- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
XYON Review: Do Their Products Actually Regrow Hair?
-
Minoxidil Before and After Photos [2026] | Does It Work?
-
How to Get Finasteride: Is It Over the Counter?
-
Keeps Review: The Truth About Their Hair Loss Treatments?
-
7 Best Oils for Hair Growth
-
Hims Hair Growth Reviews: The Pros, Cons, and Real Results
-
Topical Finasteride Before and After: Real Case Studies
-
How to Reduce the Risk of Finasteride Side Effects
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
ArticlesXYON Review: Do Their Products Actually Regrow Hair?
First Published Mar 25 2026Last Updated Mar 25 2026Company ReviewsResearched & Written By:Catherine Kennedy, PhDReviewed By:Rob English, Medical EditorWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
XYON markets itself as a science-first hair loss brand, but do its products really deliver? This review breaks down XYON’s prescription and OTC lineup, digs into the evidence behind finasteride, dutasteride, and minoxidil, and puts its SiloxysSystem™ gel claims under the microscope. Expect a clear look at efficacy, dosing (including “mega-dosed” topicals), side-effect risk, transparency, and value.
Full Article
In the rapidly growing world of telehealth brands aimed at targeting hair loss, XYON has positioned itself as a clinician-founded, science-based solution. They focus on innovation and products that prioritize safety, with the goal of empowering both men and women. In keeping with this, they have a strong selection of prescription medications to treat hair loss alongside other non-prescription hair care options.
XYON’s main selling point is its SiloxysSystem™ gel delivery system, which they position as maximizing hair growth potential while helping to reduce the risk of side effects. But how does the science back up XYON’s claims? We’ll examine the range of products offered by the brand, how their formulations stack up against the science, and whether they are as effective as they claim to be.
Interested in Topical Minoxidil?
High-strength topical minoxidil available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Key Takeaways
- XYON uses finasteride, dutasteride, and minoxidil, which are clinically proven for hair loss, but often at unusually high topical concentrations.
- The SiloxysSystem™ proprietary gel may reduce systemic absorption, but there are not yet any peer-reviewed studies that verify XYON’s data.
- Fixed formulations, sparse ingredient disclosure, and premium pricing may limit suitability for some users.
What Does XYON Offer?
XYON offers a range of prescription hair loss products in addition to over-the-counter products, such as their shampoo and conditioners. Their prescription-dependent products include reliable, clinically-proven products with a choice of delivery mechanisms – oral, gels, and solutions.
We will investigate their product range, exploring what they offer and whether the science truly supports their hair loss solutions.
Oral Finasteride
Starting off with one of the original prescription hair loss drugs, oral finasteride is an FDA-approved treatment for androgenic alopecia (AGA) in men. It was first approved in 1997 and has been widely studied, providing a wealth of information from clinical trials and experimental studies supporting its use as an effective treatment.
Finasteride works by selectively inhibiting 5-alpha reductase (5AR), the key enzyme responsible for converting testosterone to dihydrotestosterone (DHT). DHT is the primary driver of AGA. It binds to androgen receptors, resulting in the miniaturization of hair follicles. The blockage of DHT production by finasteride interrupts the hormonal processes causing hair loss, therefore helping to maintain existing hair and induce hair growth.
Clinical trials have demonstrated that finasteride treatment decreased levels of DHT in the scalp by 64-60%.[1]Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., … Continue reading They have also shown the effectiveness of finasteride in enhancing hair growth, with long-term trials of up to 5 years demonstrating that approximately 65% of men taking finasteride showed increased hair counts across the trials, including after 5-years of use.[2]Finasteride Male Pattern Hair Loss Study Group. (2002). Long-Term (5-Year) Multinational Experience With Finasteride 1 mg in the Treatment of Men With Androgenetic Alopecia. Eur J Dermatol. 12:38-49. … Continue reading,[3]Shapiro, J., Kaufman, K.D. (2003). Use of Finasteride in the Treatment of Men With Androgenetic Alopecia (Male Pattern Hair Loss). J Investig Dermatol Symp Proc. 8(1):20-23. Available at: … Continue reading
Due to finasteride’s actions on certain hormones, it can cause side effects, including libido, erectile dysfunction, breast enlargement, or mood alterations. While rare (with long-term studies suggesting that they are experienced in 2.6-13.4% of men{Olsen, E. A., Hordinsky, M., Whiting, D., Stough, D., Hobbs, S., Ellis, M. L., Wilson, T., Rittmaster, R. S., & Dutasteride Alopecia Research Team. (2006). The importance of dual 5α-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride. Journal of the American Academy of Dermatology. 55(6). 1014–1023. Available at: https://doi.org/10.1016/j.jaad.2006.05.007}}, [4]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading,[5]Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male … Continue reading), these side effects can be serious. It is for this reason that finasteride is a prescription-only product requiring medical supervision.
The oral finasteride offered by XYON is fairly standard, consisting of a 1 mg tablet taken once daily. Besides their strong branding, this pill will not differ from those offered by any other brands.
Topical Finasteride with SiloxysSystem™ Gel
Ingredients: 2.5% finasteride, SiloxysSystem™ Gel
XYON offers topical finasteride as an option to lower DHT at the scalp while reducing whole-body effects, and therefore reducing the risk of side effects. But is this true?
Topical finasteride has long been seen as the solution to side effects occasionally seen with oral treatment. Clinical studies have supported the effectiveness of finasteride in reducing scalp DHT levels and in the resultant decrease in hair loss. In one placebo-controlled study, treatment with a 0.25% finasteride solution once per day (50-200 microliter volume) resulted in significantly greater hair counts after 24 weeks compared to the placebo, and showed similar results to the oral finasteride treatment.[6]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., et al. (2021). Efficacy and Safety of Topical Finasteride Spray Solution for Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical … Continue reading
Similar results were seen in a study comparing 1% finasteride topical gel with oral finasteride. They determined that the therapeutic effects of the gel and the tablet were similar, with no significant differences seen in hair thickness or hair counts.[7]Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing the Therapeutic Effects of Finasteride Gel and Tablet in Treatment of the Androgenetic Alopecia. Indian Journal of Dermatology, … Continue reading
The effectiveness of topical finasteride is therefore supported by multiple clinical studies, but what about its side effects?
