency is about balancing efficacy with adherence: there’s no benefit to be gained from planning a treatment schedule that you can’t stick to.
Twice-daily use is the traditional standard for maximum effect, especially in people using solutions. Once-daily 5% is often good enough for many people, and can dramatically improve adherence, although studies have shown that switching to twice to once daily can slightly worsen outcomes. [5]Olsen, E. A., DeLong, E. R., & Weiner, M. S. (1987). Long-term follow-up of men with male pattern baldness treated with topical minoxidil. *Journal of the American Academy of Dermatology.* 16(3). … Continue reading
Similarly, in women, once-daily 5% foam can perform similarly to twice-daily 2% solution while being easier to sustain. Foam formulations are often recommended for women because foam tends to stay localized on the scalp and drip down less onto the face than solutions. This reduces the chance of unwanted facial hair growing as a result of treatment. Evidence also suggests that foam formulations have lower systemic absorption into the bloodstream.[6]Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., & Garcia Bartels, N. (2011). A randomized, single-blind trial of 5% minoxidil foam once daily versus 2% minoxidil solution twice daily in … Continue reading,[7]Gogtay, J. A., & Panda, M. (2009). Minoxidil topical foam: a new kid on the block. *International Journal of Trichology.* 1(2). 142. Available at: https://doi.org/10.4103/0974-7753.58560
Limitations of Topical Minoxidil
Topical minoxidil works, but it requires consistency. Results typically require months of consistent use before they become apparent, and many people struggle to maintain the routine, especially with twice-daily application.
Absorption of topical solutions can also be inconsistent, and is impacted by scalp condition, contact time, and the type of vehicle used (propylene glycol-based solutions, foams, propylene glycol-free lotions, nanoemulsions, etc.). As such, even with consistent application, results can vary.
The main side effects associated with topical minoxidil are local rather than systemic. Common issues include itching, dryness, flaking, and contact dermatitis, which are often driven by the formulation rather than the drug itself. Many users also find the product cosmetically inconvenient due to a greasy feel, visible residue, or interference with hairstyling, particularly for those with longer hair.[8]Olsen, E. A., DeLong, E. R., & Weiner, M. S. (1987). Long-term follow-up of men with male pattern baldness treated with topical minoxidil. *Journal of the American Academy of Dermatology.* 16(3). … Continue reading
Evidence for Topical Minoxidil
The use of topical minoxidil is backed up by substantial clinical data showing increased hair counts, density, and coverage in androgenic alopecia (AGA) and other alopecias, with response in a majority but not all patients. We’ll look at evidence from clinical trials involving men and women separately, as they are typically studied in different clinical trials.
Evidence in Men
The earliest significant clinical evidence supporting the use of minoxidil for hair loss is from a 1985 trial, which compared 2% and 3% solutions to placebo in 126 men with early male pattern baldness. They assessed changes in vellus hairs and terminal hairs, an important distinction as vellus hairs are fine, light colored hairs that don’t contribute to the overall appearance of hair thickness.[9]Olsen, E. A., DeLong, E. R., & Weiner, M. S. (1987). Long-term follow-up of men with male pattern baldness treated with topical minoxidil. *Journal of the American Academy of Dermatology.* 16(3). … Continue reading
They found a significant increase in terminal hair counts in 2% and 3% minoxidil-treated participants, with a greater improvement in the 3% group, though this was not statistically significant.
The study methodology switched the placebo group to 3% minoxidil after 4 months and showed that the switch increased hair counts. Unfortunately, this means that it’s not possible to assess actual changes in hair count over the 12-month study. Notably, the placebo group also showed increases in hair count during the first 4 months, which might not be expected in men with pattern baldness or AGA and could cast doubt on the hair counts.
Larger trials have been conducted since. A 48 week study compared 2% and 5% solutions to placebo and found approximately 13% and 19% increases in nonvellus hairs respectively, although hair counts did appear to start to drop after week 16.[10]Olsen, E. A., Dunlap, F. E., Funicella, T., Koperski, J. A., Swinehart, J. M., Tschen, E. H., & Trancik, R. J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical … Continue reading
Similarly, a Japanese study compared 1% and 5% formulations, and found a 26.4% increase in nonvellus hair count in the 5% topical minoxidil group and 21.2% increase in the 1% group.[11]Tsuboi, R., Arano, O., Nishikawa, T., Yamada, H., & Katsuoka, K. (2009). Randomized clinical trial comparing 5% and 1% topical minoxidil for the treatment of androgenetic alopecia in Japanese … Continue reading
Notably, these trials tend to be relatively short, with many lasting only 16 weeks. Even in these short trials, there is a noticeable drop-off in hair counts after an initial growth boost. Data suggests that topical minoxidil response rates are high in the first 3-6 months, but drop after a year or longer of use. Real-world data show that 86–95% of users discontinue topical minoxidil within one year, most commonly because the results fall short of their expectations for visible cosmetic improvement.[12]Olsen, E. A., Weiner, M. S., Amara, I. A., & DeLong, E. R. (1990). Five-year follow-up of men with androgenetic alopecia treated with topical minoxidil. *Journal of the American Academy of … Continue reading,[13]Shadi, Z. (2023). Compliance to topical minoxidil and reasons for discontinuation among patients with androgenetic alopecia. *Dermatology and Therapy.* 13(5). 1157–1169. Available at: … Continue reading
If you’re interested in topical minoxidil for men, check out our article on the best products currently available.
Evidence in Women
Most clinical trials evaluating topical minoxidil in women have traditionally used a 2% solution applied twice daily. A meta-analysis, which combines the results from multiple trials found that 2% minoxidil produced an average increase of about 12 hairs per square centimeter compared with placebo after 24 weeks of treatment.[14]Adil, A., & Godwin, M. (2017). The effectiveness of treatments for androgenetic alopecia: a systematic review and meta-analysis. *Journal of the American Academy of Dermatology.* 77(1). … Continue reading
Data also suggest that higher-strength formulations offer similar outcomes: both 5% minoxidil solution used twice daily and 5% minoxidil foam applied once daily have shown no significant difference in efficacy compared with twice-daily 2% solution use.[15]Lucky, A. W., Piacquadio, D. J., Ditre, C. M., Dunlap, F., Kantor, I., Pandya, A. G., Savin, R. C., & Tharp, M. D. (2004). A randomized, placebo-controlled trial of 5% and 2% topical minoxidil … Continue reading
As we’ve already noted, foams are often preferred by women as they can potentially limit off-target hair growth. A large, 24-week trial demonstrated that foam minoxidil formulation resulted in 9.1 hairs more per cm2 more than a placebo (foam without minoxidil), with no significant differences in irritation.[16]Lucky, A. W., Piacquadio, D. J., Ditre, C. M., Dunlap, F., Kantor, I., Pandya, A. G., Savin, R. C., & Tharp, M. D. (2004). A randomized, placebo-controlled trial of 5% and 2% topical minoxidil … Continue reading
Figure 2. Improvement in hair coverage in a woman with pattern hair loss following 6 months 5% topical minoxidil treatment. Adapted from Figure 1.[17]Ramos, P. M., Melo, D. F., Radwanski, H., Cortez de Almeida, R. F., & Miot, H. A. (2023). Female-pattern hair loss: therapeutic update. *Anais Brasileiros de Dermatologia.* 98. 506–519. … Continue reading Image used under Creative Commons License.
Oral Minoxidil
Unlike topical minoxidil, oral minoxidil is absorbed through the gastrointestinal tract and converted into its active form primarily in the liver, where sulfotransferase activity is abundant. This systemic activation largely bypasses one of the major limitations of topical therapy: variations in follicular enzyme activity. As a result, nearly all ingested minoxidil is activated and made bioavailable, which helps explain why oral formulations can work even in people who do not respond well to topical formulations.
Typical Dosing
Minoxidil was originally developed to treat hypertension, and it is still used for the condition today. Dosage for hair loss is much lower than that for hypertension, and most regimens fall within the low-dose range of 0.25 to 5 mg per day.
Within this range, efficacy appears to be dose-dependent. In men, doses between 2.5 and 5 mg daily are commonly used and tend to produce the most robust regrowth.[18]Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: … Continue reading,[19]Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O. M., Fernandez-Nieto, D., & Vaño-Galvan, S. (2019). Effectiveness and safety of low-dose oral … Continue reading
However, lower doses may still be effective for slowing progression or improving density in milder cases. In women, lower doses (0.25 to 1.25 mg daily) are preferred, as they balance efficacy with a lower risk of unwanted body hair growth and fluid retention.[20]Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse … Continue reading
To find out more about minoxidil dosing, check out our article.
Oral Minoxidil Side Effects
Because oral minoxidil is systemically active, its side effects differ from those of topical formulations. Still, at the low doses used for hair loss, oral minoxidil is generally well tolerated, and most adverse effects are reversible with adjustment or discontinuation.
The most commonly reported side effect is unwanted hair growth outside of the scalp, including on the face and arms. While this effect is often mild and reversible, it is often more of an issue for women and becomes more likely as the dose increases.[21]Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair … Continue reading
Another relatively common effect is fluid retention, which may present as mild ankle or lower-leg swelling. This occurs because minoxidil is a vasodilator and can promote sodium and water retention. Some patients also report lightheadedness or dizziness, particularly when starting treatment or increasing the dose.
Less commonly, patients may experience palpitations or changes in heart rate. These symptoms are usually short-lasting and improve with dose reduction. Importantly, serious cardiovascular complications are rare at doses of 0.25-5 mg daily and have been reported primarily in higher-dose antihypertensive use or in individuals with underlying heart disease.[22]Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse … Continue reading
Figure 3. Occurrence of adverse events in individuals taking low-dose oral minoxidil (LDOM), as reported in the FDA Adverse Event Reporting System. Adapted from Figure 1.[23]Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse … Continue reading Image used under Creative Commons License.
If you’re concerned about minoxidil side effects, check out our article on 10 ways to reduce them.
Evidence in Men
Multiple studies in men with AGA show consistent increases in total hair count and visible thickening, with reported response rates ranging from roughly 60% at very low doses (0.25 mg) to 90–100% at doses between 2.5 and 5 mg daily over 6-12 months.
The 60% response at 0.25 mg comes from a 2020 retrospective study of 25 men, looking at the effect of very-low-dose minoxidil. While there was a 60% response rate, there wasn’t any meaningful increase in terminal hair count on average.[24]Pirmez, R., & Salas-Callo, C. I. (2020). Very-low-dose oral minoxidil in male androgenetic alopecia: a study with quantitative trichoscopic documentation. *Journal of the American Academy of … Continue reading
At higher doses, response rates are higher, and hair growth is more predictable. A 24-week study using 5 mg minoxidil daily found a 19% increase in hair count. An open-label study using 5 mg minoxidil, without a placebo control, found increases in hair count and improvements in photographic assessment at 12 and 24 weeks. They also found a 19% improvement in hair counts compared to baseline.[25]Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: … Continue reading,[26]Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global … Continue reading
A retrospective study of only 41 men found a response rate of 90%, though changes were not well quantified.[27]Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O. M., Fernandez-Nieto, D., & Vaño-Galvan, S. (2019). Effectiveness and safety of low-dose oral … Continue reading
In these studies, average gains included double-digit percentage increases in hair count and noticeable improvements on global photographic assessment, indicating that oral minoxidil can produce cosmetically relevant changes rather than purely statistical ones.
Evidence in Women
In female pattern hair loss (FPHL), oral minoxidil at doses between 0.25 and 1.25 mg daily has demonstrated efficacy comparable to 5% topical minoxidil, with the added benefit of avoiding scalp irritation. Combination approaches appear particularly promising: studies pairing low-dose oral minoxidil with spironolactone report earlier reductions in shedding and greater density gains than either agent alone, although direct head-to-head comparisons remain limited.[28]Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 … Continue reading,[29]Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). … Continue reading
Oral Minoxidil in Special Populations
Evidence also supports oral minoxidil’s utility in several special populations. In people with diffuse thinning, including chronic telogen effluvium, low-dose oral minoxidil has been associated with reduced shedding and gradual improvements in density over 6–12 months.[30]Perera, E., & Sinclair, R. (2017). Treatment of chronic telogen effluvium with oral minoxidil: a retrospective study. *F1000Research.* 6. 1650. Available at: … Continue reading
While these studies are largely retrospective and lack placebo controls, the consistency of improvement across patients suggests a real biological effect, especially given minoxidil’s known ability to shorten telogen and promote anagen entry.
In alopecia areata, oral minoxidil alone produces modest response rates, but when added to immunomodulatory agents such as tofacitinib, it appears to substantially enhance regrowth without requiring higher doses of the primary drug.[31]Wambier, C. G., Craiglow, B. G., & King, B. A. (2021). Combination tofacitinib and oral minoxidil treatment for severe alopecia areata. *Journal of the American Academy of Dermatology.* 85(3). … Continue reading
Oral vs. Topical Minoxidil: Direct Comparison
Due to differences in study design and the range of doses and formulations used, it is difficult to compare the results of studies on oral and topical minoxidil. Luckily, a few trials have compared the two treatment routes directly.
Comparison Study #1
The only direct comparison of the efficacy of topical and oral minoxidil in men comes from a 2024 randomized placebo-controlled trial comparing a 5 mg oral dose (once daily) and 5% topical formulation (twice daily) for 24 weeks. They found no significant difference between the two formulations in hair, though there was a 20% increase in hair density in the oral group compared to 7% increase in the topical group. Photographic assessment indicated a significantly greater improvement in the oral group at the crown.[32]Penha, M. A., Miot, H. A., Kasprzak, M., & Ramos, P. M. (2024). Oral minoxidil vs topical minoxidil for male androgenetic alopecia: a randomized clinical trial. *JAMA Dermatology.* 160(6). … Continue reading
There was a higher rate of adverse events in the oral treatment group, though this was largely due to increased hair growth in other parts of the body. Participants taking oral minoxidil experienced significantly more headaches, while the topical group experienced scalp irritation more frequently, as we would expect.
Comparison Study #2
There are more head-to-head comparisons of oral and topical formulations in women. The first direct comparison of oral and topical minoxidil for female-pattern hair loss comes from a 24-week randomized, open comparative study conducted in Brazil. The trial compared low-dose oral minoxidil (1 mg once daily) with topical minoxidil 5% solution applied once daily in women aged 18–65 years.[33]Ramos, P. M., Melo, D. F., Radwanski, H., Cortez de Almeida, R. F., & Miot, H. A. (2023). Female-pattern hair loss: therapeutic update. *Anais Brasileiros de Dermatologia.* 98. 506–519. … Continue reading
After 24 weeks, terminal hair density increased by 12% in the oral minoxidil group and by 7.2% in the topical group. This difference did not reach statistical significance . Secondary outcomes, including hair density, global photographic assessment by dermatologists, quality-of-life scores, and hair shedding, were also evaluated, with no significant between-group differences reported.
Hypertrichosis (hair growth on other parts of the body) was reported in 27% of the oral group, compared to 4% in the topical group. This lower incidence of adverse events in the oral group in this trial compared to the study #1 is likely due to lower doses used.
Comparison Study #3
In a 6-month trial, both treatment groups demonstrated a statistically significant increase in hair diameter. Again, no significant differences between topical and oral minoxidil were reported. Photographic assessment showed significant improvement in hair densityn the topical minoxidil group, whereas no significant improvement was detected in the oral minoxidil group; however, between-group differences were not statistically significant.[34]Asilian, A., Farmani, A., & Saber, M. (2024). Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in the improvement of androgenetic alopecia: a randomized controlled … Continue reading
Patient satisfaction exceeded 60% in both groups, with no significant difference reported. Both treatments were well tolerated, and no major safety concerns were identified, though two patients in the oral group experienced hypertrichosis on their face and extremities.
Figure 4. Improvements in average hair diameter were comparable between 5% topical solution or 1 mg/day oral minoxidil over a six-month study period. Adapted from Figure 2.[35]Asilian, A., Farmani, A., & Saber, M. (2024). Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in the improvement of androgenetic alopecia: a randomized controlled … Continue reading Image used under Creative Commons License.
Comparison Study #4
Our final study again showed no significant difference between the two groups. This study used lower doses than previously seen: 0.25 mg daily with a topical placebo or topical minoxidil 2% solution with an oral placebo for 9 months.[36]Vahabi-Amlashi, S., Layegh, P., Kiafar, B., Hoseininezhad, M., Abbaspour, M., Hajebi Khaniki, S., Forouzanfar, M., & Sabeti, V. (2021). A randomized clinical trial on therapeutic effects of 0.25 … Continue reading
In the oral minoxidil group, average hair density increased from 102/cm² to 115/cm² (1.12% increase), while in the topical minoxidil group, hair density increased from 107/cm² to 113/cm² (1.06%).
These improvements are modest compared to those we have seen previously, likely due to the lower doses used. This is also reflected in the very low incidence of side effects reported.
Meta-Analysis
Meta-analyses compile numerous studies together to see if there are consistent results across different research groups and trials. A recent meta-analysis comparing topical and oral minoxidil found that there was no significant difference between the two approaches.[37]Fazal, F., Malik, B. H., Malik, H. M., Sabir, B., Mustafa, H., Ahmed, M., Abid, A., Adil, M. L., Shafi, U., & Saad, M. (2025). Can oral minoxidil be the game changer in androgenetic alopecia? A … Continue reading
This finding is apparent in the studies we’ve looked at: there are some cases where oral treatment seems to provide better results, but this is not large or consistent enough to be significant when statistical analyses are performed.
Given the trend towards greater improvements in oral formulations, it is possible that larger, longer-term studies might find significant differences. However, on an individual basis, these small, population-scale differences are unlikely to be large enough to influence treatment decisions.
Is Topical Minoxidil Safer?
Both formulations are well tolerated across studies, with the most common reported side effect for topical formulations being scalp irritation. We have seen a higher rate of side effects in groups taking oral minoxidil, though this is most commonly due to increased hypertrichosis, which may be more of a concern for women.
More serious concerns around the use of oral minoxidil are related to cardiovascular events. A large-scale retrospective study assessed the safety of 1404 people taking oral minoxidil at a range of low doses appropriate for hair loss.[38]Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair … Continue reading
Apart from hypertrichosis, adverse events were rare, with lightheadedness reported in 1.7% of participants and tachycardia (fast heart rate) reported in 0.9%. No life‑threatening cardiovascular events were reported.
Therefore, while there is an increased risk of more serious side effects in oral formulations, at the low doses used for hair loss, there is minimal risk of adverse events.
Adherence and Practical Considerations
One of oral minoxidil’s most compelling advantages is its performance in people who do not respond adequately to topical therapy. Because topical minoxidil requires local scalp activation by the sulfotransferase enzyme SULT1A1, and because only a small fraction of applied drug penetrates the skin, many users see limited benefit despite correct use. Oral minoxidil bypasses both of these bottlenecks by being converted to its active sulfate form in the liver and delivered systemically, ensuring uniform exposure to hair follicles across the scalp.
As well as efficacy, there are some other practical considerations that should influence your decisions when comparing oral and topical minoxidil.
Ease of Use
From a day-to-day standpoint, oral and topical minoxidil place very different demands on the user. Oral minoxidil is taken as a pill, usually once daily, or split into morning and evening doses, which makes it largely invisible in daily life. There is no impact on hairstyling, no waiting period before washing, and no concern about residue, odor, or transfer to pillows, clothing, or pets. For many people, this simplicity is the primary reason oral minoxidil feels sustainable.
