- About
- Mission Statement
Education. Evidence. Regrowth.
- Education.
Prioritize knowledge. Make better choices.
- Evidence.
Sort good studies from the bad.
- Regrowth.
Get bigger hair gains.
Team MembersPhD's, resarchers, & consumer advocates.
- Rob English
Founder, researcher, & consumer advocate
- Research Team
Our team of PhD’s, researchers, & more
Editorial PolicyDiscover how we conduct our research.
ContactHave questions? Contact us.
Before-Afters- Transformation Photos
Our library of before-after photos.
- — Jenna, 31, U.S.A.
I have attached my before and afters of my progress since joining this group...
- — Tom, 30, U.K.
I’m convinced I’ve recovered to probably the hairline I had 3 years ago. Super stoked…
- — Rabih, 30’s, U.S.A.
My friends actually told me, “Your hairline improved. Your hair looks thicker...
- — RDB, 35, New York, U.S.A.
I also feel my hair has a different texture to it now…
- — Aayush, 20’s, Boston, MA
Firstly thank you for your work in this field. I am immensely grateful that...
- — Ben M., U.S.A
I just wanted to thank you for all your research, for introducing me to this method...
- — Raul, 50, Spain
To be honest I am having fun with all this and I still don’t know how much...
- — Lisa, 52, U.S.
I see a massive amount of regrowth that is all less than about 8 cm long...
Client Testimonials150+ member experiences.
Scroll Down
Popular Treatments- Treatments
Popular treatments. But do they work?
- Finasteride
- Oral
- Topical
- Dutasteride
- Oral
- Topical
- Mesotherapy
- Minoxidil
- Oral
- Topical
- Ketoconazole
- Shampoo
- Topical
- Low-Level Laser Therapy
- Therapy
- Microneedling
- Therapy
- Platelet-Rich Plasma Therapy (PRP)
- Therapy
- Scalp Massages
- Therapy
More
IngredientsTop-selling ingredients, quantified.
- Saw Palmetto
- Redensyl
- Melatonin
- Caffeine
- Biotin
- Rosemary Oil
- Lilac Stem Cells
- Hydrolyzed Wheat Protein
- Sodium Lauryl Sulfate
More
ProductsThe truth about hair loss "best sellers".
- Minoxidil Tablets
Xyon Health
- Finasteride
Strut Health
- Hair Growth Supplements
Happy Head
- REVITA Tablets for Hair Growth Support
DS Laboratories
- FoliGROWTH Ultimate Hair Neutraceutical
Advanced Trichology
- Enhance Hair Density Serum
Fully Vital
- Topical Finasteride and Minoxidil
Xyon Health
- HairOmega Foaming Hair Growth Serum
DrFormulas
- Bio-Cleansing Shampoo
Revivogen MD
more
Key MetricsStandardized rubrics to evaluate all treatments.
- Evidence Quality
Is this treatment well studied?
- Regrowth Potential
How much regrowth can you expect?
- Long-Term Viability
Is this treatment safe & sustainable?
Free Research- Free Resources
Apps, tools, guides, freebies, & more.
- Free CalculatorTopical Finasteride Calculator
- Free Interactive GuideInteractive Guide: What Causes Hair Loss?
- Free ResourceFree Guide: Standardized Scalp Massages
- Free Course7-Day Hair Loss Email Course
- Free DatabaseIngredients Database
- Free Interactive GuideInteractive Guide: Hair Loss Disorders
- Free DatabaseTreatment Guides
- Free Lab TestsProduct Lab Tests: Purity & Potency
- Free Video & Write-upEvidence Quality Masterclass
- Free Interactive GuideDermatology Appointment Guide
More
Articles100+ free articles.
-
Ulo Review: #1 Customized Hair Loss Treatment in 2026
-
Does Anavar Cause Hair Loss?
-
10 Best Shampoos for Hair Loss in 2026
-
XYON Review: Do Their Products Actually Regrow Hair?
-
Minoxidil Before and After Photos [2026] | Does It Work?
-
How to Get Finasteride: Is It Over the Counter?
-
Keeps Review: The Truth About Their Hair Loss Treatments?
-
7 Best Oils for Hair Growth
PublicationsOur team’s peer-reviewed studies.
- Microneedling and Its Use in Hair Loss Disorders: A Systematic Review
- Use of Botulinum Toxin for Androgenic Alopecia: A Systematic Review
- Conflicting Reports Regarding the Histopathological Features of Androgenic Alopecia
- Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results
- A Hypothetical Pathogenesis Model For Androgenic Alopecia:Clarifying The Dihydrotestosterone Paradox And Rate-Limiting Recovery Factors
Menu- AboutAbout
- Mission Statement
Education. Evidence. Regrowth.
- Team Members
PhD's, resarchers, & consumer advocates.
- Editorial Policy
Discover how we conduct our research.
- Contact
Have questions? Contact us.
- Before-Afters
Before-Afters- Transformation Photos
Our library of before-after photos.