Topical finasteride has been shown to reduce but not eliminate the reduction of DHT throughout the rest of the body. A topical solution of 0.25% finasteride reduced serum DHT by 34.5% compared to the 55.6% reduction seen on oral finasteride use.[8]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., et al. (2021). Efficacy and Safety of Topical Finasteride Spray Solution for Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical … Continue reading Other studies have reported a range of DHT reduction with the use of topical formulations; all agree that absorption is lower than with oral administration, but not absent.
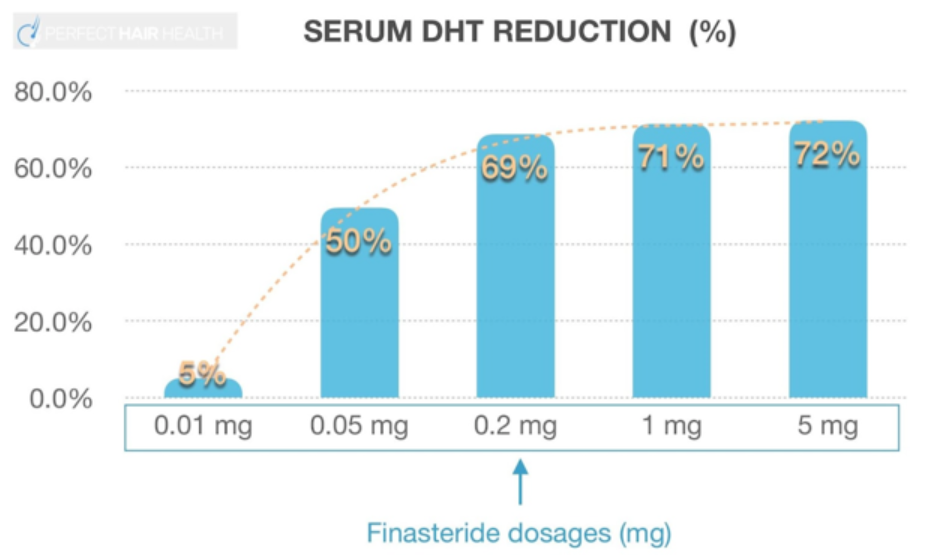
The shape of the dose-response curve for finasteride is non-linear (logarithmic), meaning that even very low levels of finasteride can produce a significant reduction in DHT. The graph below is from oral finasteride; however, the effect of the systemic absorption of topical finasteride is likely to be similar.
Figure 3. Even very low doses of finasteride (0.05 mg) can significantly reduce the amount of DHT in the serum.
As a result, while topical finasteride use may decrease side effects (due to less DHT suppression compared to oral use), it likely does not eliminate them, as serum DHT will still be lowered. One study found that 2.8% of topical finasteride users reported sexual adverse events (such as decreased libido and erectile dysfunction) compared to 4.8% on oral finasteride.[9]Piraccini, B.M., Blume-Peytavi, U., Scarci, F., et al. (2021). Efficacy and Safety of Topical Finasteride Spray Solution for Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical … Continue reading
In fact, the persistant issue of side effects from topical finasteride (despite claims from haircare brands) has resulted in the FDA releasing a warning for topical finasteride, stating that “Absorption of finasteride through the skin into the bloodstream is expected, and the reports describe adverse events that are consistent with those reported in association with the approved oral finasteride products”.[10]U.S. Food and Drug Administration. (n.d.). FDA alerts health care providers, compounders, and consumers about potential risks associated with compounded drugs. FDA. Available at: … Continue reading This further highlights the need for medical surveillance when taking topical finasteride and for care to be taken over doses.
What about XYON’s topical finasteride?
XYON uses 2.5% finasteride in its topical finasteride gel, and claims that its SiloxysSystem™ Gel is effective at maximizing results and minimizing the absorption of the medication into the whole body. But how accurate is this?
To start with, a 2.5% topical finasteride is substantially higher than any levels assessed in clinical trials and is higher than almost all other haircare brands. In general, a 0.25% formulation is used (and in some cases is considered high itself). XYON’s product is 10X stronger than this.
The non-linear dose-response curve of finasteride means that at the higher concentrations, increasing finasteride further results in negligible differences in DHT suppression and hair growth effects. The body is exposed to higher concentrations of finasteride, potentially resulting in an increased risk of side effects – and all of this with no further beneficial effects on hair growth.
XYON does not comment on the use of substantially higher levels of finasteride in their product than in any human trials. However, they do use their patented SiloxySystem™ Gel delivery system to make large claims about their ability to minimize finasteride exposure to the rest of the body – “92% less finasteride absorbed into the blood stream vs oral finasteride” and “57% less finasteride in the body after 24 hours vs other topical formulations”. This low absorption could be the reason behind the high finasteride concentration. So, what is the problem?
Firstly, due to the proprietary nature of the SiloxySystem™ Gel, there is very little information on exactly what ingredients form the gel and how it works. This means that we are unable to substantiate any of their claims or assess whether the product could work, based on their suggested mechanisms.
Secondly, despite the experiments that they have described on their website, none of their work is officially published in peer-reviewed journals.[11]XYON, (no date), Xyon Health Clinical Studies: SiloxysSystemTM Gel. Available at: https://xyonhealth.com/blogs/library/xyon-health-clinical-studies-siloxyssystem-gel (Accessed: 12 February 2026) Publishing results in these requires the work to be intensely scrutinized by unrelated scientific experts to ensure that the work is unbiased, accurate, and has complied with scientific standards. Without this, we don’t have enough information to be able to say whether XYON’s claims are in any way accurate.
Thirdly, XYON has included in their summarized findings that their SiloxysSystem™ Gel does not completely prevent finasteride from entering the rest of the body. As such, the extremely high concentration of finasteride in the gel may enter the bloodstream and cause DHT reduction in other areas of the body, potentially resulting in side effects. They report no side effects in their study, but with a sample size of only 7 participants, very little can be taken from this fact.
In summary, XYON’s topical finasteride contains a very high concentration of finasteride, and while the effects of this may be attenuated using their SiloxysSystem™ Gel delivery system, this has not been validated outside of XYON, making the use of such a high dose potentially risky.
Topical Finasteride and Minoxidil with SiloxysSystem™ Gel
Ingredients: 2.5% finasteride, 3% minoxidil, SiloxysSystem™ Gel
XYON offers its topical finasteride in a combination solution with minoxidil. They describe this product as containing finasteride to help regrow hair and stop hair loss, and minoxidil to stimulate and support continued hair growth.