Topical minoxidil, by contrast, requires direct scalp application once or twice daily, ideally to a dry scalp, with sufficient contact time before washing. Proper use often means parting hair, targeting thinning areas precisely, and tolerating cosmetic downsides such as greasiness, stiffness, or flaking. While none of these steps are difficult in isolation, the cumulative friction can become a barrier over months and years of use.
Cost and Access
Topical minoxidil has a clear accessibility advantage: it is available over the counter in standard 2% and 5% formulations, allowing immediate initiation without a prescription. For many users, this makes topical therapy an easy first-line option. Basic formulations are relatively inexpensive on a monthly basis, especially when purchased in bulk.
Oral minoxidil is prescription-only and used off-label for hair loss. This introduces an additional step and sometimes variability in access depending on region, provider comfort, and insurance coverage. That said, once prescribed, low-dose oral minoxidil is often comparable in cost and, in some cases, less expensive over time than higher-strength or specialty topical formulations.
Who Should Choose What?
Choosing between oral and topical minoxidil isn’t really about which one is “stronger” on paper. Rather, it’s important to consider which one you’re most likely to tolerate and use consistently.
Who Topical Minoxidil Is Best For
Risk-averse individuals – Topical minoxidil remains the best first-line choice for many people because it offers meaningful benefit with minimal systemic exposure. If you want the lowest-risk long-term approach and are comfortable with scalp application, topical is usually the most conservative starting point.
Cardiovascular contraindications (or uncertainty) – Anyone with a history of cardiovascular disease, kidney issues, fluid retention, low blood pressure, or unexplained palpitations should be cautious with oral minoxidil and involve a clinician if considering it. Topical use is generally preferred in these cases because systemic absorption is far lower and systemic side effects are much less common.
Localized thinning – If hair loss is limited to smaller areas (early crown thinning, mild recession, small patches of miniaturization), topical minoxidil can be a good fit. It allows you to treat targeted regions without committing to systemic exposure, and it can work well when the application is straightforward and consistent.
Who Oral Minoxidil Is Best For
Poor topical responders – If you’ve used topical minoxidil correctly for 6-12 months and seen little to no improvement, oral minoxidil may be a logical next step. Topical therapy can fail even with perfect effort due to low follicular sulfotransferase activity or limited penetration through the scalp barrier. Oral minoxidil bypasses both issues by being activated systemically and delivered more uniformly to follicles.
Diffuse or advanced thinning – Oral minoxidil can be especially appealing when thinning is widespread across the scalp (rather than concentrated at the crown or hairline), because systemic delivery doesn’t depend on targeting specific areas. It also tends to be easier to scale for advanced loss, where topical application becomes time-consuming and difficult to do thoroughly.
Convenience-focused patients – If the routine topical treatment has been a major barrier, or if you experience issues with messiness, residue, or styling disruption, oral minoxidil is often more sustainable.
Final Thoughts
There isn’t a universally superior option between oral and topical minoxidil; it’s just a matter of different trade-offs. Oral minoxidil tends to produce more consistent regrowth for many users because it avoids the two biggest bottlenecks of topical therapy: variable absorption and variable enzyme activation. The cost of that consistency is higher systemic exposure, which increases the likelihood of side effects like hypertrichosis, fluid retention, dizziness, or palpitations, even if serious complications remain rare at low doses.
Topical minoxidil offers a safer long-term profile, wide accessibility, and decades of clinical data, but its outcomes are more variable in real life. The same factors that make it safe, local delivery, and limited systemic absorption, also limit predictability, especially when adherence slips or the scalp barrier and follicular activation aren’t favorable.
In the end, the best outcomes come from individualized treatment plans that match the patient’s biology, preferences, and ability to stay consistent. Minoxidil can be a powerful tool either way, and the version you can tolerate and actually use is the one most likely to work.
References[+]
References ↑1 Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. *British Journal of Dermatology.* 150(2). 186–194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑2 https://commons.wikimedia.org/wiki/File:Minoxidil_Structural_Formula_V1.svg) ↑3 Pietrauszka, K., & Bergler-Czop, B. (2022). Sulfotransferase SULT1A1 activity in hair follicle, a prognostic marker of response to the minoxidil treatment in patients with androgenetic alopecia: a review. *Advances in Dermatology and Allergology / Postępy Dermatologii i Alergologii.* 39(3). 472–478. Available at: https://doi.org/10.5114/ada.2020.99947 ↑4, ↑10 Olsen, E. A., Dunlap, F. E., Funicella, T., Koperski, J. A., Swinehart, J. M., Tschen, E. H., & Trancik, R. J. (2002). A randomized clinical trial of 5% topical minoxidil versus 2% topical minoxidil and placebo in the treatment of androgenetic alopecia in men. *Journal of the American Academy of Dermatology.* 47(3). 377–385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑5 Olsen, E. A., DeLong, E. R., & Weiner, M. S. (1987). Long-term follow-up of men with male pattern baldness treated with topical minoxidil. *Journal of the American Academy of Dermatology.* 16(3). 688–695. Available at: https://doi.org/10.1016/S0190-9622(87)70089-9 ↑6 Blume-Peytavi, U., Hillmann, K., Dietz, E., Canfield, D., & Garcia Bartels, N. (2011). A randomized, single-blind trial of 5% minoxidil foam once daily versus 2% minoxidil solution twice daily in the treatment of androgenetic alopecia in women. *Journal of the American Academy of Dermatology.* 65(6). 1126–1134. Available at: https://doi.org/10.1016/j.jaad.2010.09.724 ↑7 Gogtay, J. A., & Panda, M. (2009). Minoxidil topical foam: a new kid on the block. *International Journal of Trichology.* 1(2). 142. Available at: https://doi.org/10.4103/0974-7753.58560 ↑8, ↑9 Olsen, E. A., DeLong, E. R., & Weiner, M. S. (1987). Long-term follow-up of men with male pattern baldness treated with topical minoxidil. *Journal of the American Academy of Dermatology.* 16(3). 688–695. Available at: https://doi.org/10.1016/S0190-9622(87)70089-9 ↑11 Tsuboi, R., Arano, O., Nishikawa, T., Yamada, H., & Katsuoka, K. (2009). Randomized clinical trial comparing 5% and 1% topical minoxidil for the treatment of androgenetic alopecia in Japanese men. *The Journal of Dermatology.* 36(8). 437–446. Available at: https://doi.org/10.1111/j.1346-8138.2009.00673.x ↑12 Olsen, E. A., Weiner, M. S., Amara, I. A., & DeLong, E. R. (1990). Five-year follow-up of men with androgenetic alopecia treated with topical minoxidil. *Journal of the American Academy of Dermatology.* 22(4). 643–646. Available at: https://doi.org/10.1016/0190-9622(90)70089-Z ↑13 Shadi, Z. (2023). Compliance to topical minoxidil and reasons for discontinuation among patients with androgenetic alopecia. *Dermatology and Therapy.* 13(5). 1157–1169. Available at: https://doi.org/10.1007/s13555-023-00919-x ↑14 Adil, A., & Godwin, M. (2017). The effectiveness of treatments for androgenetic alopecia: a systematic review and meta-analysis. *Journal of the American Academy of Dermatology.* 77(1). 136–141. Available at: https://doi.org/10.1016/j.jaad.2017.02.054 ↑15 Lucky, A. W., Piacquadio, D. J., Ditre, C. M., Dunlap, F., Kantor, I., Pandya, A. G., Savin, R. C., & Tharp, M. D. (2004). A randomized, placebo-controlled trial of 5% and 2% topical minoxidil solutions in the treatment of female pattern hair loss. *Journal of the American Academy of Dermatology.* 50(4). 541–553. Available at: https://doi.org/10.1016/j.jaad.2003.06.014 ↑16 Lucky, A. W., Piacquadio, D. J., Ditre, C. M., Dunlap, F., Kantor, I., Pandya, A. G., Savin, R. C., & Tharp, M. D. (2004). A randomized, placebo-controlled trial of 5% and 2% topical minoxidil solutions in the treatment of female pattern hair loss. *Journal of the American Academy of Dermatology.* 50(4). 541–553. Available at: https://doi.org/10.1016/j.jaad.2003.06.014 ↑17, ↑33 Ramos, P. M., Melo, D. F., Radwanski, H., Cortez de Almeida, R. F., & Miot, H. A. (2023). Female-pattern hair loss: therapeutic update. *Anais Brasileiros de Dermatologia.* 98. 506–519. Available at: https://doi.org/10.1016/j.abd.2022.09.006 ↑18 Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: https://doi.org/10.1016/j.jaad.2015.02.466 ↑19, ↑27 Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., Hermosa-Gelbard, A., Moreno-Arrones, O. M., Fernandez-Nieto, D., & Vaño-Galvan, S. (2019). Effectiveness and safety of low-dose oral minoxidil in male androgenetic alopecia. *Journal of the American Academy of Dermatology.* 81(2). 648–649. Available at: https://doi.org/10.1016/j.jaad.2019.04.054 ↑20, ↑22 Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse Event Reporting System (FAERS) with a focus on pericardial effusions. *Journal of Cosmetic Dermatology.* 24(1). e16574. Available at: https://doi.org/10.1111/jocd.16574 ↑21, ↑38 Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair loss: a multicenter study of 1404 patients. *Journal of the American Academy of Dermatology.* 84(6). 1644–1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑23 Gupta, A. K., Bamimore, M. A., Abdel-Qadir, H., Williams, G., Tosti, A., Piguet, V., & Talukder, M. (2025). Low-dose oral minoxidil and associated adverse events: analyses of the FDA Adverse Event Reporting System (FAERS) with a focus on pericardial effusions. *Journal of Cosmetic Dermatology.* 24(1). e16574. Available at: https://doi.org/10.1111/jocd.16574 ↑24 Pirmez, R., & Salas-Callo, C. I. (2020). Very-low-dose oral minoxidil in male androgenetic alopecia: a study with quantitative trichoscopic documentation. *Journal of the American Academy of Dermatology.* 82(1). e21–e22. Available at: https://doi.org/10.1016/j.jaad.2019.08.084 ↑25 Jaén, P., & Arias-Santiago, S. (n.d.). Efficacy and safety of oral minoxidil 5 mg daily during 24-week treatment in male androgenetic alopecia. Available at: https://www.researchgate.net/profile/Suparuj-Lueangarun/publication/319006716_Efficacy_and_safety_of_oral_minoxidil_5_mg_daily_during_24-week_treatment_in_male_androgenetic_alopecia/links/5b46f1690f7e9b4637cde38b/Efficacy-and-safety-of-oral-minoxidil-5-mg-daily-during-24-week-treatment-in-male-androgenetic-alopecia.pdf ↑26 Panchaprateep, R., & Lueangarun, S. (2020). Efficacy and safety of oral minoxidil 5 mg once daily in the treatment of male patients with androgenetic alopecia: an open-label and global photographic assessment. *Dermatology and Therapy.* 10(6). 1345–1357. Available at: https://doi.org/10.1007/s13555-020-00448-x ↑28 Silva, M. N. E., Ramos, P. M., Silva, M. R., Silva, R. N. E., & Raposo, N. R. B. (2022). Randomized clinical trial of low-dose oral minoxidil for the treatment of female pattern hair loss: 0.25 mg versus 1 mg. 396–399. Available at: https://doi.org/10.1016/j.jaad.2022.01.017 ↑29 Sinclair, R. D. (2018). Female pattern hair loss: a pilot study investigating combination therapy with low-dose oral minoxidil and spironolactone. *International Journal of Dermatology.* 57(1). 104–109. Available at: https://doi.org/10.1111/ijd.13838 ↑30 Perera, E., & Sinclair, R. (2017). Treatment of chronic telogen effluvium with oral minoxidil: a retrospective study. *F1000Research.* 6. 1650. Available at: https://doi.org/10.12688/f1000research.11775.1 ↑31 Wambier, C. G., Craiglow, B. G., & King, B. A. (2021). Combination tofacitinib and oral minoxidil treatment for severe alopecia areata. *Journal of the American Academy of Dermatology.* 85(3). 743–745. Available at: https://doi.org/10.1016/j.jaad.2019.08.080 ↑32 Penha, M. A., Miot, H. A., Kasprzak, M., & Ramos, P. M. (2024). Oral minoxidil vs topical minoxidil for male androgenetic alopecia: a randomized clinical trial. *JAMA Dermatology.* 160(6). 600–605. Available at: doi:10.1001/jamadermatol.2024.0284 ↑34, ↑35 Asilian, A., Farmani, A., & Saber, M. (2024). Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in the improvement of androgenetic alopecia: a randomized controlled trial. *Journal of Cosmetic Dermatology.* 23(3). 949–957. Available at: https://doi.org/10.1111/jocd.16086 ↑36 Vahabi-Amlashi, S., Layegh, P., Kiafar, B., Hoseininezhad, M., Abbaspour, M., Hajebi Khaniki, S., Forouzanfar, M., & Sabeti, V. (2021). A randomized clinical trial on therapeutic effects of 0.25 mg oral minoxidil tablets on treatment of female pattern hair loss. *Dermatologic Therapy.* 34(6). e15131. Available at: https://doi.org/10.1111/dth.15131 ↑37 Fazal, F., Malik, B. H., Malik, H. M., Sabir, B., Mustafa, H., Ahmed, M., Abid, A., Adil, M. L., Shafi, U., & Saad, M. (2025). Can oral minoxidil be the game changer in androgenetic alopecia? A comprehensive review and meta-analysis comparing topical and oral minoxidil for treating androgenetic alopecia. *Skin Health and Disease.* vzaf009. Available at: https://doi.org/10.1093/skinhd/vzaf009 Finasteride and dutasteride are prescription medications that treat androgenic alopecia (AGA) by targeting the hormone pathway that drives follicle miniaturization. Because they share the same core mechanism and have similar effects on hair loss, understanding which treatment is the best choice for you is not straightforward.
In this article, we tackle the questions that come up most often when choosing between finasteride and dutasteride. We’ll break down how they differ biologically, what head-to-head studies really show about regrowth and side effects, and how oral versus topical use changes the equation, so you can make a more informed decision about which option makes sense for your hair loss and your goals.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What do Finasteride and Dutasteride Actually Do?
AGA is not a sudden shedding event but a slow remodeling of the hair follicle driven by androgen sensitivity. Across repeated hair cycles, susceptible follicles undergo progressive miniaturization: the growth phase (anagen) shortens with each cycle, while the rest phase (telogen) remains the same or lengthens.
Dihydrotestosterone (DHT) is the key androgen driving follicular miniaturization. An enzyme called 5α‑reductase (5AR) in the hair follicle converts testosterone to DHT. Because DHT is known to drive hair loss progression, many treatments for AGA target 5AR to reduce scalp DHT levels.[1]Asfour, L., Cranwell, W., & Sinclair, R. (2023). Male androgenetic alopecia. *Endotext [Internet].* Available at: https://www.ncbi.nlm.nih.gov/books/NBK278957/
Both finasteride and dutasteride can decrease levels of DHT in the body. They were originally created to treat a condition called benign prostate hyperplasia (BPH), but have been repurposed for AGA since their impact on hair growth was noticed. While finasteride is approved by the FDA for treating both BPH, dutasteride is approved only for BPH and is therefore used off-label for hair loss.
Type I vs Type II 5α-reductase
Finasteride is an inhibitor of Type II 5AR, which is highly expressed in hair follicles in androgen‑sensitive regions such as the scalp and prostate tissue. Type II is responsible for the conversion of testosterone to DHT inside miniaturizing scalp follicles, directly driving AGA‑related changes.[2]Makridakis, N., & Reichardt, J. K. V. (2005). Pharmacogenetic analysis of human steroid 5α-reductase type II: comparison of finasteride and dutasteride. *Journal of Molecular Endocrinology.* … Continue reading
Dutasteride is a dual 5AR inhibitor, which means that it blocks Type I 5AR as well as Type II. Type I 5AR plays a more supportive role and is mainly found in sebaceous glands and skin, so it has a less direct impact on scalp follicular miniaturization.[3]Frye, S. V. (2006). Discovery and clinical development of dutasteride, a potent dual 5α-reductase inhibitor. *Current Topics in Medicinal Chemistry.* 6(5). 405–421. Available at: … Continue reading
Because of this dual action, dutasteride may decrease levels of DHT to a greater extent than finasteride. In fact, studies have shown that dutasteride may decrease DHT by 50% more than finasteride.
Figure 1. The structures of finasteride and dutasteride.[4]Wikimedia Commons. (n.d.). Azasteroid Pharmaceuticals [Image]. *Wikimedia Commons.* Available at: https://commons.wikimedia.org/wiki/File:Azasteroid_Pharmaceuticals.png (Accessed: November 2025) Image in the Public Domain.
Does that mean that dutasteride is the better option? Not necessarily. Lower DHT doesn’t necessarily translate into improved hair growth. What’s more, side effects associated with decreased DHT, particularly sexual dysfunction, can affect other aspects of health and well-being.
To understand the potential benefits and side effects of finasteride and dutasteride, we’ll dive into the science and studies that can help us make choices about treatment options.
Formulations: Topical or Oral?
It’s important to recognize that there are many different ways both finasteride and dutasteride can be taken. This includes the drug’s concentration, the frequency of administration, and the formulation used.
Oral finasteride and later oral dutasteride were designed to suppress DHT throughout the body, and they remain the most thoroughly studied options for AGA. Oral finasteride is most commonly prescribed at 1mg daily for AGA, while oral dutasteride is typically taken at 0.5mg daily. Some studies have tested a wider range of concentrations to investigate if there are any changes in benefit or risk.
Topical formulations were introduced later to change the risk profile without affecting the mechanism. Instead of lowering DHT systemically by default, topical finasteride was designed to concentrate drug activity in the scalp while limiting how much reaches the bloodstream.
Topical dutasteride represents the newest and least mature category, but evidence is promising. Because dutasteride is more potent than finasteride, even very low topical concentrations (around 0.01-0.05%) can meaningfully suppress scalp DHT. Importantly, both finasteride and dutasteride can and do enter systemic circulation when applied to the scalp, so safety concerns still need to be addressed.
Which Improves Hair Growth Most?
Because there is a range of treatment options for both finasteride and dutasteride, there is also a web of comparisons we can make when trying to compare the two. First, we’ll compare oral formulations. These are best researched, and there are a number of head-to-head comparison studies we can use to assess their effectiveness.
Oral Finasteride vs Oral Dutasteride
Here is a summary of the clinical trials that compare oral finasteride and dutasteride treatment directly:
Study Design and Population Treatments Impact on DHT Hair Growth Comparison Study #1[5]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading Randomized, placebo-controlled, double-blinded trial. 415 men with male pattern hair loss aged 21-45 years old; 24 weeks. 5 mg finasteride; 0.05, 0.1, 0.5, or 2.5 mg dutasteride; placebo. Serum DHT was significantly lower at 0.5 and 2.5 mg dutasteride than at 5 mg finasteride. 5mg finasteride decreased scalp DHT by 41%, while 0.5 and 2.5 mg dutasteride decreased it by 51% and 79% respectively. The proportion of participants with at least a 10% increase in hair counts was 41% for finasteride, and
48%, and 56% for 0.5 and 2.5 mg
dutasteride, respectively.