- Client Testimonials
Read the experiences of members
Before-Afters/ Client Testimonials- Popular Treatments
-
ArticlesUlo Review: #1 Customized Hair Loss Treatment in 2026
First Published Apr 10 2026Last Updated Apr 11 2026PharmaceuticalResearched & Written By:Michael Williams, PhDReviewed By:Catherine Kennedy, PhDWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreArticle Summary
Ulo, co-founded by Perfect Hair Health, was created to address long-standing issues in the hair loss industry, from unsafe formulations to misleading personalization claims. In this review, we examine whether Ulo’s evidence-first, highly customizable approach truly sets a new benchmark for hair loss treatment in 2026.
Full Article
The range of treatments available for hair loss can seem bewildering. Over-the-counter options range from clinically proven pharmaceuticals to ineffective fads and overpriced shampoos. Prescription options can provide robust results for many, but come with a long list of potential side effects, drawbacks, and tradeoffs.
We started Perfect Hair Health to provide clarity and evidence-based information in a space that is too often characterized by exaggerated claims and misinformation. Unfortunately, the more we learned about the hair loss industry, the more stark the reality of the problems became. While telehealth companies have made effective treatments more accessible than ever, too often brands rely on poorly formulated pharmaceuticals that present unnecessary risks to users and ignore clinical data.
We co-founded Ulo in response to these issues to provide evidence-backed hair loss treatments that can be truly customized to work for you. In this article, we’ll take a deeper look at the issues in the industry and how Ulo does things differently. We’ll review the clinical evidence supporting Ulo’s choice of pharmaceuticals and ingredients, and see how its approach translates into real-world outcomes for safety, tolerability, and hair regrowth.
Interested in Topical Finasteride?
Low-dose & full-strength finasteride available, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

What is Ulo?
After many years of consumer advocacy and watching hair loss brands provide suboptimal service and promote ineffective treatments, we co-founded Ulo.
Ulo is a telehealth provider focused on hair loss treatments. It’s committed to evidence-based treatments, real personalization, and customer safety. At its core, Ulo is built around three principles: evidence, personalization, and consumer safety. These principles are applied to ingredient selection, dosing strategies, and formulation design.
Problems in the Hair Loss Industry
There is a range of options available for hair loss that can produce real results. Best-selling pharmaceuticals like minoxidil and finasteride are backed by many years of clinical research, as well as the personal experiences of millions of users. So do you just need to pick the treatment that offers the most powerful regrowth?
Unfortunately, it’s not so simple. Finding the best treatment for you depends on finding the right dose and the most appropriate delivery method, as well as understanding potential side effects and whether combinations of treatments might optimize your results. That’s why customization is so essential for producing the best results.
However, instead of providing clear routes to effective treatments, telehealth companies sometimes compound these issues by introducing a whole new set of problems.
Problem #1: Unproven Treatments and Ingredients
The hair loss telehealth industry is a crowded one, and it’s sometimes hard to stand out. Because of this, many brands turn to unique ingredients and trends to attract new customers. Unfortunately, many of these treatments are not backed up by robust evidence.
It’s important to differentiate between strong evidence, which typically comes from multiple clinical trials conducted in humans, and weaker, limited evidence. Many drugs, compounds, and natural ingredients show some promise in petri dishes in the lab. Many even appear to regrow hair in animal models, typically mice. But this rarely translates directly into measurable outcomes in real people.
Unfortunately, some brands use this limited evidence as a basis to include these ingredients in treatments, even using them as unique selling points. If you’ve been watching progress in the hair loss industry for as long as we have, you’ll have seen these trends come and go.
Today, it’s peptides. Peptides have exploded in popularity and are touted as the solution to a wide range of health issues. You’re most likely to come across copper peptides in lists of ingredients for over-the-counter hair loss products, as well as prescription formulations.
One copper peptide, called AHK-Cu, has been shown to enhance the growth of human hair follicle cells in the lab, while another, GHK-Cu, has been shown to have anti-inflammatory properties.[1]Gruchlik, A., Jurzak, M., Chodurek, E. W. A., & Dzierzewicz, Z. (2012). Effect of Gly-Gly-His, Gly-His-Lys and their copper complexes on TNF-alpha-dependent IL-6 secretion in normal human dermal … Continue reading These experiments on cells in a petri dish are an important step in finding new drugs and understanding the molecular mechanisms behind hair loss. Unfortunately, they don’t tell us much about what will happen in actual humans.
For that, we need clinical data, preferably from placebo-controlled clinical trials. The only clinical trial we’re aware of that tested copper peptides in humans found no statistical difference in hair length or thickness after 6 months of treatment with GHK-Cu.[2]Lee, W. J., Sim, H. B., Jang, Y. H., Lee, S. J., Kim, D. W., & Yim, S. H. (2016). Efficacy of a complex of 5-aminolevulinic acid and glycyl-histidyl-lysine peptide on hair growth. *Annals of … Continue reading
That is not to say that copper peptides might not be effective at the right dose for the right person. But without clinical evidence, there’s no reason for you to expect improved outcomes, or for telehealth and hair loss brands to market them for hair loss. Copper peptides are only one example of a vast range of unproven ingredients that are regularly used in expensive hair loss treatments.