Minoxidil is another of the original FDA-approved hair loss solutions, licenced for topical use in 1988. Its mechanism of action is less obvious than finasteride, and is thought to involve a combination of enhancing blood circulation around the hair follicles to create optimal conditions for hair growth, inducing hair follicles in the resting phase (telogen) to enter the growth phase (anagen) of the hair cycle, and increasing the production of prostaglandin E2 which promotes hair growth through providing cytoprotective effects and modulating inflammation.[12]Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and Its Use in Hair Disorders: A Review. Drug Design, Development and Therapy. 13. 2777–2786. Available at: … Continue reading,[13]Patel, P., Nessel, T.A., Kumar, D.D. (2023). Minoxidil. StatPearls. Available at: http://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: 04 February 2026),[14]Michelet, J.F., Commo, S., Billoni, N., Mahé, Y.F., Bernard, B.A. (1997). Activation of Cytoprotective Prostaglandin Synthase-1 by Minoxidil as a Possible Explanation for Its Hair Growth-Stimulating … Continue reading
As an FDA-approved drug, minoxidil has a wealth of clinical evidence supporting its use. Studies have shown that pproximately 60% of men experience a slowing, stopping, or reversal of hair loss within 3-6 months of beginning treatment.[15]Asilian, A., Farmani, A., Saber, M. (2023). Clinical Efficacy and Safety of Low-Dose Oral Minoxidil Versus Topical Solution in the Improvement of Androgenetic Alopecia: A Randomized Controlled Trial. … Continue reading Increasing treatment time beyond 12 months does not normally result in continued improvements.[16]Rietschel, R.L., Duncan, S.H. (1987). Safety and Efficacy of Topical Minoxidil in the Management of Androgenetic Alopecia. Journal of the American Academy of Dermatology. 16(3). 677–685. Available … Continue reading
The majority of clinical trials using minoxidil show measurable regrowth after 4-12 months of use.[17]Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the … Continue reading,[18]Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta … Continue reading[19]Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study … Continue reading The exact level of regrowth varies; however, estimates of regrowth are approximately 50-70% over 12 months.
The combination of topical finasteride and minoxidil is a popular one. They are thought to function via two separate mechanisms, meaning that a combination of the two offers the ability to protect against hair loss from two different angles.
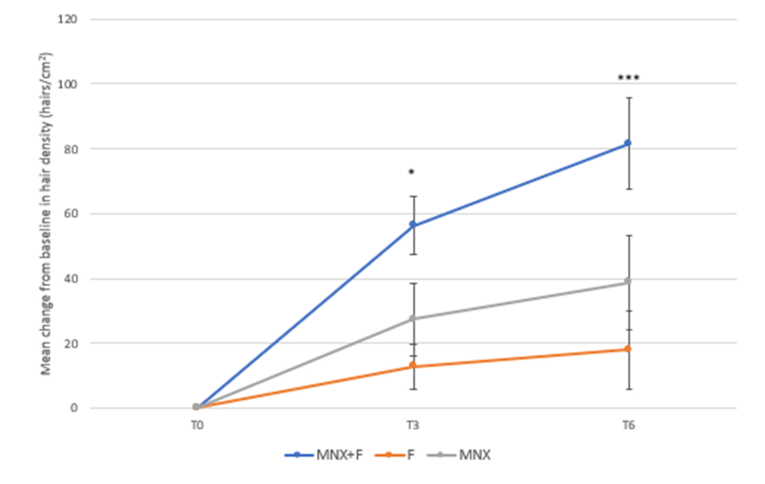
Clinical trials have demonstrated that the effectiveness of the combined therapy is higher than with minoxidil or finasteride alone.[20]Suchonwanit, P., Srisuwanwattana, P., Chalermroj, N., Khunkhet, S. (2018). A Randomized, Double-Blind Controlled Study of the Efficacy and Safety of Topical Solution of 0.25% Finasteride Admixed with … Continue reading,[21]Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, … Continue reading[22]Bharadwaj, A.V., Mendiratta, V., Rehan, H.S., Tripathi, S. (2023). Comparative Efficacy of Topical Finasteride (0.25%) in Combination with Minoxidil (5%) Against 5% Minoxidil or 0.25% Finasteride … Continue reading
Figure 5. The combination of minoxidil and finasteride (MNX+F; blue line) shows improved hair density compared to both finasteride (F; orange line) and minoxidil (MNX; gray line) after 3 months and 6 months of treatment. Adapted from Figure 2.[23]Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, … Continue reading Image used under Creative Commons license.
Are there any issues with XYON’s finasteride/minoxidil combination?
Similarly to their topical finasteride gel, XYON uses a very high concentration of finasteride in their combination gel (2.5%). As discussed above, this has the potential to increase the risk of side effects due to a greater risk of DHT suppression throughout the body. Their SiloxysSystem™ Gel is again used to minimize this absorption of the drugs systemically; however, we still lack any validated evidence supporting their claims about the gel.
Despite their using a high concentration of finasteride (presumably to counteract the slow, controlled release by their SiloxysSystem™ Gel), they actually use a lower concentration of minoxidil than the FDA recommends. Typically, 5% minoxidil is used. At this concentration, minoxidil does not require a prescription and can be bought over the counter. XYON’s product only contains 3%. While no clinical trials directly compare the efficacy of 5% versus 3% minoxidil, 5% minoxidil has clearly shown an increased effect compared to 2% solutions, whereas 2% and 3% solutions have a broadly similar effect.[24]Olsen, E.A., Dunlap, F.E., Funicella, T., et al. (2002). A Randomized Clinical Trial of 5% Topical Minoxidil Versus 2% Topical Minoxidil and Placebo in the Treatment of Androgenetic Alopecia in Men. … Continue reading,[25]Kreindler, T.G. (1987). Topical Minoxidil in Early Androgenetic Alopecia. Journal of the American Academy of Dermatology. 16(3 Pt 2). 718-724. Available at: … Continue reading
Solutions of 3% minoxidil are often offered by haircare brands as an alternative to 5% if individuals are experiencing side effects, such as scalp irritation or hypertrichosis (excessive hair growth in atypical areas). 3% minoxidil is also more commonly used when in a combination product, such as XYON is providing. However, this does not explain why in their finasteride and minoxidil gel, finasteride concentrations are boosted, but minoxidil concentrations are decreased.
Topical Finasteride and Minoxidil Solution
Ingredients: 0.25% finasteride, 6% minoxidil
In addition to offering the combination of finasteride and minoxidil with their proprietary SiloxysSystem™ Gel delivery system, XYON also offers finasteride and minoxidil in a liquid topical solution. They describe this product as a “lower-dose topical solution”.
