- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
10 Best Shampoos for Hair Loss in 2026
-
XYON Review: Do Their Products Actually Regrow Hair?
-
Minoxidil Before and After Photos [2026] | Does It Work?
-
How to Get Finasteride: Is It Over the Counter?
-
Keeps Review: The Truth About Their Hair Loss Treatments?
-
7 Best Oils for Hair Growth
-
Hims Hair Growth Reviews: The Pros, Cons, and Real Results
-
Topical Finasteride Before and After: Real Case Studies
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
ArticlesKeeps Review: The Truth About Their Hair Loss Treatments?
First Published Mar 10 2026Last Updated Mar 15 2026Company ReviewsPharmaceutical Researched & Written By:Catherine Kennedy, PhD
Researched & Written By:Catherine Kennedy, PhD Reviewed By:Michael Williams, PhD
Reviewed By:Michael Williams, PhD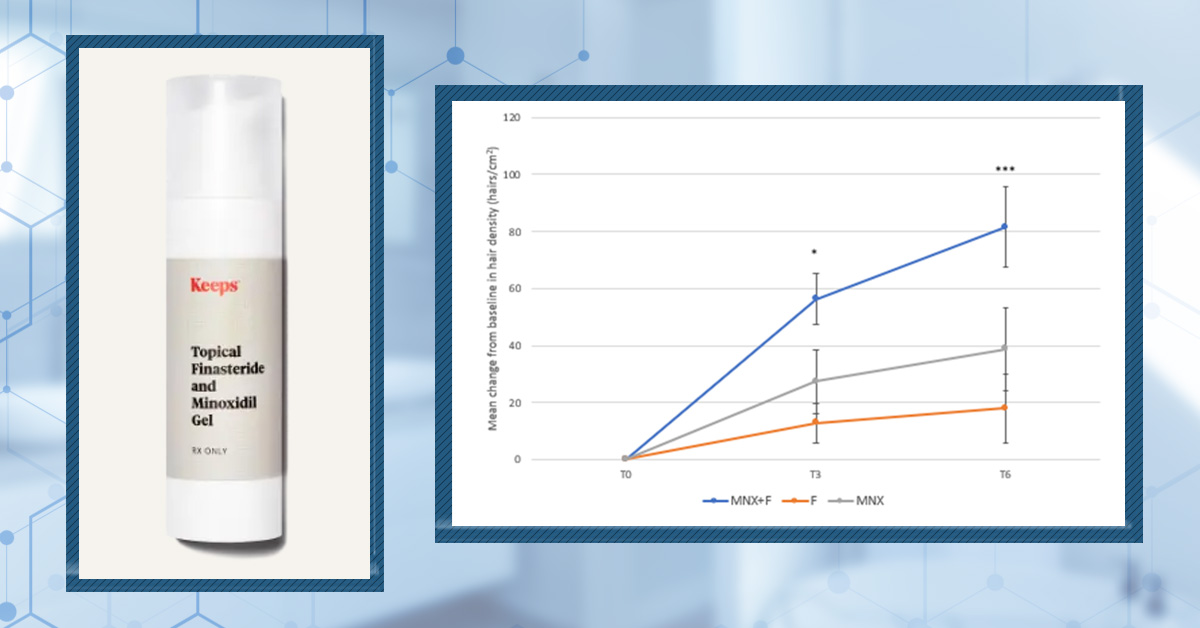
Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
Keeps is a telehealth brand offering science-backed hair loss treatments. In this review article, we’ll break down what Keeps offers, the real science behind offerings such as finasteride and minoxidil, and if the evidence aligns with (or contradicts) any marketing claims.
Full Article
Men are becoming increasingly skeptical about hair loss solutions due to the flooding of the market with pseudoscience-backed products that aren’t effective. Keeps is a telehealth brand offering hair loss and sexual health products. According to their website, their main focus is providing customers with access to proven, science-backed hair loss treatments produced using FDA-approved ingredients. They have strong claims about the effectiveness of their products, but what does the research actually say?
In this article, we’ll explore the products offered by Keeps, the science behind their hair loss treatments, whether their claims and recommendations align with current research, and any safety considerations associated with their products.
Finally, we’ll also discuss certain limitations in treatment options related to personalization. This is a problem less reflective of Keeps in particular, and more reflective of the current state of the telehealth hair care industry. It’s also why, after 10+ years of consumer advocacy, Perfect Hair Health decided to cofound the brand Ulo – a U.S.-based telehealth brand that focuses on hair loss, and also attempts to address core issues that have remained relatively neglected by other brands.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Key Takeaways
- Convenient and affordable. Keeps’ platform offers access to Rx hair loss medications throughout the U.S., along with online medical appointments to facilitate the prescription process. They also offer these products at mostly-affordable prices.
- Supported by science. The science supports the core treatments offered by Keeps – both finasteride and minoxidil reliably slow hair loss and promote regrowth for many men. Keeps also offers both medications orally and topically, which adds to the convenience and flexibility of their platform.
- Dosing concerns. While Keeps offers a range of product formulations, some active ingredients offered in their Rx topicals – such as finasteride – come at doses so high that they leak into the bloodstream and impact systemic hormones not dissimilarly to oral finasteride. As many users often turn to Rx topicals under the assumption that they’re more localized and cause fewer side effects, these larger doses may warrant revisiting by Keeps – or perhaps a change in messaging around these doses.
What Causes Male Pattern Hair Loss?
Androgenic alopecia (AGA) is the medical name of male pattern hair loss. It involves the progressive miniaturization of hair follicles, a process that is induced by androgen sensitivity and driven predominantly by interactions between genes and the hormone dihydrotestosterone (DHT).[1]Ho, C.H., Sood, T., Zito, P.M., (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026)
DHT is the key androgen driving follicular miniaturization and AGA. The 5-alpha reductase (5AR) enzyme binds to free testosterone and converts it into DHT. DHT then binds to cells via androgen receptors and influences the behavior of those cells. In AGA-sensitive hair follicles, this interaction ultimately results in increased rates of hair shedding, followed by progressive hair follicle miniaturization – both of which tend to worsen without treatment.
What Does Keeps Offer?
Keeps offers a range of treatments for hair loss, with a focus on minoxidil and finasteride. In addition, they offer various other products, including shampoos, a styling product, and supplements. Their Rx offerings include reliable, clinically-proven products delivered in multiple formats, such as sprays, gels, and pills.
We will investigate their product range, exploring what their catalog offers, and examine if the science backs their claims.
Keeps Finasteride (1mg Oral)
Formulations
Keeps offers Finasteride as an oral drug; they do not offer topical finasteride without it being in combination with minoxidil. The oral drug offered contains 1 mg finasteride.
What is Finasteride?
Finasteride is a drug originally created to treat benign prostatitic hyperplasia and now commonly used to treat AGA. It is a prescription drug that has been approved by the FDA for oral use, and is commonly also prescribed off-label for topical use.
How Does Finasteride Work?
Finasteride selectively inhibits type II 5AR, the enzyme that converts testosterone to DHT. As DHT is the primary driver of AGA, blocking its production interrupts the hormonal processes causing pattern hair loss. As such, finasteride helps to maintain existing hair and induce hair regrowth.