You can read more about understanding evidence in the hair loss industry in our article.
Problem #2: Irritants and Potentially Dangerous Ingredients
Ineffective ingredients can be an expensive, but ultimately harmless, addition to formulations. But what if ingredients went beyond useless, and were actually actively harmful? Unfortunately, that is potentially the case in some treatments currently on the market.
Propylene Glycol
On the low end of the scale are irritants, like propylene glycol. Propylene glycol is used in hair loss treatments as a carrier, meaning it’s a vehicle and helper for the active ingredients. Propylene glycol helps moisturize the skin and has been shown to improve the penetration of drugs into the skin in some models.[3]Carrer, V., Alonso, C., Pont, M., Zanuy, M., Córdoba, M., Espinosa, S., Barba, C., Oliver, M. A., Martí, M., & Coderch, L. (2020). Effect of propylene glycol on the skin penetration of drugs. … Continue reading,[4]Lessmann, H., Schnuch, A., Geier, J., Uter, W. (2005). Skin-sensitizing and irritant properties of propylene glycol. Contact Dermatitis. 53(5). 247-259. Available at: … Continue reading
Unfortunately, it’s also a known irritant and can cause contact dermatitis in a small proportion of people.[5]Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelb). 13(5). 1157-1169. Available at: … Continue reading. This might be a fair trade-off if it were an essential ingredient. However, it’s perfectly possible to produce effective topicals for hair loss without propylene glycol.
Ingredients like propylene glycol can cause inflammation and irritation, which can make hair loss worse and stop treatments from working. Some brands use additional ingredients to dampen this inflammation, rather than stopping it at the source by removing potentially inflammatory, inessential components.
Corticosteroids
Corticosteroids, for example, help lower scalp inflammation and may provide additional benefits for people with alopecia areata or scarring alopecia.[6]Alsantali, A. (2011). Alopecia areata: a new treatment plan. *Clinical, Cosmetic and Investigational Dermatology.* 4. 107–115. Available at: https://doi.org/10.2147/CCID.S22767 However, they are not designed for long-term use, and applying steroids like fluocinolone to your skin every day for a year can cause conditions like skin thinning and spider veins.[7]Torok, H. M., Jones, T., Rich, P., Smith, S., & Tschen, E. (2005). Hydroquinone 4%, tretinoin 0.05%, fluocinolone acetonide 0.01%: a safe and efficacious 12-month treatment for melasma. *Cutis.* … Continue reading What’s more, hair loss treatments are typically taken daily for many years, even decades, further increasing the likelihood of complications.
You can read more about the safety concerns behind corticosteroids in our full-length article.
Latanoprost
You might also spot hair loss formulations that include a drug called latanoprost. After initial promise in studies, latanoprost has had very mixed results from a number of clinical trials. Regardless, it was branded as new and exciting, becoming a popular treatment option.[8]Blume-Peytavi, U., Lönnfors, S., Hillmann, K., & Garcia Bartels, N. (2012). A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by … Continue reading,[9]Bloch, L. D., Escudeiro, C. C., Sarruf, F. D., & Sakai Valente, N. Y. (2018). Latanoprost and minoxidil: comparative double-blind, placebo-controlled study for the treatment of hair loss. … Continue reading
However, latanoprost was initially developed as a glaucoma treatment, and studies have shown that it can potentially change iris color in anywhere between 10% and 70% of patients.[10]Teus, M. A., Arranz-Marquez, E., & Lucea-Suescun, P. (2002). Incidence of iris colour change in latanoprost treated eyes. *British Journal of Ophthalmology.* 86(10). 1085–1088. Available at: … Continue reading This risk of permanent side effects might be reasonable for glaucoma patients, but it’s difficult to justify for those with hair loss who might not see any benefits at all.
Problem #3: Mega-Dosed Topicals
Finasteride is an effective treatment for hair loss that works by lowering the level of dihydrotestosterone (DHT) in the scalp. DHT drives changes in the follicles that lead to androgenic alopecia (AGA). However, finasteride can also lower DHT in other parts of the body, leading to side effects.[11]Zito, P. M., Bistas, K. G., Patel, P., & Syed, K. (2024). Finasteride. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: … Continue reading
Topical treatments are often presented as a way to localize drug activity to the scalp while minimizing systemic exposure. However, the extent to which drugs stay localized is heavily dependent on dosing.
Importantly, the relationship between the dose of finasteride and DHT reduction is not linear. When taken orally, even small amounts of the drug can significantly suppress serum DHT levels, reflecting systemic absorption of the drug into the blood. This means that for topical formulations applied to the scalp, even small amounts of absorption into the bloodstream can have an effect.
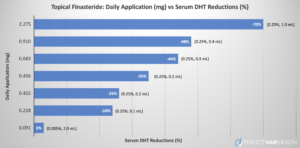
Data we’ve compiled from multiple clinical trials shows that only at doses of 0.005% is there no significant decrease in serum DHT. Despite this, many telehealth brands now offer high-strength topical finasteride formulations, often in the range of 0.25% to 0.3%, up to 60 times higher. These concentrations significantly increase the chances of side effects caused by systemic DHT reduction, while offering diminishing returns in terms of regrowth potential.