What’s the problem?
While this solution provides a lower dose of finasteride than their (very high) gel offering, 0.25% finasteride is still considered to be a relatively high dose. Haircare brands that provide more personalized options often offer low-dose topicals of below 0.25% to those who are worried about or experiencing side effects ot higher concentrations of finasteride.
The shape of the finasteride response curve means that lower doses still have a significant effect on reducing DHT. For example, one study in patients using a 0.005% finasteride solution still saw hair regrowth effects.[26]Mazzarella, G., Loconsole, G., Cammisa, G., Mastrolonardo, G., Vena, G. (1997). Topical Finasteride in the Treatment of Androgenic Alopecia. Preliminary Evaluations After a 16-Month Therapy Course. … Continue reading As such, while XYON’s 0.25% finasteride solution is a lower dose than their 2.5% finasteride gel, it should not be considered a low dose.
In addition, the minoxidil in this solution is substantially higher than in their gel, at 6% as opposed to 3%. As discussed above, haircare brands often decrease the concentrations of topicals when used in combination, so it is unclear why they have increased the concentration in this solution. This concentration is higher than the standard, FDA-approved over-the-counter dose of 5%.
While some clinical data suggest that increasing the minoxidil dose can improve results for a subset of people, there are diminishing returns once higher concentrations are reached. Side effects are thought to be more common at higher concentrations without a clear added benefit in the majority of people.[27]Singh, S., Patil, A., Kianfar, N., Waśkiel-Burnat, A., Rudnicka, L., Sinclair, R., & Goldust, M. (2022). Does topical minoxidil at concentrations higher than 5% provide additional clinical … Continue reading, [28]Ghonemy, S., Alarawi, A., & Bessar, H. (2021). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a … Continue reading
This is a particular concern for women who are at a greater risk than men of experiencing minoxidil side effects, such as hypertrichosis.[29]Dawber, R.P.R., Rundegren, J. (2003). Hypertrichosis in Females Applying Minoxidil Topical Solution and in Normal Controls. Journal of the European Academy of Dermatology and Venereology. 17(3). … Continue reading In spite of this risk, XYON’s Topical Finasteride and Minoxidil Solution for Women also contains 6% minoxidil. It is unclear how the solution differs from the men’s, despite XYON’s claim that it has been clinically formulated for women.
To summarize, XYON’s lower-dose topical solution is not actually very low-dose, and the logic behind their dosing strategy (why they increase minoxidil whilst lowering finasteride) is unclear.
Minoxidil Tablets
Ingredients: minoxidil, vitamin D
While minoxidil is most commonly offered as a topical treatment, there is also evidence supporting its use in oral form. Studies have shown high response rates to oral minoxidil, including increased total hair counts.[30]Lueangarun, S., Panchaprateep, R., Tempark, T., Noppakun, N. (2015). Efficacy and Safety of Oral Minoxidil 5 mg Daily During 24-Week Treatment in Male Androgenetic Alopecia. Journal of the American … Continue reading,[31]Panchaprateep, R., Lueangarun, S. (2020). Efficacy and Safety of Oral Minoxidil 5 mg Once Daily in the Treatment of Male Patients with Androgenetic Alopecia: An Open-Label and Global Photographic … Continue reading,[32]Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., et al. (2019). Effectiveness and Safety of Low-Dose Oral Minoxidil in Male Androgenetic Alopecia. Journal of the American Academy of … Continue reading Oral minoxidil has also been shown to be effective in women. [33]Silva, M.N.e., Ramos, P.M., Silva, M.R., Silva, R.N.e., Raposo, N.R.B. (2022). Randomized Clinical Trial of Low-Dose Oral Minoxidil for the Treatment of Female Pattern Hair Loss: 0.25 mg Versus 1 mg. … Continue reading For more information on oral minoxidil, read our article here.
The addition of vitamin D to oral minoxidil does not have any direct clinical backing. Vitamin D deficiency has been implicated in some cases of hair loss, but comprehensive research on this is incomplete. In some cases of significant deficiency, vitamin D supplementation may improve hair thinning; however, this has not been tested in a controlled clinical trial.[34]Keene, S.A. (2022). Vitamin D Deficiency and Hair Loss: A Case Report and Review of the Literature for Diagnosis and Treatment. Hair Transplant Forum International. 32(4). 113-122. Available at: … Continue reading
A small study investigating oral vitamin D supplementation in conjunction with topical minoxidil found that the combined treatment was more effective than topical minoxidil alone.[35]Hassan, G.F.R., Sadoma, M.E.T., Elbatsh, M.M., Ibrahim, Z.A. (2022). Treatment with Oral Vitamin D Alone, Topical Minoxidil, or Combination of Both in Patients with Female Pattern Hair Loss: A … Continue reading While this could indicate that a supplement with vitamin D and oral minoxidil may have beneficial effects, there are no clinical trials providing direct evidence of this.