Study #2[6]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading Randomized, placebo-controlled, double-blinded trial. 917 men with AGA aged 20-50; 24 weeks. 1 mg finasteride; 0.02, 0.1, or 0.5 mg dutasteride; placebo Not assessed. 0.5 mg dutasteride significantly improved hair growth compared to 1 mg finasteride. Study #3[7]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading Open-label, randomized study; no placebo control. 90 men with AGA aged 18-40; 24 weeks. 1 mg finasteride; 0.5 mg dutasteride. Not assessed. Increase in total hair count was significantly higher in dutasteride compared to finasteride. Study #4[8]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading Retrospective chart review. 600 men over 18 with AGA. 1 mg finasteride; 0.5 mg dutasteride. Not assessed. Dutasteride improved BASP basic M classification of AGA significantly better than finasteride. These RCTs directly comparing finasteride and dutasteride provide the best insight into their comparative efficacy. We’ll focus on 1 mg finasteride vs. 0.5 mg as these are the most commonly used doses. While these studies differ in design and duration, their findings are consistent across several clinically meaningful outcomes.
Key Takeaways from Trials
- Across head-to-head trials lasting 24 weeks, dutasteride consistently outperforms finasteride in increasing hair counts within a predefined target scalp area.[9]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading[10]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading
- Dutasteride also increases average hair shaft thickness more than finasteride 1 mg over 24 weeks.
- In trials that included investigator global photo assessment (GPA) or similar global assessments, dutasteride-treated participants were more likely to be rated as “improved” or “markedly improved” compared with those receiving finasteride.[11]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading
An important limitation to consider when interpreting these trials is the length of the studies. Most head-to-head RCTs run for 24 weeks, which may favor dutasteride if it is faster acting. Finasteride, by contrast, is known from longer studies to continue producing gains for 48 months, with hair counts typically plateauing only after around 2 years.[12]Price, V. H., Menefee, E., Sanchez, M., & Kaufman, K. D. (2006). Changes in hair weight in men with androgenetic alopecia after treatment with finasteride (1 mg daily): three- and 4-year results. … Continue reading
However, long-term data, though mostly retrospective rather than randomized, still suggest sustained superiority of dutasteride on global classification scales.[13]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading
How Big is the Difference in Reality?
On paper, dutasteride often looks clearly superior to finasteride. But translating statistical superiority from randomized trials into real-world expectations requires more nuance. The practical difference between these drugs depends less on averages and more on what kind of hair loss someone has, how fast it’s progressing, and how much regrowth is biologically possible.
Safety & Side Effects: Is Dutasteride Actually Riskier?
Blocking 5AR interferes with hormone activity and is known to cause some side effects. The most frequently discussed effects include:
- Sexual side effects – These include decreased libido, erectile dysfunction, and ejaculation disorders. Across randomized trials, these effects occur in a minority of users (generally in the single-digit percentages), appear most often early in treatment, and frequently resolve spontaneously, even while continuing therapy.[14]Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic … Continue reading
- Breast-related effects – Breast tenderness or enlargement has been reported, but rates are low across both drugs and typically comparable between finasteride and dutasteride.
- Other reported effects: Fatigue, mood changes, or nonspecific symptoms are occasionally reported, but these occur inconsistently, lack clear dose–response relationships, and are difficult to separate from background rates and expectation effects.
As we’ve already noted, the dual action of dutasteride could mean that it poses an increased risk of side effects. To see whether the evidence backs this up, we’ll take a look at what randomized trials and retrospective studies tell us about the comparative risks of each treatment. Below is a summary of the head-to-head studies to date.
Average incidence of side effects Decreased libido Ejaculation disorders Erectile dysfunction Breast enlargement and tenderness Study #1[15]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading Finasteride 2.6% vs dutasteride 4.6% Finasteride – 5 mg: 4%. Dutasteride – 0.1 mg: 3%; 0.5 mg: 1%; 2.5 mg: 13%. Dutasteride – 0.1 mg: 4%; 0.5 mg: 1%; 2.5 mg: 13%. Finasteride – 5 mg: 1% Dutasteride – 0.1 mg: 0%; 0.5 mg: 0%; 2.5 mg: 0%. Finasteride – 5 mg: 1% Not assessed. Study #2[16]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading Finasteride 10.0% vs dutasteride 10.0% Not assessed. Not assessed. Not assessed. Not assessed. Study #3[17]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading Finasteride 13.4% vs dutasteride 11.5% Finasteride – 1 mg: 6.7%. Dutasteride – 0.02 mg: 8.1%; 0.1 mg: 6.9%; 0.5 mg: 3.3%. Finasteride – 1 mg: 3.9%. Dutasteride – 0.02 mg: 2.2%; 0.1 mg: 4.8%; 0.5 mg: 3.9%. Finasteride – 1 mg: 5.6%. Dutasteride – 0.02 mg: 4.3%; 0.1 mg: 3.7%; 0.5 mg: 5.6%. Finasteride – 1 mg: 0.6%. Dutasteride – 0.02 mg: 0.5%; 0.1 mg: 0.5%; 0.5 mg: 0.6%. Study #4[18]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading Finasteride 10.5% vs dutasteride 7.6% Finasteride 0.7% vs dutasteride 1.2% Not assessed Finasteride 0% vs dutasteride 0.4% Not assessed As we can see, across randomized and controlled studies comparing dutasteride and finasteride, the overall incidence of side effects is similar between the two drugs. When averaged across trials, total reported side effects occur at nearly identical rates (approximately 8-9% for both drugs).
In some studies, finasteride shows equal or higher rates of certain sexual side effects despite producing less DHT suppression. Importantly, these findings hold even when dutasteride reduces serum DHT by an additional 19-23% beyond finasteride and scalp DHT by significantly greater margins. The most closely scrutinized adverse effects, such as reduced libido, erectile dysfunction, and ejaculation disorders, do not follow a clear dose-response pattern with dutasteride.
Why Don’t We See More Side Effects With Dutasteride?
Given that dutasteride suppresses systemic and scalp DHT substantially more than finasteride, the lack of a clear increase in side effects may seem counterintuitive.
DHT reduction alone does not appear to be a reliable predictor of side-effect risk. This suggests that once DHT is reduced beyond a certain threshold, further suppression may not meaningfully increase the likelihood of side effects, particularly if compensatory mechanisms come into play. For example, lowering of DHT below a certain level might activate hormonal mechanisms to redress the balance. Long-term studies further support this idea, showing that side-effect incidence with dutasteride tends to decrease over time rather than accumulate.
Dutasteride’s size and tissue accessibility might also play a role. It has a much higher molecular weight (around 528 daltons) than finasteride (372 daltons), which likely limits its ability to cross the blood–brain barrier. Finasteride, by contrast, is small enough to enter brain tissue more readily. Because androgen metabolism in the brain is implicated in libido and sexual function, a drug that primarily exerts its effects in peripheral tissues may suppress DHT more aggressively without proportionally increasing neurological side effects.
Why do Incidence Estimates Vary?
Reported rates of side effects with finasteride and dutasteride vary widely across studies largely because incidence is highly dependent on study design. The makeup of the participant groups can impact how likely they are to experience side effects. How side effects are defined and recorded can also influence estimates. Some studies rely on spontaneous self-reporting, while others use targeted questionnaires or direct investigator questioning. These different approaches produce different reporting rates.
Psychological and selection factors further complicate interpretation. Participants who expect problems are more likely to notice and report them, even in placebo groups (this is called the nocebo effect). This means the information that participants receive can impact side effects.
How Long-Lasting are Side Effects?
Most randomized controlled trials are short (e.g., 24 weeks), enroll relatively healthy participants, and use structured adverse-event reporting. This means they tend to capture early, transient side effects, while missing longer-term patterns such as spontaneous resolution or adaptation. In contrast, observational and retrospective studies follow broader populations over longer periods but rely on less standardized reporting, which can inflate or obscure true incidence.
As a result, short-term trials may overestimate persistent risk, while long-term datasets may underrepresent early intolerance.
Where trials discuss the resolution of side effects, one trial reported that around half of affected participants taking dutasteride reported spontaneous resolution of symptoms while continuing treatment.[19]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading By contrast, finasteride-associated side effects in these trials were less likely to resolve during ongoing treatment, even though many resolved after stopping the drug.
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Post-Finasteride Syndrome: What to Know
Post-Finasteride Syndrome (PFS) is a term used to describe persistent sexual, cognitive, or mood-related symptoms that continue for months after stopping finasteride. Its existence and prevalence remain highly debated among dermatologists, endocrinologists, and hair loss researchers.
While some clinicians argue that PFS represents a real, drug-induced condition affecting a very small subset of users, others contend that the available evidence is low quality, heavily confounded by psychological factors, media influence, and the high background rates of sexual dysfunction in adult men.[20]Traish, A. M. (2020). Post-finasteride syndrome: a surmountable challenge for clinicians. *Fertility and Sterility.* 113(1). 21–50. Available at: https://doi.org/10.1016/j.fertnstert.2019.11.030,[21]Trüeb, R. M., Régnier, A., Rezende, H. D., & Dias, M. F. R. G. (2019). Post-finasteride syndrome: an induced delusional disorder with the potential of a mass psychogenic illness?. *Skin … Continue reading
Critically, high-quality clinical trials of finasteride did not report persistent side effects after discontinuation, with symptoms resolving within three months in all affected participants.[22]Shapiro, J., & Kaufman, K. D. (2003). Use of finasteride in the treatment of men with androgenetic alopecia (male pattern hair loss). *Journal of Investigative Dermatology Symposium Proceedings.* … Continue reading Claims of PFS largely arise from case reports, small observational studies, and self-reported data, all of which suffer from major limitations such as lack of controls and small sample sizes.
One important exception is finasteride-induced gynecomastia (enlarged breast tissue in males), which is a well-documented side effect and can persist in a minority of cases even after stopping the drug, demonstrating that long-lasting tissue changes are possible, albeit rare.[23]Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has … Continue reading
If PFS is real, it is likely extremely rare, plausibly affecting fewer than 1 in 5,000 – 10,000 users. For users, the key takeaway is one of risk context: while persistent side effects cannot be ruled out entirely, their estimated risk appears very low when weighed against the well-established benefits of finasteride for AGA, especially when dosing strategies are used to minimize overall exposure.
Topical Formulations
After oral formulations, the next logical question is whether you can get the same DHT-blocking benefits where it matters most. That’s the core promise of topical finasteride and dutasteride formulations: to localize 5AR inhibition to the scalp and reduce the likelihood of systemic side effects.
The potential for using topical formulations opens up a host of new options for treating AGA. Like oral formulations, topicals can also come in a range of concentrations, but can also be delivered in a range of formulations and solutions, including:
- Solutions and Sprays – Alcohol‑ or hydroalcoholic‑based liquids applied with a dropper, spray, or pipette, sometimes combined with propylene glycol or similar penetration enhancers.
- Gels and Lotions – Semi‑solid preparations designed to increase contact time on the scalp and modulate absorption.
- Nanoparticle and Liposomal Carriers: Research formulations load finasteride or dutasteride into lipid or polymeric nanoparticles or liposomes to target follicular delivery and reduce systemic exposure.
Formulations can also be combined with physical methods, such as microneedling, or with other common pharmaceuticals, like minoxidil. This also means that the decisions we need to make when choosing the right hair loss treatment are more complicated. Again, we’ll focus on head-to-head trials that compare different topical and oral formulations, as well as what clinical studies can tell us about side effects.
Does Topical Finasteride Work?
Based on trials comparing topical dutasteride to other AGA treatments, the answer is a resounding yes. However, the challenge is that finasteride has a highly sensitive, logarithmic dose-response curve. In other words, you don’t need much finasteride in the bloodstream to create a big systemic DHT change. That’s why topical finasteride can still “leak” systemically, and why even small systemic absorption can defeat the original purpose of going topical.
To take a closer look, we’ll first see how topical formulations compare to oral.
Oral vs Topical Finasteride
Several trials directly comparing topical to oral finasteride show that topical treatment can improve target-area hair counts and other hair parameters similarly to oral dosing, while often producing less suppression of serum DHT and far lower measured systemic drug exposure.
In one Phase III trial, topical finasteride produced hair-count improvements comparable to oral finasteride. What’s more, the oral formulation reduced mean serum DHT by 55.6%, whereas topical finasteride reduced it by 34.5%, and sexual side effects were more associated with oral treatment.[24]Piraccini, B. M., Blume-Peytavi, U., Scarci, F., Jansat, J. M., Falqués, M., Otero, R., Tamarit, M. L., et al. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic … Continue reading
Other randomized comparisons similarly report meaningful regrowth with topical finasteride, with most adverse events being local (irritation) and relatively few systemic complaints.[25]Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of … Continue reading
However, even at lower concentrations, topical finasteride can reduce serum DHT depending on how much you apply and how often you apply it. For example, one versus two daily applications of the same topical concentration can produce dramatically different serum DHT suppression, illustrating that frequency and volume can matter as much as concentration.[26]Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen … Continue reading
Figure 2. Multiple daily doses of topical finasteride can reduce serum DHT more than a single dose. Adapted from Figure 3.[27]Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen … Continue reading
What Determines Systemic Absorption?
There are a few key factors that impact absorption:
- Vehicle/carrier – Alcohol- and propylene glycol–based carriers tend to enhance penetration. Stronger penetration can improve scalp delivery, but it can also increase systemic leakage.
- Amount applied per use – This is often the most overlooked variable. Two people using the same concentration can have totally different systemic exposure if one applies 0.5 mL and the other applies 2 mL.
- Frequency of application – Twice-daily use can more than double systemic effects compared to once-daily use in certain protocols.
Does Topical Dutasteride Work?
Topical dutasteride research is newer and still developing, but early findings are encouraging. The current literature suggests that topical dutasteride can meaningfully improve hair density and shaft caliber while producing minimal changes in serum DHT.
A randomized, placebo-controlled trial in 34 men found that microneedling plus 0.01% topical dutasteride produced significant improvements in hair thickness, density, and the vellus-to-terminal ratio, compared with microneedling plus saline.[28]Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M. E., Ocampo-Garza, J., Orizaga-y-Quiroga, T. L., Martínez-Moreno, A., & Ocampo-Garza, S. S. (2022). Microneedling plus … Continue reading
This is only a proof-of-concept study with a small sample size. However, it does demonstrate that topical dutasteride can drive clinically meaningful changes when follicular delivery is boostedby microneedling. But it also limits generalizability: microneedling itself is an active intervention, and it may not reflect what happens with topical dutasteride alone.
A newer Phase II trial tested 0.01%, 0.02%, and 0.05% topical dutasteride and reported dose-dependent improvements in total hair count and hair width versus placebo.[29]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading
The authors also reported that 0.05% appeared to outperform oral finasteride in total hair count. That’s an attention-grabbing claim, but it should be interpreted cautiously. Unusually high baseline counts and possible inconsistencies in target-area placement, which could inflate apparent differences, raise concerns about the methodology used.
Figure 3. Oral finasteride (yellow line) reduces serum DHT levels more than topical Dutaseride (blue lines). Adapted from Figure 3.[30]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading Image used under Creative Commons License
A common objection is that dutasteride is too large to penetrate the skin well. At around 528.5 Daltons, it sits slightly above the widely cited 500 Dalton rule, which suggests that larger molecules struggle to cross the stratum corneum. In practice, dutasteride appears capable of penetrating the scalp, as it can suppress DHT and promote hair growth.
Combination strategies: where topical regimens often perform best
Topical regimens tend to perform best when they’re used alongside other treatments, rather than as a single agent. DHT drives miniaturization, but follicular stimulation, blood-flow signaling, and wound-healing pathways all influence whether a follicle can thicken and re-enter robust growth.
Microneedling consistently appears in the literature as an accelerator of topical outcomes. Controlled micro-injury activates wound-healing signals that can independently improve density and shaft diameter, while also enhancing follicular penetration and local drug exposure.
Combination with other treatments, primarily minoxidil, can also improve outcomes. The underlying logic is straightforward: minoxidil boosts growth, while dual 5AR blockade increases the probability that follicles remain protected from hormonal changes.
One study showed that adding microneedling to topical finasteride therapy alongside minoxidil can increase hair density and shaft diameter more than minoxidil alone.[31]Chang, Y., Zhang, W., Zhou, J., Lv, L., He, Q., Chen, Y., Wang, P., & Zhai, Q. (2025). Clinical efficacy of microneedle combined with 5% minoxidil solution and finasteride in the treatment of … Continue reading
Similarly, combining dutasteride with microneedling and minoxidil has also demonstrated promising results. In a small study, all patients used topical minoxidil 5%, while adding dutasteride, either systemically or locally, significantly improved hair density and shaft caliber compared with minoxidil alone. The strongest overall results were seen with topical dutasteride 0.02% delivered via monthly microneedling, which outperformed oral dutasteride plus minoxidil on objective measures while maintaining a more favorable safety profile.[32]Abu Obeid, M. N., Abdel Fattah, N. S., Elfangary, M. M., & Al Husseni, R. M. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through … Continue reading
Overall, the evidence suggests that topical regimens may be most effective when they combine anti-androgen suppression, follicular stimulation, and delivery-enhancing strategies.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Dosing
Choosing the right dose depends on both the medication and the clinical context. Finasteride, whether oral or topical, is generally the best starting point for most men beginning pharmacologic treatment for androgenetic alopecia. Its pharmacokinetics are more forgiving, dose adjustments are easier, and when side effects occur, they are often manageable with modest reductions in dose or changes in dosing frequency.
Dutasteride, in contrast, is better suited for men with aggressive, fast-progressing AGA or for those who have not responded well to finasteride. Its greater potency comes with less flexibility, largely due to its long half-life and sustained enzyme binding. As a result, dosing with dutasteride should be conservative and deliberate, with careful consideration of long-term exposure and tolerability.
Oral Finasteride
The standard dose of oral finasteride for androgenetic alopecia is 1 mg once daily. This regimen has been used in the majority of randomized controlled trials and has consistently demonstrated improvements in key hair growth outcomes, including hair counts, hair shaft caliber, and investigator global photographic assessments.
A key pharmacologic feature of finasteride is its logarithmic dose–response curve. Most of the drug’s DHT-lowering effect occurs at relatively low doses, and by the time a daily dose of 1 mg is reached, inhibition of type II 5-alpha reductase is already close to maximal.
This explains why lower doses, such as 0.2–0.5 mg daily, can still meaningfully suppress both serum and scalp DHT, and why increasing the dose beyond 1 mg does not lead to proportionally greater hair regrowth. Instead, higher doses primarily increase systemic exposure and may raise the risk of side effects without offering additional cosmetic benefit.
Figure 4. The dose response of finasteride demonstrates that increasing the concentration of treatment doesn’t translate to increased DHT reduction.
From a practical standpoint, this dose–response relationship allows for flexibility in real-world use. Many individuals are able to maintain hair stabilization and regrowth with reduced dosing or alternate-day schedules, particularly after an initial period of stabilization.
When side effects do occur, they also tend to be dose-sensitive rather than all-or-nothing, meaning that lowering the dose or adjusting the dosing schedule is often sufficient to improve tolerability while preserving therapeutic benefit.
Oral Dutasteride
The standard oral dose of dutasteride for androgenetic alopecia is 0.5 mg once daily. This is the dose most consistently studied in clinical trials and the dose used in nearly all head-to-head comparisons with finasteride.