Figure 2. Data collected from multiple studies shows only application of very low (0.005%) concentrations of finasteride does not lower serum DHT levels.
If the aim of topical formulations is to reduce systemic absorption of DHT and minimize side effects, then mega-dose topicals run counter to this basic premise.
You can read more about finasteride dosing in our in-depth article.
Problem #4: No Customization
Hair loss is not a one-size-fits-all condition. The rate of hair loss, response to treatment, and incidence of side effects can vary significantly from person to person. Despite this, many telehealth platforms rely on highly standardized formulations.
This is a major issue when users experience side effects. The absence of options to decrease doses or look for alternatives can lead people to struggle through their treatment, or to abandon it entirely.
Conversely, if you find that your hair growth has plateaued or hair loss is increasing, there is no route to increase dosing or add additional treatments to your plan.
Problem #5 Improper Dutasteride Compounding
Dutasteride is a highly effective drug for reducing DHT, but its effectiveness depends heavily on its formulation. Unlike finasteride or minoxidil, dutasteride is poorly soluble in water and requires a lipid-based delivery system to be properly absorbed. That’s why it’s typically formulated as a soft-gel capsule
However, some providers are now compounding dutasteride into powder form in order to combine it with other ingredients, such as minoxidil or vitamins, in a single capsule. This can then be marketed as a more convenient, all-in-one solution.
Unfortunately, this may significantly reduce the drug’s bioavailability. To test this, we performed a small test. One participant took 3 pills of 0.5 mg powdered dutasteride (i.e., 1.5 mg total) and then checked changes in their blood DHT levels 12 hours later. Based on pharmacological data, we’d expect to see around an 80-90% reduction in DHT. However, we saw a reduction of only 13.6%.
Compounding of dutasteride without proper emulsifiers may be leading to lower DHT reduction, and therefore worse outcomes for hair regrowth.
Interested in Oral Dutasteride?
Oral Dutasteride Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

How Does Ulo Solve These Problems?
Rather than searching for the newest trends and gimmicks, Ulo only uses pharmaceuticals and treatments backed by robust clinical evidence. This means clinical trials performed in humans, not only preliminary data from cell-based models or animal studies.
When it comes to incorporating these clinically validated ingredients into effective treatments, Ulo prioritizes customizability rather than relying on high-strength, one-size-fits-all topical formulations. Ulo offers both low-dose and full-strength versions of topical finasteride and dutasteride, with optional additions such as higher-strength minoxidil, tretinoin, caffeine, melatonin, or cetirizine, depending on individual needs and tolerability.
What Does Ulo Offer?
Ulo offers a range of proven hair loss treatments at a range of concentrations. Here, we’ll explore the catalog in detail and look at some of the evidence supporting it.
Finasteride
As we’ve noted above, finasteride is backed by many years of clinical evidence and the experience of hundreds of thousands of users across the globe. [12]Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride … Continue reading Oral finasteride, typically taken as a 1 mg tablet daily, is one of only two FDA-approved treatments for hair loss. Ulo offers oral finasteride at the FDA-approved concentration of 1mg.
As we’ve already discussed, androgenic alopecia (AGA) can be driven by increases in DHT. Finasteride works by blocking an enzyme called 5ɑ-reductase (5AR), which converts testosterone into DHT. Therefore, by blocking this conversion, the drug can decrease DHT in the scalp, and therefore inhibit its effects on hair.[13]Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/
However, there are some downsides to oral finasteride. Because taking oral finasteride decreases DHT throughout the body, it can cause secondary side effects. This inlcludes decreased libido, erectile dysfunction, and ejaculation disorders, which affect a small proportion of users. [14]Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic … Continue reading, [15]Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has … Continue reading
Due to these side effects, topical formulations of finasteride have been developed to localize the drug to the scalp. As we’ve already seen, how localized the drug stays depends on the dose and volume of finasteride applied. Ulo offers finasteride at a very low dose of 0.005% and at 0.2%.
Very low concentrations are designed to minimize systemic exposure as much as possible. One small clinical study showed that 0.005% topical finasteride found no significant changes in serum DHT levels, suggesting minimal systemic absorption. Despite this, participants still experienced meaningful hair growth.[16]Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia: preliminary evaluations after a … Continue reading
In line with Ulo’s commitment to customization, a higher dose is available for those looking for a more powerful solution for growth. Finding the right balance of strength vs side effects is tricky. One study showed that 0.1% topical finasteride showed comparable efficacy to 1 mg oral finasteride, while a more recent trial found that 0.25% topical significantly improved target area hair count compared to placebo.[17]Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of … Continue reading
Increasing topical concentrations above 0.2% appears to provide diminishing returns for hair growth while increasing the risk of side effects. As such, this is the highest concentration available from Ulo.
Importantly, Ulo’s topical formulations are fully customizable, including the addition of growth stimulants and other add-ons to help you better find the right combination for you.