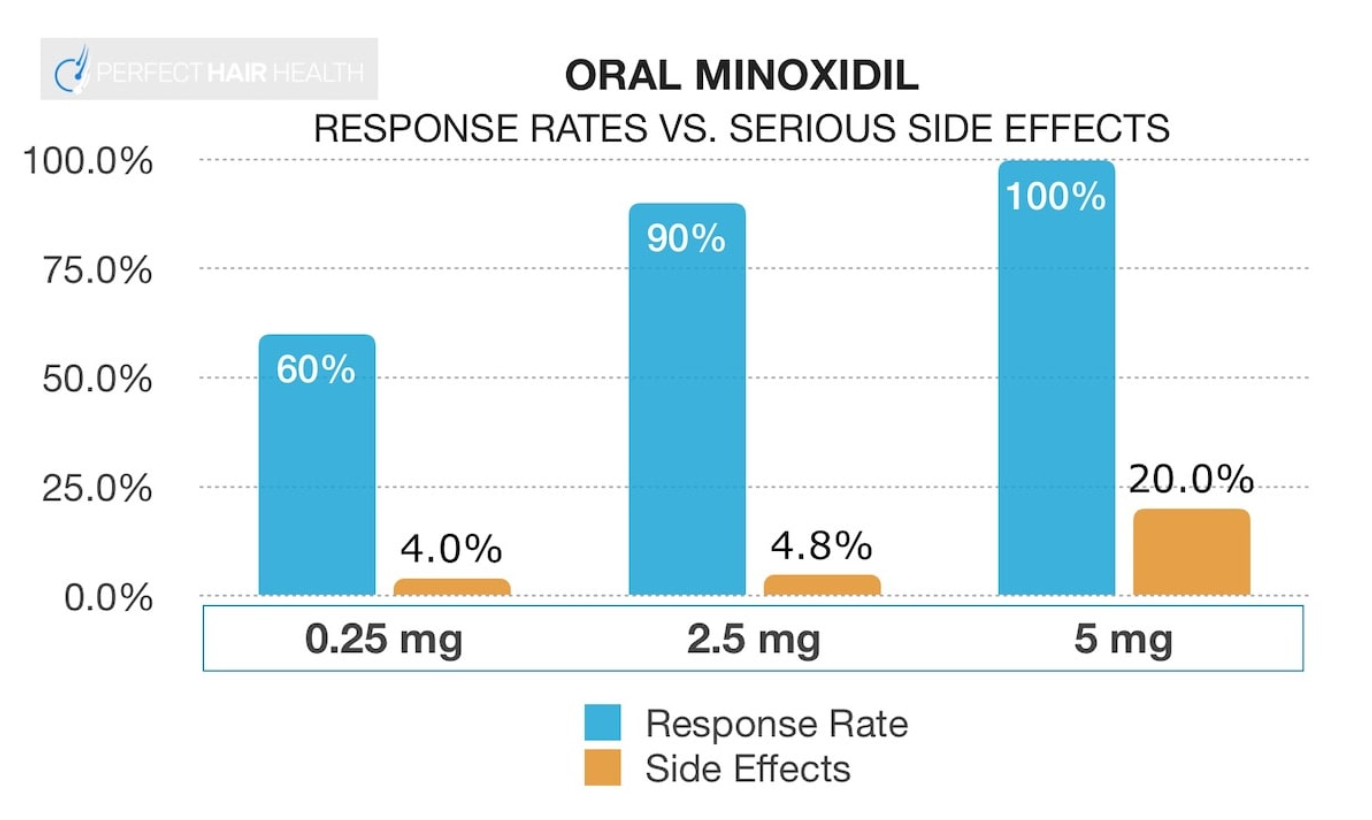
Oral minoxidil acts systemically (throughout the body), meaning that the side effects slightly differ from topical minoxidil, where its effects are largely contained to the scalp. Side effects include hypertrichosis, fluid retention, and heart palpitations. While increasing the dose of oral minoxidil increases the response rate, it also results in an increase in serious side effects.
Figure 8. Increasing the dose of oral minoxidil increases the response rate but also simultaneously increases the risk of serious side effects.
XYON offers its oral minoxidil for both men and women. They promote these as customizable in the women’s minoxidil tablets, but simply state that the minoxidil is at a lower dose in the men’s (although they do not say what this is in comparison to). Neither the webpage for the men’s nor the women’s minoxidil tablets states exactly what these doses are.
Topical Dutasteride with SiloxysSystem™ Gel
Ingredients: 2% dutasteride, SiloxysSystem™ Gel
Dutasteride is a common option for treatment escalation if treatment using finasteride or minoxidil is not working or has plateaued.
Dutasteride works via a similar mechanism to finasteride, by lowering levels of the hormone DHT. It is a more powerful 5AR inhibitor than finasteride, acting on two of the three forms of the enzyme (as opposed to finasteride, which only acts on one form), resulting in increased DHT suppression.[36]Clark, R.V., Hermann, D.J., Cunningham, G.R., Wilson, T.H., Morrill, B.B., Hobbs, S. (2004). Marked Suppression of Dihydrotestosterone in Men with Benign Prostatic Hyperplasia by Dutasteride, a Dual … Continue reading
Oral dutasteride shows increased efficacy compared to oral finasteride, with similar levels of side effects.[37]Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., Cui, Y. (2019). The Efficacy and Safety of Dutasteride Compared with Finasteride in Treating Men with Androgenetic Alopecia: A Systematic Review and … Continue reading Topical dutasteride is a more recent treatment option, and so there is less research supporting its use. However, clinical studies have shown its effectiveness, with a 0.05% solution of topical dutasteride showing significant improvement compared to 1mg oral finasteride.[38]Panuganti, V.K., Madala, P.K., Grandhi, V.R., et al. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical … Continue reading Another study showed that the combination of 0.02% topical dutasteride with microneedling showed greater improvements in hair density versus the microneedling alone group.[39]Nada, E., El Sharkawy, R., Abd El-Maged, W., Abo Elmagd, M. (2018). Topical Dutasteride with Microneedling in Treatment of Male Androgenetic Alopecia. Sohag Medical Journal. 22(1). 387-400. Available … Continue reading
There is limited research on how topical dutasteride affects serum DHT compared to oral dutasteride. One study investigating topical dutasteride reported only small changes to serum DHT levels, whereas oral finasteride saw greater changes.[40]Panuganti, V.K., Madala, P.K., Grandhi, V.R., et al. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical … Continue reading
What about XYON’s topical dutasteride?
Similarly to their topical finasteride gel, XYON uses a very high level of dutasteride in their product alongside their SiloxysSystem™ Gel, which they claim minimizes medicine absorption into the body.
Although there are relatively few studies investigating topical dutasteride, those that do exist have investigated 0.01-0.05% ranges. The XYON dose of 2% is 40X higher than the highest dose used in any clinical trial. While they do concede that this dose is higher than others on the market, they claim that their SiloxysSystem™ Gel delivers the medicine in a controlled manner to the hair follicles and limits absorption into the bloodstream. They also state that they decided on this dose with an in-house clinical trial.
While it may be true that their SiloxysSystem™ Gel has this effect, they have not published any of their results in a peer-reviewed journal, meaning that we are having to take their word for it and cannot actually check the validity of the results ourselves. In addition, although they appear to have carried out studies assessing the safety and efficacy of their SiloxysSystem™ Gel using finasteride, their study page does not contain any mention of dutasteride.[41]XYON, (no date), Xyon Health Clinical Studies: SiloxysSystemTM Gel. Available at: https://xyonhealth.com/blogs/library/xyon-health-clinical-studies-siloxyssystem-gel (Accessed: 12 February 2026)
As such, although their SiloxysSystem™ Gel may be decreasing the systemic DHT reduction, at a 2% concentration, it seems likely that some DHT suppression will be occurring, increasing the risk of side effects.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Performance DHT-Blocking Shampoo
Key ingredients: Red clover extract, Saw palmetto extract, Rosemary leaf extract, Ginseng root, Ginger root, Pea sprout extract, Avocado oil
XYON advertises its shampoo as DHT-blocking, claiming that it contains a blend of powerful natural hair-thickening ingredients, but is this based on any science?