Clinical trials have demonstrated that finasteride treatment decreased levels of DHT in the scalp by 64-60%.[2]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., … Continue reading Finasteride also contributes to anti-cell death effects, in addition to promoting pathways that are important in hair follicle maintenance.
Clinical Effectiveness
Multiple clinical trials have demonstrated the effectiveness of finasteride in improving hair growth. Long-term trials have shown its ability to enhance hair growth and reduce further hair loss across 1-year, 2-year, and 5-year clinical trials. These trials have demonstrated that approximately 65% of men show increased hair counts after 5 years of use.[3]Finasteride Male Pattern Hair Loss Study Group. (2002). Long-Term (5-Year) Multinational Experience With Finasteride 1 mg in the Treatment of Men With Androgenetic Alopecia. Eur J Dermatol. 12:38-49. … Continue reading,[4]Shapiro, J., Kaufman, K.D. (2003). Use of Finasteride in the Treatment of Men With Androgenetic Alopecia (Male Pattern Hair Loss). J Investig Dermatol Symp Proc. 8(1):20-23. Available at: … Continue reading
Are There Any Side Effects?
Finasteride works by altering certain hormones; as such, it can cause side effects throughout the body. These include altered libido, erectile dysfunction, breast enlargement, or mood alterations.
In controlled clinical trials, side effects are uncommon. A meta-analysis (a study where data from multiple independent studies are analyzed) of 15 clinical trials found that only 5.31% of users experienced sexual side effects when taking 1 mg finasteride, whereas these were seen in 3.05% of those taking a sugar pill.[5]Lee, S., Lee, Y.B., Choe, S.J., Lee, W.S. (2019). Adverse Sexual Effects of Treatment with Finasteride or Dutasteride for Male Androgenetic Alopecia: A Systematic Review and Meta-Analysis. Acta … Continue reading
Although rare, these side effects can seriously impact the lives of those experiencing them, and it is for this reason that finasteride is a prescription-only product that requires medical supervision.
Does the Science Support Keeps’ Claims?
Claim 1: Blocks the production of DHT
Conclusion: True
Finasteride has demonstrated its ability to reduce DHT in multiple clinical trials, in one study showing a 69.4% decrease in DHT in the scalp (although at a higher dose of 5 mg/day).[6]Drake, L., Hordinsky, M., Fiedler, V., et al., (1999). The Effects of Finasteride on Scalp Skin and Serum Androgen Levels in Men with Androgenetic Alopecia. Journal of the American Academy of … Continue reading
Claim 2: Works on a receding hairline and for overall hair thinning
Conclusion: True, but slightly misleading
While finasteride does “work” on a receding hairline, it often does not move a users’ hairline several inches forward – nor does it regrow hair lost 5-10 years ago. In general, drugs like finasteride typically produce a slowing, stopping, or partial reversal of hair loss in 80-90% of men affected by AGA – along with a 10% increase in terminal hair counts over 2 years. Thereafter, hair gains tend to (mostly) plateau. Without treatment, men with androgenic alopecia tend to lose ~5% hair density per year. So, with this in mind, a good rule-of-thumb is that after starting finasteride, a good responder will typically “rewind the clock” on how their hair looked 6-36 months prior. If significant hairline recession happened before then, it’s unlikely to see significant hairline recovery. For more information, see this video.
Encouragingly, finasteride tends to work in most AGA-affected scalp regions.[7]Leyden, J., Dunlap, F., Miller, B. (1999). Finasteride in the Treatment of Men with Frontal Male Pattern Hair Loss. Journal of the American Academy of Dermatology. 40(6). 930–937. Available at: … Continue reading Finasteride acts on all androgen-sensitive scalp areas – the frontal, mid-scalp, and vertex – with some evidence that the vertex (crown) may respond more robustly in some men.[8]Olsen, E.A., Whiting, D.A., Savin, R. (2012). Global Photographic Assessment of Men Aged 18 to 60 Years with Male Pattern Hair Loss Receiving Finasteride 1 mg or Placebo. Journal of the American … Continue reading
Finasteride is effective at improving overall scalp hair thinning.
Claim 3: 90% effective at treating hair loss
Conclusion: True, but requires more context
High-quality, randomized controlled trials (the optimal method of testing claims) have shown that finasteride can slow, stop, or partially reverse hair loss in 80-90% of men with AGA. This has been loosely corroborated by in-clinic, population-level data that acts as a proxy for outcomes of real-world finasteride prescriptions.[9]Sato, A., Takeda, A. (2011). Evaluation of Efficacy and Safety of Finasteride 1 mg in 3177 Japanese Men with Androgenetic Alopecia. Journal of Dermatology. 39(1). 27–32. Available at: … Continue reading,[10]Shin, J., Chung, E., Kim, M., Kim, T., Kim, W., Huh, C. (2018). Evaluation of Long-Term Efficacy of Finasteride in Korean Men with Androgenetic Alopecia Using the Basic and Specific Classification … Continue reading
Having said that, the word “effective” needs further unpacking.
Yes, finasteride is “effective” for 80-90% of men with AGA. However, in this case, the term “effective” is in reference to any slowing, stopping, or partial reversal of hair loss over placebo. That means that being effective might mean you see hair regrowth… but it also might mean you just see hair loss stabilization or, in some cases, a slowing down of your prior speed of hair loss.
For everyday consumers looking to get hair loss support, the term “effective” is more likely to be interpreted as “regrows hair”. But in the world of hair loss research – and in the way Keeps refers to “effective” – the term is actually much broader, and includes those who experience both hair loss stabilization and a slowing down of hair loss, even without reversal. This isn’t necessarily a deliberate misapplication of the term! But it is context that we want readers of this article to have when making treatment decisions.
Claim 4: Results within 4-6 months of consistent use
Conclusion: True, but that’s not the whole story.
Improved hair growth is consistently seen in controlled clinical trials within 6 months of taking finasteride.[11]Van Neste, D., Fuh, V., Sanchez-Pedreno, P. (2000). Finasteride Increases Anagen Hair in Men with Androgenetic Alopecia. British Journal of Dermatology. 143(4). 804–810. Available at: … Continue reading
However, just as with the term “effective”, we also must look at the term “results” and unpack what this means.
Two-year studies on finasteride show that microscopic hair gains typically present in the first 3 months, with those results first beginning to show cosmetically around month 6 (as the hair grows out). However, results don’t stop at month 6. Instead, they keep improving through months 18-24, at which point, hair improvement plateaus. For more information, see this video.
So, if you don’t see results within the first 6 months, please understand that you have 12-18 remaining months before results officially “peak”. There is still a lot of time!
Keeps Minoxidil Solution (5%)
Formulations
Minoxidil exists in oral and topical forms. The oral form requires a prescription, whereas the topical form can be purchased over the counter.
What is Minoxidil?