The reason dutasteride is prescribed at a lower milligram dose than finasteride comes down to its greater pharmacologic potency. Dutasteride’s pharmacokinetics are also different. The drug has a longer half-life (roughly 4–5 weeks), which means it accumulates in tissues over time.[33]Arif, T., Dorjay, K., Adil, M., & Sami, M. (2017). Dutasteride in androgenetic alopecia: an update. *Current Clinical Pharmacology.* 12(1). 31–35. Available at: … Continue reading
Because of this, missed doses tend to matter less for maintaining efficacy. However, the flip side is that if adverse effects occur, they may take weeks to months to fully resolve after discontinuation, as the drug slowly clears from the body.
These properties have important practical implications for dosing. Daily dosing is not always necessary to maintain clinical benefit, and some clinicians employ intermittent schedules, such as 0.5 mg taken two to three times per week, in an effort to balance efficacy with tolerability.
Topical Finasteride
Topical finasteride dosing is often misunderstood because the percentage concentration listed on the label is not the most important variable. What ultimately determines both efficacy and systemic risk is total daily finasteride exposure, measured in milligrams per day.
This total exposure is shaped by several interacting factors: the concentration of finasteride in the solution, the volume applied per use, how often it is applied (once versus twice daily), and the vehicle or carrier used, such as alcohol or propylene glycol, which influences skin penetration.
The key principle underlying topical finasteride dosing is that finasteride is extremely potent systemically. Only a very small fraction of the drug needs to enter circulation to meaningfully suppress serum DHT. As a result, topical formulations can unintentionally behave like oral finasteride if dosing is too aggressive, even when the product is intended to act locally on the scalp.
An evidence-informed dosing range is 1-2 mL of a 0.005%-0.02% topical finasteride solution applied once daily. This corresponds to roughly 0.1-0.2 mg of finasteride applied to the scalp per day, with estimated systemic exposure in common carriers of approximately 0.01-0.03 mg per day.
Generally, high-concentration formulations (≥0.25-1%) should be avoided, especially when combined with large application volumes or twice-daily use. These regimens can suppress serum DHT to a degree similar to 1 mg of oral finasteride, effectively defeating the purpose of choosing a topical formulation in the first place.
Topical Dutasteride
Topical dutasteride dosing is less standardized than topical finasteride, largely because the evidence base is newer and consists of fewer clinical trials. That said, several consistent patterns are beginning to emerge from the available data.
Most studies have evaluated topical dutasteride in a concentration range of 0.01% to 0.05%, typically applied once daily. In many protocols, topical dutasteride has been paired with microneedling to enhance follicular delivery, although some studies suggest that meaningful efficacy can be achieved even without microneedling.[34]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading
The practical takeaway is that low-concentration, low-volume topical dutasteride protocols appear to offer the best balance between efficacy and safety. Given the variability in formulations, vehicles, and individual skin permeability, optimal dosing is likely individualized, and careful titration remains essential as the clinical evidence continues to evolve.
Monitoring & Risk Management
Monitoring and risk management with finasteride or dutasteride are about using the minimum effective exposure while staying alert to tolerability over time. Ideally, treatment begins with a clear diagnosis of androgenetic alopecia, baseline photos, and, especially for topical or risk-averse users, optional baseline serum DHT testing to provide context for future changes.
Side effects, if they occur, should be addressed early with dose reduction, spacing doses, or switching delivery routes rather than abrupt discontinuation, since most are dose-sensitive and reversible. For topical users, repeat serum DHT testing after ~30 days can help confirm that systemic exposure remains low.
Ongoing monitoring should focus on objective progress via standardized photos every 3–6 months and general symptom awareness rather than day-to-day fluctuations.
Extra caution is warranted with dutasteride due to its long half-life and accumulation, making conservative dosing and deliberate adjustments especially important. When approached with planned checkpoints and proportional responses, both drugs can be used safely, predictably, and effectively over the long term.
Who Should Use Finasteride or Dutasteride?
Finasteride and dutasteride are among the most effective pharmacologic options for AGA, but they are not one-size-fits-all treatments. Choosing which drug to use depends on hair-loss pattern, rate of progression, prior treatment response, side-effect tolerance, and long-term goals.
Both drugs require consistent, long-term use to maintain results. Candidates should be comfortable with ongoing therapy and realistic timelines, as visible improvements often take 6-12 months, with maximal benefits closer to two years.
Consider Finasteride If…
You’re starting treatment, or your AGA is early-to-moderate. Oral finasteride is the usual baseline because it’s effective for most men and has a long track record. It’s also more forgiving: if you need to adjust dose, frequency, or route due to side effects, finasteride is generally easier to fine-tune.
If you’re risk-averse or simply want the most studied on-label option for hair loss, finasteride is usually the logical starting point, especially if you can commit to consistent use and track progress with photos over months.
Consider Dutasteride If…
Your hair loss is aggressive, or you’re losing ground despite finasteride. Dutasteride’s broader 5AR inhibition often translates into greater average gains in hair counts and thickness in shorter head-to-head trials. Clinically, it’s most often considered when progression is rapid, miniaturization is extensive, or finasteride hasn’t provided adequate stabilization after a consistent trial.
Who Should be Cautious (or Avoid)?
Men without androgenetic alopecia – Finasteride and dutasteride are ineffective for conditions such as alopecia areata, telogen effluvium, traction alopecia, or scarring alopecias. Using them in these contexts adds risk without meaningful benefit.
Those highly risk-averse to hormonal manipulation – While most users tolerate these medications well, anyone uncomfortable with altering systemic hormone levels, even at low risk, may prefer non-hormonal options or topical-only approaches with careful dosing.
Men who experienced significant adverse effects previously – Dutasteride may be more appropriate for men with rapid progression, extensive miniaturization, or those who continue to lose ground despite consistent finasteride use. Its stronger and broader DHT suppression can provide an additional margin of control in these cases.
Men trying to conceive in the near term – Although evidence of fertility harm at hair-loss doses is limited, many clinicians advise avoiding systemic 5AR inhibitors when actively trying to conceive, particularly dutasteride due to its long half-life. This decision should be individualized and discussed with a clinician.
Those unable to maintain consistency – Oral therapies require daily (or structured intermittent) dosing, while topical therapies demand regular application and attention to volume and frequency. If adherence is unlikely, expected benefits drop substantially.
Final Thoughts and Takeaways
Finasteride and dutasteride, in their different formulations, are not competing “good vs bad” options. Instead, they are different tools for different stages and severities of androgenetic alopecia.
The real decision isn’t which drug suppresses more DHT on paper, but how much suppression you need to control your hair loss, and how much systemic exposure you’re comfortable carrying long term.
Finasteride has the longest safety record, strong long-term efficacy, and a forgiving dose–response curve that allows real-world flexibility. For early-to-moderate AGA, or for men prioritizing reversibility and adjustability, finasteride, either oral or carefully dosed topical, covers the majority of clinical needs.
When hair loss is aggressive, rapidly progressive, or insufficiently controlled on finasteride, dutasteride’s broader 5AR inhibition offers a higher ceiling of protection. Head-to-head trials consistently show greater average gains, and it does not show a clearly higher overall incidence of side effects in controlled studies despite stronger systemic and scalp DHT reduction.
Topical vs oral delivery, total daily exposure, application volume, and frequency often have a larger impact on outcomes and tolerability than the finasteride-vs-dutasteride label alone. Hair loss therapy is a long game. The optimal choice is not the strongest drug you can tolerate for a few months, but the strategy you can follow consistently for years with acceptable trade-offs.
References[+]
References ↑1 Asfour, L., Cranwell, W., & Sinclair, R. (2023). Male androgenetic alopecia. *Endotext [Internet].* Available at: https://www.ncbi.nlm.nih.gov/books/NBK278957/ ↑2 Makridakis, N., & Reichardt, J. K. V. (2005). Pharmacogenetic analysis of human steroid 5α-reductase type II: comparison of finasteride and dutasteride. *Journal of Molecular Endocrinology.* 34(3). 617–623. Available at: https://doi.org/10.1677/jme.1.01725 ↑3 Frye, S. V. (2006). Discovery and clinical development of dutasteride, a potent dual 5α-reductase inhibitor. *Current Topics in Medicinal Chemistry.* 6(5). 405–421. Available at: https://doi.org/10.2174/156802606776743101 ↑4 Wikimedia Commons. (n.d.). Azasteroid Pharmaceuticals [Image]. *Wikimedia Commons.* Available at: https://commons.wikimedia.org/wiki/File:Azasteroid_Pharmaceuticals.png (Accessed: November 2025) ↑5, ↑15, ↑19 Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. *Journal of the American Academy of Dermatology.* 55(6). 1014–1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007 ↑6, ↑10, ↑16 Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 70(3). 489–498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑7, ↑17 Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. *Indian Journal of Dermatology, Venereology and Leprology.* 83. 47. Available at: https://doi.org/10.4103/0378-6323.188652 ↑8, ↑11, ↑13, ↑18 Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male androgenic alopecia in South Korea: a multicentre chart review study. *Annals of Dermatology.* 34(5). 349. Available at: https://doi.org/10.5021/ad.22.027 ↑9 Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. *Journal of the American Academy of Dermatology.* 55(6). 1014–1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007 ↑12 Price, V. H., Menefee, E., Sanchez, M., & Kaufman, K. D. (2006). Changes in hair weight in men with androgenetic alopecia after treatment with finasteride (1 mg daily): three- and 4-year results. *Journal of the American Academy of Dermatology.* 55(1). 71–74. Available at: https://doi.org/10.1016/j.jaad.2005.07.001 ↑14 Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic review. *The Journal of Clinical and Aesthetic Dermatology.* 9(7). 56–62. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC5023004/ ↑20 Traish, A. M. (2020). Post-finasteride syndrome: a surmountable challenge for clinicians. *Fertility and Sterility.* 113(1). 21–50. Available at: https://doi.org/10.1016/j.fertnstert.2019.11.030 ↑21 Trüeb, R. M., Régnier, A., Rezende, H. D., & Dias, M. F. R. G. (2019). Post-finasteride syndrome: an induced delusional disorder with the potential of a mass psychogenic illness?. *Skin Appendage Disorders.* 5(5). 320–326. Available at: https://doi.org/10.1159/000497362 ↑22 Shapiro, J., & Kaufman, K. D. (2003). Use of finasteride in the treatment of men with androgenetic alopecia (male pattern hair loss). *Journal of Investigative Dermatology Symposium Proceedings.* 8(1). 20–23. Available at: https://doi.org/10.1046/j.1523-1747.2003.12167.x ↑23 Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has comparable urinary symptom efficacy and prostate volume reduction, but less sexual side effects and breast complications than dutasteride. *International Journal of Clinical Practice.* 66(11). 1052–1055. Available at: https://doi.org/10.1111/j.1742-1241.2012.03010.x ↑24 Piraccini, B. M., Blume-Peytavi, U., Scarci, F., Jansat, J. M., Falqués, M., Otero, R., Tamarit, M. L., et al. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. *Journal of the European Academy of Dermatology and Venereology.* 36(2). 286–294. Available at: https://doi.org/10.1111/jdv.17738 ↑25 Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of Dermatology, Venereology and Leprology.* 75. 47. Available at: https://doi.org/10.4103/0378-6323.45220 ↑26 Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen levels in healthy male volunteers. *International Journal of Clinical Pharmacology and Therapeutics.* 52(10). 842–849. Available at: https://doi.org/10.5414/CP202119 ↑27 Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen levels in healthy male volunteers. *International Journal of Clinical Pharmacology and Therapeutics.* 52(10). 842–849. Available at: https://doi.org/10.5414/CP202119 ↑28 Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M. E., Ocampo-Garza, J., Orizaga-y-Quiroga, T. L., Martínez-Moreno, A., & Ocampo-Garza, S. S. (2022). Microneedling plus topical dutasteride solution for androgenetic alopecia: a randomized placebo-controlled study. Journal of the European Academy of Dermatology & Venereology. 36(10). Available at: https://doi.org/10.1111/jdv.18285 ↑29 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑30 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. *Cureus.* 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑31 Chang, Y., Zhang, W., Zhou, J., Lv, L., He, Q., Chen, Y., Wang, P., & Zhai, Q. (2025). Clinical efficacy of microneedle combined with 5% minoxidil solution and finasteride in the treatment of androgenetic alopecia in males. *Archives of Dermatological Research.* 317(1). 428. Available at: https://doi.org/10.1007/s00403-025-03891-y ↑32 Abu Obeid, M. N., Abdel Fattah, N. S., Elfangary, M. M., & Al Husseni, R. M. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through microneedling or oral 0.5 mg) in treatment of androgenetic alopecia. *QJM: An International Journal of Medicine.* 117(Supplement_2). hcae175–207. Available at: https://doi.org/10.1093/qjmed/hcae175.207 ↑33 Arif, T., Dorjay, K., Adil, M., & Sami, M. (2017). Dutasteride in androgenetic alopecia: an update. *Current Clinical Pharmacology.* 12(1). 31–35. Available at: https://doi.org/10.2174/1574884712666170310111125 ↑34 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 Finasteride is one of the most effective and widely studied treatments for androgenic alopecia (AGA). It works by lowering dihydrotestosterone (DHT) levels at the scalp to slow or halt androgen-driven hair follicle miniaturization. While most men tolerate finasteride well and experience meaningful, long-term hair preservation, side effects can occur in a minority of users and are a legitimate concern for anyone considering or already using the medication.
Importantly, tolerance to finasteride varies from person to person. Factors such as baseline hormone levels, genetics, lifestyle, and overall health can influence how an individual responds. The goal of this article is to outline practical, evidence-informed strategies to help manage or reduce side effects, enabling thoughtful, safe decisions about finasteride use with appropriate medical guidance.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Common Finasteride Side Effects
Sexual side effects, such as decreased libido, erectile dysfunction, and ejaculation disorders, occur in a small minority of users. They are most commonly reported early in treatment and often improve or resolve on their own.[1]Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic … Continue reading
Breast tenderness and enlargement (gynecomastia) are sometimes reported. Gynecomastia in particular is well-documented and can persist in a minority of cases even after stopping the drug.[2]Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has … Continue reading
Some men report cognitive symptoms while taking finasteride, most commonly described as “brain fog” or reduced mental clarity, but these complaints have not been observed at meaningful rates in controlled clinical trials. If such effects occur, they are likely subtle and gradual, which may make them difficult to quantify or consistently attribute to the medication
There is a small number of cases where sexual, cognitive, or mood-related symptoms have persisted, leading to the development of the term post-finasteride syndrome (PFS).
PFS remains controversial: some clinicians consider it a genuine but extremely rare drug-related condition, while others argue that existing evidence is low quality and heavily confounded by psychological factors, reporting bias, and the high baseline prevalence of sexual dysfunction in men.[3]Traish, A. M. (2020). Post-finasteride syndrome: a surmountable challenge for clinicians. *Fertility and Sterility.* 113(1). 21–50. Available at: https://doi.org/10.1016/j.fertnstert.2019.11.030,[4]Trüeb, R. M., Régnier, A., Rezende, H. D., & Dias, M. F. R. G. (2019). Post-finasteride syndrome: an induced delusional disorder with the potential of a mass psychogenic illness?. *Skin … Continue reading
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How common are side effects?
It’s important to remember that the majority of men who use finasteride experience no noticeable side effects,
In the original large, randomized, placebo-controlled trial that led to FDA approval for AGA, sexual side effects were reported in about 4.2% of men taking finasteride versus 2.2% on placebo in the first year. This suggests a small absolute risk, and participants in the study who stopped taking finasteride due to side effects reported that they did not persist after the medication was discontinued.[5]Kaufman, K. D., Olsen, E. A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V. H., et al. (1998). Finasteride in the treatment of men with androgenetic alopecia. *Journal of the American … Continue reading
In other trials, rates vary widely across studies depending on study design, definitions, and participant expectations.
Higher rates seen in some studies appear to be strongly influenced by the nocebo effect, where awareness of potential side effects increases reporting. More conservative analyses estimate that up to 10-15% of users may experience some form of sexual symptoms, most of which are mild, transient, and reversible.[6]Traish, A. M., Hassani, J., Guay, A. T., Zitzmann, M., & Hansen, M. L. (2011). Adverse side effects of 5α-reductase inhibitors therapy: persistent diminished libido and erectile dysfunction and … Continue reading
The Most Important Rule: Speak to a Physician
If you get severe side effects, stop and speak to your primary care physician.
Early evaluation helps rule out other medical, hormonal, or psychological contributors, establishes whether symptoms are plausibly related to finasteride, and prevents unnecessary continuation of treatment when modification or discontinuation is appropriate.
Prompt medical guidance also ensures that side effects are documented and managed correctly, rather than being compounded by anxiety, misinformation, or unsupervised dose changes. The strategies outlined below apply only to mild or moderate symptoms, should be considered only under clinician supervision, and are not appropriate for severe, rapidly worsening, or distressing effects, which warrant immediate medical review.
With that in mind, we can now look at ways to reduce the side effects of finasteride.
1. Continue Treatment and Allow Time for Adaptation (When Symptoms Are Mild)
One of the most consistently observed patterns with finasteride and other 5α-Reductase (5AR) inhibitors is that many reported side effects improve or resolve with continued use, particularly when they are mild and occur early in treatment.
Clinical trials and follow-up data also suggest the side effects are most common in the first 12 months. This pattern supports the idea that early side effects often reflect a transient adjustment phase rather than a permanent intolerance.[7]Lowe, F. C., McConnell, J. D., Hudson, P. B., Romas, N. A., Boake, R., Lieber, M., Elhilali, M., et al. (2003). Long-term 6-year experience with finasteride in patients with benign prostatic … Continue reading,[8]Hudson, P. B., Boake, R., Trachtenberg, J., Romas, N. A., Rosenblatt, S., Narayan, P., Geller, J., et al. (1999). Efficacy of finasteride is maintained in patients with benign prostatic hyperplasia … Continue reading
Several biological mechanisms may explain this improvement. Androgen receptors and downstream hormone signaling can adapt to reduced DHT levels, central nervous system sensitivity to hormonal shifts may normalize, and early symptoms may be amplified by anxiety or expectancy effects that lessen with reassurance and time.
2. Reduce the Daily Dose
The standard dose of finasteride for AGA is 1 mg once daily, a regimen supported by large randomized controlled trials showing clear improvements in hair count, hair shaft thickness, and global photographic assessments.
However, a key pharmacologic feature of finasteride is its non-linear (logarithmic) dose–response curve, meaning that most of its DHT–lowering effect occurs at relatively low doses.
Figure 1. The dose response of finasteride demonstrates that increasing the concentration of treatment doesn’t translate to increased DHT reduction.
By the time a daily dose of 1 mg is reached, inhibition of 5AR is already near maximal. As a result, lower doses, such as 0.2–0.5 mg daily, can still meaningfully suppress serum and scalp DHT, often preserving much of finasteride’s hair-protective benefit. [9]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W. P., Cotterill, P. C., Thiboutot, D. M., et al. (1999). The effects of finasteride on scalp skin and serum androgen levels in men with … Continue reading
Increasing the dose beyond 1 mg does not produce proportionally greater hair regrowth, but instead primarily increases systemic exposure, which may raise the likelihood of side effects without additional cosmetic gain.
From a practical standpoint, this dose–response relationship allows for flexibility in real-world use. Because finasteride side effects tend to be dose-sensitive rather than all-or-nothing, reducing the daily dose is often an effective first adjustment for improving tolerability while maintaining therapeutic efficacy.