While DHT-blocking shampoos may seem like a helpful addition to your haircare routine, they may be more marketing than results. There are currently no over-the-counter shampoos that have been clinically proven to lower DHT in humans. This doesn’t mean that the ingredients are not effective, but that they are not clinically proven in this format.
The majority of studies on ingredients involve their testing in an oral or leave-on topical form – with the oral form circulating through the entire body and the topical remaining on the scalp for a prolonged period of time. Shampoo, on the other hand, remains on the scalp for 1-2 minutes and is only used 3-4 times per week – insufficient for meaningful absorption.
As such, while there is evidence that some of the ingredients found in XYON’s DHT-blocking shampoo are effective at reducing DHT, these effects are only seen in topical and oral preparations, not in a shampoo.
For more information on DHT-blocking shampoos, read our article here.
The Problems with XYON’s Approach
Limited Personalization
Although XYON offers a range of products, they are all at fixed concentrations, with minimal ability to alter doses if needed. Users are unable to switch to a lower dose if side effects present themselves, except by changing products.
For example, if a user is experiencing sexual side effects from the 2.5% finasteride and minoxidil gel, to get a lower dose, they will have to switch to the 0.25% finasteride and minoxidil solution. However, due to XYON’s limited personalization ability, this will also involve increasing the minoxidil concentration from 3% to 6%, potentially resulting in other side effects.
This failure to provide a flexible, personalizable model limits the opportunity for effective hair growth, as users are unable to seek alternatives if products are causing side effects, leading either to perseverance with treatments that are causing issues or their ceasing use of treatments altogether.
Mega-Dosed Topicals
XYON’s topical offerings are all at high or very high doses, ranging from 0.25% finasteride (at the higher end of most hair care companies’ offerings) up to 2.5% finasteride, 2% dutasteride (40X higher than the dose used in any clinical trials), and 6% minoxidil (higher than the FDA-approved level).
Topicals are often advertised as a low-risk option, where application to the scalp decreases the risk of whole-body side effects. However, these treatments often leak into the blood. This is particularly seen when high concentrations are used, resulting in an increased risk of side effects.
XYON’s high drug concentrations are justified through the use of their SiloxysSystem™ Gel, which they claim minimizes the absorption of the medication into the body and lowers the risk of side effects. While this may be true, there are only XYON’s own experiments supporting this, and no scientific papers have been thoroughly reviewed and published to back up these claims.
Lack of Ingredient Transparency
XYON fails to provide a full list of product ingredients on their website, preventing users from checking for the presence of harmful ingredients often included in prescription topicals or for any ingredients to which they are sensitive.
For example, topicals often contain propylene glycol, an alcohol that helps with ingredient penetration. This ingredient has been shown to cause skin irritation in 7% of users, worsening hair growth.[42]Patel, K., Palmer, A., & Nixon, R. (2023). Allergic contact dermatitis from propylene glycol: a case series from Australia. *Contact Dermatitis.* 89(2). 79–84. Available at: … Continue reading
While XYON has stated in a blog that their gel products do not contain propylene glycol, their finasteride and minoxidil solution does. This information can only be found by hunting through their website for ingredient information, and is thus likely only found by those who know what to look for, and not by a new user.
High Costs
XYON’s products contain ingredients with a wealth of information supporting their use for hair loss. However, the inclusion of their proprietary SiloxysSystem™ Gel in their formulations has increased the price of the products to substantially higher than many other haircare brands offering similar products.
But what are you paying for? By purchasing XYON’s SiloxysSystem™ Gel products, you are paying for well-researched ingredients (finasteride/dutasteride/minoxidil) that can be found elsewhere, in addition to stylish branding and a gel formulation that may be effective but currently lacks validation.
What Makes Ulo Different?
While mega-dosed topicals, harmful ingredients, a lack of personalization, and unsubstantiated claims are unfortunately frequently seen in the hair loss industry, we cofounded Ulo to provide a hair loss solution that solves these problems.
At Ulo, we offer flexible, personalized treatment options, providing users with the choice of oral versus topical solutions at a wide range of doses, and with the ability to customize ingredients. We source only clinically tested, standardized ingredients with strong evidence supporting hair growth, and refuse to use irritants or harmful substances (such as propylene glycol).
At Ulo, we don’t make unsubstantiated claims about our products, and we provide our customers with realistic expectations based on clinical data. In addition, a physician is always on hand to provide unrivaled personal support throughout the user’s hair growth journey, at no additional cost.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Final Remarks
XYON offers multiple products using clinically proven ingredients and a range of delivery methods, providing an accessible route to hair loss treatments that are proven to work. The main ingredients used – finasteride, dutasteride, and minoxidil – are highly researched and have demonstrated the successful slowing of hair loss and increase in hair growth in many users.
However, XYON uses high concentrations of these ingredients in its topicals, increasing the risk of side effects. While their SiloxysSystem™ Gel may reduce this risk, there is a lack of verified evidence supporting these claims. In addition, XYON has limited personalization options, resulting in users being stuck using products that may be causing side effects without the option to adjust the dose.
These issues are not unique to XYON and can be seen in many other haircare brands. Ulo offers a new approach, providing flexibility and customization alongside clinically-backed ingredients and ongoing personal support to give users the optimal hair health journey.