Minoxidil was originally developed as an oral drug to treat high blood pressure. However, a side effect of this product was noted to be increased hair growth. This was corroborated in follow-up clinical studies on topical minoxidil, and in 1988, the FDA approved topical minoxidil for male pattern hair loss (AGA).[12]Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and Its Use in Hair Disorders: A Review. Drug Design, Development and Therapy. 13. 2777–2786. Available at: … Continue reading
How Does Minoxidil Work?
Minoxidil is thought to stimulate hair growth through a combination of biological mechanisms:
- Local Vasodilation: Minoxidil causes vasodilation, enhancing blood circulation around the hair follicles and delivering more oxygen and nutrients to the follicular environment, creating enhanced conditions for hair growth.[13]Patel, P., Nessel, T.A., Kumar, D.D. (2023). Minoxidil. StatPearls. Available at: http://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: 04 February 2026)
- Anagen Induction and Telogen Shortening: Minoxidil can induce the anagen phase (growth) and shorten the telogen phase (resting).[14]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study … Continue reading By influencing resting hair follicles to re-enter the growth phase, minoxidil treatment causes increased hair density and thickness.
- Prostaglandin Modulation: Minoxidil is thought to increase the production of prostaglandin E2 (PGE2), promoting hair growth, potentially through providing cytoprotective effects and modulating inflammation within the surrounding follicle environment.[15]Michelet, J.F., Commo, S., Billoni, N., Mahé, Y.F., Bernard, B.A. (1997). Activation of Cytoprotective Prostaglandin Synthase-1 by Minoxidil as a Possible Explanation for Its Hair Growth-Stimulating … Continue reading
However, it’s still unclear how, exactly, minoxidil regrows hair. If you’re interested in learning more, see this article on 3 hypotheses for how minoxidil works.
Clinical Effectiveness
The effectiveness of minoxidil can be split into response rates (proportion of men for whom minoxidil works) and regrowth rates.
Response rates
Surveys and clinical studies demonstrate that approximately 60% of men show a response to minoxidil (the slowing, stopping, or reversal of hair loss) within 3-6 months of beginning treatment.[16]Asilian, A., Farmani, A., Saber, M. (2023). Clinical Efficacy and Safety of Low-Dose Oral Minoxidil Versus Topical Solution in the Improvement of Androgenetic Alopecia: A Randomized Controlled Trial. … Continue reading However, increasing the treatment time up to over 12 months does not show continued improvements. Instead, response rates drop to approximately 30%.[17]Rietschel, R.L., Duncan, S.H. (1987). Safety and Efficacy of Topical Minoxidil in the Management of Androgenetic Alopecia. Journal of the American Academy of Dermatology. 16(3). 677–685. Available … Continue reading
After the 12-month mark, the rate of discontinuation is approximately 90%, with “low effect” as the leading reason given for stopping.[18]Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelberg). 13(5). 1157–1169. Available at: … Continue reading
Regrowth rate
Multiple clinical trials have been carried out to assess the effects of minoxidil on hair growth, with the majority demonstrating measurable regrowth after 4-12 months of use.[19]Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the … Continue reading,[20]Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta … Continue reading[21]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study … Continue reading The exact level of regrowth varies; however, estimates of regrowth are approximately 50-70% over 12 months. Few trials carry out assessments beyond 12 months. Those assessing longer-term effects see reduced regrowth after 12 months.[22]Olsen, E.A., DeLong, E.R., Weiner, M.S. (1987). Long-Term Follow-Up of Men with Male Pattern Baldness Treated with Topical Minoxidil. Journal of the American Academy of Dermatology. 16(3). 688–695. … Continue reading
Does the Science Support Keeps’ Claims?
Claim 1: Increases new hair growth in men with AGA
Conclusion: True
Minoxidil has been shown in multiple clinical trials to have a positive effect on hair growth.
Claim 2: Works at the crown and middle of the head
Conclusion: True, but it can also work at the temples
Evidence from clinical trials has not shown that minoxidil only or primarily works at the crown or middle of the head. This misconception arose due to early important trials specifically focusing on these areas, such as a 2002 trial that recruited men with a blading spot on their crown.[23]Olsen, E.A., Dunlap, F.E., Funicella, T. (2002). A Randomized Clinical Trial of 5% Topical Minoxidil Versus 2% Topical Minoxidil and Placebo in the Treatment of Androgenetic Alopecia in Men. Journal … Continue reading
More recent trials have demonstrated the effectiveness of minoxidil across the scalp, with no significant differences in effects seen between the frontotemporal and vertex areas.[24]Kanti, V., Hillmann, K., Kottner, J., Stroux, A., Canfield, D., Blume-Peytavi, U. (2016). Effect of Minoxidil Topical Foam on Frontotemporal and Vertex Androgenetic Alopecia in Men: A 104-Week … Continue reading
Claim 3: Increases hair growth by up to 35%
Conclusion: True, but also unspecific
What does it mean to increase hair growth by up to 35?% This might mean increasing hair length, or hair growth speed, or total hair counts, or vellus hair counts, or terminal hair counts, or hair density including or excluding vellus hairs, or hair diameters.
All in, the term is consumer-friendly, but also unspecific. It implies that users of minoxidil might expect a 35% visual increase in hair on their heads, when this just often isn’t the case. The improvements are often much smaller, much subtler, and shorter-lived relative to finasteride – which tends to sustain results much longer.
Keeps Minoxidil Foam (5%)
The Keeps Minoxidil Foam claims to offer the same benefits as the Minoxidil Solution. It could be assumed that the exact ingredients forming the products are not the same; however, no readily accessible list of ingredients is provided on the website.
Keeps Minoxidil+ Spray
The Minoxidil+ Spray contains additional ingredients aimed at improving minoxidil’s efficacy and supporting hair growth.
Formulation
The Minoxidil+ Spray consists of 5% minoxidil, 0.01% tretinoin, 0.2% caffeine, and 0.1% melatonin.
Clinical Effectiveness
0.01% Tretinoin
Tretinoin (also known as retinoic acid) helps to increase cell turnover in application sites and, resultantly, also helps to improve the penetration and activation of certain hair loss drugs. It has been studied in combination with other hair loss drugs, such as minoxidil.
Tretinoin is thought to increase responsiveness to minoxidil by increasing the activity of the enzyme that converts minoxidil in the body into its active form.[25]Sharma, A., Goren, A., Dhurat, R. (2019). Tretinoin Enhances Minoxidil Response in Androgenetic Alopecia Patients by Upregulating Follicular Sulfotransferase Enzymes. Dermatologic Therapy. 32(3). … Continue reading Clinical trials have shown that the once-daily application of a combination of 5% minoxidil and 0.01% tretinoin is as effective (and safe) as twice-daily application of 5% minoxidil alone.[26]Shin, H.S., Won, C.H., Lee, S.H., Kwon, O.S., Kim, K.H., Eun, H.C. (2007). Efficacy of 5% Minoxidil Versus Combined 5% Minoxidil and 0.01% Tretinoin for Male Pattern Hair Loss. American Journal of … Continue reading
See our article for a more in-depth look at tretinoin.