3. Reduce Dosing Frequency
Finasteride’s pharmacology also allows for flexibility in dosing frequency. The drug binds tightly to 5AR, and suppression of DHT persists well beyond the drug’s short plasma half-life.[10]National Center for Biotechnology Information. (n.d.). Finasteride. *NCBI Bookshelf.* Available at: https://www.ncbi.nlm.nih.gov/books/NBK513329/ (Accessed: November 2025),[11]Steiner, J. F. (1996). Clinical pharmacokinetics and pharmacodynamics of finasteride. *Clinical Pharmacokinetics.* 30(1). 16–27. Available at: https://doi.org/10.2165/00003088-199630010-00002 In practical terms, this means that daily dosing is not strictly required to maintain meaningful inhibition of scalp DHT once steady-state suppression has been achieved.
Figure 2. Multiple doses of oral finasteride (square points) do not reduce serum DHT significantly more than a single dose over 36 hours. Adapted from Figure 3.[12]Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen … Continue reading
Common clinician-guided intermittent regimens include 1 mg three times per week or 0.5 mg every other day, both of which can significantly reduce cumulative drug exposure while preserving much of finasteride’s therapeutic effect. Because DHT levels recover gradually rather than immediately, these schedules often maintain adequate androgen suppression for hair stabilization.
Intermittent dosing may be especially helpful for men who develop mild side effects on daily therapy, those who are particularly sensitive to hormonal changes, or individuals transitioning into a long-term maintenance phase after initial hair loss control.
If you want to understand more about finasteride dosing, you can read our in-depth article here.
4. Switch to Topical Finasteride
Topical finasteride is designed to reduce DHT locally within the scalp while limiting systemic exposure, making it an attractive option for men who experience side effects with oral therapy. By delivering the drug directly to the scalp, topical formulations aim to suppress DHT activity in hair follicles without relying on sustained systemic circulation.
Randomized trials comparing topical and oral finasteride show that topical treatment can produce similar improvements in target-area hair counts and other hair growth parameters, while generally causing less suppression of serum DHT and lower measured systemic drug levels.[13]Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of … Continue reading
One trial demonstrated that topical finasteride reduced DHT in the blood by 34.5%, compared to 55.6% with oral finasteride. They also found that sexual side effects were more associated with oral treatment.[14]Piraccini, B. M., Blume-Peytavi, U., Scarci, F., Jansat, J. M., Falqués, M., Otero, R., Tamarit, M. L., et al. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic … Continue reading
That said, finasteride’s highly sensitive, logarithmic dose–response curve means that even small amounts entering the bloodstream can suppress DHT. Total systemic exposure depends not only on concentration, but also on application volume, frequency, treated surface area, and formulation vehicle.
As such, topical treatments also require care when applying, and success with topical finasteride depends on consistent, measured application and adherence to the prescribed regimen.
When used thoughtfully, topical finasteride can preserve much of the hair benefit of oral therapy while reducing the risk of systemic side effects for many men.
Find out more about topical finasteride in our article.
5. Use Ultra–Low-Dose Topical Finasteride
As we’ve already seen, even low doses of finasteride can have a significant impact on hair growth. For men who are particularly sensitive to finasteride or who experience side effects even with standard topical formulations, ultra–low-dose topical finasteride may offer a further risk-reduction strategy.
These formulations typically use very low concentrations (e.g., 0.005%), aiming to provide localized scalp DHT suppression while minimizing systemic exposure as much as possible. Supporting this strategy, a small clinical study of 0.005% alcohol-based topical finasteride found appreciable changes in serum DHT levels, indicating little-to-no systemic absorption. Despite minimal systemic exposure, participants still experienced meaningful hair growth outcomes, suggesting that even ultra-low topical doses can be effective at the scalp level.[15]Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia: preliminary evaluations after a … Continue reading
Ultra–low-dose topical finasteride has not been studied extensively in large randomized controlled trials. Observationally, many users report improved tolerability and stable hair outcomes, but responses are variable, and efficacy may be more modest than with higher doses.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

6. Use PDE5 Inhibitors to Address Sexual Side Effects
For men who develop erectile dysfunction while using finasteride, PDE5 inhibitors (such as sildenafil or tadalafil) can be an effective symptom-management option. These medications are well studied in urology, including in men treated with 5AR inhibitors for benign prostatic hyperplasia (BPH).[16]Casabé, A., Roehrborn, C. G., Da Pozzo, L. F., Zepeda, S., Henderson, R. J., Sorsaburu, S., Henneges, C., Wong, D. G., & Viktrup, L. (2014). Efficacy and safety of the coadministration of … Continue reading
In that setting, PDE5 inhibitors have consistently been shown to improve erectile performance and sexual confidence, even when the underlying hormonal environment is altered.
It’s important to be clear about what this strategy does (and doesn’t) do. PDE5 inhibitors do not reverse DHT suppression or directly address libido changes or ejaculatory volume in the same way they may improve erectile rigidity. However, for many men, symptom relief can meaningfully reduce distress and help prevent performance anxiety from amplifying the problem.
Because PDE5 inhibitors are prescription medications with cardiovascular considerations and potential drug interactions, they should be used only under clinician supervision, particularly in men with heart disease, low blood pressure, or those taking nitrates or certain alpha-blockers.[17]Lui, J. L., Shaw, N. M., Abbasi, B., Hakam, N., & Breyer, B. N. (2023). Adverse reactions of PDE5 inhibitors: an analysis of the World Health Organization pharmacovigilance database. *Andrology.* … Continue reading
7. Optimize Testosterone
Testosterone plays a vital role in sexual health and, like finasteride, is associated with a wide range of symptoms. Reduced libido, erectile changes, fatigue, or low mood overlap significantly with those of hypogonadism, thyroid dysfunction, or other hormonal imbalances.[18]National Center for Biotechnology Information. (n.d.). Dutasteride. *NCBI Bookshelf.* Available at: https://www.ncbi.nlm.nih.gov/books/NBK532933/ (Accessed: November 2025)
Improving total and free testosterone levels through lifestyle changes or medical treatment when indicated can therefore restore androgen balance and reduce the relative impact of DHT suppression in sensitive individuals.
In select cases, testosterone replacement therapy (TRT) may be considered, but only with careful specialist oversight. TRT can improve sexual function, energy, and quality of life, yet it may also accelerate AGA unless combined thoughtfully with hair-loss treatments.[19]Zhang, Y., Xu, J., Jing, J., Wu, X., & Lv, Z. (2018). Serum levels of androgen-associated hormones are correlated with curative effect in androgenic alopecia in young men. *Medical Science … Continue reading For these reasons, TRT should never be initiated solely to counter finasteride side effects without a clear medical diagnosis and a comprehensive discussion of risks, benefits, and long-term goals.
8. Review Diet, Lifestyle, and Environmental Factors
While lifestyle changes cannot reverse finasteride’s pharmacologic action on DHT, they can meaningfully improve erectile function, mood, energy levels, sleep quality, and overall androgen balance, all of which overlap with the most commonly reported finasteride-related complaints.
A wide body of evidence shows that diet, exercise, and weight management play a significant role in sexual function. Randomized trials demonstrate that intensive lifestyle programs can significantly improve erectile function, with a meaningful proportion of men regaining normal erectile performance over time.[20]Esposito, K., Giugliano, F., Di Palo, C., Giugliano, G., Marfella, R., D’Andrea, F., D’Armiento, M., & Giugliano, D. (2004). Effect of lifestyle changes on erectile dysfunction in obese men: … Continue reading,[21]Hehemann, M. C., & Kashanian, J. A. (2016). Can lifestyle modification affect men’s erectile function?. *Translational Andrology and Urology.* 5(2). 187. Available at: … Continue reading
Environmental and behavioral factors can also be reviewed. Excessive heat exposure (such as frequent sauna use), high alcohol intake, cannabis use, and certain medications (including antidepressants) can independently impair sexual function or mood and may confound the interpretation of finasteride-related symptoms.
Although no studies have looked specifically at lifestyle programs for finasteride side effects, strong evidence shows that improving fitness, diet, sleep, and stress can enhance the same areas (erectile function, mood, energy, and hormone balance) that many men find most affected.
9. Consider Experimental or Adjunctive Supplements (With Caution)
Some men explore supplements marketed for testosterone support or sexual function, such as tongkat ali, in an effort to counter finasteride-related symptoms. Small studies suggest that tongkat ali may modestly increase testosterone or improve libido in certain populations, but the evidence is limited, inconsistent, and highly variable between individuals.[22]Leisegang, K., Finelli, R., Sikka, S. C., & Selvam, M. K. P. (2022). Eurycoma longifolia (jack) improves serum total testosterone in men: a systematic review and meta-analysis of clinical trials. … Continue reading
It’s critical to remember these supplements are experimental, not first-line solutions. Product quality, dosing, and purity can vary widely, and robust data on long-term safety or interactions with finasteride are lacking. While some individuals report subjective benefit, others notice no effect at all.
For these reasons, supplements should only be considered after established strategies have been explored and ideally discussed with a clinician, rather than relied upon as a primary way to manage side effects.
10. Switch Treatments or Change Strategy Entirely
If side effects persist despite adjustments to dose, frequency, or formulation, it may be appropriate to change strategies altogether. Hair loss treatment is not all-or-nothing, and finasteride, while highly effective, is not the only option.
Many men choose to rely on non-hormonal treatments entirely. Options such as topical or oral minoxidil, microneedling, low-level laser therapy devices, platelet-rich plasma (PRP), or hair transplantation can provide meaningful benefits without altering androgen pathways.
You can read more about alternative treatments in our guides and in-depth articles on minoxidil, microneedling, low-level laser therapy devices, and platelet-rich plasma (PRP).
While these approaches may not match finasteride’s disease-modifying effect on DHT, they can still preserve or improve cosmetic density for many users.
If you’re using topical finasteride, you might consider switching to low-dose topical dutasteride. offer better scalp localization at very low application frequencies due to dutasteride’s stronger and longer-lasting binding to 5AR. When used carefully and infrequently, some patients tolerate topical dutasteride well while maintaining hair stabilization.
Final Thoughts
Finasteride side effects are often manageable with thoughtful adjustments to dose, dosing schedule, formulation, and supporting lifestyle factors. There is no single correct way to use finasteride. Personalization is key, and the right approach depends on individual biology, risk tolerance, and treatment goals.
Health should always come first. Any side effects warrant open discussion with a clinician, and treatment decisions should be made collaboratively rather than in isolation. With informed, flexible planning, many men are able to find a balance that protects both their hair and their overall well-being.
References[+]
References ↑1 Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic review. *The Journal of Clinical and Aesthetic Dermatology.* 9(7). 56–62. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC5023004/ ↑2 Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has comparable urinary symptom efficacy and prostate volume reduction, but less sexual side effects and breast complications than dutasteride. *International Journal of Clinical Practice.* 66(11). 1052–1055. Available at: https://doi.org/10.1111/j.1742-1241.2012.03010.x ↑3 Traish, A. M. (2020). Post-finasteride syndrome: a surmountable challenge for clinicians. *Fertility and Sterility.* 113(1). 21–50. Available at: https://doi.org/10.1016/j.fertnstert.2019.11.030 ↑4 Trüeb, R. M., Régnier, A., Rezende, H. D., & Dias, M. F. R. G. (2019). Post-finasteride syndrome: an induced delusional disorder with the potential of a mass psychogenic illness?. *Skin Appendage Disorders.* 5(5). 320–326. Available at: https://doi.org/10.1159/000497362 ↑5 Kaufman, K. D., Olsen, E. A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V. H., et al. (1998). Finasteride in the treatment of men with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 39(4). 578–589. Available at: https://doi.org/10.1016/S0190-9622(98)70007-6 ↑6 Traish, A. M., Hassani, J., Guay, A. T., Zitzmann, M., & Hansen, M. L. (2011). Adverse side effects of 5α-reductase inhibitors therapy: persistent diminished libido and erectile dysfunction and depression in a subset of patients. *The Journal of Sexual Medicine.* 8(3). 872–884. Available at: https://doi.org/10.1111/j.1743-6109.2010.02157.x ↑7 Lowe, F. C., McConnell, J. D., Hudson, P. B., Romas, N. A., Boake, R., Lieber, M., Elhilali, M., et al. (2003). Long-term 6-year experience with finasteride in patients with benign prostatic hyperplasia. *Urology.* 61(4). 791–796. Available at: https://doi.org/10.1016/S0090-4295(02)02548-7 ↑8 Hudson, P. B., Boake, R., Trachtenberg, J., Romas, N. A., Rosenblatt, S., Narayan, P., Geller, J., et al. (1999). Efficacy of finasteride is maintained in patients with benign prostatic hyperplasia treated for 5 years. *Urology.* 53(4). 690–695. Available at: https://doi.org/10.1016/S0090-4295(98)00666-9 ↑9 Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W. P., Cotterill, P. C., Thiboutot, D. M., et al. (1999). The effects of finasteride on scalp skin and serum androgen levels in men with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 41(4). 550–554. Available at: https://doi.org/10.1016/S0190-9622(99)80051-6 ↑10 National Center for Biotechnology Information. (n.d.). Finasteride. *NCBI Bookshelf.* Available at: https://www.ncbi.nlm.nih.gov/books/NBK513329/ (Accessed: November 2025) ↑11 Steiner, J. F. (1996). Clinical pharmacokinetics and pharmacodynamics of finasteride. *Clinical Pharmacokinetics.* 30(1). 16–27. Available at: https://doi.org/10.2165/00003088-199630010-00002 ↑12 Caserini, M., Radicioni, M., Leuratti, C., Annoni, O., & Palmieri, R. (2014). A novel finasteride 0.25% topical solution for androgenetic alopecia: pharmacokinetics and effects on plasma androgen levels in healthy male volunteers. *International Journal of Clinical Pharmacology and Therapeutics.* 52(10). 842–849. Available at: https://doi.org/10.5414/CP202119 ↑13 Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of Dermatology, Venereology and Leprology.* 75. 47. Available at: https://doi.org/10.4103/0378-6323.45220 ↑14 Piraccini, B. M., Blume-Peytavi, U., Scarci, F., Jansat, J. M., Falqués, M., Otero, R., Tamarit, M. L., et al. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. *Journal of the European Academy of Dermatology and Venereology.* 36(2). 286–294. Available at: https://doi.org/10.1111/jdv.17738 ↑15 Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia: preliminary evaluations after a 16-month therapy course. *Journal of Dermatological Treatment.* 8(3). 189–192. Available at: https://doi.org/10.3109/09546639709160517 ↑16 Casabé, A., Roehrborn, C. G., Da Pozzo, L. F., Zepeda, S., Henderson, R. J., Sorsaburu, S., Henneges, C., Wong, D. G., & Viktrup, L. (2014). Efficacy and safety of the coadministration of tadalafil once daily with finasteride for 6 months in men with lower urinary tract symptoms and prostatic enlargement secondary to benign prostatic hyperplasia. *The Journal of Urology.* 191(3). 727–733. Available at: https://doi.org/10.1016/j.juro.2013.09.059 ↑17 Lui, J. L., Shaw, N. M., Abbasi, B., Hakam, N., & Breyer, B. N. (2023). Adverse reactions of PDE5 inhibitors: an analysis of the World Health Organization pharmacovigilance database. *Andrology.* 11(7). 1408–1417. Available at: https://doi.org/10.1111/andr.13430 ↑18 National Center for Biotechnology Information. (n.d.). Dutasteride. *NCBI Bookshelf.* Available at: https://www.ncbi.nlm.nih.gov/books/NBK532933/ (Accessed: November 2025) ↑19 Zhang, Y., Xu, J., Jing, J., Wu, X., & Lv, Z. (2018). Serum levels of androgen-associated hormones are correlated with curative effect in androgenic alopecia in young men. *Medical Science Monitor.* 24. 7770. Available at: https://doi.org/10.12659/MSM.913116 ↑20 Esposito, K., Giugliano, F., Di Palo, C., Giugliano, G., Marfella, R., D’Andrea, F., D’Armiento, M., & Giugliano, D. (2004). Effect of lifestyle changes on erectile dysfunction in obese men: a randomized controlled trial. *JAMA.* 291(24). 2978–2984. Available at: https://doi.org/10.1001/jama.291.24.2978 ↑21 Hehemann, M. C., & Kashanian, J. A. (2016). Can lifestyle modification affect men’s erectile function?. *Translational Andrology and Urology.* 5(2). 187. Available at: https://doi.org/10.21037/tau.2016.02.05 ↑22 Leisegang, K., Finelli, R., Sikka, S. C., & Selvam, M. K. P. (2022). Eurycoma longifolia (jack) improves serum total testosterone in men: a systematic review and meta-analysis of clinical trials. *Medicina.* 58(8). 1047. Available at: https://doi.org/10.3390/medicina58081047 Dutasteride is one of the most powerful pharmacologic treatments for androgenic alopecia (AGA). Initially approved for benign prostatic hyperplasia, dutasteride is now commonly prescribed off-label for hair loss in men and, more recently, has gained attention in topical form to minimize systemic exposure. This has created an important clinical question: how do oral and topical dutasteride compare in terms of effectiveness, safety, and real-world practicality?
This article examines what the current scientific literature reveals about oral versus topical dutasteride, reviewing mechanisms of action, efficacy data, and safety profiles to help you make more informed treatment decisions.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Dutasteride Mechanism of Action and Formulations
AGA is driven by increases in dihydrotestosterone (DHT). DHT binds to androgen receptors in hair follicles, triggering miniaturization that leads to thinner, shorter hairs. Dutasteride works by inhibiting an enzyme called 5α-reductase (5AR), which converts testosterone into DHT. Dutasteride is a dual 5AR inhibitor, meaning it blocks both type I and type II 5AR, leading to decreased DHT levels and ultimately halting the progression of AGA and restoring thicker hair.[1]Gerst, C., Dalko, M., Pichaud, P., Galey, J. B., Buan, B., & Bernard, B. A. (2002). Type-1 steroid 5α-reductase is functionally active in the hair follicle as evidenced by new selective … Continue reading,[2]Clark, R. V., Hermann, D. J., Cunningham, G. R., Wilson, T. H., Morrill, B. B., & Hobbs, S. (2004). Marked suppression of dihydrotestosterone in men with benign prostatic hyperplasia by … Continue reading
Figure 1. Structure of dutasteride, which is a dual 5AR inhibitor.[3]Wikimedia Commons. (n.d.). Dutasterid.svg [Image]. *Wikimedia Commons.* Available at: https://commons.wikimedia.org/wiki/File:Dutasterid.svg (Accessed: November 2025) Image used under Creative Commons License.
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Oral Dutasteride – Mechanism and Formulations
Oral dutasteride is absorbed through the gastrointestinal tract into systemic circulation, leading to significant and sustained reductions in DHT in the blood (serum). This includes reductions in scalp DHT, since less DHT is available systemically to diffuse into hair follicles.[4]Clark, R. V., Hermann, D. J., Cunningham, G. R., Wilson, T. H., Morrill, B. B., & Hobbs, S. (2004). Marked suppression of dihydrotestosterone in men with benign prostatic hyperplasia by … Continue reading,[5]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading
The flip side is that any symptoms tied to systemic DHT suppression, such as sexual side-effects, are driven by this same mechanism. Given the long half-life and accumulation, systemic exposure persists even if doses are missed or therapy is stopped, and any adverse effects may take weeks to resolve.[6]Tsai, T.-F., Choi, G. S., Kim, B. J., Kim, M.-B., Ng, C. F., Kochhar, P., Jasper, S., Brotherton, B., Orban, B., & Lulic, Z. (2018). Prospective randomized study of sexual function in men taking … Continue reading
Topical Dutasteride – Mechanism and Formulations
Topical dutasteride treatment might avoid some of these pitfalls: high exposure at the scalp and low exposure elsewhere can target delivery and avoid systemic issues. It is applied directly to the scalp in formulations such as:
- Simple solutions (often alcohol- or glycol-based)
- Gels or lotions
- Liposomal formulations
- Nanoemulsions and nanoemulgels
- Nanocrystalline and other particulate systems
These vehicles are designed to ensure that dutasteride can cross the stratum corneum (the outermost protective layer of the skin) and, ideally, remain localized within the scalp.