Minoxidil
Minoxidil is one such growth stimulant, which can work alone or in combination with treatments. Topical minoxidil is an FDA-approved treatment for hair loss at 5% concentrations, and it works through different mechanisms to finasteride. It alters the hair cycle, promoting entry of hair follicles into the growth phase, while shortening the resting phase. It can also increase blood flow around follicles and activate pro-growth molecular signaling pathways.[18]Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: … Continue reading
Multiple clinical trials have demonstrated the efficacy of minoxidil for increasing hair growth. [19]Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189 Because minoxidil works via different mechanisms, using the drug in combination with DHT-blockers like finasteride can promote growth by multiple routes.
Ulo offers minoxidil at 7%, higher than the over-the-counter concentration of 5%. Topical minoxidil is typically safe, with the most common side effects including scalp irritation and itching, while some users find the oily appearance of hair after use unappealing, particularly in longer hair. Many incidences of itching and irritation are likely due to the inclusion of propylene glycol, which is entirely absent from all of Ulo’s formulations.
Because the risk of adverse effects is relatively low, this higher concentration can improve results without incurring significant risk. Perfect hair health members have consistently reported strong results using 7% minoxidil, particularly when combined with enhancers like tretinoin that can help the drug to penetrate into the skin.
DHT-blockers like finasteride are not typically recommended for women who may become pregnant, due to significant danger during pregnancy. As such, minoxidil may be a better solution for pre-menopausal women, or anyone concerned about side effects.
Ulo also offers oral minoxidil in a 2.5mg tablet. This may be a good solution for users who struggle with the appearance of their hair after using topical formulations, or who find it hard to maintain consistent, daily application. Treating hair loss is a long game, and often the best solution is one that you can apply consistently every day over many years.
Oral minoxidil does come with some increased risk of side effects that users should be aware of. These include hypertrichosis (excessive hair growth outside of the scalp), mild fluid retention or ankle swelling, and lightheadedness. Cardiac issues are a more serious, but less commonly reported, issue.[20]Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair … Continue reading
Again, customization is key. Minoxidil can be effective on its own, or in combination with other therapies. Ulo offers a range of options to suit your needs and priorities.
Dutasteride
Like finasteride, dutasteride lowers levels of DHT by blocking 5AR. However, dutasteride blocks two forms of the enzyme, while finasteride only blocks one, which can lead to even greater DHT reduction. Clinical evidence consistently supports this, and across multiple studies, 0.5 mg oral dutasteride has been shown to outperform 1 mg finasteride in increasing total hair count, improving hair thickness, and reversing follicle miniaturization.[21]Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled … Continue reading
However, this mechanism of action can also result in side effects, as we saw with finasteride. For this reason, topical formulations were developed. A 2025 randomized controlled trial comparing topical dutasteride (0.01-0.05%) to oral finasteride reported greater improvements in hair density and global photographic assessments in the topical dutasteride group.[22]Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled … Continue reading
Ulo offers dutasteride at concentrations of 0.02% to 0.2%, again allowing users to balance tolerability with the strength of treatment.
Interested in Topical Dutasteride?
Hair gains bigger than finasteride? Dutasteride makes this possible, if prescribed*
Take the next step in your hair regrowth journey. Get started today with a provider who can prescribe a topical solution tailored for you.
*Only available in the U.S. Prescriptions not guaranteed. Restrictions apply. Off-label products are not endorsed by the FDA.

Tretinoin (Retinoic Acid)
Perfect Hair Health’s many years of consumer advocacy and the insights from our community have highlighted the importance of add-ons to well-established pharmaceutical options.
Tretonoin is a derivative of retinoic acid and has been shown to have a modest impact on hair growth when used alone.[23]Bazzano, G. S., Terezakis, N., & Galen, W. (1986). Topical tretinoin for hair growth promotion. *Journal of the American Academy of Dermatology.* 15(4). 880–893. Available at: … Continue reading However, it’s most effective when used alongside minoxidil.
For minoxidil to work, it first has to be activated in the scalp by an enzyme called SULT1A1. As a result, the efficacy of minoxidil is highly dependent on levels of SULT1A1. Tretinoin has been shown to increase SULT1A1 in the outer root sheath, enhancing the local activation of minoxidil. [24]Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvin, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin enhances minoxidil response in … Continue reading
Ulo allows you to add tretinoin to your formulations to optimize their efficacy and drive better results, and may be particularly beneficial for patients who have responded poorly to minoxidil or have low levels of SULT1A1.
Melatonin
Another available add-on is melatonin. While it doesn’t provide the same kind of results as pharmaceutical options, topical melatonin has been shown to reduce rates of active hair shedding and, in some cases, improve hair density and counts, particularly in individuals with early-stage AGA.[25]Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., & Elsner, P. (2012). Topical melatonin for treatment of androgenetic alopecia. *International Journal of Trichology.* 4(4). 236–245. … Continue reading
Ulo offers 0.01% melatonin to add to topical hair loss formulations. Melatonin alone is unlikely to drive significant results, but as an adjunct to drugs like finasteride and minoxidil, it can help reduce shedding and is recommended for those with early signs of AGA.