0.2% Caffeine
Caffeine is a popular (but less-supported) intervention for hair growth. It acts as a phosphodiesterase inhibitor, which may boost IGF-1 levels near hair follicles (amongst other benefits). As a result, limited research suggests that caffeine may prolong the growth stage of the hair cycle, encourage new hair growth, and reduce hair shedding. For more information, see our article assessing the effectiveness of topical caffeine.
The clinical studies supporting the use of topical caffeine (including in conjunction with minoxidil) are generally positive. However, these studies are not without their methodological flaws. For this reason, caffeine ranks lower in terms of regrowth potential and evidence quality relative to FDA-approved hair loss medications, like minoxidil and finasteride.[27]Dressler, C., Blumeyer, A., Rosumeck, S., Arayesh, A., Nast, A. (2017). Efficacy of Topical Caffeine in Male Androgenetic Alopecia. Journal der Deutschen Dermatologischen Gesellschaft. 15(7). … Continue reading For more information, see our Treatment Metrics.
For those using caffeine, please consider it an adjuvant to better-supported interventions – and not a replacement.
0.1% Melatonin
Melatonin is an endogenously-produced hormone that has long been associated with sleep quality. More recently, research suggests this hormone may also have anti-inflammatory effects that may benefit AGA-affected hair follicles.
While the mechanisms for melatonin’s impact on hair loss are not yet fully elucidated, a few clinical studies have shown that it appears to have a positive effect on hair growth and reducing hair loss.[28]Fischer, T.W., Burmeister, G., Schmidt, H.W., Elsner, P. (2004). Melatonin Increases Anagen Hair Rate in Women with Androgenetic Alopecia or Diffuse Alopecia: Results of a Pilot Randomized Controlled … Continue reading[29]Fischer, T.W., Trüeb, R.M., Hänggi, G., Innocenti, M., Elsner, P. (2012). Topical Melatonin for Treatment of Androgenetic Alopecia. International Journal of Trichology. 4(4). 236–245. Available … Continue reading
Like caffeine, the research on topical melatonin for hair regrowth is limited. Our summary of the clinical data can be found here.
However, like with all topical ingredients, there must be dosing considerations.
Currently, the only concentration of topical melatonin shown to (1) improve hair parameters and (2) not impact serum melatonin levels is a daily dose equating to 0.1 mg of topical melatonin exposure per day. This can be achieved through a variety of means: 0.0033% x 3 mL daily, 0.005% x 2 mL daily, or even 0.01% x 1 mL daily.
Keeps’ melatonin content is 10x higher than this – and likely exposes users to 1-2 mg per day topically, which likely increases circulating melatonin levels and may come with an increased risk of melatonin-produced side effects. As such, users may want to keep this in mind and apply the topical only at night – during windows close to bed. Or, they may want to opt for a minoxidil + melatonin combination product that uses lower doses of melatonin that better localize to just the scalp skin. One example would be the combination Rx topicals from Ulo, a telehealth brand we helped to cofound, in part based on these dosing considerations that seemed neglected by other brands:
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Does the Science Support Keeps’ Claims?
Claim 1: Topical minoxidil improves blood flow to hair follicles to boost hair regrowth by up to 35%
Conclusion: True, but requires a little more context
As discussed above, minoxidil might boost hair counts microscopically by 35% within 4-6 months. However, this rarely results in a 35% visual improvement to hair density – as microscopic hair count changes do not linearly track with cosmetic changes to hair volume. Consumers may want to understand this, especially those who don’t closely follow hair loss research.
Claim 2: Tretinoin gently exfoliates to enhance minoxidil’s efficacy, making for easy once-daily application
Conclusion: Mostly true
Tretinoin has been shown to enhance minoxidil’s efficacy to enable once-daily instead of twice-daily application. This comes through two factors: enhancing (1) skin penetration, and (2) drug activation. The former is a close proxy for exfoliation, and even though that’s not exactly how tretinoin works, the language is more consumer-friendly and mostly within agreement of the science.
Claim 3: Caffeine supports thicker, healthier hair growth and makes thinning hair look thicker
Conclusion: Requires larger, better-designed studies
Preliminary evidence suggests caffeine might improve hair parameters. However, the quality of evidence supporting caffeine remains incredibly low relative to the proven results of minoxidil and finasteride. For this reason, caffeine may be worth exploring as an adjuvant (i.e., addition) to be used alongside better-supported ingredients. Fortunately, Keeps’ topical formulation does this with the inclusion of minoxidil.
Claim 4: Melatonin helps increase hair growth on the scalp
Conclusion: Requires larger, better-designed studies
Studies have demonstrated that melatonin increases hair growth and reduces hair loss. But, like with caffeine, the evidence quality remains low.
Topical Finasteride and Minoxidil Gel
Formulation
Keeps offers this two-in-one prescription topical treatment consisting of 0.25% finasteride and 5% minoxidil.
Clinical Effectiveness
Finasteride and minoxidil function through two separate mechanisms. As such, their combination offers users the opportunity to attack hair loss from two different angles.
Multiple clinical trials have demonstrated the efficacy of combining finasteride with minoxidil, showing that the combined therapy is more effective than minoxidil or finasteride alone.[30]Suchonwanit, P., Srisuwanwattana, P., Chalermroj, N., Khunkhet, S. (2018). A Randomized, Double-Blind Controlled Study of the Efficacy and Safety of Topical Solution of 0.25% Finasteride Admixed with … Continue reading,[31]Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, … Continue reading[32]Bharadwaj, A.V., Mendiratta, V., Rehan, H.S., Tripathi, S. (2023). Comparative Efficacy of Topical Finasteride (0.25%) in Combination with Minoxidil (5%) Against 5% Minoxidil or 0.25% Finasteride … Continue reading, with a meta-analysis also backing up these findings.[33]Li, Y., Huang, Q., Zhou, Z., Zhang, Y. (2025). Comparing Minoxidil-Finasteride Mixed Solution with Minoxidil Solution Alone for Male Androgenetic Alopecia: A Systematic Review and Meta-Analysis of … Continue reading
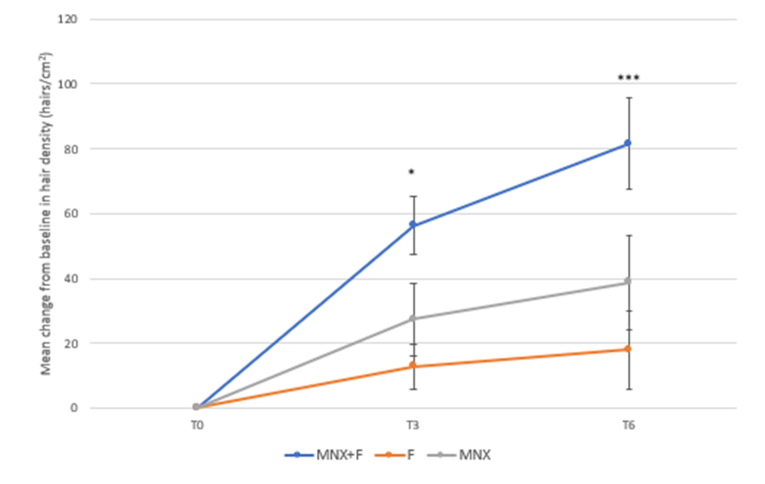
Figure 6. The combination of minoxidil and finasteride (MNX+F; blue line) shows improved hair density compared to both finasteride (F; orange line) and minoxidil (MNX; gray line) after 3 months and 6 months of treatment. Adapted from Figure 2.[34]Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, … Continue reading Image used under Creative Commons License.