What is the Available Evidence for Oral Dutasteride?
The effect of dutasteride on hair loss is well documented: multiple randomized placebo-controlled studies have been conducted over the last 25 years. Dutasteride was first developed as a treatment for benign prostatic hyperplasia and taken as a tablet. Early studies into dutasteride for hair loss therefore used oral formulations, with topical treatments developed later.
Here, we’ll take a look at the evidence supporting the use of oral dutasteride for hair loss. Many studies compare dutasteride with other treatments, most notably finasteride, or use the drug in combination with other therapies. We’ll focus on data showing the effect of dutasteride alone compared to placebo, but will also touch on comparisons with other popular treatments.
Below is a summary of the findings to date on the safety and efficacy of oral dutasteride:
Study Design and Population Treatments and Concentrations (Daily) Hair Growth Outcomes Systemic Effects Study #1[7]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading Randomized, placebo-controlled, double-blinded trial. 415 men with male pattern hair loss aged 21-45 years old; 24 weeks. 0.05, 0.1, 0.5, or 2.5 mg dutasteride; 5 mg finasteride. Dose-dependent increase in total hair count vs placebo. Improved hair count vs finasteride. Serum decrease in DHT level (92% at 0.5 mg). Study #2[8]Stough, D. (2007). Dutasteride improves male pattern hair loss in a randomized study in identical twins. *Journal of Cosmetic Dermatology.* 6(1). 9–13. Available at: … Continue reading Randomized, placebo-controlled, double-blinded trial. 17 pairs of identical twin males with AGA; 12 months.
0.5 mg dutasteride. Significant increase in total hair counts in dutasteride-treated twins. Not assessed. Study #3[9]Eun, H. C., Kwon, O. S., Yeon, J. H., Shin, H. S., Kim, B. Y., Ro, B. I., Cho, H. K., et al. (2010). Efficacy, safety, and tolerability of dutasteride 0.5 mg once daily in male patients with male … Continue reading Randomized, placebo-controlled, double-blinded trial. 153 men with male pattern hair loss aged 18-49; 6 months 0.5 mg dutasteride. Significant increase in total hair count. Sexual dysfunction was reported in 4.1% of the treatment group and 2.7% of placebo (not statistically significant). Study #4[10]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading Randomized, placebo-controlled, double-blinded trial. 917 men with AGA aged 20-50; 24 weeks. 0.02, 0.1, or 0.5 mg dutasteride; 1 mg finasteride Increase in terminal hair count and width vs placebo. Significant increase in hair width vs dutasteride. No significant changes in reported adverse events. Study #5[11]Tsunemi, Y., Irisawa, R., Yoshiie, H., Brotherton, B., Ito, H., Tsuboi, R., Kawashima, M., Manyak, M., & ARI114264 Study Group. (2016). Long-term safety and efficacy of dutasteride in the … Continue reading Open-label, prospective study, no control. 120 men with AGA aged 20-50; 52 weeks. 0.5 mg dutasteride. Increase in terminal hair count and hair width compared to baseline. 12% of men experienced libido decrease and impotence, of which 50% and 57%, respectively, were not resolved by the end of the study. Study #6[12]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading Open-label, randomized study; no placebo control. 90 men with AGA aged 18-40; 24 weeks. 0.5 mg dutasteride or 1 mg finasteride. Increase in total hair count and width compared to baseline. Increase in total hair count was significantly higher in dutasteride than finasteride group. Erectile dysfunction and loss of libido reported in 3 and 4 patients respectively. Study #7[13]Tsai, T.-F., Choi, G. S., Kim, B. J., Kim, M.-B., Ng, C. F., Kochhar, P., Jasper, S., Brotherton, B., Orban, B., & Lulic, Z. (2018). Prospective randomized study of sexual function in men taking … Continue reading Randomized, placebo-controlled, double-blinded trial. 117 men with AGA aged 23-50; 24 weeks. 0.5 mg dutasteride. Not assessed. Incidence of sexual adverse events was approximately double in the dutasteride group (16% vs 8% in placebo). Study #8[14]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading Retrospective chart review. 600 men over 18 with AGA. 0.5 mg dutasteride or 1 mg finasteride. Greater improvements in BASP classifications of AGA compared to baseline were found in dutasteride vs finasteride. Incidence of adverse events was higher in finasteride group (10.5%) than in dutasteride group (7.6%). What is the Available Evidence for Topical Dutasteride?
While evidence for the efficacy of oral dutasteride is well established, research into topical dutasteride solutions started relatively recently. Below is an overview of the studies into the efficacy of topical dutasteride:
Study Design and Population Treatments and Concentrations (Daily) Hair Growth Outcomes Systemic Effects Study #1[15]Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M. E., Ocampo-Garza, J., Orizaga-y-Quiroga, T. L., Martínez-Moreno, A., & Ocampo-Garza, S. S. (2022). Microneedling plus … Continue reading Randomized, placebo-controlled, double-blinded trial. 34 men with AGA aged 18-65; 20 weeks. Microneedling with 0.01% dutasteride solution. Placebo was microneedling with saline solution. Significant improvements in hair thickness, hair density, and vellous: terminal hair ratio. Not assessed. Study #2[16]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading Randomized, placebo-controlled, double-blinded trial. 135 adult males aged 20-60 with AGA; 24 weeks. 0.01%, 0.02%, or 0.05% topical dutasteride; 1mg oral dutasteride Dose-dependent increase in total hair count and hair width. 0.05% solution showed significant improvement over finasteride in total hair count. No change in serum DHT levels observed vs placebo. How Does Oral and Topical Dutasteride Stack Up Against Each Other?
When comparing oral and topical dutasteride, the most important point to note is that there are currently no published clinical trials directly comparing the two formulations alone head-to-head for AGA. All comparisons must be made indirectly, using separate studies with different designs, populations, timelines, and formulations.
What About the Quality of the Available Evidence?
Despite this limitation, the evidence available for each formulation allows us to evaluate its strength and predictability. The current evidence quality for each formulation is as follows:
- Oral dutasteride — 59/100
- Topical dutasteride — 55/100
By our metrics, this means that both treatments have similar evidence quality overall. Longer-term data support oral dutasteride, while topical dutasteride remains less validated, largely due to fewer studies and limited long-term follow-up.
We’ll take a closer look at the strength of the evidence supporting both formulations.
Oral Dutasteride Studies
Oral dutasteride can reduce serum DHT levels by up to 90% and scalp DHT levels by 51-79%.[17]Clark, R. V., Hermann, D. J., Cunningham, G. R., Wilson, T. H., Morrill, B. B., & Hobbs, S. (2004). Marked suppression of dihydrotestosterone in men with benign prostatic hyperplasia by … Continue reading,[18]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading Randomized controlled trials consistently demonstrate that dutasteride, particularly at the standard 0.5 mg daily dose, suppresses serum and scalp DHT and delivers some of the most robust regrowth outcomes documented for AGA.
A number of the studies outlined above include large sample sizes, often over 100 participants, with the largest including over 900.[19]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading Importantly, these cohorts should be large enough to detect changes in the incidence of adverse events, even if they are relatively rare. The replication of results in multiple trials using similar endpoints is a strong indicator of the efficacy of the drug.
Unfortunately, the longest of these RCTs only lasted 6 months, which may mean that lasting impacts on growth are overlooked. Similarly, side effects associated with long-term use over many years may be missed.
We can turn to longer studies using finasteride for some indication of how sustained benefits may be, given the similar mechanism of action of the two treatments. A 3-year trials using 1 mg daily doses of finasteride showed sustained improvement in hair growth over the course of the study, while placebo groups saw decreased hair counts.[20]Price, V. H., Menefee, E., Sanchez, M., & Kaufman, K. D. (2006). Changes in hair weight in men with androgenetic alopecia after treatment with finasteride (1 mg daily): three- and 4-year results. … Continue reading
A 10-year follow-up study concluded that improvements in hair growth, as determined by expert analysis of hair photographs, from finasteride do not worsen over time, and in 21% of cases, hair growth got even better after 5 years of treatment.[21]Rossi, A., Cantisani, C., Scarnò, M., Trucchia, A., Fortuna, M. C., & Calvieri, S. (2011). Finasteride, 1 mg daily administration on male androgenetic alopecia in different age groups: 10-year … Continue reading
While we can’t draw any firm conclusions about the long-term efficacy of dutasteride from studies using finasteride, these findings can indicate the impact of long-term inhibition of 5AR, which is also seen in dutasteride treatment.
Topical Dutasteride Studies
Evidence for topical dutasteride is promising but remains less mature. Early clinical research indicates that topical formulations can meaningfully improve hair density, hair shaft diameter, and overall investigator-assessed regrowth, with efficacy in trials approaching that of oral therapy.
One small study with only 34 participants showed significant improvements in hair growth and thickness when combined with microneedling, compared to microneedling alone. Another study combined dutasteride formulations with minoxidil and also found that topical dutasteride delivered via microneedling produced hair density and thickness improvements similar to oral dutasteride, with fewer systemic side effects.[22]Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M. E., Ocampo-Garza, J., Orizaga-y-Quiroga, T. L., Martínez-Moreno, A., & Ocampo-Garza, S. S. (2022). Microneedling plus … Continue reading,[23]Abu Obeid, M. N., Abdel Fattah, N. S., Elfangary, M. M., & Al Husseni, R. M. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through … Continue reading
A recently published Phase II trial, the first to test topical dutasteride without microneedling, reported that all concentrations improved hair growth versus placebo, produced minimal changes in serum DHT, and that 1 ml daily of 0.05% appeared to outperform oral finasteride. However, concerns about unusually high baseline hair counts and inconsistencies in the placement and size of the 1 cm² target areas used for hair measurements raise questions about the reliability of these results.[24]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading
Figure 2. Oral dutasteride (yellow line) reduces serum DHT levels more than topical Dutaseride (blue lines). Adapted from Figure 3.[25]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading Image used under Creative Commons License.
Real-world outcomes from low-dose topical dutasteride users (0.01–0.02% applied daily or near-daily) suggest more modest benefits, with good scalp localization and stable serum DHT noted as positives, rather than regrowth exceeding that with oral finasteride.
There have been some concerns raised about the ability of dutasteride to penetrate into the scalp. Dutasteride is a relatively large molecule, with one molecule weighing 528.5 Daltons (Da). There is a commonly cited rule, called the 500 Dalton Rule, that states that molecules should be under 500 Da in order to penetrate the skin. However, dutasteride has been demonstrated to be effective in topical formulations and can measurably lower DHT concentrations. What’s more, a small, unpublished study has also shown that the drug does penetrate into both the scalp and serum, even at low concentrations. As such, in this case, dutasteride does seem to penetrate the skin in the scalp, without the need for microneedling or injection.
How Safe are Dutasteride Formulations?
Because dutasteride produces sustained suppression of DHT, safety considerations, particularly regarding hormonal and sexual side effects, are central to treatment selection. Most clinical trials record adverse events, while some studies are focused specifically on side-effects.[26]Tsai, T.-F., Choi, G. S., Kim, B. J., Kim, M.-B., Ng, C. F., Kochhar, P., Jasper, S., Brotherton, B., Orban, B., & Lulic, Z. (2018). Prospective randomized study of sexual function in men taking … Continue reading
Oral Dutasteride
Across randomized trials and long-term observational studies, the most consistently reported adverse effects of oral dutasteride involve sexual function, including:
- Decreased libido
- Erectile dysfunction
- Ejaculation disorders
- Gynecomastia and breast tenderness (rare)
Trials report overall total adverse event rates between 4 and 12%, with sexual adverse events such as decreased libido as high as 13%, although this was reported in higher doses (2.5mg daily).[27]Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase … Continue reading At the standard 0.5mg daily dose, sexual side effects typically occur at a rate of between 1 and 5%. In the context of other popular treatments, comparisons with finasteride show no consistent increase in sexual side effects with dutasteride, despite dutasteride lowering serum DHT by an additional 20-30% beyond finasteride.[28]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading
Most controlled studies show that the majority of sexual side effects resolve either during treatment or after discontinuation. However, due to dutasteride’s long half-life (4-5 weeks) and high tissue binding, systemic effects can persist. At standard 0.5mg daily dosing, serum DHT generally returns to 80% of baseline within 6 months of discontinuation.
Topical Dutasteride
Topical dutasteride is generally well tolerated, and reported adverse events are usually mild and localized, including:
- Scalp irritation
- Erythema
- Contact dermatitis
- Pruritus or dryness
These occur inconsistently and are formulation-dependent, influenced by solvent systems, penetration enhancers, and application volume.
The primary safety advantage of topical dutasteride is reduced systemic exposure. In trials, topical regimens in the 0.01–0.05% range produce minimal or no significant changes in serum DHT or testosterone.[29]Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study … Continue reading When decreases in serum DHT are observed with topical use, they are generally smaller than those seen with oral dosing.[30]Abu Obeid, M. N., Abdel Fattah, N. S., Elfangary, M. M., & Al Husseni, R. M. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through … Continue reading
Adherence and Practical Considerations
Dutasteride, whether oral or topical, is primarily used for AGA in men, though its use for hair loss remains off-label in the United States. Oral dutasteride is formally FDA-approved only for benign prostatic hyperplasia, while topical formulations exist solely through compounding pharmacies.
Adherence and Route of Application
Oral dutasteride offers the simplest adherence burden: one capsule daily (or a modified intermittent schedule). This ease is a major factor behind its consistent outcomes. Topical dutasteride, while potentially safer from a systemic standpoint, requires regular scalp application and attention to dosing volume.
The effectiveness of topical formulations is heavily influenced by factors such as vehicle, scalp contact time, treated surface area, and individual skin permeability. These practical considerations can meaningfully shape both efficacy and systemic absorption.
Advanced vehicles such as nanoemulsions or nanoemulgels can increase scalp penetration by up to 1.5-fold compared to conventional solutions.[31]Ali, M. S., Alam, M. S., Alam, N., & Siddiqui, M. R. (2014). Preparation, characterization and stability study of dutasteride loaded nanoemulsion for treatment of benign prostatic hypertrophy. … Continue reading However, this may also increase systemic exposure. Application frequency can be adjusted to balance efficacy and hormonal safety. Because dutasteride binds to 5AR for prolonged periods, daily application is not always necessary.
Time on the scalp also matters. The longer a topical formulation remains in contact with the skin, the more dutasteride can penetrate the follicle. Most protocols recommend allowing at least four hours before washing the hair. Rinsing too soon can reduce efficacy, while prolonged contact increases both potency and the possibility of systemic absorption.
The size of the application area affects systemic exposure. Treating a small region, such as the crown, introduces less drug into circulation than applying the same concentration across the entire scalp. Patients with diffuse thinning often benefit from using lower volumes or concentrations to counterbalance the increased surface area.
As we can see, there is a wide range of factors to consider when making a treatment plan with topical dutasteride, which may make consistent application a challenge.
Use in Women
Off-label use of dutasteride in women is limited and generally reserved for postmenopausal women or women with documented hyperandrogenism or a strong family history of AGA. Due to the systemic androgenic impacts of the drug, it’s typically only used in cases where other therapies (minoxidil, spironolactone) are inadequate.
Clinical Interpretation and Decision-Making
The key trade-off between oral and topical formulations is systemic potency versus localized targeting, balanced against differences in side-effect risk, convenience, and cost. Oral therapy is simpler and less variable; topical therapy requires consistent application but offers greater hormonal safety for risk-averse patients.
Combination therapy, most commonly topical dutasteride with minoxidil, can enhance outcomes through complementary mechanisms. Oral dutasteride may also be paired with topical agents in some cases.
Significant research gaps remain, including the absence of direct head-to-head oral-versus-topical trials, limited long-term safety data for topical dutasteride, and the lack of standardized dosing guidelines for topical formulations.
Final Thoughts
Dutasteride, whether oral or topical, represents one of the most powerful tools currently available for treating AGA. Oral dutasteride offers the most consistent and robust regrowth outcomes, supported by the strongest body of clinical evidence, but with predictable systemic hormonal suppression and long persistence after discontinuation.
Topical dutasteride, while still supported by a smaller and newer evidence base, offers a compelling alternative for patients seeking meaningful scalp-level efficacy with reduced systemic exposure. Its ultimate success depends heavily on formulation quality, dosing strategy, and adherence.
There is no universally “best” option: only the best option for specific circumstances. Treatment selection should be guided by hair-loss severity, prior treatment response, risk tolerance, reproductive plans, and long-term adherence capacity. As research continues to evolve, clinical guidance will become more precise. For now, optimal results come from choosing the formulation that best aligns with your goals and comfort with risk.