Caffeine
Caffeine is commonly found in shampoos and conditioners, though it’s unlikely to have any significant impact on hair growth in these wash-off treatments. There is some evidence to suggest that topical solutions of 0.2% can help boost growth, though caffeine hasn’t been as thoroughly investigated as the other ingredients we see here.[26]Dhurat, R., Chitallia, J., May, T. W., Jayaraaman, A. M., Madhukara, J., Anandan, S., Vaidya, P., & Klenk, A. (2018). An open-label randomized multicenter study assessing the noninferiority of a … Continue reading
Instead of acting as the primary active ingredient, caffeine is also best used as an add-on. Clinical evidence suggests that 5% minoxidil with 1% caffeine and 1.5% azelaic acid can be more effective than minoxidil alone.[27]Pazoki-Toroudi, H., Moghadam, R. M., Ajami, M., Nassiri-Kashani, M., Ehsani, A., Tabatabaie, H., Khani, S., Abbasi, Z., Shizarpour, M., Firooz, A., & Dowlati, Y. (2013). The efficacy and safety … Continue reading
Caffeine, therefore, is probably best understood as a relatively low-risk addition that may provide modest benefits when used alongside better-supported therapies. For this reason, Ulo offers 0.2% caffeine as an add-on to prescription therapies.
Cetirizine
Cetirizine is an antihistamine best known as a treatment for allergies. There is, however, encouraging clinical evidence suggesting it can help treat hair loss. In one placebo-controlled trial, 43.3% of participants using topical cetirizine showed hair regrowth, compared to none in the placebo group. In another trial, cetirizine improved hair density, although minoxidil performed better overall.[[Zaky, M. S., Abo Khodeir, H., Ahmed, H. A., & Elsaie, M. L. (2021). Therapeutic implications of topical cetirizine 1% in treatment of male androgenetic alopecia: a case‐controlled study. Journal of Cosmetic Dermatology. 20(4). 1154-1159. Available at: https://doi.org/10.1111/jocd.13940}}
It’s unclear whether cetirizine will be effective as a stand-alone treatment. Following this evidence, Ulo offers it as an add-on to better evidenced treatments to take advantage of its hair growth potential. What’s more, because it’s so widely used in allergy medications, the safety profile of cetirizine is very well understood.
Is Ulo the #1 Customized Hair Loss Treatment in 2026?
There is a range of telehealth brands that offer great solutions for many people. Some companies have made amazing progress in improving access to hair loss treatments, offering combinations of oral and topical therapies, simplified prescription processes, and greater flexibility in delivery.
However, these solutions are often still built around standardized formulations and rigid ingredient lists. For many people, these platforms can still deliver solid results, particularly for those who respond well to first-line therapies and don’t require fine-tuning.
Ulo builds on the strengths of these platforms, including accessibility, convenience, and clinically validated treatments, but expands them by offering genuine flexibility in treatment design. Users can choose between oral and topical treatments, low-dose and full-strength formulations, and can add evidence-supported adjuncts like tretinoin, melatonin, caffeine, or cetirizine depending on their goals, tolerability, and stage of hair loss.
Some users need to start cautiously to minimize side effects, while others need stronger interventions or add-ons to push beyond a plateau. Ulo’s range of oral and topical finasteride and dutasteride, higher-strength minoxidil, and customizable adjuncts allows treatment to be adjusted around the individual.
Ulo distinguishes itself by combining a foundation of evidence-based therapies with customization, formulation control, and evidence-based decision-making that are still largely absent elsewhere.
Looking to the Future of Ulo
Ulo’s approach is not limited to its current product line. A core part of its long-term strategy is continued investment in research and development, with the goal of improving both the effectiveness and safety of hair loss treatments over time. This includes work on new proprietary base formulations designed to enhance localization, helping active ingredients remain targeted to the scalp while minimizing systemic exposure.
Future treatments also aim to provide more options for individuals with sensitivities, as well as a broader range of concentrations, formulations, and ingredient combinations tailored to different stages of hair loss and treatment goals.
Final Thoughts
At Perfect Hair Health, our goal has never been to criticize the industry without cause. However, we continue to highlight issues because, as they persist, they continue to provide a less effective, less safe treatment space for consumers.
Ultimately, we would prefer a world where these issues no longer exist, where treatments are consistently evidence-based, properly formulated, and tailored to the individual. That would be better for consumers, and it would mean there’s less need to constantly question the quality of what’s being offered.
Ulo is our attempt to move in that direction. It reflects the standards we’ve been advocating for over the past decade: better formulations, better dosing strategies, and a more thoughtful approach to customization.