Does the Science Support Keeps’ Claims?
Claim 1: Finasteride stops hair loss by blocking the production of DHT, the hormone responsible for male pattern baldness
Conclusion: True
Studies have shown that the topical application of finasteride also successfully lowers DHT levels in the scalp.[35]Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in … Continue reading
Claim 2: Minoxidil increases blood flow to hair follicles to stimulate hair regrowth
Conclusion: True
Minoxidil has been shown in multiple clinical trials to have a positive effect on hair growth.
Keeps Daily Hair Defense Supplement
Formulation
The Daily Hair Defense Supplement contains saw palmetto, vitamins A, B6, B12, C, D, calcium, selenium, zinc, and biotin.
Clinical Effectiveness
Saw Palmetto
Saw palmetto is a popular hair growth supplement as it does not require a prescription to obtain. But is it actually effective?
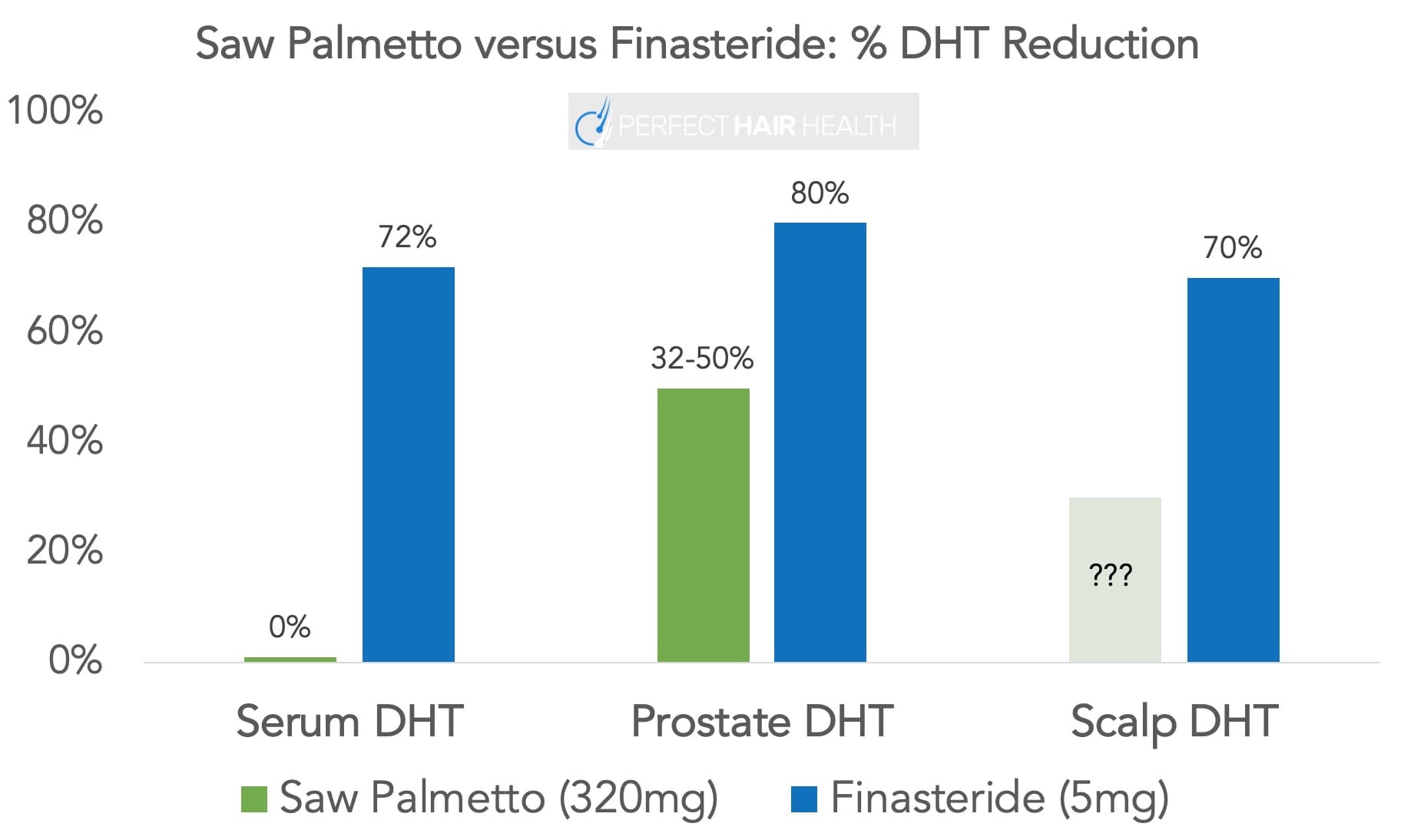
Early studies demonstrated positive results, showing that 60% of men taking 200 mg of saw palmetto experienced some degree of hair regrowth compared to 11% in the control group, although the researchers agreed that more work needed to be done given the small size of the trial.[36]Prager, N., Bickett, K., French, N., Marcovici, G. (2002). A Randomized, Double-Blind, Placebo-Controlled Trial to Determine the Effectiveness of Botanically Derived Inhibitors of 5-Alpha-Reductase … Continue reading
Saw palmetto is able to reduce DHT, although not to the same degree as finasteride, resulting in reduced effectiveness at inducing hair growth compared to finasteride.[37]Rossi, A., Mari, E., Scarnò, M. (2012). Comparative Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A Two-Year Study. International Journal of Immunopathology and … Continue reading However, hair growth was still seen on saw palmetto supplementation, indicating its potential for use in a supplement.
For more information, see our article on saw palmetto. Or review saw palmetto inside our Ingredient Database.
Vitamins A, B6, B12, C, and D
There is little-to-no convincing clinical evidence that supplementation with Vitamins A, B6, B12, C, and D has any effect on hair growth, although they may have positive effects in cases where people have a severe deficiency.[38]Cheung, E.J., Sink, J.R., English, J.C. (2016). Vitamin and Mineral Deficiencies in Patients With Telogen Effluvium: A Retrospective Cross-Sectional Study. Journal of Drugs in Dermatology. 15(10). … Continue reading
Calcium
No strong clinical trials have shown any role of calcium in hair growth.[39]Almohanna, H.M., Ahmed, A.A., Tsatalis, J.P., Tosti, A. (2018). The Role of Vitamins and Minerals in Hair Loss: A Review. Dermatology and Therapy (Heidelberg). 9(1). 51–70. Available at: … Continue reading
Selenium
Both severe selenium deficiency and too much selenium can cause hair loss. A clinical trial in chemotherapy patients receiving a selenium supplement showed reduced hair loss, but no successful clinical trials have been carried out in people representative of the general population (i.e., individuals most likely to buy the Keeps supplements).[40]Almohanna, H.M., Ahmed, A.A., Tsatalis, J.P., Tosti, A. (2018). The Role of Vitamins and Minerals in Hair Loss: A Review. Dermatology and Therapy (Heidelberg). 9(1). 51–70. Available at: … Continue reading
Biotin
Biotin deficiencies also cause hair loss. However, they have to be extreme – often caused by rare genetic mutations (i.e., biotinidase deficiencies), long-term abuse of alcohol, and/or irritable bowel disease. To date, no high-quality clinical trials have demonstrated any effectiveness for otherwise-healthy people supplementing with biotin to improve androgenic alopecia.[41]Yelich, A., Jenkins, H., Holt, S., Miller, R. (2024). Biotin for Hair Loss: Teasing Out the Evidence. Journal of Clinical and Aesthetic Dermatology. 17(8). 56–61. Available at: … Continue reading
Does the Science Support Keeps’ Claims?