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

References[+]
References ↑1 Gerst, C., Dalko, M., Pichaud, P., Galey, J. B., Buan, B., & Bernard, B. A. (2002). Type-1 steroid 5α-reductase is functionally active in the hair follicle as evidenced by new selective inhibitors of either type-1 or type-2 human steroid 5α-reductase. *Experimental Dermatology.* 11(1). 52–58. Available at: https://doi.org/10.1034/j.1600-0625.2002.110106.x ↑2, ↑4, ↑17 Clark, R. V., Hermann, D. J., Cunningham, G. R., Wilson, T. H., Morrill, B. B., & Hobbs, S. (2004). Marked suppression of dihydrotestosterone in men with benign prostatic hyperplasia by dutasteride, a dual 5α-reductase inhibitor. *The Journal of Clinical Endocrinology & Metabolism.* 89(5). 2179–2184. Available at: https://doi.org/10.1210/jc.2003-030330 ↑3 Wikimedia Commons. (n.d.). Dutasterid.svg [Image]. *Wikimedia Commons.* Available at: https://commons.wikimedia.org/wiki/File:Dutasterid.svg (Accessed: November 2025) ↑5, ↑18 Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. *Journal of the American Academy of Dermatology.* 55(6). 1014–1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007 ↑6, ↑13, ↑26 Tsai, T.-F., Choi, G. S., Kim, B. J., Kim, M.-B., Ng, C. F., Kochhar, P., Jasper, S., Brotherton, B., Orban, B., & Lulic, Z. (2018). Prospective randomized study of sexual function in men taking dutasteride for the treatment of androgenetic alopecia. *The Journal of Dermatology.* 45(7). 799–804. Available at: https://doi.org/10.1111/1346-8138.14329 ↑7, ↑27 Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. *Journal of the American Academy of Dermatology.* 55(6). 1014–1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007 ↑8 Stough, D. (2007). Dutasteride improves male pattern hair loss in a randomized study in identical twins. *Journal of Cosmetic Dermatology.* 6(1). 9–13. Available at: https://doi.org/10.1111/j.1473-2165.2007.00297.x ↑9 Eun, H. C., Kwon, O. S., Yeon, J. H., Shin, H. S., Kim, B. Y., Ro, B. I., Cho, H. K., et al. (2010). Efficacy, safety, and tolerability of dutasteride 0.5 mg once daily in male patients with male pattern hair loss: a randomized, double-blind, placebo-controlled, phase III study. *Journal of the American Academy of Dermatology.* 63(2). 252–258. Available at: https://doi.org/10.1016/j.jaad.2009.09.018 ↑10, ↑19, ↑28 Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 70(3). 489–498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑11 Tsunemi, Y., Irisawa, R., Yoshiie, H., Brotherton, B., Ito, H., Tsuboi, R., Kawashima, M., Manyak, M., & ARI114264 Study Group. (2016). Long-term safety and efficacy of dutasteride in the treatment of male patients with androgenetic alopecia. *The Journal of Dermatology.* 43(9). 1051–1058. Available at: https://doi.org/10.1111/1346-8138.13310 ↑12 Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. *Indian Journal of Dermatology, Venereology and Leprology.* 83. 47. Available at: https://doi.org/10.4103/0378-6323.188652 ↑14 Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male androgenic alopecia in South Korea: a multicentre chart review study. *Annals of Dermatology.* 34(5). 349. Available at: https://doi.org/10.5021/ad.22.027 ↑15, ↑22 Sánchez-Meza, E., Ocampo-Candiani, J., Gómez-Flores, M., Herz-Ruelas, M. E., Ocampo-Garza, J., Orizaga-y-Quiroga, T. L., Martínez-Moreno, A., & Ocampo-Garza, S. S. (2022). Microneedling plus topical dutasteride solution for androgenetic alopecia: a randomized placebo-controlled study. *Journal of the European Academy of Dermatology & Venereology.* 36(10).. Available at: https://doi.org/10.1111/jdv.18285 ↑16, ↑25, ↑29 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. *Cureus.* 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑20 Price, V. H., Menefee, E., Sanchez, M., & Kaufman, K. D. (2006). Changes in hair weight in men with androgenetic alopecia after treatment with finasteride (1 mg daily): three- and 4-year results. *Journal of the American Academy of Dermatology.* 55(1). 71–74. Available at: https://doi.org/10.1016/j.jaad.2005.07.001 ↑21 Rossi, A., Cantisani, C., Scarnò, M., Trucchia, A., Fortuna, M. C., & Calvieri, S. (2011). Finasteride, 1 mg daily administration on male androgenetic alopecia in different age groups: 10-year follow-up. *Dermatologic Therapy.* 24(4). 455–461. Available at: https://doi.org/10.1111/j.1529-8019.2011.01441.x ↑23, ↑30 Abu Obeid, M. N., Abdel Fattah, N. S., Elfangary, M. M., & Al Husseni, R. M. (2024). Comparison between topical minoxidil 5% alone versus combined with dutasteride (topical 0.02% through microneedling or oral 0.5 mg) in treatment of androgenetic alopecia. *QJM: An International Journal of Medicine.* 117(Supplement_2). hcae175–207. Available at: https://doi.org/10.1093/qjmed/hcae175.207 ↑24 Panuganti, V. K., Madala, P. K., Grandhi, V. R., Alluri, C. V., Mohammad, J., Kssvv, S. R., & Dundigalla, M. R. (2025). A randomized, double-blind, placebo- and active-controlled phase II study to evaluate the safety and efficacy of novel dutasteride topical solution (0.01%, 0.02%, and 0.05% w/v) in male subjects with androgenetic alopecia. *Cureus.* 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑31 Ali, M. S., Alam, M. S., Alam, N., & Siddiqui, M. R. (2014). Preparation, characterization and stability study of dutasteride loaded nanoemulsion for treatment of benign prostatic hypertrophy. *Iranian Journal of Pharmaceutical Research (IJPR).* 13(4). 1125. Available at: https://doi.org/10.3390/pharmaceutics14061152 IngredientsPharmaceuticalsNatural RemediesCompany ReviewsBy Cassie Hopton, PhDFeb 3, 202610 Best DHT-Blocking Shampoos