References[+]
References ↑1 Gruchlik, A., Jurzak, M., Chodurek, E. W. A., & Dzierzewicz, Z. (2012). Effect of Gly-Gly-His, Gly-His-Lys and their copper complexes on TNF-alpha-dependent IL-6 secretion in normal human dermal fibroblasts. *Acta Poloniae Pharmaceutica.* 69(6). 1303–1306. Available at: https://pubmed.ncbi.nlm.nih.gov/23285694/ ↑2 Lee, W. J., Sim, H. B., Jang, Y. H., Lee, S. J., Kim, D. W., & Yim, S. H. (2016). Efficacy of a complex of 5-aminolevulinic acid and glycyl-histidyl-lysine peptide on hair growth. *Annals of Dermatology.* 28(4). 438–443. Available at: https://doi.org/10.5021/ad.2016.28.4.438 ↑3 Carrer, V., Alonso, C., Pont, M., Zanuy, M., Córdoba, M., Espinosa, S., Barba, C., Oliver, M. A., Martí, M., & Coderch, L. (2020). Effect of propylene glycol on the skin penetration of drugs. *Archives of Dermatological Research.* 312(5). 337–352. Available at: https://doi.org/10.1007/s00403-019-02017-5 ↑4 Lessmann, H., Schnuch, A., Geier, J., Uter, W. (2005). Skin-sensitizing and irritant properties of propylene glycol. Contact Dermatitis. 53(5). 247-259. Available at: https://doi.org/10.1111/j.0105-1873.2005.00693.x. ↑5 Shadi, Z. (2023). Compliance to Topical Minoxidil and Reasons for Discontinuation among Patients with Androgenetic Alopecia. Dermatology and Therapy (Heidelb). 13(5). 1157-1169. Available at: https://doi.org/10.1007/s13555-023-00919-x ↑6 Alsantali, A. (2011). Alopecia areata: a new treatment plan. *Clinical, Cosmetic and Investigational Dermatology.* 4. 107–115. Available at: https://doi.org/10.2147/CCID.S22767 ↑7 Torok, H. M., Jones, T., Rich, P., Smith, S., & Tschen, E. (2005). Hydroquinone 4%, tretinoin 0.05%, fluocinolone acetonide 0.01%: a safe and efficacious 12-month treatment for melasma. *Cutis.* 75(1). 57–62. Available at: https://pubmed.ncbi.nlm.nih.gov/15732437/ ↑8 Blume-Peytavi, U., Lönnfors, S., Hillmann, K., & Garcia Bartels, N. (2012). A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by latanoprost 0.1% on hair growth and pigmentation in healthy volunteers with androgenetic alopecia. *Journal of the American Academy of Dermatology.* 66(5). 794–800. Available at: https://doi.org/10.1016/j.jaad.2011.05.026 ↑9 Bloch, L. D., Escudeiro, C. C., Sarruf, F. D., & Sakai Valente, N. Y. (2018). Latanoprost and minoxidil: comparative double-blind, placebo-controlled study for the treatment of hair loss. *Surgical & Cosmetic Dermatology.* 10(1). 39–43. Available at: https://doi.org/10.5935/scd1984-8773.20181011015 ↑10 Teus, M. A., Arranz-Marquez, E., & Lucea-Suescun, P. (2002). Incidence of iris colour change in latanoprost treated eyes. *British Journal of Ophthalmology.* 86(10). 1085–1088. Available at: https://doi.org/10.1136/bjo.86.10.1085 ↑11 Zito, P. M., Bistas, K. G., Patel, P., & Syed, K. (2024). Finasteride. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK513329/ (Accessed: October 2025) ↑12 Kaufman, K.D., Olsen, E.A., Whiting, D., Savin, R., DeVillez, R., Bergfeld, W., Price, V.H., Van Neste, D., Roberts, J.L., Hordinsky, M., Shapiro, J., Binkowitz, B., Gormley, G.J. (1998). Finasteride In The Treatment Of Men With Androgenetic Alopecia. Journal Of The American Academy Of Dermatology. 39(4 Pt 1). 578-589. Available at: https://doi.org/10.1016/s0190-9622(98)70007-6 ↑13 Ho, C.H., Sood, T., Zito, P.M. (2024). Androgenetic Alopecia. StatPearls. Available at: https://www.ncbi.nlm.nih.gov/books/NBK430924/ ↑14 Hirshburg, J. M., Kelsey, P. A., Therrien, C. A., Gavino, A. C., & Reichenberg, J. S. (2016). Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic review. *The Journal of Clinical and Aesthetic Dermatology.* 9(7). 56–62. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC5023004/ ↑15 Kaplan, S. A., Chung, D. E., Lee, R. K., Scofield, S., & Te, A. E. (2012). A 5-year retrospective analysis of 5α-reductase inhibitors in men with benign prostatic hyperplasia: finasteride has comparable urinary symptom efficacy and prostate volume reduction, but less sexual side effects and breast complications than dutasteride. *International Journal of Clinical Practice.* 66(11). 1052–1055. Available at: https://doi.org/10.1111/j.1742-1241.2012.03010.x ↑16 Mazzarella, G. F., Loconsole, G. F., Cammisa, G. A., Mastrolonardo, G. M., & Vena, G. A. (1997). Topical finasteride in the treatment of androgenic alopecia: preliminary evaluations after a 16-month therapy course. *Journal of Dermatological Treatment.* 8(3). 