Claim 1: The key ingredient, saw palmetto, stabilizes hair loss and promotes healthy hair growth
Conclusion: Potentially misleading
Saw palmetto is thought to support hair growth and reduce hair loss, although not as successfully as finasteride. While short-term (i.e., 6-month) clinical studies have shown that saw palmetto can improve hair loss in those with AGA, and long-term (i.e., 2-year) studies show hair stabilization – the long-term studies did not rely on objective hair counting metrics, such as a phototrichogram. Instead, they relied on visual observations made by the investigators. For these reasons, the evidence supporting long-term stabilization of saw palmetto remains limited. Our personal opinion is that it likely helps, but not nearly as much as more powerful DHT reducers, and its durability long-term remains a subject of debate. For more information, see this article.
Claim 2: Vitamins A, B6, B12, C, D, Calcium, Selenium, Zinc, and Biotin support overall health, immune function, and vitality
Conclusion: True
These vitamins and minerals do not have any significant effects on hair growth on otherwise-healthy people. But they may contribute to improved overall health by reducing the risk of deficiency.
Problems with Keeps’ Approach: Our Recommendations
Overall, Keeps provides a range of comprehensive and affordable offerings for those with androgenic alopecia – including OTC products and Rx medications. Their range of topical formulations (gels, sprays, serums) also offer several alternatives for those still looking to use topicals, but may need to make adjustments due to having a sensitive scalp and/or longer hair. In our eyes, these are all steps in the right direction. The brand is a positive force in the telehealth haircare space.
Having said that, we also see areas of opportunity for Keeps to improve both personalization and outcomes for consumers. Here are a few:
1. Better Guidance On How To Improve Results
In the world of hair loss treatments, many consumers often quit therapies due to the perception of “no effect”. This often happens because of an educational gap:
- Consumers don’t often have clear guidance on how long to use products before expecting results. In Keeps’ marketing language on finasteride, there is an emphasis on results occurring within 6 months. While this is often true microscopically and, in some cases, cosmetically – the totality of clinical data on finasteride show that results continue to improve through 18-24 months before plateauing. For this reason, we see an opportunity for Keeps to update their marketing language to specify that, if someone isn’t seeing results in 6 months, that doesn’t mean they’re not getting results. They still have 12-18 more months of progress before being in a fully-informed position to judge. Results will happen sooner for some, and later for others.
- Consumers don’t often have clear guidance on “escalation” protocols. Non-responders of Keeps’ 5% minoxidil might want to learn that the addition of topical tretinoin might amplify their results and put them in a better position for success. But for those purchasing Keeps’ 5% minoxidil, this escalation protocol isn’t always clearly stated. Similarly, for true non-responders to finasteride – or for those whose goals are to maximize hair gains – oral dutasteride can help achieve this.[42]Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study … Continue reading This option is also not available with Keeps, limiting the opportunity for effective hair growth. However, at the time of this writing, Keeps does not yet offer oral dutasteride to its customers.
2. More Flexibility With Mega-Dosed Topicals
One of Keeps’ strengths is its flexibility of its topical base formulas – including gel, spray, and serum options. But there remains an area of opportunity to expand this flexibility not just to its base formulas, but also its ingredient doses.
This is especially true with two ingredients that have a high capacity to travel from the scalp into the body and potentiate side effects: namely, finasteride and melatonin.
Finasteride Dosing Concerns
Keeps offers combination topicals (if prescribed) of 5% minoxidil and 0.25% finasteride. Many consumers turn to hair loss topicals because they’re hoping to avoid the systemic effects of drugs. However, what isn’t always communicated to consumers seeking Rx topicals is that high-strength concentrations of finasteride (i.e., 0.25%) are virtually guaranteed to go systemic, and lower blood levels of DHT (our best proxy for systemic absorption) by nearly the same amount as oral finasteride.{{Piraccini, B. M., Blume-Peytavi, U., Scarci, F., Jansat, J. M., Falqués, M., Otero, R., Tamarit, M. L., et al. (2022). Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial. *Journal of the European Academy of Dermatology and Venereology.* 36(2). 286–294. Available at: https://doi.org/10.1111/jdv.17738}}
This issue has been recognized by the FDA, who have recently issued a warning for topical finasteride, stating that “Absorption of finasteride through the skin into the bloodstream is expected, and the reports describe adverse events that are consistent with those reported in association with the approved oral finasteride products”.[43]U.S. Food and Drug Administration. (n.d.). FDA alerts health care providers, compounders, and consumers about potential risks associated with compounded drugs. *FDA.* Available at: … Continue reading
Encouragingly, there is a clinical study showing that an ultra low-dose of topical finasteride (0.005% x 2 mL daily) improved hair parameters without affecting serum DHT levels. We discuss the study in this article.
At the time of this writing, Keeps does not offer this ultra-low dose of topical finasteride. They only offer the high-strength topical finasteride. And while an ultra-low dose of topical finasteride does not guarantee zero systemic absorption or complete protection of any side effect, we think the lower-dose is better-aligned with the ethos of why hair loss sufferers pursue topicals in the first place: an attempt to localize a medication’s impact mainly to the scalp.
Melatonin Dosing Concerns
Similarly, Keeps offers an Rx topical containing 0.1% melatonin – which is supported by the clinical literature and may improve hair parameters in those with androgenic alopecia.
However, this dose of melatonin (i.e., 1 mg per mL) has also been shown to raise circulating levels of melatonin throughout the body – with no large, randomized clinical trials to tease out whether these elevated melatonin levels interfere were sleep quality or cause drowsiness.
At the same time, there are studies showing that low-dose topical melatonin (i.e., 0.1 mg per day, or 1/10th the concentration offered by Keeps if applying 1 mL daily) can improve also hair parameters, but without significantly impacting serum melatonin levels. At least for us, the lower concentration of melatonin appears to better-align with the goals of most topical users: to localize a medication’s effects and reduce the potential for side effects. And so we see another area of opportunity for Keeps to revisit the concentration of this ingredient, as well.
For those interested in more customization, more flexible doses of topical finasteride, and lower concentrations of melatonin that better localize to the scalp – we created the telehealth brand, Ulo, to finally offer consumers this level of personalization.