DHT-blocking shampoos are widely available, but do they actually work? While blocking DHT is a proven strategy for treating androgenic alopecia, most evidence comes from oral medications and leave-on topicals, not rinse-off shampoos. In this article, we separate science from marketing by breaking do...By Sarah King, PhDJan 31, 2026Best Minoxidil for Men: Top Picks for 2026
With countless hair loss products on the market, choosing the right minoxidil can be overwhelming. This evidence-based guide ranks the best minoxidil treatments for men in 2026, including prescription and over-the-counter options, based on effectiveness, safety, customization, and value. Discover wh...By Sophie Grice, PhDJan 27, 2026Switching From Finasteride to Dutasteride
Switching from finasteride to dutasteride is often framed as a simple upgrade or downgrade. In reality, each switch changes not only DHT suppression but also exposure patterns, timelines for results, washout dynamics, and evidence quality. This article breaks down realistic switching scenarios, oral...By Perfect Hair Health TeamJan 21, 2026Best Minoxidil for Women: Top 6 Brands of 2026
With so many hair-thinning treatments marketed to women, it can be difficult to separate what truly works from what’s just hype. This evidence-based guide ranks the best minoxidil options for women in 2026, including prescription and over-the-counter formulas, based on effectiveness, tolerability, c...By Perfect Hair Health TeamJan 17, 2026Oral Minoxidil: Before and After Photos [2026]
Oral minoxidil is emerging as a popular off-label treatment for hair loss, offering an option for those who can’t use or don’t respond to topical formulas. While some achieve dramatic regrowth, most see more modest improvements. Discover why results vary, how to interpret before-and-after photos rea...By Perfect Hair Health TeamJan 16, 2026Best Topical Finasteride: 5 Top Products of 2026
Topical finasteride is exploding in popularity, but not all formulas are created equal. This guide breaks down the 5 best topical finasteride treatments on the market, comparing strength, safety, customization, and real-world value. From ultra-tailored prescriptions like Ulo to big telehealth brands...By Perfect Hair Health TeamJan 16, 2026Oral Finasteride Before and After: Real Case Studies
Oral finasteride remains the gold-standard treatment for male pattern hair loss, with decades of data showing consistent improvement in density and prevention of further thinning. However, dramatic online regrowth photos can lead to unrealistic expectations. In your regrowth journey, it’s important ...By Perfect Hair Health TeamJan 16, 2026Best Topical Dutasteride: 8 Choices for 2026
Topical dutasteride is emerging as a powerful next-generation option for treating hair loss. This expert-reviewed guide ranks the 8 best topical dutasteride treatments, comparing strength, safety, and customization, to help you find the most effective DHT-blocking solution for your needs. From Ulo’s...By Catherine Kennedy, PhDJan 12, 2026Does Finasteride Lower Testosterone?
Finasteride’s reputation for affecting hormones has led many to assume it suppresses testosterone, but the biology is far more nuanced. In this article, we unpack what happens to testosterone when DHT is inhibited, why levels often rise instead of fall, and how this squares with the sexual side effe...By Cassie Hopton, PhDJan 12, 2026Saw Palmetto Shampoo: Is It Effective for Hair Loss?
Saw palmetto is hailed as a natural hair loss treatment, promising hair growth and strength. It is incorporated into all manner of hair loss products, including shampoos. But is the hype backed by science? This article dissects the science of saw palmetto as a hair growth ingredient, as well as sham... - Mission Statement
 Scroll Down
Scroll Down
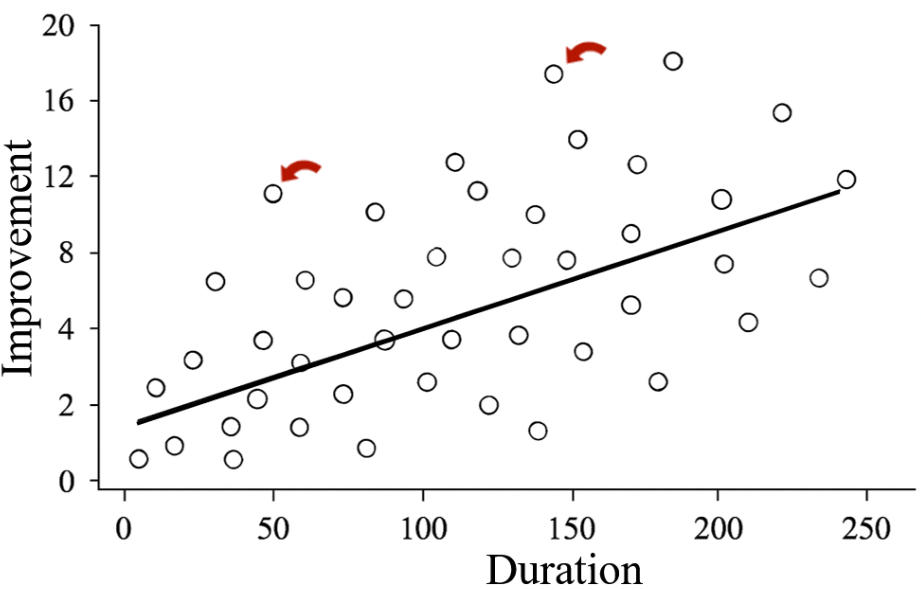
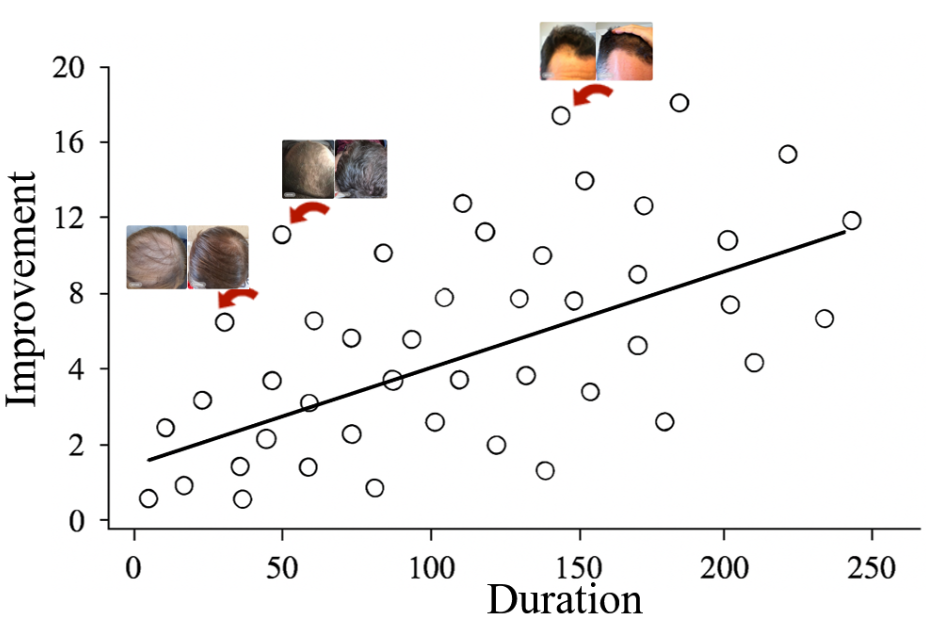
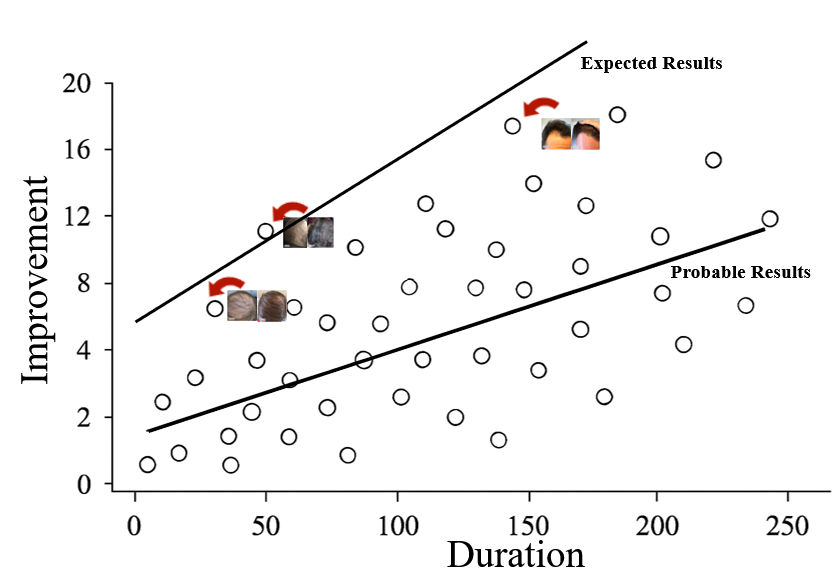
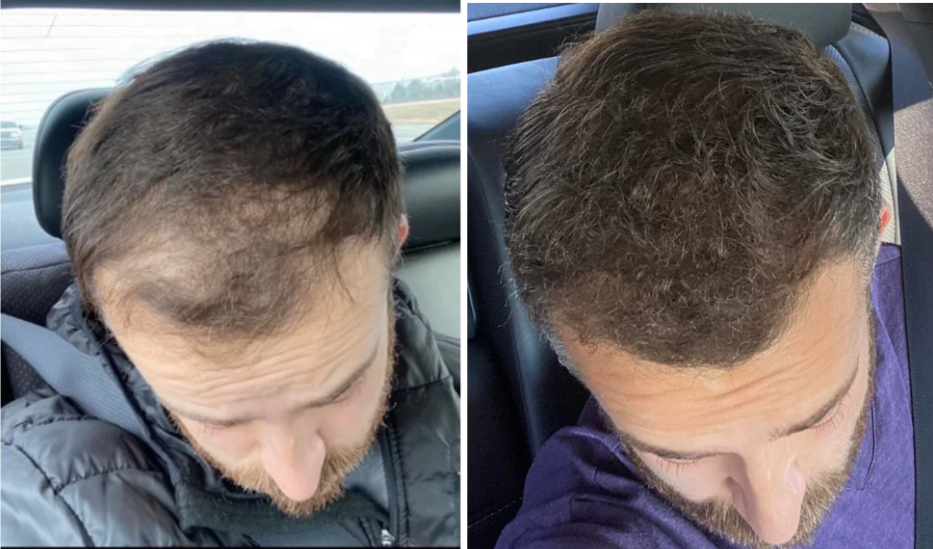

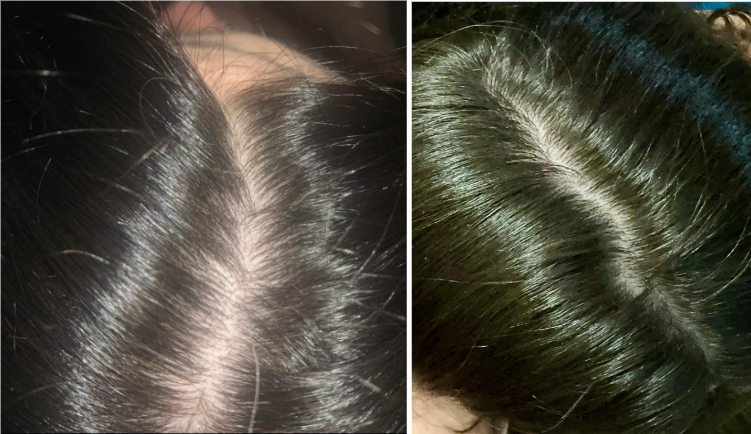
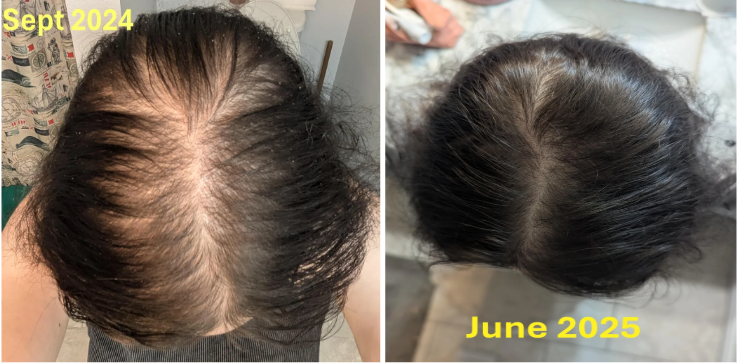
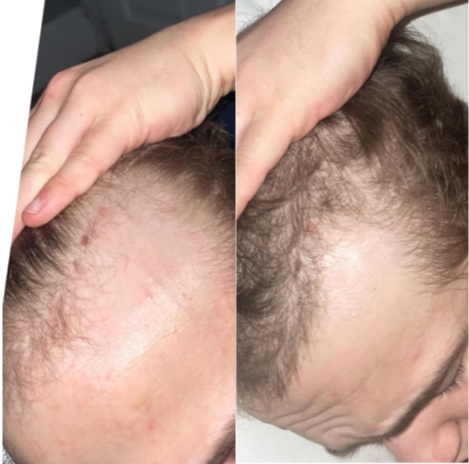

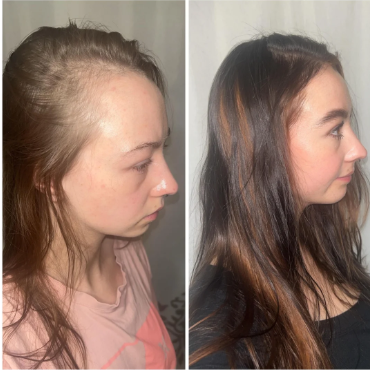

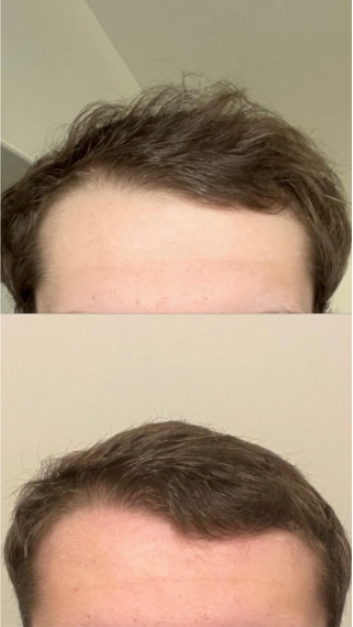
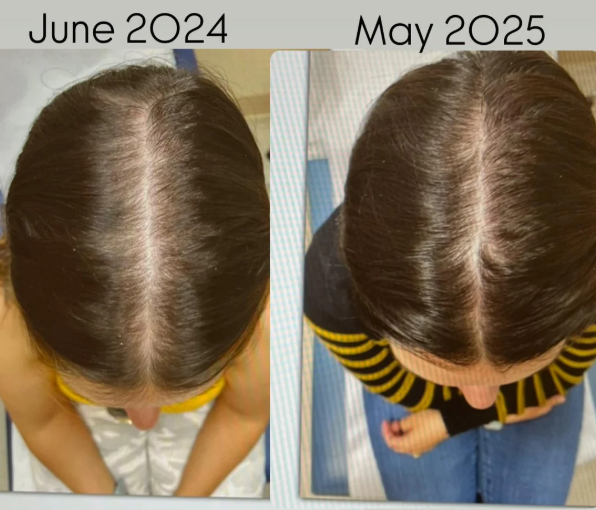
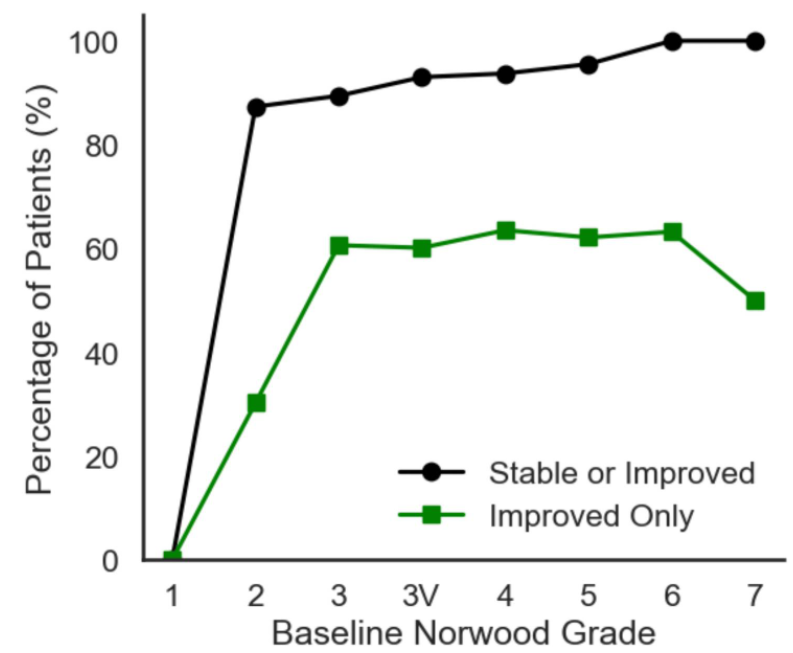







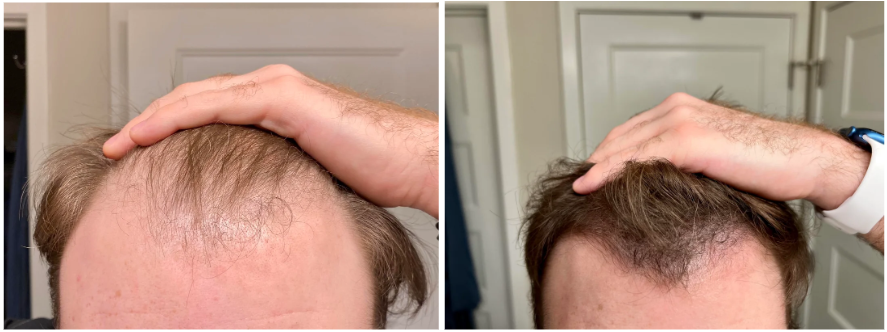
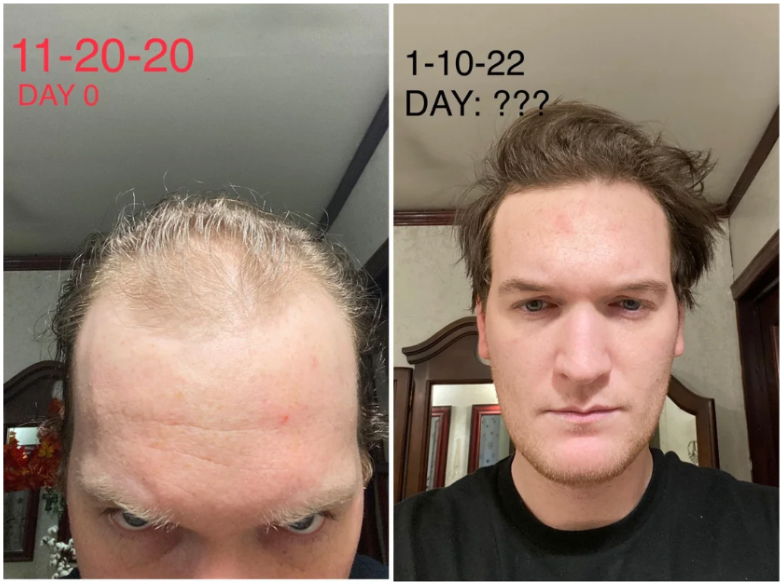

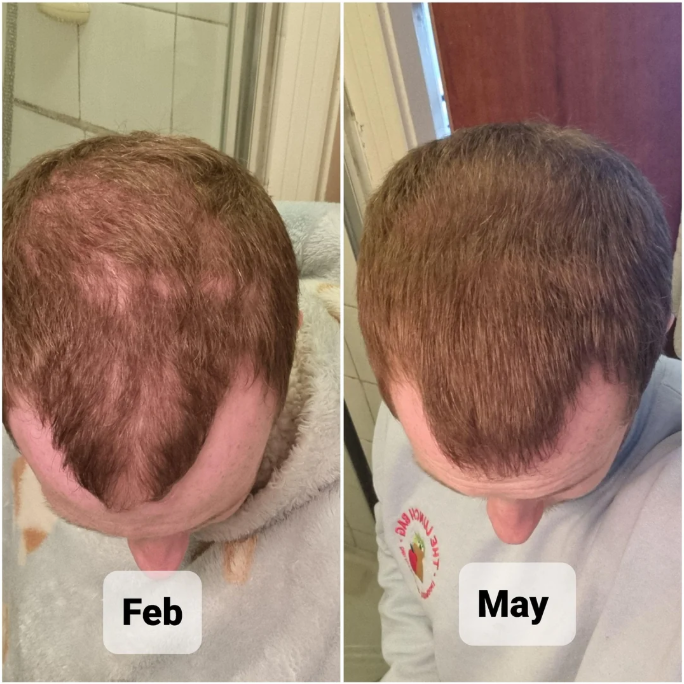
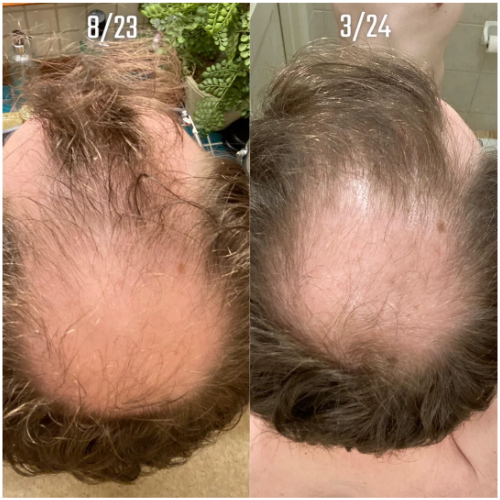
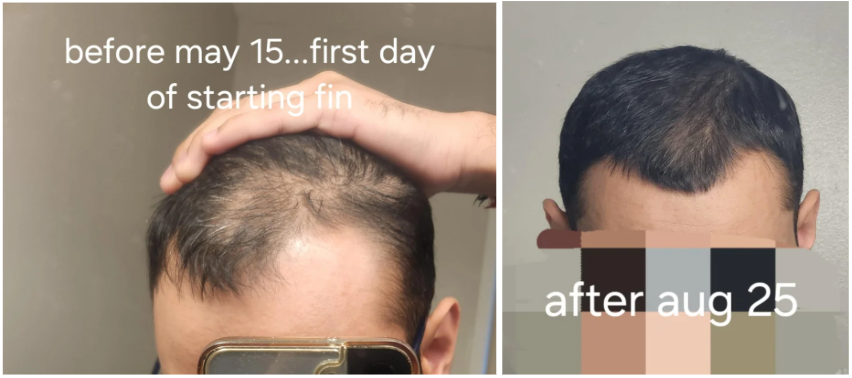

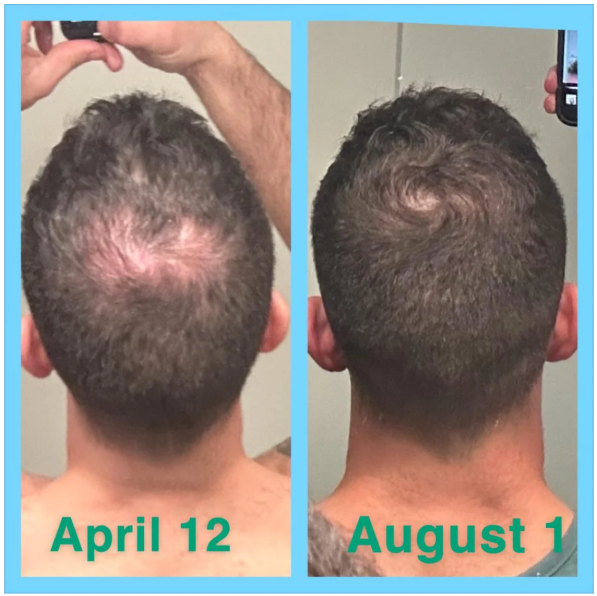
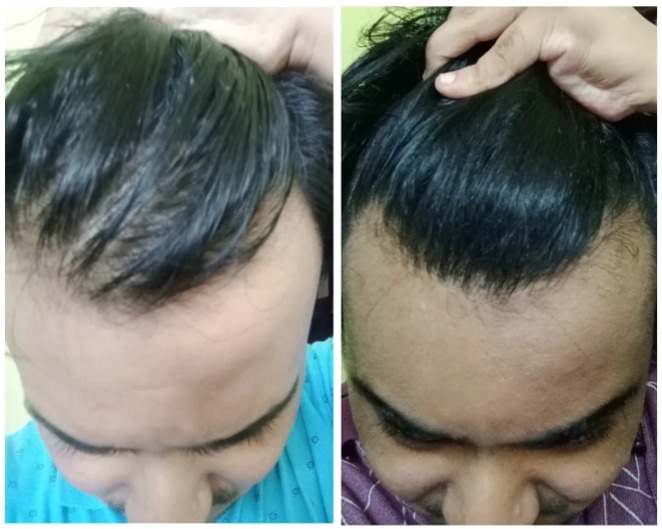
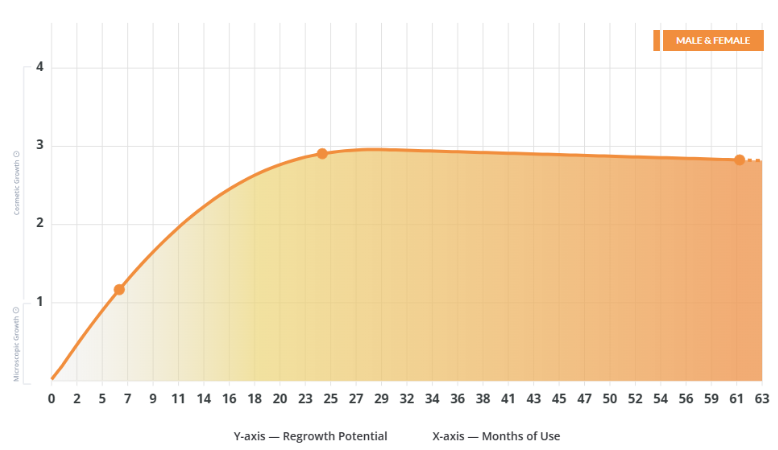








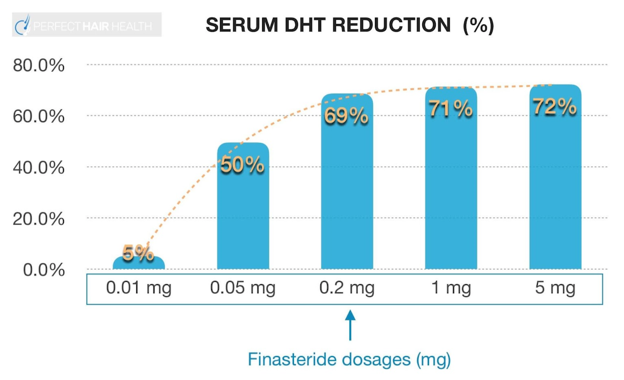
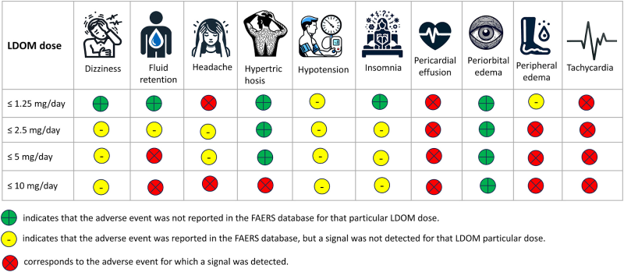

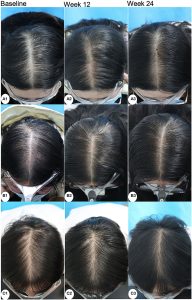
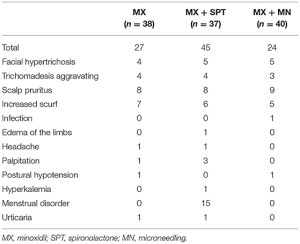
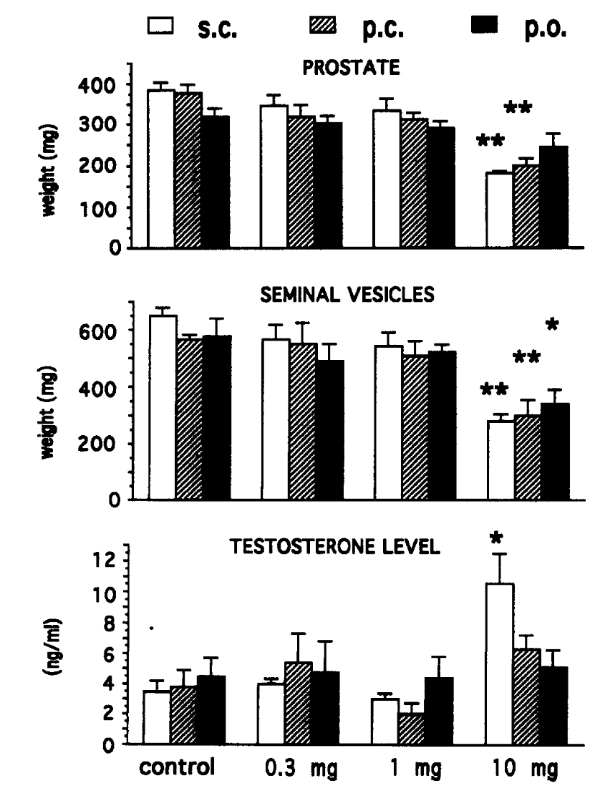

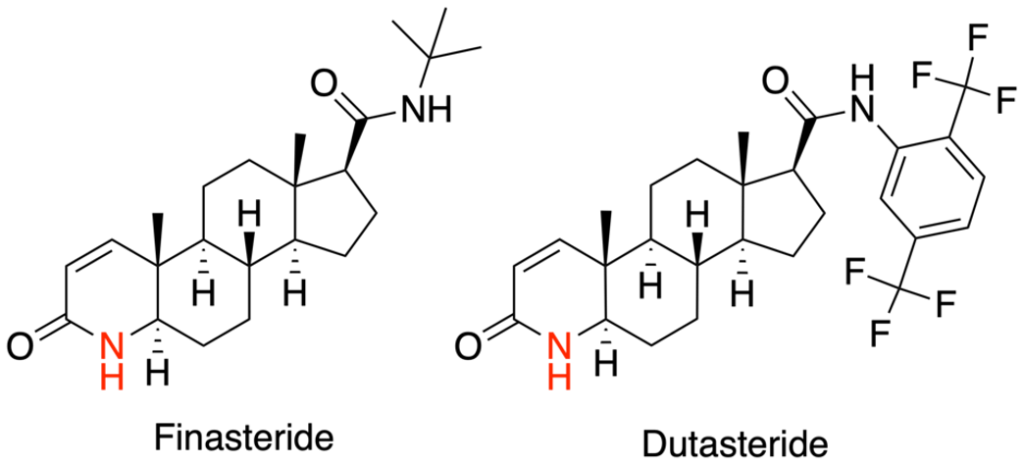
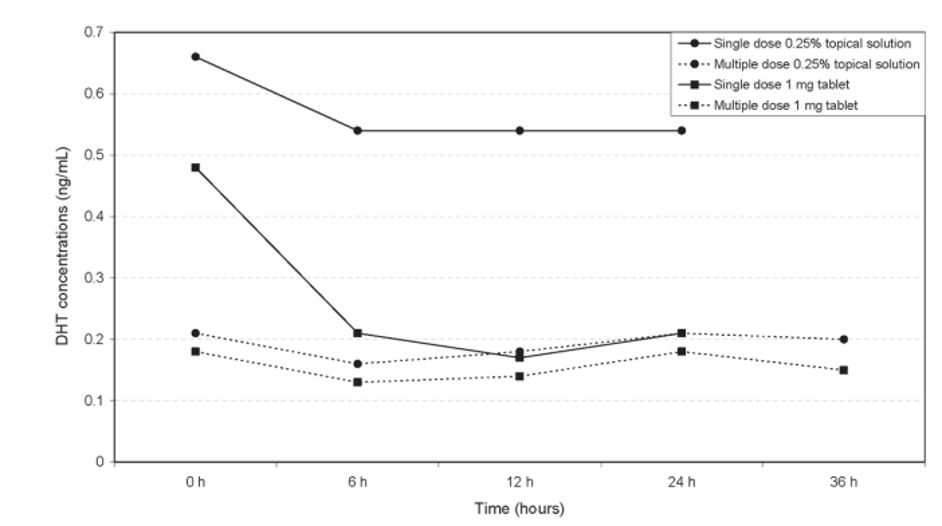
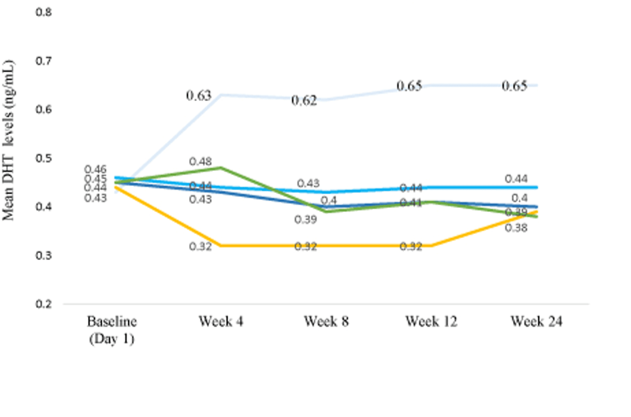
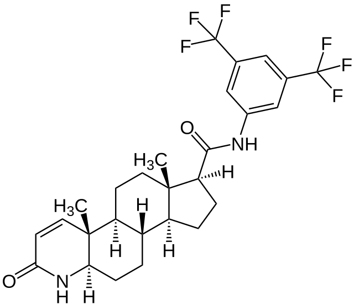
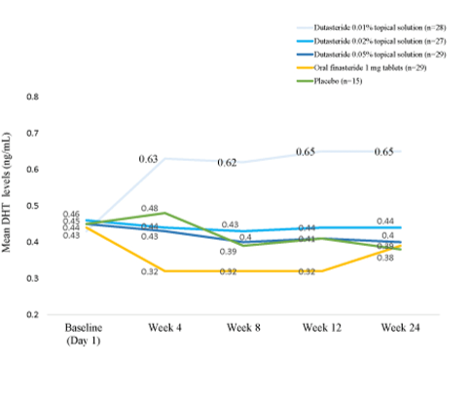


![Oral Minoxidil: Before and After Photos [2026]](https://perfecthairhealth.com/wp-content/uploads/2025/10/93-OM.jpg)