References[+]
References ↑1 Drake, L., Hordinsky, M., Fiedler, V., Swinehart, J., Unger, W.P., Cotterill, P.C., Thiboutot, D.M., Lowe, N., Jacobson, C., Whiting, D., Stieglitz, S., Kraus, S.J., Griffin, E.I., Weiss, D., Carrington, P., Gencheff, C., Cole, G.W., Pariser, D.M., Epstein, E.S., Tanaka, W., Dallob, A.D., Vandormael, K., Geissler, L. and Waldstreicher, J. (1999). The Effects of Finasteride on Scalp Skin and Serum Androgen Levels in Men with Androgenetic Alopecia. J Am Acad Dermatol. 41(4), pp.550-554. Available at: https://pubmed.ncbi.nlm.nih.gov/10495374/ ↑2 Finasteride Male Pattern Hair Loss Study Group. (2002). Long-Term (5-Year) Multinational Experience With Finasteride 1 mg in the Treatment of Men With Androgenetic Alopecia. Eur J Dermatol. 12:38-49. Available at: https://pubmed.ncbi.nlm.nih.gov/11809594/ ↑3 Shapiro, J., Kaufman, K.D. (2003). Use of Finasteride in the Treatment of Men With Androgenetic Alopecia (Male Pattern Hair Loss). J Investig Dermatol Symp Proc. 8(1):20-23. Available at: https://www.sciencedirect.com/science/article/pii/S0022202X15529357 ↑4 Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. Indian Journal of Dermatology, Venereology and Leprology. 83. 47. Available at: https://doi.org/10.4103/0378-6323.188652 ↑5 Choi, G.-S., Sim, W.-Y., Kang, H., Huh, C. H., Lee, Y. W., Shantakumar, S., Ho, Y.-F., et al. (2022). Long-term effectiveness and safety of dutasteride versus finasteride in patients with male androgenic alopecia in South Korea: a multicentre chart review study. Annals of Dermatology. 34(5). 349. Available at: https://doi.org/10.5021/ad.22.027 ↑6, ↑8, ↑9 Piraccini, B.M., Blume-Peytavi, U., Scarci, F., et al. (2021). Efficacy and Safety of Topical Finasteride Spray Solution for Male Androgenetic Alopecia: A Phase III, Randomized, Controlled Clinical Trial. Journal of the European Academy of Dermatology and Venereology. 36(2). 286-294. Available at: https://doi.org/10.1111/jdv.17738 ↑7 Hajheydari, Z., Akbari, J., Saeedi, M., Shokoohi, L. (2009). Comparing the Therapeutic Effects of Finasteride Gel and Tablet in Treatment of the Androgenetic Alopecia. Indian Journal of Dermatology, Venereology and Leprology. 75. 47. Available at: https://ijdvl.com/comparing-the-therapeutic-effects-of-finasteride-gel-and-tablet-in-treatment-of-the-androgenetic-alopecia/ ↑10 U.S. Food and Drug Administration. (n.d.). FDA alerts health care providers, compounders, and consumers about potential risks associated with compounded drugs. FDA. Available at: https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-consumers-potential-risks-associated-compounded (Accessed: November 2025) ↑11, ↑41 XYON, (no date), Xyon Health Clinical Studies: SiloxysSystemTM Gel. Available at: https://xyonhealth.com/blogs/library/xyon-health-clinical-studies-siloxyssystem-gel (Accessed: 12 February 2026) ↑12 Suchonwanit, P., Thammarucha, S., Leerunyakul, K. (2019). Minoxidil and Its Use in Hair Disorders: A Review. Drug Design, Development and Therapy. 13. 2777–2786. Available at: https://doi.org/10.2147/DDDT.S214907 ↑13 Patel, P., Nessel, T.A., Kumar, D.D. (2023). Minoxidil. StatPearls. Available at: http://www.ncbi.nlm.nih.gov/books/NBK482378/ (Accessed: 04 February 2026) ↑14 Michelet, J.F., Commo, S., Billoni, N., Mahé, Y.F., Bernard, B.A. (1997). Activation of Cytoprotective Prostaglandin Synthase-1 by Minoxidil as a Possible Explanation for Its Hair Growth-Stimulating Effect. Journal of Investigative Dermatology. 108(2). 205–209. Available at: https://doi.org/10.1111/1523-1747.ep12334249 ↑15 Asilian, A., Farmani, A., Saber, M. (2023). Clinical Efficacy and Safety of Low-Dose Oral Minoxidil Versus Topical Solution in the Improvement of Androgenetic Alopecia: A Randomized Controlled Trial. Journal of Cosmetic Dermatology. 23(3). 949–957. Available at: https://doi.org/10.1111/jocd.16086 ↑16 Rietschel, R.L., Duncan, S.H. (1987). Safety and Efficacy of Topical Minoxidil in the Management of Androgenetic Alopecia. Journal of the American Academy of Dermatology. 16(3). 677–685. Available at: https://doi.org/10.1016/S0190-9622(87)70087-5 ↑17 Olsen, E.A., Whiting, D., Bergfeld, W. (2007). A Multicenter, Randomized, Placebo-Controlled, Double-Blind Clinical Trial of a Novel Formulation of 5% Minoxidil Topical Foam Versus Placebo in the Treatment of Androgenetic Alopecia in Men. Journal of the American Academy of Dermatology. 57(5). 767–774. Available at: https://doi.org/10.1016/j.jaad.2007.04.012 ↑18 Hasanzadeh, H., Nasrollahi, S.A., Halavati, N., Saberi, M., Firooz, A. (2016). Efficacy and Safety of 5% Minoxidil Topical Foam in Male Pattern Hair Loss Treatment and Patient Satisfaction. Acta Dermatovenerologica Alpina, Pannonica et Adriatica. 25(3). 41–44. Available at: https://doi.org/10.15570/actaapa.2016.12 ↑19 Van Neste, D. (2020). Placebo-Controlled Dose-Effect Studies with Topical Minoxidil 2% or 5% in Male-Patterned Hair Loss Treated with Oral Finasteride Employing an Analytical and Exhaustive Study Protocol. Skin Research and Technology. 26(4). 542–557. Available at: https://doi.org/10.1111/srt.12827 ↑20 Suchonwanit, P., Srisuwanwattana, P., Chalermroj, N., Khunkhet, S. (2018). A Randomized, Double-Blind Controlled Study of the Efficacy and Safety of Topical Solution of 0.25% Finasteride Admixed with 3% Minoxidil vs 3% Minoxidil Solution in the Treatment of Male Androgenetic Alopecia. Journal of the European Academy of Dermatology and Venereology. 32(12). 2257–2263. Available at: https://doi.org/10.1111/jdv.15171 ↑21 Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, Controlled, Assessor Blinded, 3-Arm, Pilot Trial. Journal of Cosmetic Dermatology. 23(2). 502–509. Available at: https://doi.org/10.1111/jocd.15953 ↑22 Bharadwaj, A.V., Mendiratta, V., Rehan, H.S., Tripathi, S. (2023). Comparative Efficacy of Topical Finasteride (0.25%) in Combination with Minoxidil (5%) Against 5% Minoxidil or 0.25% Finasteride Alone in Male Androgenetic Alopecia: A Pilot, Randomized Open-Label Study. International Journal of Trichology. 