Keeps does a lot of things right, and we’re thrilled that they exist to serve hair loss sufferers. But for those particularly interested in topical localization, side effect mitigation, and an added level of personalization – Ulo was built to fill this large gap in the marketplace. Not only do we offer clear escalation protocols for non-responders to treatments (i.e., topical minoxidil + tretinoin, oral dutasteride, etc.), but we also offer lower-dose topicals that may finally enable those with higher drug sensitivities to finally begin to tolerate (and benefit from) hair growth medications. For these reasons, we’re thrilled to be cofounders of the brand. You can learn more here:
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Final Remarks
Keeps offers multiple clinically-backed medications via a range of delivery methods, providing a broad and accessible entry point for hair loss treatments that are proven to work. These treatments have the potential to successfully slow hair loss and increase hair growth in many users.
Even still, there are areas of opportunity where Keeps may want to expand their core offerings for hair loss sufferers. This may include lower-dose finasteride and melatonin topical options that better localize to the scalp. It may also include the addition of oral dutasteride for those looking to unlock bigger hair gains than finasteride.
All in, Keeps enables easier access to clinically-proven hair loss medications via their telehealth model. We hope they consider our above recommendations, and in the interim, we wish them continued success.
References[+]
References ↑1 Ho, C.H., Sood, T., Zito, P.M., (2024), Androgenetic Alopecia. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ (Accessed: 07 January 2026) ↑2 Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., Carrington, P., Gencheff, C., Cole, G.W., Pariser, D.M., Epstein, E.S., Tanaka, W., Dallob, A.D., Vandormael, K., Geissler, L. and Waldstreicher, J. (1999). The Effects of Finasteride on Scalp Skin and Serum Androgen Levels in Men with Androgenetic Alopecia. J Am Acad Dermatol. 41(4), pp.550-554. Available at: https://pubmed.ncbi.nlm.nih.gov/10495374/ ↑3 Finasteride Male Pattern Hair Loss Study Group. (2002). Long-Term (5-Year) Multinational Experience With Finasteride 1 mg in the Treatment of Men With Androgenetic Alopecia. Eur J Dermatol. 12:38-49. Available at: https://pubmed.ncbi.nlm.nih.gov/11809594/ ↑4 Shapiro, J., Kaufman, K.D. (2003). Use of Finasteride in the Treatment of Men With Androgenetic Alopecia (Male Pattern Hair Loss). J Investig Dermatol Symp Proc. 8(1):20-23. Available at: https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑5 Lee, S., Lee, Y.B., Choe, S.J., Lee, W.S. (2019). Adverse Sexual Effects of Treatment with Finasteride or Dutasteride for Male Androgenetic Alopecia: A Systematic Review and Meta-Analysis. Acta Dermato-Venereologica. 99(1). 12–17. Available at: https://doi.org/10.2340/00015555-3035 ↑6 Drake, L., Hordinsky, M., Fiedler, V., et al., (1999). The Effects of Finasteride on Scalp Skin and Serum Androgen Levels in Men with Androgenetic Alopecia. Journal of the American Academy of Dermatology. 41(4). 550–554. Available at: https://doi.org/10.1016/S0190-9622(99)80051-6 ↑7 Leyden, J., Dunlap, F., Miller, B. (1999). Finasteride in the Treatment of Men with Frontal Male Pattern Hair Loss. Journal of the American Academy of Dermatology. 40(6). 930–937. Available at: https://doi.org/10.1016/S0190-9622(99)70081-2 ↑8 Olsen, E.A., Whiting, D.A., Savin, R. (2012). Global Photographic Assessment of Men Aged 18 to 60 Years with Male Pattern Hair Loss Receiving Finasteride 1 mg or Placebo. Journal of the American Academy of Dermatology. 67(3). 379–386. Available at: https://doi.org/10.1016/j.jaad.2011.10.027 ↑9 Sato, A., Takeda, A. (2011). Evaluation of Efficacy and Safety of Finasteride 1 mg in 3177 Japanese Men with Androgenetic Alopecia. Journal of Dermatology. 39(1). 27–32. Available at: https://doi.org/10.1111/j.1346-8138.2011.01378.x ↑10 Shin, J., Chung, E., Kim, M., Kim, T., Kim, W., Huh, C. (2018). Evaluation of Long-Term Efficacy of Finasteride in Korean Men with Androgenetic Alopecia Using the Basic and Specific Classification System. Journal of Dermatology. 46(2). 139–143. Available at: https://doi.org/10.1111/1346-8138.14719 ↑11 Van Neste, D., Fuh, V., Sanchez-Pedreno, P. (2000). Finasteride Increases Anagen Hair in Men with Androgenetic Alopecia. British Journal of Dermatology. 143(4). 804–810. Available at: https://doi.org/10.1046/j.1365-2133.2000.03780.x ↑12 Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and Its Use in Hair Disorders: A Review. Drug Design, Development and Therapy. 13. 2777–2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑13 Patel, P., Nessel, T.A., Kumar, D.D. (2023). Minoxidil. StatPearls. Available at: http://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: 04 February 2026) ↑14, ↑21 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study Protocol. Skin Research and Technology. 26(4). 542–557. Available at: https://doi.org/10.1111/srt.12827 ↑15 Michelet, J.F., Commo, S., Billoni, N., Mahé, Y.F., Bernard, B.A. (1997). Activation of Cytoprotective Prostaglandin Synthase-1 by Minoxidil as a Possible Explanation for Its Hair Growth-Stimulating Effect. Journal of Investigative Dermatology. 108(2). 205–209. Available at: https://doi.org/10.1111/1523-1747.ep12334249 ↑16 Asilian, A., Farmani, A., Saber, M. (2023). Clinical Efficacy and Safety of Low-Dose Oral Minoxidil Versus Topical Solution in the Improvement of Androgenetic Alopecia: A Randomized Controlled Trial. Journal of Cosmetic Dermatology. 23(3). 949–957. Available at: https://doi.org/10.1111/jocd.16086 ↑17 Rietschel, R.L., Duncan, S.H. (1987). Safety and Efficacy of Topical Minoxidil in the Management of Androgenetic Alopecia. Journal of the American Academy of Dermatology. 16(3). 677–685. Available at: https://doi.org/10.1016/S0190-9622(87)70087-5 ↑18 Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelberg). 13(5). 1157–1169. Available at: https://doi.org/10.1007/s13555-023-00919-x ↑19 Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the Treatment of Androgenetic Alopecia in Men. Journal of the American Academy of Dermatology. 57(5). 767–774. Available at: https://doi.org/10.1016/j.jaad.2007.04.012 ↑20 Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta Dermatovenerologica Alpina, Pannonica et Adriatica. 25(3). 