189–192. Available at: https://doi.org/10.3109/09546639709160517 ↑17 Hajheydari, Z., Akbari, J., Saeedi, M., & Shokoohi, L. (2009). Comparing the therapeutic effects of finasteride gel and tablet in treatment of the androgenetic alopecia. *Indian Journal of Dermatology, Venereology and Leprology.* 75. 47. Available at: https://doi.org/10.4103/0378-6323.45220 ↑18 Messenger, A. G., & Rundegren, J. (2004). Minoxidil: mechanisms of action on hair growth. British Journal of Dermatology. 150(2). 186–194. Available at: https://doi.org/10.1111/j.1365-2133.2004.05785.x ↑19 Gupta, A.K., Charrette, A. (2015). Topical Minoxidil: Systematic Review And Meta-Analysis Of Its Efficacy In Androgenetic Alopecia. Skinmed. 13(3). 185-189 ↑20 Vañó-Galván, S., Pirmez, R., Hermosa-Gelbard, A., Moreno-Arrones, Ó. M., Saceda-Corralo, D., Rodrigues-Barata, R., Jimenez-Cauhe, J., et al. (2021). Safety of low-dose oral minoxidil for hair loss: a multicenter study of 1404 patients. Journal of the American Academy of Dermatology. 84(6). 1644–1651. Available at: https://doi.org/10.1016/j.jaad.2021.02.054 ↑21 Shanshanwal, S. J., & Dhurat, R. S. (2017). Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: a randomized controlled open-label, evaluator-blinded study. *Indian Journal of Dermatology, Venereology and Leprology.* 83. 47. Available at: https://doi.org/10.4103/0378-6323.188652 ↑22 Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Varma Alluri, C., Mohammad, J., Rao, K.S.S.V.V., Reddy Dundigalla, M., (2025). A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia. Cureus. 17(8). e89309. Available at: https://doi.org/10.7759/cureus.89309 ↑23 Bazzano, G. S., Terezakis, N., & Galen, W. (1986). Topical tretinoin for hair growth promotion. *Journal of the American Academy of Dermatology.* 15(4). 880–893. Available at: https://doi.org/10.1016/S0190-9622(86)80024-X ↑24 Sharma, A., Goren, A., Dhurat, R., Agrawal, S., Sinclair, R., Trueb, R.M., Vano-Galvin, S., Chen, G., Tan, Y., Kovacevic, M., Situm, M., McCoy, J. (2019). Tretinoin enhances minoxidil response in androgenetic alopecia patients by upregulating follicular sulfotransferase enzymes. Dermatologic Therapy. 32(3). 12915. Available at: https://doi.org/10.1111/dth.12915 ↑25 Fischer, T. W., Trüeb, R. M., Hänggi, G., Innocenti, M., & Elsner, P. (2012). Topical melatonin for treatment of androgenetic alopecia. *International Journal of Trichology.* 4(4). 236–245. Available at: https://doi.org/10.4103/0974-7753.111199 ↑26 Dhurat, R., Chitallia, J., May, T. W., Jayaraaman, A. M., Madhukara, J., Anandan, S., Vaidya, P., & Klenk, A. (2018). An open-label randomized multicenter study assessing the noninferiority of a caffeine-based topical liquid 0.2% versus minoxidil 5% solution in male androgenetic alopecia. *Skin Pharmacology and Physiology.* 30(6). 298–305. Available at: https://doi.org/10.1159/000481141 ↑27 Pazoki-Toroudi, H., Moghadam, R. M., Ajami, M., Nassiri-Kashani, M., Ehsani, A., Tabatabaie, H., Khani, S., Abbasi, Z., Shizarpour, M., Firooz, A., & Dowlati, Y. (2013). The efficacy and safety of minoxidil 5% combination with azelaic acid 1/5% and caffeine 1% solution on male pattern hair loss. *Journal of Investigative Dermatology.* 133. S84–S84. Available at: https://doi.org/10.1038/jid.2013.97 Want help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Learn MoreMichael Williams, PhD
Michael is a researcher and writer who holds a BSc in Bioscience, an MSc in Regenerative Medicine, and a PhD in Translational Biomedicine. He undertook his PhD research at Houston Methodist Research Institute, Texas, focusing on cell signaling in the ovarian cancer tumor microenvironment. He conducted postdoctoral research at Barts Cancer Institute in London, exploring cellular metabolism in acute myeloid leukemia. He has published work in a range of fields, including oncology, nanomedicine, and cell-based therapeutics.
"... Can’t thank @Rob (PHH) and @sanderson17 enough for allowing me to understand a bit what was going on with me and why all these [things were] happening ... "
— RDB, 35, New York, U.S.A."... There is a lot improvement that I am seeing and my scalp feel alive nowadays... Thanks everyone. "
— Aayush, 20’s, Boston, MA"... I can say that my hair volume/thickness is about 30% more than it was when I first started."
— Douglas, 50’s, Montréal, CanadaWant help with your hair regrowth journey?
Get personalized support, product recommendations, video calls, and more from our researchers, trichologists, and PhD's dedicated to getting you the best possible outcomes.
Join Now - Mission Statement
 Scroll Down
Scroll Down