15(2). 56–62. Available at: https://doi.org/10.4103/ijt.ijt_72_22 ↑23 Rossi, A., Caro, G. (2023). Efficacy of the Association of Topical Minoxidil and Topical Finasteride Compared to Their Use in Monotherapy in Men with Androgenetic Alopecia: A Prospective, Randomized, Controlled, Assessor Blinded, 3-Arm, Pilot Trial. Journal of Cosmetic Dermatology. 23(2). 502–509. Available at: https://doi.org/10.1111/jocd.15953 ↑24 Olsen, E.A., Dunlap, F.E., Funicella, T., et al. (2002). A Randomized Clinical Trial of 5% Topical Minoxidil Versus 2% Topical Minoxidil and Placebo in the Treatment of Androgenetic Alopecia in Men. Journal of the American Academy of Dermatology. 47(3). 377-385. Available at: https://doi.org/10.1067/mjd.2002.124088 ↑25 Kreindler, T.G. (1987). Topical Minoxidil in Early Androgenetic Alopecia. Journal of the American Academy of Dermatology. 16(3 Pt 2). 718-724. Available at: https://doi.org/10.1016/s0190-9622(87)70093-0 ↑26 Mazzarella, G., Loconsole, G., Cammisa, G., Mastrolonardo, G., Vena, G. (1997). Topical Finasteride in the Treatment of Androgenic Alopecia. Preliminary Evaluations After a 16-Month Therapy Course. Journal of Dermatological Treatment. 8(3). 189-192. Available at: https://doi.org/10.3109/09546639709160517 ↑27 Singh, S., Patil, A., Kianfar, N., Waśkiel-Burnat, A., Rudnicka, L., Sinclair, R., & Goldust, M. (2022). Does topical minoxidil at concentrations higher than 5% provide additional clinical benefit?. Clinical and Experimental Dermatology. 47(11). 1951–1955. Available at: https://doi.org/10.1111/ced.15338 ↑28 Ghonemy, S., Alarawi, A., & Bessar, H. (2021). Efficacy and safety of a new 10% topical minoxidil versus 5% topical minoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic evaluation. Journal of Dermatological Treatment. 32(2). 236–241. Available at: https://doi.org/10.1080/09546634.2019.1654070 ↑29 Dawber, R.P.R., Rundegren, J. (2003). Hypertrichosis in Females Applying Minoxidil Topical Solution and in Normal Controls. Journal of the European Academy of Dermatology and Venereology. 17(3). 271-275. Available at: https://doi.org/10.1046/j.1468-3083.2003.00621.x ↑30 Lueangarun, S., Panchaprateep, R., Tempark, T., Noppakun, N. (2015). Efficacy and Safety of Oral Minoxidil 5 mg Daily During 24-Week Treatment in Male Androgenetic Alopecia. Journal of the American Academy of Dermatology. 72(5). AB113. Available at: https://doi.org/10.1016/j.jaad.2015.02.466 ↑31 Panchaprateep, R., Lueangarun, S. (2020). Efficacy and Safety of Oral Minoxidil 5 mg Once Daily in the Treatment of Male Patients with Androgenetic Alopecia: An Open-Label and Global Photographic Assessment. Dermatology and Therapy. 10(6). 1345-1357. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC7649170/ ↑32 Jimenez-Cauhe, J., Saceda-Corralo, D., Rodrigues-Barata, R., et al. (2019). Effectiveness and Safety of Low-Dose Oral Minoxidil in Male Androgenetic Alopecia. Journal of the American Academy of Dermatology. 81(2). 648-649. Available at: https://doi.org/10.1016/j.jaad.2019.04.054 ↑33 Silva, M.N.e., Ramos, P.M., Silva, M.R., Silva, R.N.e., Raposo, N.R.B. (2022). Randomized Clinical Trial of Low-Dose Oral Minoxidil for the Treatment of Female Pattern Hair Loss: 0.25 mg Versus 1 mg. Journal of the American Academy of Dermatology. 87(2). 396-399. Available at: https://doi.org/10.1016/j.jaad.2022.01.017 ↑34 Keene, S.A. (2022). Vitamin D Deficiency and Hair Loss: A Case Report and Review of the Literature for Diagnosis and Treatment. Hair Transplant Forum International. 32(4). 113-122. Available at: https://doi.org/10.33589/32.4.113 ↑35 Hassan, G.F.R., Sadoma, M.E.T., Elbatsh, M.M., Ibrahim, Z.A. (2022). Treatment with Oral Vitamin D Alone, Topical Minoxidil, or Combination of Both in Patients with Female Pattern Hair Loss: A Comparative Clinical and Dermoscopic Study. Journal of Cosmetic Dermatology. 21(9). 3917-3924. Available at: https://doi.org/10.1111/jocd.14743 ↑36 Clark, R.V., Hermann, D.J., Cunningham, G.R., Wilson, T.H., Morrill, B.B., Hobbs, S. (2004). Marked Suppression of Dihydrotestosterone in Men with Benign Prostatic Hyperplasia by Dutasteride, a Dual 5α-Reductase Inhibitor. Journal of Clinical Endocrinology & Metabolism. 89(5). 2179-2184. Available at: https://doi.org/10.1210/jc.2003-030330 ↑37 Zhou, Z., Song, S., Gao, Z., Wu, J., Ma, J., Cui, Y. (2019). The Efficacy and Safety of Dutasteride Compared with Finasteride in Treating Men with Androgenetic Alopecia: A Systematic Review and Meta-Analysis. Clinical Interventions in Aging. 14. 399-406. Available at: https://doi.org/10.2147/CIA.S192435 ↑38, ↑40 Panuganti, V.K., Madala, P.K., Grandhi, V.R., et al. (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia. Cureus. 17(8). Available at: https://doi.org/10.7759/cureus.89309 ↑39 Nada, E., El Sharkawy, R., Abd El-Maged, W., Abo Elmagd, M. (2018). Topical Dutasteride with Microneedling in Treatment of Male Androgenetic Alopecia. Sohag Medical Journal. 22(1). 387-400. Available at: https://doi.org/10.21608/smj.2018.42083 ↑42 Patel, K., Palmer, A., & Nixon, R. (2023). Allergic contact dermatitis from propylene glycol: a case series from Australia. *Contact Dermatitis.* 89(2). 79–84. Available at: https://doi.org/10.1111/cod.14325 Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreCatherine Kennedy, PhD
Catherine is a scientist and researcher with a background in biosciences and clinical neuroscience. She completed an integrated master’s degree in Biosciences (MBiol) before earning a PhD in Clinical Neurosciences at the University of Cambridge. Throughout her career, she has contributed to the scientific literature through the publication of numerous peer-reviewed research papers.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down