41–44. Available at: https://doi.org/10.15570/actaapa.2016.12 ↑22 Olsen, E.A., DeLong, E.R., Weiner, M.S. (1987). Long-Term Follow-Up of Men with Male Pattern Baldness Treated with Topical Minoxidil. Journal of the American Academy of Dermatology. 16(3). 688–695. Available at: https://doi.org/10.1016/S0190-9622(87)70089-9 ↑23 Olsen, E.A., Dunlap, F.E., Funicella, T. (2002). A Randomized Clinical Trial of 5% Topical Minoxidil Versus 2% Topical Minoxidil and Placebo in the Treatment of Androgenetic Alopecia in Men. Journal of the American Academy of Dermatology. 47(3). 377–385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑24 Kanti, V., Hillmann, K., Kottner, J., Stroux, A., Canfield, D., Blume-Peytavi, U. (2016). Effect of Minoxidil Topical Foam on Frontotemporal and Vertex Androgenetic Alopecia in Men: A 104-Week Open-Label Clinical Trial. Journal of the European Academy of Dermatology and Venereology. 30(7). 1183–1189. Available at: https://doi.org/10.1111/jdv.13324 ↑25 Sharma, A., Goren, A., Dhurat, R. (2019). Tretinoin Enhances Minoxidil Response in Androgenetic Alopecia Patients by Upregulating Follicular Sulfotransferase Enzymes. Dermatologic Therapy. 32(3). e12915. Available at: https://doi.org/10.1111/dth.12915 ↑26 Shin, H.S., Won, C.H., Lee, S.H., Kwon, O.S., Kim, K.H., Eun, H.C. (2007). Efficacy of 5% Minoxidil Versus Combined 5% Minoxidil and 0.01% Tretinoin for Male Pattern Hair Loss. American Journal of Clinical Dermatology. 8(5). 285–290. Available at: https://doi.org/10.2165/00128071-200708050-00003 ↑27 Dressler, C., Blumeyer, A., Rosumeck, S., Arayesh, A., Nast, A. (2017). Efficacy of Topical Caffeine in Male Androgenetic Alopecia. Journal der Deutschen Dermatologischen Gesellschaft. 15(7). 734–741. Available at: https://doi.org/10.1111/ddg.13271 ↑28 Fischer, T.W., Burmeister, G., Schmidt, H.W., Elsner, P. (2004). Melatonin Increases Anagen Hair Rate in Women with Androgenetic Alopecia or Diffuse Alopecia: Results of a Pilot Randomized Controlled Trial. British Journal of Dermatology. 150(2). 341–345. Available at: https://doi.org/10.1111/j.1365-2133.2004.05685.x ↑29 Fischer, T.W., Trüeb, R.M., Hänggi, G., Innocenti, M., Elsner, P. (2012). Topical Melatonin for Treatment of Androgenetic Alopecia. International Journal of Trichology. 4(4). 236–245. Available at: https://doi.org/10.4103/0974-7753.111199 ↑30 Suchonwanit, P., Srisuwanwattana, P., Chalermroj, N., Khunkhet, S. (2018). A Randomized, Double-Blind Controlled Study of the Efficacy and Safety of Topical Solution of 0.25% Finasteride Admixed with 3% Minoxidil vs 3% Minoxidil Solution in the Treatment of Male Androgenetic Alopecia. Journal of the European Academy of Dermatology and Venereology. 32(12). 2257–2263. Available at: https://doi.org/10.1111/jdv.15171 ↑31 Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, Controlled, Assessor Blinded, 3-Arm, Pilot Trial. Journal of Cosmetic Dermatology. 23(2). 502–509. Available at: https://doi.org/10.1111/jocd.15953 ↑32 Bharadwaj, A.V., Mendiratta, V., Rehan, H.S., Tripathi, S. (2023). Comparative Efficacy of Topical Finasteride (0.25%) in Combination with Minoxidil (5%) Against 5% Minoxidil or 0.25% Finasteride Alone in Male Androgenetic Alopecia: A Pilot, Randomized Open-Label Study. International Journal of Trichology. 15(2). 56–62. Available at: https://doi.org/10.4103/ijt.ijt_72_22 ↑33 Li, Y., Huang, Q., Zhou, Z., Zhang, Y. (2025). Comparing Minoxidil-Finasteride Mixed Solution with Minoxidil Solution Alone for Male Androgenetic Alopecia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Frontiers in Medicine. 12. 1632139. Available at: https://doi.org/10.3389/fmed.2025.1632139 ↑34 Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, Controlled, Assessor Blinded, 3-Arm, Pilot Trial. Journal of Cosmetic Dermatology. 23(2). 502–509. Available at: https://doi.org/10.1111/jocd.15953 ↑35 Lee, S.W., Juhasz, M., Mobasher, P., Ekelem, C., Mesinkovska, N.A. (2018). A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. Journal of Drugs in Dermatology. 17(4). 457–463. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC6609098/ ↑36 Prager, N., Bickett, K., French, N., Marcovici, G. (2002). A Randomized, Double-Blind, Placebo-Controlled Trial to Determine the Effectiveness of Botanically Derived Inhibitors of 5-Alpha-Reductase in the Treatment of Androgenetic Alopecia. Journal of Alternative and Complementary Medicine. 8(2). 143–152. Available at: https://doi.org/10.1089/acm.2002.8.143 ↑37 Rossi, A., Mari, E., Scarnò, M. (2012). Comparative Effectiveness of Finasteride vs Serenoa Repens in Male Androgenetic Alopecia: A Two-Year Study. International Journal of Immunopathology and Pharmacology. 25(4). 1167–1173. Available at: https://doi.org/10.1177/039463201202500435 ↑38 Cheung, E.J., Sink, J.R., English, J.C. (2016). Vitamin and Mineral Deficiencies in Patients With Telogen Effluvium: A Retrospective Cross-Sectional Study. Journal of Drugs in Dermatology. 15(10). 1235–1237. Available at: https://pubmed.ncbi.nlm.nih.gov/27741341/ ↑39, ↑40 Almohanna, H.M., Ahmed, A.A., Tsatalis, J.P., Tosti, A. (2018). The Role of Vitamins and Minerals in Hair Loss: A Review. Dermatology and Therapy (Heidelberg). 9(1). 51–70. Available at: https://doi.org/10.1007/s13555-018-0278-6 ↑41 Yelich, A., Jenkins, H., Holt, S., Miller, R. (2024). Biotin for Hair Loss: Teasing Out the Evidence. Journal of Clinical and Aesthetic Dermatology. 17(8). 56–61. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC11324195/ ↑42 Harcha, W. G., Barboza Martínez, J., Tsai, T.-F., Katsuoka, K., Kawashima, M., Tsuboi, R., Barnes, A., Ferron-Brady, G., & Chetty, D. (2014). A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 70(3). 489–498. Available at: https://doi.org/10.1016/j.jaad.2013.10.049 ↑43 U.S. Food and Drug Administration. (n.d.). FDA alerts health care providers, compounders, and consumers about potential risks associated with compounded drugs. *FDA.* Available at: https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-consumers-potential-risks-associated-compounded (Accessed: November 2025) Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn More
Catherine Kennedy, PhD
Catherine is a scientist and researcher with a background in biosciences and clinical neuroscience. She completed an integrated master’s degree in Biosciences (MBiol) before earning a PhD in Clinical Neurosciences at the University of Cambridge. Throughout her career, she has contributed to the scientific literature through the publication of numerous peer-reviewed research papers.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down